Abstract
Social jetlag, a misalignment between sleep timing on the weekend and during the work week, is associated with depressive symptoms among adults across both sexes. A previous study found that later sleep timing was associated with depressive symptoms in women but not men. To date, however, no research has investigated whether the association between social jetlag and depression varies by sex among adolescents. The current study assessed self-reported sleep, depressive symptoms, and demographic information from 3058 adolescents (48% female, mean [SD] age 15.59 [0.77] years) from the age 15 wave of the Fragile Families and Child Wellbeing Study (FFCWS). Social jetlag was calculated as the absolute value of the midpoint of sleep on the weekend minus the midpoint of sleep during the school week. Depressive symptoms were measured through a modified 5-item version of the Center for Epidemiologic Studies Depression Scale (CES-D). We assessed whether the associations among sleep duration on school nights, social jetlag, and depressive symptoms were similar between male and female adolescents using multiple linear regression. In fully adjusted models, sex moderated the association between school night total sleep time and depressive symptoms (p < 0.001) and between social jetlag and depressive symptoms (p = 0.037). In females, but not in males, school night total sleep time was negatively associated with depressive symptoms (p < 0.001), whereas social jetlag (p < 0.001) was positively and independently associated with depressive symptoms. The results indicate the importance of regular sleep timing across the week and adequate sleep duration for the maintenance of optimal emotional health among female adolescents.
Major depressive disorder is the most common type of mood disorder in the United States (Substance Abuse and Mental Health Services Administration, 2014). The average age of onset of major depression is between 11 and 14 years (Merikangas et al., 2011), indicating that depressive symptoms may emerge well before adulthood. Indeed, nearly 12% of American adolescents aged 13 to 18 have manifested clinical depression at some point in their lifetime, with 9% of these adolescents reporting severe depression-related impairment (Merikangas et al., 2011). Thus, identifying risk factors for depressive symptoms before adulthood, such as sleep behaviors, may be critical for the early detection and treatment of major depressive disorder.
Over 70% of adolescents in the United States report fewer than the minimum recommended amount of 8 hour sleep per night (Wheaton et al., 2018). Furthermore, insufficient total sleep time (TST) is associated with depressive symptoms (Fernandez-Mendoza et al., 2016; Raniti et al., 2016; Roberts and Duong, 2014), with one longitudinal study finding a reciprocal relationship between short sleep and depressive symptoms across one year (Roberts and Duong, 2014). Given the high prevalence of insufficient sleep and the bidirectional association with depressive symptoms during a period in which depressive symptoms emerge (Merikangas et al., 2011), sleep health is a priority area for adolescent mental health research.
Adolescents experience a shift toward later preferred timing of daily activities (e.g., sleep), or later chronotype, as compared with children and adults (Borisenkov et al., 2012; Carskadon et al., 1993; Randler et al., 2017; Roenneberg et al., 2004). During the school week, adolescents with later chronotype tend to have later bedtimes but relatively fixed early wake times due to school and other obligations (Roenneberg et al., 2003; Roepke and Duffy, 2010; Vitale et al., 2015). Late bedtimes and early wake times result in shorter TST during the school week as compared with the weekend when adolescents are “free” to wake up later. As a result, adolescents may attempt to compensate for short sleep during the school week with longer TST on the weekend (Roenneberg et al., 2003; Roepke and Duffy, 2010; Vitale et al., 2015). Later chronotypes thus tend to have a sleep interval with a later midpoint on the weekend relative to the school week (Wittmann et al., 2006). This misalignment in sleep-wake timing between the school week and the weekend is known as “social jetlag” (Wittmann et al., 2006). Adolescents experience short sleep during the school week and more social jetlag compared with adults (Touitou, 2013; Wittmann et al., 2006) due to the combination of both a shift toward later chronotype during adolescence (Borisenkov et al., 2012; Carskadon et al., 1993; Randler et al., 2017; Roenneberg et al., 2004) and early school start times (Vollmer et al., 2016; Wheaton et al., 2015). Therefore, both short school night TST and social jetlag are prevalent in adolescents.
Among adults, social jetlag is associated with negative emotional health (Lau et al., 2013; Randler and Vollmer, 2013; Yong et al., 2016). For example, adults with social jetlag self-report greater depressive symptoms (Levandovski et al., 2011; Polugrudov et al., 2016). However, a separate study among adolescents and young adults aged 12 to 21 years failed to replicate this association (de Souza and Hidalgo, 2014); in that analysis, the potential moderating effect of sex was not investigated. Female adolescents are more than twice as likely to exhibit major depressive disorder or dysthymia as compared with male adolescents (Avenevoli et al., 2015; Merikangas et al., 2011), suggesting potential sex differences in the etiology of depressive symptoms. Interestingly, one study found that adult females, but not males, with sleep midpoints later than 0445 hour were more likely to exhibit depressive symptoms (Morita et al., 2015). As later chronotype is associated with increased social jetlag (Wittmann et al., 2006), a positive relationship may similarly exist between social jetlag and depressive symptoms in females, but not males. However, no studies have examined whether sex moderates the relationship between social jetlag and depressive symptoms in an adolescent population specifically. Female adults exhibit a shorter free-running circadian period, or tau, as compared with male adults (Duffy et al., 2011). Research in adolescents is more limited but suggests a similar pattern of shorter tau in females vs. males (Crowley and Eastman, 2018). Corroborating these studies, females also exhibit an earlier dim light melatonin onset (DLMO) phase and wider DLMO phase angle (relative to bedtime) than do males (Van Reen et al., 2013), indicating sex differences in the relationship of circadian phase to sleep timing behaviors. The greater prevalence of depressive symptoms and earlier circadian phase in females suggest potential sex differences in the association among sleep timing misalignment and depressive symptoms. Furthermore, it is unknown whether any relationship between social jetlag and depressive symptoms remains after accounting for TST during the school week, which is shorter in individuals with social jetlag (Touitou, 2013; Wittmann et al., 2006).
The current study had 2 aims: 1) to investigate the relationships between social jetlag and TST on school nights (TST School) with depressive symptoms among adolescents, and 2) to determine whether these relationships are moderated by sex. We hypothesized that TST School would be negatively associated with depressive symptoms in both sexes. Furthermore, considering previous research among adults, we hypothesized that greater social jetlag would be associated with depressive symptoms in females but not in males.
Materials and Methods
Participants and Design
Data for the current analyses come from the Fragile Families and Child Wellbeing Study (FFCWS; www.fragilefamilies.princeton.edu). The original birth cohort consisted of ~4900 children born between 1998 and 2000 in 20 US cities, obtained through stratified random sampling. Non-marital births were over-sampled to obtain a sample of low socio-economic status (SES) (Wagmiller, 2010). To date, data have been collected in 6 waves: at birth (Reichman and Teitler, 2001) and at ages 1 (Noonan et al., 2010), 3 (Wang and Maguire-Jack, 2018), 5 (Hale et al., 2011), 9 (Hunt et al., 2017), and 15 (Mathew et al., 2019; Nahmod et al., 2018). The current study examines the cross-sectional survey responses of 3444 youth at age 15 years and their primary caregivers (PCGs), defined as the person with whom the youth lives for “half the time” or more. Youth missing data on demographic, sleep, or depressive symptoms were excluded from the current study, yielding a final analytical sample of 3058 youth (88.8% of in-wave total and 62.4% of initial birth cohort). Families were compensated $100 for completion of the PCG questionnaire and $50 for completion of the youth questionnaire. Researchers in the FFCWS are located at multiple institutions, and approval has been obtained from each site’s Institutional Review Board.
Youth Age 15 Questionnaire
Data used in statistical analyses were drawn from 2 sources at the age 15 wave: a questionnaire administered to youth, and a separate questionnaire administered to their primary caregivers.
This questionnaire was administered to youth at age 15. Teens were asked to report sleep variables, including bedtime/wake time on school days and weekends, depressive symptoms, and demographic information, including race/ethnicity, height, and weight (used to calculate body mass index, BMI), and number of hours spent working outside the home weekly.
Sleep Measures
“Bedtime on school nights” and “bedtime on weekend nights” were assessed through the questions, “What time do you usually go to bed on school nights? By school night, we mean Sunday night through Thursday night,” and “What time do you usually go to bed on weekend nights? By weekend night, we mean Friday and Saturday night,” respectively. Youth were prompted to report the hour, minutes, and AM/PM for bedtimes.
“Wake time on school mornings” and “wake time on weekend mornings” were assessed through the questions, “What time do you usually wake up on school day mornings?” and “What time do you usually wake up on weekend mornings?” respectively. Youth were prompted to report the hour, minutes, and AM/PM for wake times.
The following variables were calculated using bedtime and wake time measures collected from the Youth Age 15 Questionnaire. “Total sleep time (TST) on school nights” (TST School) was calculated as the interval between reported bedtime and wake time on school nights (Sunday through Thursday) in hours. “TST on weekend nights” (TST Weekend) was calculated as the interval between reported bedtime and wake time on weekend nights (Friday and Saturday) in hours. “Weekly average TST” was calculated through the following formula: ([TST School × 5] + [TST Weekend × 2]) / 7. “Midpoint of sleep on school nights” was calculated through the following formula: Bedtime School + (TST School × 0.5) (Roenneberg et al., 2003). “Midpoint of sleep on weekend nights” was calculated through the following formula: Bedtime Weekend + (TST Weekend × 0.5) (Roenneberg et al., 2003). “Chronotype” was calculated according to the formula created by Roenneberg et al. (2007), which uses the midpoint of sleep on weekend nights, corrected for accumulated sleep debt, to determine chronotype: Sleep Midpoint on Weekend Nights − 0.5 × (TST Weekend − (5 × TST School + 2 × TST Weekend) / 7. (A higher sleep debt-corrected sleep midpoint on weekend nights indicates later chronotype.)
In primary regression models, “social jetlag” in hours was calculated through the following formula: | sleep midpoint on weekend nights–sleep midpoint on school nights | (Wittmann et al., 2006). We conducted a separate regression analysis using social jetlag categorized as low (≤2 hour), moderate (>2 hour and ≤4 hour), and high (>4 hour), as defined by Levandovski et al. (2011), to test the possibility of a non-linear relationship between social jetlag and depressive symptoms (see Supplemental Information).
Depressive Symptoms Measure
Depressive symptoms were assessed through a modified 5-item version of the Center for Epidemiologic Studies Depression Scale (Perreira et al., 2005). The five-item CES-D scale has previously shown superior cross-cultural comparability to the full 20-item scale. For 5 statements expressing depressive symptoms, youth selected a level of agreement based on the following scale: 1 = strongly agree, 2 = somewhat agree, 3 = somewhat disagree, and 4 = strongly disagree. The statements were as follows: “I feel I cannot shake off the blues, even with help from my family and my friends,” “I feel sad,” “I feel happy” (reverse-scored), “I feel life is not worth living,” and “I feel depressed.” A depressive symptoms score was calculated as the average of items scored and reverse scored, with a greater composite score indicating greater depressive symptomology (i.e., 0 = strongly disagree, 3 = strongly agree). Youth with 2 or more missing items were not included in the analytical sample. The coefficient alpha for the items on the scale was 0.76, indicating acceptable reliability.
Covariate Measures from Youth Questionnaire
“Race and ethnicity” consisted of 4 exclusive categories: “White/Caucasian” (reference); “Black/African American,” “Hispanic and/or Latino,” or a “mixed, other, or none” category.
“Body mass index (BMI) percentile” at age 15 was assessed using self-reported height and weight. BMI was calculated through the following formula: (weight in pounds / [height in inches2] × 703) (Keys et al., 1972) and a percentile was calculated based on 2000 CDC growth charts (Kuczmarski et al., 2002). The 50th percentile indicates the median BMI for the adolescent’s age and sex, and a percentile greater than 68% indicates the adolescent is overweight based on the World Health Organization growth reference (De Onis et al., 2007).
“Number of hours of work weekly” was used to assess the amount of time (in hour) the youth spent working for pay outside of the household.
Primary Caregiver (PCG) Questionnaire
Covariate Measures from PCG Questionnaire
Covariates for statistical analyses were additionally drawn from a questionnaire administered to PCGs: relationship to youth, PCG employment status, annual household income level, PCG highest level of education completed, number of children in household other than the youth, and whether the PCG (if a biological parent) was married or cohabiting with the youth’s other biological parent (but not if married to someone else). PCG depressive symptoms were scored through the Composite International Diagnostic Interview – Short Form (CIDI-SF), Section A (Kessler et al., 1998) and consisted of 0, does not meet criteria for major depressive disorder, and 1, meets criteria for major depressive disorder.
Statistical Analyses
Analyses were conducted in SAS 9.4 (SAS Institute Inc., Cary, NC). Cases were excluded from all analyses if missing any of the following: bedtime or wake time on either school or weekend nights (n = 63), more than 2 items from the CES-D (n = 1), and/or covariate questions (n = 322), yielding 386 excluded adolescents and a total analytic sample of N = 3058.
Sex differences in sleep timing and TST variables were assessed through independent-groups t tests. Effect sizes (d) were interpreted based on Cohen’s cutoffs for small (d = 0.20), medium (d = 0.50), and large (d = 0.80) (Cohen, 1992).
Separately, 3 linear regression analyses were conducted to determine the interactions of TST School and social jetlag with sex on depressive symptoms. Model 1 included the following predictors: sex, TST School and its interaction with sex, and social jetlag (measured continuously) and its interaction with sex. Models 2 and 3 tested the associations of TST School and social jetlag with depressive symptoms separately within females and males, respectively. Additional analyses tested the associations of TST School and categories of social jetlag (low, moderate, or high) with depressive symptoms (see Supplemental Information). Given that individuals with African ancestry exhibit shorter circadian periods compared with individuals with European ancestry (Eastman et al., 2017), we additionally ran models stratified by Black (n = 1416) and non-Black (i.e., White/Caucasian, Hispanic and/or Latino, or mixed, other, or none; n = 1642) race/ethnicity. Model 4 tested the 3-way interactions for each sleep variable (social jetlag and TST School) with sex and Black race/ethnicity. Models 5 and 6 were similar to Model 1 except conducted only in non-Blacks or in Blacks, respectively: predictors included TST School and its interaction with sex, and social jetlag and its interaction with sex. Effect sizes (∆R2) were interpreted based on Cohen’s cutoffs for small (∆R2 = 0.01), medium (∆R2 = 0.09), and large (∆R2 = 0.25) (Cohen, 1992).
Covariates were selected a priori based on previous literature (Chase-Lansdale et al., 2003; Jung et al., 2017; Kim, 2014; Park et al., 2013; Schubert et al., 2017; Wang et al., 2016; Zimmerman and Katon, 2005) and were included in analyses as they were significantly correlated with CES-D depressive symptoms score: reported race/ethnicity, age in years, BMI percentile, number of hours of work outside of the home per week, number of other children below the age of 18 in the household, annual household income, and PCG relationship to youth, married/cohabiting with youth’s biological parent, employment status, education level, and meeting depression criteria as indicated by the CIDI-SF, Section A (Kessler et al., 1998). Categorical covariates were dummy-coded and entered into the regression models. The variance inflation factor (VIF) did not exceed 2.5 for any predictor in the regression model, indicating no multicollinearity (i.e., no predictor was strongly correlated with another predictor) (Menard, 1995).
Results
The final analytical sample consisted of 3058 youths, 88.8% of the age 15 wave (see Table 1 for sample demographic information). Among adolescents in the entire sample, the mean depressive symptoms (CES-D) score was 0.59 with a range of 0 to 3 (SD = 0.60); 81% of adolescents had a score ≤1 (indicating an average response of “disagree” with each of 5 depressive statements). Female adolescents had significantly higher depressive symptom scores (0.66 [0.63]) than did males (0.52 [0.56]), t(2946) = 6.22, p < 0.001, d = 0.23.
Demographic information from analytical sample (N = 3058) of the age 15 wave of the Fragile Families and Child Wellbeing Study (FFCWS).
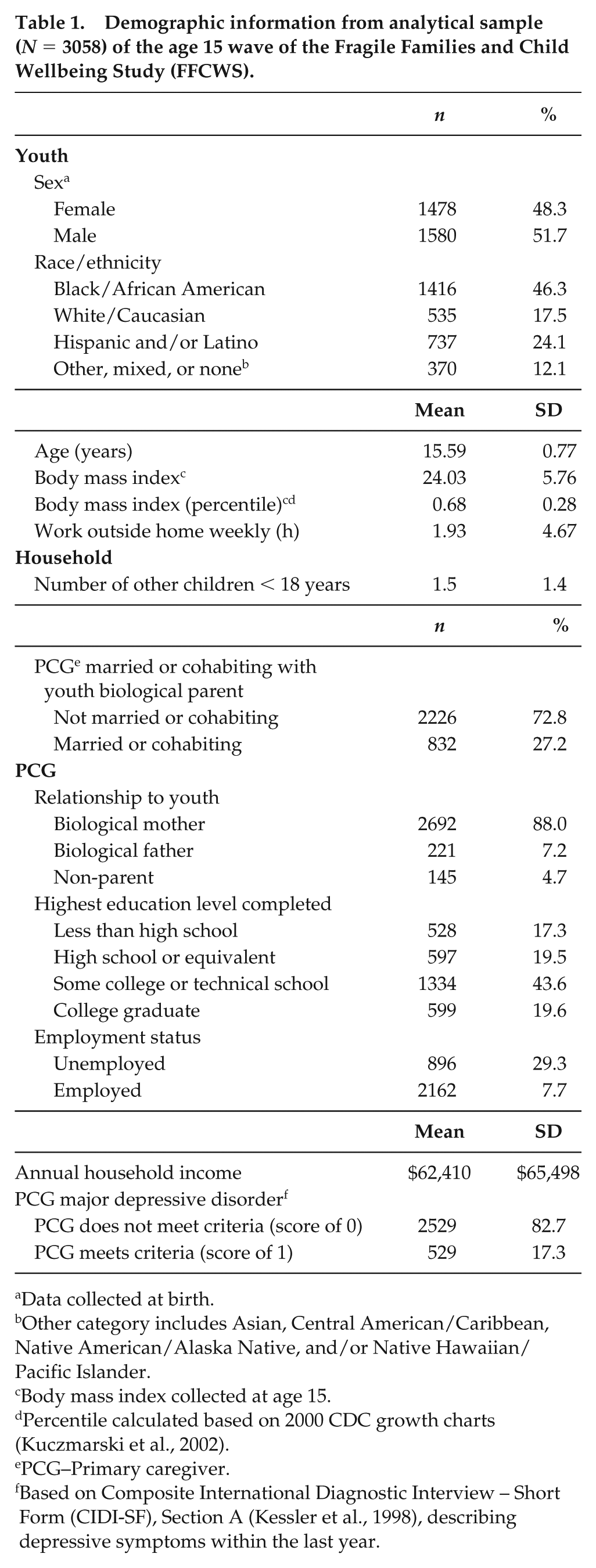
Data collected at birth.
Other category includes Asian, Central American/Caribbean, Native American/Alaska Native, and/or Native Hawaiian/Pacific Islander.
Body mass index collected at age 15.
Percentile calculated based on 2000 CDC growth charts (Kuczmarski et al., 2002).
PCG–Primary caregiver.
Based on Composite International Diagnostic Interview – Short Form (CIDI-SF), Section A (Kessler et al., 1998), describing depressive symptoms within the last year.
Between-Sex Differences in Sleep Variables
Females reported significantly less sleep on school nights (7.93 [1.23] hour) than did males (8.02 [1.24] hour), t(3056) = −2.02, p = 0.044, d = −0.07, driven by an earlier wake time in females compared to males (p < 0.001). The mean social jetlag in the entire sample was 2.80 [1.46] hour with no difference in between the sexes (p > 0.05). Furthermore, compared to males, females reported more sleep on the weekends (p = 0.001), driven by females going to bed earlier than males on the weekend (p = 0.001) and reporting earlier chronotype as indicated by the sleep midpoint on weekend nights, corrected for sleep debt (p = 0.001). Additional sleep variables for the between-sex comparisons are presented in Table 2.
Between-sex t-tests in sleep measures (N = 3058).
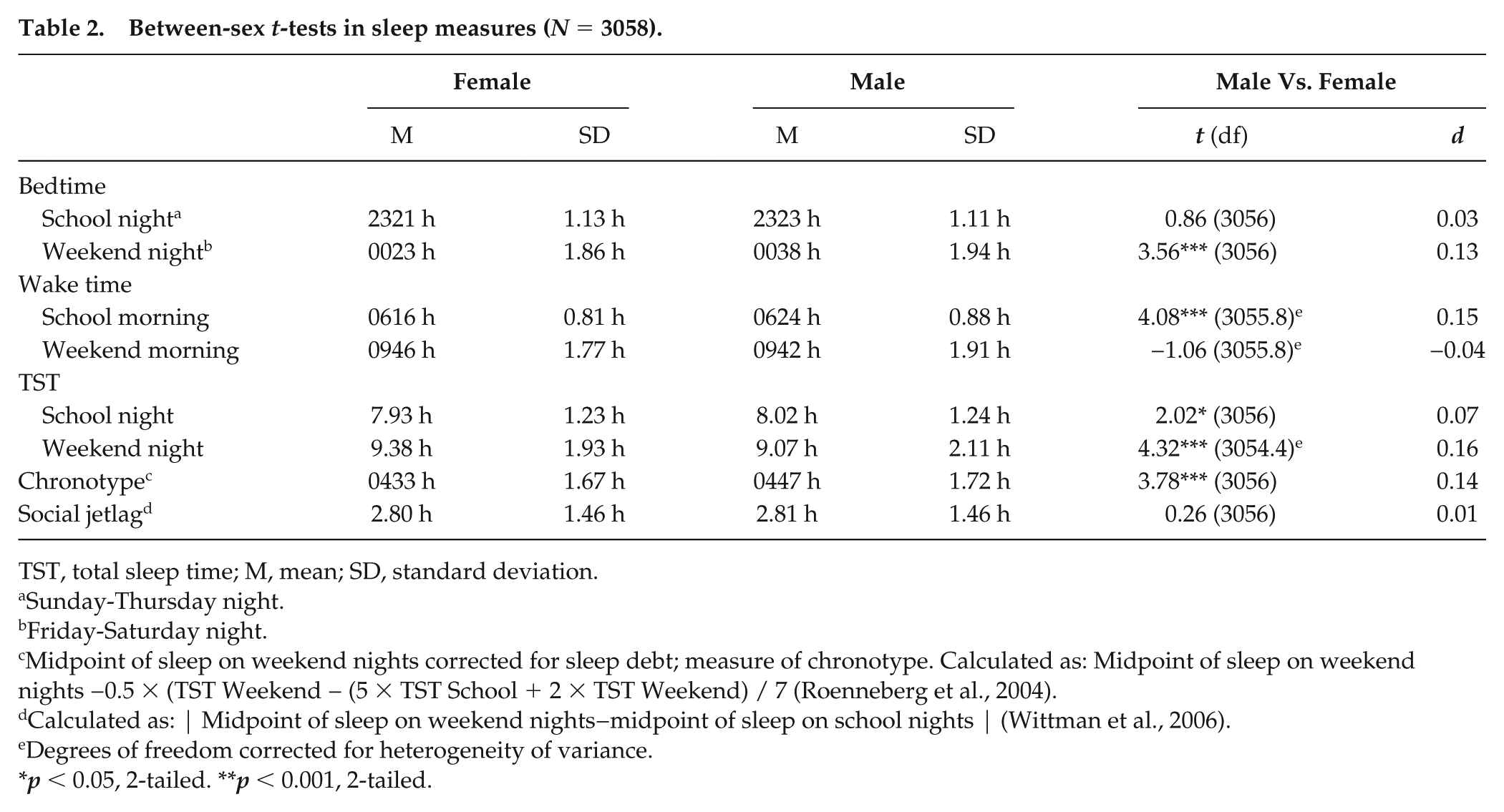
TST, total sleep time; M, mean; SD, standard deviation.
Sunday-Thursday night.
Friday-Saturday night.
Midpoint of sleep on weekend nights corrected for sleep debt; measure of chronotype. Calculated as: Midpoint of sleep on weekend nights −0.5 × (TST Weekend − (5 × TST School + 2 × TST Weekend) / 7 (Roenneberg et al., 2004).
Calculated as: | Midpoint of sleep on weekend nights−midpoint of sleep on school nights | (Wittman et al., 2006).
Degrees of freedom corrected for heterogeneity of variance.
p < 0.05, 2-tailed. **p < 0.001, 2-tailed.
Between-Sex Associations of TST School and Social Jetlag with Depressive Symptoms Score
The full model including the entire sample, N = 3058 (Model 1), was used to investigate the associations of TST School, social jetlag, and their sex interactions with depressive symptoms score (see Table 3) and explained 5.8% of the variance in the depressive symptoms score. There were significant interactions between sex and TST School (b = 0.07, ∆R2 = 0.011, p < 0.001) and between sex and social jetlag (b = 0.03, ∆R2 = 0.003, p = 0.037) on depressive symptoms.
Regression models predicting depressive symptoms (CES-D Score a ) from TST School and social jetlag (N = 3058)

CI, confidence interval; Ref, reference group; TST, total sleep time.
CES-D subscale range: 0 = low depressive symptoms; 3 = high depressive symptoms (Perreira et al., 2005).
95% confidence interval for the unstandardized coefficient, b.
Mean-centered.
Calculated as: | Midpoint of sleep on weekend nights−midpoint of sleep on school nights | (Wittmann et al., 2006).
Adjusted for reported race/ethnicity, age in years, body mass index percentile, number of other children below the age of 18 in the household, number of hours youth works outside home per week, annual household income, youth relationship to primary caregiver (PCG), PCG married/cohabiting with youth’s biological parent, PCG employment status, PCG education level, and PCG meeting clinical depression criteria for past year.
p < 0.01, 2-tailed. ***p < 0.001, 2-tailed.
Post hoc analyses were conducted to investigate the associations of TST School and social jetlag with depressive symptoms within each sex separately. Model 2 (in females only) explained 7% of the variance in the depressive symptoms score and showed that TST School was negatively associated with depressive symptoms (b = −0.08, ∆R2 = 0.027, p < 0.001); thus, females with shorter TST School reported more depressive symptoms. Social jetlag was positively associated with depressive symptoms in females (b = 0.04, ∆R2 = 0.009, p < 0.001), indicating females with greater social jetlag expressed more depressive symptoms. Model 3 (in males only) explained 3.1% of the variance in depressive symptoms score and found no association between TST School and depressive symptoms or between social jetlag and depressive symptoms (p > 0.05). The results indicated that females with short sleep during the school week and with greater social jetlag, but not males, expressed more depressive symptoms. See Figure 1 for a depiction of the associations of TST School and social jetlag with depressive symptoms within each sex.
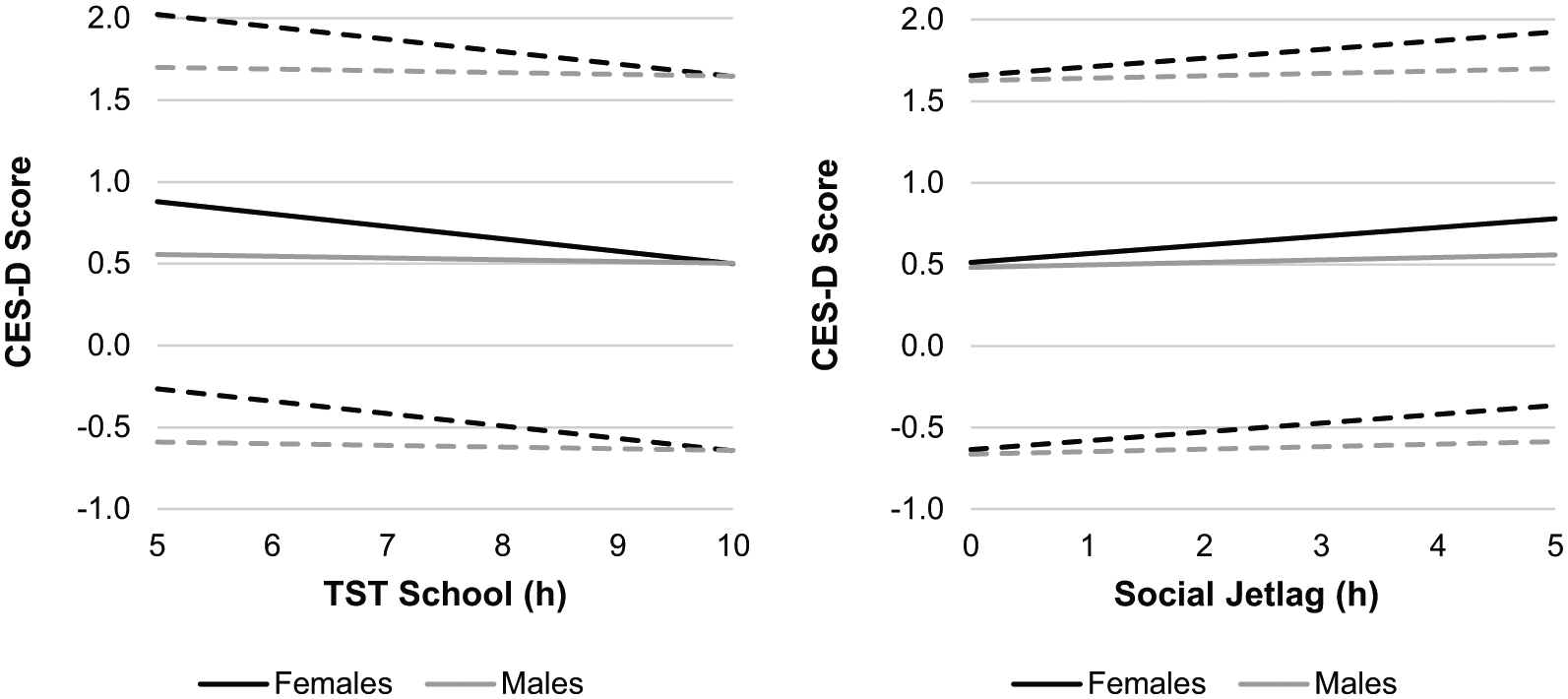
According to Model 1, the interactions between school night total sleep time (TST School) and sex (b = 0.07, ΔR2 = 0.011, p < 0.001), and between social jetlag (| Midpoint of sleep on weekend nights−midpoint of sleep on school nights | ; Wittman, 2006) and sex (b = −0.03, ΔR2 = 0.003, p = 0.037), on depressive symptoms (Center for Epidemiologic Studies Depression Scale; CES-D) score (Perreira, 2005) in adolescents (N = 3058) were significant. In females (black lines), TST School (left panel; b = −0.08, ΔR2 = 0.027, p < 0.001) and greater social jetlag (right panel; b = 0.04, ΔR2 = 0.009, p < 0.001) were associated with a higher depressive symptoms score. In males (gray lines), neither TST School nor social jetlag (p > 0.05), were associated with a higher depressive symptoms score according to Model 3. Dashed lines represent 95% CI around the mean.
Stratification by Black Race/Ethnicity
Model 4 tested the 3-way interactions of each sleep variable (social jetlag and TST School) with sex and Black race/ethnicity and explained 5.9% of the variance in the depressive symptoms score. The interaction among social jetlag, Black race/ethnicity, and sex was significant (b = 0.07, ∆R2 = 0.008, p = 0.011); there was no significant interaction among TST School, Black race/ethnicity, and sex (p > 0.05).
Models 5 (in non-Blacks) and 6 (in Blacks) examined interactions between sleep variables and sex, stratified by Black race/ethnicity, with the following predictors: TST School and its interaction with sex, and social jetlag and its interaction with sex. Model 5 in non-Blacks explained 7.0% of the variance in depressive symptoms and indicated a significant interaction between social jetlag and sex on depressive symptoms (b = −0.07, ∆R2 = 0.014, p < 0.001). Post hoc analyses in non-Blacks were conducted to investigate the associations of TST School and social jetlag with depressive symptoms within each sex separately. A model in non-Black females explained 9.1% of the variance in depressive symptoms and indicated a significant positive association between social jetlag and depressive symptoms (b = 0.06, ∆R2 = 0.014, p = 0.001). A model in non-Black males explained 4.6% of the variance in depressive symptoms and indicated no significant association between social jetlag and depressive symptoms (p > 0.05). Model 6 in Black adolescents (male and female) explained 5.4% of the variance in depressive symptoms and indicated no significant interaction between social jetlag and sex on depressive symptoms (p > 0.05). According to Model 6, social jetlag was not significantly associated with depressive symptoms (p > 0.05) in Black adolescents (both male and female).
In summary, the results indicate a negative association between TST School and depressive symptoms in both Black and non-Black females (b = −0.08, ∆R2 = 0.027, p < 0.001), a positive association between social jetlag and depressive symptoms in non-Black females only (b = 0.06, ∆R2 = 0.014, p = 0.001), and no significant associations between either social jetlag or TST School and depressive symptoms in males (Blacks and non-Blacks).
Discussion
The findings from the current study indicate that in female adolescents, greater social jetlag and shorter TST School were independently associated with higher depressive symptoms. However, in males, neither social jetlag nor TST School were associated with depressive symptoms. Furthermore, the amount of social jetlag did not differ between male and female adolescents; TST School was significantly higher in males. Together, these findings indicate that differences in the amounts of TST and social jetlag between males and females do not account for between-sex differences in adolescent depressive symptoms. Rather, the associations of TST School and social jetlag with depressive symptoms differ between the sexes; that is, female adolescents exhibit greater vulnerability to experiencing depressive symptoms accompanying short TST School and social jetlag. Thus, male and female adolescents may have different risk factors and require different treatment for depressive symptoms.
Our findings indicate significant, albeit small, differences in TST and sleep timing between male and female adolescents. Compared with males, females have a 5.4-min shorter TST School (d = 0.07) driven by an earlier school day wake time (d = 0.15), and a 19-min longer TST Weekend (d = 0.16) driven by a 14-min earlier chronotype (d = 0.14) and earlier weekend night bed time (d = 0.13). Although they have a similar bedtime during the school week to their male counterparts, females wake up earlier on school mornings, obtaining less school night sleep and compensating by going to bed earlier on the weekend and obtaining more weekend night sleep. Previous research supports our findings of shorter TST School for female vs. male adolescents (Jiang et al., 2015; Ming et al., 2016; Ojio et al., 2016) but more research is needed on the between-sex differences in adolescent TST Weekend. The 5.4-min shorter TST School in females compared with males may seem like a modest difference but translates to a 27-min reduction in sleep duration across one week. Moreover, research suggests that even a 4-min increase in TST per night is linked to improved self-reported mental health and fewer emotional problems in adolescents (Chan et al., 2017). Our findings of between-sex differences in sleep duration and timing highlight that female adolescents may be more vulnerable to experiencing sleep insufficiency during the school week than males. Future studies should replicate these sex differences in adolescent sleep duration and timing to determine whether these constitute meaningful differences with regard to emotional health.
We found a mean social jetlag of 2.80 hour in this large sample of adolescents, with no between-sex differences (2.80 hour in females and 2.81 hour in males). Population-level studies of social jetlag in adolescents demonstrate average social jetlag levels around 2.50 hour (Haynie et al., 2018; Vollmer et al., 2017), similar to the current sample. These levels of social jetlag in adolescents contrast sharply with population-level studies in adults, which find that most adults have a social jetlag ≤1 hour (Islam et al., 2018; Koopman et al., 2017; Parsons et al., 2015), translating to about 250% greater social jetlag in adolescents vs. adults. As social jetlag increases with later chronotype (Wittmann et al., 2006) and given that adolescents tend to have a later chronotype than adults (Borisenkov et al., 2012; Carskadon et al., 1993; Randler et al., 2017; Roenneberg et al., 2004), it is unsurprising that adolescents would have greater levels of social jetlag. The evidence indicates that adolescents are a critical population with respect to targeted interventions to lengthen sleep during the school week, decrease social jetlag, and improve associated health outcomes.
Corroborating previous research, we found that females self-reported greater depressive symptoms compared with male adolescents (Avenevoli et al., 2015; Frost et al., 2015; Merikangas et al., 2011; Salk et al., 2016). Though the effect size for the between-sex difference was small (d = 0.23), the mean CES-D depressive symptoms score for female adolescents was 0.66, nearly 27% higher than males (mean score, 0.52). Furthermore, most of our sample (81%) scored ≤1 on the CES-D, which ranges from 0, “strongly disagree,” to 3, “strongly agree” (with depressive statements). Therefore, the level of depressive symptomology was relatively low in our sample. Given that the CES-D does not have a clinical cutoff for depression (Perreira et al., 2005), we cannot conclude that levels of clinical depression meaningfully differed between male and female adolescents in our sample. Moreover, some research suggests females are more likely to self-report depressive symptoms than males, even when clinician-scored depressive symptoms do not differ between the sexes (Compas et al., 1997; Frank et al., 1988; Perugi et al., 1990). Nevertheless, these between-sex differences emphasize the need to address the potential greater prevalence of depressive symptoms in females when implementing interventions for depression in adolescents.
In females, TST School was negatively associated with self-reported depressive symptoms. The results are consistent with studies indicating an association between short sleep and depression in adolescents (Fernandez-Mendoza et al., 2016; Raniti et al., 2016). Contrary to our expectations, males did not exhibit an association between TST School and depressive symptoms. Previous studies have indicated sex differences in the cortisol stress response to sleep loss, which may explain the observed sex differences in the current study. For example, adult women exhibit greater cortisol awakening response (CAR) to sleep loss (Eek et al., 2012; Vargas et al., 2017), whereas adult men exhibit lower CAR after sleep loss (Vargas et al., 2017). Individuals who ruminate before sleep exhibit elevated CAR, indicating that CAR may be a marker of psychological stress (Schlotz et al., 2004). Thus, females, but not males, may experience elevated CAR in response to sleep loss, which is linked to greater psychological stress. There is only one study examining CAR in response to sleep loss in adolescents, the findings of which suggest that short sleep duration is associated with lower CAR in males but not in females (Pesonen et al., 2014). More research is needed regarding the association between sleep insufficiency and CAR in male and female adolescents. These sex differences, along with the findings from the current study, indicate that females may be more vulnerable to experiencing depressive symptoms with insufficient sleep than males.
Interestingly, we found that beyond TST School, social jetlag was positively associated with depressive symptoms in female but not male adolescents. Poor sleep quality is associated with depressive symptoms in females but not males (van Zundert et al., 2013); the current study corroborates the notion that poor sleep health (i.e., social jetlag) affects mood to a greater degree in females than males. Alternatively, given the cross-sectional nature of the current study, it is plausible that females who have later sleep timing on the weekend relative to the school week (i.e., higher social jetlag) exhibit behaviors that are simultaneously associated with later weekend sleep timing and with depressive symptoms. For example, adolescent females tend to ruminate (i.e., dwell on negative thoughts) more than males (Rood et al., 2009); and the association between rumination and depressive symptoms is stronger among adult females than males (Polanco-Roman et al., 2016). Females who exhibit a delayed bedtime on the weekend (relative to the school week) and thus, greater social jetlag, may also ruminate near bedtime on the weekend more than males with a delayed bedtime on the weekend. These bedtime behaviors may result in simultaneous delayed bedtime, social jetlag, and depressive symptoms for females rather than social jetlag causing depressive symptoms per se. Sex differences in rumination near bedtime may be explained by differences in online social media use. Female adolescents are more likely to make technology-based social comparisons than males, and the association between technology-based social comparisons and depressive symptoms is stronger in females than males (Nesi and Prinstein, 2015). Indeed, having an electronic device (such as a mobile phone) in the bedroom or internet use in the hour before bedtime predicts greater social jetlag, and these behaviors are more prevalent in female adolescents (Nesi and Prinstein, 2015; Spilková et al., 2017). Females may be more likely to use social media and delay bedtime on weekend nights (when they are free to stay up later) rather than during the school week. Therefore, in female adolescents, social jetlag may increase the risk for depressive symptoms, be a consequence of depressive symptoms, or have a reciprocal relationship. Future empirical work should determine whether the contributors to social jetlag (e.g., use of social media before bedtime) differ between male and female adolescents.
We found that the moderating influence of sex on the association between social jetlag and depressive symptoms was present only in non-Black adolescents. The disparity between Blacks and non-Blacks in these associations may be explained by differences in the length of the free-running circadian period; in individuals of European ancestry, but not of African ancestry, females exhibit shorter circadian period compared with males (Eastman et al., 2017). It is possible that differences in circadian period between non-Black males and females results in between-sex differences in the associations between social jetlag and emotional health in this population, which do not exist in Blacks. Future studies should probe the interactions between sleep deficiency, sex, and race/ethnicity on other domains of emotional health.
Our findings indicate that shorter TST School and greater social jetlag are associated with an increased risk for depressive symptoms in female but not male adolescents. The current study indicates that female youth with later chronotype who must conform to early school schedules are particularly at risk for social jetlag (Touitou, 2013; Wittmann et al., 2006) and, potentially, depressive symptoms; social jetlag is not associated with depressive symptoms in male adolescents. Future studies should target short TST School and social jetlag to identify and subsequently treat female adolescents at risk for depressive symptoms. The risk of depressive symptoms may be reduced through lengthening sleep on school nights (e.g., going to bed earlier or delaying school start times). Lengthening sleep on school nights may in turn reduce the amount of sleep debt across the week, resulting in lower levels of social jetlag and further reducing the risk of depressive symptoms.
Limitations of the current study include a cross-sectional study design, restricting our ability to determine a temporal order of sleep patterns and depressive symptoms. As mentioned, research demonstrates a reciprocal relationship between short sleep and depressive symptoms (Roberts and Duong, 2014); social jetlag may cause depressive symptoms or may result from other factors, such as greater rumination near bedtime in female adolescents (Polanco-Roman et al., 2016; Rood et al., 2009). Future longitudinal research should determine whether sleep behaviors precede, follow, or have a reciprocal relationship with depressive symptoms in adolescents of both sexes. Furthermore, the depressive symptoms scale employed in the current study (CES-D) does not have a clinical cutoff for major depressive disorder (Perreira et al., 2005). However, due to the large sample size, we were able to identify associations of small effect size according to Cohen’s guidelines (Cohen, 1992) among TST School and depressive symptoms (∆R2 = 0.027) and between social jetlag and depressive symptoms (∆R2 = 0.009) in females. Additionally, we calculated TST as the elapsed time between self-reported bedtime and wake time. However, sleep latency may be longer on school days due to adolescents going to bed before their biological night; thus, it is likely that we overestimated TST, particularly on school nights. Finally, we used survey sleep data, which may be prone to self-report bias, particularly in individuals with poor emotional health (Baillet et al., 2016). The strengths of the current study are its large sample size and the inclusion of demographic and household covariates in regression models (e.g., race and ethnicity, income), which may have been confounding variables (Xing et al., 2011).
In conclusion, we found that shorter sleep duration during the school week and greater social jetlag were associated with depressive symptoms in females, but not males, in a large national sample of adolescents. Though effect sizes were small, our findings indicate female adolescents may be more vulnerable to experiencing negative emotional health accompanying short sleep and social jetlag than are males. Our findings may help explain the nearly two-fold greater prevalence of depression in female compared with male adolescents (Avenevoli et al., 2015; Merikangas et al., 2011). Future experimental and longitudinal research should determine whether increasing sleep duration during the school week and decreasing social jetlag reduce depressive symptoms in female adolescents.
Supplemental Material
JBR_Supplemental_Material_(1) – Supplemental material for Sex Moderates Relationships Among School Night Sleep Duration, Social Jetlag, and Depressive Symptoms in Adolescents
Supplemental material, JBR_Supplemental_Material_(1) for Sex Moderates Relationships Among School Night Sleep Duration, Social Jetlag, and Depressive Symptoms in Adolescents by Gina Marie Mathew, Lauren Hale and Anne-Marie Chang in Journal of Biological Rhythms
Footnotes
Conflict of Interest Statement
The authors have indicated no financial conflicts of interest relevant to the current study. Dr. Lauren Hale receives an honorarium from the National Sleep Foundation for serving as Editor-in-Chief of the journal Sleep Health. Research reported in this publication was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) of the National Institutes of Health under award numbers R01HD073352 (to LH), R01HD36916, R01HD39135, and R01HD40421, as well as a consortium of private foundations. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
