Abstract
Age-related disrupted sleep has been associated with modifications in the timing of endogenous circadian rhythms. There are studies suggesting a decreased sensitivity of the aging circadian pacemaker to light. In this study, we aimed to test whether a moderate illuminance light stimulus would produce significant phase shifts in older adults, and whether those would fall in a range consistent with the illuminance response curve (IRC) we previously reported in older adults. We conducted an 8-day in-patient study with 12 healthy older participants (mean age [SD], 58.3 [4.2] y, 5 females). A 6.5-h polychromatic white light exposure with a target illuminance of 270 lux was administered beginning in the early biological night. Before and after the light exposure, a circadian phase estimation procedure was carried out, with plasma melatonin data used as a circadian phase marker. The mean phase delay produced by the light stimulus was 1.78 ± 0.77 h. Ten of the 12 phase delay shifts at ~270 lux fell within the 95% predictive interval of the model fit to data from our previous IRC in older adults. This finding demonstrates that the circadian system of healthy older adults is sensitive to the phase-delaying effects of a moderate intensity light stimulus. The magnitude of the observed phase shifts is consistent with the previously described IRC in older adults.
The circadian system regulates many rhythmic functions in humans, including the timing of sleep, REM sleep propensity, and sleep efficiency (Dijk et al., 1999). With aging, there is a shift in sleep timing to earlier hours, sleep is more fragmented, and it becomes more difficult to maintain sleep during the biological daytime (Dijk et al., 1999). However, it is not understood whether changes in the circadian system contribute to these age-related changes in sleep.
In humans, as in many other organisms, the circadian system is entrained to the 24-h day by regular exposure to light and darkness. Light has a phase-dependent effect on the human circadian system (Khalsa et al., 2003; Rüger et al., 2013), with light exposure during the early subjective night causing phase-delay shifts. In addition to the timing of light exposure, the intensity, wavelength, and duration of light (Chang et al., 2012; Dewan et al., 2011; Lockley et al., 2003; Rimmer et al., 2000; Zeitzer et al., 2000), as well as previous light exposure history (Chang et al., 2011; Hébert et al., 2002; Smith et al., 2004), influence the human circadian system. It has been hypothesized that age-related changes in circadian sensitivity to light might contribute to some of the observed sleep changes in older adults.
There is some evidence to support this idea. In rodents, aging is associated with a reduced phase-shifting response to light, and higher levels of light are necessary for stable entrainment (Labyak et al., 1998; Weinert 2000). This reduced circadian sensitivity may be caused by an alteration in light transmission through the lens, which naturally “yellows” with age and therefore transmits less short wavelength light (Brainard et al., 1997; Charman, 2003). There are also reports of structural changes in the retinal ganglion cell layer and fewer circadian photoreceptors with age (Semo, Peirson, et al., 2003), as well as alterations in the function of the circadian system with aging, with a reduction in the synchrony of the SCN cell population (Meijer et al., 2012).
We previously studied the phase-delaying effects of a broad range of illuminances in healthy older individuals to determine whether the phase-shifting sensitivity of the human circadian timing system to a 6.5-h polychromatic white light stimulus was reduced with age (Duffy et al., 2007). After fitting the illuminance-phase-shift data with a 4-parameter model, we compared that model to one fit to data from a previous study in young adults (Zeitzer et al., 2000). We found that older and young participants did not show differences in sensitivity to very dim or very bright light, or in the steepness of the curve. However, we did find that the model’s sensitivity term was significantly greater in the older subjects (mean [SD], 263 [88.9] lux) than in the young subjects (119 [43.1] lux), suggesting that older subjects were less sensitive to moderate intensity illuminances.
Several previous studies in humans have reported results consistent with our findings. Three studies used polychromatic white light of similar or brighter levels than the saturating levels used for the illuminance response curve (IRC) of older adults, and each found no significant difference in the magnitude of the response between older and young participants (Benloucif et al., 2006; Kim et al., 2014; Klerman et al., 2001).
A study using a short (30-min) monochromatic light exposure (456 and 548 nm) in the evening found that the melatonin suppression response of older women was reduced compared with that of young women, but only for short wavelength light, and this effect was greater for the lower of the two irradiances tested (3.8 µW/cm2 and 9.8 µW/cm2) (Herljevic et al., 2005). Studies that have applied light in the phase-advance region are more mixed, with some suggesting reduced sensitivity among older participants under some conditions (Jud et al., 2009; Klerman et al., 2001; Sletten et al., 2009).
In the present study, we aimed to test whether a moderate illuminance light stimulus would produce significant phase shifts in older adults. We chose a target illuminance based on the sensitivity term from the model in our previous study to test the hypothesis that the range of phase-shifting responses in healthy older adults would lie within the 95% predictive interval (PI) of that model fit.
Volunteers aged 55 y and older were recruited from the community for this study. All were medically and psychologically healthy and were free from prescription medications and clinically significant sleep disorders. Ophthalmologic screening was performed to rule out the presence of cataracts, color-blindness, glaucoma, or a history of eye trauma. None reported recent night work or rotating shift work or crossing more than one time zone. Thirteen participants began the study, and one withdrew on the second study day. Findings from the remaining 12 participants (mean age [SD] 58.3 [4.2] years, 7 males, 5 females) are reported here.
The Partners Health Care Human Research Committee approved the protocol, and each participant gave written informed consent. The study was conducted according to the principles expressed in the Declaration of Helsinki.
Participants were instructed to maintain a regular, self-selected sleep-wake schedule of 8 h in bed each night for at least a week before admission to the laboratory, and to refrain from napping. This strict sleep-wake schedule was to ensure a stable entrainment to the light-dark cycle before study commencement. During this time, participants were also instructed to abstain from caffeine, alcohol, nicotine, and over-the-counter medications.
Throughout the 8-day study, each participant lived in a private study room in the Brigham and Women’s Hospital Center for Clinical Investigation. During the 3 baseline days, 8-h sleep episodes were scheduled at each participant’s habitual times. The baseline was followed by a constant routine (CR) circadian phase estimation procedure, which lasted for 27 h. Following an 8-h sleep episode, on the following intervention day, a 6.5-h experimental light exposure (LE) was administered in the middle of the inverted 16-h wake episode. The LE day was followed by an 8-h sleep episode, and a second CR lasting 30 h was carried out (See Figure 1 and Supplemental Online Methods).
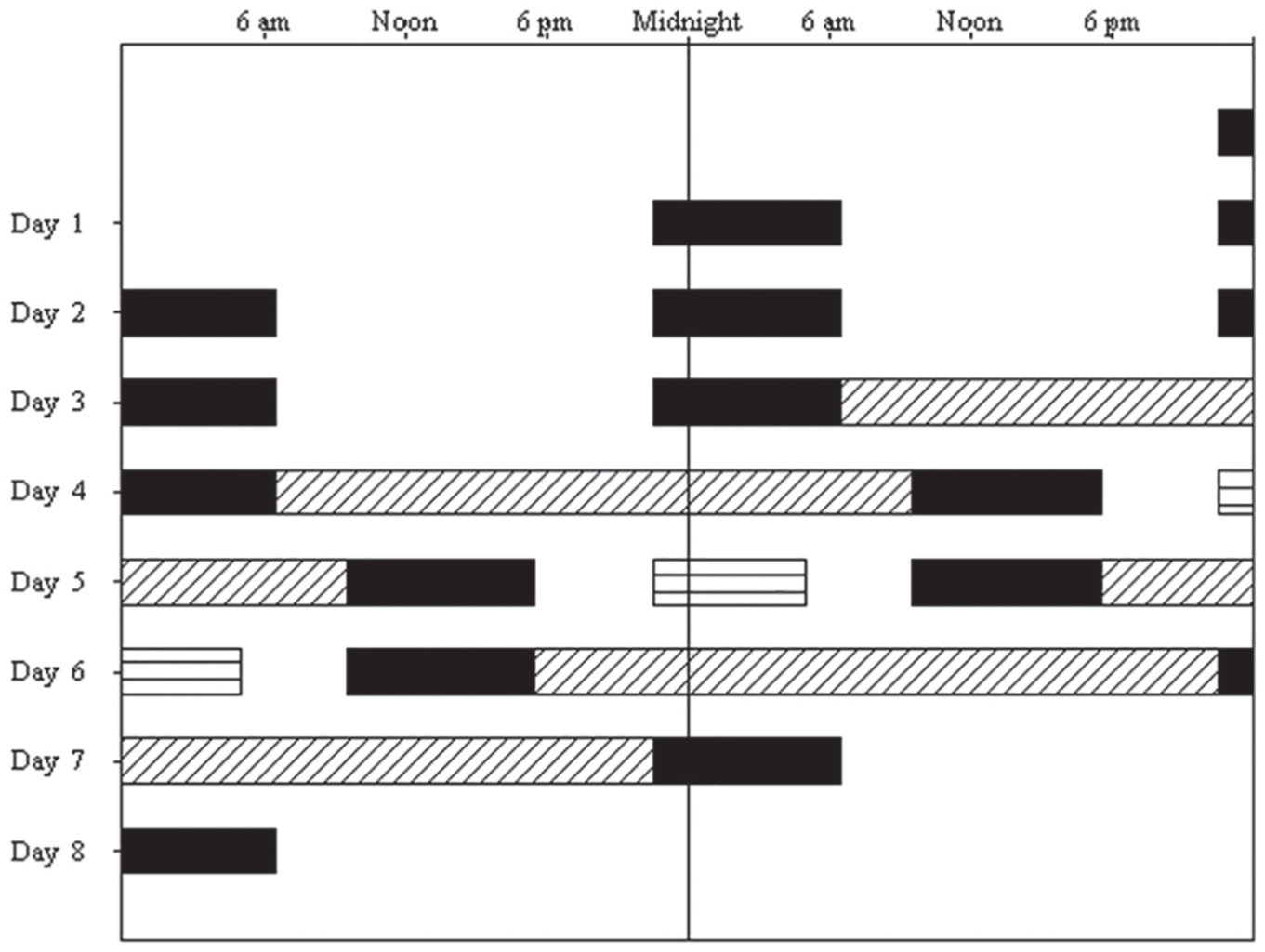
Double raster plot of the study protocol. Time of day is presented on the horizontal axis, with each day of the experiment shown both to the right of, and beneath, the previous day. Sleep episodes (black bars) were scheduled at the subject’s habitual times during baseline (Days 1-4), and on Days 5 and 6 to be centered 12 h opposite the 6.5-h experimental light exposure (LE). The 6.5 h LE is shown as horizontal lines on the night between Day 5 and 6. Before and after LE, constant routine (CR) phase estimate procedures are shown as hashed bars on Days 4 to 5 and Days 6 to 7.
The target illuminance level for the experimental LE was 270 lux, selected based on our previous study, and corresponding to the illuminance that elicits a half-maximal phase delay response in older adults (Duffy et al., 2007; see Supplemental Online Methods). The LE lasted for 6.5 h and was timed to be centered in the middle of the inverted waking episode; this means that it started in the early biological nighttime.
Two constant routine circadian phase estimation procedures (CR) were conducted, one before and one after the LE experimental day. Each CR began at wake time. The CR was designed to minimize or distribute evenly across the 24-h day periodic changes in posture, activity, sleep-wake state, environment, and feeding-fasting (Duffy and Dijk 2002) (see Supplemental Methods Online).
Blood samples were collected twice per hour during the CRs via an indwelling intravenous catheter inserted into a forearm vein and connected to a long tubing that allowed for sampling from outside the participant’s room. Samples were put into EDTA-containing microtubes and kept on ice for up to 1 h before processing, after which the plasma was frozen. Plasma samples were assayed for melatonin (Solidphase, Inc., Portland, ME) using the Bühlmann Melatonin Direct RIA kit (Bühlmann Laboratories, Schönenbuch, Switzerland).
The melatonin data from each CR were fit with a 3-harmonic regression model, and the maximum of the fitted waveform was used as the melatonin phase marker. Phase shift was defined as CR2 phase − CR1 phase. We also determined the timing of the LE relative to the melatonin peak (as inferred from CR1’s melatonin peak timing) for each individual. We then examined the relationship between the phase of the LE and the resulting phase shift magnitude using Spearman’s correlation.
We extracted the values of the 95%PI at an illuminance of 270 lux from the 4-parameter logistic models derived from data collected previously in older (Duffy et al., 2007) and young (Zeitzer et al., 2000) adults (Figure 2). Both data sets had been fit to the Michaelis-Menton-derived equation y = [(a–c)/(1+(x/b)d)]+c; where
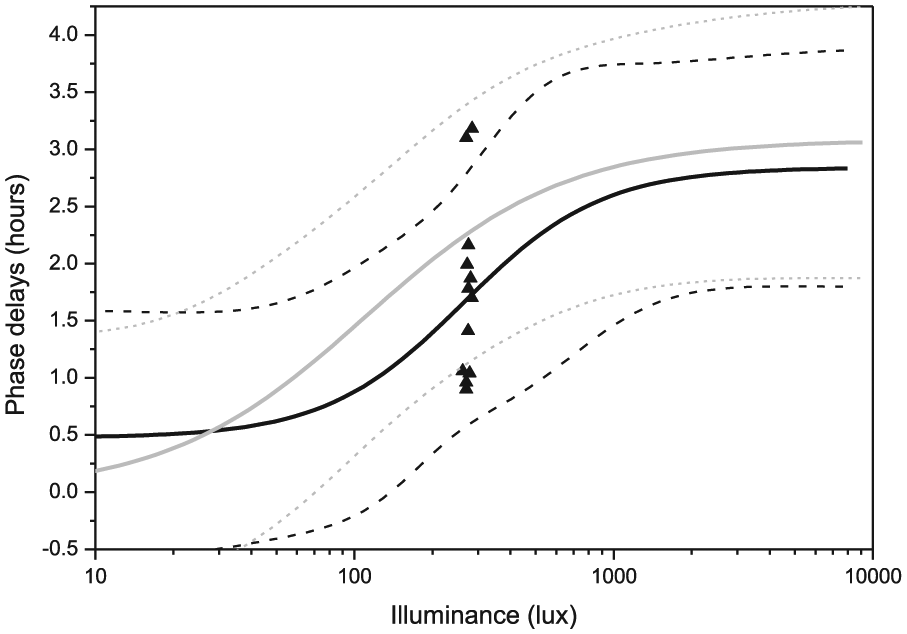
Phase shifts of the fitted plasma melatonin peak (MELmax) by the 6.5-h experimental light exposure of 270 lux. Data from each of the 12 subjects are plotted individually and shown with triangle symbols. The solid black line represents the 4-parameter logistic model fit in older subjects from our previous study (Duffy et al., 2007), with the 95% predictive interval (PI) of the model shown as black dashed lines. For comparison, the 4-parameter logistic model fit to the data from our previous study in young subjects (Zeitzer et al., 2000) is shown as a solid grey line, with its 95%PI shown as grey dashed lines.
We next counted the number of phase delay shifts for those whose magnitude fell: 1) within the 95%PI at ~270 lux of the model fit to the IRC data from the older adults only; 2) within the 95%PI at ~270 lux of the model fit to the IRC data from the young adults only; 3) within the 95%PI at ~270 lux of both IRCs; and 4) outside the 95%PI of both IRCs. We then ran a McNemar’s Chi-squared test with continuity correction to test the hypothesis that older adults in the current study were more likely to fall within the older adults’ IRC 95%PI than within the young adults’ IRC 95%PI at the tested illuminance of ~270 lux.
The average bed- and wake times of the subjects were 2234 (0057) h and 0636 (0051) h, respectively. The average timing of the fitted melatonin peak was 0137 (0127) h during CR1.
Actual LE levels were 272.68 (13.17) lux (range, 233.43 to 283.85 lux). On average, the LE began 2.7 (1.35) h (range, 0.45 to 4.6 h) before the melatonin peak, at a circadian time corresponding to the phase delay portion of the human phase-response curve.
The average timing of the melatonin peak during CR2 was 0324 (0141) h, with a mean phase shift of 1.78 (0.77) h (range, 0.9 to 3.18 h). The mean and SD fell within the 95%CI of the IRC reported from our previous study (Duffy et al., 2007). There was no significant correlation between the timing of the LE relative to CR1’s calculated melatonin peak and the magnitude of the phase delay (Spearman’s correlation,
The IRC model fit from the previous study predicted that phase-delay shifts to a 270-lux stimulus in older adults would lie 95% of the time between 0.58 h and 2.79 h (PI, not confidence intervals, which are narrower), whereas the IRC derived from young adults (Zeitzer et al., 2000) predicted that the phase-delay shifts to an identical stimulus would lie between 1.13 h and 3.39 h (95%PI). Of the 12 phase delay shifts obtained in the current study, 4 fell within the 95%PI of the older adults’ IRC model fit only, 2 fell within the 95%PI of the young adults’ model fit only, 6 fell in an overlapping region of both the young and the older adults’ model fits, and none were outside the 95%PI for either the young or the older adults’ curves (McNemar’s Chi-squared test with continuity correction
In summary, we found significant phase shifts in the melatonin rhythm in response to a 6.5-h exposure to ~270 lux of polychromatic white light in healthy older adults. The mean and SD of the phase shifts fell within the 95%CIs of the 4-parameter logistic model fit to data from our previous IRC (Duffy et al., 2007), and 10 of the 12 phase shifts in the group fell within the 95%PI derived from the same model. These current findings thus support the hypothesis that older adults can phase delay significantly at a moderate intensity of light.
In our previous study (Duffy et al., 2007), the model fit to data from older subjects was compared with a model fit to a similar series of studies in young adults, and the model terms indicated that older adults were less sensitive to low-to-moderate levels of light (~50 to 1000 lux), typical of levels that would be experienced indoors. Our current finding shows that, at moderate light intensity, the range of phase delay shifts is consistent with the predicted responses from our previously described IRC in older adults. However, as the 95%PIs are large, and the PIs derived from the IRCs of older and young adults overlapped, the current range of responses to ~270 lux is also consistent with previously described IRCs in young adults (Zeitzer et al., 2000).
There are numerous reports that aging is associated with an earlier timing of circadian phase and earlier awakening within the circadian cycle (Duffy et al., 1998; Duffy et al., 2002; Kripke et al., 2007), and that the sleep of older adults is more sensitive to circadian timing than the sleep of younger adults (Dijk et al., 1999). Evening bright light treatment can sometimes improve the sleep of older adults (Campbell et al., 1993); although, not always (Friedman et al., 2009). These inconsistent findings may be due to adherence issues, hypothesized to be induced by the glare discomfort from bright light (Campbell et al., 1993; Friedman et al., 2009). Our finding confirms that long-exposure evening light treatment using moderate intensity light, such as that used in Palmer et al. (2003), can effectively delay circadian rhythms in older adults, and may therefore be used in cases where compliance with brighter light is a problem.
Phase shifts in the current study are consistent with the previously described IRCs from older adults that suggested an age-related decreased circadian sensitivity to light. Several possible mechanisms for such an age-related reduced circadian sensitivity to light have been described. Changes in pupil diameter and lens opacity with aging (Kessel et al., 2011; Nylen et al., 2014; Zhang et al., 1998) could reduce the amount of corneal light reaching the retina, leading to a reduction in effective retinal illumination, rather than representing a true reduction in circadian light sensitivity. Another possibility is an age-related reduction in the detection of light by the circadian system. There is evidence from animals of a reduction in the number of melanopsin-expressing retinal ganglion cells with age (Semo, Lupi, et al., 2003), and this reduction is associated with a weaker circadian activity pattern (Lax et al., 2016). There is also evidence of reduced amplitude neuronal activity and mRNA rhythms in the SCN (Kawakami et al., 1997; Watanabe et al., 1995), as well as altered phasing of rhythms within the SCN (Sellix et al., 2012) with age, leading to the hypothesis that reduced light input to the central pacemaker with age can lead to reduced coupling of SCN cells (Ramkisoensing and Meijer, 2015). This, in turn, can lead to altered entrainment (Sellix et al., 2012) and/or alterations in the amplitude of output rhythms (Weinert 2000). It has also been shown that the range of entrainment of the circadian pacemaker decreases as the cellular coupling strength increases (Gu et al., 2017).
Our finding that the range of phase delays to the same ~270 lux stimulus could vary so drastically (from 0.9 to 3.18 h) may be further indirect proof of decreased coupling strength of SCN neurons with age, as shown by Farajnia et al. (2012) in rodents. We took efforts to ensure that our participants’ sleep-wake cycles would be optimally entrained to their light-dark cycle when they started the study by having them adhere to a consistent 16:8 h light-dark, wake-sleep schedule for at least 2 weeks before the study. In the post-hoc analysis, we confirmed that the LE (timed relative to the participants’ pre-study sleep wake times), were appropriately centered on the phase delay portion of the phase-response curve. We also found no relationship between the circadian phase of the LE and the magnitude of the resulting phase delays. This could be due to the (intentionally) narrow range of phases and small sample size of our study, or it may reflect a more variable coupling strength of the pacemaker with age. In a previous study, where we compared light exposure patterns of healthy older and young community-dwelling adults, the older adults experienced significantly greater light exposure in the evening (Scheuermaier et al., 2010). Taken together with our current finding, it is unclear what the consequences of age-related decreased sensitivity to evening light might be in cases where older adults self-select exposure to brighter artificial light that may compensate for any functional deficit of the circadian system.
There is ample evidence that chronic circadian disruption, such as that which occurs in shift work, can predispose the individual to disorders of metabolic, immune, and cardiovascular systems (Dochi et al., 2009; Gu et al., 2015; Wegrzyn et al., 2017). There is also evidence that even moderate circadian misalignment can disrupt sleep, and chronic insufficient sleep is also linked with many similar disorders (Aho et al., 2016; Rao et al., 2015). Thus, a better understanding of whether there is reduced circadian sensitivity to light with aging, and the functional consequences of such a change, may have important health consequences for older adults.
The main limitations of this study were the small sample size and the absence of a control group of young adults undergoing the same protocol to directly test whether the phase-delay shifting response of these older adults to a moderate intensity light stimulus was indeed reduced. We carried out sample size calculations to determine how many participants would be necessary to detect a difference between age groups and found a sample size of 24 in each group would be required. While we are unable to carry out additional studies at this time, given the potential importance of this finding, we believe further investigation of this issue, perhaps using other less-expensive protocols, should be pursued.
Supplemental Material
JBR_18-0037_Supplemental_Materials_2018-10-31 – Supplemental material for Phase Shifts to a Moderate Intensity Light Exposure in Older Adults: A Preliminary Report
Supplemental material, JBR_18-0037_Supplemental_Materials_2018-10-31 for Phase Shifts to a Moderate Intensity Light Exposure in Older Adults: A Preliminary Report by Karine D. Scheuermaier, Jung Hie Lee and Jeanne F. Duffy in Journal of Biological Rhythms
Footnotes
Acknowledgements
The authors wish to thank the study participants; the technical, nursing, and dietary staff of the Brigham and Women’s Center for Clinical Investigation; and the technical staff of the Division of Sleep and Circadian Disorders Sleep Core and Chronobiology Core for their assistance with data collection and participant monitoring.
This study was supported by the US National Institutes of Health [R01 AG06072] and was carried out in the Harvard Clinical and Translational Science Center [UL1 RR025758]. JHL was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (MSIT) [2017R1A2B4003493]. KS was supported by fellowships from the US National Institutes of Health [T32 HL07901, F32 AG031690] during the data collection and analysis phase, and by the South African Medical Research Council while writing the paper; JFD was supported by grants from the US National Institutes of Health [P01 AG09975, R01 AG044416] while writing the paper.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
