Abstract
Eating at night time, as is frequent in shift workers, may contribute to increased cardiovascular disease (CVD) risk through a disruption in usual lipid metabolism, resulting in repeated and sustained hyperlipidemia at night. This systematic review aimed to investigate the impact of eating a meal at night compared with the same meal eaten during the day on postprandial lipemia. Six databases were searched: CINAHL Plus, Cochrane Library, EMBASE, Ovid MEDLINE, Informit, and SCOPUS. Eligible studies were original research cross-over design with a minimum fasting period of 5 h before testing preceded by a standardized control meal; measured postprandial triacylglycerol (TAG) for 5 h or greater; had meal time between 0700 h and 1600 h for day time and between 2000 h and 0400 h for night time; and had within-study test meals (food or drink) that were identical in macronutrient composition and energy. Two authors independently completed eligibility and quality assessment using the American Dietetic Association Quality Criteria Checklist for Primary Research. After removing duplicates, 4,423 articles were screened, yielding 5 studies for qualitative synthesis. All studies identified at least one parameter of the postprandial TAG response that was different as a result of meal time (e.g., the total concentration or the time course kinetics). Two studies reported a greater total TAG concentration (area under curve) at night compared with day, and 3 studies found no difference. Four studies reported that the kinetics of the postprandial time course of TAGs was different at night compared with during the day. Inconsistent reporting in the primary studies was a limitation of the review. Night eating may negatively affect postprandial lipemia and this review shows there is a need to rigorously test this using standardized methods and analysis with larger sample sizes. This is critical for informing strategies to lower CVD risk for shift workers.
The increasing demands of globalization and our rapidly evolving 24-h society have resulted in approximately 20% of the population in industrialized countries engaging in shift work (Boivin and Boudreau, 2014). Shift work is defined as working outside the conventional hours of 0600 h to 1800 h and may include work in the early morning, evening, and overnight (Institute for Work and Health, 2003; McMenamin, 2007). Shift work is associated with numerous negative health outcomes and has been identified as an independent risk factor for cardiovascular disease (CVD) (Brown et al., 2009; Tuchsen et al., 2008). Compared with day workers, shift workers are also at an increased risk of vascular events (Vyas et al., 2012). The evidence that shift work increases the risk of type 2 diabetes is also compelling, with a recent meta-analysis reporting that male shift workers have a 37% increased risk of diabetes development compared with day workers (Gan et al., 2015). Health problems within this working population are therefore of economic, industrial, and community importance.
Humans are diurnal (active during the day and rested during the night) with internally generated circadian rhythms of approximately 24 h (Antunes et al., 2010). These circadian rhythms are hierarchically organized, with a master circadian clock located in the suprachiasmatic nuclei (SCN) of the hypothalamus and peripheral clocks located in many other tissues and cells of the body (Gooley et al., 2006). The SCN is entrained by the light/day cycle in the external environment and plays an important role in synchronizing behavioral and metabolic rhythms, such as the sleep/wake cycle, hormone secretion, and the fast/feed cycle (Hastings et al., 2003; Schibler et al., 2003) via the coordination of peripheral circadian clocks. This high level of synchronization between central and peripheral clocks ensures that these aforementioned rhythms are optimal for physiological functions occurring at the most appropriate time of the day (Johnston, 2014).
Many nutritionally driven metabolic processes, such as digestion, follow a circadian pattern, and changes in the timing of food intake can influence internal clocks in peripheral organs involved in nutrient metabolism, such as the liver, kidney, and pancreas (Damiola et al., 2000). This uncoupling of the central clock from these peripheral clocks can disrupt the circadian system, which manifests as an abnormal metabolic response to food intake (Johnston et al., 2016). In a small number of well-controlled simulated shift-work studies, eating at night time compared with during the day were associated with poor glucose tolerance and dampened insulin sensitivity (Gibbs et al., 2014; Leung et al., 2017; Morgan et al., 1998; Saad et al., 2012) possibly contributing to the increased risk of type 2 diabetes observed in the shift working population. The impact of night-time eating on lipid metabolism is less clear.
Under normal circadian alignment, circadian genes control lipid absorption and are involved in the regulation of lipid biosynthesis (Gnocchi et al., 2015). Data from animal studies (circadian mutant mice) show that the adipose tissue “clock” regulates lipid storage and mobilization (Paschos et al., 2012; Shostak et al., 2013), ensuring that daily rhythms in lipid absorption, storage, and transport are temporally coordinated with activity and rest cycles (Bailey et al., 2014). In humans, circadian disruption impacts circulating triacylglycerol (TAG) levels, lipid transport, and fat storage (Hampton et al., 1996; Romon et al., 1997), and, under simulated shift work conditions, participants present with a prolonged postprandial lipid response at night (Al-Naimi et al., 2004). The effects of a delayed removal of TAG-rich lipoprotein in the postprandial period (Groot et al., 1991) may contribute to the elevated risk of CVD observed in shift workers (Knutsson et al., 1986). The exact physiological mechanisms are not yet fully understood, and there are no data examining time of day differences in postprandial lipemia in animal studies, but potential mechanisms proposed include meal-induced inflammation and infiltration of the vascular wall by remnant triglyceride lipoproteins (Emerson et al., 2016; Jackson et al., 2012).
Given the large proportion of individuals undertaking shift work, and the increasing number of people who eat at irregular times, research is warranted to understand the interaction between meal timing on postprandial lipemia (i.e., high blood TAG or other lipids) owing to its identification as an independent risk factor for CVD (Chan et al., 2013). This systematic review, therefore, aimed to investigate the impact of eating at night compared with eating during the day on postprandial lipemia.
Methods
This review is reported according to the Preferred Reporting Items for Systematic reviews and Meta-Analysis statement (PRISMA) (Liberati et al., 2009).
Literature Search
The search terms (Table 1) were developed in conjunction with the subject librarian for the Faculty of Medicine, Nursing and Health Sciences at Monash University (April 16, 2016). Three sets of search terms were used (Table 1), combined with “AND”. This review was commenced on April 23, 2016, when the initial searches were undertaken. After this initial screening, one inclusion criteria set a priori was adjusted to maximize the inclusion of relevant studies. This criterion was the minimum fasting time prior to postprandial testing, which was revised from a minimum of 8 h to a minimum of 5 h. Due to a change in staff, there was a delay in re-running the searches. The search was re-run on June 13, 2018, to identify articles investigating the impact of night time eating on postprandial lipemia in healthy participants using 6 electronic databases: CINAHL Plus, Cochrane Library, EMBASE, Ovid MEDLINE, Informit, and SCOPUS. The full search details in the Medline dataset are presented in Supplemental Table 1.
Systematic literature review search terms.

represents any group of characters, including no character that is a variant of the word, for example, Eat* would search for eat and eating.
Eligibility Criteria
Original research conducted in healthy adults with a BMI in the range of 18.5 - 30 and not known to have any metabolic diseases published in peer-reviewed journals in English were included for review. Abstracts, conference posters, reviews, and unpublished research were excluded. Retrieved abstracts were reviewed against the following criteria: 1) crossover design with an identical night time and day time test meal; 2) postprandial TAG were measured for a minimum of 5 h, as the usual duration of the postprandial hyperlipidemic period is 5 to 8 h (Lairon et al., 2007); 3) fasting period before the intervention meal was a minimum of 5 h; and 4) control and test meals (food or drink) were identical in macronutrient and energy composition (isocaloric). For a study to be eligible, the day time point was between 0700 h and 1600 h and the night time point was between 2000 h and 0400 h.
Selection Process
Duplicate and irrelevant articles were excluded based on abstract and title by one author (EK). Articles deemed eligible for full-text review were retrieved and screened against inclusion criteria by 2 authors (EK and MPB). Articles were included if they met all the above mentioned inclusion criteria. If consensus between the 2 reviewers could not be reached, a third reviewer made a final decision (CEH).
Data Extraction
Data were extracted independently by 2 authors (EK and TLS) and entered into a spreadsheet for analysis. Information extracted included study design, sample size, demographic characteristics of the participants, control and test meal composition for day and night time comparator, time of test meal, postprandial duration and period of fasting before test meals, baseline and postprandial TAG concentration estimated using the area under the curve (AUC), the method used to determine AUC or incremental area under the curve (iAUC) and the P value pertaining to time of meal (day vs. night) difference. Secondary data included the kinetics of the postprandial time course and summary measures (P value) of changes in other circulating lipids that were reported in the included studies to contribute to characterizing the postprandial lipemic profile (total cholesterol, LDL and HDL cholesterol).
To obtain data that were not available in the published paper, authors were contacted twice; however, no additional data were obtained, including fasting TAG in 2 studies (Hadjadj et al., 1999; Romon et al., 1997) and postprandial AUC TAG concentrations for 2 studies (Le Fur et al., 1999; Lund et al., 2001). If data were not available, but differences between day and night responses were reported narratively in the text, studies were retained for the qualitative synthesis.
Risk of Bias Assessment
Risk of bias within studies was assessed using the American Dietetic Association (ADA) Quality Criteria Checklist for Primary Research (Williams et al., 2011). Studies were classified as positive, neutral, or negative after assessment. For the study to be classified as positive, 4 major items required a positive response: 1) freedom from selection bias; 2) comparable study groups; 3) clear description of exposure factor and comparison, which, in this case, was day and night; and 4) clearly defined outcomes determined by valid and reliable measurements. Studies graded as negative quality were deemed ineligible for inclusion in the qualitative synthesis. Two reviewers (EK and CEH) independently assessed the quality of each included study and when consensus was not reached this was resolved by a third reviewer (MPB).
Clear criteria to assess the risk of bias across studies was not defined a priori, as the type of studies included in this review are not routinely registered with trial registries. All included studies were carefully reviewed for evidence of selective reporting of outcomes; i.e., outcomes listed in the methods section of the included studies compared with those for which results are presented, and this was included in assessment of Item 7 of the quality criteria checklist assessment.
Qualitative Data Synthesis
The primary outcome was differences in postprandial (AUC or iAUC) TAG concentrations between day and night. Fasting TAG values are reported, as they are known to affect postprandial levels. Where studies also reported on the kinetics of the time course, any significant differences are reported as secondary outcomes. If the included studies also reported on other postprandial lipids, the key changes (i.e., significantly different, as determined by P value) are presented to provide a more complete picture of this impact on lipemia. The probability value (P value), where available, was used to report significant differences between day and night outcomes for each study. Narrative descriptions of differences were assumed to be accurate when P values were not reported.
Results
Study Selection
Figure 1 outlines the selection process of the included studies. The search yielded 6,706 results. After removing duplicates and articles that did not meet the eligibility criteria by title and abstract screening, 75 studies were retrieved for full-text review and assessed for inclusion. Six of these papers were identified as having met the eligibility criteria, representing 5 studies, as one study was reported in 2 papers (Guerci et al., 2000; Hadjadj et al., 1999). The data from the study by Hadjadj et al. (1999) was used in the qualitative synthesis, as they reported the AUC data. The reasons for excluding articles at the full-text review stage included: population characteristics, meal times (night meal time before 2000 h or daytime meal after 1600 h), meals not being isocaloric, fasting time <5 h, TAG not reported as an outcome, and postprandial measurement times <5 h (Refer to Figure 1).
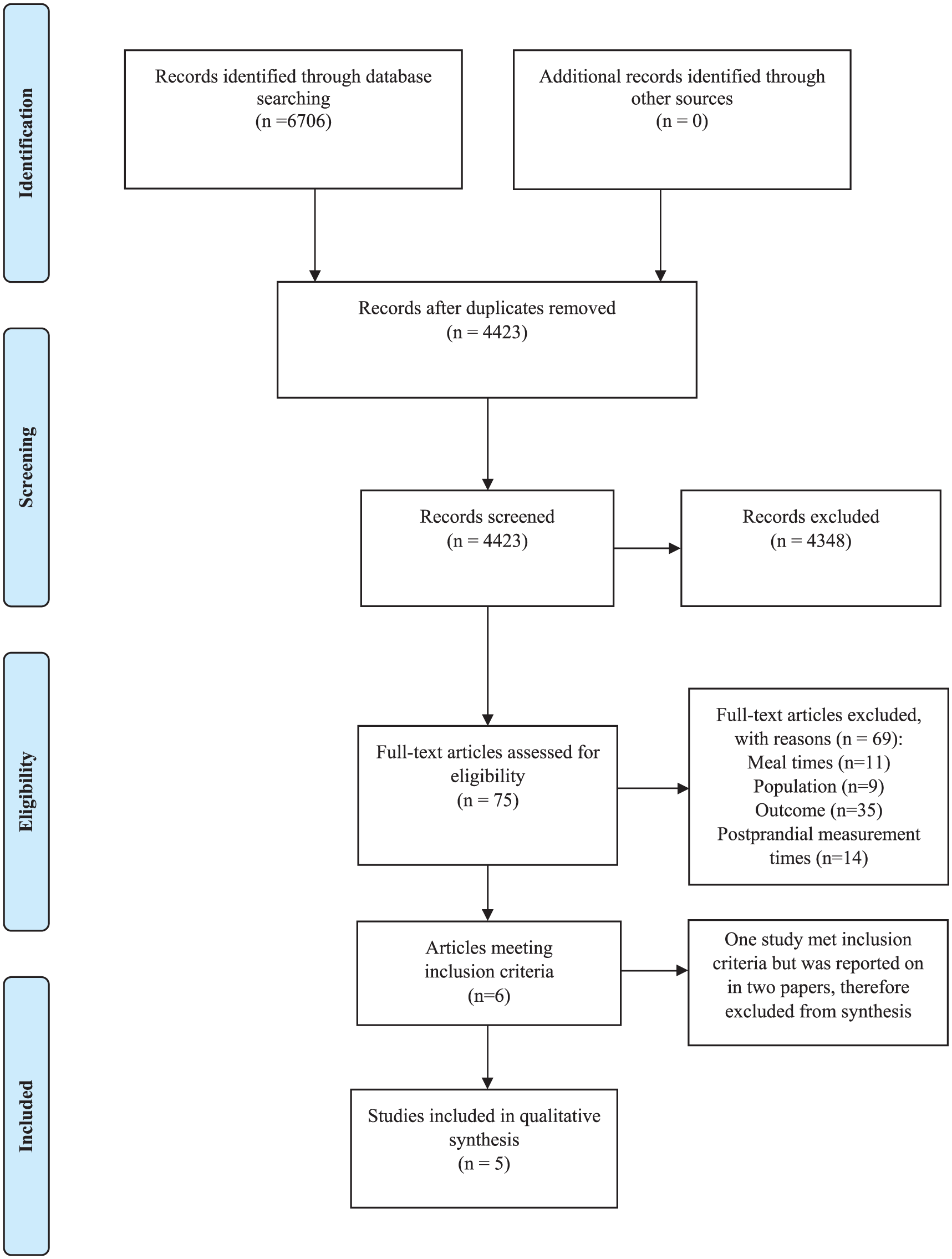
PRISMA flow chart of the review process. Six papers were identified as having met the study criteria, representing 5 studies, as one study was reported in 2 papers.
Study characteristics, including study design, participant characteristics and intervention protocols were extracted for each included study. The high levels of heterogeneity between the 5 study protocols and missing data precluded a meta-analysis.
Description of Included Studies
The characteristics of the included studies are presented in Table 2. The included studies were all of a cross-over design with one study having multiple interventions (Le Fur et al., 1999). Le Fur et al. (1999) compared day and night time eating with and without a mental stressor; in this review, we only report the day and night comparisons without the mental stress. Of the 5 studies included, 3 were conducted in men. Two studies included both males and females, of which one aimed to investigate sex-differences in postprandial TAG (Sopowski et al., 2001). Sample size ranged from 9 to 25; this results in a combined total of 73 participants, of which most were male (n = 61). One of the studies was undertaken in a shift working population in field conditions, and meal responses were tested during day shift, during night shift at the beginning of a period of night-shift work (second night shift), and on day shift 2 days following the night shift period. Three studies simulated night shift and kept participants awake overnight, whereas the study by Hadjadj et al. (1999) permitted participants to sleep during the night session. Two studies specifically excluded shift workers and 2 did not specify working shift work as an exclusion criterion. Blood sample collection varied between 7 and 10 h, with blood collection time points differing between studies. The age range of participants was 19 to 33 years.
Study characteristics of acute postprandial studies included in qualitative synthesis
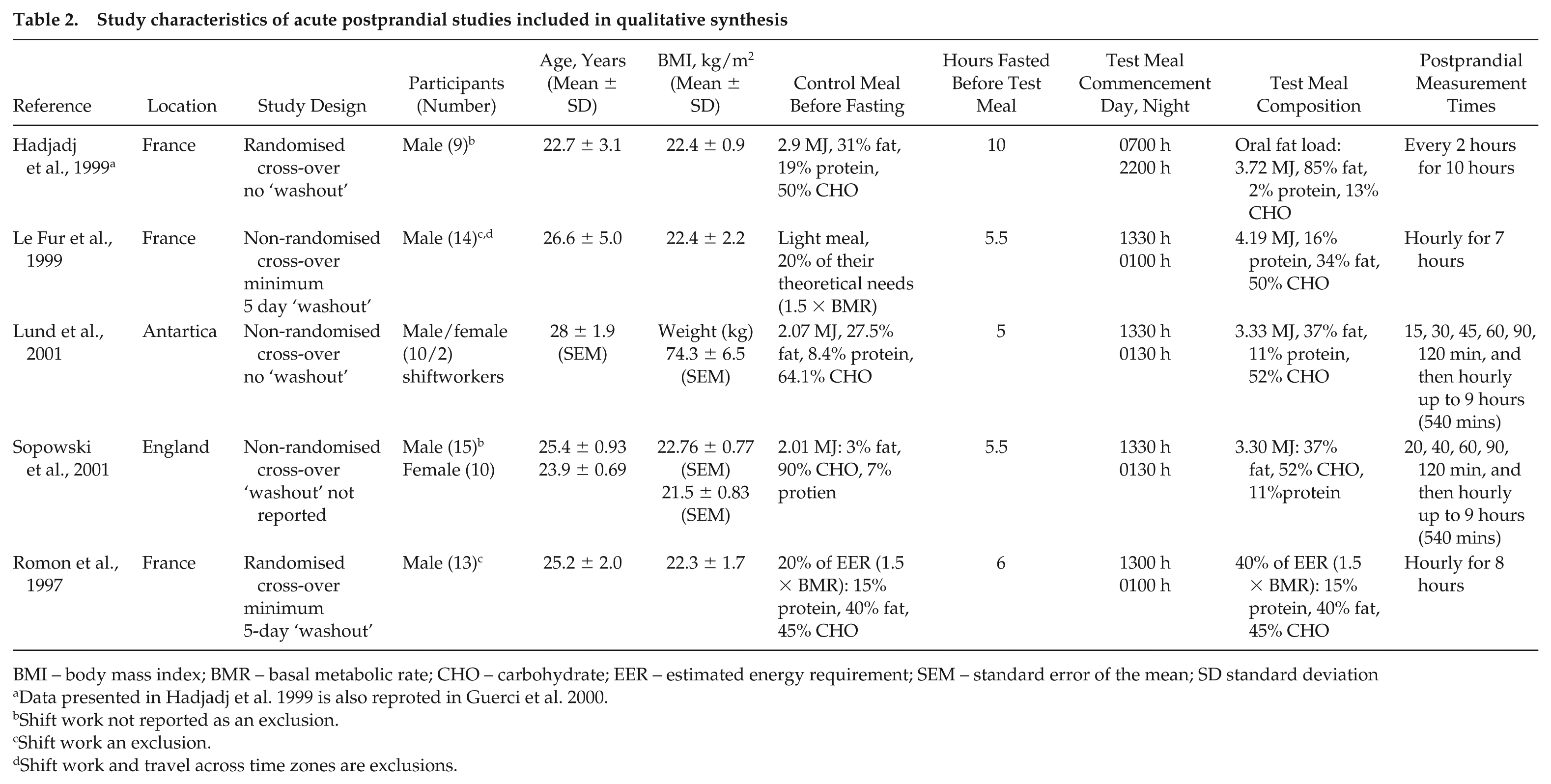
BMI – body mass index; BMR – basal metabolic rate; CHO – carbohydrate; EER – estimated energy requirement; SEM – standard error of the mean; SD standard deviation
Data presented in Hadjadj et al. 1999 is also reproted in Guerci et al. 2000.
Shift work not reported as an exclusion.
Shift work an exclusion.
Shift work and travel across time zones are exclusions.
All studies included the administration of a standardized meal before commencement of the fasting period and a test meal at the completion of the fasting period. Four studies used solid test meals and one used an oral fat load (Hadjadj et al., 1999). The energy content of the control meals and test meals varied between the studies (Table 2). The times of meal administration also varied between the studies, but all were within the day and night timeframes specified in the inclusion criteria. The fasting periods also varied across studies.
Quality Assessment (Risk of Bias)
Based on the Academy of Nutrition and Dietetics (2016) quality assessment checklist, 3 studies received a positive quality rating (Hadjadj et al., 1999; Le Fur et al., 1999; Romon et al., 1997) and 2 were rated neutral (Lund et al., 2001; Sopowski et al., 2001). Selection bias and not specifying the inclusion criteria were the main factors separating neutral from positive studies. Table 3 summarizes the information on the quality assessment process.
Quality assessment of studies investigating the effect of night time and daytime eating on postprandial lipid metabolism.
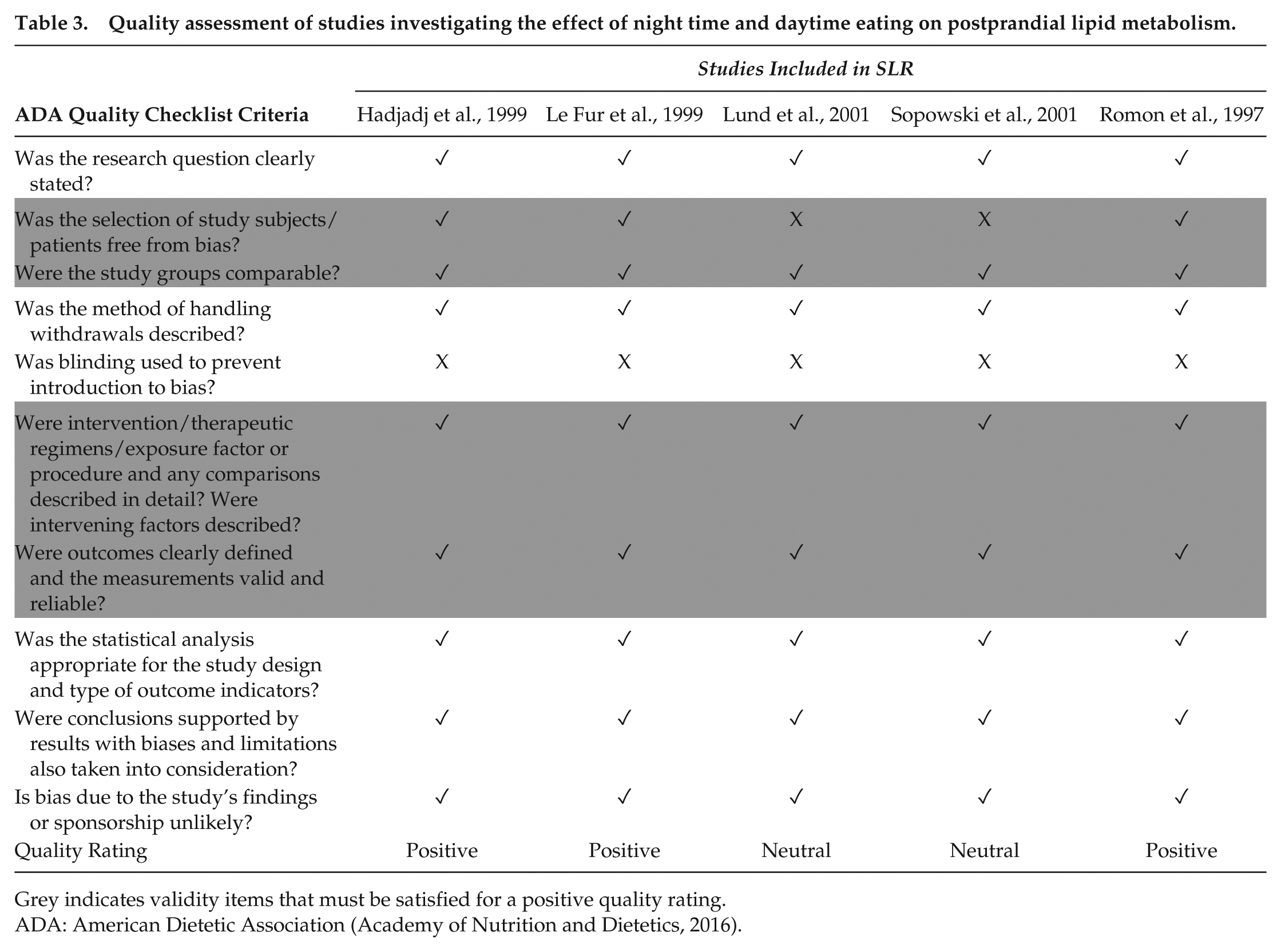
Grey indicates validity items that must be satisfied for a positive quality rating.
ADA: American Dietetic Association (Academy of Nutrition and Dietetics, 2016).
Comparison of Diurnal and Nocturnal Postprandial Circulating TAG Concentration
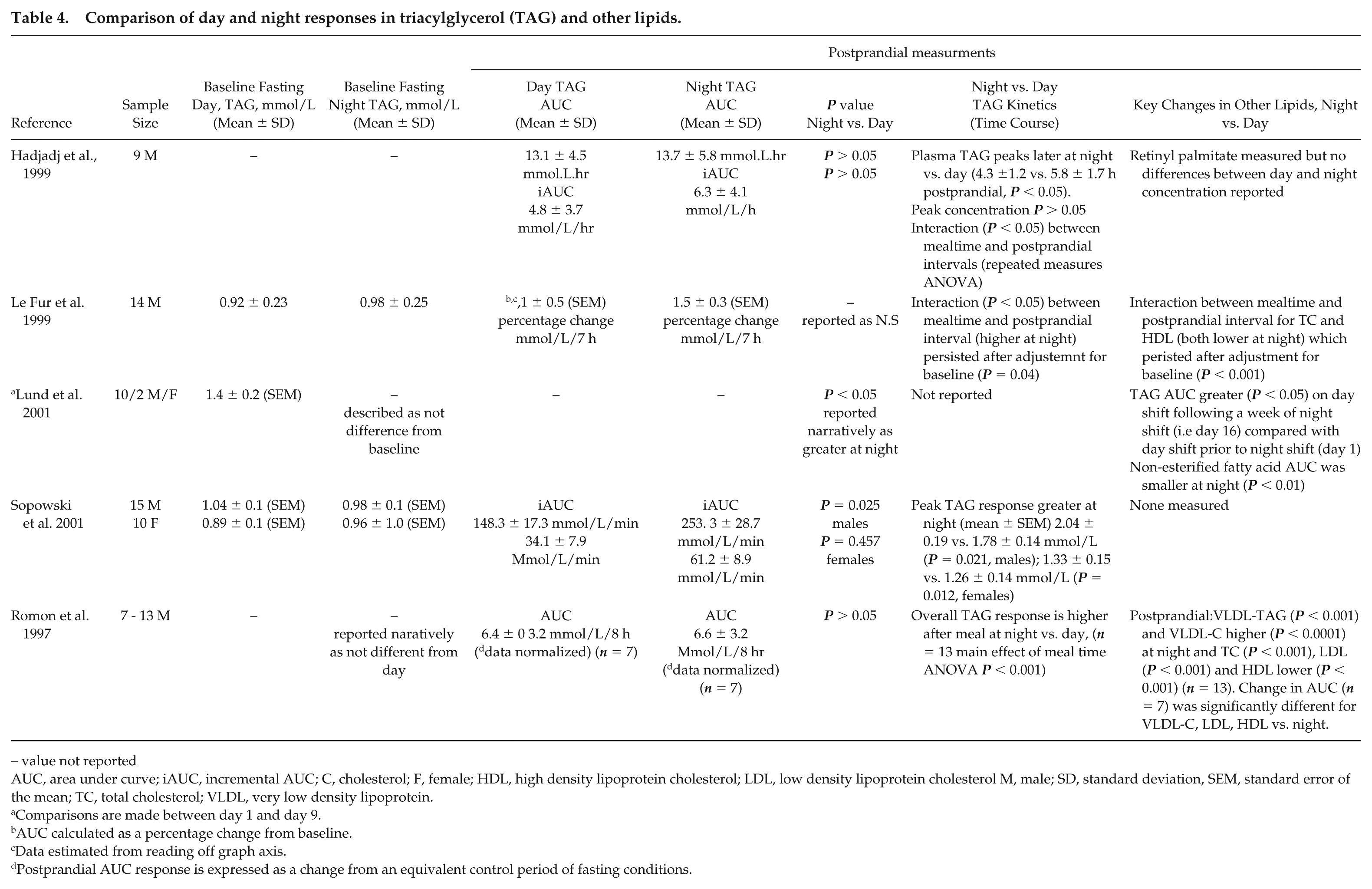
Four of the 5 included studies quantified circulating TAG concentrations by deriving the AUC (summarized in Table 4). Two studies (Lund et al., 2001; Sopowski et al., 2001) found a statistically significant difference in AUC, both reporting a higher concentration at night compared with during the day (Table 4); however, this difference was only significant in males and not females in the study by Sopowski et al. (2001). Le Fur et al. (1999) and Romon et al. (1997) reported no significant difference in AUC. The methods used to calculate AUC varied across studies. Hadjadj et al. (1999) and Lund et al. (2001) applied the trapezoidal rule. Hadjadj et al (1999) and Sopowski (2001) calculated incremental AUC (iAUC). The study by Le Fur et al. (1999) calculated AUC as a percentage change from baseline (i.e., fasting value at time point zero) before test meal consumption. In the study of Romon et al. (1997), the postprandial TAG concentration was compared in 2 ways: the first compared the postprandial time course at night with day (n = 13) and reported a significant main effect of meal time (P < 0.001). Visual inspection of the time course curve shows that the night time concentrations are higher than the day time concentrations. In the same study, 7 participants also completed an 8-h fasting protocol during the day and during the night. These responses were used as a control condition to compare with postprandial responses. AUC was calculated for all 4 experiments and the postprandial AUC was normalized to the fasting AUC for each day and night condition. There was no difference in the change in AUC (fasting-postprandial) between day and night.
Comparison of day and night responses in triacylglycerol (TAG) and other lipids.
– value not reported
AUC, area under curve; iAUC, incremental AUC; C, cholesterol; F, female; HDL, high density lipoprotein cholesterol; LDL, low density lipoprotein cholesterol M, male; SD, standard deviation, SEM, standard error of the mean; TC, total cholesterol; VLDL, very low density lipoprotein.
Comparisons are made between day 1 and day 9.
AUC calculated as a percentage change from baseline.
Data estimated from reading off graph axis.
Postprandial AUC response is expressed as a change from an equivalent control period of fasting conditions.
Comparison of Diurnal and Nocturnal Postprandial Circulating TAG Time Course Kinetics
Four of the 5 studies reported on differences in the postprandial TAG time course curve kinetics between day and night (summarized in Table 4). Two studies (Le Fur et al., 1999; Hadjadj et al., 1999) reported a significant interaction between meal time and postprandial interval, indicating differences in the kinetics of the TAG postprandial curve between day and night. Conversely, Romon et al. (1997) did not find a significant interaction, only a main effect of meal time (as described above). For the remaining 2 studies, it is unclear if the interaction between meal time and postprandial interval was assessed. Two studies investigated the peak TAG response (Hadjadj et al., 1999; Sopowski et al., 2001). Hadjadj et al (1999) analyzed the time taken (h) to reach the peak concentration, and found the peak was reached significantly later than during the day, but found no difference in the peak concentration (mmol/L). In contrast, Sopowski et al. (2001) reported the peak concentration (mmol/L) of circulating TAG was higher at night for both males and females, compared with during the day.
Comparison of Fasting TAG Response in the Morning Compared with the Night Time
Four studies reported on fasting TAG under diurnal and nocturnal conditions (Le Fur et al., 1999; Lund et al., 2001; Romon et al., 1997; Sopowski et al., 2001) (Table 4). Measurements were made from 1 or 2 samples (<15 min apart and averaged) and no significant differences were found. Hadjadj et al. (1999) only reported pooled fasting values. In the study of Romon et al. (1997), a control fasting experiment was performed under diurnal and nocturnal conditions in half of the participants (n = 7). Blood samples were taken over 8 h and a significant interaction between time of day and fasting response was found.
Other Notable Lipid Responses between Diurnal and Nocturnal Conditions
Table 4 also summarizes postprandial changes in other key lipids. Three studies (Le Fur et al., 1999; Lund et al., 2001; Romon et al., 1997) reported on other lipid changes during the postprandial period. Two studies (Le Fur et al., 1999; Romon et al., 1997) reported a significantly lower postprandial total cholesterol and HDL-cholesterol at night compared with during the day. Lund et al. (2001) reported lower non-esterified fatty acids circulating at night compared with during the day.
Discussion
Long-term postprandial lipemia is an independent CVD risk factor (Karpe, 1999). Metabolism is, in part, regulated by circadian rhythms, which predict the feed/fast cycle. Consuming food outside of the predicted cycles can negatively impact on postprandial metabolic responses. The aim of this systematic review was to explore the effect of time of day (day vs. night) on postprandial circulating TAG response. Overall, this search found that few studies (n = 5) have been published characterizing the postprandial TAG response to meals consumed at night compared with day. All included studies identified at least one parameter of the postprandial TAG response (e.g., the total concentration or the time course kinetics) that changed when the meal was consumed at night compared with during the day, but a definitive day vs. night response cannot be clearly determined.
Lipid metabolism is under circadian control in several metabolic organs, including the liver and adipose tissue (Gooley, 2016). Disruption of these molecular clocks can result in lipid dysregulation, which may manifest as an increased production of lipoproteins rich in triglycerides, decreased clearance of lipoprotein-rich TAGs, increased TAG synthesis as a result of an increased availability of fatty acids from peripheral tissue (Barter et al., 1971; Schlierf and Dorow, 1973) and/or a slower clearance of lipoproteins (Fainaru et al., 1994). Furthermore, the relative insulin resistance observed at night time—well described in similar acute trials (Van Cauter et al., 1997)—has been associated with a reduced catabolism of lipoprotein-rich TAGs by lipoprotein lipase (Cassader et al., 2001) and may favor VLDL secretion (Choi and Ginsberg, 2011), which underpins the increased circulating TAG levels of hepatic origin (Morgan et al., 1998). Circulating levels of TAG that exceed concentrations of 1.5 mmol/l are reported to precipitate the generation of atherogenic small LDLs (Griffin et al., 1999). The study by Lund et al. (2001), which was undertaken in shift workers, reported mean postprandial TAG levels immediately after night shift that exceeded this threshold of 1.5 mmol/l (Table 4). These findings support the epidemiological data indicating shift work is a risk factor for vascular disease (Vyas et al., 2012).
The studies included in this review examined the impact of the solar clock time (i.e., day and night) on postprandial responses and are in agreement with other studies that examined postprandial responses at the one clock time (1330 h) before and after experimentally induced circadian disruption (Hampton et al., 1996; Ribeiro et al., 1998). In these experimental studies, bright light administration and altered sleep times are used to gradually advance or “phase shift” over 3 days and then sustained for a further 3 days. Both these studies reported a delay in the postprandial peak of TAG after the phase shift compared with the normal circadian phase.
Overall, whilst all included studies found some difference in the postprandial lipid response (AUC or time course kinetics), there was not a single parameter that changed consistently across studies. Moreover, there was no consistency in the reporting of these outcomes; i.e., some studies found difference in AUC, whereas others reported differences in kinetics. The different findings across studies may in part, be related to the different meals provided in each study. Across studies, the macronutrient profiles and total energy content of the test meal and the control meal differed. TAG metabolism can be influenced by previous macronutrient consumption, particularly fat and carbohydrate intake, if consumed on the same day or even consumed the night before a test meal challenge (Lambert and Parks, 2012). Previous studies have identified that males and females have different postprandial responses to high fat meals (Kolovou et al., 2006; Kovar and Poledne, 2000); yet only one of the included studies recruited an approximately equal numbers of male (n = 15) to female (n = 10) participants (Sopowski et al., 2001). Sopowaki et al. (2001) found that males (but not females) had a higher postprandial TAG concentration (iAUC) at night compared with day; however, the peak TAG concentration was greater at night compared with day for both sexes. As such, the impact of meal timing on postprandial lipid metabolism in females is poorly described in the literature.
The data analysis approach differed across studies and this may also contribute to the inconsistent findings on estimates of total concentration. For example, 4 different approaches to calculating AUC were found across the 5 studies and one study did not present AUC data. The time-course kinetic data were also not consistent, with some studies only reporting the overall pattern of the time course and other studies examining specific parameters such as peak amplitude and time to peak amplitude. The physiological consequences of the altered postprandial responses at night have not been elucidated; however, other studies of lipemia indicate increased peroxidation and inflammatory responses, which can contribute to CVD risk (Dias et al., 2017). This review is limited in terms of the generalizability of the findings due to the small number of studies (mostly from Europe) investigating the impact of eating at night on postprandial lipids.
The inconsistencies in study design, including period of fasting, nutrient composition of the test meals, meal timing and participant characteristics, have hampered the elucidation of the effect of night time eating on postprandial TAG metabolism. The results from this review provide some indication of an impairment of lipid metabolism throughout the night (relative to day responses) and generate a potential mechanism for the increased vascular risk observed in shift workers; however, a comprehensive study is needed to confirm this proposition. Importantly, this review establishes a need for standardized protocols for investigating postprandial lipid metabolism in order for results to be used meaningfully within a preventative context. For example, the development of dietary advice that may mitigate the increased risk of CVD that shift workers experience purely as a result of their occupation.
Supplemental Material
SupplementaryTable1 – Supplemental material for Effect of Night Time Eating on Postprandial Triglyceride Metabolism in Healthy Adults: A Systematic Literature Review
Supplemental material, SupplementaryTable1 for Effect of Night Time Eating on Postprandial Triglyceride Metabolism in Healthy Adults: A Systematic Literature Review by Maxine P. Bonham, Elleni Kaias, Iona Zimberg, Gloria K. W. Leung, Rochelle Davis, Tracey L. Sletten, Hazel Windsor-Aubrey and Catherine E. Huggins in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors would like to acknowledge Professor Andrew Sinclair for editorial assistance with the manuscript. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest Statement
The authors alone are responsible for the content and writing of this article. The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
