Abstract
Various intestinal functions exhibit circadian rhythmicity. Disruptions in these rhythms as in shift workers and transcontinental travelers are associated with intestinal discomfort. Circadian rhythms are controlled at the molecular level by core clock and clock-controlled genes. These clock genes are expressed in intestinal cells, suggesting that they might participate in the circadian regulation of intestinal functions. A major function of the intestine is nutrient absorption. Here, we will review absorption of proteins, carbohydrates, and lipids and circadian regulation of various transporters involved in their absorption. A better understanding of circadian regulation of intestinal absorption might help control several metabolic disorders and attenuate intestinal discomfort associated with disruptions in sleep-wake cycles.
Several physiological, biochemical, and behavioral activities in living organisms cycle with a period of 24 h and are referred to as circadian rhythms. Such rhythms are the overt manifestation of endogenous, self-sustained oscillators that can be synchronized to the 24-h day. At the molecular level, mRNA and proteins exhibit circadian rhythms, as do the activities of individual cells. Daily changes in light provide powerful external cues to entrain circadian activities. In addition, food consumption that occurs regularly may also act as an entraining agent. Here, we will review how light and food signals regulate intestinal food absorption. Specifically, we will summarize the circadian regulatory mechanisms that control absorption of macronutrients with an emphasis on transport proteins involved in the absorption of amino acids, sugars, fatty acids, and cholesterol. Understanding the molecular mechanisms involved in the circadian regulation of nutrient uptake might provide new targets in the treatment of diabetes, obesity, and hyperlipidemia.
Central and Peripheral Circadian Regulation by Clock Genes
Daily changes in light are sensed by retinal ganglion cells (Berson et al., 2002), and this information is transmitted to the suprachiasmatic nuclei (SCN) in the brain, site of the master circadian clock in mammals. The basic intracellular oscillatory mechanism is a transcriptional-translational autoregulatory feedback loop. Two transcription factors, Bmal1 and Clock, dimerize and bind to
Besides these transcriptional changes, circadian regulation is also modulated by posttranslational modifications of the clock proteins (Bass and Takahashi, 2010; Dibner et al., 2010; Bass, 2012). Both Cry and Per proteins undergo posttranslational phosphorylation, and these modifications affect the stability and half-lives of these proteins. Thus, changes in protein levels via protein degradation pathways also participate in the circadian control of clock genes.
Apart from these core clock genes, Bmal1:Clock heterodimers also interact with E-boxes present in the promoters of other clock-controlled genes that regulate metabolism. Therefore, core clock and clock-controlled genes represent the upper echelon of a hierarchical top-down system of regulation that translates changes in the environment into protein expression, leading to alterations in physiological, biochemical, and behavioral activities.
Although circadian rhythms are centrally controlled by the SCN, core clock and clock-controlled genes are also expressed in almost all cells. We and others have shown that clock genes are rhythmically expressed in intestinal cells (Stokkan et al., 2001; Sladek et al., 2007; Hoogerwerf et al., 2007; Froy and Chapnik, 2007; Pan and Hussain, 2009). The clock genes in the enterocytes are not in contact with neurons originating from the eye, and therefore, their rhythmicity is most likely entrained by hormonal and neuronal signals emanating from the SCN (Welsh et al., 2010; Mourad and Saade, 2011; Schwartz and Zeltser, 2013; Hussain and Pan, 2014; Hussain, 2014b; Gnocchi et al., 2015). This is supported by the observation that the peaks and nadirs in the expression of clock genes in the peripheral tissues are time delayed compared with their expression in the SCN. At this time, the molecular and neuronal mechanisms that entrain the expression of clock genes in the enterocytes are unknown.
Expression of Clock Genes in the Intestine
Various functions of the intestine exhibit circadian activities (Scheving, 2000; Hoogerwerf, 2006; Hussain and Pan, 2009; Sotak et al., 2011; Hussain, 2014b). It is well known that the major complaints of shift workers and transcontinental travelers are gastrointestinal disturbances (Vener et al., 1989). Hence, it is possible that many of the intestinal activities are regulated by clock genes, and disruptions in these activities might cause gastrointestinal discomfort. Indeed, the expression of various clock genes has been documented in various parts of the intestine. It has been shown that jejunum and colon exhibit high expression of clock genes (Pan and Hussain, 2007; Sladek et al., 2007; Hoogerwerf et al., 2007; Froy and Chapnik, 2007; Pan et al., 2010; Pan et al., 2013a; Pan et al., 2013b). Except for
Regulation and Expression of Clock Genes by Food
Apart from the mechanisms that are mainly triggered by light, expression of clock genes in peripheral tissues, such as in the intestine and liver, is also affected by food availability (Damiola et al., 2000; Stokkan et al., 2001; Hara et al., 2001; Stephan, 2002; Feillet et al., 2006; Mendoza, 2007; Hussain and Pan, 2009; Patton and Mistlberger, 2013; Hussain, 2014b). This can be easily demonstrated by providing food to rodents for a limited time instead of ad libitum feeding. Under conditions of restricted food availability at certain times, animals modify their behavior, resulting in maximal expression of peripheral tissue clock genes around the mealtime. Such changes in clock gene expression do not occur in the SCN. Thus, expression of clock genes in the peripheral tissues responds to various stimuli, and these stimuli supersede the central signals arising from the SCN. How food entrainment actually regulates peripheral clock gene expression is not well understood.
Changes after food entrainment have characteristic features of a circadian rhythm. The rhythms are sustained for some time after the food is removed. The phases of these rhythms are shifted by different food-entrainment protocols. Ablation of the SCN has no significant effect on food entrainment, indicating that it might be SCN independent (Stenvers et al., 2012; Schwartz and Zeltser, 2013; Hussain, 2014b; Asher and Sassone-Corsi, 2015). It is likely that the food-entrained oscillator is a network of several neural sites in the brain that cooperate to elicit a behavioral and physiologic response to the altered timing of food availability (Bechtold and Loudon, 2013; Hussain, 2014b).
Although the food-entrained oscillator might be independent of the SCN, the clock genes involved in light-induced circadian rhythms also respond to food entrainment in the peripheral tissues (Stokkan et al., 2001). Feeding mice for 10 d in the daytime significantly alters the expression pattern of clock genes (Pan and Hussain, 2009), with most of the genes showing peak expression around the time of food availability instead of the normal peak expression at the nighttime. Thus, food availability can alter the expression pattern of clock genes in the intestine. However, there seems to be an important coordination between SCN-regulated mechanisms and food entrainment as mice kept in constant dark or constant light fail to respond properly to food entrainment (Pan and Hussain, 2007, 2009). Thus, normal functioning of the light-entrained response might be necessary for adaptation to restricted food availability.
Clock genes probably also participate in food entrainment of lipid absorption. For example,
Structure of the Intestine and Polarity of Nutrient Absorption Across the Intestine
In this review, we will mainly focus on the absorption of food that occurs in the small intestine. Partially digested food arrives in the small intestine from the stomach. In addition, the small intestine obtains pancreatic secretions that are rich in various proenzymes involved in the digestion of nutrients. Since proteins and carbohydrates are water miscible, they are hydrolyzed by enzymes in the lumen of the small intestine. For lipids that are immiscible in water, an additional step of solubilization is needed before they can be acted upon by enzymes. For this purpose, the small intestine also receives bile acids from the gall bladder. In the lumen, these bile acids form micelles consisting of a hydrophobic core and a hydrophilic charged surface that can interact with water. Lipids are solubilized in these micelles and are hydrolyzed by pancreatic lipases, phospholipases, and cholesterol esterase, resulting in the generation of free fatty acids, monoacylglycerol, lysophospholipids, and cholesterol. Very little is known whether the intraluminal hydrolysis of these macronutrients shows any daily variation.
The hydrolyzed products of these macronutrients are taken up by enterocytes, highly differentiated intestinal epithelial cells that show polarity. The apical surfaces of these cells face the lumen and have microvilli to increase the surface area for absorption. The basolateral side faces the tissues. Nutrients are taken up by enterocytes from the apical side and transported to the basolateral side, and this process of transporting molecules across the enterocytes is referred to as absorption.
Regulation of Amino Acid and Peptide Uptake
Proteins are hydrolyzed in the intestinal lumen to amino acids and small di- and tripeptides. Different amino acids are taken up from the apical side (Fig. 1) by enterocytes involving several transporters (Broer, 2008). However, uptake of di- and tripeptides is mediated by a high-capacity, low-affinity peptide transporter 1 (PEPT1) present in the apical membranes. It is also involved in the uptake of different drugs such as β-lactam antibiotics, selected angiotensin-converting enzyme inhibitors, and protease inhibitors, mainly due to their structural resemblance to di- and tripeptides (Rubio-Aliaga and Daniel, 2002; Daniel, 2004; Spanier, 2014). This transporter does not recognize free amino acids or peptides containing 4 or more amino acids (Daniel, 2004). PEPT1 is a proton-dependent peptide co-transporter; therefore, peptide uptake is coupled to the proton gradient and is independent of the intracellular peptide concentrations. The proton gradient is maintained by another sodium-proton exchanger, NHE3, that is also located in the apical membranes of the enterocytes (Fig. 1). This exchanger pumps out H+ and brings Na+ into the enterocytes and has been shown to exhibit circadian expression in the colon (Sladek et al., 2007). The NHE3 promoter contains a putative E-box and has been speculated to be a clock-controlled gene (Sladek et al., 2007). The intracellular sodium concentration is kept low by exchanging it with potassium ions by the Na+, K+ ATPase located on the basolateral side of the enterocytes. After uptake, all the peptides are hydrolyzed within enterocytes, and free amino acids are transported to the blood circulation by transporters present on the basolateral side.
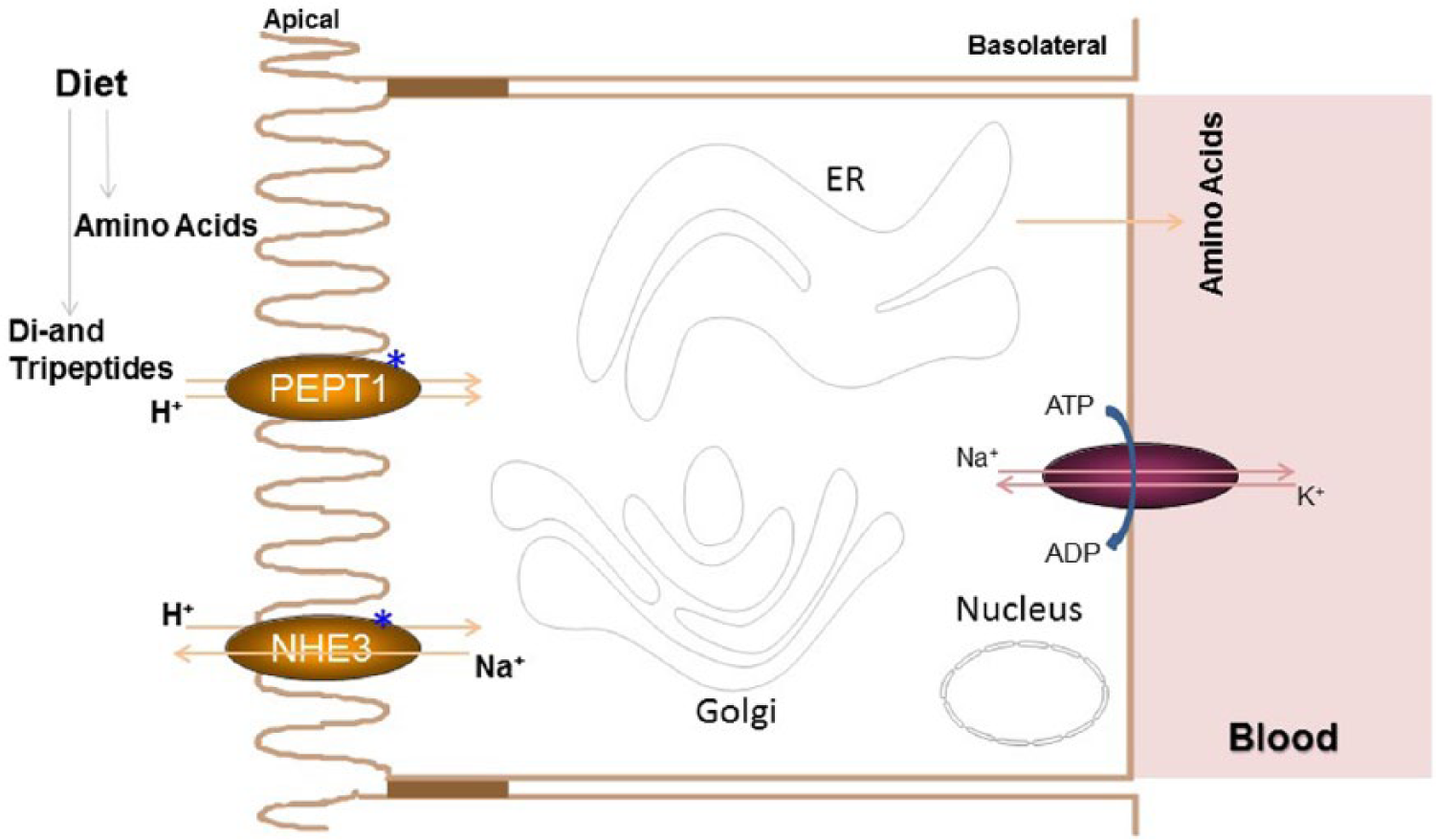
Protein absorption. Dietary proteins are digested in the intestinal lumen to amino acids, dipeptides, and tripeptides. Amino acids are taken up by several transporters (not shown). However, di- and tripeptides are taken up by peptide transporter 1 (PEPT1). This is a co-transporter that transports H+ and peptides into enterocytes. As the peptide transport is linked to the H+ gradient, peptide uptake is independent of lumenal concentrations. For efficient peptide uptake, intracellular H+ concentrations are kept low by another transporter, NHE3. This transporter exports H+ to the intestinal lumen in exchange for Na+. Intracellular Na+ levels are kept low in enterocytes by a basolateral Na+, K+-ATPase that exports Na+ and imports K+. Thus, these transporters act in concert to optimize peptide uptake by enterocytes. All the peptides are hydrolyzed in the cytosol of enterocytes, and amino acids are transported across the basolateral side into the blood circulation by amino acid transporters. Proteins known to exhibit rhythms are identified with *.
Circadian regulation of PEPT1 has been studied by several groups (Pan et al., 2002, 2003, 2004; Saito et al., 2008; Qandeel et al., 2009). Pan et al. examined the daily rhythm of the uptake of glycosyl-sarcosine by PEPT1 in the small intestinal loops and everted sacs of rats fed ad libitum and kept in a 12 h:12 h light-dark cycle; they observed that the highest initial rate of uptake was in the dark phase (Pan et al., 2002). Further, protein and mRNA levels of PEPT1 were highest at 2000 h and lowest at 0800 h (Pan et al., 2002; Qandeel et al., 2009). These studies suggest that PEPT1 expression changes over 24 h mainly due to changes in the transcription of the gene that appears to be highest in the dark phase. Next, they showed that prolonged fasting abolished daily rhythmic changes in PEPT1 protein levels, with levels at 0800 h as high as at 2000 h. These studies indicate that prolonged fasting increases PEPT1 expression and results in sustained high expression levels (Pan et al., 2003). Further, restricted feeding during the day shifted the expression pattern of PEPT1 (Pan et al., 2004). Now the highest expression of PEPT1 was observed in the light phase when food was provided. Thus, food availability is a significant determinant of peak PEPT1 expression.
Molecular studies of the circadian regulation of PEPT1 have been conducted by 2 groups, resulting in 2 different proposed mechanisms (Saito et al., 2008; Okamura et al., 2014). The basal expression of PEPT1 is dependent on Sp1 and Cdx2 transcription factors. Saito et al. (2008) observed that these transcription factors do not show variations over 24 h, suggesting that they might not be involved in the circadian regulation of PEPT1 (Saito et al., 2008). Another transcription factor that is known to regulate PEPT1 is PPARα, which shows modest circadian variation. PPARα deficiency in mice reduces the basal expression levels of PEPT1 but has no effect on daily changes in expression. Thus, these authors concluded that PPARα might not be involved in the circadian regulation of PEPT1. Next, they studied the circadian changes in the expression of clock genes in mice fed ad libitum or for a few hours during the day. They found that the daily rhythm in mRNA levels of albumin D site binding protein (DBP) is similar to that of PEPT1 mRNA. Further, they identified a DBP binding site in the distal promoter region of PEPT1 and concluded that DBP might contribute to rhythmic changes in the expression of PEPT1. However, the authors did not study the daily expression of PEPT1 in DBP-deficient mice.
Okamura et al. (2014) also studied the expression of PEPT1 in wild-type (WT) and PPARα null mice. In WT mice, PEPT1 mRNA levels were high in the daytime and during fasting conditions, probably secondary to increased availability of fatty acids, agonists of PPARα, and increased binding of PPARα to the peroxisome proliferator response elements (PPREs) in the PEPT1 promoter. However, in
Regulation of Glucose Uptake
Carbohydrates consist of mono- (glucose, fructose, and galactose), di- (lactose, sucrose), and polysaccharides (starch, cellulose). Polysaccharides are broken down to monosaccharides by the action of lumenal and brush border hydrolases. Enterocytes take up monosaccharides from the intestinal lumen involving 2 transporters found in the apical membrane. These monosaccharides are then exported out of the enterocytes from the serosal side into the blood circulation by a transporter present on the basolateral side of the enterocytes (Drozdowski and Thomson, 2006).
The first transporter present on the apical membranes of enterocytes is the sodium-dependent glucose transporter 1 (SGLT1) that is mainly involved in the uptake of glucose and galactose (Fig. 2). This co-transporter has 2 binding sites, one for Na+ and one for glucose/galactose. The transport of glucose/galactose is dependent on the concentration of Na+ across the apical membranes and is independent of glucose/galactose concentrations. The Na+ concentration is kept low within the enterocytes by an energy-dependent exchange of Na+ for K+ via a membrane bound Na+, K+ ATPase present on the basolateral side of the enterocytes. Thus, maintenance of low intracellular Na+ helps in facilitated uptake of glucose/galactose.
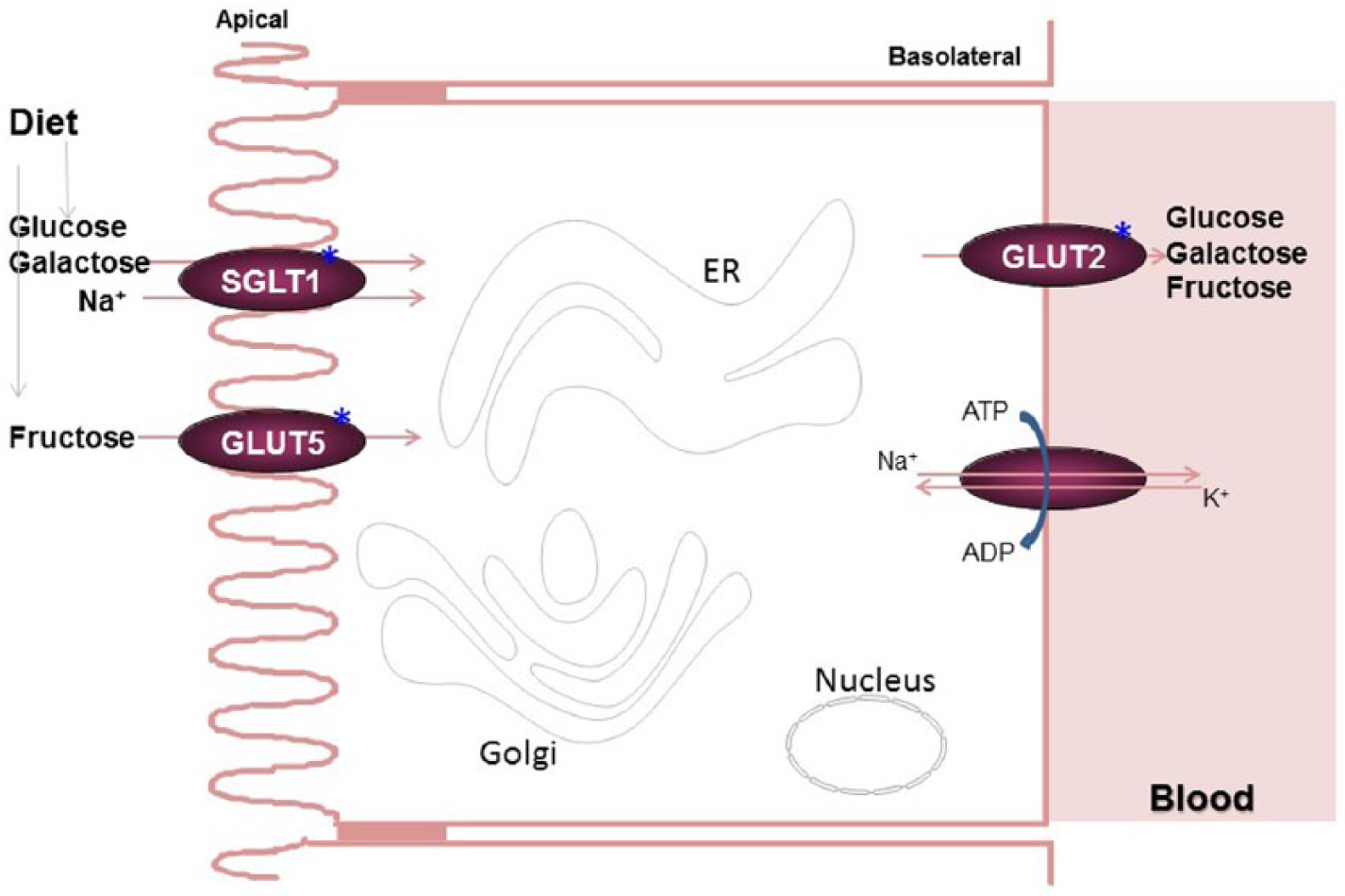
Carbohydrate absorption. A combination of lumenal and brush border enzymes hydrolyze dietary carbohydrates to glucose, galactose, and fructose. Glucose and galactose are taken up by enterocytes by SGLT1. It is a co-transporter that transports glucose along with Na+. Thus, glucose uptake is independent of its concentrations; instead, it is dependent on low intracellular Na+. Intracellular Na+ concentrations are kept low by the basolateral Na+, K+-ATPase that pumps Na+ out and brings K+ in. Glut5 is mainly involved in concentration-dependent fructose uptake into enterocytes. Glucose, galactose, and fructose are transported out of the cells from the basolateral side by another transporter, Glut2, which effluxes these sugars based on their concentration gradients. *Denotes proteins that show circadian changes.
The second transporter found on the apical membranes is the glucose transporter 5 (GLUT5). This low-affinity, high-capacity facilitative transporter is mainly involved in the uptake of fructose by enterocytes across the concentration gradient. Therefore, fructose is absorbed when there are high concentrations of this sugar in the lumen.
GLUT2, the third major transporter of sugars, is also a facilitative transporter present on the basolateral membranes of enterocytes and responsible for the transport of large amounts of all monosaccharides from the intracellular compartment to the outside toward the serosal side (Fig. 2). GLUT2 appears to be unique in that it can also be found on the apical side under certain conditions to facilitate the absorption of dietary sugars (Leturque et al., 2009; Jones et al., 2011). When lumenal concentrations of sugars are high and uptake by SGLT1 and GLUT5 are at the maximum, higher amounts of sugars can be absorbed by the translocation of GLUT2 to the apical membrane. Thus, GLUT2 functions to optimize sugar uptake depending on sugar availability.
Circadian rhythms in the absorption of sugars were observed even before knowledge of the molecular mechanisms of clock genes (Balakrishnan et al., 2012). Early studies showed that peak glucose absorption in rodents during the nocturnal period can be shifted by altering food availability (Fisher and Gardner, 1976). Subsequent studies showed that the daily expression of all 3 major hexose transporters is influenced more by food availability than by rhythms imposed by light and darkness (Pan et al., 2004; Balakrishnan et al., 2010; Iwashina et al., 2011).
Several investigators have studied the circadian regulation of these hexose transporters, as previously reviewed (Tavakkolizadeh et al., 2001; Balakrishnan et al., 2012). Rhoads and associates (1998) have shown that increases in SGLT1 are due to increased transcription at the time of food availability. Iwashina et al. (2011) studied changes over 24 h in the expression of the mRNA levels of the 3 hexose transporters in the jejunum of C57Bl6J mice fed ad libitum or for 8 h during the day. SGLT1, GLUT5, and GLUT2 mRNA levels showed peak expression at the onset of the dark in ad libitum fed mice. In mice fed for 8 h during the day, expression of these transporters was highest just before the anticipated time of food availability. Thus, in both ad libitum fed and food-entrained mice, the highest levels of the hexose transporter mRNAs are found prior to food availability, indicating that these genes are part of the food-anticipatory response.
These authors further provided evidence for the possible direct regulation of these genes by Bmal1 by studying its binding to the promoters of these genes by chromatin immunoprecipitation (Iwashina et al., 2011). Bmal1 was found to associate with the promoter regions of these genes during the light phase, earlier than the peak levels of mRNA found in the jejunum. This increased binding was concurrent with peak expression of Bmal1 mRNA. Thus, it is possible that when Bmal1 levels are high, it interacts with the promoter/enhancer sequences of the hexose transporters as it does with the Per2 promoter. In mice subjected to restricted feeding, Bmal1 interacted with the promoters of these transporters just before the anticipated availability of food instead of its usual time of interaction in ad libitum fed mice. These studies indicate that hexose transporters might be direct targets of Bmal1:Clock heterodimers and their regulation directly controlled by core clock genes. However, more detailed studies are needed to explain how circadian rhythms modulate SGLT1, GLUT2, and GLUT5 expression.
Intestinal Lipid Absorption
Predominant dietary lipids are triacylglycerols, phospholipids, and cholesterol. Several reviews have extensively discussed different steps involved in lipid absorption (Tso, 1985; Hussain et al., 1996; Hussain, 2000; Phan and Tso, 2001; Hussain et al., 2005; Mansbach and Gorelick, 2007; Hussain and Pan, 2009; Iqbal and Hussain, 2009; Pan and Hussain, 2012; Abumrad and Davidson, 2012). Here, we will briefly summarize these steps and then discuss what is known about the circadian regulation of the different steps involved in lipid absorption (Figs. 3 and 4).
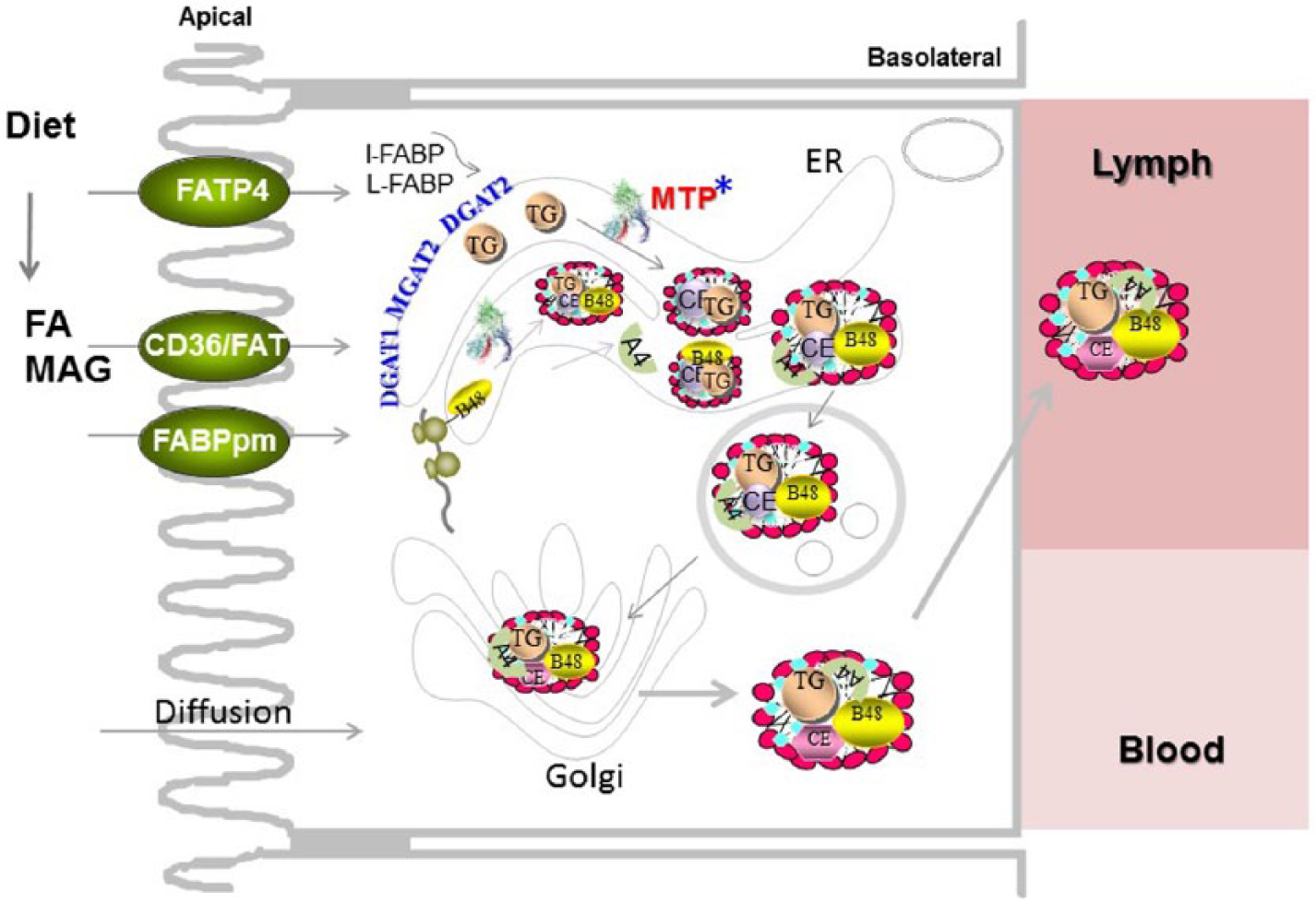
Fat absorption. Dietary triglyceride and phospholipids are broken down to free fatty acids, lysophospholipids, and monoacylglycerols in the intestinal lumen. These lipids enter enterocytes via diffusion and protein-mediated facilitated processes. Diffusion occurs across the concentration gradient. Protein-mediated processes can help in the uptake of fatty acids against the concentration gradient. Several transporters have been implicated in protein-mediated facilitated uptake of fatty acids, such as CD36/FAT, FATP4, and FABPpm. Inside the cell, fatty acids are transported to the endoplasmic reticulum, where they are used for the synthesis of triglycerides by several enzymes such as DGAT1, MGAT2, and DGAT2. Triglycerides are then added to newly synthesized apoB48 by microsomal triglyceride transfer protein (MTP). In addition, other apolipoproteins, such as apoAIV, associate with these particles. The newly synthesized lipoproteins are packaged in transport vesicles and delivered to the Golgi complex for further maturation, and they are secreted toward the basolateral side. They are concentrated in lacteals and delivered to the blood at the thoracic duct. *Proteins known to show daily changes in their expression.
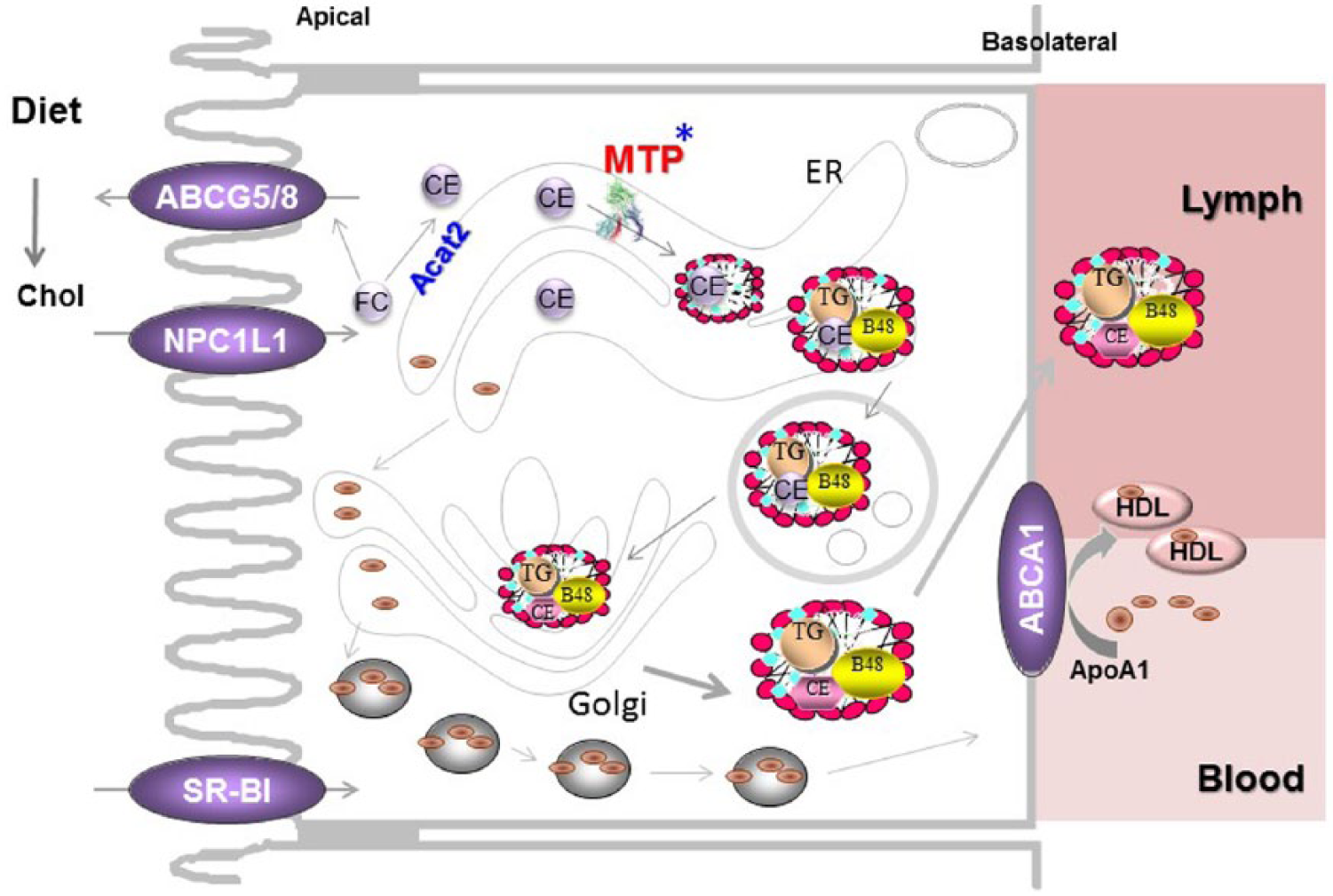
Cholesterol absorption. Diet contains free cholesterol and cholesterol esters. Cholesteryl esters are hydrolyzed in the intestinal lumen to free cholesterol and fatty acids. Free cholesterol is taken up by enterocytes via protein-mediated uptake involving NPC1L1. In the cells, free cholesterol has 2 fates. It can be exported back to the lumen via ABCG5 and ABCG8 or delivered to the endoplasmic reticulum for esterification by ACAT2. Both free and esterified cholesterol are added to chylomicrons and secreted with these particles. Besides chylomicrons, cholesterol can also be secreted via the high-density lipoprotein pathway. In this process, ABCA1 and apoAI play a significant role. *Proteins under circadian control.
Fatty Acid Absorption
Since lipids are water-insoluble, they are emulsified with bile salts in the intestinal lumen (Phan and Tso, 2001; Mu and Hoy, 2004; Hussain and Pan, 2014; Hussain, 2014a). In this process, hydrophobic lipids are partitioned into bile salt micelles, making them water miscible. The end products of intestinal triglyceride digestion are unesterified fatty acids and monoacylglycerols (Iqbal and Hussain, 2009). They are taken up by enterocytes via diffusion and protein-mediated transporters (Fig. 3; Iqbal and Hussain, 2009; Pan and Hussain, 2012; Abumrad and Davidson, 2012). Uptake via diffusion occurs when fatty acid concentrations are high in the lumen. FAT/CD36 and several other fatty acid transport proteins have been shown to be involved in protein-mediated uptake (Abumrad et al., 1998; Iqbal and Hussain, 2009; Pan and Hussain, 2012; Abumrad and Davidson, 2012). Thus, the major steps in the uptake of dietary fat from the intestinal lumen to enterocytes are emulsification with bile, hydrolysis by lipases, and uptake by transporters (Fig. 3).
After uptake, fatty acids are transported within the cells by fatty acid binding proteins. In the endoplasmic reticulum, fatty acids are used for the synthesis of triacylglycerols, phospholipids, and cholesterol esters. These lipids are then packaged into lipoproteins called chylomicrons (Hussain et al., 2005; Mansbach and Siddiqi, 2010). Chylomicrons are very large, spherical triacylglycerol-rich particles that also contain phospholipids and cholesterol. The surface of these particles is covered with a phospholipid monolayer, free cholesterol, and several apolipoproteins. Chylomicron assembly requires 2 proteins: apolipoprotein B (apoB) and microsomal triglyceride transfer protein (MTP). ApoB is a structural protein that is co-translationally lipidated by MTP into lipoproteins that are secreted by enterocytes (Hussain et al., 2003, 2012). Several studies indicate that intestinal lipid absorption via chylomicrons varies significantly over 24 h.
Lipid absorption by enterocytes is maximal at nighttime and lowest in daytime in rodents (Pan and Hussain, 2007, 2009; Pan et al., 2010; Pan et al., 2013a). We showed that both uptake and secretion of fatty acids by enterocytes show temporal changes over 24 h (Pan and Hussain, 2007, 2009). Further, we reported that MTP expression shows significant daily variation (Pan and Hussain, 2007, 2009; Pan et al., 2010). In rodents, MTP mRNA and protein levels are high in the nighttime and low in the daytime. However, MTP expression is significantly changed when mice are subjected to food entrainment. Mechanistic studies showed that
We have also studied the expression of other genes involved in lipid synthesis and absorption (Pan and Hussain, 2009). Most of the genes examined showed daily changes in their expression. These include
Disruptions in circadian rhythms, as in
Besides the genes directly involved in lipid absorption, other genes have also been shown to affect daily absorption of lipids. For example,
Cholesterol Absorption
The diet contains free cholesterol and cholesteryl esters. Cholesteryl esters are hydrolyzed to yield free cholesterol and fatty acids. Free cholesterol is taken up by NPC1L1. Net cholesterol absorption appears to involve 3 key transporters (Fig. 4). NPC1L1 is involved in cholesterol entry into enterocytes, whereas ABCG5/ABCG8 heterodimers export cholesterol. We showed that both the uptake and secretion of cholesterol by enterocytes vary significantly over 24 h (Pan and Hussain, 2009; Pan et al., 2013a). After uptake, cholesterol is transported to the endoplasmic reticulum, where it is converted to cholesteryl esters. Both free and esterified cholesterol are incorporated into chylomicrons and secreted with these particles.
While triglycerides are exclusively secreted with chylomicrons, phospholipids and cholesterol can also be absorbed via the high-density lipoprotein (HDL) pathway. In this pathway, ATP-binding cassette transport family A member 1 (ABCA1) plays a role. Plasma HDL levels do not show significant daily variation, although it is unknown if HDL secretion by enterocytes does so.
In hepatocytes, cholesterol is converted to bile acids or excreted as such to the bile. Bile synthesis exhibits circadian rhythmicity (Gnocchi et al., 2015). Recently, trans intestinal cholesterol excretion has also been shown to contribute to the elimination of cholesterol from the body (Jakulj et al., 2013). Very little is known about the circadian regulation of this pathway involved in cholesterol excretion from the body. In contrast, it is well known that cholesterol biosynthesis exhibits significant daily variation (Gnocchi et al., 2015).
Since cholesterol is not metabolized by peripheral cells, it is brought back to the liver from these cells by reverse cholesterol transport for excretion from the body. As a first step, peripheral cells release their cholesterol to plasma acceptors (apoAI, HDL, etc.) via ABCA1 and ABCG5 transporters in a process called cholesterol efflux (Baldan et al., 2006; Phillips, 2014). Defects in cholesterol efflux result in cellular cholesterol accumulation. Excess accumulation of cholesterol in macrophages results in foam cell formation and contributes to atherogenesis (Tabas, 2002). We showed that macrophages in
Future Studies/Perspectives
As summarized above, attempts are being made to understand molecular mechanisms involved in the circadian regulation of macronutrient absorption. We hope that this review may stimulate more research in this direction. Knowledge about the circadian regulation of nutrient absorption might allow us to better control intestinal disorders.
Work at night due to voluntary sleep curtailment or misalignment with the day/night cycle might override neuroendocrine entrainment and disrupt various tissue rhythms such as those of the intestine. This might result in several intestinal disturbances seen in night-shift workers and transcontinental travelers. Therefore, studies are needed in model systems to simulate these disruptions to fully understand why such changes cause intestinal discomfort.
While
Footnotes
Acknowledgements
This work was supported in part by a National Institutes of Health grant (DK-81879) and a VA Merit Award (BX001728) to M.M.H.
Abbreviations
apo, apolipoprotein; Bmal1, brain and muscle ARNT-like protein 1; Clock, circadian locomotor output cycles kaput; Cry, cryptochrome; HDL, high-density lipoprotein; MTP, microsomal triglyceride transfer protein; NPC1L1, Niemann-Pick C1-Like 1; Per, period; Rev-erbα, reverse erythroblastosis virus α; Rorα, retinoic acid receptor-related orphan receptor α; SHP, small heterodimer partner.
