Abstract
Synchronization of male and female locomotor activity plays a critical role in ensuring reproductive success, especially in semelparous species. The goal of this study was to elucidate the effects of individual chemical signals, or pheromones, on the locomotor activity in the sea lamprey (Petromyzon marinus). In their native habitat, adult preovulated females (POF) and ovulated females (OF) are exposed to sex pheromone compounds that are released from spermiated males and attract females to nests during their migration and spawning periods. In this study, locomotor activity of individual POF and OF was measured hourly in controlled laboratory conditions using an automated video-tracking system. Differences in the activity between a baseline day (no treatment exposure) and a treatment day (sex pheromone compound or control exposure) were examined for daytime and nighttime periods. Results showed that different pheromone compound treatments affected both POF and OF sea lamprey (p < 0.05) but in different ways. Spermiated male washings (SMW) and one of its main components, 7α,12α,24-trihydroxy-5α-cholan-3-one 24 sulfate (3kPZS), decreased activity of POF during the nighttime. SMW also reduced activity in POF during the daytime. In contrast, SMW increased activity of OF during the daytime, and an additional compound found in SMW, petromyzonol sulfate (PZS), decreased the activity during the nighttime. In addition, we examined factors that allowed us to infer the overall locomotor patterns. SMW increased the maximum hourly activity during the daytime, decreased the maximum hourly activity during the nighttime, and reduced the percentage of nocturnal activity in OF. Our findings suggest that adult females have evolved to respond to different male compounds in regards to their locomotor activity before and after final maturation. This is a rare example of how species-wide chemosensory stimuli can affect not only the amounts of activity but also the overall locomotor pattern in a vertebrate species.
For animals to survive, it is vital for them to be synchronized with their external environments by coordinating the timing of behavioral and physiological cycles. This is especially true for those animals that undergo seasonal reproduction. Diel rhythms are used as a timekeeping system that allows an animal to anticipate and prepare for changes in the physical environment. Diel locomotor rhythmicity is prevalent in nature and can be influenced by several exogenous and endogenous factors. Studies of activity rhythms traditionally focus on the environmental effects of photoperiod or temperature, less commonly looking into other sensory modalities, such as olfaction. The sea lamprey, Petromyzon marinus, of the Laurentian Great Lakes has a life cycle consisting of several stages in which changes in locomotor activity patterns occur concurrently with changes in exposure to conspecific signals in the form of pheromones that can modulate behavior and provide information to the receivers, such as sex or reproductive status (Beamish, 1980; Albone et al., 1984; Johnston, 2000; Meckley et al., 2012). This allows for a rare opportunity to investigate the effects of an environmental exogenous cue on locomotor activity and patterns.
Previous studies have documented locomotor patterns in particular life stages of the sea lamprey. Lamprey larvae live for several years in river and sediment beds, and although they spend most of their time burrowed, they are primarily nocturnal when active (Hardisty and Potter, 1971a; Potter, 1980; Almeida et al., 2005). Larvae metamorphose into a juvenile parasitic form and migrate downstream to lakes, during which they have been observed to be behaviorally arrhythmic and feed on larger fish (Hardisty and Potter, 1971b). After feeding in open waters for a little over a year (Bergstedt and Seelye, 1995), immature (prespermiated male and preovulated female) lampreys undertake a spring migration through rivers and streams to appropriate spawning grounds, guided by larval olfactory cues (Sorensen et al., 2003; Sorensen and Vrieze, 2003; Sorensen et al., 2005; Wagner et al., 2009). Petromyzonol sulfate (PZS or PS) is one of the metabolites released by the larvae that act as a potent odorant (Teeter, 1980; Venkatachalam, 2005; Fine and Sorensen, 2010). During this upstream migration, the immature lampreys exhibit a nocturnal locomotor activity pattern, being active at night and seeking refuge during the day (Manion and McLain, 1971; Kelso and Gardner, 2000; Vrieze et al., 2011; Binder and McDonald, 2007). Males make this migration prior to the females, and as they mature, they become arrhythmic, showing nest-building activity during both the day and night (Applegate, 1950; Manion and Hanson, 1980). Because sea lampreys are semelparous and die shortly after spawning (Applegate, 1950), and during migration and spawning periods, sea lampreys do not eat and rely on nutrients stored during their parasitic phase for gonad maturation and spawning behaviors, the timing of reproduction in this species is especially crucial (Beamish, 1979; Madenjian et al., 2003).
In this species, mature (spermiated) males release an array of sex pheromones that influence the activity of mature (ovulated) females. This includes the pheromone 7α,12α,24-trihydroxy-5α-cholan-3-one 24 sulfate (3kPZS), which guides ovulated females upstream over long distances to nests (Li et al., 2002; Siefkes et al., 2005; Johnson et al., 2006; Johnson et al., 2009; Johnson et al., 2012a). Washings collected from spermiated males (SMW), which include 3kPZS and several other active compounds, have been shown to have similar effects as 3kPZS and also induce additional spawning behaviors (Siefkes et al., 2005; Johnson et al., 2006; Johnson et al., 2009; Johnson et al., 2014). In a preceding study (Walaszczyk et al., 2013), we identified the effects of SMW and maturity stage on locomotor activity by showing that SMW reduced total activity in preovulated and ovulated females during the normally active nighttime hours. In addition, ovulated females, which are overall less active than preovulated females, showed an increase in daytime activity and a disruption of their nocturnal activity, which led to overall arrhythmic locomotion (Walaszczyk et al., 2013).
What remains unclear from the preceding study are the specific chemical compounds in SMW that contribute to the interaction between maturation and locomotor pattern changes. We hypothesize that preovulated and ovulated females have evolved to respond differently to specific signal components identified in SMW, which will be reflected in their locomotor responses. We speculate that this is due to a difference in life history, as preovulated females must be guided long distances to appropriate spawning grounds, whereas ovulated females must coordinate their reproductive activity with spermiated males at the spawning grounds. To test this hypothesis, we measured diel locomotor activity as well as changes in locomotor pattern characteristics in the form of nocturnal activity percentages and the maximum hourly activity during the daytime and nighttime periods of adult female sea lampreys in controlled laboratory conditions during exposure to compounds found within SMW.
Materials and Methods
Animals
Standard operating procedures for transporting, handling, maintaining, and euthanizing sea lampreys were approved by the Institutional Committee on Animal Use and Care of Michigan State University (AUF 02/13-041-00). Sea lampreys were captured from late April through mid-July 2013 and 2014 in standard mechanical traps operated by the U.S. Fish and Wildlife Service and Department of Fisheries and Oceans, Canada, from several tributaries of Lakes Huron and Michigan. Sea lampreys were transported to the laboratory of the University Research Containment Facility of Michigan State University each year after collection.
Females and males were separated based on the protocol described by Vladykov (1949) and held in chilled water (6-8 °C) in large flow-through tanks provided with air stones. All sea lampreys were held in laboratory conditions for less than 30 days prior to experimental trial acclimation. Maturity stages were separated by using the protocol established by Siefkes et al. (2003). Ovulated females were classified as such if a defined keel was present and eggs were expressed by manual pressure. Spermiated males (n = 20) were used for washings collection and were identified as such based on the presence of a dorsal ridge and sperm expression when manual pressure was applied.
Experimental Condition and Treatments
Experiments were conducted at the University Research Containment Facility of Michigan State University. Preovulated (weight: mean ± SE = 264.5 ± 7.1 g, n = 24; length: mean ± SE = 50.5 ± 0.5 cm, n = 24) and ovulated (weight: mean ± SE = 229.9 ± 9.8 g, n = 38; length: mean ± SE = 46.1 ± 0.6 cm, n = 38) females were acclimated together (4 or 8 females) for 6 days prior to each experiment in a large circular, aerated flow-through tank (120.1 cm diameter; 50.2 cm water height). Animals were held individually in experimental tanks, which were identical to the acclimation tank, for the duration of trials. A PVC pipe (10 cm diameter, 35.5 cm long) at the bottom of the tank provided a refuge from the light. Tanks were adjusted to a constant temperature of 15 °C and equipped with an aquarium air stone (25.4 cm; Penn-Plax, Hauppauge, NY, USA). Temperature was checked at least twice daily using a digital thermometer (Extech, South Burlington, VT, USA). Temperature loggers (Hobo Temperature Data Logger; Onset, Bourne, MA, USA) were placed in each tank during a subset of trials and recorded the temperature every 30 min to ensure the temperature remained constant. Each tank was held under a 14:10-h light-dark cycle (lights-on, 0600 h; lights-off, 2000 h) using light timers and exposed to approximately 500 lux on average across the surface of the water of 6500K light during daytime hours and <1 lux dim red light (740 nm) during nighttime hours. Modified trolling motors were used (Minn Kota Model Endura C2 Transom-Mount; Johnson’s Outdoor, Racine, WI, USA) to imitate slow-moving stream conditions (mean ± SE = 0.04 ± 0.01 m/sec). Flow rates were determined using a portable flow meter (Model 2000; Hach Marsh-McBirney, Loveland, CO, USA). Trolling motors were turned on for 24 h every 3 days in the acclimation tank and remained on for the experiment duration.
Preovulated female trials were conducted between 3 June 2013 and 26 June 2013, whereas ovulated female trials were conducted between 28 July 2013 and 27 August 2013 or 14 July 2014 and 3 September 2014. These dates are representative of the sea lamprey adult life cycle and, therefore, when each maturity stage was available for experimentation. On the morning of the start of each experiment, female lampreys were placed individually in 1 of 4 randomly selected experimental tanks at 0800 h and allowed to acclimate for 4 h. At 1200 h, the trial began, and 24 h of activity (baseline period) were recorded using the video-tracking program EthoVision XT with the Multiple Arena Module (Version 9; Noldus Information Technology, Asheville, NC, USA) coupled to 4 CCD cameras (Model EQ-610; EverFocus, Taipei, Taiwan). The system ran on a Dell Precision Intel-based computer with digitizing board (Euresys H264; San Juan Capistrano, CA, USA) to enable simultaneous live tracking and digital video recording with the EthoVision software. After 24 h, a treatment was introduced to each tank (a final tank concentration of 10–10 M 3kPZS [Bridge Organics, Vicksburg, MI, USA], 10–10 M PZS [Bridge Organics], SMW with a concentration of 10–10 M 3kPZS, or an equivalent amount of methanol [MeOH, vehicle; Sigma-Aldrich, St. Louis, MO, USA]). The treatment selected for each tank was randomized prior to each trial. SMW used during all experiments were collected by placing 20 spermiated males into a tank of 200 L of aerated water with no flow for 36 h. Then, 1-L water samples were collected and frozen at −20 °C until used. Concentrations of natural 3kPZS in SMW were quantified using ultra-high-performance liquid chromatography–tandem mass spectrometry according to the protocol described by Li et al. (2011). At the start of the treatment day, a dose of the desired compound(s) was added to bring concentrations up to the appropriate levels. Solutions based on each tank’s volume and flow velocity were made and pumped into each tank via reef dosing pumps (Model The Sentry; Innovative Aquatics, Cumming, GA, USA) to account for outflow and keep the treatment in the tank held at a constant concentration throughout the trial duration. An additional 24 h of activity were then recorded and deemed the treatment period. At the end of each experiment, females were euthanized with 0.5% MS222 (Tricaine methanesulfonate; Sigma-Aldrich), and tissues were collected for future experiments.
Locomotor Activity Recording and Data Analysis
Using EthoVision XT 9, we tracked movement defined as the seconds the animal was active per hour in 1-h bins across the 48-h trial. Hour times represent the start of the 1-h time bin. To account for individual variation and small sample sizes, differences between the movement time of the baseline control day and treatment day were used for all analyses. To determine if pheromone treatments influenced the diel pattern of locomotor activity, movement time data were separated by hour and determined to be either daytime (0600-2000 h) or nighttime (2000-0600 h). To account for unanticipated, artificial bursts of activity when the lights turned on and off, hours 0600 and 2000 were not included in the analyses. Occasionally, ovulated females, which are more fragile because they are physically degrading as they approach the final act of spawning, died during the trial. If this occurred, the trial was stopped and data were not included in the analysis. All statistical analyses were performed in SAS version 9.4 (SAS Institute, Cary, NC, USA).
Effects of the pheromone compounds and maturity on movement were analyzed for each time period (daytime or nighttime) using repeated-measures general linear models implemented within the PROC MIXED function of SAS. The fixed effects of treatment (MeOH control vs. SMW, 3kPZS, and PZS), maturity (preovulated females [POF] vs. ovulated females [OF]), and their 2-way interaction were fit to the model, with the random terms controlling for repeated measures within the individual number (ID) of each animal. Akaike information criteria (AIC) were used to select the best-fitted model prior to examining the hypothesis tests. The autoregressive covariance structure was selected for the model, which has homogeneous variances and correlations that decline exponentially with distance, due to the nature of the repeated data collected across time. Post hoc comparisons were performed with the Dunnett test, which allows for comparisons between treatment groups and a control (reference) group, for each maturity stage (Zar, 1999) or with a Bonferroni correction when comparing 2 treatment groups. To test that there were no tank effects for the preovulated and ovulated female groups, we compared the baseline movement from each treatment group using the same model.
To investigate how pheromone compounds and maturity influenced the diel locomotor pattern, we examined changes in daily movement by calculating the differences in percentage of nocturnal activity after exposure to each treatment. This was calculated by taking the amount of activity during the nighttime period, dividing it by the total amount of activity during the daytime and nighttime, and turning it into a percentage. We then calculated the differences of these percentages between the baseline and treatment days, comparing among different treatment groups and maturity stages. In addition, we tested the effects of each of these factors on the maximum hourly activity during the daytime and nighttime. These were defined as the maximum movement during a 1-h time period within the daytime or the nighttime. The corresponding maximum hourly activity differences were then calculated by taking the difference between the treatment period maximum hourly activity and the baseline period maximum hourly activity. We conducted 2-way analyses of variance (ANOVAs) using PROC GLM of SAS to account for our unbalanced design for the fixed effects of treatment (MeOH control vs. SMW, 3kPZS, and PZS), maturity (POF vs. OF), and their 2-way interaction on nocturnal activity differences and each time period’s maximum hourly activity. When significant (α < 0.05), the Dunnett post hoc test was used to test for differences between groups for each maturity stage.
Results
After transfer to the tanks, sea lampreys had transient bursts of activity but settled down to normal behavior prior to the start of the trial. Temperatures remained constant during the experiments (temperature: mean ± SE = 15.1 ± 0.01 °C across the duration of testing of all tanks, n = 7 trials). There were no treatment effects during the first 24 h of the experiment in which no treatment odor was administered, neither for the nighttime (preovulated females: Figure 1, F3, 189 = 0.21, p = 0.89; ovulated females: Figure 2, F3, 279 = 0.31, p = 0.82) nor for the daytime (preovulated females: Figure 1, F3, 109 = 0.77, p = 0.51; ovulated females: Figure 2, F3, 82 = 1.17, p = 0.32). These data were used as a baseline control day to make comparisons with the treatment day.
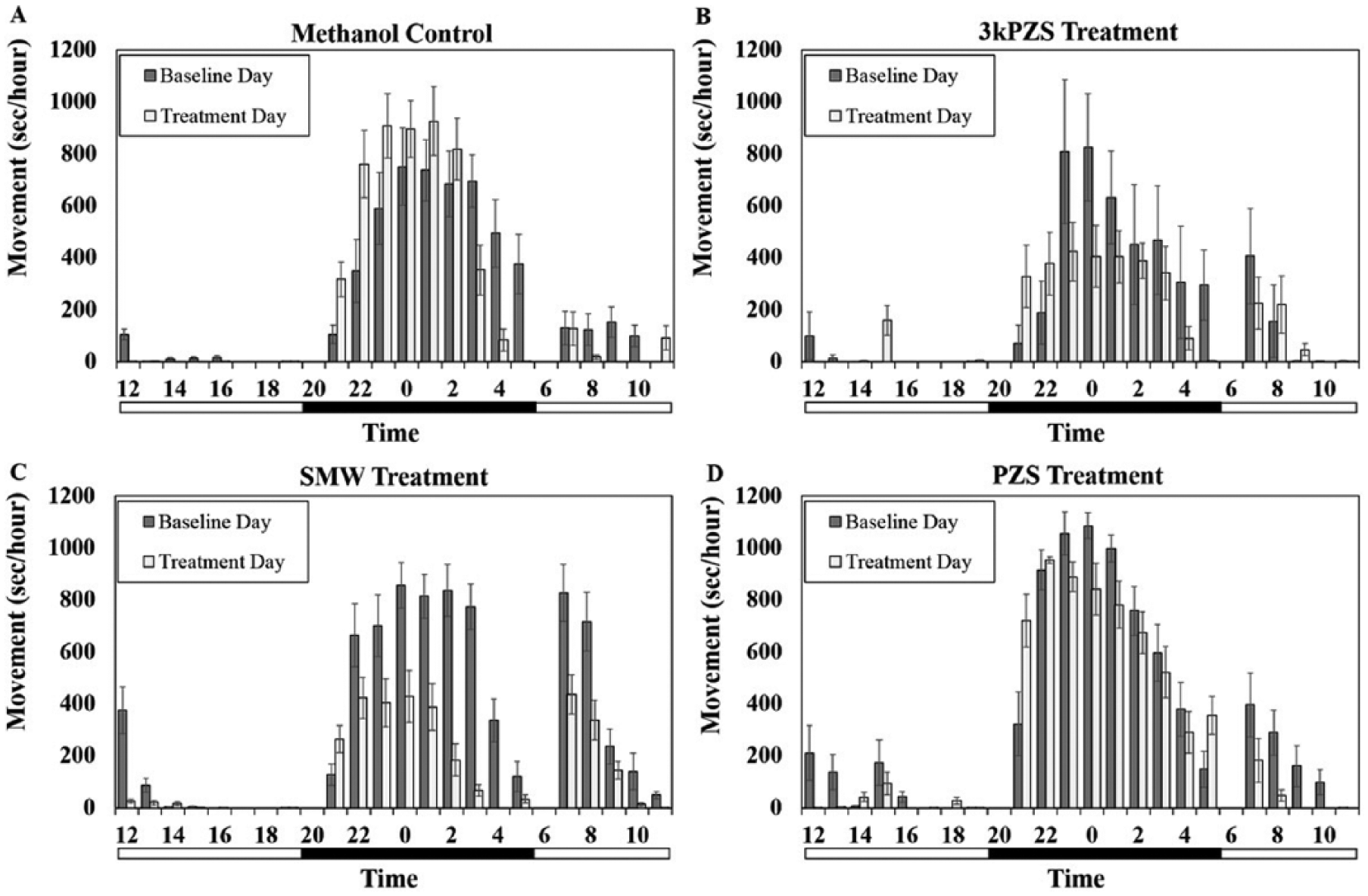
Effects of sex pheromone compounds on locomotor activity in preovulated females. Vertical bars denote 1 standard error. The bar below the graph is split into black for nighttime and white for daytime.
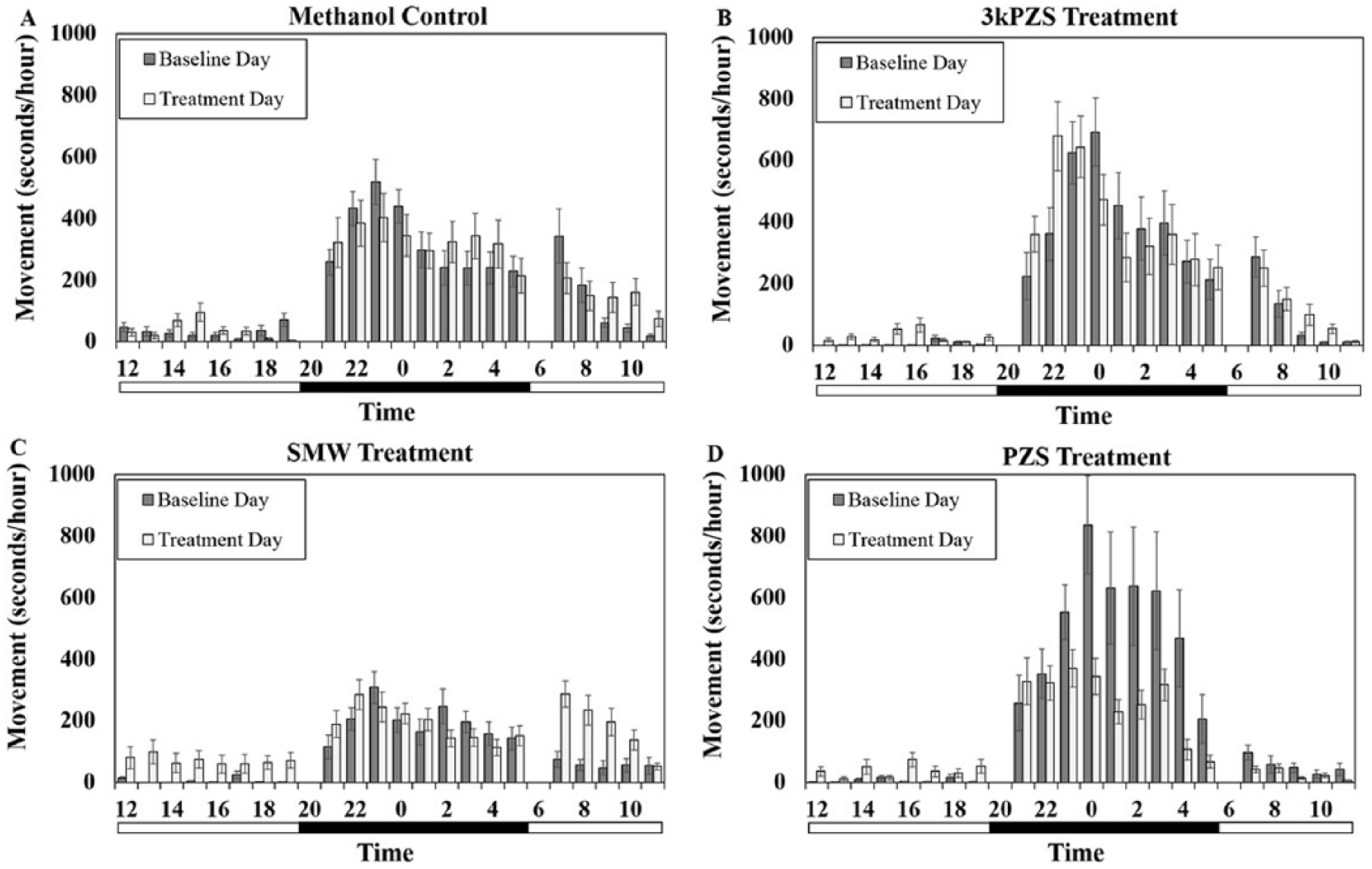
Effects of sex pheromone compounds on locomotor activity in ovulated females. Vertical bars denote 1 standard error. The bar below the graph is split into black for nighttime and white for daytime.
Nighttime Movement
Preovulated and ovulated females both demonstrated a nocturnal locomotor pattern during the baseline day. There was a significant effect of the treatment by maturity interaction (F3, 143 = 3.53, p = 0.017) on movement during the nighttime period (Figure 3B). During the nighttime, SMW reduced locomotor movement in preovulated females compared to the methanol control (Dunnett post hoc test: t = −4.04, p = 0.00061). Additionally, the pheromone compound 3kPZS produced a reduction in locomotor movement in this maturity stage (Dunnett post hoc test: t = −2.22, p = 0.028) and had the same effect on movement as SMW (Bonferroni correction: 3kPZS vs. SMW: t = 1.79, p = 0.63) There was no effect in preovulated females exposed to PZS (Dunnett post hoc test: t = −1.41, p = 0.55).
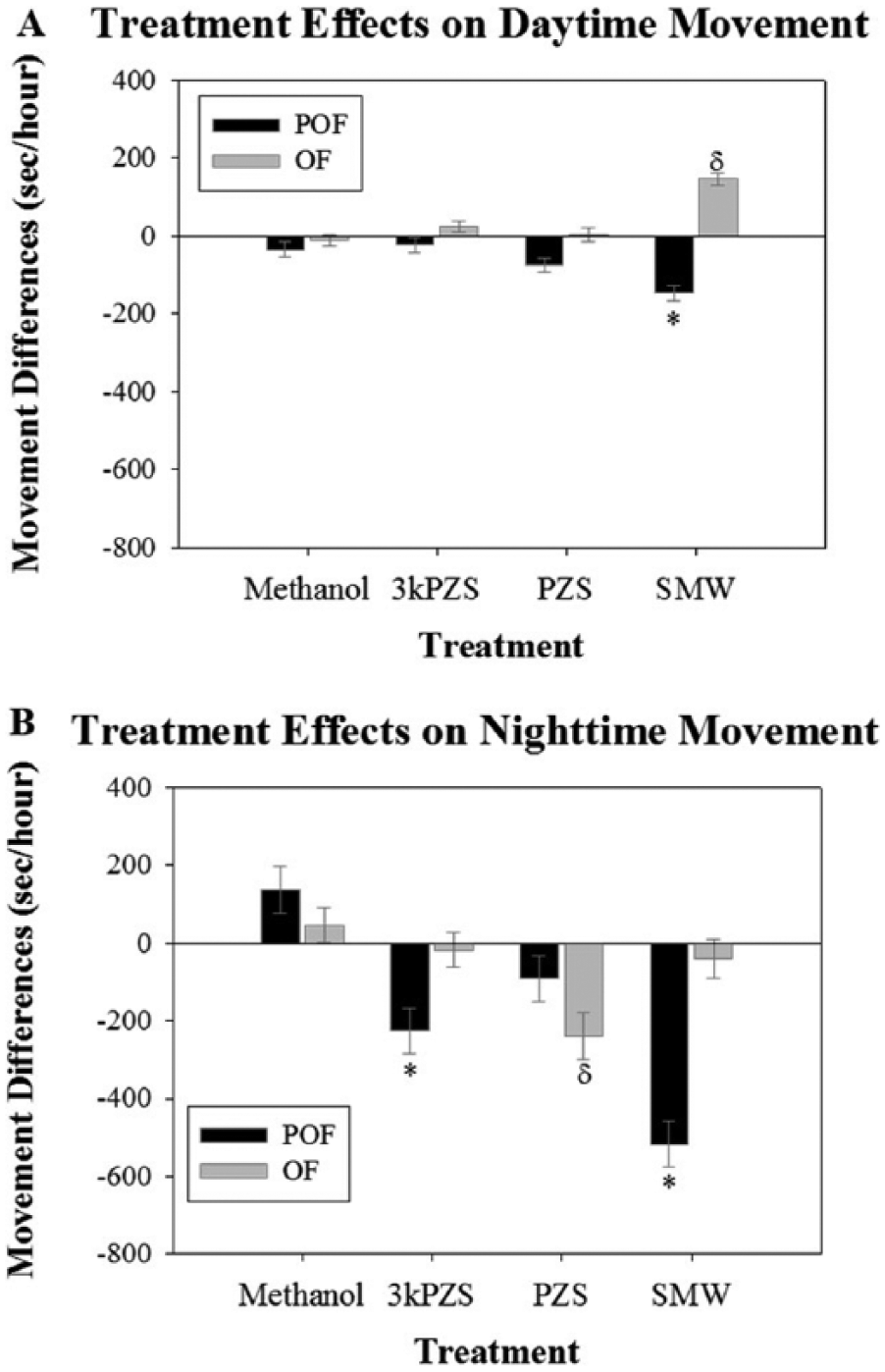
Effects of sex pheromone compounds on (A) daytime movement and (B) nighttime movement differences between the baseline and treatment days of adult female sea lampreys. POF denotes preovulated females and OF denotes ovulated females. The asterisk (*) represents groups that are significantly different from the preovulated, methanol control. δ represents groups that are significantly different from the ovulated, methanol control. Vertical bars denote 1 standard error.
Interestingly, as opposed to preovulated females, in ovulated females, the pheromone compound PZS reduced the activity during the nighttime hours compared to the methanol control. (Figure 3B, Dunnett post hoc test: t = −3.11, p = 0.013). There were no effects documented from exposure to 3kPZS (Dunnett post hoc test: t = −0.52, p = 0.99) or SMW (Dunnett post hoc test: t = −0.68, p = 0.985).
Daytime Movement
During the daytime hours, the treatment-by-maturity interaction (F3, 135 = 6.44, p = 0.00040) affected movement (Figure 3A). While treatment did not affect daytime movement within the preovulated females (F3, 144 = 2.22, p = 0.089), treatment did affect ovulated female locomotor activity (F3, 128 = 5.41, p = 0.0015). Locomotor movement increased during the daytime when ovulated females were exposed to SMW (Figure 3A, Dunnett post hoc test: t = 3.78, p = 0.00020) compared to the methanol, ovulated female control. In contrast, 3kPZS (Dunnett post hoc test: t = 0.91, p = 0.93) and PZS (Dunnett post hoc test: t = 0.28, p = 0.99) had no effect during this time period.
Diel Movement
The treatment-by-maturity interaction affected the change in the percentage of nocturnal activity (F3, 52 = 3.97, p = 0.013). Further analysis revealed that in ovulated females, the percentage of nocturnal activity was reduced compared to the ovulated, methanol control (Figure 4A, Dunnett post hoc test: t = −2.84, p = 0.03). Similarly, the daytime and nighttime maximum hourly activities were affected by the interaction between treatment and maturity stage (F3, 53 = 3.9, p = 0.013 and F3, 53 = 4.85, p = 0.0047, respectively). Ovulated females exposed to SMW showed an increase in movement during the daytime maximum hourly activity (Figure 4B, Dunnett post hoc test: t = 3.95, p = 0.0016) and a reduction in movement during the nighttime maximum hourly activity (Figure 4C, Dunnett post hoc test: t = −2.41, p = 0.019) compared to the ovulated, methanol control. There were no effects on the movement during the maximum hourly activity in preovulated females for daytime (Dunnett post hoc tests; 3kPZS: t = −0.57, p = 0.99; PZS: t = −0.13, p = 0.90; SMW: t = −1.31, p = 0.63) or nighttime (Dunnett post hoc tests; 3kPZS: t = −1.4, p = 0.557; PZS: t = −1.3, p = 0.63; SMW: t = −2.26, p = 0.13). Other treatments had no effect on movement during the maximum hourly activity in ovulated females during the daytime (Dunnett post hoc tests; 3kPZS: t = 2.26, p = 0.13; PZS: t = 1.79, p = 0.37) or nighttime (Dunnett post hoc tests; 3kPZS: t = 0.21, p = 0.83; PZS: t = −2.35, p = 0.07).
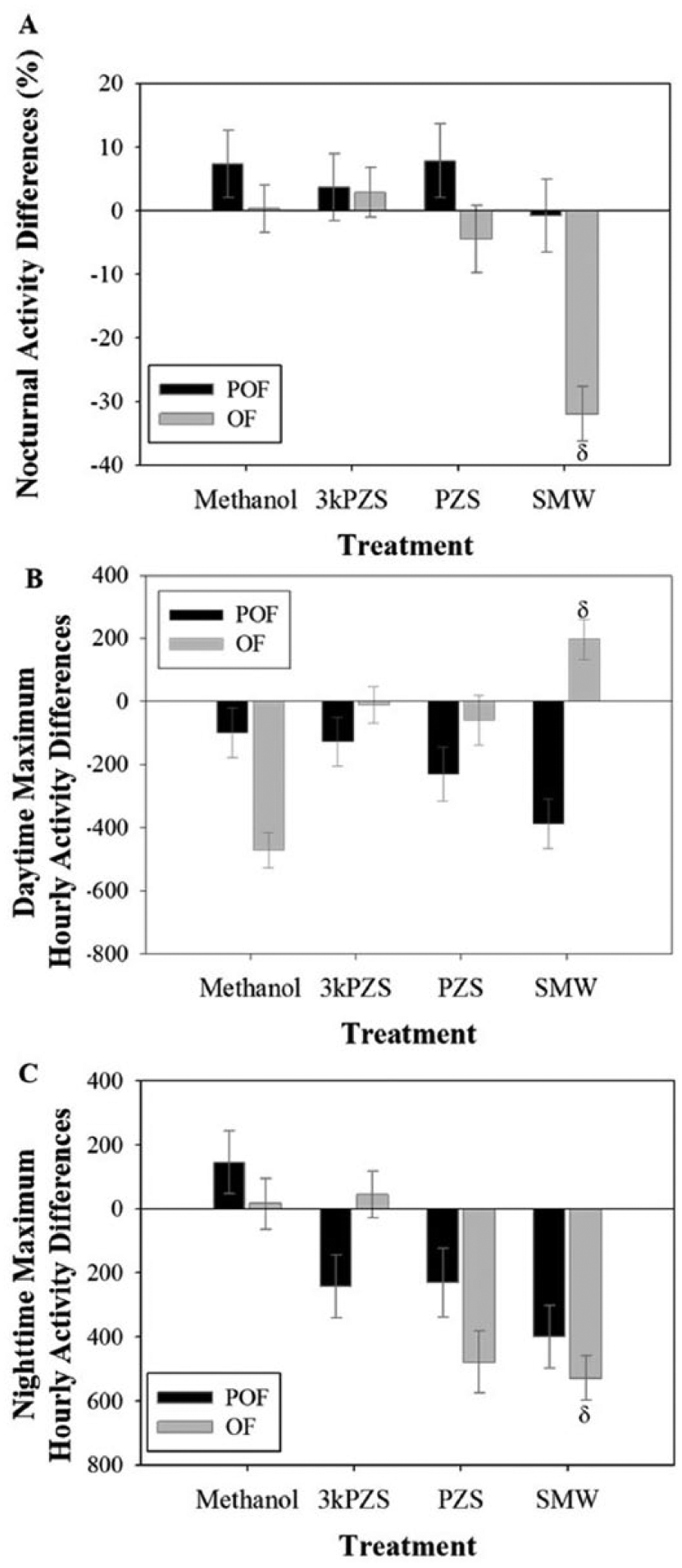
Effects of sex pheromone compounds on locomotor pattern characteristics of adult female sea lampreys. (A) The differences in percentage nocturnal activity between the baseline and treatment days. (B) The differences in movement during the daytime maximum hourly activity between the baseline and treatment days. (C) The differences in movement during the nighttime maximum hourly activity between the baseline and treatment days. POF denotes preovulated females, and OF denotes ovulated females. δ represents group that is significantly different from the ovulated, methanol control. Vertical bars denote 1 standard error.
Discussion
We have identified 2 specific molecules released from spermiated male sea lampreys that have diverse effects on the locomotor activity of different stages of adult females. Our results show that the response to specific compounds of SMW is maturity dependent, which is reflected in the distinct responses during the nighttime of preovulated females to 3kPZS and ovulated females to PZS. Additionally, we have confirmed our previous results (Walaszczyk et al., 2013) that SMW have a differential effect on each maturity stage. While SMW reduce nighttime activity in preovulated females, they do not have an effect on the percentage of nocturnal activity or the daytime maximum hourly activity, thus not affecting the overall diel pattern. Contrastingly, SMW contribute to the alteration of the diel pattern of locomotor activity in ovulated females, reducing the percentage of nocturnal activity and nighttime maximum hourly activity as well as increasing the daytime maximum hourly activity. This is consistent with our previous studies, in which ovulated females become arrhythmic in their locomotor activity when exposed to SMW in semi-natural field conditions (Walaszczyk et al., 2013). Although these results are also congruent with previous literature in other species documenting that sex pheromones can affect locomotor activity amounts in insects (August, 1971; Rust and Bell, 1976; Hawkins, 1978; Pontes et al., 2014) as well as fish (Bjerselius et al., 1995; Amcoff et al., 2014), we have provided a rare example of how a specific, exogenous chemical cue can alter not only the amounts of activity but also the overall pattern of locomotor activity in a vertebrate species.
In natural environments, adult female lampreys are exposed to a complex array of pheromone plumes, which change as females make their upstream migration and mature. The olfactory system of sea lampreys contains very sensitive and specific receptor mechanisms for both 3kPZS and PZS (Li et al., 1995; Li and Sorensen, 1997; Siefkes and Li, 2004). 3kPZS is released in high levels from spermiated males, whereas PZS in the environment comes from lamprey larvae (Haslewood and Tökés, 1969; Sorensen et al., 2005) and from spermiated males (Brant et al., 2013). While PZS can also be derived from a larval population, the spawning males are often upstream of these populations. Therefore, when females get close to the nests, the influences of PZS are most likely from spermiated males. It is possible that 3kPZS is also released by sea lampreys of other stages, such as larvae, although no report has documented this possibility yet. When making their upstream migration, preovulated females are at a further distance from the spermiated males and exposed to more diluted pheromone concentrations than ovulated females. In this ethological context, it makes sense these preovulated females would respond to 3kPZS, which is more highly concentrated than PZS in this environment. Ovulated females, however, being closer to the spawning grounds, would be exposed to more concentrated levels of PZS at the spawning grounds compared to downstream, where it becomes diluted as the distance is increased away from the sources of the compound. We speculate that preovulated and ovulated females have evolved responses to different chemical signals at different concentrations or in different ratios in regards to their biological locomotor activity and pattern. Our results provide evidence that PZS is a contributing factor to the documented ovulated female switch in activity from nocturnal to arrhythmic, which assists in coordinating the vital, limited timing of activity between mature males and females, increasing the chance for reproduction. PZS may contribute to a signal for ovulated females to stay or resample a specific area that represents spawning territory with appropriate mates, thus reducing the energy expenditure on locomotor activity to reserve energy for gonadal development and the act of reproduction.
We have additionally provided evidence that several compounds within SMW are interacting to modulate overall locomotor activity and patterns, as the reduction in both diurnal and nocturnal activity by SMW for preovulated females and the increase in diurnal activity in ovulated females cannot be solely explained by 3kPZS or PZS. This is consistent with insect studies that have shown that most characterized pheromones are complex mixtures that only produce strong behavioral responses when all compounds are present or represented in relative proportions of different components (Linn et al., 1987). These results suggest that there are additional effective compounds and are concurrent with preceding studies on sea lampreys that document that more than one component of SMW is needed to trigger the entire array of mature female sexual behaviors (Siefkes et al., 2005; Johnson et al., 2009; Johnson et al., 2012a; Li et al., 2013). In these experiments, although 3kPZS elicits the same robust upstream movement to an odorant release point as SMW do, ovulated females show a preference for the full compound suite of SMW, spend more time in nests baited with SMW, and show more spawning behaviors (Johnson et al., 2012b). Potential candidates for additional, effective odor cues include alternative bile salt compounds released by spermiated males, such as 3 keto-1-ene PZS or 3,12-diketo-4,6-petromyzonene-24-sulfate (DKPES), which attract ovulated females (Li et al., 2013; Johnson et al., 2014; Brant, 2015).
Our results are supportive of previous field tests, in the fact that ovulated females exposed to SMW show increased activity during the day (Walaszczyk et al., 2013). In these previous studies, however, ovulated females exposed to SMW also had reduced activity during the nighttime hours, whereas the results presented here are not significant. One explanation is that previous studies were conducted in the field, which has drastically different conditions compared to the laboratory setting. Behavior was also measured over several days as females matured and were exposed to a SMW or a control, whereas in the current study, the duration of exposure is much shorter, allowing us to examine the short-term, continuous, and high concentration effects of each compound. Longer treatment exposure may have revealed a gradual switch to an arrhythmic state. An additional explanation is that full effects are dependent on the mixture of compounds found within SMW. Each batch gathered from spermiated males has its own unique compound profile. To control for this, the SMW are standardized to an equal amount of 3kPZS but not PZS. It is possible that the batch we used in the present study had a lower concentration of PZS compared to that of previous studies and, therefore, did not produce a significant effect during the nighttime hours.
We have revealed the behavioral effects of individual pheromone compounds on locomotor activity, and while it has been demonstrated from a neurological standpoint how pheromones affect locomotion in the sea lampreys (Derjean et al., 2010), the mechanism in which pheromones interact with endogenous biological clocks that control rhythmicity remains to be elucidated. This could involve neuronal activation of the pineal complex, which is vital for maintaining the timing of locomotor movement (Morita et al., 1992; Binder and McDonald, 2007) in this species. This complex indirectly connects to the pheromone-sensitive olfactory bulb and projects to the dorsal mesencephalic tegmentum, which is an important motor region in this species (Yáñez et al., 1993; Yáñez et al., 1999). Additionally, circadian locomotor activity could be regulated through influences of the molecular circadian clock transcriptional and translational feedback loops consisting of several clock genes found within vertebrates that are critical for adapting an organism’s body to the external environment (Zhang and Kay, 2010). Future work should aim to investigate the roles of these systems in locomotor activity alterations via pheromone exposure.
This study has revealed 2 specific pheromone compounds released by spermiated male sea lampreys that produce different effects on locomotor activity and patterns in adult preovulated and ovulated females. In addition to these 2 compounds, we have demonstrated there are others that contribute to changes in locomotor activity yet to be revealed. These results provide a rare example of an olfactory stimulus that affects the interaction between the overall locomotor activity pattern and maturity state in a vertebrate species.
Footnotes
Acknowledgements
The authors thank the staffs of the U.S. Geological Survey Hammond Bay Biological Station, U.S. Fish and Wildlife Service Marquette Biological Station, and Canada Department of Fisheries and Ocean Sea Lamprey Control Station for facilities, sea lamprey, and equipment. They also thank Dr. Cory Brant for his assistance in acquiring animals, Dr. Ke Li for her assistance in measuring pheromone concentrations within washings, and Anne Scott for providing feedback on the manuscript. This work was supported by a grant from the Great Lakes Fishery Commission.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
