Abstract
Circadian clocks are inherent to most organisms, including cryptozoic animals that seldom encounter direct light, and regulate their daily activity cycles. A conserved suite of clock genes underpins these rhythms. In this study, we explore the circadian behaviors of the red flour beetle Tribolium castaneum, a significant pest impacting stored grain globally. We report on how daily light and temperature cues synchronize distinct activity patterns in these beetles, characterized by reduced morning activity and increased evening activity, anticipating the respective environmental transitions. Although less robust, rhythmicity in locomotor activity is maintained in constant dark and constant light conditions. Notably, we observed more robust rhythmic behaviors in males than females with individual variation exceeding those previously reported for other insect species. RNA interference targeting the Clock gene weakened locomotor activity rhythms. Our findings demonstrate the existence of a circadian clock and of clock-controlled behaviors in T. castaneum. Furthermore, they highlight substantial individual differences in circadian activity, laying the groundwork for future research on the relevance of individual variation in circadian rhythms in an ecological and evolutionary context.
Keywords
Virtually all lifeforms from bacteria to humans express daily patterns in their physiological and behavioral processes, known as circadian rhythms, which enable them to accommodate the recurring changes in their environment (Saunders, 2002). These rhythms are not only reactions to the environment but are endogenously controlled by molecular pacemaker systems called circadian clocks, which can be entrained to the external periodic cues such as light and temperature, collectively known as zeitgebers (Bell-Pedersen et al., 2005). Coordinating activities in anticipation to the periodic environmental changes has obvious adaptive functions such as escaping adverse environmental conditions, ensuring proper availability of food and mates, synchronizing behaviors with conspecifics, and reducing competition or predation risks (Favreau et al., 2009; Vaze and Sharma, 2013). Apart from these extrinsic advantages, a circadian clock confers an intrinsic advantage of maintaining a stable and optimal temporal compartmentalization of biological processes regardless of any irregular fluctuations in the environment. Even animals living in aperiodic conditions are known to have a functioning molecular clock (Trajano and Menna-Barreto, 1996) which points toward its intrinsic adaptive values.
Circadian clocks operate similarly across different taxa. Their cellular mechanism is comprised of transcription-translation feedback loops of clock genes and their protein products, capable of producing self-sustained circadian oscillations (Doleela, 2023; Helfrich-Förster, 2004). Detailed knowledge about the molecular mechanisms of animal clocks is available mainly from the studies in mouse and fruit fly as representatives of mammals and insects, respectively. In the main circadian feedback loop of fruit flies, the clock proteins PERIOD (PER) and TIMELESS (TIM) represent the transcriptional repressors, whereas in mammals, CRYPTOCHOROMES (CRY1 and CRY2) dimerize with the PERIOD proteins (mPER1, mPER2, and mPER3) to perform the equivalent role. This transcriptional inhibition occurs by blocking of their transcriptional activators CLOCK and CYCLE (BMAL1 is the mouse homolog of CYCLE). A striking difference between the mammalian and Drosophila clocks lies in the mechanism by which light resets and synchronizes the clock with the environment. Light induces TIM degradation in flies, whereas it leads to the transcriptional activation of the Period genes in mice (Helfrich-Förster, 2004).
Insect clocks are best documented in Drosophila. However, given the enormous diversity of insect species and their habitats, it is plausible that their molecular clock mechanisms have diversified in response to their specific environmental conditions (Kotwica-Rolinska et al., 2022; Tomioka and Matsumoto, 2015; Doležel, 2023). Diversities in some clock components are already known, for instance, certain insects like beetles and honeybees possess the mammalian light-insensitive cryptochrome, CRY2, unlike Drosophila, which possesses only the photosensitive CRY1. On the other hand, mosquitoes and monarch butterflies are known to possess both CRY1 and CRY2 (Yuan et al., 2007). This underscores the significance of investigating a broad range of insect models for gaining a comprehensive understanding of various aspects of the circadian clock.
Tribolium castaneum, the red flour beetle, is a widely found pest of stored grains and an important model organism of increasing relevance in many fields such as classical genetics, population genetics, insect development, and epigenetics (Campbell et al., 2022). These flour beetles are cryptozoic, meaning that they are animals living mostly in darkness (Brown et al., 2009). They have very little exposure to light in their supposed ancestral or current habitat behind tree barks or grain stores, respectively (Good, 1936; Sokoloff, 1974). Their cryptozoic adaptations include loss of photoreceptors sensitive to blue light (Jackowska et al., 2007) and an increase in odorant and gustatory receptors (Richards et al., 2008). Even though the adults are normally known for being negatively phototactic (Park, 1934), they crawl to flour surfaces from time to time where they can get direct light exposure as observed in laboratory populations (Misra and Englert, 1985). Copulation usually occurs on the flour surface as well (Park, 1934). They are also likely to experience temperature variations in their natural habitats. Despite spending most of their life in the absence of light, we know that they still possess the genes of a functional molecular clock (Li et al., 2018). However, whether T. castaneum indeed displays rhythmic activity profiles, driven by a functional circadian clock, has not been convincingly demonstrated.
T. castaneum has homologs for all clock genes, known in D. melanogaster (Richards et al., 2008). First studies on the molecular clock in T. castaneum revealed that Clock (Clk), cycle (cyc), period (per), and timeless (tim) are all rhythmically expressed (Li et al., 2018). Instead of the Drosophila-type cryptochrome, CRY1, these beetles possess the light-insensitive CRY2, the function of which remains unclear. It has been suggested that CRY2 plays a role in inhibiting CLK and CYC, thus acting as a negative element of the feedback loop as known in other insects such as Hymenoptera and Heteroptera (Kotwica-Rolinska et al., 2022; Yuan et al., 2007). However, T. castaneum also possesses the Drosophila-type TIM (Kotwica-Rolinska et al., 2022) along with the mammalian CRY2, making it a unique model to study insect clocks, also possibly in an evolutionary context of transition from the Drosophila-type to mammalian-type clocks.
In insects, functional circadian clocks and the rhythms they produce are traditionally evaluated by measuring an individual’s variation in locomotor activity during the course of the day (Chiu et al., 2010). It has been shown that T. castaneum exhibits rhythmic locomotor activity in light-dark cycles (LD; Rath et al., 2021), as well as in the absence of external cues in constant darkness (DD; Abe et al., 2021). However, both studies report only average data of individually kept beetles, without determining the overall rhythmicity (i.e., how many beetles behaved rhythmically in a certain condition), nor showing individual actograms, which would allow to estimate the individual variation within the population. It is important to investigate these behavioral variations as an organism reacts to and modifies its environment through its behavior (Duckworth, 2009), and therefore, having sufficient behavioral variation can readily help populations to adapt faster to changing environments (Bolnick et al., 2003). Furthermore, to our knowledge, there are also no studies exploring whether the circadian clock in these beetles can be synchronized by temperature fluctuations.
In the present study, we monitored the locomotor activity patterns of T. castaneum to characterize in detail the circadian profiles in their movements. We studied light as well as temperature entrainment and the effect of constant conditions on an individual level to characterize the behavioral variation existing within the population. We also performed RNAi treatment to study the changes in activity patterns upon knockdown of the Clock gene (Clk) encoding the key circadian transcription factor CLK. As T. castaneum is an important model organism and also a pest, knowledge about its biological clock and how it influences behavior and physiology is important. It can serve as an exceptional model system for investigating the molecular clock mechanisms of Coleoptera, where our current understanding is comparatively limited and thereby also advancing our overall comprehension of insect clocks.
Materials and Methods
Beetle Cultures
For this study, the Croatia 1 (Cro1) strain, collected in Croatia in 2010, and a commonly used lab-adapted strain San Bernardino (SB), from San Bernardino, California, USA, established by Sokoloff in 1960s were used (Milutinović et al., 2013). The populations were kept at standard laboratory conditions of 30 °C, 70% humidity, and a 12 h:12 h light:dark cycle (lights on at 08:00 and lights off at 20:00; light from flourescent bulbs, 4000 K, cool white, 3350 lumen) in plastic boxes or glass jars with foam stoppers to allow air circulation. The standard beetle diet consisted of heat-sterilized (75 °C for at least 24 h) organic wheat flour (Bio Weizenmehl Type 550, dm—Drogerie Market) mixed with 5% brewer’s yeast powder (referred to as “flour” from now on).
Locomotor Activity
To monitor locomotor activity patterns, we used the Drosophila Activity Monitoring System (DAM5 H-4; Trikinetics Inc., Waltham, MA, USA; referred to as “DAM5 system” in the following). Individual beetle’s movements interrupt one of four infrared light beams intersecting the glass tubes housing single beetles, at equal distances from each other (Rosato and Kyriacou, 2006; Suppl. Fig. S1), which enables the recording of an individual’s movements. Sexually matured virgin beetles of known sex were placed into an individual glass tube (diameter: 5 mm, length: 65 mm) with a flour disk, 40 µl of flour/water solution (0.15 g/ml) dried overnight, as food inside on one end and sealed with cotton plugs on both ends. Thirty-two glass tubes were assembled into each DAM5 monitor, which were placed in environmentally controlled incubators (Percival) for the recording of activity. The standard rearing conditions were maintained in the incubators throughout the period of recording unless stated otherwise. Before the first day of recording, beetles were accustomed for at least 6 hours in the activity monitors.
Circadian Activity Patterns in LD Cycles Followed by Constant Darkness
For the general characterization of the circadian activity patterns in 12:12 LD and subsequent DD, we used the Cro1 population (the beetles used belong to the handling control of a knockdown experiment). After sex discrimination, we individualized the pupae in wells of a 96-well flat-bottom microtiter plate (Type F, Sarstedt) containing 0.08 g of flour. Thirty-two adults per sex, which were at least 2-week-old post eclosion, were used for monitoring locomotor activity for an initial 7 days during LD at 30 °C (normal rearing temperature) at a light intensity of 20 lux followed by 7 days of DD.
Circadian Activity Patterns in Constant Light After Light Entrainment (LD)
To study the effect of constant light (LL) on the circadian activity patterns, we monitored beetles of the Cro1 population in LL after entrainment to LD, at two different light intensities: 20 lux and 100 lux. Around 2 weeks post eclosion, we used 32 adults per sex, which were individualized since sex discrimination at pupal stage, for each light intensity. We recorded their activity for an initial 7 days in LD, followed by 6 days in LL.
Comparison of Activity Profiles Between Two Laboratory Populations
For comparing the circadian activity patterns of the two laboratory populations Cro1 and SB, we separated the pupae according to sex and kept them in groups of 24 individuals in petri dishes (∅10 cm, Waldeck) with ad libitum flour. For each population, 24 adults per sex which were at least 11 days post eclosion (SB population develops slower than Cro1) were used. Activity recordings were carried out in LD (20 lux) for 7 days, followed by another 7 days in DD. During LD, a technical issue caused the temperature and humidity to drop from 30 °C to 25 °C and from 70% to 50%, respectively, for 8 hours (from midnight until lights on at 8 am).
Circadian Activity Patterns During and After Temperature Cycles
To test if daily temperature cycles can entrain the locomotor activity patterns in the absence of light, we recorded the beetles’ behavior in oscillating temperatures as the zeitgeber. Beetles from the Cro1 population were reared on flour-disks prepared in 96-well plates from day 15 post oviposition onwards. During this time, they were not exposed to light until 21 days post oviposition (as they also served as a control group in a separate experiment, which is not described here). Around 2 weeks post eclosion, beetles were sexed, and 24 adults per sex were used for the activity recording for an initial 7 days of temperature cycling (12 h at 25 °C/12 h at 30 °C), followed by another 7 days of constant 30 °C in DD. Activity recordings were carried out in complete darkness in both thermal phases to make sure that the regularly fluctuating temperature is the only zeitgeber. The experiment was repeated a second time with the same conditions (n = 48 per sex during temperature entrainment, and n = 32 per sex during constant temperature, sample size reduced due to recording error) except that the temperature change was set to ramp for 2 hours to create a more natural condition of temperature variations.
Activity Profiles After Knockdown of Clk
To test, if rhythmic behavior depends on the canonical molecular clock mechanism, we performed RNAi-knockdown (Tomoyasu et al., 2008) of the key circadian clock gene Clk at the pupal stage using the Cro1 population.
After sex discrimination, pupae were glued with their last abdominal segment onto a glass slide for the dsRNA injection. The pupae were injected between the second and third lowest segments of their abdomen with dsRNA of either Clk or jellyfish eGFP, which served as the control for the dsRNA injection treatment (Schulz et al., 2022). The dsRNA construct of Enhanced Green Fluorescent Protein (eGFP) was produced in our lab (via cloning followed by PCR and in vitro transcription using the T7 MEGAscript Kit [Ambion by Life Technologies GmbH, Darmstadt, Germany]). The dsRNA construct for Clk was obtained from EupheriaBiotech (Dresden, Germany) and has previously been used in the ibeetle RNAi screen (Schmitt-Engel et al., 2015; Suppl. Table S1). A third treatment group served as the pure handling control (non-injected control) where the pupae glued on to the glass slides did not receive any injection. dsRNA was injected at a concentration of 1000 ng/μL dissolved in phosphate-buffered saline (PBS) using a microliter injector (FemtoJet; Eppendorf AG, Hamburg, Germany) and borosilicate glass capillaries (100 mm length, 1.0 mm outside diameter, 0.021 mm wall thickness; Hilgenberg GmbH, Malsfeld, Germany). Upon eclosion, the beetles were kept individually in 96-well plates with 0.08 g of flour until the start of the activity measurements in the DAM5 system.
The activity measurements started 2 weeks after eclosion. The activity of beetles from all three groups were monitored in LD (light intensity of 100 lux) for 7 days, followed by 7 days in DD.
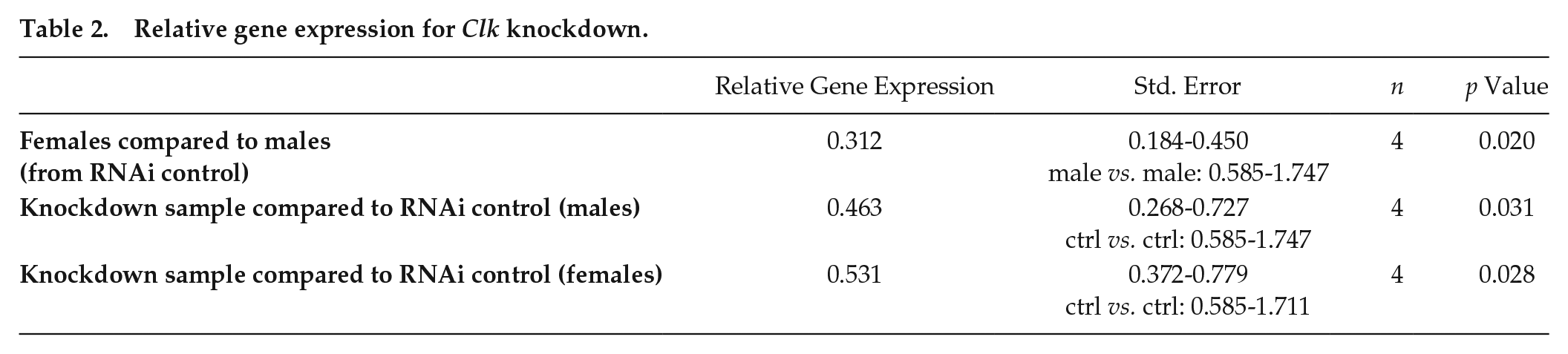
To confirm the successful downregulation following the RNAi treatment, we measured Clk expression by qPCR. Fourteen days after eclosion, we shock froze the individuals in liquid nitrogen. Furthermore, we usually pooled 3-4 whole bodies per replicate. All samples were stored at −80 °C until the RNA extraction.
We extracted total RNA by combining a TRIZOL-chloroform approach with a spin column kit (SV Total RNA Isolation System; Promega [Peuß et al., 2016]). In short, we ground the TRIZOL-submerged samples with a sterile pestle until no larger tissue parts were visible. Samples then went into an ultrasound bath to further ensure disintegration of tissue. After 10 min of incubation, samples were spun down, and the supernatant was mixed with chloroform. Another centrifugation step ensured formation of phase separation. We then transferred the upper aqueous phase mixed with a lysis buffer onto the spin column and proceeded according to the manufacturer’s protocol. Finally, RNA was eluted with 80 µl of nuclease-free water and stored at −80 °C until further use.
For the qPCR, we first reverse transcribed RNA to cDNA using the RevertAid First Strand cDNA Synthesis Kit (Thermo Scientific) and the provided oligo(dT)18 primer, following the manufacturer’s instructions. Before the cDNA was used in the qPCR, it was diluted 1:5 with nuclease-free water.
We performed qPCRs with technical duplicates and two housekeeping genes rp49 and RpL13 a to normalize target gene expression. Amplification efficiency of primers was previously established (Suppl. Table S2), as well as the uniform expression of the housekeeping genes across sexes and treatments. The qPCR ran on a LightCycler 480 System (Roche) with KAPA SYBR Fast mastermix (Sigma-Aldrich).
For each RNAi treatment and control, we measured gene expression separately for males and females in 4 biological replicates. We included samples in the downstream analysis if the standard deviation between the technical replicates remained below 0.5 and the melting curves showed only one clear peak.
Behavior Analyses
After 2 weeks of recording, we extracted the raw data of locomotor activity in activity bins of 30 min using DAMFileScan software (TriKinetics). We looked for activity patterns from the average histograms during different entrainment and constant conditions, differences in average activity levels, and proportion of rhythmic beetles using data from individual beetles.
For the average comparison of diel patterns in movement, average histograms of mean activity in 30-min intervals for all individuals belonging to the same treatment and sex group across the days of the same environmental condition were generated using the Fly-toolbox (Levine et al., 2002b).
Individual beetles were scored to be rhythmic or arrhythmic based on a combination of qualitative and quantitative measures using double-plotted actograms, autocorrelation (Levine et al., 2002b), and χ2-periodograms (Sokolove and Bushell, 1978) generated using Fly-toolbox in MATLAB (MATLAB and Statistics Toolbox Release, 2021b). Double-plotted actograms were used for the visual analysis to identify rhythmic patterns. Each row in a double-plotted actogram displays the activity on 2 consecutive days for which the activity of the second day is re-plotted in the left part of the next row to inspect for the activity patterns horizontally and vertically. A beetle is categorized as rhythmic if activity patterns are repeated for at least half of the days of activity recording. To reduce the subjectivity from the visual detection of rhythmicity, quantitative thresholds from autocorrelation and periodograms were also used in combination with the visual inspection of actograms (Levine et al., 2002b). Autocorrelation graphs identify patterns in the data by examining the correlation between the movement signal and a time-shifted version of itself. This analysis helps detect both the strength of the rhythm (RS-value) and the duration of the identified period (p). The χ2-periodogram assesses rhythmicity by testing various potential periods within the data and evaluating their fit, represented by the Qp value. The dominant signal period is determined based on the highest Qp value, while a 95% confidence interval is used to evaluate the significance of the chosen period. In addition, the h-value indicates to which extent the Qp value of the selected period exceeds this confidence interval. A beetle was categorized as rhythmic only if it met all three criteria: having a clear rhythmic pattern in the actogram, RS > 1 in the autocorrelation and h > 10 in the periodogram.
The circular phase plots showing the phase of individual beetles (Levine et al., 2002b) were also plotted using MATLAB. For the analysis of activity levels, activity in 1-min intervals were averaged over all monitoring days of a specific condition for individual beetles. The bar graphs, plotting proportion of rhythmic beetles and boxplots for the average activity were generated using ggplot2 (Wickham, 2016) in RStudio version 2021.09.1 (RStudio Team, 2021) under R version 4.1.2 (R Core Team, 2021). We did some final editing of supplementary figure panels using Inkscape (Developers, Inkscape Website, 2020).
Statistics
All the statistical analysis for the rhythmicity and average activity levels were performed using R Studio employing the additional package glmmTMB (Brooks et al., 2017).
The analysis for rhythmicity was done on binary data indicating either presence or absence of consistent rhythmic patterns for individual beetles. The analysis for the activity levels was done on the activity in 1-min bins averaged over all the days of a condition for individual beetles. The differences in the proportion of rhythmic beetles and activity levels were analyzed in generalized linear mixed-effect models (GLMMs; Suppl. Table S3) with the corresponding error distributions. The final models were derived after p-value–based stepwise model simplification. Mean phase values of rhythmic beetles during LD and DD were compared using paired t-test on individual phase values obtained from circular phase plots.
For the statistical analysis of gene expression after RNAi treatment, we used the Relative Expression Software Tool (REST2009; Pfaffl et al., 2002). The tool is designed to compare the gene expression between two groups—in our case, expression in the knockdown treatment was compared to the expression in the RNAi control group. REST employs a mathematical model, which takes the PCR efficiencies of the primers into account when calculating expression ratios from the mean crossing point deviation. It then performs a randomization test to determine statistically significant differences between the treatment groups.
Results
To foster our understanding of the workings of the molecular clock in flour beetles and to further characterize the circadian patterns in their behavior, we first monitored and analyzed the locomotor activity rhythms of an outbred population (Cro1) of T. castaneum. We tested the beetles’ behavior in different light and temperature cycles for both sexes. In addition, we compared the outbred strain (Cro1) to an inbred strain (SB), which has adapted to laboratory conditions for decades, to infer strain-specific behavioral differences. Finally, we downregulated Clk gene expression via RNAi to investigate the role of this key circadian clock gene in generating the behavioral patterns.
Locomotor Activity Patterns in LD Cycles
We monitored the locomotor activity patterns of beetles from the Cro1 population in LD (20 lux) conditions followed by DD. The average activity profiles of beetles showed a clear and repeatable diurnal rhythm (Figure 1a and 1d and Suppl. Fig. S2). The daily average activity patterns were characterized by a consistent reduction in activity levels anticipating the onset of morning light in all the experiments, which we refer to as “morning dip” (Figure 1a). The average value of the morning anticipation index, MAI = total activity between ZT 21.5 and ZT 24 (window of morning dip)/total activity between ZT14 and ZT16.5 (a different 2.5-h window during night), calculated from last 3 days in LD, was 0.83 ± 0.53 for males and 0.67 ± 0.74 for females. They reacted with a startle response of increased activity observed in response to the onset of the morning light stimulus (Figure 1a). This response fades quickly within 30 min and is not observed in DD conditions. Upon more natural-like conditions with a linear ramping, extending the lights-on transition to 2 h, the startle response either disappeared or appeared at the end of the ramping period, but as a lower peak than the non-ramping conditions (Suppl. Fig. S3). Furthermore, beetles gradually increased their activity levels throughout the late afternoon, a behavior which we accordingly (and in analogy to a similar pattern observed in D. melanogaster (Hamblen-Coyle et al., 1992)) call evening anticipation (Figure 1a). The average value of the evening anticipation index, EVI = total activity between ZT 9.5 and ZT 12 (2.5-h window during evening anticipation)/total activity between ZT 4 and ZT 6.5 (a different 2.5-h window during day), calculated from last 3 days in LD, was 7.48 ± 14.70 for males and 1.90 ± 1.59 for females. With the start of the dark phase in LD, the beetles slightly reduced their activity levels from their highest peak during the evening anticipation but remained highly active throughout the night until they reduced their activity considerably during the morning dip (Figure 1a and 1c).
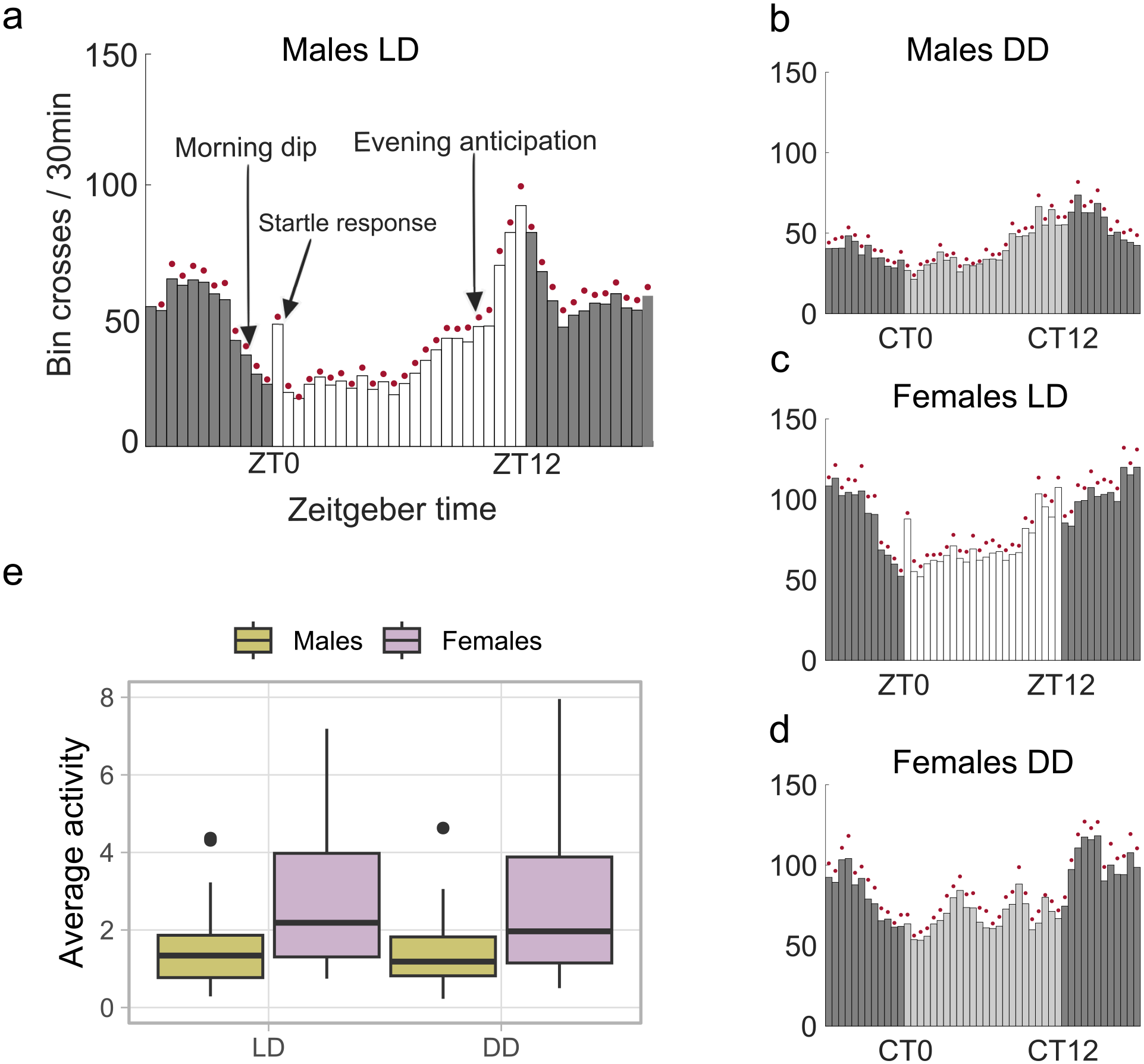
Locomotor activity patterns of T. castaneum in LD and DD. (a-d) Average histograms showing mean activity (bin crosses/30 min) of Cro1 beetles during 7 days in LD (L = 20 lux) and DD. The red dot above each bar denotes the standard error of mean (SEM); (e) boxplots showing average activity per min for individual beetles during 7 days in LD and DD (males: n = 31, females: n = 32). Outliers are marked with black dots.
Locomotor Activity Patterns in DD
In DD, the beetles’ average activity profiles largely mirrored those observed in LD, except for the obvious absence of the startle response. Before the start of the subjective morning, both males and females showed a clear reduction of activity similar to the morning dip in LD (average MAI calculated from first 3 days in DD: males = 0.78 ± 0.75, females = 0.83 ± 0.73). However, the gradual increase of activity throughout the subjective late afternoon that resembled the evening anticipation was clearly observed only in males (Figure 1b and 1d, average EAI calculated from first 3 days in DD: males = 11.03 ± 41.21, females = 2.13 ± 3.65). Moreover, the beetles that continued their rhythmic behavior in DD following LD exhibited the same activity peak phase in both conditions, indicating stable entrainment of their circadian clock (Suppl. Fig. S4). In addition to the generally higher levels of activity observed in females than in males, the average histograms also illustrate the irregular and noisy patterns exhibited by the females (Figure 1a-1d). The analysis of individual average activity in 1-min bins also reveals a significantly lower average activity for males (GLMM, estimate = −0.588, p < 0.001, Figure 1e). In terms of individual rhythmicity, 52% of males and 22% of females showed rhythmic patterns in LD, whereas the proportion of rhythmic beetles was similar (32% for males and 28% for females) in DD conditions (Table 1). However, upon statistical analysis of individual rhythmicity, there was no significant effect for the sex (GLMM, estimate = 0.338, p = 0.665) or the light condition (GLMM, estimate = −0.491, p = 0.489), whereas there was a weak trend for the interaction between the effects of sex and light condition (GLMM, estimate = 1.727, p = 0.089, Suppl. Table S4).
Proportion of rhythmic beetles in different experiments.
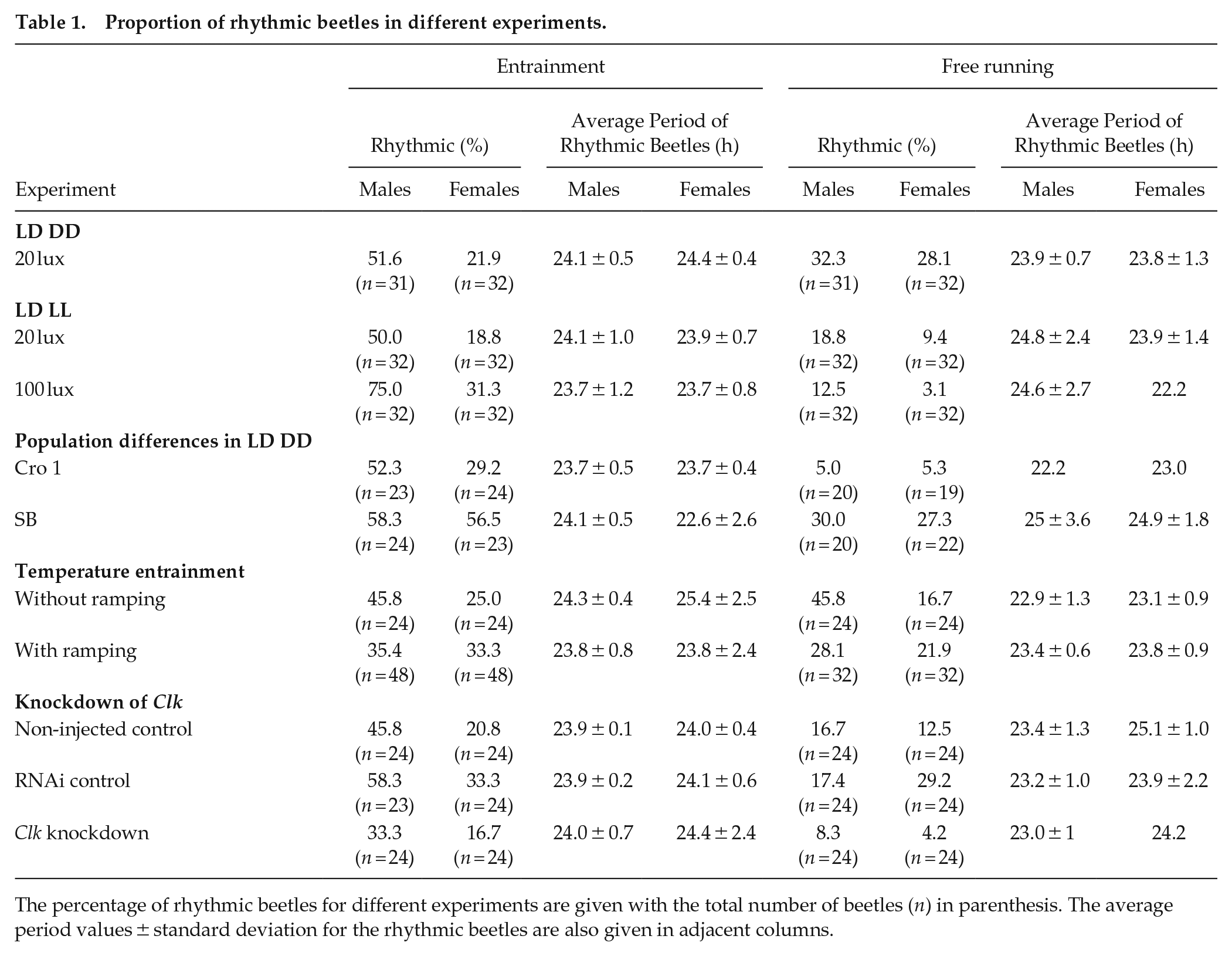
The percentage of rhythmic beetles for different experiments are given with the total number of beetles (n) in parenthesis. The average period values ± standard deviation for the rhythmic beetles are also given in adjacent columns.
Variation in Locomotor Activity Patterns
A substantial amount of individual variation can be seen in the rhythmic patterns in LD (Figure 2a-2f). Among the rhythmic beetles, not all show identical patterns. Activity peaks of many beetles start just before the lights turn off, and among these, some have their peak of activity for a short duration before the lights turn off (Figure 2c) or extending further throughout the dark phase of the LD (Figure 2a). The individual variation in phase of their activity can also be seen from the circular phase plot (Figure 2g). There was variation in terms of whether the beetles could consistently maintain the rhythmicity throughout both light conditions as well (Figure 3a-3d). Not all beetles that were rhythmic in LD were able to maintain their rhythmicity during DD. Nine out of 31 males and 2 out of 32 females were rhythmic in LD but showed arrhythmic behavior in DD. There was a small proportion of beetles (3 out of 31 for males and 4 out of 32 for females) that showed rhythmic behavior only in DD (Figure 3e).
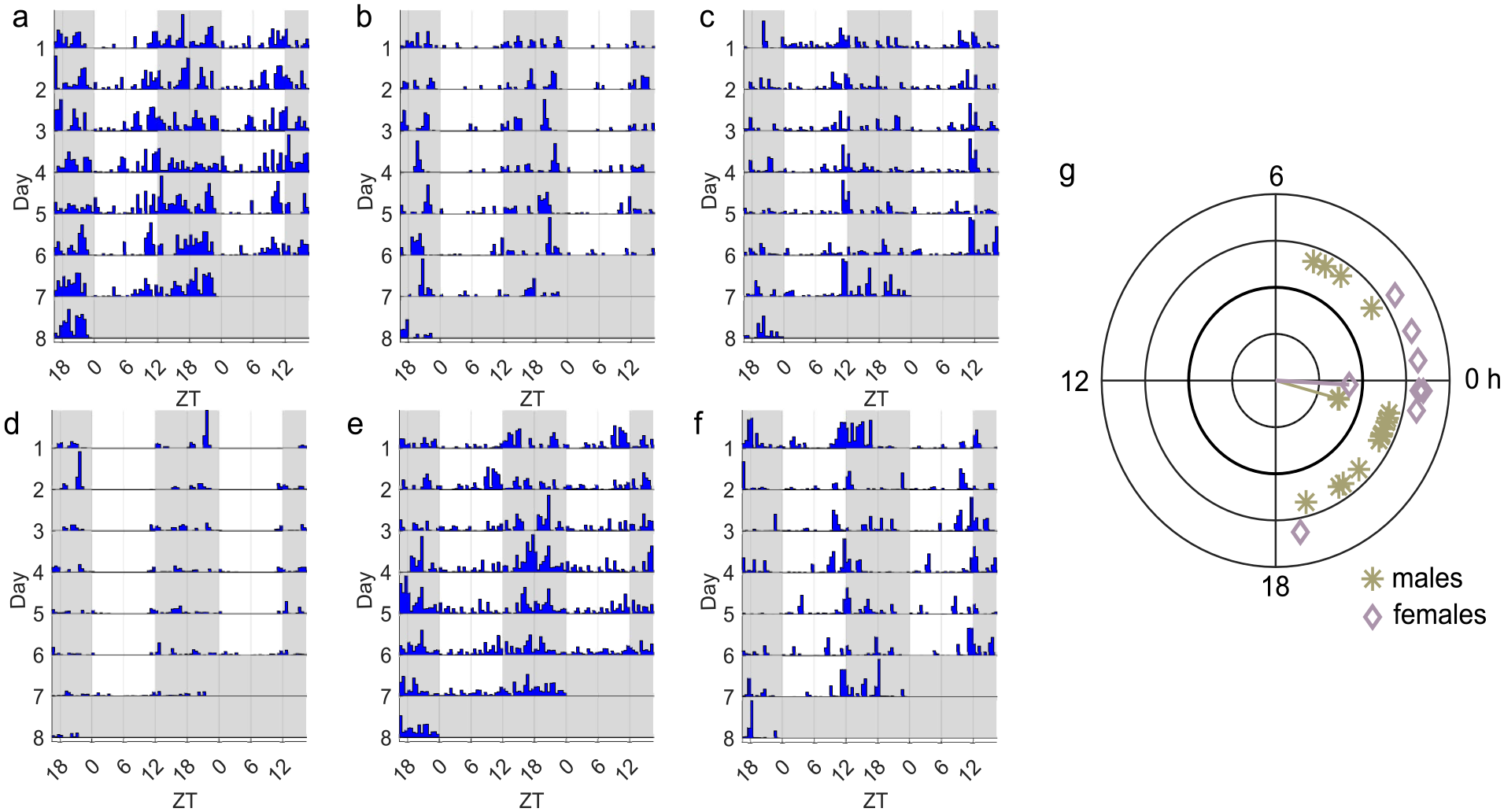
Variation among individuals. (a-f) Individual actograms of selected beetles showing variation in behavioral patterns during LD. (g) Circular phase plot showing activity phases of rhythmic beetles in LD based on data from the first 3 days. Average activity peak phase is indicated by the direction of the vector, while the vector length reflects phase coherence within each group (Levine et al., 2002a).
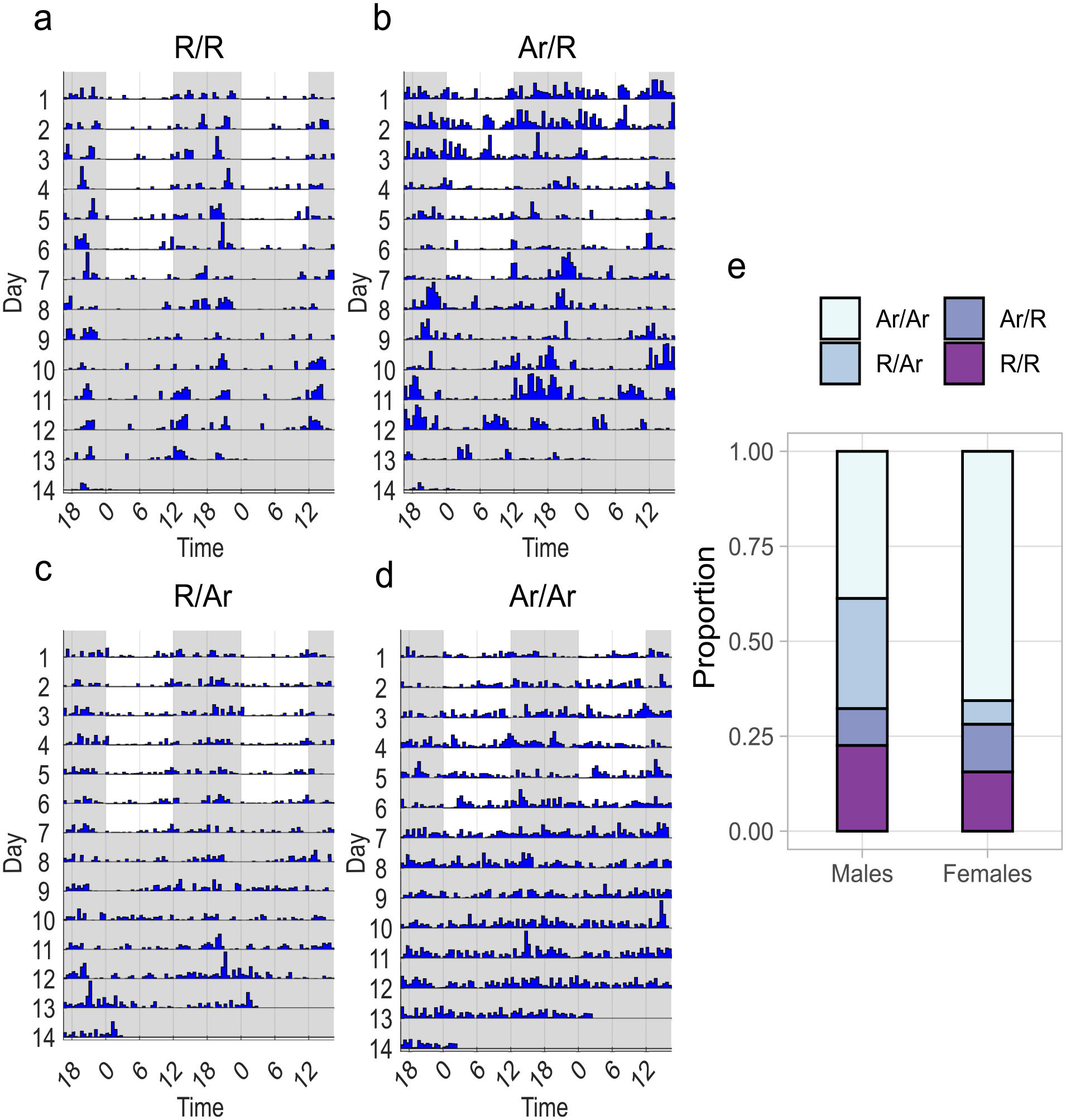
Maintenance of rhythmicity in LD and DD. (a-d) Individual actograms showing different categories of beetles according to their status of being rhythmic (R) or arrhythmic (Ar) during LD (first 7 days) and DD (last 7 days) (R/R—rhythmic in LD and DD, R/Ar—rhythmic in LD but not in DD, Ar/R—rhythmic in DD but not in LD, Ar/Ar—arrhythmic in both LD and DD). (e) Stacked bar plot showing the proportion of these categories separately for males and females.
Locomotor Activity Patterns in LL Conditions
When LD was followed by LL conditions, the average activity profiles of beetles resembled those observed in DD; however, overall rhythmicity compared to DD was reduced in both light intensities tested (20 lux and 100 lux; Figures 4 and Suppl. Fig. S5, respectively, Table 1). When examining individual rhythmicity, males showed a significantly higher proportion of rhythmic individuals than females (GLMM, estimate = 1.711, p < 0.001, Figure 4e, Suppl. Table S4). Furthermore, the proportion of rhythmic beetles experienced a significant decrease in LL conditions compared to LD (GLMM, estimate = −3.193, p < 0.001, Figure 4e, Suppl. Table S4). There was an overall significant effect of light intensity on the rhythmicity (GLMM, estimate = −1.002, p = 0.03, Figure 4e, Suppl. Table S4). There was a significant interaction between light intensity and the light condition, with a sharper drop in rhythmicity from LD to LL for the higher light intensity (GLMM, estimate = 1.772, p = 0.026, Figure 4e, Suppl. Table S4).
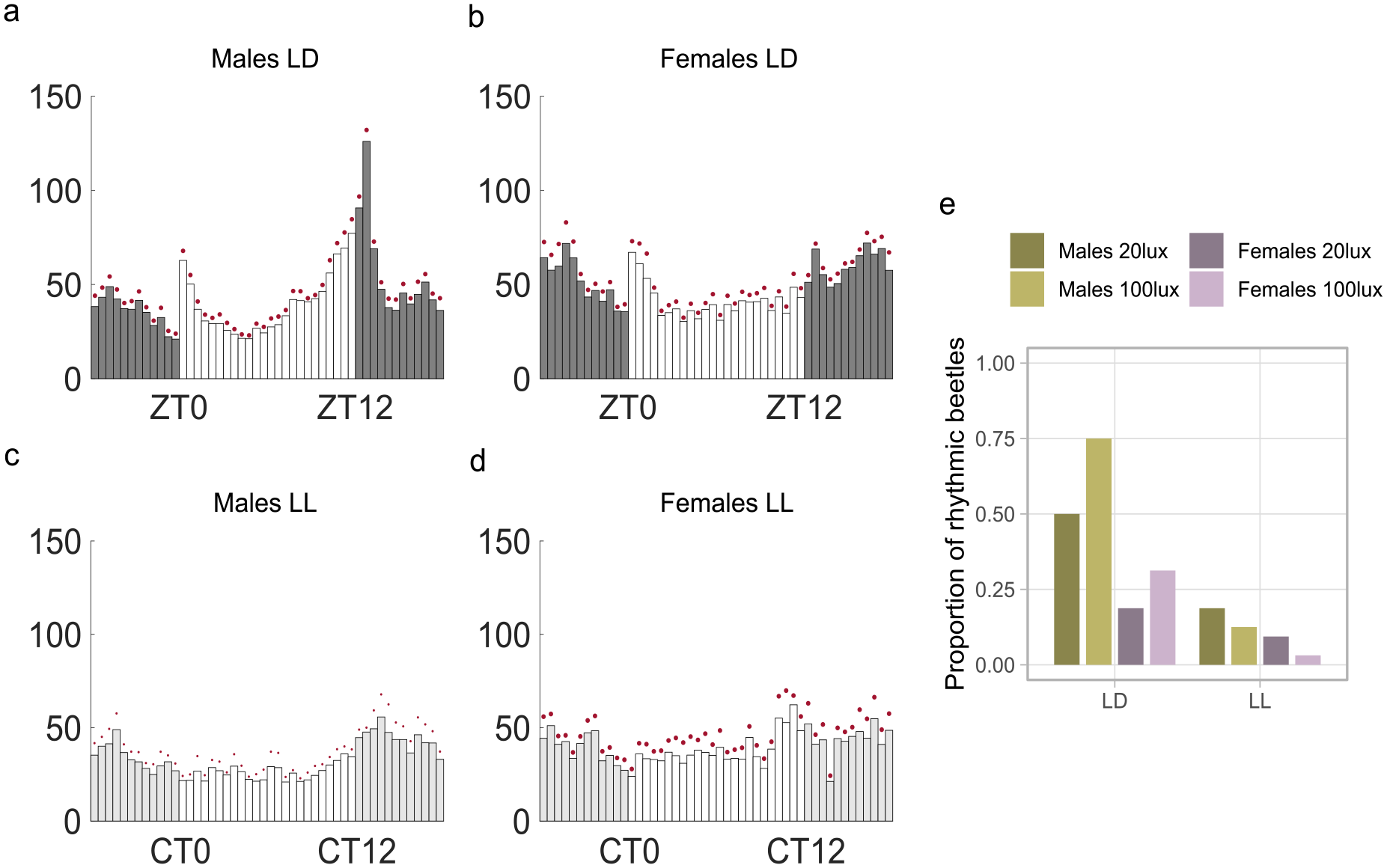
Rhythmicity in LL conditions. (a-d) Average histograms showing mean activity for all beetles during 7 days in LD and LL (L = 20 lux). For average histograms with L = 100 lux, see Supplementary Figure S5. (e) Proportion of rhythmic beetles in LD and LL for the two light intensities (20 lux and 100 lux) (males: n = 32 per light intensity, females: n = 32 per light intensity).
Population Differences in Locomotor Activity and Behavioral Patterns
Both Cro1 and SB populations had similar behavioral patterns (Figure 5a-5h), but the average activity was significantly lower for the SB population during LD (GLMM, estimate = −0.667, p < 0.001, Figure 5j) and DD (GLMM, estimate = −0.368, p = 0.04, Figure 5k, Suppl. Table S4). The average activity of females was significantly higher than that of males in LD (GLMM, estimate = −0.624, p < 0.001, Figure 5j, Suppl. Table S4) but not in DD (GLMM, estimate = −0.118, p = 0.512, Figure 5k, Suppl. Table S4). While examining individual rhythmicity, the SB population showed a significantly higher proportion of rhythmic beetles (GLMM, estimate = 1.116, p = 0.006, Figure 5i, Suppl. Table S4). The effect of light condition was also significant with a higher proportion of beetles observed in LD (GLMM, estimate = 1.761, p < 0.001, Figure 5i, Suppl. Table S4). However, there was no significant effect of sex on rhythmicity (GLMM, estimate = 0.413, p = 0.279, Figure 5i, Suppl. Table S4).
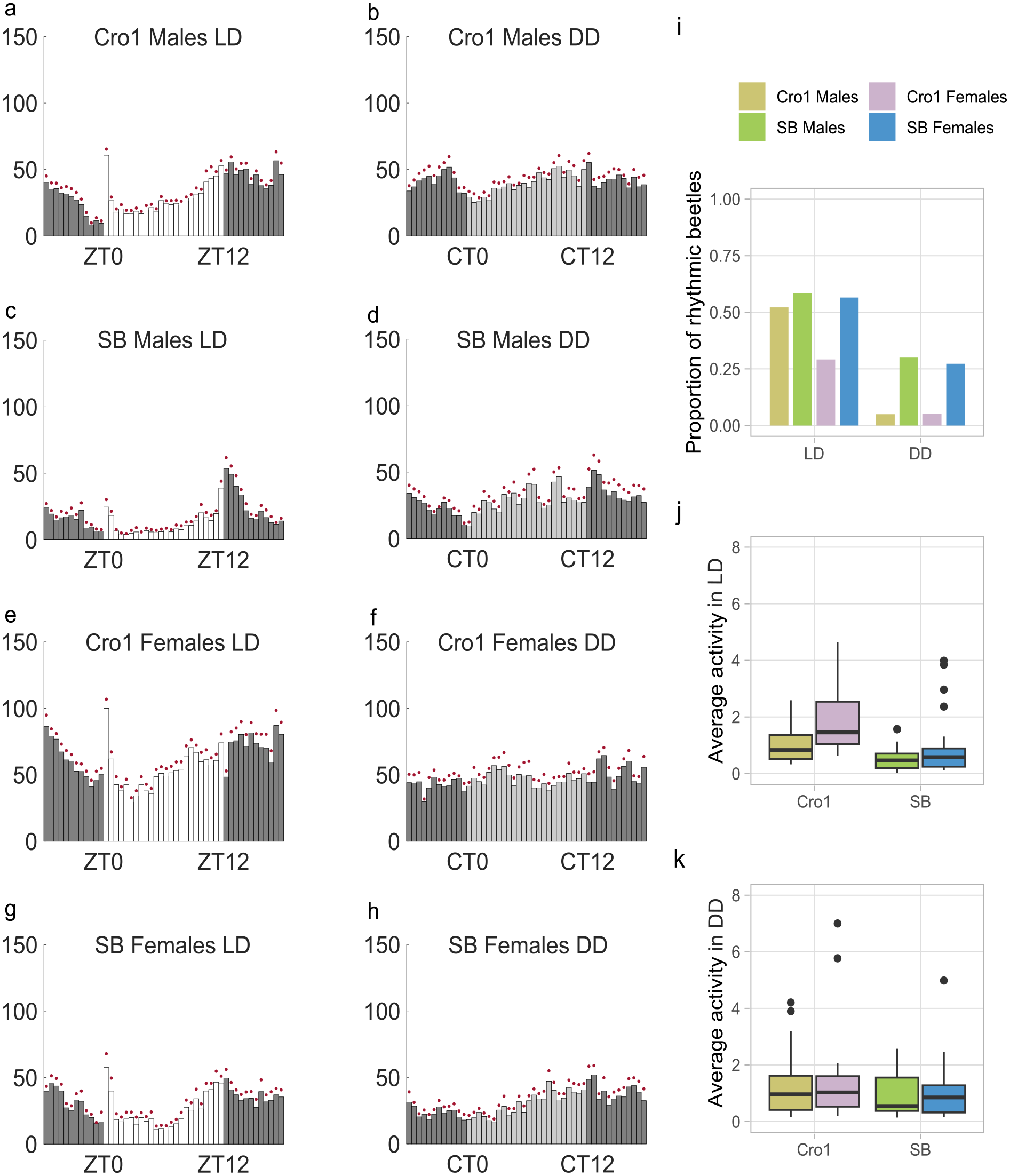
Population differences in rhythmicity. (a-h) Average histograms showing mean activity for all beetles during 7 days in LD (L = 20 lux) and DD for Cro1 and SB populations. (i) Proportion of rhythmic beetles in LD (Cro1 males: n = 23, Cro1 females: n = 24, SB males: n = 24, SB females: n = 23) and DD (Cro1 males: n = 20, Cro1 females: n = 19, SB males: n = 20, SB females: n = 22). (j and k) Boxplots showing activity in 1-min bins for individual beetles averaged over 7 days in LD (j, Cro1 males: n = 23, Cro1 females: n = 24, SB males: n = 24, SB females: n = 23) and DD (k, Cro1 males: n = 22, Cro1 females: n = 20, SB males: n = 21, SB females: n = 22).
Locomotor Activity Patterns During and After Temperature Cycles
Similar to the behavior in LD, the beetles showed activity rhythms in a 12 h:12 h rectangular temperature cycle of 25 C:30 C in DD (Figure 6a and 6e). However, rhythmic behavioral patterns, apart from the prominent peaks of activity in response to the sudden temperature change in the morning and evening, were less prominent than LD. These peaks of activity at the temperature transitions resembled the startle response observed after lights-on in LD and were not retained in constant temperature conditions (Figure 6a, 6b, 6e, and 6f).
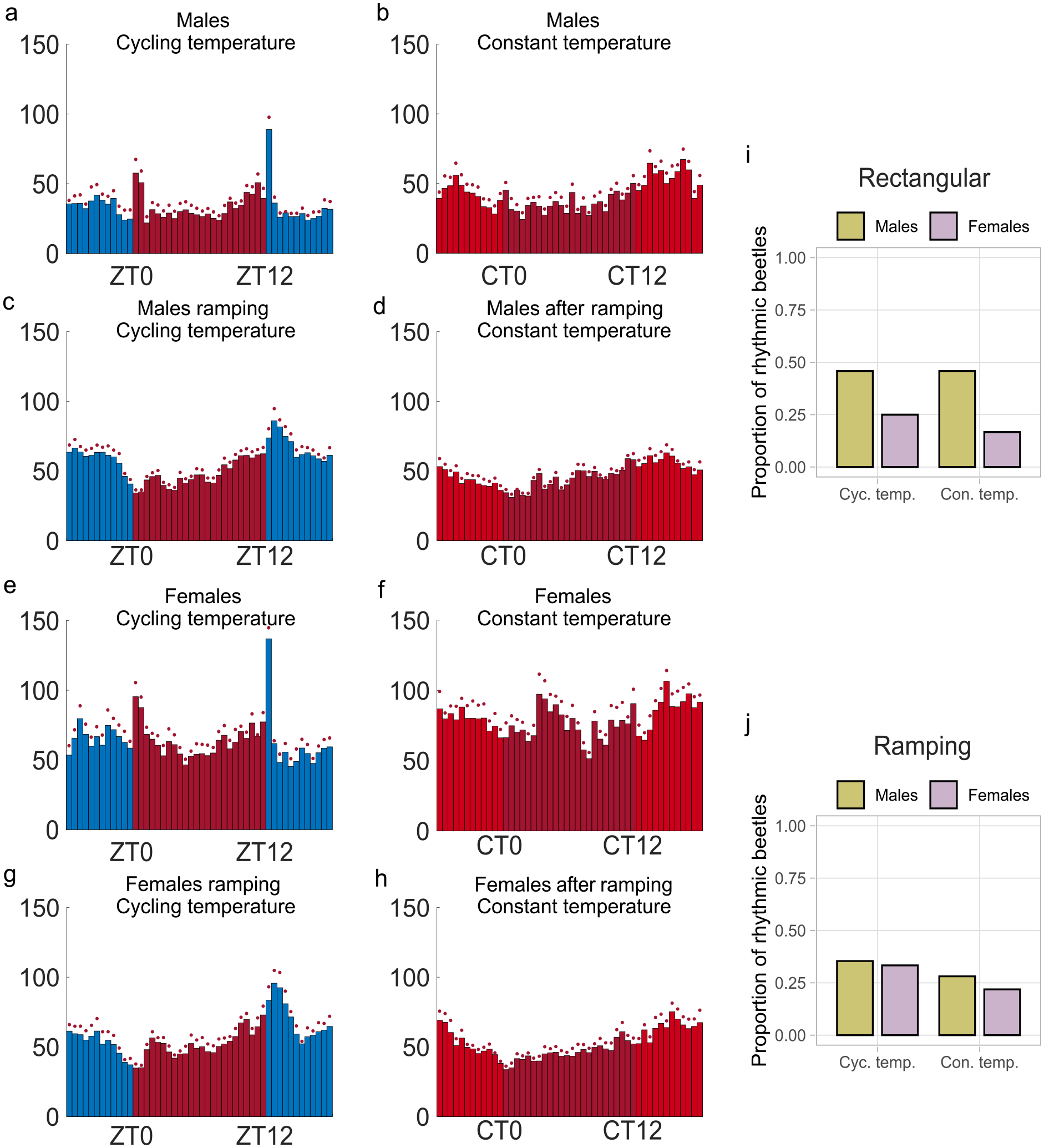
Temperature entrainment. (a-h) Average histograms showing mean activity for all beetles during 7 days of rectangular (a, e) or ramped (c, g) 25 °C: 30 °C temperature cycles and a subsequent constant temperature (30 °C) (b, d, f, h), in DD. (i, j) Proportion of rhythmic beetles during rectangular (i) and ramped (j) temperature cycles and subsequent constant temperatures (rectangular: n = 24 per temperature condition and sex, ramping: n = 48 in temperature cycling and n = 32 in constant temperature for both sexes).
After the temperature cycles, only males showed a clear rhythmic activity pattern in constant temperature conditions (Figure 6b). The proportion of rhythmic beetles significantly differed between sexes (GLMM, estimate = 1.418, p = 0.018, Figure 6i, Suppl. Table S4).
Upon the more natural-like temperature cycling in DD with a ramping of temperature change within 2 hours, the startle responses disappeared (Figure 6c and 6g). Similar as in LD cycles, there was a clear dip in the morning activity as well as evening anticipation visible during the ramped temperature cycles (Figure 6c and 6g). In the subsequent constant temperature conditions, both males and females retained these activity patterns (Figure 6d and 6 h). There was no difference in the proportion of rhythmic beetles between the sexes (GLMM, estimate = 0.185, p = 0.634, Figure 6j, Suppl. Table S4), as well as between temperature cycles and constant temperature conditions (GLMM, estimate = −0.427, p = 0.283, Figure 6j, Suppl. Table S4). Overall, these results show that T. castaneum can entrain their circadian clock and locomotor activity to both ramped and rectangular temperature cycles.
Locomotor Activity Patterns Upon Knockdown of Clk
We downregulated the key circadian clock gene Clk using RNAi in the pupal stage to study the subsequent changes in activity profiles.
There was a significant reduction in Clk gene expression compared to the RNAi control, indicating that the dsRNA-mediated knockdown was efficient (Table 2). Clk knockdown was correlated with a significant reduction in rhythmicity (GLMM, estimate = −1.206, p = 0.006, Suppl. Table S4) in comparison to the RNAi control beetles (gfp-RNAi and non-injected control), indicating that Clk function is important for behavioral rhythmicity (Figure 7). There was a significant interaction effect of sex and light condition as well on the rhythmicity (GLMM, estimate = 1.362, p = 0.04, Figure 7, Suppl. Table S4). The Clk gene expression was also significantly different between sexes in the RNAi control (Table 2).
Relative gene expression for Clk knockdown.
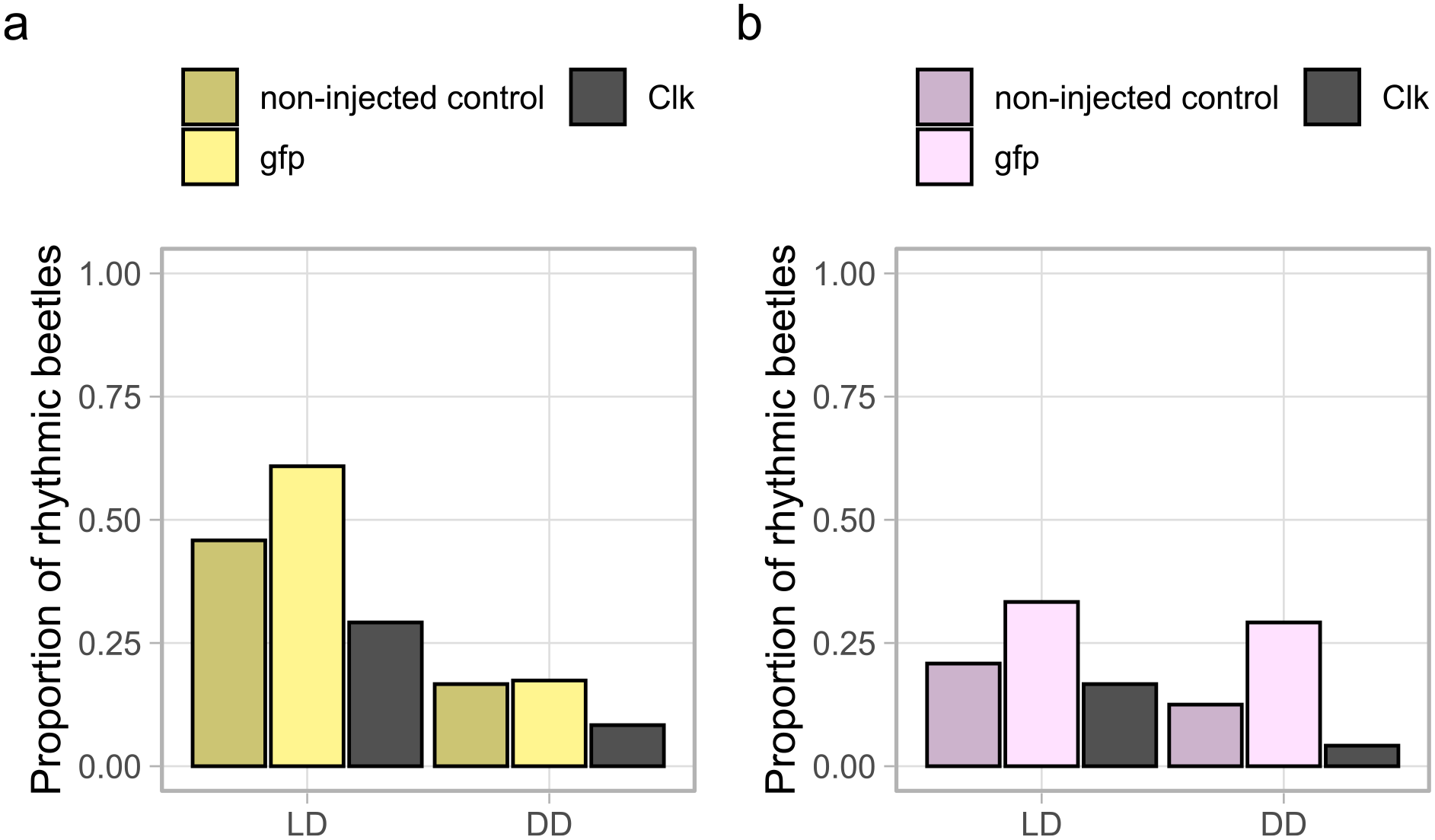
Rhythmicity upon knockdown of Clk. Proportion of rhythmic individuals in adults following the knockdown treatment at pupal stage for Clk in males (a) and females (b), LD: n = 24 per sex and treatment groups, DD: n = 24 per sex and treatment groups except for RNAi control males (n = 23).
Discussion
We used the DAM system to perform a detailed characterization of the average as well as individual locomotor activity profiles of adult T. castaneum, extending its application to study behavioral patterns in another important model organism. We recorded the activity of lab-adapted populations with high temporal resolution and described the activity patterns under light entrainment, constant dark conditions, and for the first time to our knowledge, under thermal oscillations and LL conditions. The beetles anticipated environmental transitions and, even though less robust, maintained their rhythmicity in DD and LL. We showed that a substantial amount of individual variation exists in the diel patterns of this cryptozoic species. The knockdown of Clk significantly reduced rhythmicity, indicating the existence of a circadian clock in T. castaneum and its control of behaviors.
The finding that temperature cycling acts as a zeitgeber for the beetle clock, in addition to light, is consistent with our expectations, considering that beetles are exposed to cycling temperatures in their human-associated (synanthropic) habitats of grain silos (Ziegler et al., 2021), as well as in their supposed natural habitats under tree bark (Good, 1936; Sokoloff, 1974). Studies in many other insects including fruit flies (Glaser and Stanewsky, 2005) and honeybees (Giannoni- Guzmán et al., 2021) have documented the role of temperature oscillations as zeitgebers, in addition to LD cycles. The notable patterns of the average activity profiles under both zeitgebers included a consistent drop of activity just before the change of conditions in the morning, which we refer to as “morning dip,” and the gradual increase of activity throughout the late afternoon, known as evening anticipation. These patterns emerged even before the zeitgeber conditions change and were maintained in constant conditions, indicating an anticipatory response to the predicted changes resulting from an entrained molecular clock. Other insects such as D. melanogaster (Dubowy and Sehgal, 2017) and Plutella xylostella (D. Wang et al., 2021) also show such gradual increase in activity anticipating the transitions of environmental conditions. However, a gradual decrease of activity (morning dip) toward the time of dark-light transition is rather atypical.
The beetles reacted with a startle response of heightened activity to the light turning on in the morning, as well as to the change of temperature in the subjective morning and evening. The absence of these startle responses in constant conditions as well as their disappearance or strong reduction under more natural-like conditions of ramping temperature or light conditions confirm that they are not mediated by the circadian clock but are instead reactions to sudden changes in environmental conditions. A similar startle response immediately following the dark-light transition is commonly observed, for instance, in Aedes aegypti, and consistent with our study, these responses were not present in DD (Gentile et al., 2009).
Average histograms consistently exhibited evening anticipation in constant conditions as well as during the entrainment phase for both light and temperature as zeitgebers. A steep evening anticipation was characteristic of the average activity profiles under light entrainment, especially for males. The evening anticipation aligns well with the time of day when mating rates of T. castaneum reach their maximum (Z. Wang et al., 2024). In red flour beetles, males initiate the copulatory behavior and must mate multiple times to ensure fertilization success, especially because of the last male sperm precedence (Fedina and Lewis, 2008). An increased activity of males in search of mates could be a potential reason why males possess a strong and clear evening anticipation peak compared to the females. Similarly, in P. xylostella, males were shown to have better rhythmic features than females in DD in low-temperature conditions (D. Wang et al., 2021). Such sex-specific differences in activity profiles are commonly observed in many insect species and are often associated with their mating behavior. For example, in Anopheles gambiae, males have an earlier flight activity peak than females, consistent with the fact that males aggregate to form swarms to which females subsequently enter (Rund et al., 2012). In addition to the difference in evening anticipation, the overall activity levels of females were significantly higher than those of males, as was also reported recently (Benita et al., 2024). In addition, males generally exhibited a higher proportion of rhythmicity, which may be attributed again to the necessity for males to initiate copulation behavior, thereby requiring precise timing of such behaviors.
In addition to the individual variation in phase, there was substantial variation in the individual activity patterns including differences in the duration of peak activity, levels of activity at different times, and the ability to maintain rhythmic behavior in constant conditions following entrainment. A study in the cave beetle species, Laemostenus latialis, also showed similar variability in individual behavioral patterns (Pasquali and Sbordoni, 2014). Another study observed significant diversities in circadian behaviors in social insect species compared to the non-social fruit flies (Giannoni-Guzmán et al., 2014). Since T. castaneum is a non-social yet group-living insect, one could speculate that the dual role of the pheromone, 4,8-dimethyldecanal (DMD) as an aggregation and a sex attractant for females (Levinson and Levinson, 1995), enables individuals to rely on their conspecifics for timing their own mating rather than a strict reliance on their circadian clock. Another possibility is that variability in their peak activity timings could help in avoiding any possible male-male competition. The activity patterns were similar between the Cro1 strain and the long-term laboratory-adapted SB strain. However, the SB population showed significantly higher rhythmicity than Cro1, probably attributed to the inbred nature of SB.
The proportion of rhythmic beetles considerably decreased in DD conditions. Nevertheless, the presence of all clock gene homologs known in Drosophila (Richards et al., 2008) indicates that T. castaneum possesses a functional circadian clock. Therefore, the decrease in rhythmicity in DD compared to that under light entrainment could possibly indicate a weak link or uncoupling between the endogenous clock and the locomotor behavior in DD (Abhilash et al., 2017), as seen in the case of blind mole rats (Beale et al., 2016) and in Drosophila species inhabiting higher latitudes with weakly rhythmic environments (Bertolini et al., 2019). The interindividual variability seen in locomotor activity profiles could also possibly be the result of a weak behavioral regulation by the endogenous clock (Beale et al., 2016). One could speculate that such a weak coupling or even uncoupling might be due to the absence or reduced selective pressure for circadian functions in their possible ancestral natural dark habitats such as rotting logs and tree barks, as well as in their current, also largely dark habitat of grain silos (Good, 1936; Sokoloff, 1974). The possible chance of relying on olfactory cues from conspecifics for mating initiation might have also resulted in a weak selection pressure for the evolution of a strong endogenous clock or a strong coupling of the clock with behavior.
There was a significant decrease in the proportion of rhythmic beetles in LL conditions, more pronounced in higher light intensity, even though rhythmic patterns were observable in the average histograms. In wild-type D. melanogaster, strong LL leads to an arrhythmic behavior (Konopka et al., 1989; Matsumoto et al., 1994) because of the light-dependent TIM degradation by CRY1 which conveys photic signals to the circadian clock (Ceriani et al., 1999; Emery et al., 2000). The beetles are not expected to have such an arrhythmic behavior as they possess only the light-insensitive cryptochrome (CRY 2; Yuan et al., 2007). However, most likely, retinal and/or potentially also unknown extraretinal photoreceptors play a role (Helfrich-Förster, 2020) in photic entrainment, which could then possibly affect the rhythmicity in such LL conditions. A similar reduction of rhythmicity in LL conditions was reported for Nasonia vitripennis, which also possesses only the light-insensitive CRY 2 (Bertossa et al., 2014), whereas some other closely related Nasonia species retained their rhythmicity (Bertossa et al., 2013). A study in linden bug, which also possesses only CRY 2, revealed a deteriorating effect of LL on rhythmicity even though around 40% of individuals showed strong rhythmicity in such LL conditions (Kaniewska et al., 2020).
To test if the observed behavioral rhythms depend on a molecular circadian clock, we attempted the knockdown of the key circadian clock gene Clk in the pupal stage and subsequently monitored locomotor activity profiles at the adult stage. Indeed, rhythmicity in Clk dsRNAi animals was significantly reduced compared to the control GFP dsRNAi beetles, indicating a role for a canonical CLK-dependent transcriptional/translational circadian feedback loop in T. castaneum. Considering the high proportion of arrhythmic beetles within the beetle population, knockdown experiments performed on already identified rhythmic adults could provide further support for this finding.
Since T. castaneum is a non-social but gregarious species, further experiments to study their circadian rhythms in a group setting would be interesting. The social influence on circadian rhythms has not only been demonstrated in social insects but also for species like Drosophila for which this is most likely mediated by olfactory cues (Levine et al., 2002a). Studies of circadian rhythms of T. castaneum in a social setting are especially important because it is already known that an exposure to the male aggregation pheromone increases the expression of the timeless clock gene in these beetles (Rath et al., 2021). It is possible that social interaction strengthens molecular clock function and synchronization, leading to improved behavioral rhythmicity in beetles kept in a group setting compared to those housed as individuals.
As T. castaneum is a ubiquitous pest, knowledge about its diel patterns in behavior under different external environmental cues can be useful in pest-management strategies, for instance, in light traps. Recently, circadian clock research is gaining more importance in immune system homeostasis (Haspel et al., 2020). Improved knowledge about the circadian clock and the rhythms it controls in T. castaneum, which is an excellent model to study insect immunity (Altincicek et al., 2008; Ferro et al., 2019), can be beneficial for the emerging research in the intersection of circadian function and immune regulation which has potential clinical relevance. It is remarkable that despite the absence of strong light cues in their natural habitats, the molecular clock of flour beetles still shows a strong entrainment to light as well as that many beetles can retain this rhythmicity in constant conditions. The considerable variation seen in different aspects of the diel patterns along with its status as a group-living yet non-social insect renders this organism a unique model for investigating circadian clocks and the different behavioral and physiological parameters they control. This complements studies in other insect groups, contributing to a comprehensive understanding of circadian rhythms across different insect species.
Supplemental Material
sj-zip-1-jbr-10.1177_07487304241263619 – Supplemental material for Deciphering a Beetle Clock: Individual and Sex-Dependent Variation in Daily Activity Patterns
Supplemental material, sj-zip-1-jbr-10.1177_07487304241263619 for Deciphering a Beetle Clock: Individual and Sex-Dependent Variation in Daily Activity Patterns by Reshma R, Tobias Prüser, Nora K. E. Schulz, Paula M. F. Mayer, Maite Ogueta, Ralf Stanewsky and Joachim Kurtz in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank Hannah Goldgräbe for helping with the experiments. This research was funded by the German Research Foundation (DFG) as part of the SFB TRR 212 (NC3, Project number 316099922)—Project number 396780003 (to Joachim Kurtz)
Author Contributions
JK and RS conceived the ideas. JK, RS, MO, RR, NKES, and TP planned the experimental procedure. RR, TP, NKES, and PMFM carried out the experiments, and RR carried out the statistical analyses together with NKES, MO, TP, and PMFM. RR wrote the first draft which was revised by NS and TP. RR and TP produced the figures. All authors contributed to writing of the article in further revisions and approved the submitted version.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
