Abstract
Artificial light in modern society has led to the ubiquity of light exposure at night as individuals work night shifts and use light-emitting electronic devices before bedtime. These aberrant light conditions have detrimental consequences on cognitive and mental health, compelling the need to understand the mechanisms by which light affects brain functions. Although it was believed that aberrant light impairs health by first disrupting circadian rhythms and sleep, we showed that chronic exposure to a light cycle termed T7 (3.5 h of darkness, 3.5 h of light) caused mood and learning dysfunction in adult mice independent of sleep deprivation or circadian arrhythmicity, suggesting the direct effects of aberrant light on brain function. However, the mechanisms by which light directly causes mood and learning dysfunction remain poorly understood. In this study, we sought to determine whether exposure to the T7 cycle disrupts adult hippocampal neurogenesis, given that suppressed neurogenesis has been correlated with mood and learning dysfunction. After exposing adult mice to the T7 light cycle, we analyzed adult hippocampal neurogenesis by examining cellular proliferation and number of adult-born neurons. Contrary to our hypothesis that T7 would suppress neurogenesis, we found that adult mice exposed to 2 or 10 weeks of the T7 light cycle did not exhibit deficits in hippocampal neurogenesis. Our findings suggest that the direct effects of light on mood and learning do not depend on adult hippocampal neurogenesis.
Light is one of the most fundamental sensory inputs that influence all aspects of day-to-day health and physiology. The presence of natural light from the sun calibrates internal circadian rhythms to the external 24-h light/dark cycle, allowing organisms to prepare for expected changes in the environment. Health problems arise when the internal clock is not synchronized to the outside light/dark cycle. Such misalignments are common in modern society because of the advent of artificial light. Aberrant light schedules such as those due to shift work cause sleep loss, cognitive and psychiatric deficits, and increased risk of cardiovascular disease (Wagge et al., 2014; Chang et al., 2015; Scheer et al., 2009). Approximately 7% of all wage and salary workers work the evening shift (McMenamin, 2007), and 9 out of 10 Americans reported using electronic devices an hour before bed (Gradisar et al., 2013), indicating that a significant portion of Americans are at risk of developing health problems associated with exposure to aberrant light schedules. The ubiquity of aberrant light schedules and their destructive effects on health compel the public health need to investigate the influence of aberrant light on physiology.
It has been widely assumed that aberrant light affects health by first disrupting circadian rhythms and sleep, which are well-known mediators of mood and cognition (Pilcher and Huffcutt, 1996; Emens et al., 2009). However, we discovered a light cycle that challenges this assumption. Termed T7 (3.5 h of dark, 3.5 h of light), this aberrant light cycle presents light at all circadian phases without causing sleep deprivation or circadian arrhythmicity (LeGates et al., 2012), allowing us to assess the effects of direct light exposure independent of sleep and circadian rhythms. We found that chronic exposure to the T7 cycle caused depression-like behaviors and learning deficits in adult mice (LeGates et al., 2012), highlighting the direct effects of light on mood and learning function. However, the mechanisms by which the T7 cycle causes mood and learning dysfunction remain poorly understood.
Several studies in rodents have implicated adult hippocampal neurogenesis in mood and learning functions. Chronic exposure to stressful conditions that lead to depression-like behavior and learning impairments, such as physical restraint or forced swimming, reduces adult hippocampal neurogenesis (Pham et al., 2003; Heine et al., 2004). Suppressed adult hippocampal neurogenesis has also been found in mice exposed to aberrant light schedules such as constant darkness (DD), constant light (LL), and experimental jet lag (Gibson et al., 2010; Fujioka et al., 2011; Monje et al., 2011). These correlations between suppressed neurogenesis and behavioral dysfunctions suggest that aberrant light may use adult hippocampal neurogenesis as a neural pathway to affect mood and learning. However, such results have been complicated by contradictory findings, such as a study by Mueller et al. (2011) that showed no changes in adult neurogenesis following exposure to constant light. Furthermore, Gibson et al. (2010) used hamsters to study jet lag, while other studies used mice to study DD (Monje et al., 2011) or LL (Fujioka et al., 2011; Mueller et al., 2011). Thus, the methods and models used by the respective studies differ in too many ways to conclude whether aberrant light environments affect adult neurogenesis. The presence of circadian arrhythmicity following LL can also complicate interpretations about the relationship between light and neurogenesis. To date, no published study has examined whether light can directly affect adult neurogenesis independent of circadian arrhythmicity.
In this study, we sought to examine the effects of the T7 light cycle on adult hippocampal neurogenesis. We first exposed adult wild-type mice to 2 weeks of the T7 cycle and then collected histological brain sections to examine hippocampal cell proliferation and number of immature neurons (for detailed experimental methods, see the Supplementary Material; Fig. 1A). On the 14th day of the T7 cycle, mice were injected with 5-ethynyl-2′-deoxyuridine (EdU) to label proliferating cells and were perfused on the 15th day (Fig. 1A). Control mice were kept in the standard T24 cycle (12 h of darkness, 12 h of light; Fig. 1A). In the T24 group (n = 9), we found 1781 ± 67 EdU+ cells in the dentate gyrus per animal (Figure 1B, F). In the T7 group (n=10), we found 1555 ± 92 EdU+ cells in the dentate gyrus per animal (Fig. 1C, F). Thus, there appears to be a trend toward a decreased number of EdU+ cells in the T7 group (13% decrease compared with T24); however, this effect did not reach statistical significance (Fig. 1F). We then examined endogenous markers of cell proliferation and number of young neurons, including Ki67 (a marker of proliferating cells) and DCX (a marker of immature neurons). In the T24 group (n = 5), we found 1982 ± 276 Ki67+ cells and 7174 DCX+ cells in the dentate gyrus per animal (Fig. 1B, D, G, H). In the T7 group (n = 4), we found 1967 ± 252 Ki67+ cells and 7691 ± 363 DCX+ cells in the dentate gyrus per animal (Fig. 1C, E, G, H). There were no statistically significant differences in the number of Ki67+ or DCX+ cells in the dentate gyrus between the T24 and T7 group (Fig. 1G, H). These results suggest that 2 weeks of exposure to the T7 light cycle did not affect cell proliferation and number of immature neurons.
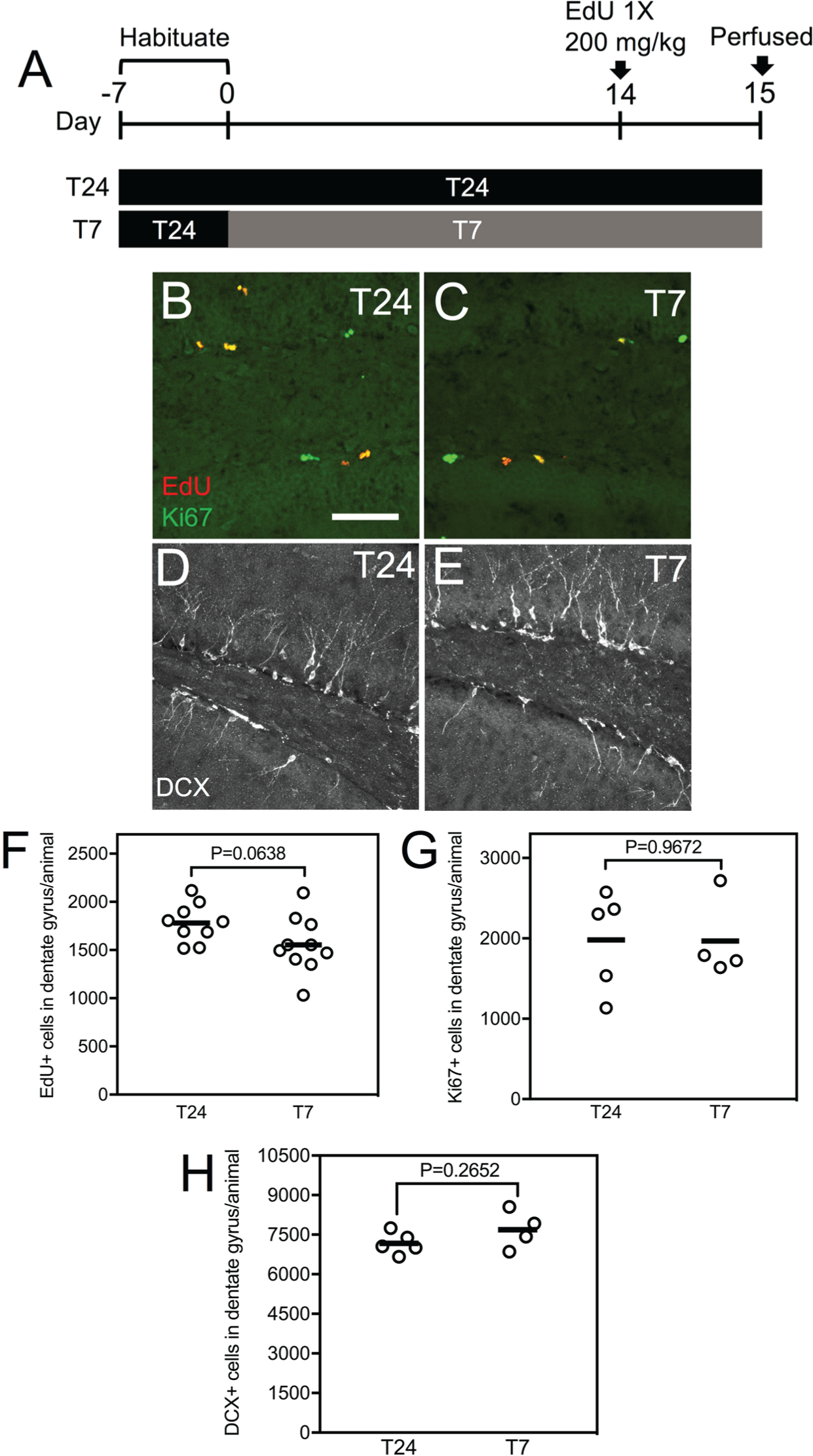
Two weeks of exposure to the T7 aberrant light cycle do not affect adult hippocampal neurogenesis. (A) Experimental design. (B, C) Confocal images of Ki67 staining in the dentate gyrus from mice exposed to either the T24 (B) or T7 cycle (C). (D, E) Confocal images of DCX staining in the dentate gyrus from mice exposed to either the T24 (D) or T7 cycle (E). (F) EdU+ cell counts in the dentate gyrus from mice exposed to either the T24 or T7 cycle. No statistically significant differences were found in the number of EdU+ cells between the T24 and T7 group (P > 0.05, ns, unpaired Student t test). (G) Ki67+ cell counts in the dentate gyrus from mice exposed to either the T24 or T7 cycle. No statistically significant differences were found in the number of Ki67+ cells between the T24 and T7 group (P > 0.05, ns, unpaired Student t test). (H) DCX+ cell counts in the dentate gyrus from mice exposed to either the T24 or T7 cycle. No statistically significant differences were found in the number of DCX+ cells between the T24 and T7 group (P > 0.05, ns, unpaired Student t test). Individual data points and mean (horizontal bar) are shown. Scale bar: 94 µm.
Since we did not find any effects of T7 on cell proliferation and number of immature neurons after 2 weeks of exposure, we lengthened the duration of exposure to 10 weeks. To label newborn cells, EdU was injected once daily for 7 days starting on the sixth week of the T7 cycle, and mice were perfused at the end of the 10th week (Fig. 2A). In the T24 group (n = 7), we found 1537 ± 60 EdU+ cells in the dentate gyrus per animal (Fig. 2F, J). In the T7 group (n = 8), we found 1411 ± 64 cells in the dentate gyrus per animal (Fig. 2G, J). There were no statistically significant differences in the number of EdU+ cells in the dentate gyrus between the T24 and T7 group (Fig. 2J). To examine specifically adult-born neurons, we co-stained EdU with NeuN (a marker of mature, differentiated neurons). In the T24 group (n = 5), we found 370 ± 57 EdU+/NeuN+ cells in the dentate gyrus per animal (Fig. 2F, K). In the T7 group (n = 5), we found 345 ± EdU+/NeuN+ cells in the dentate gyrus per animal (Fig. 2G, K). There were no statistically significant differences in the number of EdU+/NeuN+ cells in the dentate gyrus between the T24 and T7 group (Fig. 2K). Similar to the previous experiment involving 2 weeks of exposure to T7, we also examined Ki67 and DCX as endogenous markers of cell proliferation and number of immature neurons. In the T24 group (n = 7), we found 1690 ± 115 Ki67+ cells and 8369 ± 985 DCX+ cells in the dentate gyrus per animal (Fig. 2B, D, H, I). In the T7 group (n = 8), we found 1793 ± 122 Ki67+ cells and 9345 ± 445 DCX+ cells in the dentate gyrus per animal (Fig. 2C, E, H, I). There were no statistically significant differences in the number of Ki67+ or DCX+ cells in the dentate gyrus between the T24 and T7 group (Fig. 2H, I). We also performed double-labeling to examine co-localization between DCX/NeuN to quantify the number mature newborn neurons. However, the percentage of DCX cells that co-localized with NeuN was low (7.48% ± 1.3% for the T24 group, n = 5 animals; 8.04 ± 0.41 for the T7 group, n = 5 animals), and there were no statistically significant differences between the T24 and T7 groups. These results suggest that up to 10 weeks of exposure to the T7 light cycle did not affect adult hippocampal neurogenesis.
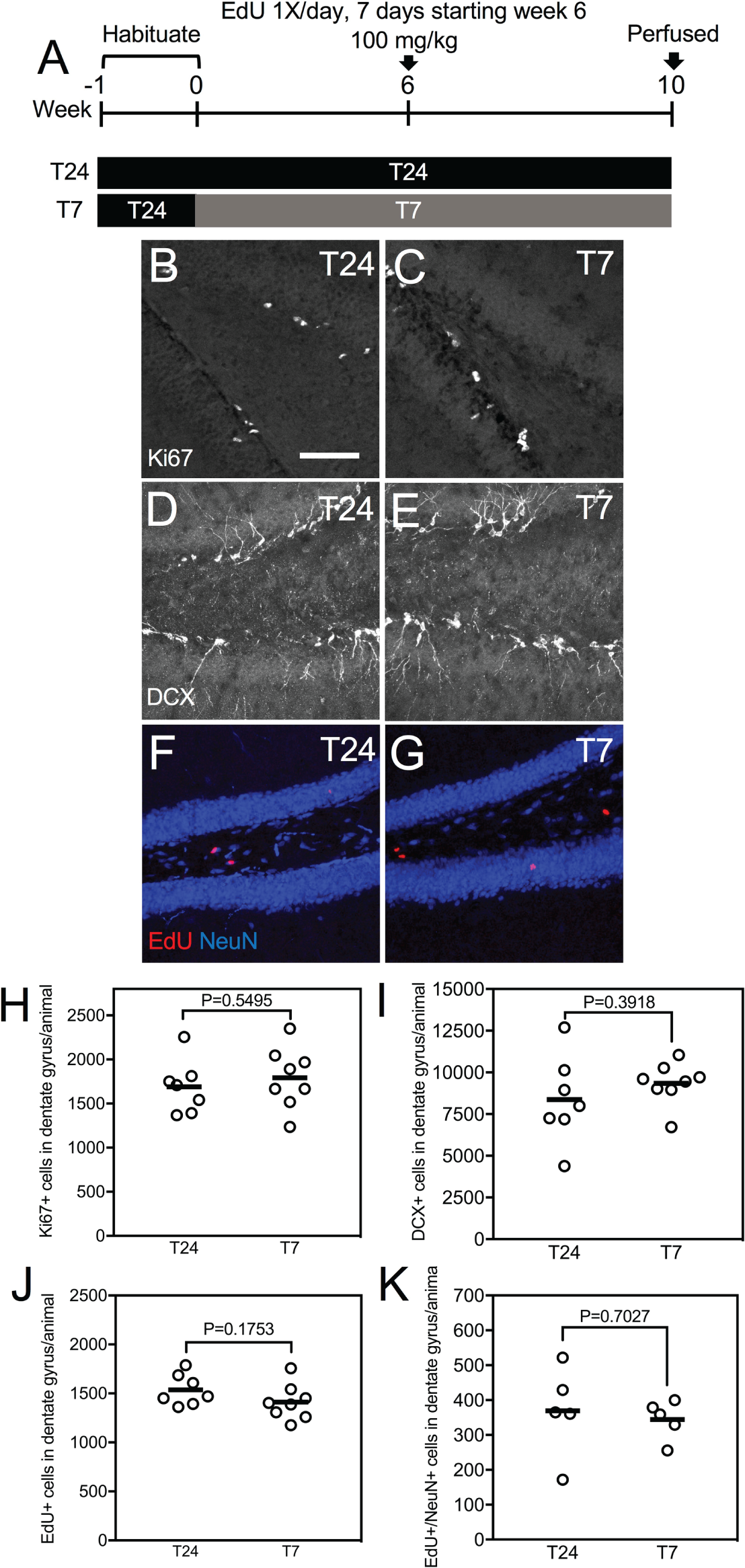
Ten weeks of exposure to the T7 aberrant light cycle do not affect adult hippocampal neurogenesis. (A) Experimental design. (B, C) Confocal images of Ki67 staining in the dentate gyrus from mice exposed to either the T24 (B) or T7 cycle (C). (D, E) Confocal images of DCX staining in the dentate gyrus from mice exposed to either the T24 (D) or T7 cycle (E). (F, G) Confocal images of EdU/NeuN staining in the dentate gyrus from mice exposed to either the T24 (F) or T7 cycle (G). (H) Ki67+ cell counts in the dentate gyrus from mice exposed to either the T24 or T7 cycle. No statistically significant differences were found in the number of EdU+ cells between the T24 and T7 group (P > 0.05, ns, unpaired Student t test). (I) DCX+ cell counts in the dentate gyrus from mice exposed to either the T24 or T7 cycle. No statistically significant differences were found in the number of Ki67+ cells between the T24 and T7 group (P > 0.05, ns, unpaired Student t test). (J) EdU+ cell counts in the dentate gyrus from mice exposed to either the T24 or T7 cycle. No statistically significant differences were found in the number of EdU+ cells between the T24 and T7 group (P > 0.05, ns, unpaired Student t test). (K) EdU/NeuN+ cell counts in the dentate gyrus from mice exposed to either the T24 or T7 cycle. No statistically significant differences were found in the number of EdU/NeuN+ cells between the T24 and T7 group (P > 0.05, ns, unpaired Student t test). Individual data points and mean (horizontal bar) are shown. Scale bar: 94 µm.
As a whole, our results show that chronic exposure to the T7 light cycle does not suppress adult hippocampal neurogenesis. Given that the T7 light cycle causes mood and learning dysfunction independent of sleep deprivation and circadian arrhythmicity (LeGates et al., 2012), the lack of an effect on neurogenesis following T7 exposure suggests that the direct effects of aberrant light are not dependent on adult neurogenesis to affect mood and learning. One caveat in our study is that we did not perform behavioral analysis to check whether the mice exposed T7 developed mood or learning deficits. In our previous study, we found that 2 weeks of exposure to T7 was sufficient to induce mood and learning deficits (LeGates et al., 2012), but we have not analyzed behavior at 10 weeks of exposure. In this study, we extended the exposure to 10 weeks since we did not find deficits in cell proliferation and number of immature neurons at 2 weeks. It is not known whether the animals still exhibit mood and learning deficits after 10 weeks of exposure to T7 and thus would be important to explore for future studies.
To conclude, our study highlights that the direct effects of aberrant light exposure on mood and learning may not depend on adult hippocampal neurogenesis and that neurogenesis-independent forms of hippocampal plasticity such as dendritic morphology should be explored as potential neural pathways by which aberrant light directly affects brain function.
Footnotes
Acknowledgements
We thank all members of the Hattar lab for their guidance and support. This work was supported by R21EY024452 (to S.H.), Woodrow Wilson Undergraduate Research Fellowship (to P.Q.D.), Michael R. Bloomberg Scholarship (to P.Q.D.), Barry M. Goldwater Scholarship (to P.Q.D.), and the NIH Undergraduate Scholarship (to P.Q.D.).
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
