Abstract
The possession of a rhythm is usually described as an important adaptation to regular changing environmental conditions such as the light-dark cycle. However, recent studies have suggested plasticity in the expression of a rhythm depending on life history and environmental factors. Barn owl (
Keywords
Rhythmicity is occurring at every level in nature, from the rotation of the earth causing day-night patterns to the behavioral activity patterns of most animals, body temperature fluctuations, and rhythmic expression patterns of hormones such as melatonin and cortisol (Daan and Aschoff, 1975; Cassone et al., 1993; Zeman and Gwinner, 1993; Ingram et al., 1999; Foster and Kreitzmann, 2004; Williams et al., 2012). An adaptation to predictable changes in the environment allows animals to conduct appropriate behavior at the ecologically best time (Aschoff, 1981a), which can confer survival (DeCoursey et al., 2000; Spoelstra et al., 2016) and reproduction advantages (Greives et al., 2015).
Variability of rhythmicity in behavior and physiology has recently become the focus of many studies. For example, reindeer (
The existence of ultradian rhythms, that is, rhythms shorter than 24 h, and underlying physiological mechanisms are not well understood (Stephenson, 2013). Whereas circadian rhythms with a period length of about 24 h follow the light-dark cycle, ultradian rhythms do not correspond to any known environmental periodicity (Daan and Aschoff, 1981). Ultradian rhythms can be observed for behavior such as feeding, foraging, or locomotion, but they also occur in releasing patterns of hormones and in heartbeat frequency (Kimura and Tsai, 1984; Gerkema et al., 1993; Tornatzky et al., 1998). They are thought to closely express the internal physiological state of an individual (Berger et al., 2002). In mammals, the adjustment of circadian rhythms strongly relies on maturation of the hypothalamic suprachiasmatic nucleus (SCN), which is responsible for entrainment to the light-dark cycle, and on the development of corresponding neural and humoral signaling pathways (Weinert, 2005). However, the role of the SCN for the development of ultradian rhythms is not clear. In rats, a lesion of the SCN resulted in a complete loss of circadian and ultradian rhythms for wheel-running activity, indicating ultradian rhythms as submultiples of the circadian period (Wollnik and Turek, 1989). In contrast, ultradian rhythmicity in wheel-running activity and feeding of the common vole (
Recent studies in the barn owl (
Methods
Study Species and Area
We studied a population of wild barn owls in Switzerland breeding in nest boxes fixed to the external wall of barns. Hence, nestlings can perceive the light-dark cycle through the nest box’s entrance, and they can hear natural and human-made noise. Data were collected between May and October 2011 and between April and October 2012. We took a small blood sample to identify the nestling sex using molecular markers (Py et al., 2006), and the hatching date was determined using the wing length measured in the first days after hatching (Roulin, 2004b). To disentangle genetic and environmental effects on sleep states (Scriba et al., 2014), we exchanged the same number of eggs or hatchlings between randomly chosen pairs of nests with similar hatching dates (in 2011, we left some eggs in the nest of origin, while the other eggs of the clutch were exchanged between pairs of nests, whereas in 2012, we swapped entire clutches between pairs of nests). In 2011, 41 (20 males and 21 females) of the 66 nestlings were raised by foster parents, while 25 nestlings (11 males and 14 females) were not cross-fostered and hence raised by their biological parents. In 2012, 36 (11 males and 25 females) of the 42 nestlings were cross-fostered, and 6 (2 males and 4 females) nestlings stayed in their original nest. Barn owls in this area produce between 1 and 9 nestlings, which fledge at an age of approximately 55 days. Because incubation starts as soon as the first egg has been laid, nestlings hatch asynchronously about 2.5 days apart, generating a pronounced age hierarchy. The mother stays in the nest box throughout 24 h until the offspring are approximately 4 weeks of age, when they are able to thermoregulate and eat without maternal help. At that time, the parents come to the nest only at night to bring small mammals (Roulin, 2004a). Nestlings are active both during the light and dark period (Scriba et al., in press), and they spread their meals throughout 24 h by eating food that has been stored at night by their parents mainly during the first 4 h of the night (Roulin, 2001; Roulin, 2004a; Michelat and Giraudoux, 1992). Nestlings show complex social interactions not only by communicating vocally to negotiate priority access to food resources (Johnstone and Roulin, 2003) but also by sharing food actively and preening each other (Roulin et al., 2012, 2016). At night, each nestling produces in the absence of the parents, on average, 1279 ± 122 calls (up to 10,245 calls per night), implying that there is the potential for substantial noise disturbance (Roulin, 2002).
Assessment of Plumage Spottiness
Barn owls display eumelanin-based black spots of varying size on the tip of their ventral feathers. The diameter of black spots in a reference area (60 × 40 mm) on the breast was measured with a caliper to the nearest 0.1 mm at the age of about 55 days, when feathers are fully developed. This method is repeatable (0.84-0.92) (Roulin, 2004b). The mean spot diameter was calculated and used in statistical analyses.
Brain Activity Recordings
Brain activity was recorded in 2011 in 31 male and 35 female nestlings from 29 nests aged between 27 and 48 days (mean = 38.2 ± 0.5 days), and in 2012, an electroencephalogram (EEG) was recorded in 13 male and 29 female nestlings from 30 nests aged between 36 and 49 days (mean = 42.8 ± 0.6 days). We used minimally invasive subcutaneous wire electrodes in conjunction with a small neurologger (Neurologger 2; www.vyssotski.ch/neurologger2) (Vyssotski et al., 2009; Scriba et al., 2013a, 2013b). Briefly, part of the feathers on top of the head were removed, the skin was locally anesthetized (Gingicaine/tetracaine 754 mg/65 g; Gingi-Pak, Camarillo, CA, USA), and 5 stainless steel wire electrodes (0.13 mm diameter, with 2 mm of the insulation removed for exposure; Cooner Wire, Chatsworth, CA, USA) were inserted with a 23-gauge needle under the superficial layers of the skin. One electrode was placed over the posterior part of the visual hyperpallium (visual Wulst) of each brain hemisphere and referenced to a posterior electrode placed over the caudal nidopallium of the same hemisphere. The ground electrode was centered between the other electrodes. We connected all 5 electrodes to a data logger glued to the head and covered them with plastic protection to prevent the birds from removing the logger and electrodes. The data logger additionally contained a 3-dimensional accelerometer, which measured behavioral state–related head movements in all 3 axes (X, Y, Z). The logger was powered by 2 batteries (each 1.4 V and 310 mAh; zinc-air 13; Renata, Itingen, Switzerland) connected in series. The total weight was about 5 g, which represents 2.1% of the nestlings’ body mass. The EEG of both brain hemispheres was recorded at 200 Hz for up to 5 days; during the entire period, the nestlings stayed undisturbed together with siblings in the nest box and were naturally provisioned by their parents. The last 24-h period of the EEG was analyzed for each year by a different person using Somnologica software (Medcare, Reykjavik, Iceland). The EEG was scored for wakefulness, non-REM sleep, and REM sleep in 4-sec epochs (Scriba et al., 2013a, 2013b). As in earlier studies with barn owls, interhemispheric asymmetries in non-REM sleep intensity were very rarely observed (Scriba et al., 2013b), and usually, both hemispheres showed some level of non-REM sleep–related EEG activity simultaneously. Epochs with more than one state were scored according to the predominant state.
Rhythmicity Analysis
The time spent in each state (i.e., wakefulness, non-REM sleep, and REM sleep) was calculated for each 5-min period (rather than shorter periods of time) because this was shown to remove noise and clarify any periodicity in short time series (Dowse and Ringo, 1994; Dowse et al., 2010). The time series, consisting of the percentage of time spent in each state for each 5-min period over 24 h (Fig. 1), were then analyzed for the intensity of rhythmicity and the period length (τ, tau) of the sleep-wakefulness pattern by performing autocorrelations and maximum entropy spectral analysis (MESA) (Burg, 1967) implemented in a Fortran program (prediction error filter coefficient was set to 90) (Dowse, 2007). Autocorrelograms can be used to visualize periodic signals out of time series, and MESA is a high-resolution technique that is well suited for the analysis of short noisy time series (Ables, 1974; Dowse, 2009; Dowse et al., 2010). Tau values indicate the period length or duration of the rhythmic motive, that is, the time between 2 peak values of a parameter (Fig. 1). The maximal spectral density of the periodograms, produced by fast Fourier transformations with the software R (R Core Team, 2012), relates to the variance explained by a single frequency and hence is a measure of rhythmicity in time series, with higher values showing stronger single rhythmicity in the time series (Fig. 1). We used 24-h periods to determine ultradian rhythmicity in each individual. We did not analyze circadian periodicity since we only extracted 24-h periods of each recording.
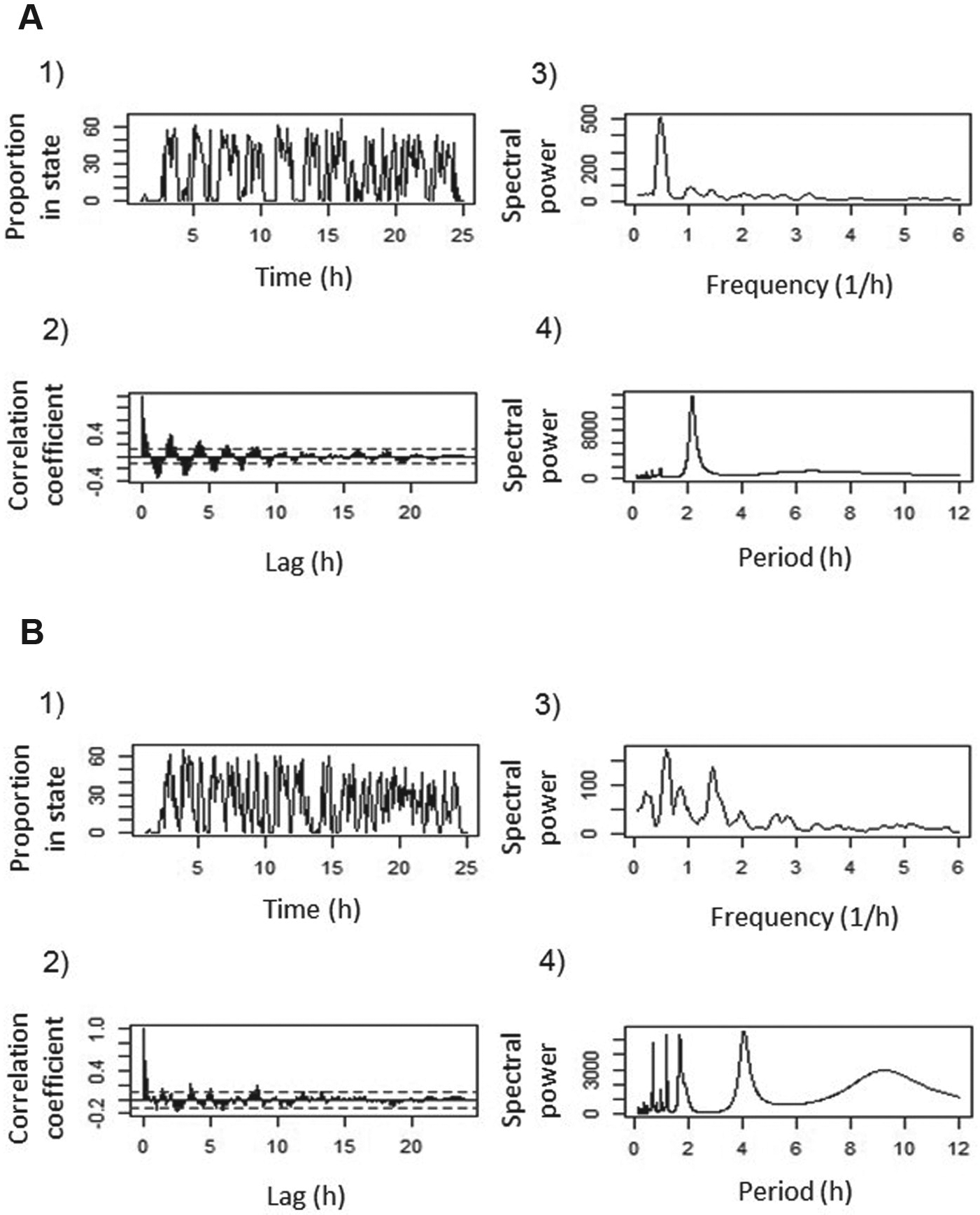
Non-REM sleep pattern (1) and corresponding autocorrelogram (2), periodogram (3), and MESA spectrogram (4) of a clearly rhythmic (A) and arrhythmic/multirhythmic (B) barn owl nestling. The period length tau can be determined from the autocorrelogram as the distance between 2 positive or negative peaks (for A, tau is 2.17 h, and for B, no single period length can be determined because several different oscillations appear to overlap). The spectral power of the highest peak in the periodogram represents the intensity of rhythmicity.
We considered a time series as significantly rhythmic when the maximal observed spectral density outranged the maximal spectral densities from 1000 data permutations of the same series. The calculation of data permutations was established as an effective method to detect periodicity in short time series (Dallmann et al., 2012; Krawczyk and Krapiec, 2010; Pardo-Iguzquiza and Rodriguez-Tovar, 2000; Ptitsyn et al., 2006; Mueller et al., 2012).
Statistical Procedure
Statistical analyses were conducted using the software JMP (version 10.0.0; SAS Institute, Cary, NC, USA). First, we compared rhythmicity values between years using the Student
Results
Rhythmicity in Relation to Cerebral States
To test whether sleep architecture (i.e., percentage of time spent in one cerebral state, bout duration, and number of bouts) (Table 1) was related to rhythmicity, we standardized values for each year so that we could pool the data obtained in 2011 and 2012. For all 3 states, the intensity of rhythmicity in each cerebral state was positively associated with the percentage of time spent in this state and the mean bout duration but negatively with the number of bouts per 24 h (Table 1). These relationships were particularly strong for REM sleep. In contrast, for all 3 cerebral states, the period length of rhythmicity was not significantly correlated with the 3 measures of sleep architecture (Table 2).
Sleep architecture in nestling barn owls.
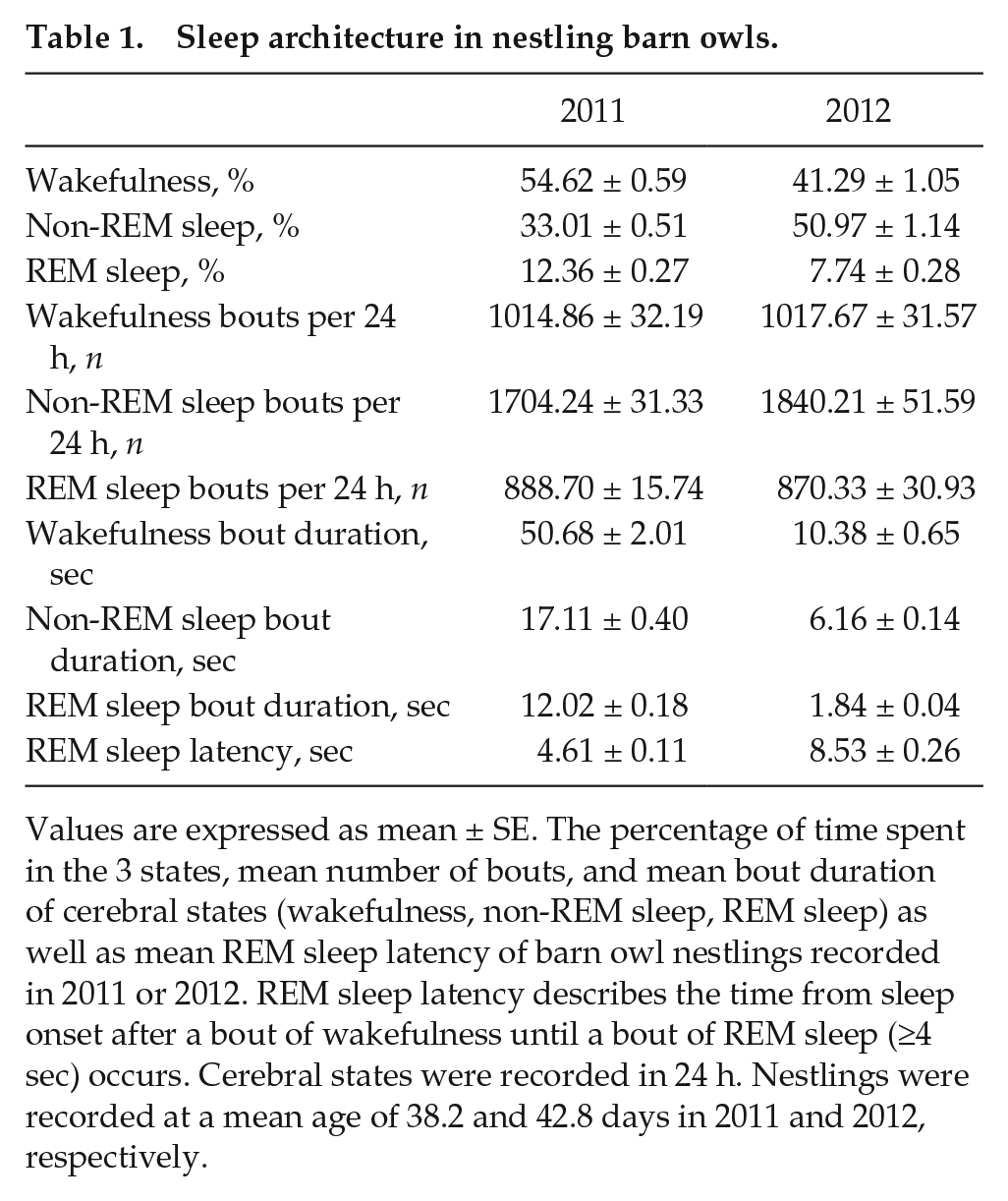
Values are expressed as mean ± SE. The percentage of time spent in the 3 states, mean number of bouts, and mean bout duration of cerebral states (wakefulness, non-REM sleep, REM sleep) as well as mean REM sleep latency of barn owl nestlings recorded in 2011 or 2012. REM sleep latency describes the time from sleep onset after a bout of wakefulness until a bout of REM sleep (≥4 sec) occurs. Cerebral states were recorded in 24 h. Nestlings were recorded at a mean age of 38.2 and 42.8 days in 2011 and 2012, respectively.
Pearson correlations between the intensity and period length of rhythmicity for the 3 cerebral states in nestling barn owls.
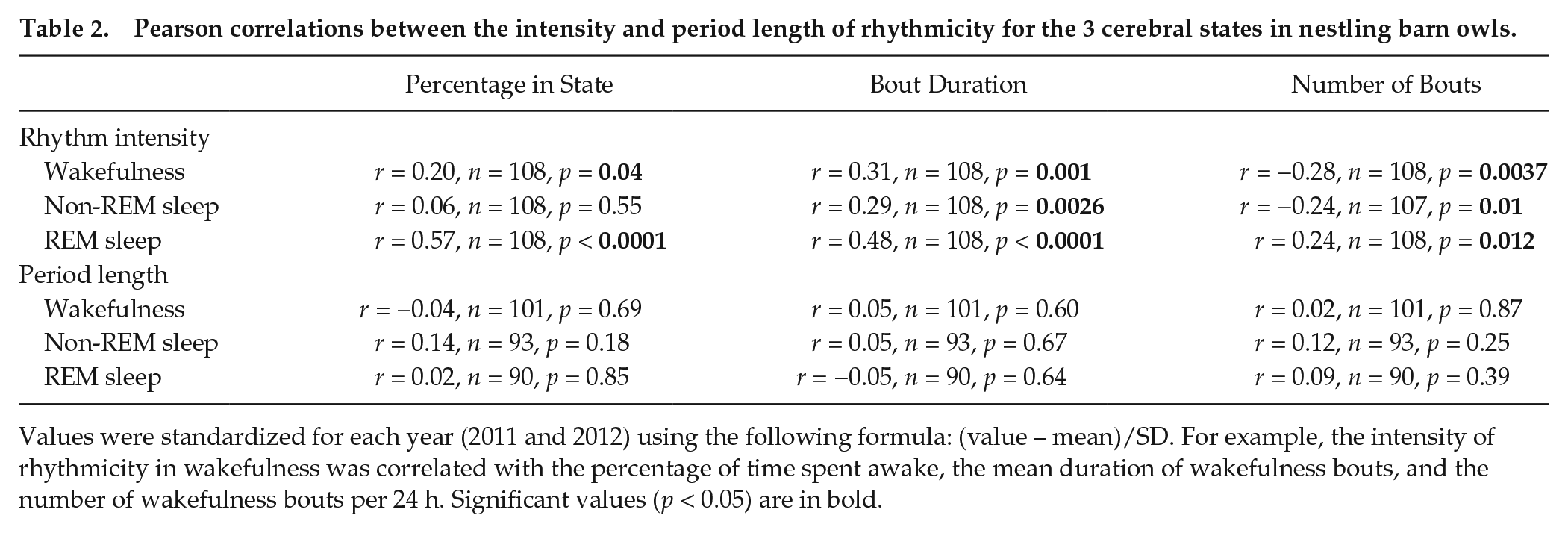
Values were standardized for each year (2011 and 2012) using the following formula: (value – mean)/SD. For example, the intensity of rhythmicity in wakefulness was correlated with the percentage of time spent awake, the mean duration of wakefulness bouts, and the number of wakefulness bouts per 24 h. Significant values (
Period Length of Rhythmic Brain Activity
Among the 66 nestlings recorded in 2011, 60 were rhythmic with respect to wakefulness, 59 to REM sleep, and 58 to non-REM sleep; overall, 44 individuals (67%) were rhythmic with respect to all 3 sleep-wakefulness components. From the 42 nestlings recorded in 2012, 41 were rhythmic in wakefulness, 39 in non-REM sleep, and 35 in REM sleep; overall, 32 nestlings (76%) were rhythmic in all 3 states. We combined the 2 years because period lengths measured in 2011 and 2012 were not significantly different (Student
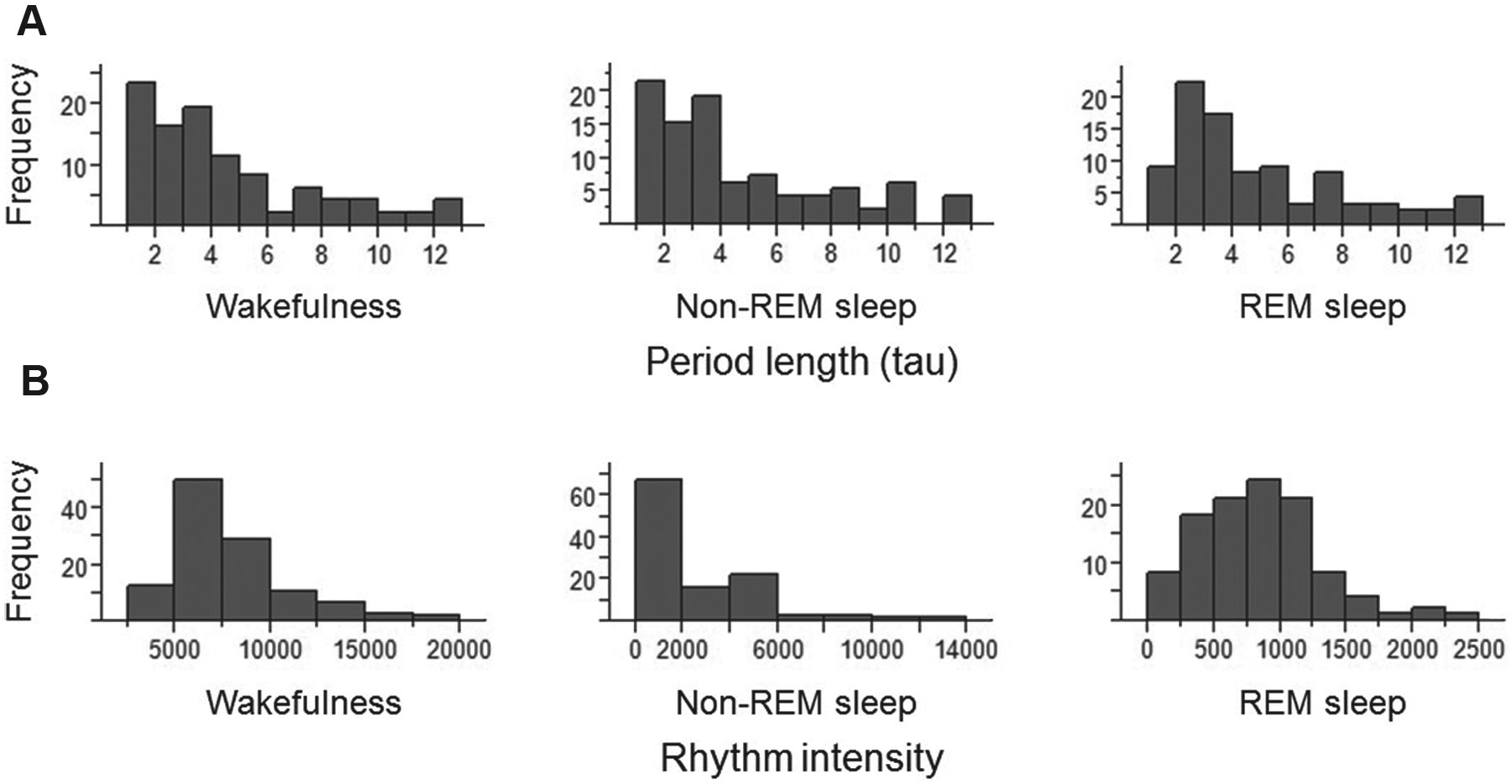
Histogram of the frequency (i.e., number of individuals) of the (A) period length and (B) intensity of rhythmicity in sleep-wakefulness states measured in barn owl nestlings.
Because the number of individuals that were rhythmic differed between cerebral states, we performed separate analyses to investigate whether the period lengths were associated with the size of black feather spots. In linear mixed models with the nest of origin as a random variable, the period length of wakefulness was significantly related to the interaction between nestling spot diameter and nestling sex (Table 3). Larger-spotted females showed a shorter period length of rhythmicity in wakefulness than smaller-spotted females (
Linear mixed models for the relationships between period length and intensity of rhythmicity in sleep-wakefulness states and age, sex and spot diameter in barn owl nestlings.

Significant values (
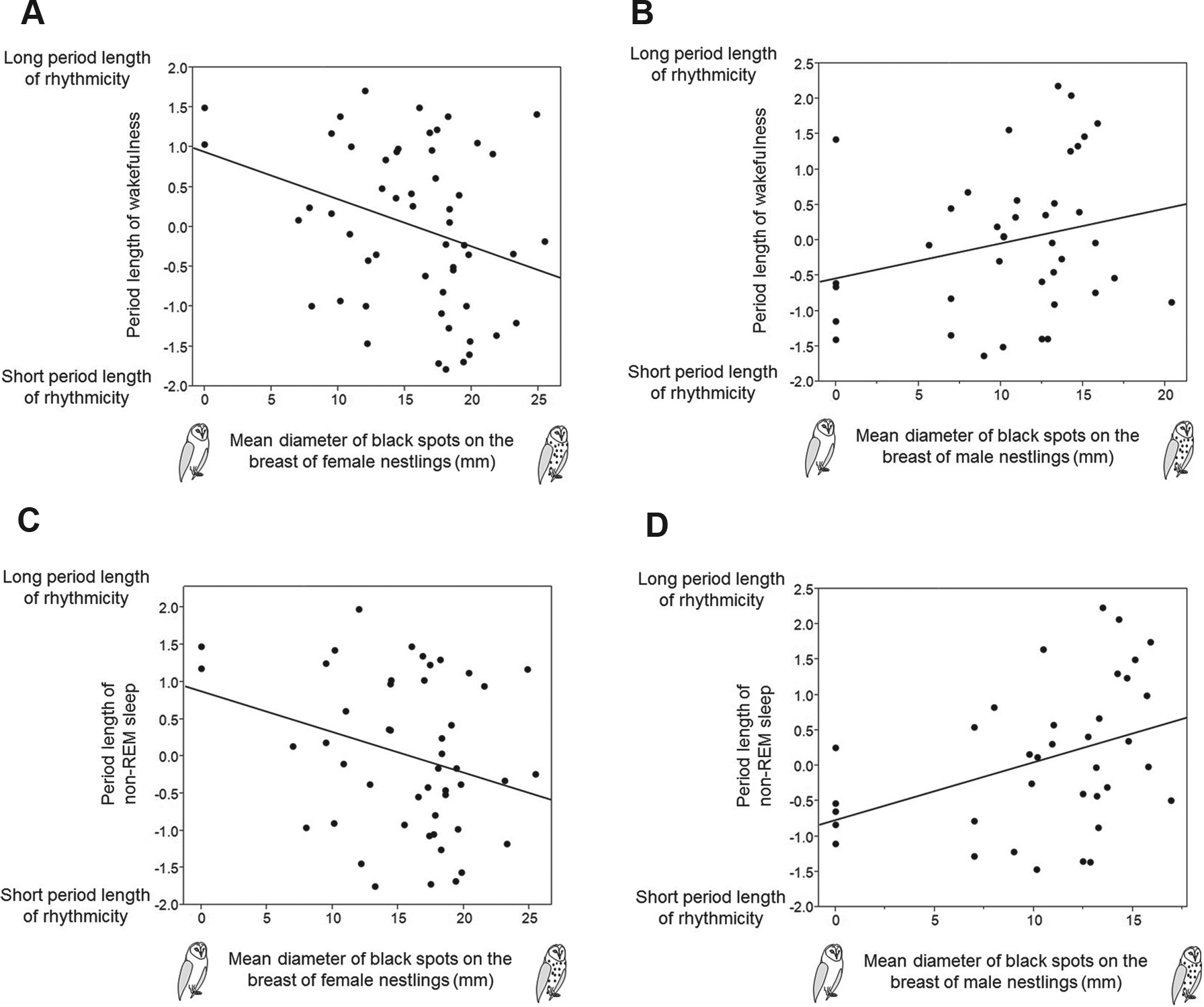
Relationship between the diameter of black feather spots and the period length of rhythmicity (log-transformed standardized tau values) of wakefulness (A, B) and non-REM sleep (C, D) in nestling male (B, D) and female (A, C) barn owls. Only rhythmic individuals were considered.
Intensity of Rhythmicity
The rhythm intensity in wakefulness did not differ between years (Student
The intensity of rhythmicity in wakefulness was positively correlated with the intensity of non-REM (
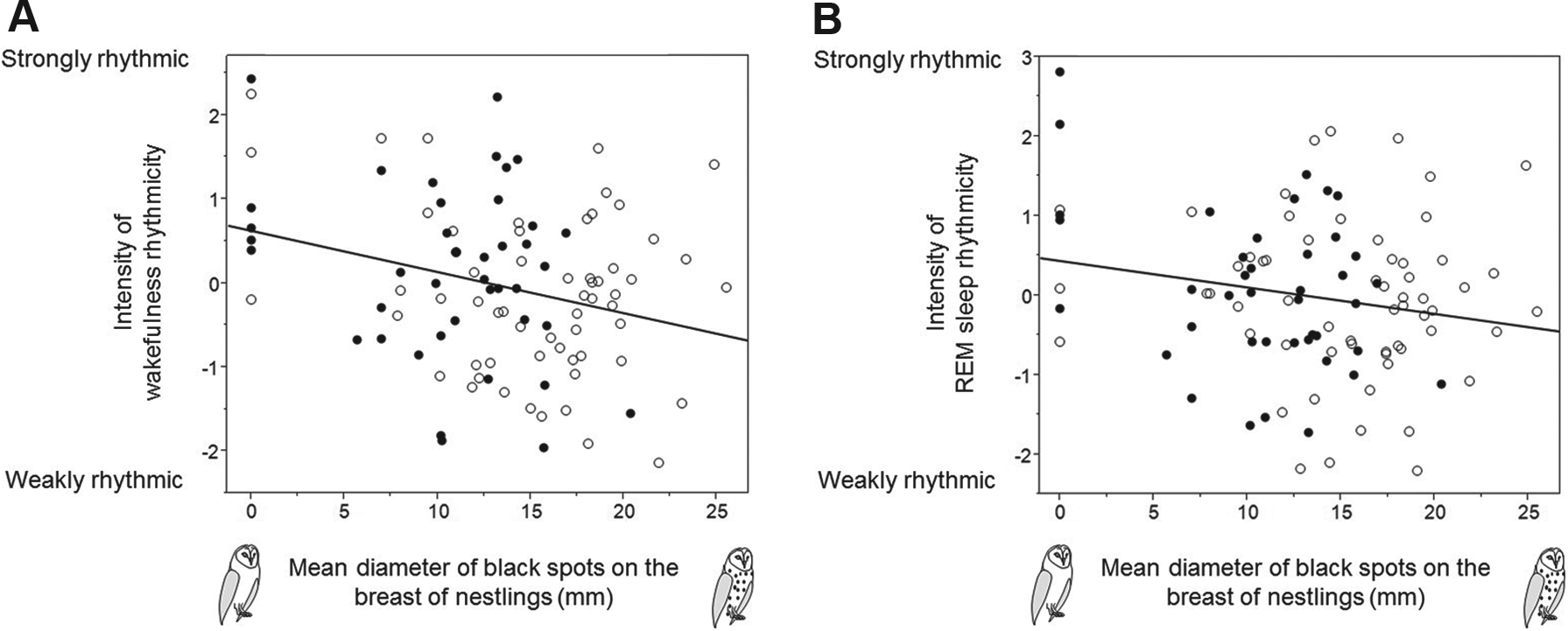
The intensity of rhythmicity in wakefulness and REM sleep in relation to spot diameter in male (closed circles) and female (open circles) barn owl nestlings. The intensity of rhythmicity in wakefulness (A) and REM sleep (B) declined with increasing spot size.
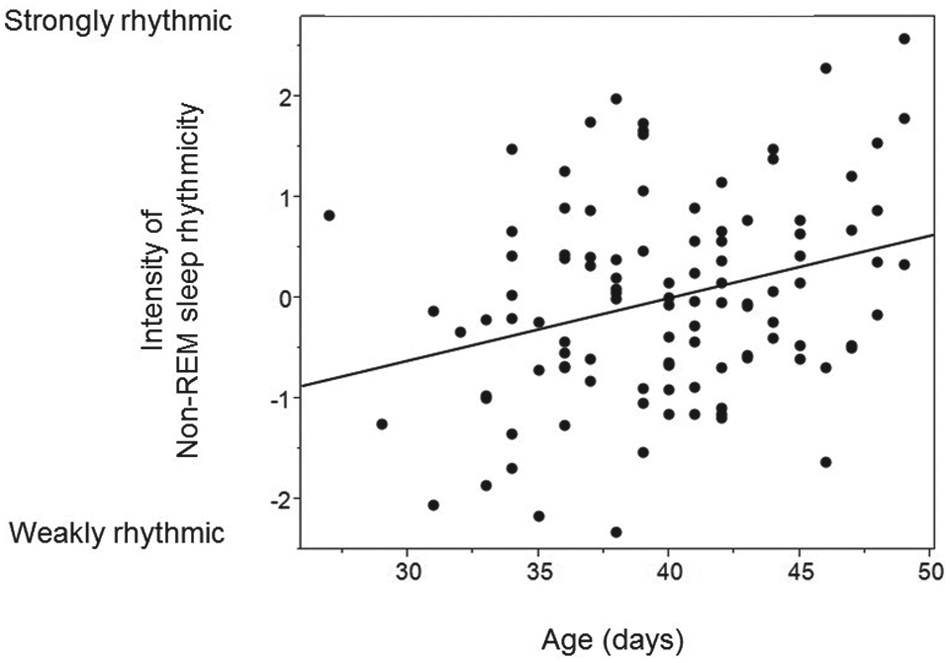
The intensity of rhythmicity in non-REM sleep increased with age in nestling barn owls.
In linear mixed models with the nest of origin and year as random variables, the size of black spots did not differ between rhythmic and arrhythmic individuals after controlling for sex (separate models for each cerebral state; wakefulness:
Discussion
Barn owl nestlings were mostly rhythmic, with mean period lengths of 4.5 h for wakefulness, 4.6 h for non-REM sleep, and 4.9 h for REM sleep. Larger-spotted females had a shorter period length of wakefulness and non-REM sleep than smaller-spotted females, whereas larger-spotted males displayed a longer period length of non-REM sleep than smaller-spotted males. Regardless of sex, smaller-spotted nestlings were more strongly rhythmic with respect to wakefulness and REM sleep. The intensity of rhythmicity in non-REM sleep increased significantly with age, whereas the period length was not associated with age.
Development of Rhythmicity
In mammals and birds, sleep architecture changes during development. The time spent in REM sleep is highest in young animals and progressively decreases to reach low levels in adults (Roffwarg et al., 1966; Jouvet-Mounier et al., 1970; Frank and Heller, 1997; Thurber et al., 2008). This decrease is pronounced, and in the barn owl, we found in an earlier study a strong decline in the amount of REM sleep (from ~17% to 7%) over a relatively short period of time between 27 and 48 days of age, which was concomitant with an increase in the duration of wakefulness and non-REM sleep bouts (Scriba et al., 2013a). Here, we show that the intensity of rhythmicity, but not the period length, in non-REM sleep increased with age in barn owl nestlings. In Japanese quails (
Social Aspects of Rhythmicity
We found individual differences in the intensity of ultradian sleep-wakefulness rhythmicity, with some nestlings displaying an intense rhythm characterized by a high amplitude and other nestlings showing a less intense rhythm with a low amplitude of rhythmicity. The period length and intensity of rhythmicity in sleep-wakefulness states were different between nest mates. This indicates that brain activity is an individual-specific trait, which might be to a less degree heritable and weakly sensitive to the social environment. Furthermore, in adult blue tits (
Rhythm and Melanism
Previous studies showed that offspring born from larger-spotted mothers switch more frequently between wakefulness and sleep and are more vigilant to watch out for the arrival of a parent bringing food to the nest (Scriba et al., 2014). These results suggest that larger-spotted nestlings are better able to anticipate any change in environmental conditions than smaller-spotted individuals, raising the hypothesis that they are less rhythmic. Our results reported in the present study are consistent with this idea. Thus, what could be the benefit for smaller-spotted individuals to be more rhythmic? Being less behaviorally flexible might be advantageous when environmental conditions are stable. Another hypothesis is that larger-spotted owls are more often looking at the nest box’s entrance, and since this is the source of light in the nest, it might weaken the robustness of the rhythm. Since smaller-spotted nestlings are less attentive towards the nest box’s entrance, this may explain why they display a more robust rhythm. More data are required to discuss further the potential behavioral consequences of variations in the degree of rhythmicity.
We did not find a difference in spot size between rhythmic and arrhythmic individuals. It might be that we do not have enough power to detect an effect because from the 108 nestlings, only 7 were arrhythmic in wakefulness, 15 in non-REM sleep, and 18 in REM sleep. Also, we cannot distinguish between arrhythmic individuals and those showing several rhythms. Arrhythmic birds do not have a defined period length, and therefore, we cannot compare the period length of rhythmic and arrhythmic birds. We only compared the period length among rhythmic birds.
Barn owl nestlings with more black spots on the feathers have a lower body temperature and higher oxygen consumption compared to owlets with fewer spots (Dreiss et al., 2016). These physiological differences resulted in color-specific behavioral strategies, with heavily spotted nestlings huddling with siblings more rapidly to reduce heat loss compared to nestlings with fewer spots. Furthermore, larger-spotted individuals consumed less food under ad libitum conditions, but when food deprived, they lost less weight (Dreiss et al., 2010). These studies suggest that eumelanin-based coloration is associated with a better ability to maintain energy balance between food intake and energy expenditure (Dreiss et al., 2016; Ducrest et al., 2008; Roulin, 2009; Roulin et al., 2010). Therefore, large- and small-spotted owls may differentially handle the trade-off between rhythmicity and energy homeostasis because the maintenance of a rhythm may be energetically intense. Accordingly, in eyeless cavefish, the absence of a circadian rhythm in metabolism allows them to save energy (Moran et al., 2014). This raises the possibility that owlets with large spots might save energy by exhibiting a weak rhythm for sleep-wakefulness.
In red deer, the ultradian period length is longer in winter, while in all other seasons, when environmental conditions are less harsh, the period length of activity is shorter (Berger et al., 2002). The annual dynamics in period length has been interpreted as a nutritional strategy depending on food quality and quantity as in winter, food is scarce and of high fiber content, taking longer to digest (Berger et al., 2002). In owlets, we did not find any association with the date, but the period length was related to the size of black feather spots in interaction with sex. Smaller-spotted female nestlings showed a longer period length for wakefulness and non-REM sleep than larger-spotted females, whereas in male nestlings, the opposite relationship was observed for non-REM sleep. This rather surprising result could be interpreted in light of the fact that selection exerted on spot size is sex dependent, with selection being positive in females and negative in males (Roulin et al., 2010). Female nestlings are larger than males (Almasi and Roulin, 2015), and hence, the physiological requirements of differently spotted individuals may differ between males and females, with a possible result that individuals show different period lengths. Although the daily variation in temperature and human-made noise occurs predictably, on a smaller scale, parental visits as well as social interactions with siblings occur probably with unpredictable frequency at night. Therefore, female nestlings with large eumelanic spots might have a shorter period length to benefit from a higher investment in vigilance, which might bring the advantage of a faster reaction to parents bringing food. Finally, the different pattern of rhythmicity in differently spotted individuals might reflect differences in the maturation of neuronal tissue involved in rhythmicity.
Footnotes
Acknowledgements
This research was supported by the Swiss National Science Foundation, the Société Académique Vaudoise, and the Max Planck Society. The authors thank Paul Béziers and Céline Simon for their help in the field and laboratory. They are grateful to William J. Schwartz and to 2 anonymous reviewers for helpful comments on a previous version of the article.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
