Abstract
Mice are used widely for research on circadian, molecular and other processes; however, little is known of circadian age- and, particularly, sex-related changes that occur over the entire lifespan of this species. To shed light on this question, the authors used a longitudinal design for the first continuous actogram measurements of general circadian locomotor activity rhythms unperturbed by photocycle or other experimental manipulations over the lifespan in male and female C57BL/6J mice. These weaning-to-death actograms are the most inclusive undertaken to date. Comparisons of circadian parameters (phase angle of entrainment, length of daily activity, bout length/intensity) were made among 4 life stages (adolescence, adult, middle age, and senescence). The present data reveal the progressive and sex-related changes in general locomotor activity pattern that occur throughout the lifespan. From the overall perspective of this study, there appears to be a need for wider age and sex representation in circadian research.
Age-related changes in numerous physiological and behavioral aspects of mammalian circadian timekeeping function have long been studied. However, this research has been limited in part to unpaired comparisons between independent age-selected cohorts, which can introduce potential confounds related to exposure to different environmental conditions (i.e., photocycle, food type, chamber noise, laboratory personnel, etc.) prior to experimentation. Another shortcoming is that although mice are used widely for research on circadian, molecular and other processes, little is known of age- and sex-related circadian changes that occur throughout the lifespan. Here, a longitudinal design was used as the first-ever attempt to continuously and noninvasively monitor the daily general locomotor activity pattern of individual male and female mice from weaning (21 days of age) to natural death. Rhythm traits, including length of daily activity (alpha), phase-angle of entrainment, and activity bout intensity and duration, were measured in individually housed mice under a 12:12 LD photocycle using passive infrared detectors interfaced with a ClockLab data acquisition system. Statistical analyses of these traits were undertaken over 4 life stages based on the classification scheme by Flurkey et al. (2007): adolescence (weeks 3-8), adult (weeks 12-24), middle age (weeks 40-60), and senescence (weeks 72-96).
Life-long general locomotor activity measurements were recorded from an initial group of 6 male and 6 female C57BL/6J mice. Representative actograms are presented in Figure 1. Several salient features are evident from these records. First, there is a significant life-stage variation in alpha in both sexes, with increased activity duration during the adult versus other life stages (Fig. 2). Notably, alpha is consistently longer in females over all stages due to a significantly earlier activity onset (especially during adolescence) and later offset throughout life. Second, the phase-angle of behavioral entrainment to lights-off also varies with respect to life stage and sex. During adolescence, activity onset in both sexes occurred ~50 min prior to lights-off. This decreases to ~20 min over subsequent life stages, during which females exhibit a significantly larger phase angle of photic entrainment than males, particularly during middle age (Fig. 2). Third, activity bout analysis also reveals marked life stage and sex differences in activity intensity and duration for both sexes, which decrease significantly during aging (Fig. 3).
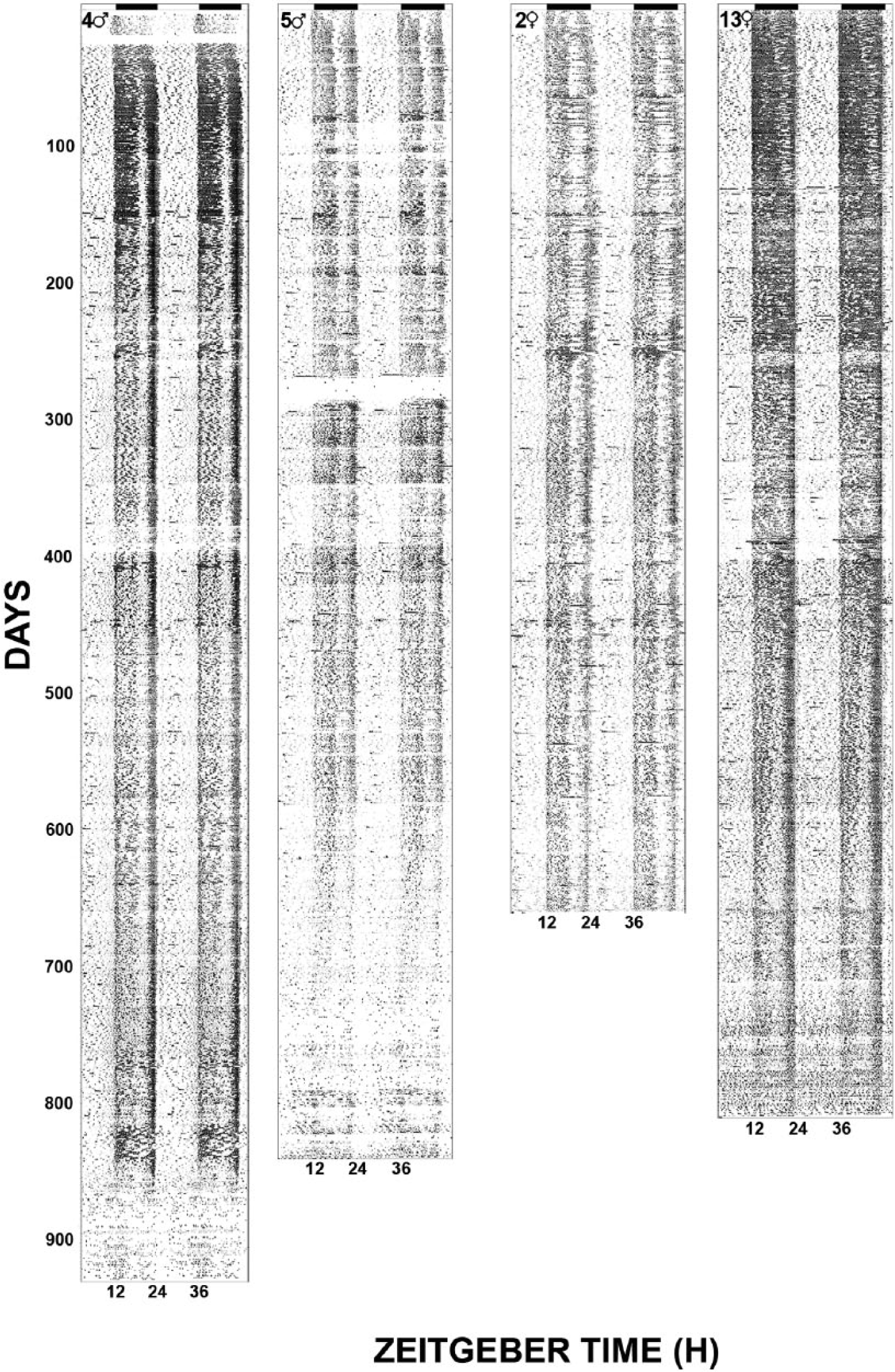
Representative double plotted actograms showing continuously monitored locomotor activity for female and male mice from weaning to natural death. Horizontal filled bars represent the dark phase of the 24-h LD cycle.
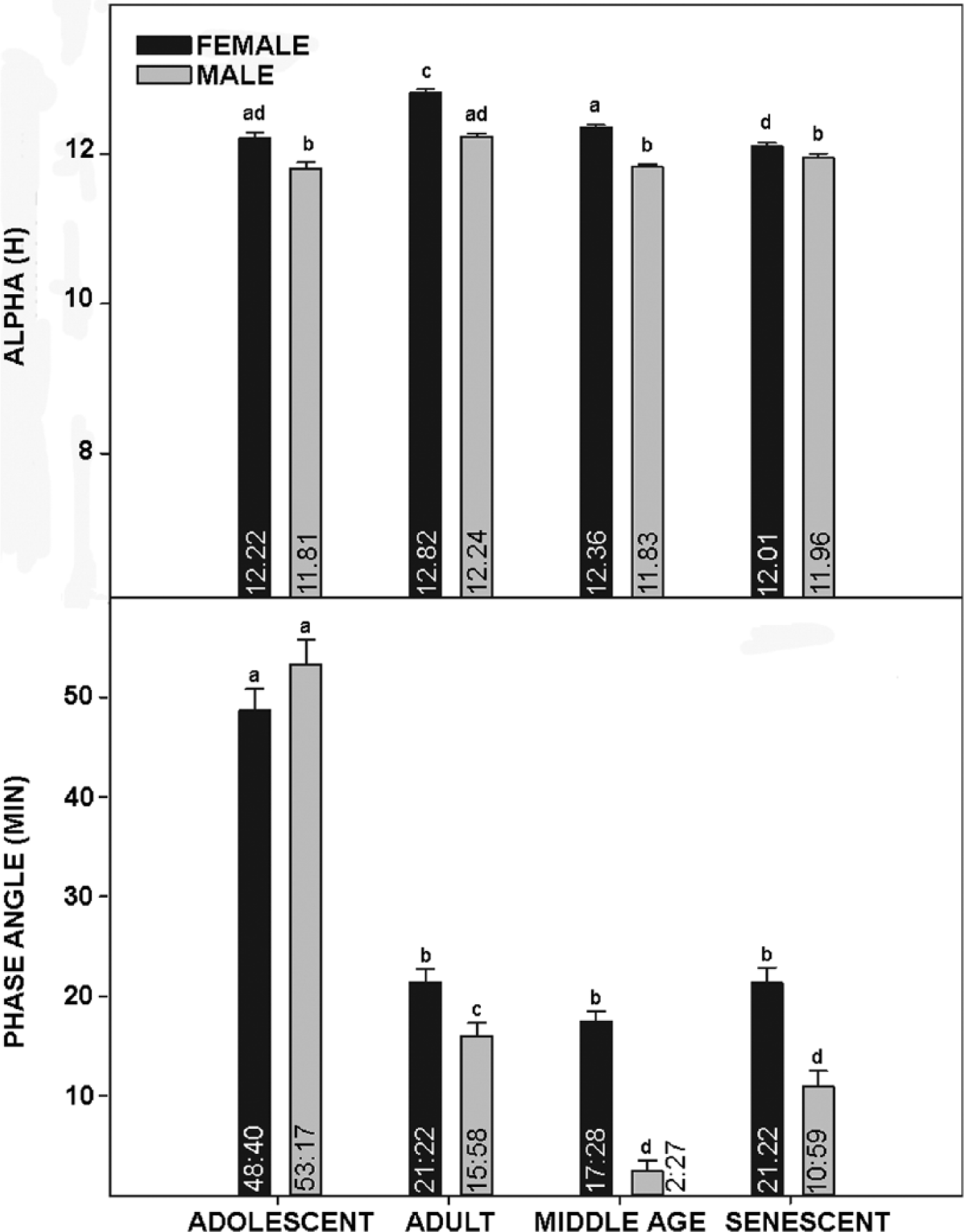
Top: Bar graphs depicting mean length of daily activity (alpha) of female and male mice at 4 life stages. Bottom: Bar graphs depicting phase angle of entrainment for female and male mice at 4 life stages. Means for each group are included inside of the respective bars, and vertical lines above the bars are standard error of the mean. Bars with different letters are significantly different, p < 0.01.
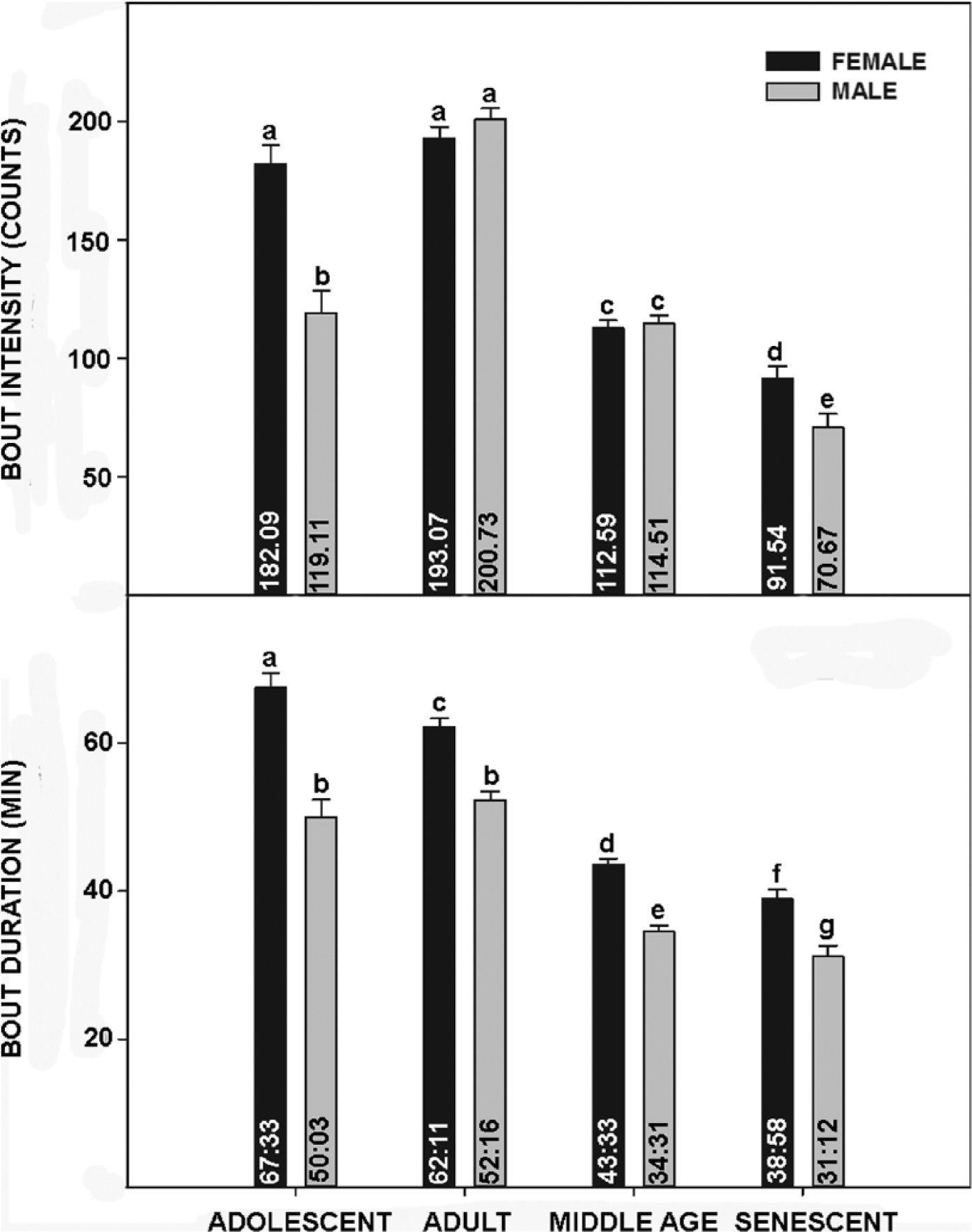
Top: Bar graphs depicting mean bout intensity of female and male mice at 4 life stages. Bottom: Bar graphs depicting bout duration for female and male mice at 4 life stages. Means for each group are included inside of the respective bars, and vertical lines above the bars are standard error of the mean. Bars with different letters are significantly different, p < 0.01.
The present aging data are consistent with those of previous work showing that the mammalian circadian system undergoes significant aging-related changes in the temporal regulation of behavioral and physiological processes. For example, significant alterations in the expression of the human sleep-wake cycle are associated with aging (Witting et al., 1990; van Coevorden et al., 1991; Van Someren, 2000), and the number and duration of activity bouts also vary between young and old people (Lieberman et al., 1989) and mice (Valentinuzzi et al., 1997). Circadian control of the endocrine system is affected by aging, with altered rhythmic expression of testosterone (Bremner et al., 1983), melatonin (Sharma et al., 1989), cortisol, thyroid-stimulating hormone, prolactin, and growth hormone (Sharma et al., 1989; van Coevorden et al., 1991). Other longitudinal circadian analyses have been undertaken to characterize aging effects on free-running rhythms in male (Davis and Viswanathan, 1998) and female (Duffy et al., 1999) Syrian hamsters and in the SAMP8 Alzheimer’s model mouse (Pang et al., 2004). Consistent with the present results, male and female hamsters continuously monitored for approximately 90 weeks displayed decreases in the average level of activity, time active, and bout intensity as they aged (Davis and Viswanathan, 1998; Duffy et al., 1999), although in contrast to the present study the hamsters were maintained in constant dim lighting conditions and assessment of changes was performed only on hamsters older than 30 weeks (to avoid potential confound by gonadal regression/recrudescence) as opposed general locomotor rhythms under LD. These differences are significant because the congruence of these findings suggests that decreasing level, length, and intensity of locomotor activity are independent of the disrupted secretions of melatonin or testosterone. Also consistent with the present findings, female hamsters are more active than males throughout life (Duffy et al., 1999). In the senescence-accelerated SAMP8 mice monitored at 2, 7, and 12 months of age, there was a progressive decrease in general wheel running activity (Pang et al., 2004). Given that these mice are a model of accelerated aging, these findings are consistent with the present results showing age-related reduction in locomotor activity (bout intensity) and support the validity of using SAMP8 mice for aging research, as the same changes occur in normally aging mice and are therefore unlikely to be an artifact of accelerated aging only. The actograms of Figure 1 also indicate that there are apparent aging-related losses in circadian amplitude. However, unlike wheel running, the passive infrared system is subject to variances due to overhead positioning alterations caused by cage changing and possible long-term differences in sensor reception, which confound the quantification of longitudinal amplitude changes.
In the present study, adolescent mice of both sexes exhibited a larger phase angle of entrainment to lights-on compared with all other age-groups, a result that differs from previous measurements on this strain of mice (Valentinuzzi et al., 1997). However, it is consistent with developmental circadian studies undertaken in Degus (Lee et al., 2006) and may reflect a maturation of the circadian timing system that is not completed until the mice reach sexual maturity. In contrast with the findings of Vaentinuzzi et al. (1997), activity bout intensity and duration are greater during adolescence and mature adulthood and decrease substantially with age. These inconsistences may be explained by the use of running wheels to measure activity versus passive infrared used in the present study or the use of shifting LD cycles versus constant LD 12:12 used here.
The present measurements reveal pronounced sex differences in all rhythm traits investigated. Notably, there were significant differences in the expression of these traits throughout all 4 life stages, including alpha, phase angle, and activity profiles. In both sexes, although length of daily activity followed a similar pattern (shorter duration during the adolescence, which increased during the adult stage and decreased back to near adolescent levels in middle age and senescence), females had a longer alpha for all aging stages. Activity bout intensity (episodes per bout across the 24-h day) is also greater in adolescent females versus males. Finally, the duration of activity bouts is significantly greater in females throughout life, and for both sexes this also decreased from adulthood to senescence. In terms of other sex-related circadian effects of aging, males and females exhibit differential age-associated changes in daily cortisol expression (Van Cauter et al., 1996), vasoactive intestinal peptide (VIP) expression within the SCN (Zhou et al., 1995), and the daily sleep-wake cycle (Campbell et al., 1989). It is possible that the sex differences reported here, particularly in alpha and phase-angle, result from fluctuating female sex steroids associated with the estrous cycle. Unfortunately, we could not undertake long-term daily vaginal smear analysis to correlate estrus with behavior, as this intervention would have distorted the general locomotor activity rhythm. However, for 2 reasons we do not think this is the basis for the larger female alpha or phase-angle. First, estrogen treatment does not affect alpha in female mice (Royston et al., 2014). Second, most C57BL female mice are effectively sterile after 12 months (Franks and Payne, 1970), but the increased alpha reported here persists well after reproductive senescence until death. Also, although estrogen does advance activity onset in female mice (Blattner and Mahoney, 2014), the larger phase-angle similarly persists permanently after female reproductive senescence.
A final consideration is that the majority of circadian studies in mice are conducted in young adult males (20+2 weeks [data from Journal of Biological Rhythms publications over the last 5 years]). From the perspective of the present study, it is apparent that this literature, skewed heavily toward young animals, represents only a small portion of total lifespan (see Fig. 1) and is certainly not representative of females. From a translational standpoint, as women live on average ~7 years longer than men and therefore constitute a majority of the most elderly population, it would seem beneficial to consider the inclusion of older mice of both sexes in circadian studies.
Footnotes
Acknowledgements
The authors thank Alex Yaw for her assistance with the preparation of the figures, Caitlin Crosier for her expert animal husbandry, and Bianca Simonassi, Brandon Boesiger, and Lauren Misja for their technical assistance in data analysis.
Conflict of Interest Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
