Abstract
The daily pattern of animal behavior is thought to be of potential enormous importance for survival. Here, we compared the daily activity pattern of Drosophila melanogaster wild-type flies and the clock-impaired mutants, per01 and ClkJrk, under pseudo-natural conditions and laboratory conditions with natural-like temperature profiles. We found that clock-impaired flies respond stronger to changes in the environment, namely temperature increases, than wild-type flies. We hypothesize that the circadian clock may suppress unproductive activity in response to temperature fluctuations but that such suppression can be overcome in extreme conditions that are likely life-threatening for the flies. Thus, possessing a clock seems to be of adaptive significance.
Recently, it was observed for mice and fruit flies that activity patterns in nature are quite different from those reported in the laboratory and that some canonical clock mutants do not differ very much from wild-type animals (Daan et al., 2011; Vanin et al., 2012). In a heroic study, Vanin et al. (2012) monitored thousands of fruit flies during spring, summer, and autumn outside the laboratory at 2 geographical locations, Leicester (United Kingdom) and Treviso (Italy), over 3 seasons. One important finding was that wild-type flies and clock-impaired per mutants had similarly timed morning (M) activity bouts but, in addition, developed a third activity bout in the afternoon (A) under warmer conditions that had hitherto not been described in laboratory studies. The A peak seemed to be clock-controlled because it occurred earlier in the short period mutants pers, but its nature is unclear. Vanin et al. (2012) speculated that it might be a clock-controlled escape response of the flies. Even more astonishing than the emergence of the A peak is the fact that there was virtually no difference in the phasing of the activity pattern of per01 mutants and wild-type flies in nature, particularly for the M component. per01 mutants have been shown to possess a residual clock in several studies (Bywalez et al., 2012; Collins et al., 2005; Dowse et al., 1987; Helfrich-Förster, 2001; Helfrich and Engelmann, 1987; Kempinger et al., 2009), so could this residual clock be robust enough to produce wild-type activity patterns under natural conditions? These outdoor experiments challenge our assumptions about the necessity of a circadian clock for the locomotor phenotype and question the usefulness of laboratory studies for providing adaptive explanations for behavioral rhythms.
Here, we provide a possible explanation for the A peak and show the first evidence that the activity patterns of per01 mutants and wild-type flies are actually different under pseudo-natural conditions. We hypothesize that the circadian clock is required to suppress futile activity in response to increasing temperatures during the day, nevertheless permitting escape responses to high hazardous temperatures. Our view is based on laboratory studies with simulated natural-like temperature cycles and on outdoor experiments under changing weather conditions.
Materials and Methods
Fly Strains
CantonS and wild-type Alto-Adige (WTALA) flies (Sandrelli et al., 2007; Tauber et al., 2007) were used as the wild-type flies. WTALA consists of a mixed culture of 37 isofemale lines caught in Val Venosta (Alto-Adige) in North Italy (45°N) that was regularly refreshed by adding few flies from the isofemale lines that were maintained separately. Vanin et al. (2012) used the same mixed culture for their outdoor experiments. The clock mutants per01 and ClkJrk are described in Konopka and Benzer (1971) and Allada et al. (1998), respectively. The per01 mutants were obtained from C. P. K. Kyriacou and have previously been isogenized with the CantonS wild-type strain (Vanin et al., 2012). To judge the behavior of per01 mutants additionally in a different genetic background, we used w1118 per01;+;uas-per16 flies (Grima et al., 2004). These show no period expression and have previously been demonstrated to behave identical to per01 mutants, at least under laboratory conditions (Rieger et al., 2009). All strains were reared in conventional 3 × 9-cm food vials on Drosophila medium (0.8% agar, 2.2% sugar-beet syrup, 8.0% malt extract, 1.8% yeast, 1.0% soy flour, 8.0% corn flour, and 0.3% hydroxybenzoic acid). Rearing occurred either in the lab under LD 12:12 cycles at 25 °C (for the experiments carried out in Italy) or under the same outdoor conditions that were used for monitoring the flies (for the experiments carried out in Germany).
Recording of Activity Rhythms
Locomotor activity of individual male flies was recorded photoelectrically in 1-min intervals using the Drosophila Activity Monitoring (DAM) System (Trikinetics, Inc., Waltham, MA). At the age of 3 to 5 days, individual male flies were transferred into the recording tubes, which were filled one-third with agar/sugar medium (2% agar; 4% sucrose) and were closed by an air-penetrable plug.
For the laboratory experiments, activity monitors were put into a temperature-controllable incubator (I-36NL; Percival Scientific, Inc., Perry, IA) to enable simulation of natural temperature cycles (according to Yoshii et al., 2009; Bywalez et al., 2012). Temperature linearly increased/decreased between the lowest temperature in the morning and the highest temperature 3 h after noon in 1-min intervals. Lowest and highest temperatures were set to 16 °C and 26 °C (for 16 °C/26 °C temperature-cycles), to 20 °C and 30 °C (for 20 °C/30 °C temperature-cycles), or to 25 °C and 35 °C (for 25 °C/35 °C temperature-cycles). Day length was set to either 12 h or 16 h. Illumination, derived from “white” LEDs (Lumitronix LED-Technik, GmbH, Jungingen, Germany), was adjusted to 100 lux (19 µW/cm2). Dawn and dusk were simulated by increasing/decreasing irradiance in 1-min steps within 1.5 h (Rieger et al., 2007, 2012).
For the outdoor experiments, activity monitors were put in countryside areas without urban light, well protected from direct sun and rain. One experiment was performed in Treviso (Italy) at the beginning of June 2010 under the same conditions used by Vanin et al. (2012). The other experiments were performed in a garden in the countryside around Würzburg (Germany) from June to August 2012. Temperature and light intensity (>10 lux) were monitored by environmental monitors (Trikinetics) in parallel to the activity of the flies.
Recording of the Flies’ Activity Level (Distance Walked) during Increasing Environmental Temperatures
To examine the amount and the kind of activity the flies showed when temperatures rose during midday, we video-monitored the flies’ activity in the lab (video camera: Panasonic HDC-SD1, Panasonic, Secaucus, NJ), first in response to a 10 °C temperature increase from 25 °C to 35 °C at zeitgeber time 8 (8 h after dawn) over 30 min and second during a natural-like light and slow temperature increase taking 11 h as under a 16-h photoperiod. The latter experiment was carried out in a climate chamber at the Department of Animal Ecology and Tropical Biology (Uni Würzburg). Flies were kept individually in the same glass tubes in which they were recorded previously, and locomotor activity was video-monitored for 10 min in regular time intervals. Movies were imported into the Ethovision 8.5 software (Noldus Information Technology, Leesburg, VA), and the distance walked was determined for each fly at each temperature. Walking distance was plotted against temperature and a correlation coefficient calculated. Furthermore, the temperature was determined at which the distance walked increased significantly in comparison to the previous time intervals. During the fast temperature increase, 25 flies were kept additionally together in conventional food vials (3 cm diameter, 9 cm height), and snapshots of their position were taken at the different temperatures.
Determination of Activity Onsets and Offsets under Natural Conditions
Onsets and offsets of morning activity, afternoon activity, and evening activity under pseudo-natural conditions were basically determined as described in Vanin et al. (2012). Briefly, these were determined fly-by-fly for each day of every experiment, as shown in Figure 1. Activity onset was set if continuous movement with a steady increase of activity (and no more than 1 half-hour time bin without any or lower activity than the previous bin) led to a peak, followed by a steady decrease of activity defining the offset. Figure 1 shows representative examples for a wild-type and per01 fly. Special care was taken not to count sudden increases in activity level due to strong changes in illumination as activity onset of the afternoon peak (e.g., Fig. 1, on day 2 for per01).
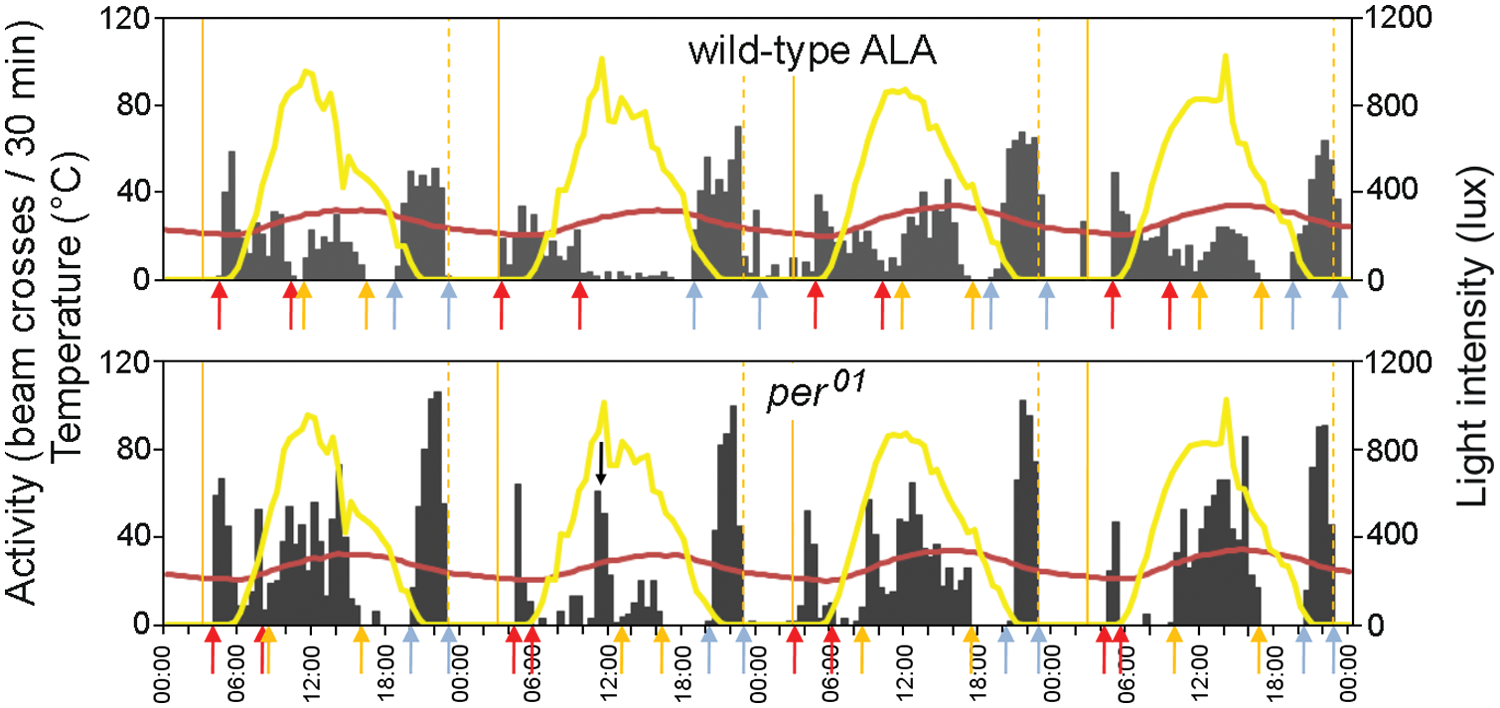
Determination of on- and offset of morning (M) activity (red arrows), afternoon (A) activity (orange arrows), and evening (E) activity (blue arrows) of individual flies. Representative examples of a wild-type fly (ALA) and a per01 mutant are shown. Environmental temperature cycles (red) and light cycles (yellow) as well as nautical dawn (solid orange line) and dusk (broken orange line) are indicated. Activity onsets were defined as onsets of continuous movements with a steady increase of activity (and no more than 1 time bin with lower activity than the previous bin) led to a peak, followed by a steady decrease of activity defining the offset. Special care was taken not counting sudden increases in activity level due to strong changes in illumination as activity onset of the afternoon peak (e.g., arrow, on day 2 for per01).
Statistics
Data were analyzed for statistical differences by a 1- or 2-way analysis of variance (ANOVA) followed by a Bonferroni post hoc test after testing for normal distribution (Kolmogorov-Smirnov). Significant differences in the frequency of the A peak between wild-type flies and per01 mutants were tested by the χ2-test.
Results
Activity Recordings in the Laboratory
In our first experiment, laboratory activity of wild-type flies (CantonS) and the clock-mutants per01 and ClkJrk was monitored for 7 days under LD 12:12 with simulated twilight and maximal light intensity 100 lux, superimposed on natural-like low temperature cycles (16 °C to 26 °C) as well as under temperature cycles alone (in constant darkness DD) (Fig. 2). Under these conditions, wild-type flies show the usual bimodal activity patterns with M and evening (E) peaks, whereas the clock mutants developed an additional A peak. This A peak was most pronounced in ClkJrk mutants. Under temperature cycles alone, the activity of ClkJrk mutants appeared to track the temperature increase. Under combined LD and temperature cycles, ClkJrk still followed the temperature increase but responded additionally to dusk. Wild-type flies did not show such a temperature response at all, indicating that a functional clock suppresses the activating effects of increasing temperatures. The behavior of per01 mutants was intermediate between that of ClkJrk mutants and wild-type flies, suggesting again that per01 mutants are not completely clock-less but still possess a residual clock (see also Bywalez et al., 2012). In the next step, we aimed to provoke the A peak also in wild-type flies by exposing them to long days (16 h of light and 8 h of darkness, LD 16:8) and temperature cycles with higher temperatures. At 20 °C/30 °C temperature cycles, we could only elicit a tiny A peak in 2 of 24 flies that is not visible in the average activity profile, but at 25 °C/35 °C, all 41 flies revealed the A peak (Fig. 2).
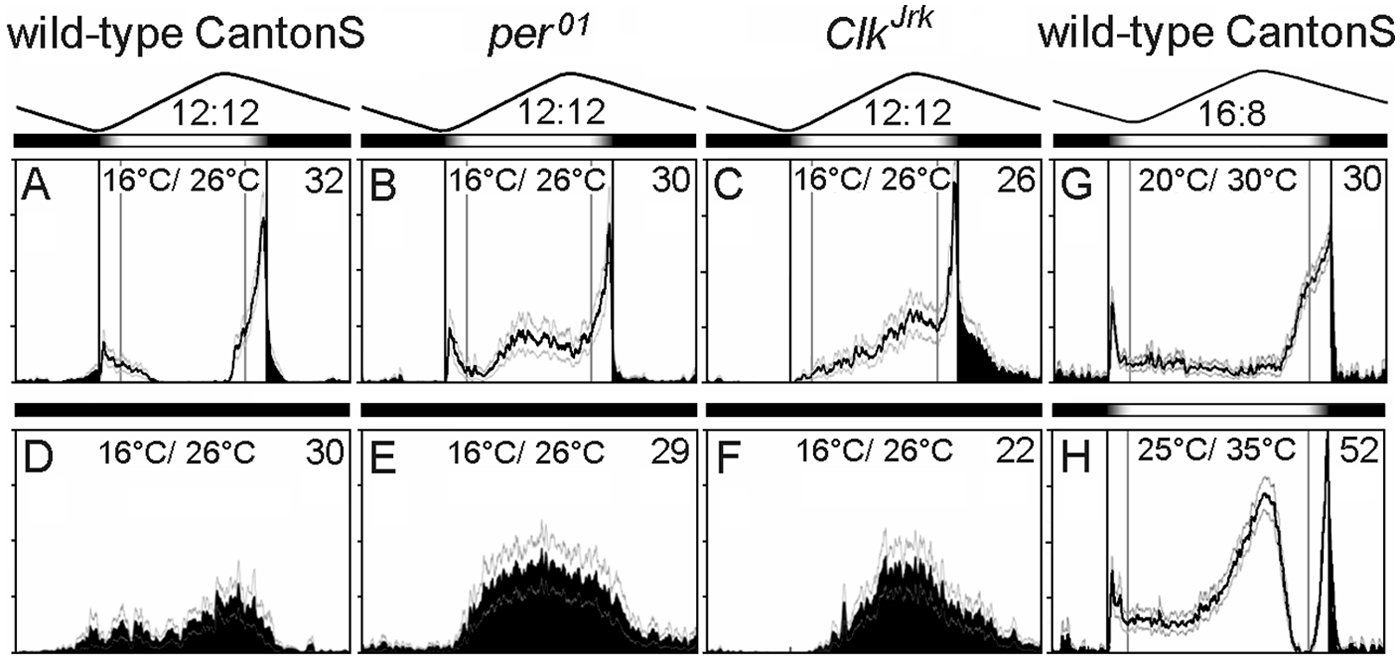
Average activity profiles (
We then examined the kind of activity wild-type flies show at temperatures exceeding 30 °C. We video-monitored the flies’ activity in response to a 10 °C temperature increase from 25 °C to 35 °C at zeitgeber time 8 (8 h after dawn) in 30 min. Flies were either kept individually in the same glass tubes in which they were recorded previously, or 25 flies were kept together in conventional food vials (3 cm diameter, 9 cm height; Fig. 3). Although the temperature increase was much faster than in nature, the flies did not change their behavior when the temperature increased from 25 °C to ~29 °C; they quietly moved around or remained immobile. When the temperature reached 30 °C to 31 °C, they became immobile, and above 31 °C to 32 °C, they became hyperactive. The flies’ immobile phase was more pronounced in the food vial than in the single glass tubes: At ~30 °C, all flies moved to the bottom of the vial and remained immobile on the food (Fig. 3B), perhaps because the food was still cooler than the air. Fifty percent of the flies even stayed there when the temperature increased further (Fig. 3C). The other 50% of flies in the vial, as well as all the flies housed in the individual glass tubes, moved toward the Styrofoam plugs (Fig. 3C). We conclude that this high activity may represent an escape response as suggested by Vanin et al. (2012). However, in contrast to Vanin et al. (2012), we propose that the response to temperature is different between wild-type flies and clock mutants and that an intact clock will suppress activity at increasing temperatures as long as the latter are tolerable for the flies.
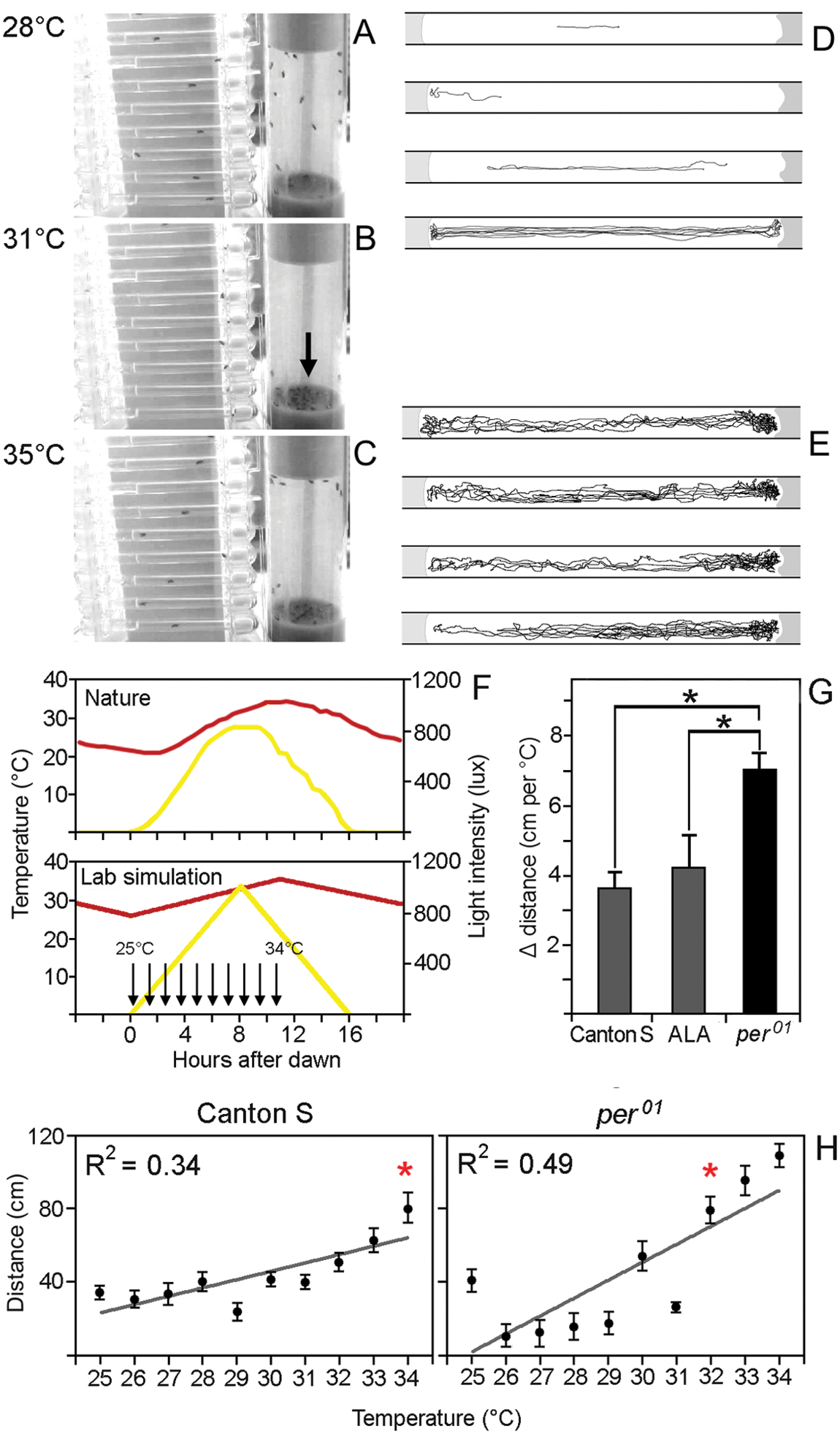
Behavioral changes provoked by a temperature increase from 25 °C to 35 °C within 30 min (A-E) and a natural-like temperature increase of the same magnitude within 11 h (F-H). For the fast temperature increase, the flies were kept either individually in glass tubes, filled with food at the left and closed by a Styrofoam plug at the right, or in a group of 25 flies in regular food vials. At 28 °C, the flies were randomly placed in the tubes and the vial (A) and either moved around or were rather inactive. At 31 °C, all flies remained immobile on the food in the vial (arrow) or close to the foam plug in the tubes (B). At 35 °C, the flies in the tubes moved around quickly, whereby they spent the most time at the foam plugs (C). In the food vial, half of the flies moved around quickly, spending most time close to the foam plug. The other half remained immobile on the food. (D, E) Tracks of 4 individual flies during 1-min intervals. At 28 °C, the flies did not show any preference for a certain spot in the tube and moved rather straight, if they moved at all (D). At 35 °C, the movements of the flies were less straight and clearly directed against the foam plug (E). For the natural-like temperature increase, the flies were kept individually in glass tubes and their activity was filmed for 10 min when the temperature had increased by 1 °C (see arrows in F). Light (yellow) and temperature (red) cycles in the lab mimicked the natural ones (F) as closely as possible. The distance the flies walked during the filmed 10 min increased with increasing temperature in all genotypes (H; ANOVA: CantonS: F(9, 90) = 10.780, p < 0.001; WTALA: F(9, 90) = 4.216, p < 0.001; per01: F(9, 90) = 30.481,
Since the temperature increase of 10 °C in 30 min is much faster than in nature, we repeated the same experiment with a slow temperature increase that represents the natural situation, and we included per01 mutants in the experiment (Fig. 3F-H). If our hypothesis is true, per01 mutants should show an earlier and faster activity increase with increasing temperature than wild-type flies. This was indeed the case: per01 mutants started with a lower initial activity (perhaps because their M peak terminated earlier; see below) than wild-type flies, but they increased their activity significantly more when the temperature rose (Fig. 3H). As a consequence, the correlation coefficient between temperature and traveled distance was higher in per01 mutants than in wild-type flies. A significant increase in the walked distance occurred in wild-type flies only when the temperature rose to 34 °C, whereas in per01 mutants, the walking distance significantly increased already at 32 °C (Fig. 3H).
Activity Recordings Outdoors
If an intact clock suppresses activity in response to moderate temperature increases, the A peak of wild-type flies should look different from that of per01 mutants, even under pseudo-natural conditions. To test this, we monitored outdoor activity in wild-type flies and per01 mutants (including w1118 per01;+;uas-per16 flies) in Treviso and Würzburg using the same environmental and activity monitors (Trikinetics) and the same wild-type strain “Alto Adige” (WTALA) as used by Vanin et al. (2012). We did not find any significant differences between the 2 per01 strains. Therefore, we will simply talk about per01 mutants in the following.
The first experiment was performed for 9 days in Treviso (45°40′N) at the beginning of June 2010. During these June days, photoperiod (sunrise to sunset) was close to 16 h, with sunrise at 0500 h and sunset at 2100 h. The first 7 days were predominantly sunny, and temperatures varied roughly between ~20 °C and ~30 °C on days 1 to 3 and between ~25 °C and ~35 °C on days 4 to 6. In the evening on day 6, a thunderstorm occurred, and temperatures dropped back to ~20 °C/~30 °C on day 7 (Fig. 4). Day 8 was rainy, with light intensity never exceeding 200 lux and temperatures staying rather constant around ~20 °C. On day 9, the rain ceased and the temperature rose to ~26 °C in the afternoon (Fig. 4). As reported by Vanin et al. (2012), M, A, and E activity peaks were visible in both strains, but we detected clear differences. Wild-type flies showed the A peak mainly during the warm days, whereas the mutants revealed it more frequently (χ² = 42.59, p < 0.001; n = 307; Fig. 4C). To quantify M, A, and E components in more detail, we determined the onsets and offsets of each component for each individual fly (as was done by Vanin et al., 2012, for the onset for M and E components). Furthermore, we calculated the percentage of activity the flies spent during M, A, and E components during 4 representative days (days 2-5) by summing the number of activity counts during each component, dividing it by the total activity counts for that day, and multiplying it by 100. We found that wild-type flies started the A peak significantly later than per01 mutants (ANOVA: F(1, 150) = 16.150, p < 0.001; Fig. 4C) and spent a significantly lower relative amount of daily activity in the A component (ANOVA: F(1, 89) = 84.944, p < 0.001; Fig. 4B). These results suggest that wild-type flies suppress the A peak even in nature unless they are at risk of overheating. On the last 2 rainy days, the activity resembled very much the activity pattern known from the laboratory. Wild-type flies were bimodal and per01 mutants showed a more uniform diurnal activity. This observation suggests that the activity pattern of flies in nature is not as different from that in the laboratory as long as they experience an “English” summer environment (i.e., cooler days with little temperature cycling on overcast days, with no obvious requirement for escape responses). Consequently, the comparison of laboratory and natural experiments is useful for dissecting out putative adaptive responses.
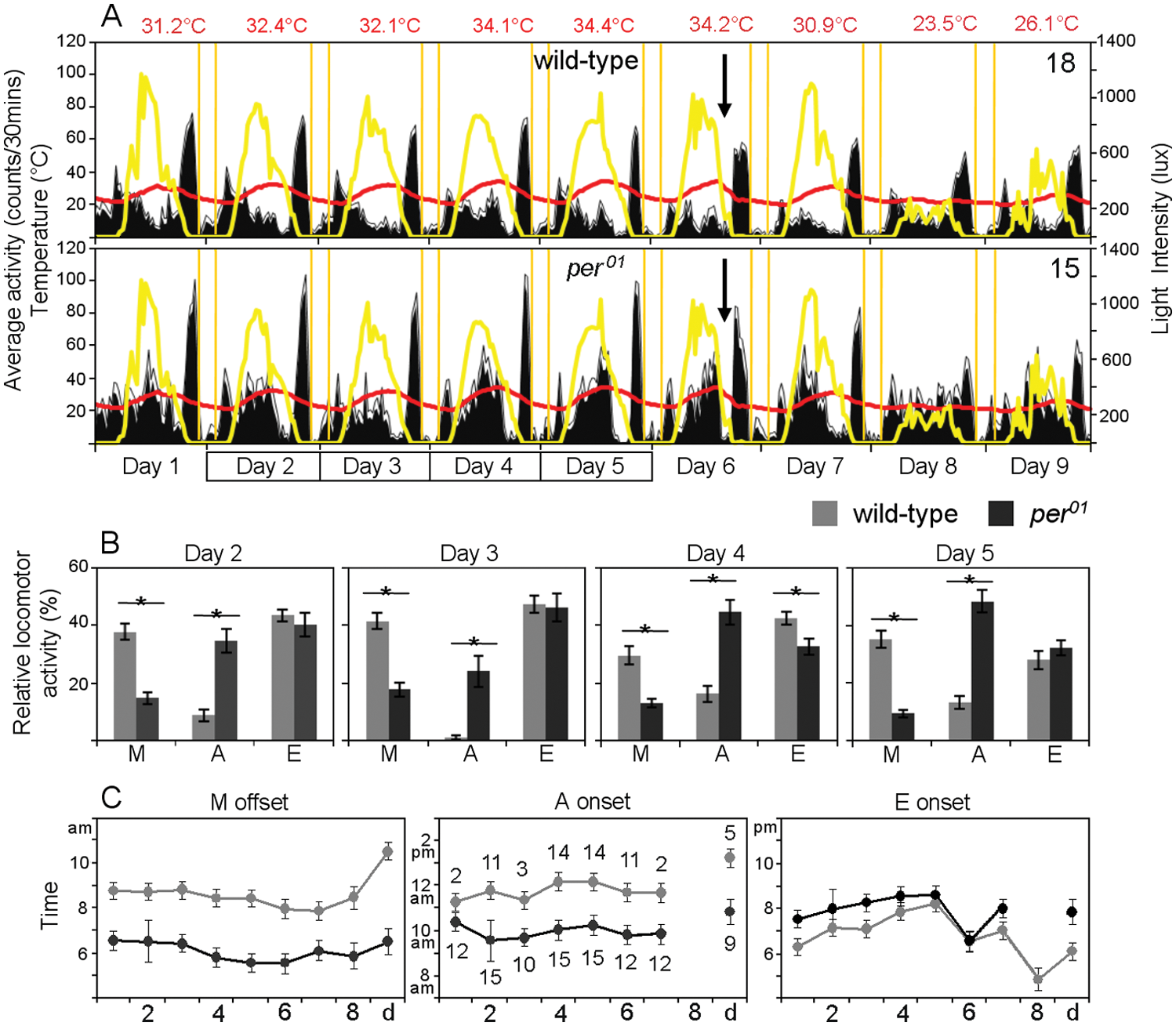
Average activity pattern of wild-type flies and per01 mutants monitored in parallel for 9 days in June (June 5-13, 2010) at Treviso; number of recorded flies is given in the right corner. The red lines represent the temperature cycle and the yellow shading the light cycle. The red numbers on top indicate the maximal temperatures on the respective days. Vertical yellow lines indicate beginning and end of nautical twilight. The relative amount of activity shown during morning, afternoon, and evening activity components (indicated as M, A, and E, respectively) was calculated for days 2 to 5 for both wild-type and per01 (B). Asterisks indicate statistically significant differences between the 2 genotypes. The amount of activity displayed by the 2 strains during the E component was rather similar, but wild-type flies moved more during the M and less during the A component than per01 mutants. Furthermore, per01 mutants showed earlier Moffsets and Aonsets (C;
We also found significant differences between wild-type flies and per01 mutants for the M component and, to a lesser extent, also for the E component. The M peak of per01 mutants seemed sharper than that of wild-type flies, and the quantitative analyses showed that per01 mutants spent a significantly lower relative amount of daily activity in the M component (ANOVA: F(1, 123) = 145.46, p < 0.001; Fig. 4B). Whereas the M activity onset occurred at similar times in both strains, the offset of M activity was much later in wild-type flies (Fig. 4C). Wild-type flies’ M peak lasted on average 2 h longer than that of per01 mutants (ANOVA: F(1, 225) = 129.49, p < 0.001). In agreement with Vanin et al. (2012), we propose that the onset of M activity is predominantly triggered by external cues (i.e., nautical twilight and temperature) and that this is the same in wild-type flies and per01 mutants. Nevertheless, in wild-type flies, M activity is still under clock control, and therefore it continues after the stimulus itself (nautical twilight) has stopped. The relative amount of activity during the E component was similar between the 2 genotypes, but the onset of E activity was significantly dependent on the genotype (Fig. 4C) (ANOVA: F(1, 265) = 106.86, p < 0.001). Bonferroni post hoc analysis revealed that wild-type flies had a significantly earlier onset of E activity (Eonset) than per01 mutants on all days except days 5 and 6. Eonset of wild-type flies depended strongly on temperature (F(8, 46) = 25.50, p < 0.001) occurring later at higher temperatures (Fig. 4C), whereas that of per01 mutants was more constant and remained closer to dusk (with the exception of day 6; Fig. 4C). Day 5, with a temperature maximum of 34.4 °C, was the hottest day during the recorded period. On this day, wild-type flies delayed Eonset toward dusk, and consequently, it did not differ from per01 mutants. Day 6 was exceptional in the sense that both genotypes showed simultaneously a very early Eonset (Fig. 4C). The latter coincided with a sudden decrease in illumination and temperature caused by a thunderstorm (arrows in Fig. 4A). Thus, Eonset may have occurred in response to these environmental changes in both genotypes.
The next experiments were carried out in Würzburg (49°41′N) during June to August 2012. In comparison to Treviso, the climate in Würzburg is more continental, resulting in temperature amplitudes larger than 10 °C (see Fig. 5). As expected from the results of Vanin et al. (2012), this affected the flies’ timing of M, A, and E activity. Here, we focused only on evident differences between wild-type flies and per01 mutants. We found that the differences in occurrence and timing of the A peak between the 2 genotypes observed in Treviso were similarly present in Würzburg. We chose one experiment that was performed for 11 days in August for a more detailed analysis (Fig. 5), because this experiment included variable weather conditions as did the experiment in Treviso. During these August days, photoperiod (sunrise to sunset) was close to 14 h with sunrise at ~0520 h and sunset at ~1920 h. Some days were rather hot, with the maximal daily temperature between 33.2 °C and 36.8 °C (days 3-6), and others were colder, with the maximal daily temperature remaining below 30 °C (days 1-2, 7-11). The coolest night, at 13.0 °C, occurred between days 7 and 8. During this night, the flies, especially per01 mutants, were rather inactive. Unlike Treviso, there was no day on which temperatures stayed constantly at ~20 °C, but the last 3 days were rainy with only moderate temperature changes. On days 10 and 11, per01 mutants showed no clear M and E peaks, whereas wild-type flies were bimodal. Thus, the outdoor activity of the flies came again close to the activity pattern reported for laboratory conditions (Fig. 5A). As found for Treviso, phase analysis of the Würzburg data revealed that Monset was the same in both genotypes, whereas Moffset occurred significantly later in wild-type flies than in per01 mutants (Fig. 5B). Similarly, Aonset occurred significantly earlier in per01 mutants. Under Würzburg conditions, we even found that per01 mutants showed a longer A peak than wild-type flies (Aoffset was significantly later in per01 mutants; Fig. 5B). These results clearly underline our finding that per01 mutants have a more pronounced A peak than wild-type flies. Regarding E activity, we found differences between Treviso and Würzburg: per01 mutants showed an earlier Eoffset instead of a later Eonset than wild-type flies under Würzburg conditions (Fig. 5B).
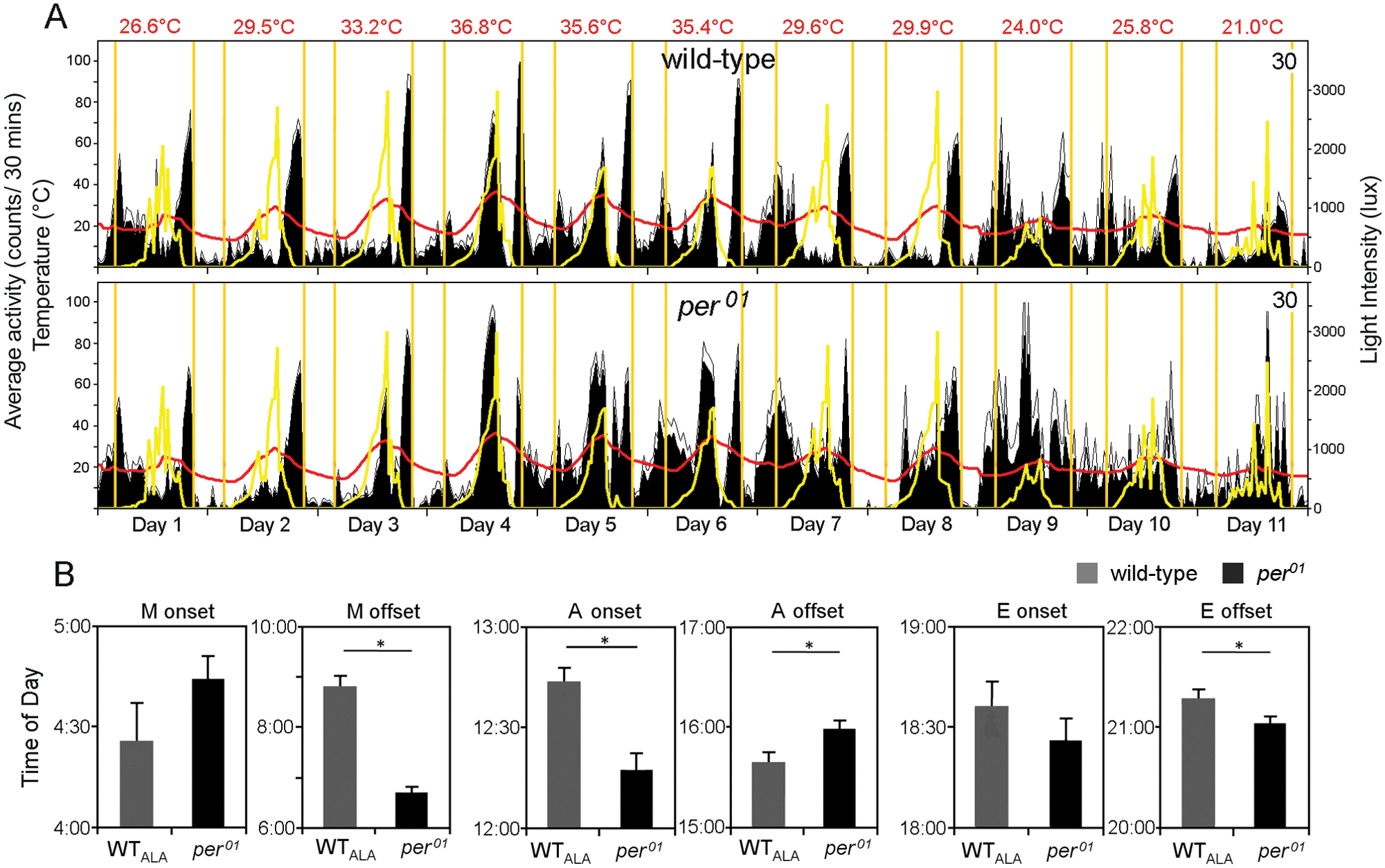
Average activity pattern of wild-type flies and per0 mutants monitored in parallel for 11 days in August (August 16-26, 2012) at Würzburg (A); number of recorded flies is given in the right corner (labeling as in Fig. 4). Activity on- and offsets were determined for morning (M), afternoon (A), and evening (E) peaks and averaged over all days for wild-type flies and per01 mutants (B;
Discussion
Here, we provide an alternative view on the results of Vanin et al. (2012) that favors the adaptive value of possessing a circadian clock. Such a clock may help animals save energy. Being active at too high temperatures is costly and should be avoided unless it is necessary to move for reasons of hunger or to avoid predators or hazardous environmental conditions. We assume that clock-impaired flies will waste more energy than wild-type flies by responding to temperature increases, and in the long run, they may be at a fitness disadvantage to wild-type flies. A similar conclusion was drawn by Daan et al. (2011).
Our views confer with those of Vanin et al. (2012) that the A peak is influenced by the clock, since they showed that A onset occurs earlier in pers with a fast endogenous clock as well as in per01 mutants, which may also have a faster residual clock. Indeed, our results described above reveal this to be so for per01. As temperature sensitivity of flies is modulated by the clock causing different temperature preferences during the course of the day (Kaneko et al., 2012), pers mutants may become more sensitive earlier than wild-type flies to the stimulus of increasing temperature. The question remains why perL mutants with a slow endogenous clock show the A onset at approximately the same time as wild-type flies and not later (Vanin et al., 2012). A possible explanation is that there is a temperature limit above ~32 °C at which the flies have to respond independently of the signals from the clock. Most interestingly, perL mutants show a later offset of A activity than wild-type flies (Vanin et al, 2012), and this fits again to a slow endogenous clock. Nevertheless, we have to note that the onsets of activity are also context dependent and that the just described differences between wild-type flies and pers and per01 mutants were not always found. For example, Vanin et al. (2012) could not reproduce the earlier Aonset in pers and per01 mutants under lab conditions. They did also not find a more frequent occurrence of the A peak in per01 mutants as compared with wild-type flies under lab conditions, at least not under simulated summer conditions (1600 lux, 25 °C/35 °C). The latter is consistent with our results: We found that wild-type flies always revealed the A peak at 25 °C/35 °C temperature cycles in the lab (at 100 lux). The difference between both genotypes was only evident under lower temperatures. In Supplementary Figure S9, Vanin et al. (2012) show the frequency of the A peak also under 20 °C/30 °C. Under these conditions, a higher percentage of per01 mutants showed the A peak as compared with wild-type flies. Although this difference seemed not to be significant, it is in line with our findings. We did only use 100 lux during our 20 °C/30 °C temperature cycles. Since the flies also respond to changes in illumination (Vanin et al., 2012; Rieger et al., 2007; Fig. 1 in this article), it is well conceivable that higher light intensities applied in a natural-like manner, as done by Vanin et al. (2012), promote the occurrence of the A peak and thus diminish differences between per01 mutants and wild-type flies.
Furthermore, the phasing of the E peak seems to be strongly context dependent. In contrast to Vanin et al. (2012), we found that the onset of the E peak was later in per01 than in wild-type flies, but this was only true on most days under Italian and not at all under German outdoor conditions. Vanin et al. (2012) reported that Eonset was even earlier in per01 mutants than in wild-type flies. They have analyzed thousands of flies in Treviso and Leicester, and it is well possible that on average, Eonset is earlier in per01 mutants than in wild-type flies.
Nevertheless, more important than the just discussed small discrepancies is the fact that per01 mutants and wild-type flies behaved significantly different under conditions that mimic nature more closely than the usual rectangular LD cycles (e.g., simulated dawn and dusk together with moderate temperature cycles in the lab as well as the pseudo-natural conditions of the present study). It will be most rewarding to look at such differences under even more natural conditions. In nature, flies live not isolated in glass vials but in groups on or in rotten fruits. Without doubt, social, auditory, olfactory, and gustatory cues influence their activity pattern (Kent et al., 2008; Krupp et al., 2008; Billeter et al., 2012). This has clearly been shown in laboratory experiments, in which male and female flies were allowed to interact (Fujii et al., 2007).
In summary, we show here that the locomotor activity of flies is composed of direct responses to environmental changes and the output from the circadian clock. We reveal that significant differences in the rhythmic behavior of wild-type and per01 flies are present under pseudo-natural conditions; the activity of per01 mutants responds more directly to the environment than does that of wild-type flies. Certainly, we have analyzed only a relatively small data set. It will be most interesting to mine the large data set obtained by Vanin et al. (2012) for such differences. If our hypothesis is correct, completely clock-less flies such as ClkJrk or cyc01 mutants should respond even more to the environmental changes than the per01 mutants that have a residual clock.
Footnotes
Acknowledgements
We thank C.P. Kyriacou and R. Costa and 2 unknown reviewers for comments on the manuscript, Stefano Vanin for help with the experiments in Treviso, Dirk Rieger for help in Würzburg, Atefeh Pooryasin for help with the Noldus software, and Jochen Krauss and Ingolf Steffan-Dewenter for letting us use their climate chamber for simulating the natural-like temperature and light cycles. C.H.-F. acknowledges financial support from the European Community (the 6th Framework Project EUCLOCK no. 018741) and the DFG (Fo207/12-3; 13-1). T.Y. acknowledges the JSPS for a Grant-in-Aid for Research Activity Start-up.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
