Abstract
Photoperiodic reproductive diapause is an essential part of female life cycle in several insect species living on high latitudes, where overwintering in reproductive stage involves high risks for survival and progeny production. The sensitive period (SP), during which photoperiodic cues can trigger the switch from direct development to diapause, can last from a few hours or days after emergence to the entire life span of females. Moreover, in some species, sexually mature females can enter post-reproductive diapause as a response to decreasing day length and/or temperature. We studied the duration of SP for diapause induction and the females’ ability to enter post-reproductive diapause at short day lengths in Drosophila montana strains from different latitudes in Europe, North America, and Japan. Our study shows that the females of this species have a life-long SP and that they retain an ability to switch between reproduction and diapause as a response to back-and-forth changes in day length for at least 3 months. D. montana strains from different latitudes showed high variation in females’ ability to enter post-reproductive diapause; females of the southern strains generally requiring longer time and/or lower temperature to enter this stage than those of the northern strains. Moreover, the proportion of females that switched to post-reproductive diapause in 3 weeks in short day conditions at 16 °C showed positive correlation with the critical day length (CDL) for diapause induction and the latitudinal and continental origin of the strains. Life-long SP increases females’ flexibility to respond to short-term changes in environmental conditions and enables reproducing females to switch to post-reproductive diapause when the days get shorter and colder toward the autumn. This ability can play a major role in species phenology and should be taken into account in theoretical and empirical studies on insect adaptation to seasonal variation.
Keywords
Insect phenology, involving repeated seasonal life cycle events, such as development from egg to adult, reproduction and diapausing, is sensitive to seasonal and latitudinal variation in climatic conditions. In species with an overwintering reproductive diapause, the mating period typically starts in spring so that the next generation(s) emerge during the summer. The summer generation females, which emerge before the critical day length (CDL; a photoperiod where 50% of the females of a given population enter diapause), develop ovaries and start to reproduce, while the ones that emerge after this time point enter diapause and typically develop ovaries only during the next spring. However, in some insect species females show plasticity in their diapause response so that also the females with fully developed ovaries may enter diapause (post-reproductive diapause), when the days get shorter and cooler toward the autumn.
Most of the phenotypic variation detected in insect diapause within populations is likely to be due to genetic variation in the plasticity of diapause responses under different environmental conditions (Hopper, 1999). Developmental plasticity can lead to the production of alternative phenotypes (e.g., reproducing and diapausing females), when individuals experience discretely different environments and/or when continuously varying environment induces a discrete threshold-like switch from one developmental pathway to another (Nijhout, 2003). In environments where the beginning and ending date of the favorable season varies among years, variation in diapause responses may also involve a component of risk spreading (“bet-hedging”; Kivelä et al., 2016). Here, the diapausing females can act as a buffer against environmental uncertainty and facilitate population (or genotype) survival, if the non-diapausing ones fail to reproduce (Hopper, 1999). Insect decision-making may also favor strategies that are intermediate between phenotypic plasticity and bet-hedging, when the time lag between information sensing and phenotype induction is large and/or when environmental cues are only partially predictive of future conditions (Joschinski and Bonte, 2020).
The sensitive period (SP), during which the switch to diapause can be triggered, ranges from various periods within the parental generation through different stages of embryonal, larval, and pupal development to the adult stage (Koštál, 2006). Taylor (1986) and Taylor and Spalding (1988) have divided the SPs involved in adult reproductive diapause into “determinate” ones, which end at a particular age, and “indeterminate ones,” which can extend indefinitely through insect life-span. Indeterminate/lifelong SP gives the females flexibility in responding to changing environmental cues and even allows them to reproduce before entering diapause (Bean et al., 2007; Taylor and Spalding, 1989). For example, in the tamarisk leaf beetle, Diorhabda elongata, reproducing females cease oviposition, resorb oocytes, and enter reproductive diapause when switched from long to short days (Bean et al., 2007). Increased oocyte resorption has been detected in some species also as a response to low temperatures (Singh, 1958), host unavailability (Wang and Horng, 2004), and low food quality (Moore and Attisano, 2011). In genus Drosophila, ovarian development has long been believed to be irreversible, once induced (Lumme, 1978), and post-reproductive diapause has so far not been reported in any species.
Our study species, Drosophila montana, offers a good opportunity to trace the duration of SP and to study the plasticity of female diapause responses in different photoperiods and temperatures, as the females’ developmental pathway (sexual maturation or diapause) is determined by photoperiodic cues received after eclosion (Salminen et al., 2012). This species also shows robust latitudinal clines in CDL and its temperature-sensitivity (Tyukmaeva et al., 2020), as well as in the photoperiodic time measuring system underlying the switch to diapause (Lankinen et al., 2021). In the present study, we addressed the following questions: (1) Are D. montana females able to switch from reproduction to diapause, and vice versa, when transferred between long- and short-day conditions? (2) How long do the females retain an ability to enter reproductive diapause, and can their SP be classified as determinate or indeterminate? (3) Does D. montana females’ ability to enter post-reproductive diapause show latitudinal variation and/or correlation with the strain CDL? Our study is the first one to demonstrate post-reproductive diapause in Drosophila, and it brings new insights into the plasticity of insect photoperiodic responses in seasonally varying environmental conditions at different latitudes.
Material and Methods
Study Strains
We used in the present study D. montana strains from Europe, North America, and Asia. Most of these strains have been established from the progenies of single overwintered fertilized females collected in the wild (information on the 2 oldest strains is lacking). In the lab, the strains were maintained in bottles containing malt medium (Lakovaara, 1969) in continuous light (LL) at 19 °C, which helps to prevent changes in their CDL (see Lankinen et al., 2013).
We have used in this and other ongoing studies the strain codes, which give information on the continental and latitudinal origin of the strains. We show also the previously used codes for each strain in Table 1 to enable comparisons with earlier studies.
Collecting site, latitude, establishing year, the codes used for D. montana strains in the present and earlier studies and CDL (Critical Day Length for diapause induction) for the study strains.
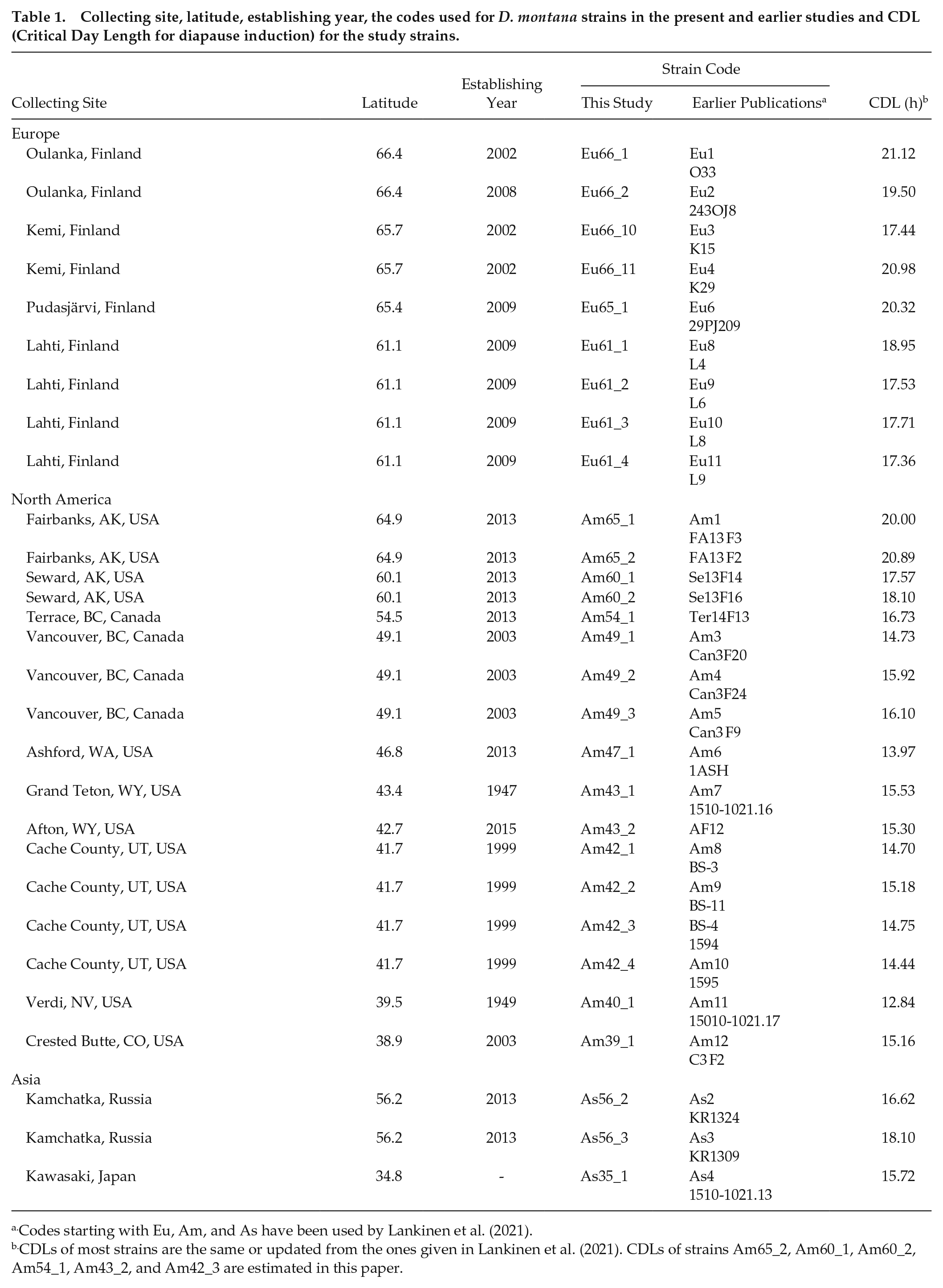
Codes starting with Eu, Am, and As have been used by Lankinen et al. (2021).
CDLs of most strains are the same or updated from the ones given in Lankinen et al. (2021). CDLs of strains Am65_2, Am60_1, Am60_2, Am54_1, Am43_2, and Am42_3 are estimated in this paper.
Experimental Procedures
All experiments were performed in the University of Oulu, Finland. During the experiments, we kept the flies in malt media bottles in wooden or plastic, light-insulated and actively ventilated boxes in the temperature-controlled rooms at 16 °C ± 0.3 °C or at 13 °C ± 0.3 °C. The boxes were illuminated with 1 white fluorescent lamp per chamber (9 W, Megaman, Germany) with the light intensity of 300-1000 lux corresponding about 5-16 W/m2. The flies were transferred into the first experimental photoperiods in malt media bottles already as pupae (females and males together) and into the next photoperiods in malt vials at adult stage. Flies of each strain were divided into several bottles/vials to prevent slight variation in the lightning conditions from increasing variation between strains. In all experiments, the adults were transferred into fresh vials weekly until they reached the age where their reproductive stage could be determined. In CDL studies, female reproductive stage was determined at the age of 21 ± 1 days, and in the transfer experiments at ages ranging from 21 to 126 days at 16 °C.
Ovarian development of Drosophila species is usually divided into 4 stages based on the developmental condition of the most posterior egg chamber (Watabe and Beppu, 1977; Kimura et al., 1978); this categorization is based on 14 stages of ovarian development in Drosophila melanogaster (see, e.g., Mirth et al., 2019). Stages I and II involve females with undeveloped ovaries (young and diapausing females), stage III females with mature ovaries, and stage IV very old post-reproductive females that are finishing or have already finished egg-laying. We added a new, undescribed stage in this classification: females that are in post-reproductive diapause (stage V).
We determined the reproductive stage of the ovaries of ether anesthetized D. montana females under a light microscope. Females were considered as sexually immature or diapausing, if their ovaries were small and transparent and/or if they contained no or only small amount of yolk. Ovaries of a few days old female (stage I) were tiny and they were surrounded by up to 300 roundish small and loose fatbody cells. In females that emerged in diapause-inducing conditions, the ovaries stayed small, the loose fatbody cells disappeared and the fatbody tissue gradually filled most of the abdomen (stage II). Ovaries of sexually mature females contained fully developed eggs (stage III), and the ovaries of post-reproductive females (stage IV) had elongated vitellarium portion and flattened appearance and they could contain a few eggs. Finally, the ovaries and the fatbody of the females that were in post-reproductive diapause (stage V) had the same appearance as the ovaries of stage II females. Photographs of the 3 first development stages of D. montana females’ ovaries are presented in Salminen and Hoikkala (2013). In the present study, “diapause proportion” measured during the first 3 weeks includes immature females with developing ovaries and diapausing females with undeveloped ovaries (stages I and II). After 3 weeks, “diapause proportion” refers to the proportion of females that are in diapause (stage II) or in post-reproductive diapause (stage V).
Measuring the Critical Day Length (CDL) for Diapause
We estimated the CDLs from the strains’ photoperiodic response curves (PPRCs) based on female diapause proportions in several photoperiods as in Lankinen et al. (2021). PPRCs were estimated for each strain with function drm (using option type = “binomial”) from R package drc (v3.0-1; Ritz et al., 2015) using a 4-parameter log-logistic model.
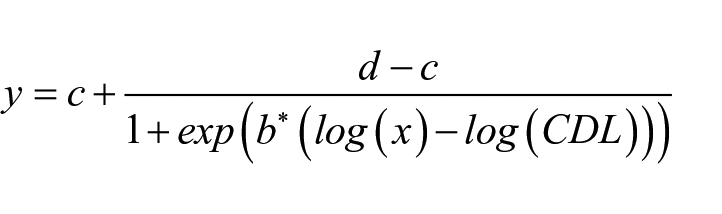
In this formula, y is the proportion of diapausing females, x is the number of light hours per day, c and d are the lower and upper limits (bound between 0 and 1), when x approaches 0 and 24, respectively, b is the slope around CDL (the point of inflection of the PPRC; bound between 0.001 and 100,000), and CDL is the critical day length defined as the dosage of light hours per day inducing reproductive diapause for 50% of the females (bound between 0 and 24).
CDL of most strains has already been estimated by Lankinen et al. (2021), but we recalculated these values for some strains after measuring their diapause proportions in 1-2 additional photoperiods and/or dissecting more females in some photoperiods (differences between strains’ old and new CDL values were ≤0.12 h). We also estimated the CDLs for 6 new strains: Am65_2, Am60_1, Am60_2, Am54_1, Am43_2, and Am42_3. Sample sizes for all strains are given in Supplementary Table S1.
Determining the Length of Sensitive Period (SP) for Diapause Induction and Females’ Ability to Switch Between Reproduction and Diapause
In D. montana, eclosing females have undeveloped ovaries and the females develop sexually mature or enter reproductive diapause depending on the photoperiodic conditions after emergence (Salminen et al., 2012); for example, in LL the females develop ovaries and in LD 12:12 they enter diapause.
In the first set of experiments, we studied whether and how D. montana females’ ability to enter diapause is affected by the number of days spent in continuous light (LL) before being transferred into short days (LD12:12) and/or by the number of days in short days after the transfer. Here, we transferred the females of 3 strains, Eu66_1, Eu61_1, and Eu61_2, from LL to LD12:12 1-13 days after emergence and maintained them there until the day 21 or 42 of the experiment. In the 21-day experiment, female reproductive stage was determined after 8-20 days and in the 42-day experiment after 29-41 days in LD12:12. Both experiments were performed in 16 °C.
In the second set of experiments, we first maintained groups of newly emerged females of strains Eu66_1 and Eu61_1 in LD12:12 or LL for 16-18 weeks at 16 °C to find out whether they are able to maintain their reproductive stage (diapausing or reproducing) over long periods in stable photoperiods. We determined the reproductive stage of female samples from both conditions once a week, starting from week 3 in LD12:12 and from week 1 in LL (see Figure 2a and 2b). Then we performed a transfer experiment, which lasted for 12 weeks and where female reproductive stage was determined from the samples collected at 1-3 weeks intervals (see Figure 2c-2e). In this experiment, we maintained all females for the first 3 weeks in LL and the next 3 weeks in LD12:12. After this, we left a group of females for another 3 weeks in LD12:12 (Figure 2c) and transferred the rest of females into LL for 3 weeks. Finally, we divided the females that had been transferred into LL into 2 groups; one group was left in LL for another 3 weeks (Figure 2d), while the second group was transferred again into LD12:12 (Figure 2e).
In the third set of experiments, we studied whether the photoperiod of LD12:12 is adequate for performing comparisons between the post-reproductive diapause proportions of D. montana strains from different latitudes, given considerable variation detected in their CDLs. The most important requirement for these comparisons was that the day length must be shorter than the CDL of all study strains (this was fulfilled). Moreover, given the large difference between the 12-h day length and the CDL of the northernmost strains (up to 21 h), we tested whether the proportion of females in post-reproductive diapause differs between LD12:12 and LD18:6 in the 2 most northern strains, Eu66_1 and Eu66_2 with high CDLs. In the first experiment, we maintained freshly emerged females (sample size 2187) either in LD 12:12 or in LD 18:6 for 14 weeks (Figure 3a). In the second experiment, we maintained the females (sample size 1 988) the first 3 weeks in LL to allow them to develop ovaries, and then transferred them for 6 weeks into LD 12:12 or in LD 18:6 to see how efficiently these photoperiods induce post-reproductive diapause (Figure 3b). Both experiments were performed at 16 °C and female reproductive stage was determined at 1-3 weeks intervals.
Studying Geographic Variation in Females’ Tendency to Enter Post-Reproductive Diapause
We first studied females’ tendency to switch from reproduction to diapause at 16 °C and/or 13 °C in 29 D. montana strains from different latitudes in Finland, North America, and Japan. Females of 26 strains were maintained the first 3 weeks in LL and the last 3 weeks in LD12:12 at 16 °C, and the reproductive stage of female samples was determined 3, 4, 5, 6, and/or 9 weeks after the transfer. Strain comparisons in females’ ability to enter post-reproductive diapause were based the diapause proportions of 6-week-old females at 16 °C (3 weeks in LL + 3 weeks in LD 12:12), where the strains showed maximal differences. We also determined the diapause proportion of 18 strains at 13 °C after an initial period of 4 weeks in LL to allow females enough time for ovary development (this was done especially for the strains whose diapause proportion remained low at 16 °C). In this temperature, we determined the reproductive stage of females 4, 6, 7, 8, and/or 10 weeks after the transfer.
We used 2 kinds of tests to study females’ ability to enter (or not enter) post-reproductive diapause. First, we tested whether a significant proportion of females had entered post-reproductive diapause within different D. montana strains by comparing the number of females with developed ovaries at the beginning (after 3, 4, or 5 weeks, depending on strains) and at the end of the experiment (after 3-12 weeks, depending on strains) at 13 °C or 16 °C, using a chi-square test. The purpose of this test was to find out whether the females of given strains enter post-reproductive diapause when given enough time given and maintained in a suitable temperature. Second, we used a general linear model (glm) to test whether strains’ CDL, latitude, and/or continent of origin explains variation in the proportion of females that enter post-reproductive diapause when maintained 3 weeks in LL and 3 weeks in LD12:12 at 16 °C. We modeled the number of diapausing females as a binomial response (with a logit link function) and used an analysis of deviance with a chi-square test to identify the best model explaining the observed data. Using the best model, we then calculated the correlations between D. montana strains’ post-reproductive diapause proportion with CDL and latitude.
Results
D. montana Females’ Ability to Switch Between Reproduction and Diapause
We started the present study by testing whether and how the experiment duration affects our ability to define the length of the sensitive period (SP) for diapause induction. The main question in this set of experiments was whether and how D. montana females’ ability to enter post-reproductive diapause is affected by the number of days spent in continuous light (LL) before being transferred into short days (LD12:12) and/or by the number of days in short days after the transfer. This is important to know, as too short total experimental time (e.g., 18 days in Salminen and Hoikkala, 2013) may give misleading results.
We made this part of studies on 3 D. montana strains, Eu66_1, Eu61_1, and Eu61_2, by transferring groups of females from LL to LD12:12 1-13 days after emergence and determining the stage of female ovaries in samples collected after the experiment had lasted for 21 or 42 days (Figure 1). In Eu61_1, practically all females entered diapause regardless of the number of days spent in either condition (288/295 females in 21-day experiments and all 447 females in 42-day experiment), which could be due to females’ slow ovary development and/or to their rapid response to altered photoperiodic conditions. In the 2 other strains, the proportion of diapausing females decreased in 21-day experiment along with an increase in the number days in LL and decrease in LD12:12 (in Eu61_1 and Eu61_2 only 809/1135 and 259/519 females entered diapause, respectively; Figure 1b and 1d). In 42-day experiment, longer time in LD12:12 enabled also these females to enter diapause (349/371 of Eu61_1 females and 309/328 of Eu61_2 females entered diapause; see Supplementary Table S2 for exact sample sizes). Overall, these studies show that D. montana females’ SP lasts at least 13 days, that the females have to be maintained more than 13 days in LL to be sure that their ovaries are fully developed and that they will enter post-reproductive diapause when given enough time in LD12:12. Thus, in the rest of the experiments, we maintained the females first 3 weeks in LL at 16 °C (or 4 weeks at 13 °C) to ensure that all of them had time to develop ovaries and at least 3 weeks in short day conditions so that they had time to enter post-reproductive diapause.
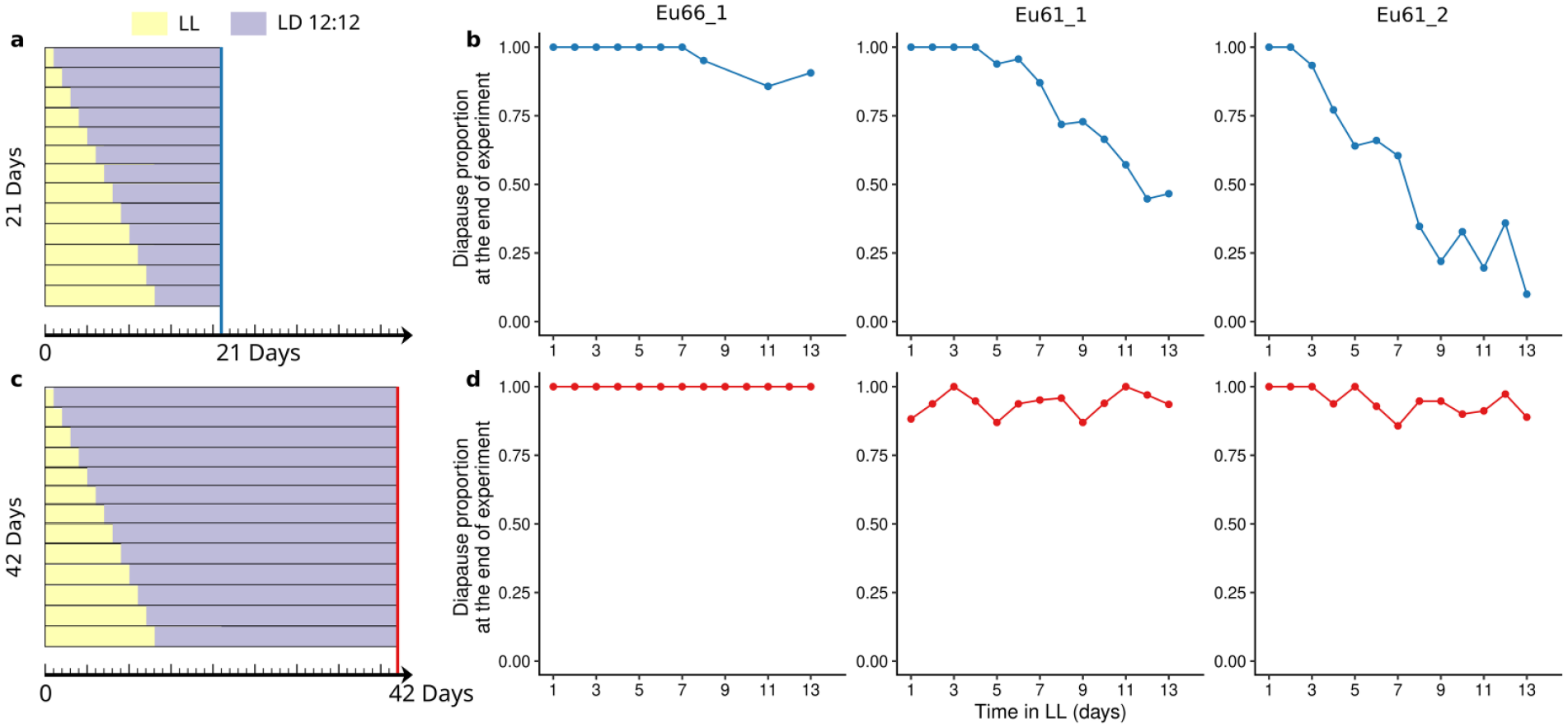
Study protocol for the 21-day (a) and 42-day (c) transfer experiments and the diapause proportions of the females of D. montana strains Eu66_1, Eu61_1, and Eu61_2 measured in these experiments (b and d; the top rows in a and c correspond the first time points in b and d, etc.). Females were maintained 1-13 days after their emergence in LL (number of days in LL is shown on the x-axis) and then transferred for the remaining days in LD12:12 (for 9-20 days in 21-day experiments and for 30-41 days in 42-day experiments). In the 21-day experiments, the total number of females was 295 for Eu66_1, 1135 for Eu61_1, and 519 for Eu61_2, and the average numbers of females per estimate 34, 87, and 9, respectively. In the 42-day experiments, the total number of females was 447 for Eu66_1, 371 for Eu61_1, and 328 for Eu61_2, and the respective average numbers of females per estimate 9 and 40 and 25. See Supplementary Table S2 for exact sample sizes.
In the next set of experiments, we first studied females’ ability to remain in diapause or reproductive state in constant photoperiods (LD12:12 and LL) in 2 D. montana strains. In Eu66_1, female diapause proportion remained close to 100% in LD12:12 for up to 18 weeks, while in Eu61_1 it decreased to 50% after 15 weeks (Figure 2a). In LL, the females of both strains developed ovaries in 3 weeks and then remained at reproductive stage until the end of the period (Figure 2b). We then performed transfer experiments, where practically all females developed ovaries in 3 weeks in LL and entered post-reproductive diapause after 3 weeks in LD12:12 (Figure 2c-2e). After the next transfer, the females that had been left in LD12:12 remained in diapause (Figure 2c), while the ones that were transferred into LL developed ovaries in 1-2 weeks, which suggests that their ovaries were already in pre-vitellogenic state during the transfer (Figure 2d). Finally, transferring a group of reproducing females on week 9 into LD12:12 for 3 weeks induced diapause, but the diapause proportion remained at 75% in Eu66_1 and at 25% in Eu61_1 (Figure 2e). These findings show that D. montana females can remain in diapause or reproductive state and respond to back-and-forth changes in day length by switching between these states for at least 3 months.
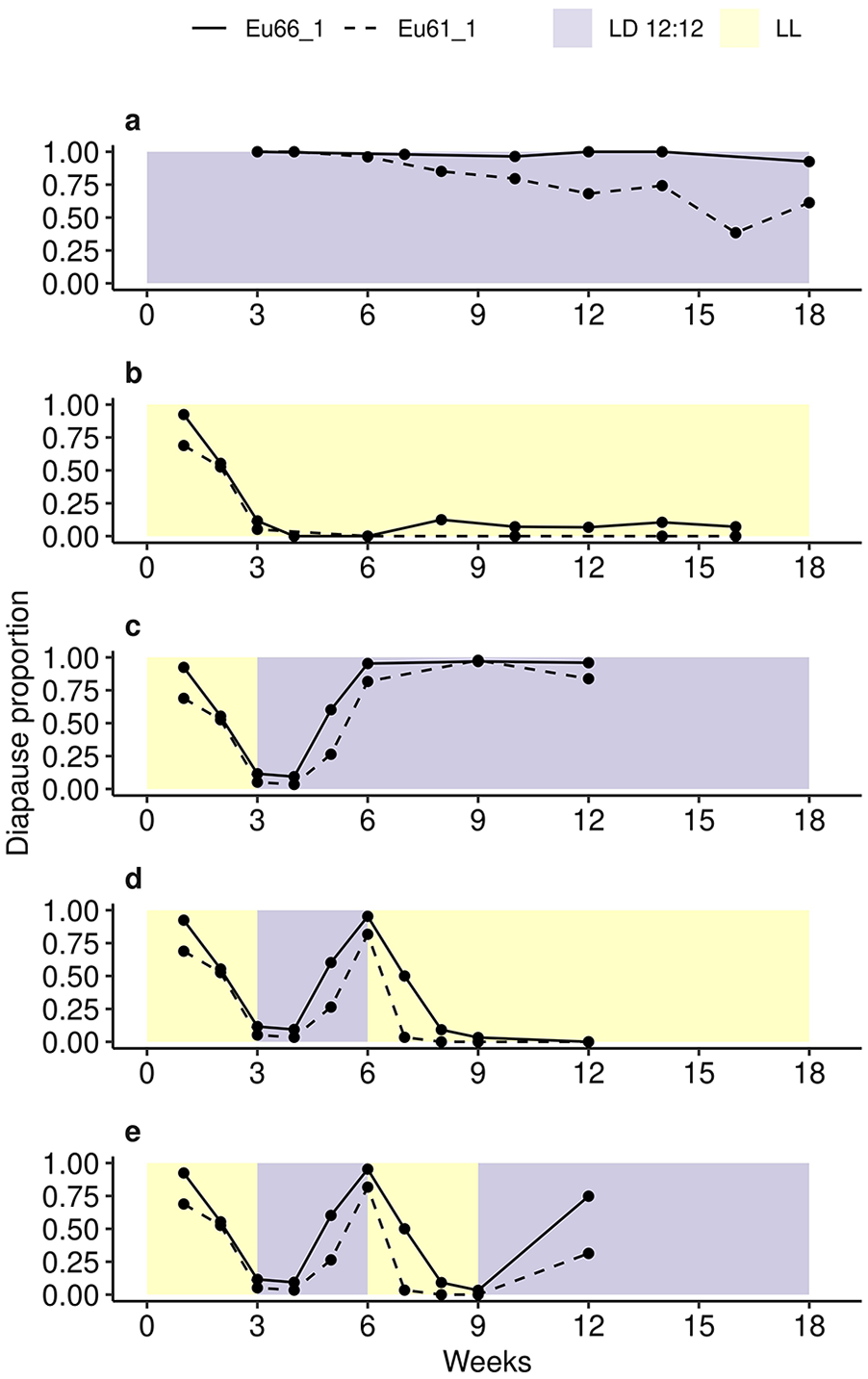
Constant condition and transfer experiments performed on the females of D. montana strains Eu66_1 and Eu61_1. In constant condition experiments (a and b), females’ ovary development was traced in LL and LD 12:12 until the females were 18 and 16 weeks old, respectively. In the transfer experiments, all females were maintained the first 3 weeks in LL and the next 3 weeks in LD 12:12. Then a group of females was left in LD 12:12 (c), and the rest of the females were transferred on week 6 into LL (d). Finally, a group of females was transferred on week 9 back to LD 12:12 (e). Experiments involved 5115 females from 2 strains, across 5 treatments and 18 possible time points (~98 females per estimate).
Before tracing geographical variation in female tendency to enter post-reproductive diapause, we wanted to verify that the photoperiod of LD12:12 is adequate for strain comparisons given considerable variation detected in strain CDLs. The first requirement, that the CDLs of all study strains are longer than 12 h, was fulfilled (see Table 1). The second requirement, the appropriateness of using LD12:12 as a diapause inducing photoperiod in all strains, was studied in the 2 northernmost strains Eu66_1 (CDL 21.12) and Eu66_2 (CDL 19.5). In the first part of these experiment, the females were maintained since their emergence either in LD 12:12 or in LD 18:6 at 16 °C (Figure 3a). In Eu66_1, female diapause proportions were similar in both photoperiods during the first 6 weeks, but after that the diapause was maintained more effectively in LD 12:12 than in LD18:6, while in strain Eu66_2 the 2 photoperiods were equally effective, but less effective than LD 12:12 in Eu66_1 (Figure 3a). In the second part of the experiment, the females were maintained the first 3 weeks in LL to allow them to develop ovaries, after which they were transferred into LD 12:12 or in LD 18:6 for the next 6 weeks. As Figure 3b shows, both photoperiods were about equally effective in inducing post-reproductive diapause and maintaining it for 6 weeks in both strains. Nearly all females of strain Eu66_1 entered post-reproductive diapause in both photoperiods in 3 weeks. However, in Eu66_2, LD12:12 appeared to be more effective in inducing post-reproductive diapause than LD18:6, which was close to the strain CDL (in the latter photoperiod only half of the females entered diapause in 3 weeks and female diapause proportion remained low even after 6 weeks). Accordingly, we decided to use LD12:12 as a diapause inducing photoperiod when studying geographic variation in females’ tendency to enter post-reproductive diapause.
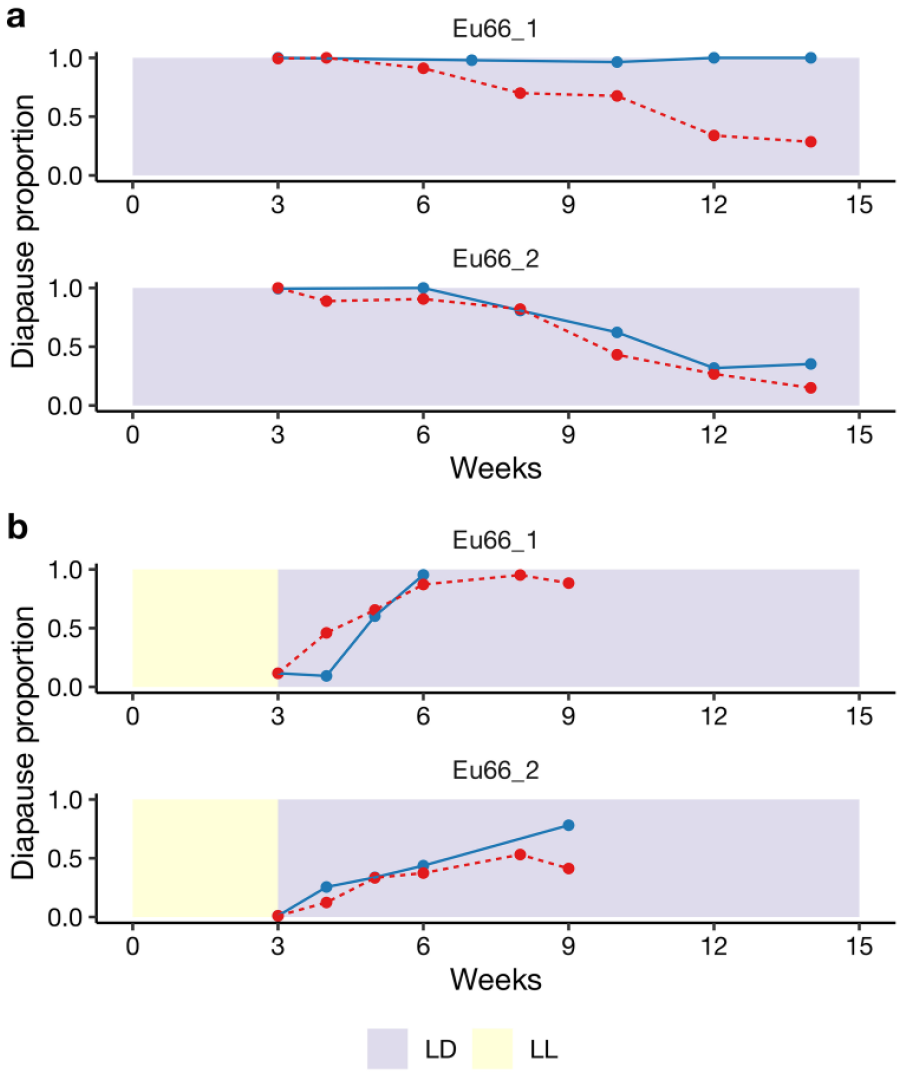
Diapause proportions of the females of the North European strains Eu66_1 and Eu66_2 (a) when maintained from the first day in LD12:12 (blue, solid curve) or LD18:6 (red, dashed curve) and (b) when maintained the first 3 weeks in LL and the next 6 weeks in LD12:12 blue, solid curve and LD18:6 (red, dashed curve). We used in these experiments 2187 (a) and 1988 (b) females from 2 strains across 11 time points (average numbers of females per time point were 84 and 117, respectively).
Geographic Variation in D. montana Females’ Ability to Enter Post-Reproductive Diapause
In the first part of experiments, our purpose was to find out whether the females of D. montana strains enter post-reproductive diapause when maintained in suitable conditions. Here, the basic transfer experiment (the first 3 weeks in LL and the next 3 weeks in LD12:12 at 16 °C) on 26 strains from Europe, North America, and Asia revealed remarkable variation in females’ tendency to enter post-reproductive diapause (Figure 4). Nearly all females of 8 strains, including 4 strains from Europe, 3 from North America and 1 from Asia, entered post-reproductive diapause. Maintaining 22 strains in LD12:12 for another 3 weeks raised the diapause proportion of 11 strains close to 100%. Comparing the number of reproducing females against the total number of females at the start or soon after the start of the diapause-inducing LD (weeks 3-5 depending on the strain), and after maintaining the females in LD12:12 for 3-9 weeks (weeks 6-12) with a chi-square test, showed that a significant proportion (at α < 0.05) of females in 22 out of 26 strains had entered post-reproductive diapause (see Figure 4). The 4 strains with no significant post-reproductive diapause included Am60_1, Am49_1, Am40_1, and Eu66_10. Of these strains, Am49_1 and Am60_1 showed a slight trend toward post-reproductive diapause, while Eu66_10 and Am40_1 had the lowest diapausing ratio at week 9.
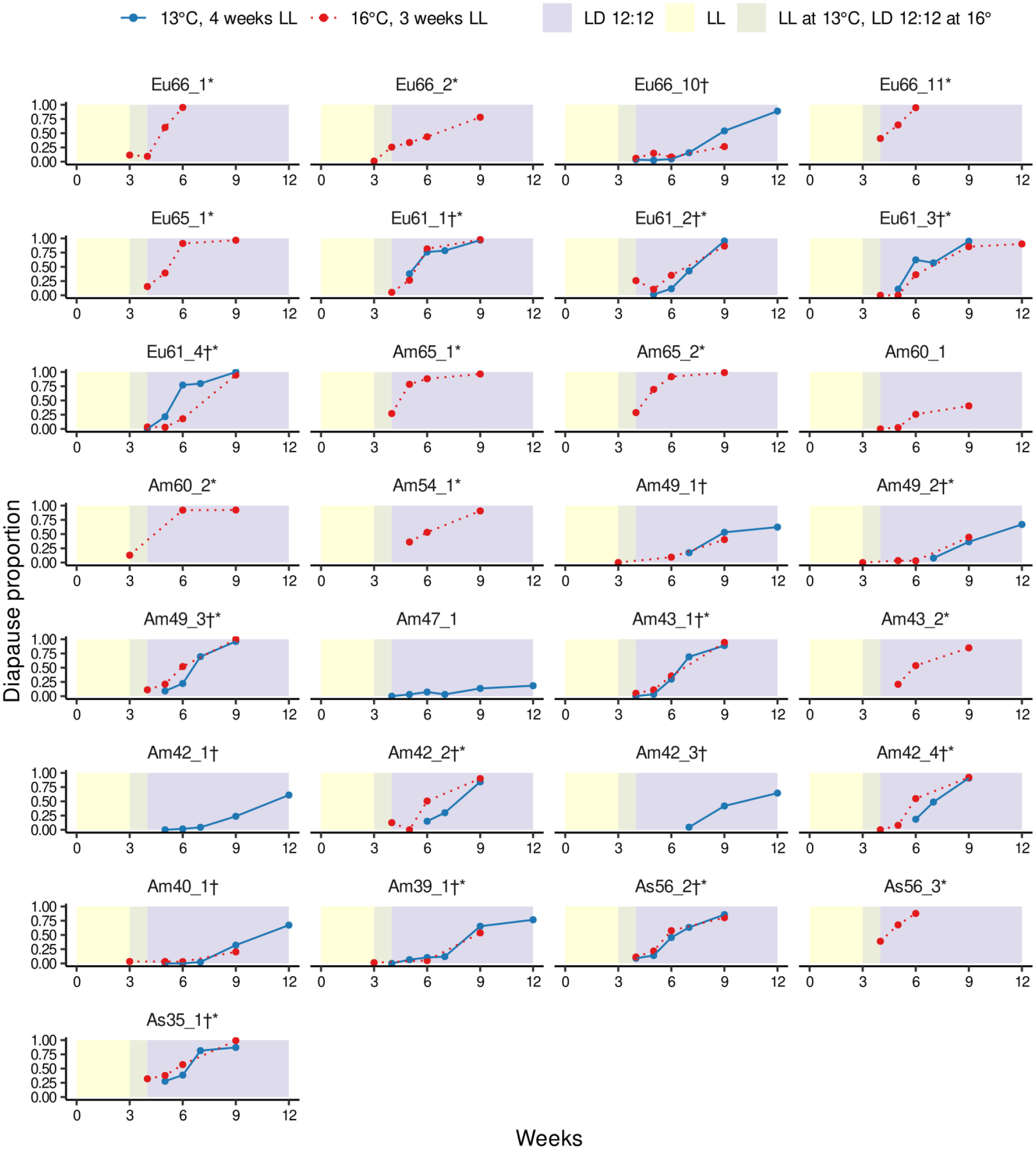
Females of the study strains were first maintained 3 weeks in LL and then transferred into LD 12:12 for another 3 weeks in 16 °C (red, dashed curves). For the strains where female diapause percentage had not reached 100% during the 3 weeks in LD 12:12, the experiment was continued for another 3 weeks (in some cases for 6 weeks). Finally, the experiment was repeated for 18 strains in 13 °C (blue, solid curves) . In this temperature, the females were maintained the first 4 weeks in LL to allow them enough time for ovary development. * and † indicate strains where we detected a post-reproductive diapause at 16 °C and 13 °C, respectively (see material and method). We used in these experiments a total of 14573 females, across 29 lines, 2 experimental temperatures, and 7 possible time points (average number of females per time point 82.3). See Supplementary Table S3 for exact information on sample sizes.
We then performed the same experiment on 18 strains at 13 °C, including strains Am47_1, Am42_1, and Am42_3 which had not been used in 16 °C experiments (Figure 4). At this temperature, the diapause proportion of nearly all strains raised close to 100% by the end of week 9 or week 12. However, in 5 strains (Eu66_10, Am49_1, Am49_2, Am42_3, and Am40_1) the diapause proportion was only ~50% at the end of week 9 and in 2 strains even lower (in Am42_1 ~20% and in Am47_1 ~10%; these 2 strains have a very weak and temperature-sensitive diapause response; Lankinen et al., 2021). A chi-square test, similar to the one explained above, showed that only 1 strain (Am47_1) did not have significant post-reproductive diapause at 13 °C.
Females’ age distribution varied in this experiment between 21 and 70 days, but this is not a problem as the females can retain their reproductive stage at least for 4 months in constant photoperiods (see Figure 2b). In addition, to make sure that a 3-degree temperature difference (13 °C vs. 16 °C) does not affect female diapause induction in LL, we determined female reproductive stage at the time points between 3 and 12 weeks using the data published here and some additional data collected by Lankinen (unpublished). In the combined data over these time points, the average proportion of diapausing females in LL was 3.8% (n = 1 541) at 16 °C and 4.0% (n = 2 322) at 13 °C.
Finally, we used a glm model to identify the factors that could best explain variation in the proportion of post-reproductive diapause observed across D. montana strains on week 6 (3 weeks in LL and 3 weeks in LD12:12) at 16 °C. We used a binomial model (link function “logit”) with the ratio of diapausing females as the response variable and tested the models with the CDL and the latitude or the continental origin of the strains as explanatory variables. We then compared the fit of models adding each variable successively in all possible orders. The analysis of deviance showed that the model using all 3 explanatory variables explains the observed data significantly better than a model with only 1 or 2 variables (p value < 0.05). The best model showed an effect of the continent of origin (estimates = −11.78, −2.17, and 0.92 for Asia, Europe, and Northern America strains, respectively, all with p values < 0.05), the CDLs (estimate = 0.75, p value < 2 × 10−16), and latitude (estimate = −0.070, p value = 2.09 × 10−13). Scaling the latitude and CDL variables to account for the different units resulted in qualitatively the same observations although the scaled CDLs had the biggest estimated coefficients (coefficients respectively for CDLs and latitude: 2.2963, −0.7240, and the estimates for the continent of origin: Asia, Europe, and North America, respectively, 0.9406, −1.7192, and −0.7567, with all estimates with p value < 0.05). We illustrate the correlation between the strains’ post-reproductive diapause proportion with CDLs in Figure 5 and with latitude in Supplementary Figure S2. While CDL contributed more to the fit of our model (see Supplementary Table S5) than latitude, the fact that latitude still contributed significantly in explaining the proportion of diapause in post-reproductive diapause of our strains suggests that the proportion of post-reproductive diapause and the CDL reflect slightly different aspects of female reproductive diapause.
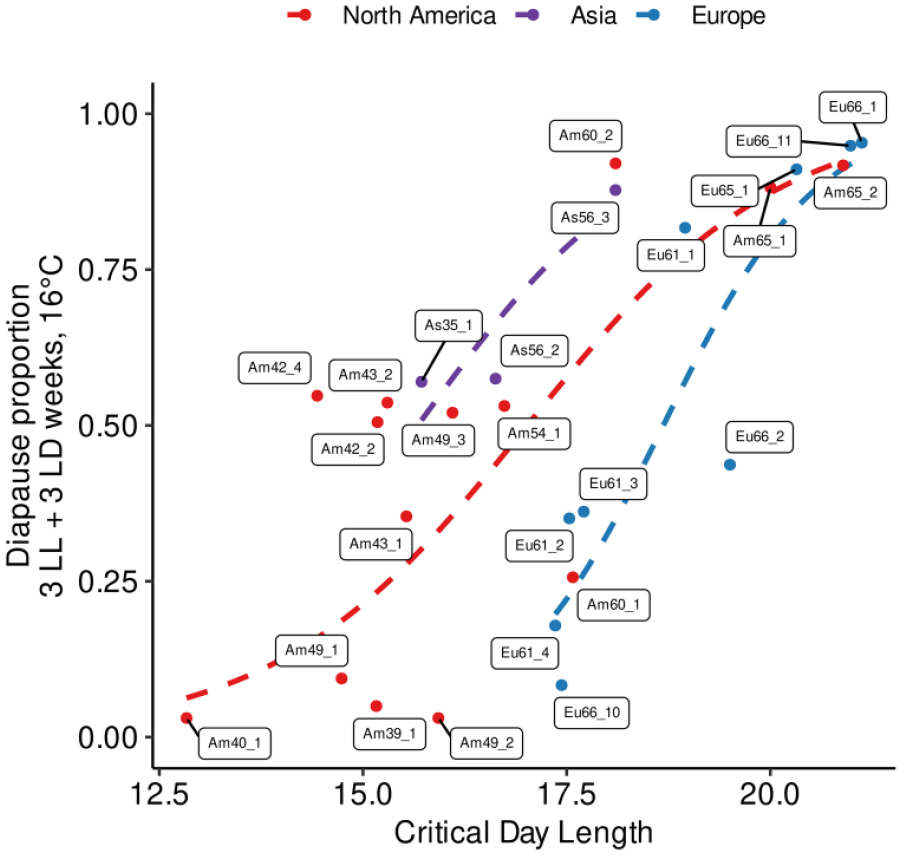
Correlation between the proportion of females that had entered post-reproductive diapause proportion when maintained 3 weeks in LL and 3 weeks in LD 12:12 (y-axis) and the critical day length (CDL; x-axis) for D. montana strains from different continents. We used in these experiments a total of 2979 females across 26 strains (average number of females per strain 115).
Discussion
Reproductive diapause plays an important role in insect ability to survive over the cold season and produce progeny during the next spring. We showed that D. montana females have a lifelong SP, that they are able to switch between reproduction and diapause according to prevailing photoperiodic and temperature conditions, and that this ability is shared between strains from Europe, North America, and Japan. Moreover, females’ sensitivity to environmental cues triggering the switch to post-reproductive diapause showed remarkable latitudinal variation and correlated with the strain CDL. This is the first demonstration of a lifelong/indeterminate SP in genus Drosophila, and the conditions invoking post-reproductive diapause, and it opens road for similar studies in other species of this genus.
Our study shows that D. montana females are able to switch between reproduction and diapause as a response to day length for at least 3 months, which suggests indeterminate SP and indicates high plasticity in female diapause response. This kind of plasticity may evolve when variation in the developmental responses to environmental cues is heritable and some responses confer greater reproductive success than the others, and when environments vary predictably and frequently enough to make the ability to produce alternative phenotypes advantageous (Davis and Wund, 2016). In high latitude populations, natural selection is also suggested to favor broad “safety margins” so that the diapause is initiated when days are relatively long (long CDL), while in the low-latitude populations the strong time stress for producing several generations per year may postpone the diapause toward the autumn (short CDL). On high latitudes, the time lag between D. montana females’ emergence and CDL is less than 3 months even if the females emerge in early summer, and the present study shows they can enter post-reproductive stage at any time during their reproductive life-span. Also, Aspi et al. (1993) have shown that in a partially bivoltine D. montana population (1 full and 1 partial generation per year) in northern Finland, females with fully developed ovaries disappear in late summer when the temperature is still relatively high, which confirms that reproducing females enter post-reproductive diapause in late summer conditions.
D. montana possesses robust latitudinal clines in CDL and its temperature-sensitivity (Tyukmaeva et al., 2020), as well as in females’ susceptibility to enter diapause in continuous darkness and in non-circadian photoperiods and in correlation between these traits and CDL (Lankinen et al., 2021). In the present study, we detected latitudinal variation in D. montana females’ susceptibility to enter post-reproductive diapause; females of the southern strains requiring longer time and/or lower temperature to enter this state than those of the northern strains. Moreover, the proportion of females that switched to post-reproductive diapause in 3 weeks in short day conditions at 16 °C showed positive correlation with CDL and the latitudinal and continental origin of the strains. Overall, these studies show that D. montana females from high latitudes have lower requirements for the photoperiodic and temperature cues invoking direct or post-reproductive diapause even though variation in these traits is high also within the populations.
Indeterminate/lifelong SP of D. montana females can have far-reaching consequences on species survival in cold environments, but it also raises several questions for further studies. First, it would be interesting to know whether the CDL where females enter post-reproductive diapause differs from the CDL of freshly emerged females. For example, in Diorhabda elongata leaf beetles possessing post-reproductive diapause, CDL of the females switching from reproduction to diapause is nearly the same as that measured for newly emerged females (Bean et al., 2007). Another interesting question is whether and how the switch to post-reproductive diapause affects D. montana females’ overwinter survival and total offspring production. Reproducing females are often suggested to contribute to the overwintering population only via their offspring (Taylor, 1980), but the situation changes if they are still able to enter diapause and continue progeny production in spring (Taylor and Spalding, 1988), like in D. montana. Switching to post-reproductive diapause in late summer also involves several risks that may decrease females’ overwinter survival. Most importantly, the females with post-reproductive diapause have used a lot of energy for ovary development and egg production and they have only a short time to collect energy reservoirs for the winter even if they can get some of this energy back through egg resorption. The risks involved in post-reproductive diapause are to be assessed against the risk of surviving over winter with ovaries full of eggs, as the cold-tolerance of reproducing D. montana females is significantly lower than that of the diapausing ones (Vesala and Hoikkala, 2011). Studying the trade-offs underlying post-reproductive diapause would help to better understand the adaptation of insect species into environments with high seasonal variations and to identify the selection pressures affecting this trait in uni- and multivoltine (1 or several generations per year) populations along latitudes.
Conclusion
This study revealed in a cold-adapted Drosophila species, D. montana, unsuspected complexity in the regulation of reproductive diapause that is likely to play an important role in species adaptation to seasonal variations. It also shows that studying complex regulatory systems, such as diapause, can be challenging and requires careful experiment planning, since seemingly small differences in the experimental design can yield very different results and lead to erroneous conclusions. Moreover, the study raised several topics, which we hope to inspire studies in a wider range of species. First, are other Drosophila species able to enter post-reproductive diapause and is this behavior commonly occurring in natural conditions? Second, given that females have the flexibility to switch back and forth between diapause and reproduction, what are the trade-off involved with reproducing in late summer when the eggs and larvae might have poor chances to survive the winter? Answers to these questions could bring new light to the understanding of the adaptation of insects to seasonal variation in temperate habitats.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304221108968 – Supplemental material for Plasticity in Photoperiodism: Drosophila montana Females Have a Life-Long Ability to Switch From Reproduction to Diapause
Supplemental material, sj-pdf-1-jbr-10.1177_07487304221108968 for Plasticity in Photoperiodism: Drosophila montana Females Have a Life-Long Ability to Switch From Reproduction to Diapause by Pekka Lankinen, Chedly Kastally and Anneli Hoikkala in Journal of Biological Rhythms
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
