Abstract
Synchronization of male and female locomotor rhythmicity can play a vital role in ensuring reproductive success. Several physiological and environmental factors alter these locomotor rhythms. As sea lamprey, Petromyzon marinus, progress through their life cycle, their locomotor activity rhythm changes multiple times. The goal of this study was to elucidate the activity patterns of adult female sea lamprey during the sexual maturation process and discern the interactions of these patterns with exposure to male pheromones. During these stages, preovulated and ovulated adult females are exposed to sex pheromone compounds, which are released by spermiated males and attract ovulated females to the nest for spawning. The locomotor behavior of adult females was monitored in a natural stream with a passive integrated tag responder system as they matured, and they were exposed to a sex pheromone treatment (spermiated male washings) or a control (prespermiated male washings). Results showed that, dependent on the hour of day, male sex pheromone compounds reduce total activity (p < 0.05) and cause increases in activity during several daytime hours in preovulated and ovulated females. These results are one of the first examples of how sex pheromones modulate a locomotor rhythm in a vertebrate, and they suggest that the interaction between maturity stage and sex pheromone exposure contributes to the differential locomotor rhythms found in adult female sea lamprey. This phenomenon may contribute to the reproductive synchrony of mature adults, thus increasing reproductive success in this species.
Synchronization of internal and external environments by coordinating the proper timing of physiological and behavioral cycles is vital to enhance the survival and reproductive fitness of an organism. Diel locomotor rhythmicity is common in nature and can be influenced by several internal and external environmental stimuli; these stimuli may act simultaneously, or one stimulus may dominate and mask the effects of the others to produce a final locomotor rhythm (Aschoff, 1960). During seasonal reproduction, coordination of locomotor rhythmicity between males and females within a species becomes increasingly important to ensure reproductive success. The phenomenon of locomotor activity alterations as a response to pheromones has been documented in insects (Shorey and Gaston, 1965; Baker and Cardé, 1979; Liang and Schal, 1990), and although in fish, pheromones have been shown to produce activity responses (LaBerge and Hara, 2003), effects on overall locomotor rhythmicity are less well described.
The sea lamprey, Petromyzon marinus, has a life cycle consisting of several behaviorally different stages, offering a unique opportunity to study the factors that alter locomotor rhythmicity. Sea lamprey ammocoetes (larvae) are nocturnal, emerging at night to filter feed (Hardisty and Potter, 1971a). Ammocoetes metamorphose into a behaviorally arrhythmic juvenile stage, during which they parasitize large fish (Hardisty and Potter, 1971b). After feeding in open waters for a little longer than a year (Bergstedt and Seelye, 1995), immature (preovulated and prespermiated) sea lamprey undergo an upstream spring migration through river and stream channels to appropriate spawning grounds, during which they are nocturnal (Manion and McLain, 1971) and are guided by larval lamprey pheromone cues (Teeter, 1980; Wagner et al., 2009). Prespermiated male sea lamprey make this migration first, arriving at nests before females (Applegate, 1950). As sea lamprey mature, they become arrhythmic, beginning to build nests during the day and night (Applegate, 1950; Manion and Hanson, 1980). Another pheromone, 7α, 12α, 24-trihydroxy-5α-cholan-3-one 24-sulfate (3kPZS), is released by spermiated males and initiates searching behaviors, guiding ovulated female sea lamprey to nests (Li et al., 2002; Li et al., 2003; Siefkes et al., 2005; Johnson et al., 2006; Johnson et al., 2009). Sea lamprey are semelparous and die shortly after spawning.
Until animals are sexually mature for reproduction, remaining nocturnal could be a benefit to escape predator pressure, as has previously been suggested (Semlitsch, 1987; Keitt et al., 2005). Additionally, timing of reproduction in this species is especially important because during the migration and spawning periods, sea lamprey do not eat and rely on the storage of nutrients acquired during the parasitic phase as energy for the maturation of gonads and the act of spawning (Beamish, 1979; Madenjian et al., 2003).
Our overarching hypothesis is that synchronized locomotor activity of ovulated female and spermiated male sea lamprey may be mediated by sexual pheromones, thus increasing the chances for successful reproduction. Specifically, we hypothesize that while sexual maturation and sex pheromone exposure will each have independent effects, it is their interaction that will have an effect on the overall locomotor rhythmicity. Therefore, we predict that preovulated females will have a nocturnal rhythm, whereas ovulated females will increase their activity to the daytime hours, which likely will increase the chances for encountering spermiated males, which are arrhythmic. Furthermore, we speculate that females respond behaviorally to male sex pheromones, and exposure to these compounds acts as a signal to females that they have reached appropriate spawning grounds and mates, which will cause a decrease in locomotor activity of female sea lamprey to reserve energy for reproduction.
In this study, we aim to document the effects of sexual maturation and determine the direct effects of sex pheromone exposure on female locomotor activity and rhythms. Here, we present observations on the diel locomotor activity of preovulated and ovulated female sea lamprey in field conditions during exposure to a suite of pheromone compounds from washings of spermiated males or a control (prespermiated washings). Sexual maturation and sex pheromone exposure had independent effects on locomotor activity in adult female sea lamprey, but it was their interaction that disrupted the overall locomotor rhythms of the female sea lamprey, altering them from a nocturnal state when preovulated to an arrhythmic state once reaching maturity.
Materials and Methods
Animals
Sea lamprey were captured in mechanical traps operated by the U.S. Fish and Wildlife Service and Department of Fisheries and Oceans, Canada, in the St. Marys River, which is an outflow of Lake Superior. Standard operating procedures for transporting, maintaining, handling, anesthetizing, and euthanizing sea lamprey were approved by the Institutional Committee on Animal Use and Care of Michigan State University. Males and females were separated based upon the protocol conducted by Vladykov (1949). Maturity stages were separated based upon the protocol established by Siefkes et al. (2003). Preovulated females matured during the experiment. Ovulated females were classified as such if animals had a defined keel and eggs were expressed by manual pressure. Prespermiated and spermiated males were used for collecting washings and were separated based on spermiated males having a dorsal ridge and expressing sperm when manual pressure was applied.
Experimental Design
This study was conducted in the Little Ocqueoc River in Millersburg, Michigan, between 15 and 25 July 2008. The Little Ocqueoc River is a tributary of the Ocqueoc River, which is above the sea lamprey barrier. A portion of the river with similar cross-sectional stream flow, shady conditions, slow-moving current, and depth was selected. The river was separated into 2 channels naturally with fallen trees, and additional sandbags were added to ensure there was no cross-flow. Temperature loggers (Hobo Temperature Data Logger, Onset, Bourne, MA) were placed in the river near the cages to monitor the temperature throughout the duration of the experiment. Sea lamprey were exposed to natural lighting conditions. During this time, sunrise occurred at 0600 h and sunset at 2100 h, exposing the animal to approximately 15 h of daylight, 9 h of darkness, and 2 transitional twilight periods.
Two PVC cages 1 × 1 × 0.5 m lined with steel wire mesh were placed into each channel in a line parallel to stream flow (Fig. 1A). Each cage had a hinged lid and contained a Plexiglas rectangle covering the back half as an attachment surface as well as a refuge area from light created from a cement block placed on top of the Plexiglas. Each cage was equipped with a circular passive integrated transponder (PIT) antenna connected to a multiplexer equipped with an internal system to record signals from PIT tags (Oregon RFID, Portland, OR). The PIT antenna was placed in a line situated 0.25 m back from the front of each cage and 0.25 m below the top of the cage and tuned to ensure that the sea lamprey would not rest in a position that would continually set off the system (Fig 1B). Each time a sea lamprey passed near the antenna, the time and the uniquely identifiable tag number were recorded and used as a measure of locomotor activity. Because of the antenna placement, this system tracked a relative measure of activity, as some movements of each animal were inevitably missed. All of the animals within each cage, however, did show activity and provided large enough sample size numbers that we are confident that these behaviors are representative of this species. This measurement system allowed us to infer activity patterns through comparisons of activity between maturity states, comparisons of activity between treatment groups, and the overall rhythms of activity in each group.
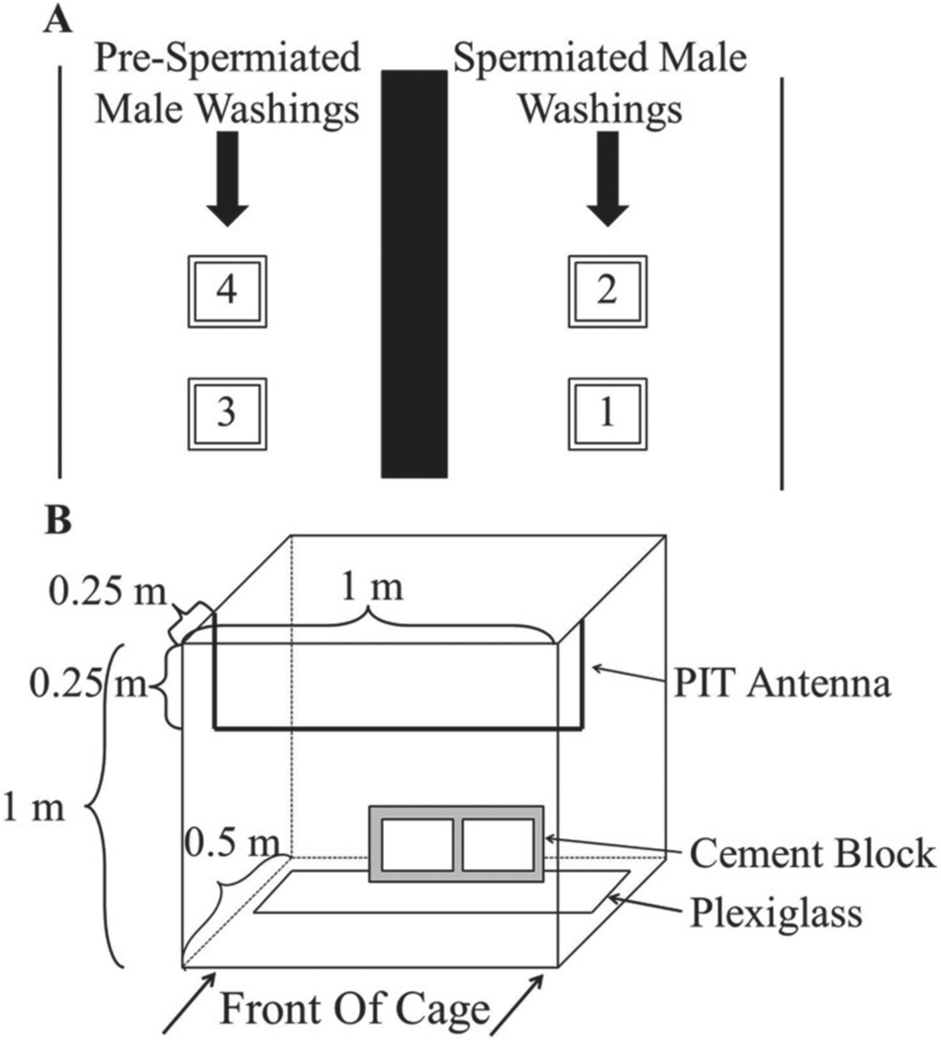
Experimental stream channel design. (A) Cages 1 and 2 were exposed to spermiated male washings. Cages 3 and 4 were exposed to prespermiated male washings. Channels were separated by a line of sandbags. Twenty preovulated females were placed in each cage and exposed to treatment for the duration of the experiment. (B) Individual experimental cage. Plexiglas and cement block were placed in the back half of the cage. Bold line denotes PIT antenna placed in top, front portion of cage. Stream current flowed from front to back of cage.
Twenty preovulated female sea lamprey equipped with a PIT tag placed within the gill slit openings were placed into each cage. A double-headed peristaltic, battery-powered pump (Master-Flex 7553-70, Cole-Parmer, Vernon Hills, IL) was used to pump either spermiated male washings (SMW) or prespermiated male washings (PSMW) into either side of the channel. Washings were collected by placing 20 spermiated or prespermiated males into a tank of 200 L of aerated water with no flow for 36 h. Water samples were collected in 1-gallon containers and frozen at temperatures below –20 °C until experimentation. Concentrations of natural 3kPZS in SMW and PSMW were quantified using ultra-high-performance liquid chromatography–tandem mass spectrometry according to the protocol described by Xi et al. (2011). The SMW contained 2.33 mg of 3kPZS/L and the PSMW had no 3kPZS, so the PSMW could be used as a control. Each pump was set at a constant rate of 30 mL/min. For cages 1 and 2, SMW was thawed and diluted with river water to create a final concentration of 10–12 M 3kPZS once mixed with stream water within the channel. The amount that the SMW was diluted was calibrated for every 24 h and dependent on the stream flow discharge, which was measured with a Marsh-McBirney flow meter (Frederick, MD) once a day. An equivalent volume of PSMW was added into the other channel as a control for cages 3 and 4.
Preovulated females were placed into each cage at the start of the experiment and checked for maturity every 2 days. Blood samples of 1 mL were collected from a selected group of half of the animals in each cage via the caudal vein every third night of the experiment starting 15 July 2008, between 0000 and 0400 h for potential future experiments. The order in which cages were selected for blood drawing was rotated every night that blood was drawn. Cages were checked twice a day and dead sea lamprey were removed. The experiment continued until all of the sea lamprey expired.
Data Analysis
A generalized linear mixed model with a Poisson distribution and a log-link function was fit to the count data of movements. Fixed effects included were treatment (SMW vs. PSMW), maturity stage (preovulated vs. ovulated), hour of the day, and the interactions of treatment by hour, maturity by hour, and maturity by treatment by hour. Fixed effects of blood drawing, blood within treatment, and cage within treatment were also added into the model to account for any background effects that may be present. Random effects included in the model were animal ID, day, animal ID by day, and hour by day, to properly account for correlations due to the double repeated measure sampling process. Proc Glimmix of SAS (Littell et al., 2006) was used to fit the model. Our goal was to test the interaction of hour by maturity, the interaction of hour by treatment, and the triple interaction of hour by maturity by treatment. We first compared levels of maturity (Fig. 2) at each hour by treatment and subsequently compared treatment at each combination of hour and maturity (Fig. 3). To clearly show the interaction of treatment and sexual maturation, we replotted the data from Figure 3 for each maturity stage treated with pheromone for comparison (Fig. 4). We performed these comparisons based on the least squares means of the linear predictor and back-transformed point estimates to the original count scale (Poisson variable) for our presentation and interpretation of results.
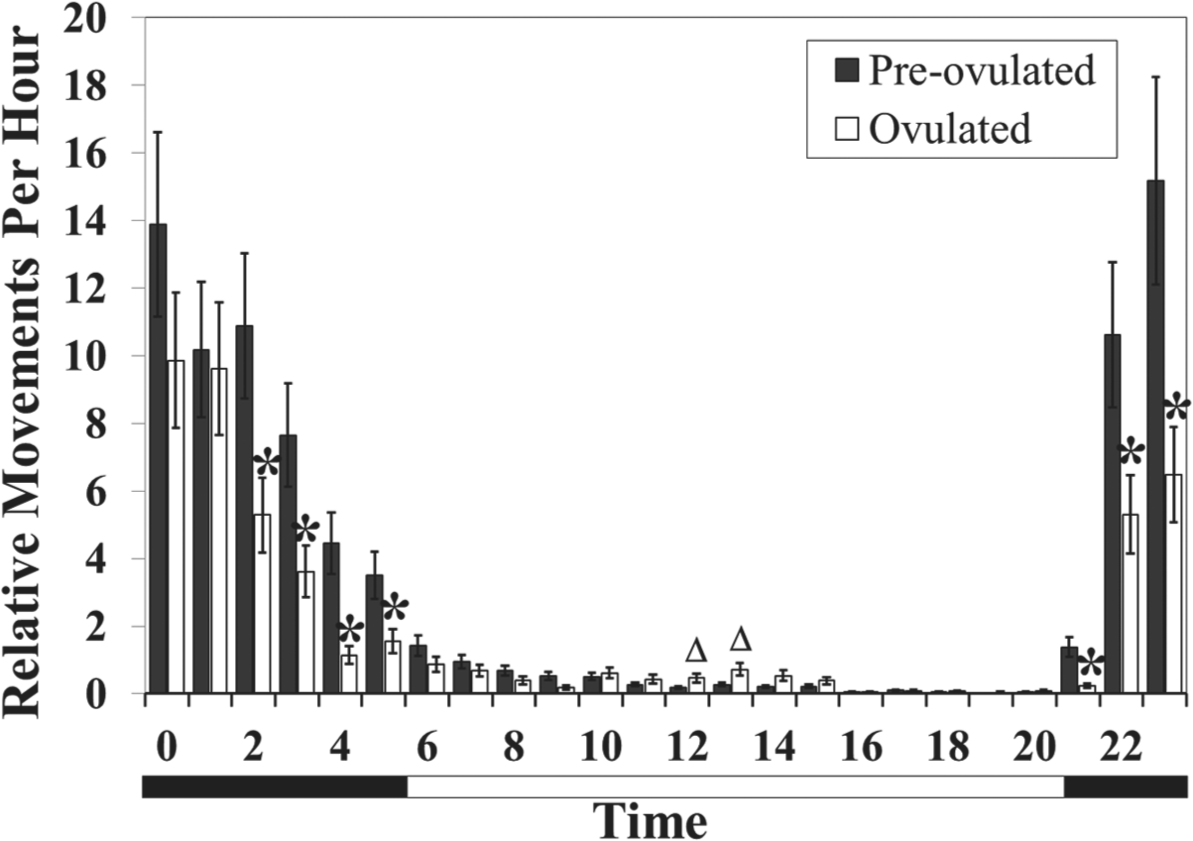
Effects of maturity on locomotor activity. Asterisks denote that activity is significantly lower in ovulated females (p < 0.05). Triangles represent the hour at which activity is significantly higher in ovulated females (p < 0.05). Vertical bars denote 1 standard error. The bar below the graph is split into black for nighttime and white for daytime.
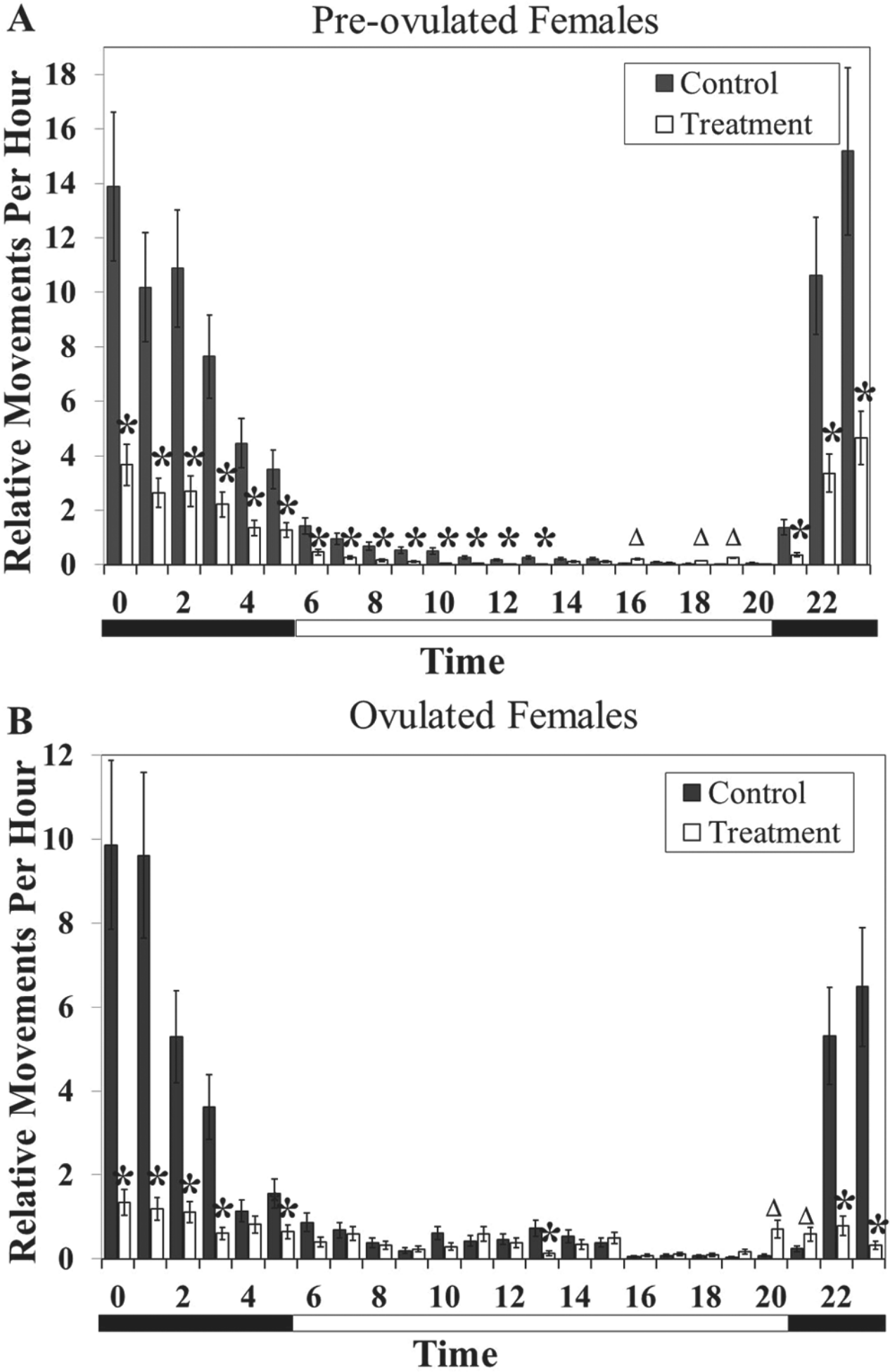
Effects of sex pheromones on locomotor activity and rhythmicity in adult females. (A) Preovulated female activity is altered in animals exposed to treatment. (B) Ovulated female rhythmicity and activity are altered in treated animals. Asterisks represent hours at which activity is significantly lower in treated females (p < 0.05). Triangles denote hours at which activity is significantly higher in treated females (p < 0.05). Vertical bars denote 1 standard error. The bar below the graph is split into black for nighttime and white for daytime.
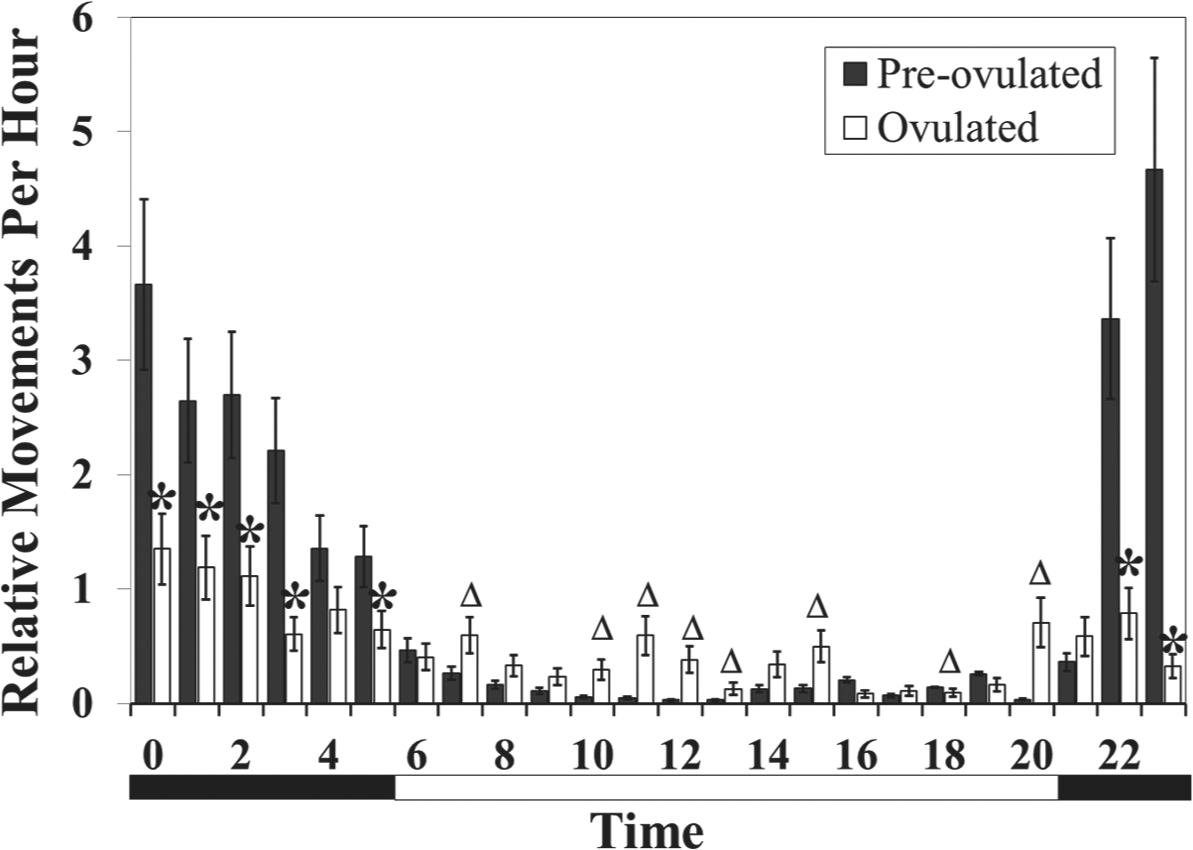
Effect of sex pheromones on rhythmicity in adult females. Preovulated females are nocturnal and ovulated females are arrhythmic when exposed to sex pheromones in sea lamprey. Asterisks denote that activity is significantly lower in ovulated females (p < 0.05). Triangles represent the hour at which activity is significantly higher in ovulated females (p < 0.05). Vertical bars denote 1 standard error. The bar below the graph is split into black for nighttime and white for daytime. Data presented in this figure are replotted from those of Figure 3.
Results
After transfer to the tanks or the cages, sea lamprey demonstrated transient bursts of activity but settled down to normal behavior after a few hours. When not moving, lamprey attached themselves to the sheets of Plexiglas provided in each cage. Temperatures ranged between 15 and 19 °C during the duration of the experiment but remained relatively stable near 17 °C.
Response to Sexual Maturation
Maturity stage affected locomotor activity, which was dependent on the hour of the day (F23,7748 = 8.67, p < 0.0001). When preovulated and ovulated females were exposed to the PSMW control, both had a nocturnal locomotor rhythm; however, ovulated animals showed a clear reduction in activity during several hours of the dark period (0200-0500 h and 2100-2300 h) as well as an increase in activity during multiple hours (1200-1300 h) of the midday period (Fig. 2). The peaks of locomotor activity in the diel profile also differed between the 2 maturity stages, with preovulated females peaking in locomotion 2 h after sunset (2300 h) and ovulated females peaking 3 to 4 h after sunset (0000-0100 h).
Response to Sex Pheromones
Locomotor activity was affected by exposure to SMW, which was dependent on hour (F23,7748 = 8.37, p < 0.0001) in preovulated as well as ovulated females (Fig. 3). Preovulated females were nocturnal whether they were exposed to the PSMW control or to SMW, and the peak of activities did not change (Fig. 3A). Exposure to SMW, however, caused a decrease in locomotion during all hours of the night (0000-0500 h and 2100-2300 h) as well as several hours of the day (0600-1300 h). Additionally, activity was increased in these animals during several hours of the daytime period (1600 h and 1800-1900 h).
Ovulated females showed not only a change in activity levels but also a change in rhythm when exposed to the SMW (Fig. 3B). The females exposed to the control were nocturnal, with a peak at 0000 to 0100 h; however, treated females showed no clear rhythmic pattern or peak in diel locomotor activity. These treated females had a reduction in activity during several hours of the night (0000-0300 h, 0500 h, 1300 h, and 2200-2300 h) as well as an increase in activity 1 hour before and after sunset (2000-2100 h), which contributed to the overall loss of rhythm.
Interaction of Maturity and Sex Pheromone
The 3-way interaction of maturity stage, sex pheromone exposure, and hour altered the locomotor rhythmicity (F24,7748 = 5.24, p < 0.0001). Preovulated and ovulated female sea lamprey exposed to SMW had differential locomotor rhythms (Fig. 4). Preovulated females had a strong nocturnal pattern, peaking 2 h after sunset (2300 h), while ovulated females had no clear locomotor rhythm or peak (hour had no significant effect on locomotor activity in ovulated females exposed to treatment, F7,88 = 0.76, p = 0.6249). Additionally, locomotor activity was reduced in ovulated females during several hours of the night (0000-0300 h, 0500 h, 2200-2300 h) and was increased during several hours of the day (0700 h, 1000-1300 h, 1500 h, 1800 h, and 2000 h), demonstrating the alteration of the locomotor rhythm between these 2 life stages.
Blood collection was not significant in affecting movement (F1,7748 = 1.21, p = 0.271), nor was there a significant blood within treatment interaction (F1,7748 = 0.8, p = 0.773). While cage within treatment was significant (F2,7748 = 5.6, p = 0.0037) and these cage differences were incorporated into the model, this additional effect did not change the results and conclusions of the effects of maturity, treatment, and hour or their interactions.
Discussion
The results presented here document that the locomotor rhythm of the adult female sea lamprey is modulated by the interaction of sexual maturation and exposure to the male sex pheromone. We have shown that each factor has an independent effect on the locomotor activity; however, it is the interaction of the 2 factors that produces the switch in rhythm from a nocturnal state shown in preovulated females to what is representative of an arrhythmic state with the addition of daytime activity exhibited in ovulated females. Conducting research in the quasi-natural environment required that activity be subsampled with PIT antennas instead of being monitored by cameras in a laboratory. The overall low activity recorded was a result of the subsampling. As each sea lamprey within each cage did show activity in our experiment, we are confident that the subsampled data, although not containing all activity, correctly characterized the locomotor activity of female sea lamprey. It is not uncommon for fishes of different maturation stages to have differential locomotor phases in the field (Emery, 1973; Johnson and Müller, 1978; Helfman, 1978, 1981; Helfman et al., 1982; Magnan and FitzGerald, 1984; Reebs et al., 1995); however, to our knowledge this is the first demonstration of sex pheromones altering a locomotor rhythm in a vertebrate species.
For many decades, changes in the locomotor rhythms across the sea lamprey life cycle have been of interest. Several factors have been investigated to determine the cause of these changes. The corneal degradation that occurs with maturation was initially hypothesized to cause a loss of vision and the onset of daytime activity (Applegate, 1950; Manion and Hanson, 1980); however, this idea was put to rest when it was shown that the diel activity rhythm of sea lamprey was not affected by blindness (Binder and McDonald, 2007). Later, it was discovered that the dominant photosensory organ involved in circadian locomotor control in lamprey species is the light-sensitive pineal gland (Tamotsu and Morita, 1986). The locomotor rhythm is lost in the river lamprey, Lampetra japonica, when the pineal gland is removed, implicating its role in rhythmicity control (Morita et al., 1992). The pineal gland can detect the day length photosensory changes (Morita and Dodt, 1971; Tamotsu and Morita, 1986) across the spawning season, and it was hypothesized that these alterations in the environment were responsible for the addition of daytime activity. However, in a study in which river lamprey were kept in cold water under natural lighting conditions, the onset of daytime activity in addition to sexual maturation was delayed (Sjoberg, 1977), suggesting that day length is not the key to controlling the locomotor rhythm. This is consistent with our presented results in which the locomotor rhythm was altered even though the day length remained consistent over each experimental period.
More recently, temperature has been implicated in the control of locomotor rhythmicity. Binder and McDonald (2007) demonstrated that preovulated adult female sea lamprey exposed to low temperatures (7 °C) or high temperatures (20 °C) had a reduction in their nocturnal locomotor activity peak, and an addition of activity during the daytime hours was observed at high temperatures. Additionally, the investigators showed that rapid increases in temperature of 7 or 8 °C over a 4-h period caused a brief increase in daytime activity. These results suggest that temperature is an important factor in modulating locomotor activity; however, sexual maturation was not examined, as only preovulated females were used, and these results cannot be extrapolated to the change in locomotor rhythmicity demonstrated across adult life stages. Furthermore, in our study the temperature was consistent between a range of 15 to 19 °C, with little change from the first to the final days of the experiment. The alterations in locomotor activity and rhythms that we have demonstrated provide evidence to suggest that other factors are contributing to these changes. It is possible that locomotor rhythms are only overt in an optimal temperature range. Sea lamprey migration upstream begins at approximately 10 °C, and the intensity of the migration varies with temperature (Applegate, 1950). Once within this thermal range, however, it is likely that sexual maturation and exposure to sex pheromones dominate the effects of temperature and, subsequently, are the determining factors of the locomotor rhythm. As a mostly nocturnal rhythm was still present in ovulated females exposed to a control, it is possible that sex pheromones are masking the effects of temperature and sexual maturation, contributing to the arrhythmic state that is displayed. Evidence has suggested in mammals that olfactory stimuli can alter circadian rhythms and that these 2 systems are linked functionally (Honrado and Mrosovsky, 1991; Fluxman and Haim, 1993; Haim and Rozenfeld, 1993; Goel and Lee, 1995). Therefore, an alternative explanation is that olfactory stimuli may influence circadian rhythms by modulating the activity of the circadian clock in the pineal gland through stimulation of structures that innervate this gland directly.
One possible rationale for the changes seen in locomotion as sea lamprey mature is that once sea lamprey reach spawning maturity, intraspecific communication becomes essential for the coordination of reproduction. The sex pheromone released by spermiated males may act as a signal to females that they are in appropriate spawning nest territory and to reduce energy expenditure on locomotion to promote energy for reproduction or gonadal development. In this species, only a finite amount of energy and, therefore, a narrow time range exist for reproduction due to the cessation of eating after the juvenile stage (Beamish, 1979; Madenjian et al., 2003). Furthermore, sea lamprey only spawn once in their life. Due to the limited amount of energy and time available for reproduction, the reproductive benefits that come from a reduction in overall activity as well as an extension of activity to daytime hours outweigh the diurnal predation risks. Our results are consistent with previous studies in which adult fishes become arrhythmic during the spawning phase (see Reebs, 2002, for review).
Communication is also important to coordinate locomotor activities between mature male and female sea lamprey. Spermiated males are arrhythmic (Applegate, 1950), and as females mature, they may switch from a nocturnal to an arrhythmic state to optimally match the locomotor activity of the males. The sex pheromone cue may be the key to facilitate this synchronization of behavior, improving reproductive fitness through an increased chance of a spawning event to occur. This idea is consistent with previous studies in diverse fish species exhibiting that chemical signals participate in the synchronization between males and females by affecting both behavior and physiology (Partridge et al., 1976; Liley, 1982; Stacey and Sorenson, 1991; Sorenson, 1992; Olsen and Liley, 1993).
The idea that male sex pheromone compounds can alter female sea lamprey locomotor activity is not surprising. The male sex pheromone component 3kPZS alters behaviors in ovulated female sea lamprey (Siefkes et al. 2005, Johnson et al. 2009). Ovulated females exposed to 3kPZS show robust upstream movements, and when this compound was applied to a stream at a concentration greater than that of SMW, females were drawn to the source of 3kPZS (Johnson et al., 2009). Additionally, exposing ovulated females to SMW, which consists of a suite of pheromone compounds, retained them on the nests (Johnson et al., 2009). 3kPZS has also been shown to have effects on differential systems in male and female sea lamprey. In male sea lamprey, it has been shown that 3kPZS has a greater effect on the endocrine system (Chung-Davidson et al., 2013; Siefkes et al., 2005), whereas in females it has a greater effect on the locomotor system (Li et al., 2002; Johnson et al., 2009). In our study, the measured change between the treatment and control was the presence of 3kPZS. Due to this and previous evidence, we hypothesize that this sex pheromone may be the main component of spermiated male pheromone compounds affecting locomotor rhythmicity; however, further studies using targeting 3kPZS are needed to confirm this.
Overall, this study has provided evidence that sexual maturation and exposure to sex pheromones are contributing factors to locomotor activity rhythms in adult female sea lamprey. Pheromones have been implicated in locomotor rhythm changes in insects and changes in amounts of activity in other fish species, but this is one of the first examples of the effects of sex pheromones on the locomotor rhythm in a vertebrate species. Moreover, we have demonstrated that these key factors have independent effects that cause reductions in activity and an extension of activity into the daytime but that it is their interaction that produces a shift in the locomotor rhythm. Future work should determine whether the pheromones are having direct effects on the circadian clock system within the pineal gland and other circadian or locomotor control portions of the nervous system, are exerting masking effects, or both.
Footnotes
Acknowledgements
The authors thank the staffs of U.S. Geological Survey Hammond Bay Biological Station and U.S. Fish and Wildlife Service Marquette Biological Station for facilities, sea lamprey, and equipment. Thanks to Cory Brant, Nicole Griewahn, David Partyka, Aaron Smuda, Trevor O’Meara, and Henry Thompson for field assistance. Additional thanks to Henry Thompson for technological assistance in sorting behavioral data. They thank Dr. Xiaodan Xi for her assistance in measuring pheromone concentrations within washings, as well as Dr. Yu-Wen Chung-Davidson for thoughtful comments during the development of this manuscript. This work was supported by grants from the Great Lakes Fishery Commission and NIGMS grant number: 1R24GM083982 to W.M.L. This article is contribution number 1754 of the U.S. Geological Survey Great Lakes Science Center. Use of trademark names does not represent endorsement by the U.S. Government.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
