Abstract
The measurement of circulating levels of brain-derived neurotrophic factor (BDNF) has been proposed to be a marker of disease and an indicator of recovery. Thus, knowing the temporal pattern and influence of potential circadian rhythms is important. Although several studies have measured BDNF at different times of day, no studies have done so while controlling for potential masking influences such as sleep and activity. Further, no previous study has examined circadian rhythms within individuals. We examined circadian rhythms in plasma BDNF while minimizing masking from behavioral and environmental factors using a 30-h constant routine (CR) protocol. In a sample of 39 healthy adults, we found significant circadian rhythms in 75% of women and 52% of men. The timing of the acrophase of the BDNF rhythm, however, was unrelated to clock time in women, while it was related to clock time in men. These results indicate that the use of single-sample measures of plasma BDNF as a marker of disease will be unreliable, especially in women. Repeated plasma BDNF samples over a 24-h period within individuals would be needed to reveal abnormalities related to disease states.
Brain-derived neurotrophic factor (BDNF) is a member of the neurotrophin family, a group of polypeptide growth factors that influence proliferation, differentiation, and survival of neuronal cells (Ghosh et al., 1994; Henderson, 1996; McAllister et al., 1999; Sohrabji and Lewis, 2006). BDNF is highly expressed in the central nervous system, regulating synaptic plasticity and connectivity in the adult brain, primarily by acting upon tropomyosin receptor kinase B (TrkB) (Bramham and Messaoudi, 2005). BDNF from the brain has been found to enter general circulation (Krabbe et al., 2007; Rasmussen et al., 2009), and altered circulating levels have been reported in several diseases such as Alzheimer’s (Laske et al., 2006), Parkinson’s (Scalzo et al., 2010), schizophrenia (Fernandes et al., 2015), type 2 diabetes (Krabbe et al., 2007), and heart disease (Takashio et al., 2015). Circulating BDNF levels have been proposed as a biomarker for major depression and successful treatment, with lower levels in depression and normalization after treatment (Polyakova et al., 2015). Given the potential for circulating BDNF levels to inform the progression of disease states, it is important to understand how these levels are controlled and change over the day, as circadian variability in plasma BDNF levels may lead to the misinterpretation of an individual’s basal BDNF levels as normal or abnormal due to the time of sampling.
BDNF has been found to be centrally rhythmic and to be a potent modulator of the circadian system. BDNF is highly expressed in the suprachiasmatic nucleus (SCN) (Liang et al., 1998b), the seat of the master circadian clock in mammals (Moore and Eichler, 1972; Ralph et al., 1990). Expression of BDNF within the SCN is rhythmic, with highest mRNA expression during the biological night and highest protein expression during the biological day (Liang et al., 1998b). TrkB, the primary receptor for BDNF, is also expressed throughout the SCN, with especially high expression in the region receiving retinal input via the retinohypothalamic tract (Liang et al., 1998a; Allen and Earnest, 2005). Manipulations of BDNF and TrkB have been found to affect the response of the circadian system to light, with decreased BDNF signaling associated with decreased phase shifting to light (Liang et al., 2000; Michel et al., 2006). Recently, structural plasticity within the SCN has been found to be dependent on BDNF/TrkB signaling (Girardet et al., 2013). Circadian rhythms in BDNF and/or TrkB receptors have been demonstrated in several brain regions, including the hippocampus, frontal cortex, visual cortex, superior colliculus, and cerebellum (Bova et al., 1998; Berchtold et al., 1999; Schaaf et al., 2000; Pollock et al., 2001; Dolci et al., 2003).
Several studies have attempted to examine daily rhythms in circulating plasma levels of BDNF (Begliuomini et al., 2008; Piccinni et al., 2008; Pluchino et al., 2009; Choi et al., 2011). Levels have been reported to be high in the morning for men (Begliuomini et al., 2008; Piccinni et al., 2008; Choi et al., 2011) but not women (Piccinni et al., 2008; Choi et al., 2011), although levels have been shown to drop over the waking day in women, depending on menstrual phase (Pluchino et al., 2009). These studies, however, have some important limitations. All studies to date have examined circulating BDNF levels across the waking day (3-5 samples over 16 h), excluding samples during the typical sleep period, in ambulatory participants. Thus, these studies have not controlled for potential masking influences (e.g., activity, sleep, meals, light, etc.) on circulating BDNF levels. Further, no study has directly examined circadian rhythms in plasma BDNF within participants. In previous studies, conclusions were drawn about plasma BDNF rhythms in women and men from the grouped data only (overall patterns in women and men from data grouped at each time point). This approach, however, cannot reveal whether individuals display rhythms, and it is not appropriate if the measure is likely to be highly variable in its timing, as greater interindividual variability in a group would appear as a flattened amplitude or a lack of rhythms for the group. To accurately assess potential circadian rhythms of plasma BDNF while controlling for masking effects induced by day-night changes in posture, behavior, and environmental influences, the constant routine (CR) protocol is used (Minors and Waterhouse, 1984). The CR consists of a regimen of semirecumbent wakefulness in constant dim lighting conditions for at least 1 circadian cycle (>24 h), with food and fluid spread evenly across day and night. The CR eliminates periodic changes in behavior and holds environmental conditions constant, allowing the endogenous circadian variation in physiologic measures to be precisely assessed within individuals (Klerman et al., 1999). In order to evaluate sex differences in plasma BDNF rhythms, we examined women and men on a 30-h CR, examining each individual for significant circadian rhythms in BDNF using cosinor analysis.
Further, we examined whether a common polymorphism of the BDNF gene influences plasma BDNF levels and rhythms. In humans, a naturally occurring, common single nucleotide polymorphism of the human BDNF gene resulting in a valine to methionine substitution at codon 66 (Val66Met) has been identified and has been associated with a decrease in activity-dependent secretion of BDNF (Egan et al., 2003).
Materials and Methods
Participants
We examined plasma BDNF levels in 39 healthy adults (18-36 years old; 16 women) studied at the Center for Clinical Investigation of Brigham and Women’s Hospital. All participants were medically and psychologically healthy and were required to have a body mass index (BMI) between 18 and 30. They were asked to refrain from using alcohol, nicotine, caffeine, prescription and nonprescription drugs, and dietary supplements both prior to and throughout the course of the study, and compliance was verified using urinary toxicological analysis during screening and upon admission to the laboratory. To ensure that each participant was stably entrained to his or her schedule, no participant who reported recent (within 1 year) regular night shift work or recent (with 3 months) travel across more than 1 time zone was studied. Participants maintained a regular sleep-wake schedule for at least 1 week immediately prior to the study, such that their bedtimes and wake times occurred at the same time (±30 minutes) each day and were 8 h apart. The timing of the 8-h sleep opportunity was self-selected based on the participants’ habitual sleep-wake times. Compliance with the sleep-wake schedule during the week immediately prior to the study was verified by time-stamped call-ins for wake times and bedtimes and wrist actigraphy.
The studies were approved by the Human Research Committee of the Partners HealthCare System and were conducted in accordance with the Declaration of Helsinki. Each participant gave written informed consent.
Experimental Protocol
The data described in this manuscript were part of a larger 8-day inpatient study protocol. Participants were studied for 8 days in an environment free of time cues (no windows, clocks, radio, live TV, newspapers) and were continually monitored by staff. The protocol consisted of (a) 1 baseline day-night (16:8 wake-sleep); followed by (b) 5 days of timed light exposure designed to phase advance the sleep episode by 8 h while maintaining 8-h sleep episodes; followed by (c) a 30-h constant routine and final 8-h recovery sleep episode. There were 3 distinct light protocol designs: (1) a gradual shift in which the sleep episode was advanced by 1.6 h each day for 5 days (using either white or green light) (6 men, 4 women); (2) a “slam” shift in which the sleep episode was abruptly advanced by 8 h (using either white or green light) (11 men, 8 women); and (3) a modified slam shift in which the extended wake period prior to the 8-h advance of the sleep period included 2 short nap opportunities (green and white light combined) (6 men, 4 women). Participants underwent a 30-h extended wake episode under constant routine conditions after the advanced schedule. During the CR, participants remained in a semirecumbent posture (lying in bed with the head of the bed elevated ~45°) in dim light (approximately 0.0048 W/m2 [~1.8 lux]) and were fed equicaloric snacks each hour. Participants maintained this posture throughout the CR, and a staff member remained in the room to help maintain wakefulness. Blood samples were collected every hour via an indwelling catheter inserted into a forearm vein.
BDNF and Melatonin Assays
Plasma was separated by centrifuging blood samples at 2200 to 2800 rpm for 10 min at 4 °C and was stored at −80 °C. BDNF was measured in duplicate by ELISA (Quantikine; R&D Systems, Minneapolis, MN) (interassay coefficient of variation [COV], 7.6%-11.3%; intra-assay COV, 3.8%-6.2%). All samples for the same participant were assayed in duplicate on the same plate to minimize interassay variability. Samples were preprocessed by centrifugation at 10,000 rpm for 10 min at 4 °C, and the protein assay was performed using clean supernatant (this additional centrifugation step minimized COV with no substantial change in BDNF concentration). Plasma melatonin samples were assayed using the Bühlmann melatonin radioimmunoassay (Alpco Diagnostics, Salem, NH), which had a functional sensitivity of 0.9 pg/mL and an analytical sensitivity of 0.3 pg/mL, an intra-assay precision of 7.9% to 8.2%, and an interassay precision of 11.7% (SolidPhase Inc., Portland ME).
Genotyping
Participants were genotyped for the rs6265 (coding DNA variant Val66Met) variant from the BDNF gene. DNA was extracted from whole blood and genotyped using TaqMan SNP genotyping assays (Applied Biosystems, Assay ID: C1159275810). PCR amplification and allele specific discrimination was performed using LightCycler 480 (Roche Applied Science, Pleasanton, CA). The reaction consisted of 10 ng of DNA, 6.25 µL of 2X master mix (Applied Biosystems, Catalog No. 4371357), and 0.65 µL of 20X TaqMan SNP genotyping assay in a PCR reaction volume of 12.5 µL. Annealing was performed at 60 °C. The LightCycler 480 software was used to detect specific SNP alleles using the end-point detection method. Genotyping was performed in duplicate to ensure reproducibility, and a call rate greater than 95% was obtained. For 12 samples, DNA was extracted from frozen saliva and genotyped using the same TaqMan SNP genotyping assays. Genotypes for 3 samples with poor DNA quality were obtained by PCR amplification followed by Sanger sequencing using an ABI 3170 DNA sequencer (rs6265: 385bp amplicon using forward primer 5′ GAGTGATGACCATCCT-TTTCCT3′ and reverse primer 5′ CGGACCCTCATGGACATGTT3′).
Data Analysis
Cosinor analysis
The single cosinor method (Nelson et al., 1979) was used to determine the presence of circadian rhythmicity in BDNF expression, within individuals. For each individual’s data, a cosinusoid of a predefined period (24 h) was fit by the method of least squares. This analysis derives estimations of several rhythm characteristics including the mesor (the rhythm-adjusted average cycle value), the amplitude (half the distance between the peak and trough of the fitted waveform), and the acrophase (timing of peak). A zero-amplitude test was used to determine the presence of circadian rhythmicity (α < 0.05).
Melatonin phase
We defined melatonin onset as the time at which plasma melatonin levels rose to 25% of the fitted nightly peak (termed here DLMO). The nightly melatonin peak was determined by fitting a 3-harmonic waveform to the data from the CR (after eliminating the initial 5 h and last 30 min). The amplitude of the fitted waveform (maximum minus minimum of fitted waveform) was used to derive the 25% crossover threshold (Klerman et al., 2002). We calculated 25% of the amplitude and interpolated between adjacent samples to determine the minute when the plasma levels rose to 25% of that nightly peak level. Phase angles for DLMO and BDNF acrophase were calculated as DLMO-BDNF acrophase for each individual participant.
Two-tailed Student t tests were used to compare data from women and men and to make comparisons by BDNF genotype (Val/Val vs. Val/Met). A Pearson correlation was used to compare melatonin and plasma BDNF amplitude.
Results
Women and men did not differ in age (23.4 ± 4.7 years vs. 23.8 ± 4.8 years; p = 0.77). Consistent with our previous work (Cain et al., 2010), women had a significantly earlier DLMO at baseline than men (2120 h ± 1 h 0 min vs. 2223 h ± 1 h 46 min; p < 0.05) but did not differ from men in habitual bedtime (2305 h ± 58 min vs. 2337 h ± 51 min; p = 0.08). Average BDNF levels during the CR did not significantly differ for women and men (258.82 pg/mL ± 174.95 vs. 360.99 pg/mL ± 353.80; p = 0.24).
Of 39 participants, 24 individuals (62%) demonstrated significant circadian rhythms in plasma BDNF levels as determined by cosinor analysis. A greater proportion of women displayed significant circadian rhythms than men. Twelve of 16 women (75%) displayed significant 24-h rhythms, whereas 12 of 23 men (52%) displayed significant rhythms. The type of advancing protocol used had no influence on the presence or absence of a rhythm (p = 0.98).
The following results were based on the 24 participants (12 women) with significant circadian rhythms (see Table 1 for summary data). The clock time of BDNF acrophase was significantly earlier in women than men (p < 0.01). Relative to the beginning of the CR (hours awake), women displayed a significantly earlier acrophase than men (p < 0.01). Relative to each individual’s DLMO, women were significantly earlier, with a BDNF acrophase that was 7.53 h (±6.84 h) before DLMO, while men had a BDNF acrophase that was 1.79 h (±6.04 h) before DLMO (p < 0.05). The timing of BDNF acrophases relative to clock time and individual DLMO time is presented in Figure 1. A comparison of the pattern of BDNF levels across the day in men and women is presented in Figure 2.
Summary data of melatonin and plasma BDNF rhythms in men and women.
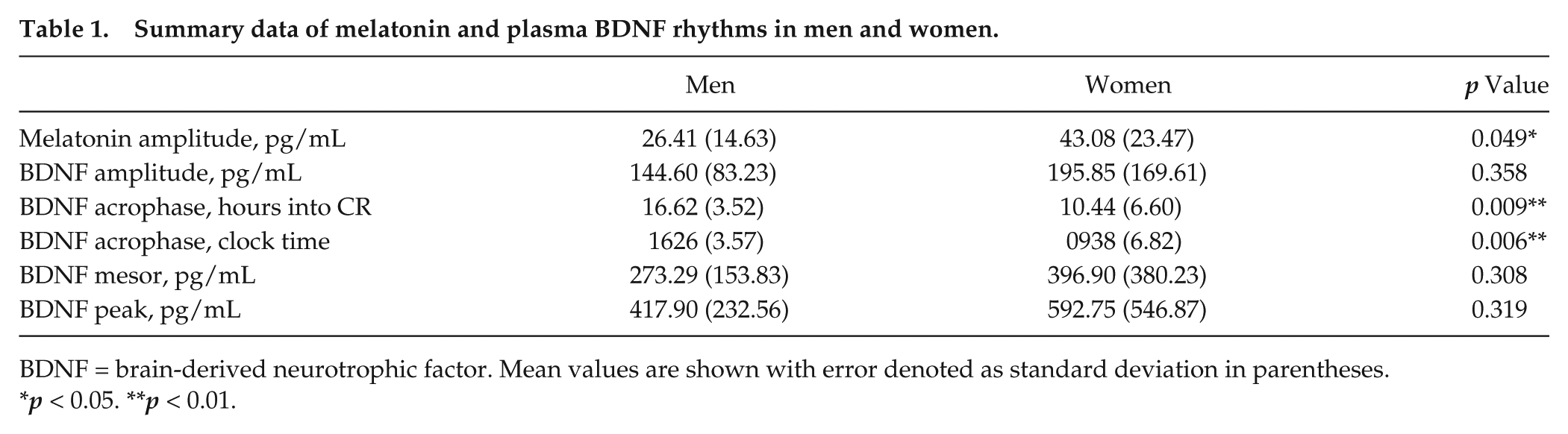
BDNF = brain-derived neurotrophic factor. Mean values are shown with error denoted as standard deviation in parentheses.
p < 0.05. **p < 0.01.
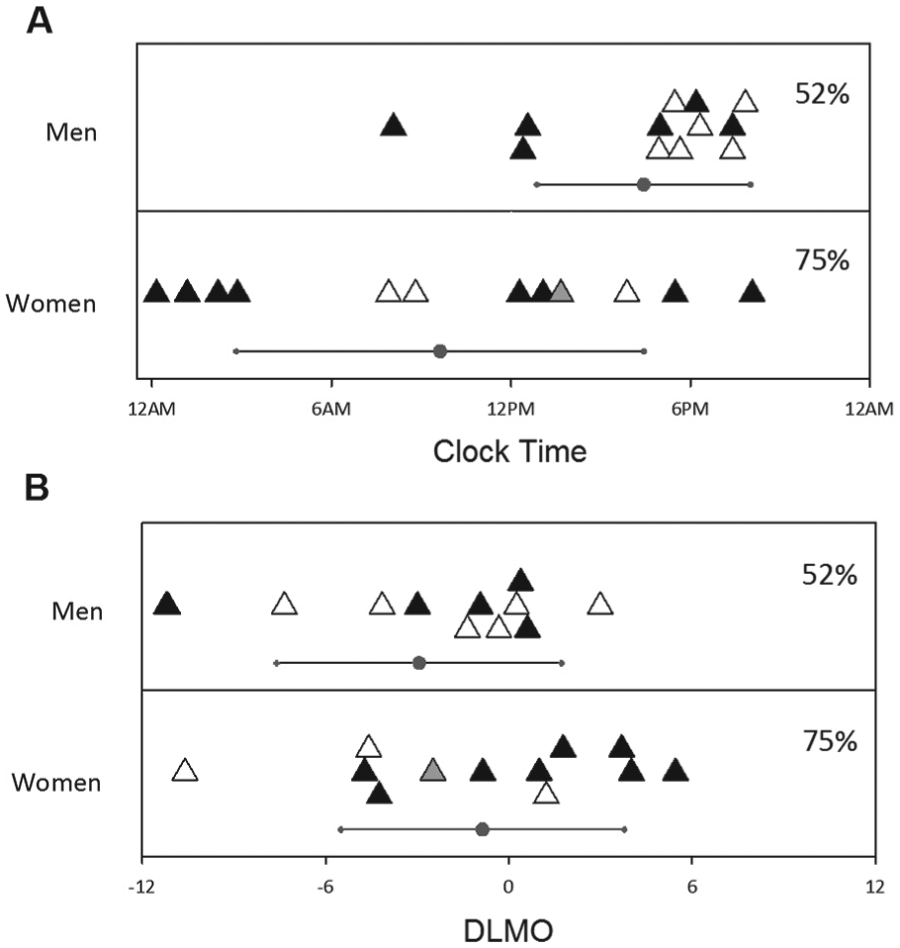
Acrophase of plasma BDNF in men and women. Acrophase data are plotted according to clock time (A) and relative to DLMO (B), with 0 indicating the timing of DLMO. Val/Val genotype is indicated by filled triangles, while open triangles indicate Val/Met genotype. No genetic sample was obtained from the participant represented by the gray triangle. Circles indicate the group mean. Error bars indicate standard deviation. Percentages are the proportion of the whole group of each sex showing significant rhythms.
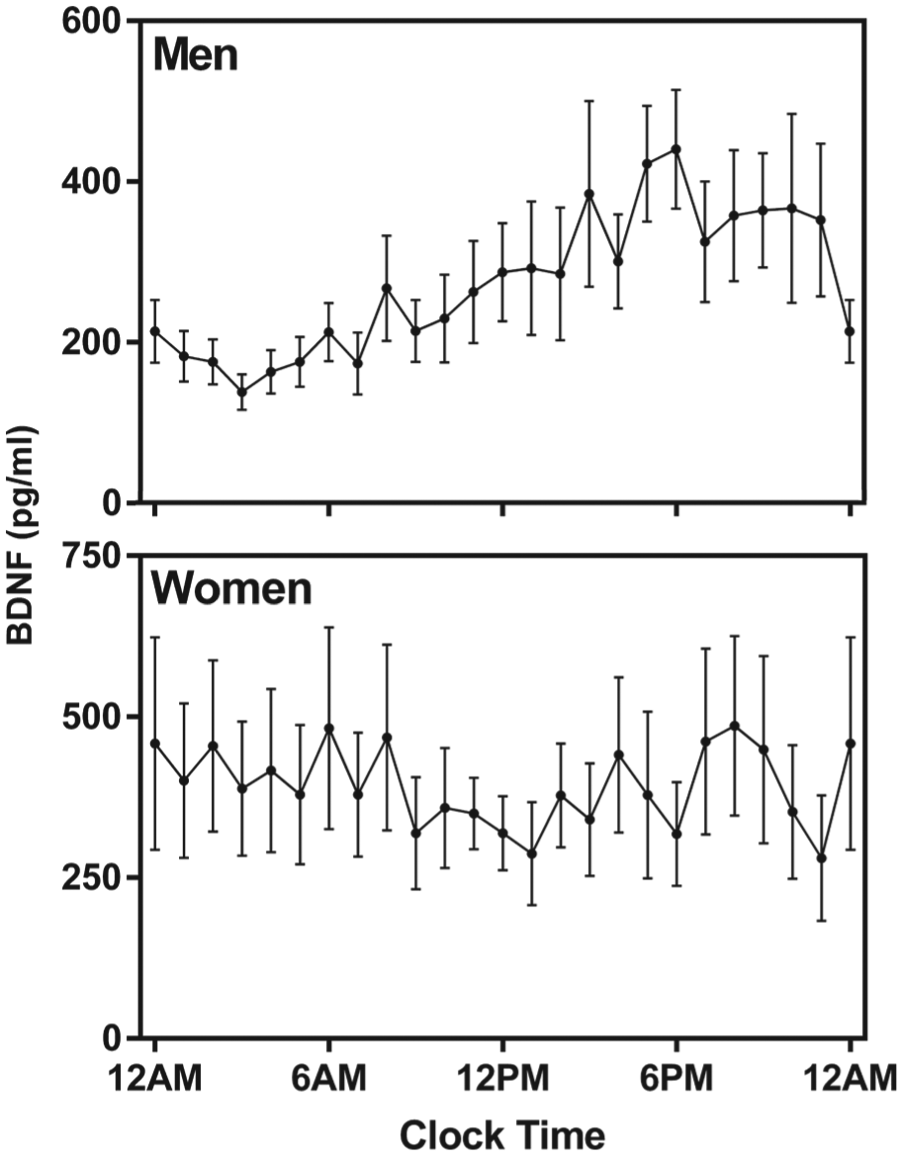
Average (±standard error) plasma BDNF waveforms for men (top) and women (bottom) who displayed significant individual rhythms. Data are plotted according to clock time. Plasma BDNF assay values were averaged per hour.
The mesor (weighted mean) of BDNF levels was not different between women and men (p = 0.31). The amplitude of BDNF was also not different between women and men (p = 0.36). Consistent with our previous work (Cain et al., 2010), we found that melatonin amplitude (using a 3-harmonic fit) was significantly greater in women (43.08 pg/mL ± 23.47) than in men (26.41 pg/mL ± 14.63; p < 0.05). A Pearson correlation revealed no relation of melatonin and BDNF amplitude in men (r = 0.05, p = 0.88), although there was a trend in women (r = 0.57, p = 0.05).
The Val66Met genotype did not influence the timing or levels of plasma BDNF. Genetic samples were obtained for 38 of 39 participants. For all participants (n = 38), those with the Val/Val genotype (n = 22) were not different from the Val/Met genotype (n = 16) in average BDNF levels over the CR (359.97 pg/mL ± 321.89 vs. 225.88 pg/mL ± 140.38; p = 0.13). For the 24 participants showing significant circadian rhythms in BDNF, those with the Val/Val genotype (n = 14) were not different from the Val/Met genotype (n = 9) in mesor (372.38 pg/mL ± 356.03 vs. 290.44 pg/mL ± 174.17; p = 0.53) or amplitude (192.09 pg/mL ± 161.42 vs. 141.13 pg/mL ± 81.69; p = 0.39), although there was a trend toward Val/Val genotype participants having an earlier acrophase than Val/Met participants relative to wake on CR (11.57 ± 1.82 h vs. 16.46 ± 1.23 h; p = 0.06).
Discussion
Previous studies examining rhythms in circulating BDNF have not tested for circadian rhythms within individuals but examined rhythmic patterns of grouped data. Based on these methods, it has been concluded that women have no rhythms in circulating BDNF but that men do (Piccinni et al., 2008; Choi et al., 2011). We examined circadian rhythms within individuals under highly controlled conditions and found not only that women displayed circadian rhythms in plasma BDNF but that individual rhythms were more common in women than in men. Although individual women were more likely to display significant circadian rhythms in plasma BDNF, the timing of acrophase was unrelated to clock time. In men, however, acrophase was related to the clock time. For clock time, all but 3 of the 12 men with rhythmic BDNF levels had an acrophase that was within a 3-h window, while women had acrophases that appeared to be randomly distributed across time. Thus, previous conclusions that men had BDNF rhythms, but women did not, appear to be an artefact of the use of grouped data, taken according to clock time. As demonstrated in Figure 2, when grouped data are examined, men appear to demonstrate a rhythm as a group, whereas women do not.
While women appeared random in the acrophase of BDNF relative to clock time, there appeared to be more grouping when women’s acrophases were analyzed relative to circadian timing (DLMO). Of the 12 women with significant rhythms in BDNF, 11 had acrophases within 6 h of their individual DLMOs (a 12-h window). This finding underscores the importance of measuring plasma BDNF relative to a circadian marker rather than a clock time for all individuals.
For both men and women, the timing of plasma BDNF acrophase relative to DLMO was quite variable compared with other rhythms, such as cortisol and core body temperature, which tend to have very predictable timing, relative to melatonin (e.g., Van Cauter et al., 1994; Czeisler et al., 1999). Thus, it appears that plasma BDNF rhythms are loosely coupled to the circadian pacemaker. This loose coupling may relate the varying sources of circulating BDNF, which include vascular endothelial and smooth muscle cells (Nakahashi et al., 2000), macrophages or lymphocytes (Kerschensteiner et al., 1999), and neurons and glia in the central nervous system (Pan et al., 1998; Karege et al., 2002). Thus, plasma BDNF is likely to be a poor marker of circadian time in individuals. These findings are reminiscent of those of Chua et al. (2013), who found that there were significant individual rhythms in plasma lipids but that these rhythms were largely unpredictable relative to an individual’s melatonin timing, with some lipid peaks that were in antiphase between individuals. Why women in the current study were more variable in the timing of their BDNF rhythms is unknown, but a general tendency for more variable peripheral rhythms in women may relate to such phenomena as the tendency for the timing of chronomodulated chemotherapy to be beneficial for men but not for women (Giacchetti et al., 2012).
Plasma BDNF has been shown to be influenced by menstrual phase, with levels increasing as estrogen increases (Begliuomini et al., 2007), likely due to the interaction with an estrogen response element on the BDNF gene (Sohrabji et al., 1995). Thus, changes in estrogen levels in women across the menstrual phase may have further impaired the ability of previous studies to demonstrate rhythms in plasma BDNF from grouped data. Although estrogen has been found to influence overall levels, it is unlikely to have affected the presence or absence of significant circadian rhythms as measured within individuals. Further, the influence of estrogen on plasma BDNF was unlikely to differentially affect the ability to detect a rhythm between sexes, as amplitude and overall levels were similar in the men and women in our study. In our cohort, we found no significant differences in BDNF acrophase based on self-reported menstrual phase, but the small group size precludes any confident conclusions.
An important limitation of our study is that the 30-h CR protocol was near the end of an 8-day in-laboratory study where circadian phase was advanced using environmental light. We do not know the influence of advancing these schedules on circulating BDNF or whether such an influence might differ between women and men. Thus, no conclusions should be drawn about the specific timing of plasma BDNF relative to either clock time or DLMO. Indeed, our finding of a BDNF acrophase in men at ~1800 h is different from previous reports using grouped data (e.g., ~0800-0900 h; Piccinni et al., 2008; Choi et al., 2011). It seems likely that the timing of plasma BDNF might be different relative to clock time and DLMO under stable light-dark and sleep-wake schedules. Future studies will be needed to clarify this timing.
Previous studies using clock time to collect group data have concluded that women have no rhythms in plasma BDNF, while our study examining individual rhythms demonstrated that women do exhibit circadian rhythms but that the timing of plasma BDNF acrophases are highly variable. In both men and women, plasma BDNF can vary greatly across the day, with peak timing that is very different between individuals. These results suggest that the use of single-sample plasma BDNF as a biomarker of disease state may be problematic, especially when studying women. Multiple samples taken within individuals across the day, however, may reveal important links to disease and recovery.
Footnotes
Acknowledgements
We thank the study participants as well as the staff of the Harvard Clinical and Translational Science Center (CTSC) and the Chronobiology Core of the Division of Sleep Medicine of Brigham and Women’s Hospital (BWH) for their contributions. The work presented in this manuscript was performed at the Division of Sleep and Circadian Disorders, Department of Medicine and Department of Neurology, Brigham and Women’s Hospital, Boston, Massachusetts; the Division of Sleep Medicine, Harvard Medical School, Boston, Massachusetts; and the Center for Human Genetic Research, Massachusetts General Hospital, Boston, Massachusetts. This study was supported by the National Space Biomedical Research Institute (NSBRI) grant HFP01601. Inpatient studies were conducted in the CTSC and supported in part by UL1 RR025758 from the National Center for Research Resources (NCRR) of the National Institutes of Health (NIH). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NSBRI, NCRR, NIH or BWH.
Conflict of Interest Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
