Abstract
Fatty acids (FAs) can exert important physiological effects: for example, as precursors of eicosanoids, as signaling molecules, and, in particular, as parts of phospholipids, the major constituents of cell membranes. Animals can remodel cell membranes in terms of their FA composition in response to environmental conditions, and even endothermic mammals exhibit seasonal cycles in the FA makeup of membranes. Previous evidence pointed to the existence of both seasonal and daily cycles in phospholipid composition of human cell membranes. Therefore, we used a noninvasive method to collect human mucosa cells over 1 year in 20 healthy subjects, and we determined seasonal and daily rhythmicity of phospholipid FA content. Our results show that significant daily rhythms were detectable in 11 of 13 FAs and were largely synchronous among subjects. Also, these daily rhythms showed stable phase relationships between different FAs within subjects. In contrast, yearly rhythms in phospholipid FA content were statistically significant in only ~50% of subjects and were asynchronous between subjects. These results support the view that while human physiology is still dominated by geophysical sunrise and sunset, resulting in strong daily cycles, seasonal rhythms are less well defined, at least in Western societies. We suggest that the main physiological function underlying rhythms in cell membrane composition is the regulation of the activity of transmembrane proteins, such as ion pumps, which can be strongly affected by the fatty acyl chains of phospholipids in the surrounding membrane bilayer. Hence, among a multitude of other functions, cycles in membrane FA composition may be involved in generating the daily rhythm of metabolic rate. Rhythms in certain membrane FAs, namely polyunsaturated and monounsaturated FAs that are known to affect health, could be also involved in daily and seasonal rhythms of diseases and death.
Lipids are the most abundant organic components in human cells, with their frequency exceeding that of all other organic molecules combined (Freitas, 1999). A large fraction of these lipids are phospholipids, the major constituents of cellular membranes. Membranes can differ in their fatty acid composition, and these differences are known to affect transmembrane proteins, such as the Ca2+-ATPase, rhodopsin, Na+/K+-ATPase, or cytochrome c oxidase (reviewed in Arnold et al., 2015). Fatty acids (FAs) derived from phospholipids also play a role as precursors of prostaglandins, thromboxanes, and leukotrienes (Funk, 2001) and can affect gene expression by acting as ligands to activation factors (review in Bordoni et al., 2006). Not surprisingly then, FAs are involved in the regulation of diverse physiological processes including cardiovascular function, muscle contraction, and thermogenesis (Arnold et al., 2015). A particularly important class of FAs are polyunsaturated fatty acids (PUFAs). A multitude of effects on human health have been attributed to PUFAs, in particular to the highly unsaturated docosahexaenoic acid (DHA). These include positive effects of DHA on the cardiovascular system, anti-inflammatory effects, and beneficial effects for neural function (Lakatta and Sollot, 2002; Wall et al., 2010; Anderson and Taylor, 2012). Whereas mammals, including humans, can produce saturated and monounsaturated FAs (SFAs and MUFAs) they cannot synthesize PUFAs de novo. Thus, the 2 essential PUFAs, linoleic acid (LA) and α-linolenic acid (ALA), must be obtained from the diet. Mammals can, however, prolong and further desaturate these essential PUFAs to longer chained PUFAs and substantially remodel the phospholipid FA composition of membranes (Arnold et al., 2015).
The mechanisms by which PUFAs, or other FAs, affect health are only partly understood. The current evidence, however, points to an important role of FAs in membrane phospholipids, rather than free FAs. For example, prostaglandins, thromboxanes, and leukotrienes that regulate inflammation are all derived from FAs that are released from membranes by phospholipases (Wall et al., 2010; Anderson and Taylor, 2012). In fact, incorporation of certain PUFAs into cell membranes is required for anti-inflammatory, antiatherogenic effects (De Caterina, 2011). Further, dietary FAs with known beneficial effects on cardiovascular function such as DHA are incorporated in membrane phospholipids and are thought to directly affect cardiomyocyte plasma membrane ion channel activity, which may explain their antiarrhythmic properties (Anderson and Taylor, 2012). The importance of FAs as membrane components is further supported by the time course of DHA incorporation into cell membranes, which matches the appearance of antiarrhythmic effects after only 2 weeks of dietary intervention in humans (McLennan and Abeywardena, 2005). Yet another example is the beneficial effect of oleic acid (OA), which reduces blood pressure by altering membrane lipid structure, leading to hypotensive effects via G-protein-mediated signaling (Terés et al., 2008).
Some of the known effects of FAs involve physiological functions that undergo daily rhythms. A prime example is the cardiovascular system, which shows a strong daily rhythmicity of various parameters such as heart rate and blood pressure (reviewed by Guo and Stein, 2003). Endogenous daily rhythms in cardiovascular function are apparently partly responsible for the finding that pathophysiological events, such as myocardial ischemia/infarction or sudden cardiac death, show daily rhythms with a peak at ~0900 h (Guo and Stein, 2003; Scheer et al., 2010). There are not only daily rhythms but also seasonal peaks in death caused by various diseases, such as cardiovascular disease and heart failure as well as cancer and nephritis (overview in Nelson, 2004).
Given the known impact of FAs, especially of PUFAs, on various physiological functions and health, we hypothesized that humans may exhibit daily and/or seasonal (i.e., yearly) cycles in their cellular membrane FA composition. Surprisingly little is known on this subject. Dallmann et al. (2012) found that several free FAs in human plasma (as well as other metabolites) showed clear circadian rhythmicity, but membrane composition was not investigated in that study. We are aware of only a single investigation (Yoshida and Yoshida, 2004) that provided some evidence for a diurnal change in FA composition of human mucosa cells, but it was limited to data collection at 3 time points during the day. Similarly, there seems to be only a single study of seasonal changes in FA composition (of serum phospholipids) in humans (De Vriese et al., 2004). This study was restricted to the analysis of PUFAs only but provided evidence for seasonal changes in certain PUFAs, in particular arachidonic acid (AA) and DHA. These findings led us to re-address the question of daily and seasonal rhythms in cellular FA composition using a noninvasive method of mucosa cell sampling, which allowed frequent measurements, approximately 6 times per day and 12 times per year. In contrast to previous studies on this topic, we determined the full range of phospholipid FAs (SFA, MUFA, and PUFA). Further, we analyzed the results with a focus not only on the presence or absence of rhythms but also on the degree of synchrony of possible rhythms between subjects and on the phase relation between FA rhythms. This was because both synchronous rhythms and stable phase relations between FA cycles would point to the physiological importance of FA rhythmicity.
Material and Methods
Data Collection
Twenty healthy Caucasian volunteers (10 females, 10 males; age 39.6 ± 2.25 years, range 25-55 years) participated in this study. All subjects were informed about the purpose of the study and signed a form giving their consent to analyze their samples and to publish the anonymized data. This study was conducted according to the Declaration of Helsinki and was approved by the Ethics Committee of the University of Veterinary Medicine Vienna, Austria (license 12-09-97-2013).
Data were collected over a period of 1 year at monthly intervals. Participating subjects were instructed to collect data and samples at or close to the 15th of each month and to enter the exact dates and sampling times on a data sheet. On each measurement day, subjects collected samples at approximately 3-h intervals during their daily activity phase, but without altering their sleeping patterns, and noted the exact times of day. Some subjects did not adhere to the 3-h interval schedule but did protocol the times of data collection (e.g., Fig. 3a, subject 19). Immediately prior to or after mucosa collection, subjects measured their body temperature below the tongue. For this purpose, participants were provided with digital thermometers (Thermoval, Hartmann, Heidenheim, Germany, resolution 0.1 °C, precision ±0.1 °C) that give a temperature record only after readings are stable over time. Participants were asked not to eat for at least 30 min prior to sample collection. To obtain buccal mucosa samples, subjects were provided with plastic flasks (VWR International, Vienna, Austria) containing 25 mL of water for each time point. Participants were asked to clean their mouths thoroughly (with water but without toothpaste to avoid possible effects on lipid analysis), to then intensively rinse their mouths for 30 sec with 25 mL of water provided in laboratory flasks, and to subsequently spit the fluid into provided glass jars. After transfer of the fluid into plastic flasks with screw tops, samples were immediately frozen (at −18 °C) until transfer to the laboratory for further analysis (within 72 h).
Lipid Extraction and Analysis
Samples were thawed and centrifuged for 10 min at 1200 g and 4 °C. The supernatant was discarded. The pellet was resuspended in 1.5 mL of distilled water. After potential food remnants were allowed to sediment, the supernatant was removed, and lipids were extracted by adding 2 mL of chloroform-methanol (2:1) and mixing on a vortex for 4 min. Subsequently, 100 µL of NaCl solution (18%) was added, the mixture was vortex-mixed for 2 min and centrifuged at 2200 g for 10 min, and the chloroform-methanol phase was retrieved. This step was repeated after adding 2 mL of chloroform-methanol to the sample. The merged chloroform-methanol phases were concentrated and resuspended in 100 µL of chloroform-methanol, and phospholipids were separated by TLC (solvent: n-hexan/diethyl ether/formic acid 40:10:1). Subsequently, the phospholipid band was scraped off, 1 mL of methanol and 22 µL of H2SO4 were added, and the sample was boiled in a water bath for 30 min. Phospholipids were extracted by twice addition of 400 µL of hexane, vortex-mixing for 1 min, and centrifugation at 2200 g.
Phospholipid extracts were transesterified under nitrogen, extracted into hexane, and analyzed by GLC (Perkin Elmer Autosystem XL with autosampler and FID, Norwalk, CT) using a capillary column (HP INNOWax, 30 m × 0.25 mm, Hewlett Packard, Palo Alto, CA). Fatty acid methyl esters were identified by comparing retention times with those of standards (Sigma-Aldrich, St. Louis, MO), and peaks were integrated using the Turbochrom 4.1 Software (Perkin Elmer). For all tissue samples, we determined the proportions of the fatty acids listed in Table 1. For more details on the GLC analysis, see Valencak and Ruf (2013).
Names, abbreviations, and mean proportions (%) of fatty acids and fatty acid classes investigated; SEMs reflect the variation among subjects.
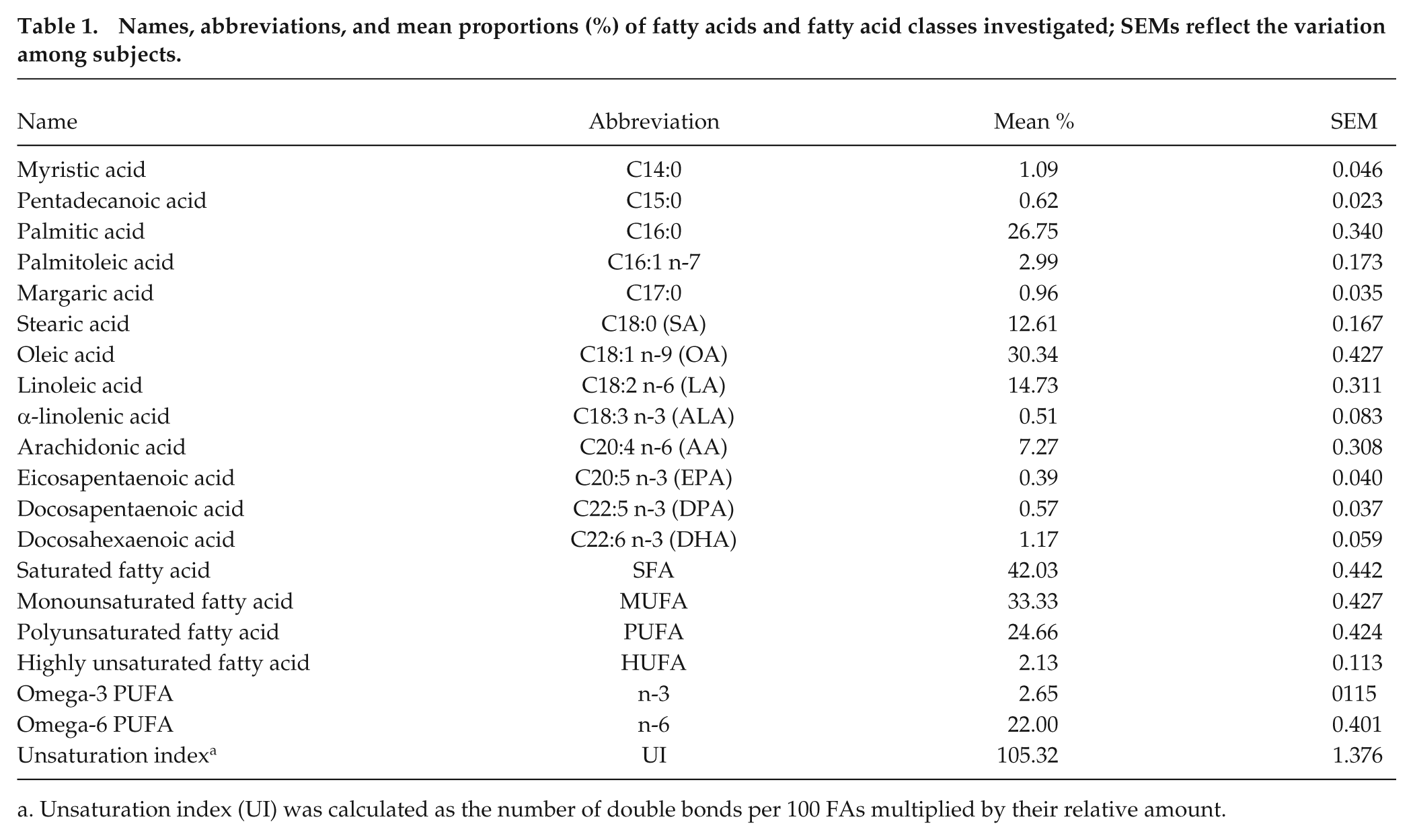
Unsaturation index (UI) was calculated as the number of double bonds per 100 FAs multiplied by their relative amount.
Statistical Analysis
Data were analyzed using the statistical package R 3.1.2 (R Core Team, 2014). The distributions of fatty acid proportions were characterized by leptokurtic shapes (i.e., an excess of both high and low values) or skewness to the right only. To achieve approximate normal distribution of residuals for subsequent parametric analyses, these data were transformed using
To determine possible seasonal and daily rhythms of FA proportions (as well as of body temperature, as a reference), we fitted the following model to data from each subject:
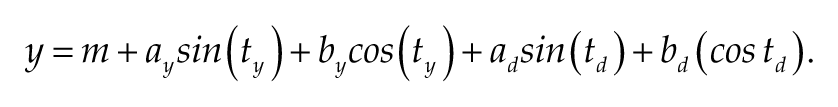
Here, y represents the predicted value at each time of the year (
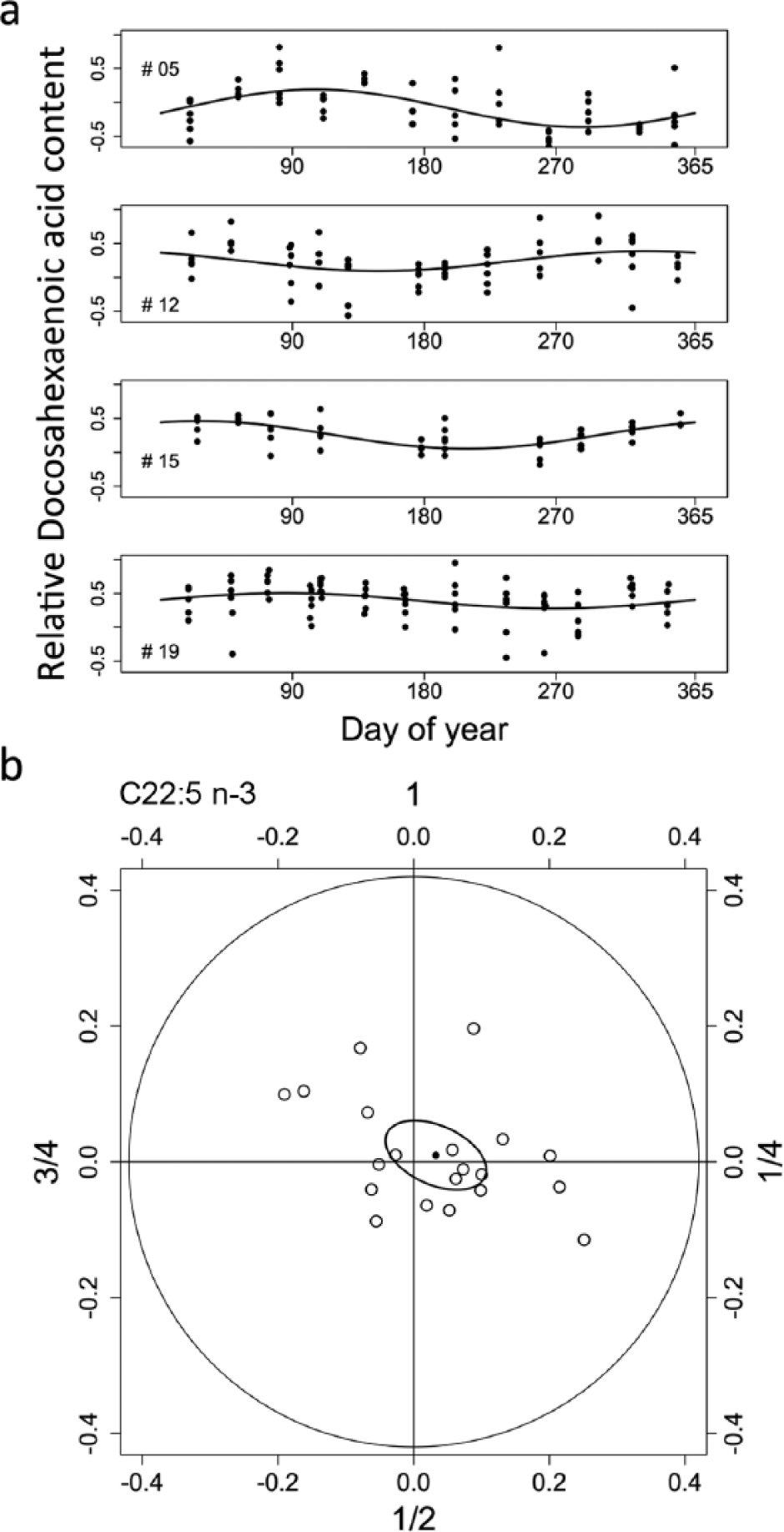
(a) Examples for yearly rhythms in docosahexaenoic acid (C22:6 n-3) in 4 subjects. Data points show relative contents, that is, transformations of original proportions (see Methods). Rhythms were significant (ANOVA; p < 0.01) in all 4 subjects, but peaks occurred at different times of the year. Missing days of data collection (subjects 12 and 15) were due to sickness or absence of participants. (b) Individual amplitudes and acrophases in 20 human subjects (white circles) as well as the population mean (black circle) and 95% confidence ellipse of the mean for yearly rhythms in docosahexaenoic acid (C22:6 n-3) proportions in human mucosa membrane phospholipids. As yearly rhythms had highly variable acrophases, the confidence ellipse overlaps the origin.
However, to see whether rhythms not only were detectable but had similar acrophases and amplitudes among subjects, we used the following second-order statistic (Batschelet, 1981): Individual rhythm amplitudes were plotted at their respective acrophases on a circular scale (e.g., Fig. 1b). We then determined population mean rhythm characteristics as well as 95% confidence ellipses (based on Hotelling’s T2), using equations given in Batschelet (1981). This procedure rejects the significance of rhythmic components with very low amplitudes and/or highly variable acrophases if they lead to a 95% confidence ellipse that overlaps the origin. When the significance of rhythms was not rejected, confidence intervals for mean amplitude and acrophase were determined from the intersection of the ellipse with a radius through the population mean and from tangents to the ellipse through the origin, respectively.
To illustrate average rhythms and residual errors, we plotted predictions computed from population mean amplitudes (
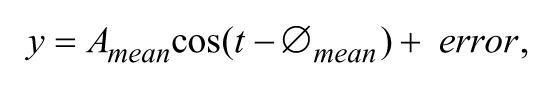
for the range t =
To see whether the obtained seasonal and daily rhythm amplitudes and acrophases were affected by age or sex, we used ANOVA. Given a sample size of 20 subjects, this analysis was limited to main effects only. ANOVA was also used to assess possible effects of age or sex on mean individual levels of FA proportions (i.e., means of untransformed data for each subject), which were normally distributed. Further, to see whether there were stable temporal relations between certain FA rhythms, we computed Pearson’s correlation coefficients for pairs of acrophases of FA cycles within subjects. In several cases, visual inspection indicated close correlations, with single data points, however, appearing to be obvious outliers. All of these apparent outliers were at phases very close (<0.5 units) to either 0 or 2π
To limit the multiplicity of p values, statistical analyses were restricted to the 13 FAs measured. However, we also present some descriptive statistics on FA classes (such as SFA, MUFA, PUFA). Among PUFAs we differentiated between n-3 and n-6 PUFAs, which are classified according to the location of the first double bond with reference to the terminal methyl group. If not stated otherwise, data are given as mean ± SEM.
Results
Mean Levels of FA Contents
The mean levels of FA contents (Table 1) were unaffected by sex, but proportions of several FAs changed with age. Pentadecanoic acid (C15:0) and margaric acid (C17:0) as well as palmitoleic acid (C16:1 n-7) all significantly decreased as age increased (all F3,16≥ 5.65, P≤ 0.030). In contrast, AA (C20:4 n-6) content increased with age (F3,16 = 5.11, p = 0.038), as did DPA (C22:5 n-3) content (F3,16 = 8.21, p = 0.011).
Yearly Rhythms
Yearly rhythms in all membrane phospholipid FAs determined, as well as body temperature (Tb), were significant in approximately half of the 20 subjects (median 10, range 7-15 depending on the FA tested). However, except for OA, yearly FA rhythms (as well as Tb) had vastly different acrophases. An example for these asynchronous seasonal rhythms, for the case of DHA, is shown in Figure 1. Evidence for a yearly rhythm that was significantly synchronous among subjects was detectable only for OA (C18:1 n-9). The amplitude of the OA rhythm was relatively small (0.087; 95% CI 0.016-0.16); its mean peak occurred at day of year 306, that is, in early November. Even for OA, the time of its acrophase was quite variable among subjects (95% CI 240-347).
Daily Rhythms
Significant daily rhythms, with a much higher degree of synchrony than yearly rhythms, were observed in Tb as well as in 11 of the 13 FAs determined (Figs. 2 and 3). The amplitudes and acrophases of these rhythms are given in Table 2. Even for those FAs (C20:4 n-6 and C20:5 n-3) that showed increased variability among acrophases, fits of a periodic function with a 24-h period were significant (ANOVA) in 12 and 13, respectively, of 20 subjects. Daily rhythms occurred in all FA subclasses: SFAs, MUFAs, and PUFAs (Figs. 2-4). The largest amplitude of daily rhythms was observed in stearic acid (SA; C18:0), which peaked around 0700 h (Figures 3 and 4). The most consistent rhythm, that is, the cycle with the least variance in both amplitude and acrophase, was that of palmitoleic acid (C16:1 n-7; see confidence intervals in Table 2).
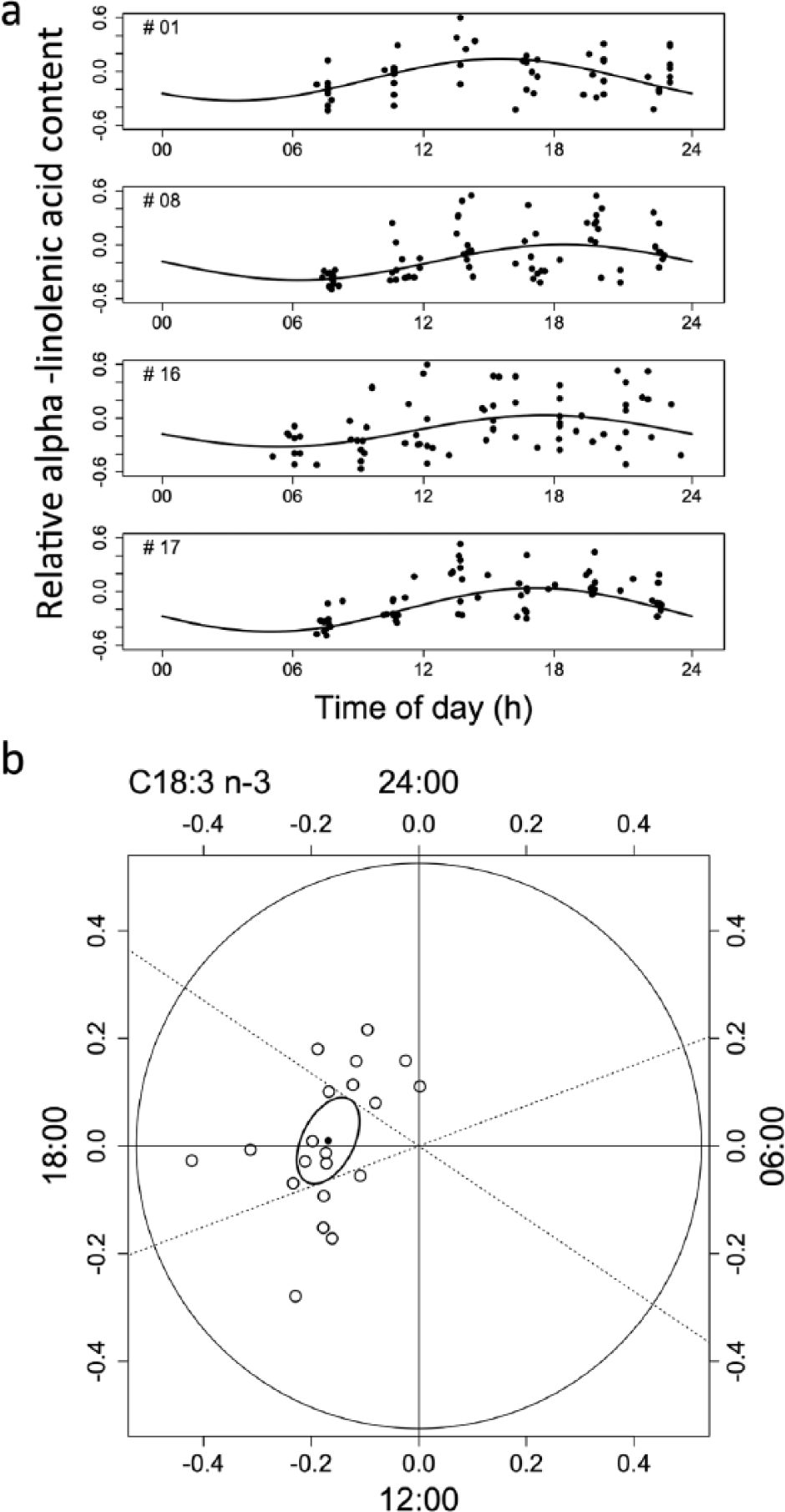
(a) Examples for daily rhythms in relative content (cf. Fig. 1) of a polyunsaturated fatty acid, α-linolenic acid (C18:3 n-3), in 4 subjects, with peaks occurring around 1800 h. (b) Individual amplitudes and acrophases (white circles) as well as the population mean (black circle) and 95% confidence ellipse of the mean for daily rhythms in α-linolenic acid (C18:3 n-3). Dashed lines show 95% confidence intervals of the mean phase of the peak.
Mean amplitudes (relative scale) and acrophases (time of day) as well as 95% confidence intervals of both parameters for daily rhythms in body temperature (Tb) and the proportion of FAs or FA classes in mucosa membrane phospholipids.
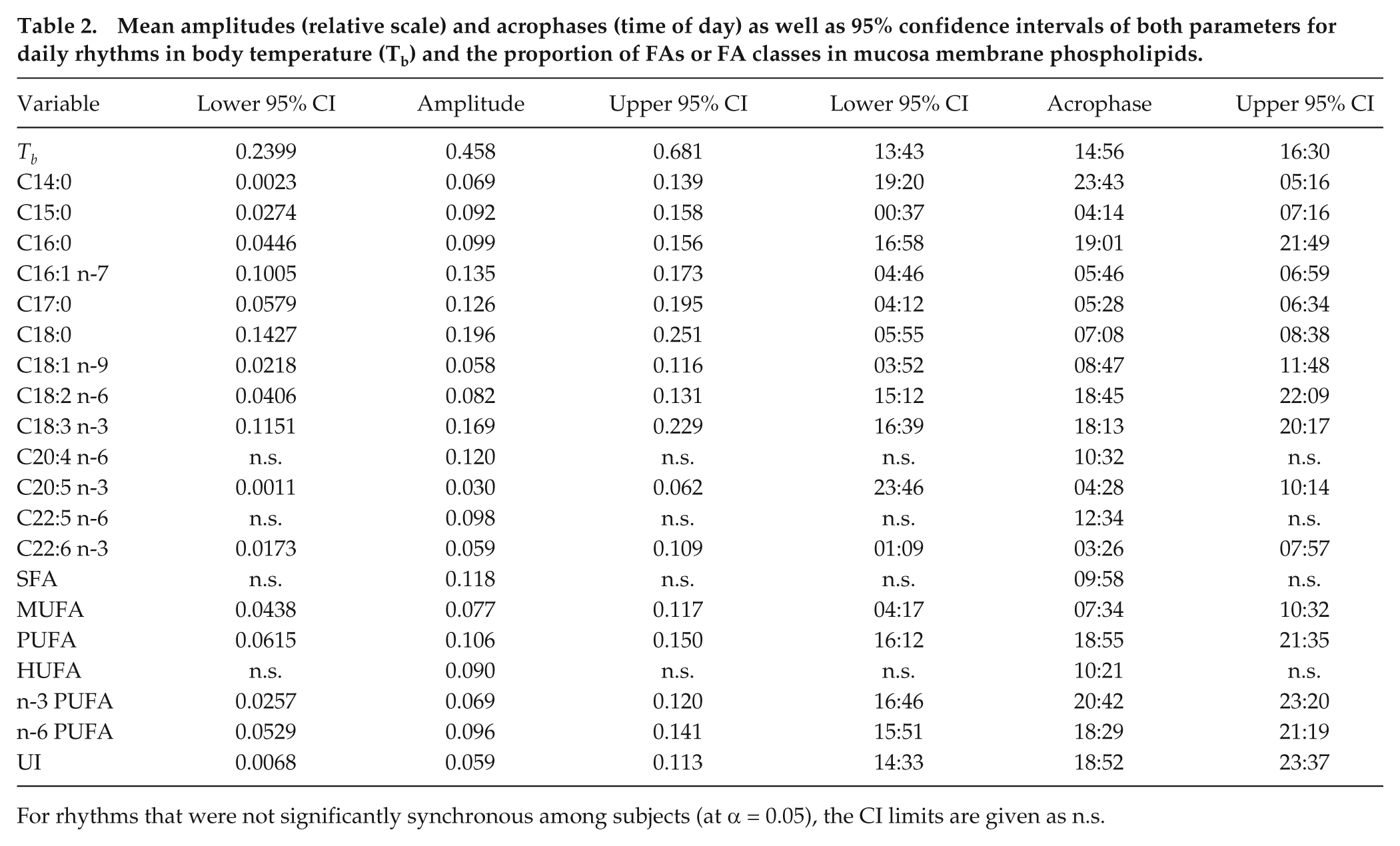
For rhythms that were not significantly synchronous among subjects (at α = 0.05), the CI limits are given as n.s.
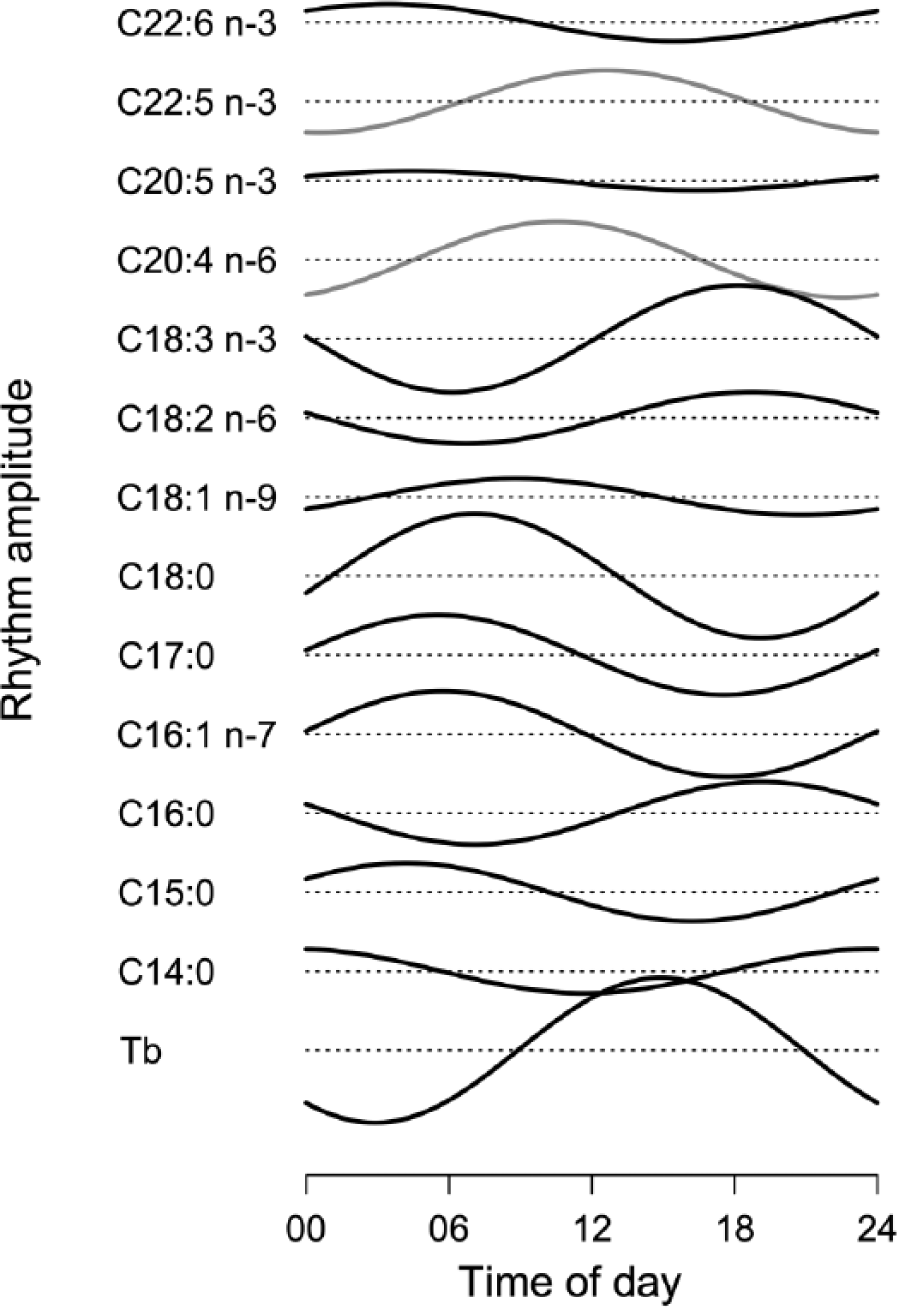
Daily rhythms of body temperature (Tb) (lowest panel) and of membrane phospholipid fatty acid composition. Curves were computed from population means of individual amplitudes and acrophases. Rhythms were significantly synchronous (at α = 0.05, black curves) in all fatty acids determined, except for AA and DPA (gray curves). Dashed lines show mean levels for each curve. To allow the comparison of amplitudes, all graphs of fatty acid rhythms are plotted on the same scale.
The timing of peaks in FA rhythms often tended to be characteristic for subjects, with stable phase relationships, as indicated by correlations between rhythm acrophases of different FAs. Figure 5 shows this for the example of the phase relation between in LA (C18:2 n-6) and ALA (C18:3 n-3). Notable correlations were found mainly between FAs of similar chain length (Table 3). There were no significant correlations, however, between acrophases or amplitudes of FA rhythms and daily (or yearly) rhythms in Tb.
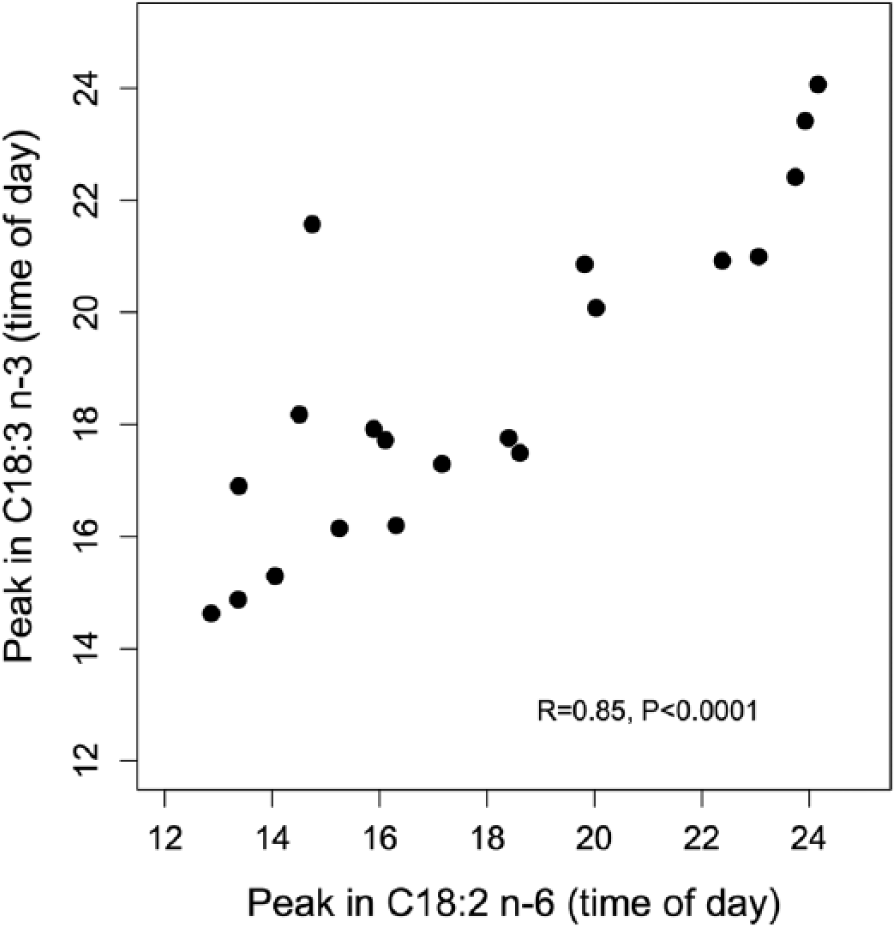
An example for stable phase angles between membrane FA rhythms: relationship between the times of daily peaks in linoleic acid (C18:2 n-6) and peaks in α-linolenic acid (C18:3 n-3) among the 20 subjects. Individuals with early peaks in linoleic acid also showed early peaks in α-linolenic acid, and vice versa.
Fatty acid pairs showing significant (p < 0.05) correlations (or tendencies for a correlation; 0.05 < p < 0.10) between the acrophases of daily rhythms.
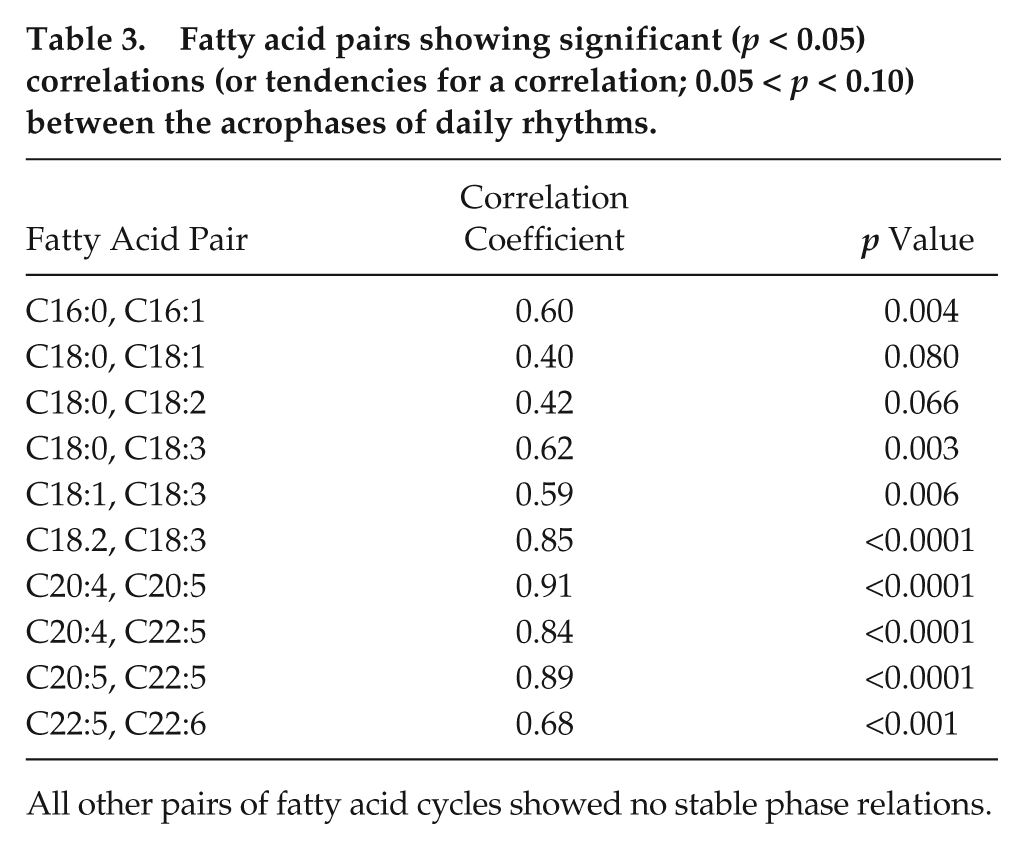
All other pairs of fatty acid cycles showed no stable phase relations.
Discussion
Daily and Seasonal Rhythms in Phospholipid Fatty Acid Composition
Our results show that all FAs determined in human mucosa phospholipids exhibit daily cycles in their relative concentrations, and the vast majority of these rhythms were significantly synchronous among subjects (Fig. 4). In contrast, significant yearly rhythms of phospholipid composition were detectable only in approximately 50% of the subjects and were largely asynchronous between subjects. This discrepancy in the strength of seasonal versus daily rhythms is fully in line with previous research on other cycles. Human behavior and physiology are still dominated by geophysical sunrise and sunset, resulting in strong daily cycles. However, seasonal patterns, such as in reproduction, death, and disease, are much more difficult to detect and have shown decreasing amplitudes over the last decades (Roenneberg and Aschoff, 1990; Roenneberg, 2004; Foster and Roenneberg, 2008). Decreased rhythmicity on the population level could be due to both reduced amplitude of cycles within individuals and loss of synchrony between individuals. The latter phenomenon, asynchronous seasonal cycles, has even been observed in tropical deer kept in a wildlife park in England (Loudon and Curlewis, 1988). Our results suggest that both factors, dampening and asynchrony, contributed to weak seasonal FA rhythms. A loss of seasonality in humans has been attributed to decreased exposure of modern human populations to environmental cues such as changes in photoperiod and ambient temperature (Roenneberg, 2004) or even to an increased genetically determined nonresponsiveness to such signals (Bronson, 2004).
The Origin of Phospholipid FA Cycles
One source of FAs in the synthesis of phospholipids is fats taken up with the diet. In the case of PUFAs, which humans and other vertebrates cannot synthetize de novo, diet is the only source (although a small fraction of longer chained FAs are derived from the elongation of the essential PUFAs LA and ALA). Not surprisingly, then, there is ample evidence in mammals and birds that diet affects phospholipid FA composition (Geiser, 1991; Andersson et al., 2002; Maillet and Weber, 2006; Maillet and Weber, 2007; Valencak and Ruf, 2011). The effect of dietary FA uptake on membrane phospholipid FA composition is much weaker than that on body fat stores but still statistically significant (Abbott et al., 2012). Thus, it is conceivable that the FA rhythms observed here were at least partly caused by cycles in dietary FA composition. There are strong daily rhythms in the general meal composition of humans, with lower proportions of fat and carbohydrate in the morning (e.g., De Castro, 2004). To our knowledge it is not known, however, whether daily cycles in food composition also include rhythms in the uptake of certain FAs. Following up on this question would require detailed analyses of the FA composition of meals. The same is true for seasonal rhythms in meal composition, which are also known to undergo slight variation, with a spring peak in carbohydrate and a fall or winter peak in fat intake (Kräuchi and Wirz-Justice, 1988; De Castro, 1991; Ma et al., 2005). In other mammals, in particular in herbivores, there is evidence for pronounced seasonal changes in the FA composition of diets (Frank, 1994; Mel’uchová et al., 2008; Ruf and Arnold, 2008; Popescu et al., 2011). If seasonal changes in dietary FA content also affect membrane composition in humans, changes in dietary habits, namely a loss of seasonality in food availability in modern societies, may well have contributed to a weakening of yearly FA cycles.
In animals, cellular membranes can be remodeled in response to environmental conditions. Especially in ectotherms, this process is evoked by cold exposure and can occur within hours (review in Hazel and Williams, 1990). Among mammals, there are also pronounced seasonal changes in membrane FA composition, in particular—but not exclusively—in heterothermic species that undergo daily torpor or hibernation (e.g., Geiser, 1991; Valencak et al., 2003; Arnold et al., 2011). In hibernators, membrane remodeling can occur while the animals do not take up any food but entirely rely on body energy reserves (Arnold et al., 2011). Hence, these alterations can be completely independent from the diet. The biochemical pathways underlying phospholipid remodeling are quite well understood, and involve specificity of enzymes for certain FAs or for FA groups, i.e. SFA versus PUFA (Yamashita et al., 1997; Henneberry et al., 2002). However, there are still many open questions in this area, particular with respect to the specificity of enzymes for individual FAs within the PUFA class (Arnold et al., 2015). Further, it seems entirely unknown which signal pathways could lead to daily or seasonal cycles in membrane phospholipid remodeling.
The Function of Phospholipid FA Cycles
We determined phospholipid FA composition in mucosa cells only—a single tissue. However, while tissues, for instance in heart, skeletal muscle, liver, or brain, may differ substantially in their phospholipid FA composition, the current evidence suggests that changes induced either by diet or by endogenous remodeling alter FA composition in the same direction in various tissues (e.g., Valencak et al., 2003; Arnold et al., 2011; Abbott et al., 2012). Hence, we assume that daily and seasonal rhythms as observed here in mucosa reflect similar cycles—although perhaps phase shifted and with different amplitudes—in various organs and tissues.
We can only speculate on the function of cycles in cell membrane FA composition. One explanation that immediately comes to mind is adaptation of membranes to changing tissue temperature, which in humans shows a clear rhythm, at least on a daily basis (Fig. 4). Membrane remodeling in response to changing temperature not only is common in ectotherms and endothermic heterotherms but also is a well-known phenomenon in bacterial membranes (Sinensky, 1974). This membrane remodeling has been called “homeoviscous adaptation” (Sinensky, 1974; Hazel and Williams, 1990; Cossins, 1994) because it was thought to serve as a mechanism to maintain optimal viscosity at different temperatures. However, the validity of this idea has been questioned, mainly because several observed membrane properties and membrane protein-interactions cannot be explained in terms of viscosity alone (Hazel, 1995; Lee, 2004). Still, even if the underlying function is more complex than just maintaining stable membrane fluidity, the fact that even in bacteria cell membrane composition is altered as temperature changes indicates that this response is an ancient mechanism that may have been retained even in endothermic mammals, such as humans.
We are aware of only one study that has shown a clear involvement of a membrane-derived phospholipids in circadian rhythms. In mice, the phosphatidylcholine PC(18:0/18:1) (i.e., a phospholipid containing fatty acyl chains derived from SA and OA) is part of a daily rhythm that serves to promote nighttime lipogenesis in the liver and subsequent fatty acid breakdown in skeletal muscles (Liu et al., 2013). PC(18:0/18:1) is transported from the liver to muscles, where it activates the nuclear receptor PPARα, which leads to lipid uptake and fatty acid oxidation. This role of PC(18:0/18:1), which it exerts only during the active phase of the animals, may be related to a daily rhythm in membrane remodeling. However, it seems unlikely that phospholipids acting as signaling molecules explain daily or seasonal cycles in membrane FA composition. In our view, a much more likely candidate for such a function of rhythmicity is the interaction between membrane FA and transmembrane proteins.
Numerous transmembrane proteins are affected by the FA composition of the surrounding bilayer, including rhodopsin, Na+/K+-ATPase, cytochrome c oxidase, and the sarcoplasmatic reticulum Ca2+-ATPase (SERCA) (Lee, 2004; Arnold et al., 2015). It seems that FAs in the bilayer influence conformational changes of transmembrane proteins via several pathways such as binding directly, altering membrane thickness, and exerting lateral pressure (Lee, 2004; Arnold et al., 2015). The activity of SERCA in muscle cells, for instance, can be increased several-fold by diet-induced changes of the membrane phospholipid composition (Swanson et al., 1989). Ca2+ pumping by SERCA is specifically enhanced by increased LA content and inhibited by high DHA content (Giroud et al., 2013). These differential effects of certain FAs point to the importance of maintaining stable phase relationships between rhythms of different FAs (Table 3), if the function of these cycles is the regulation of transmembrane proteins. Such a function seems plausible, because it should be adaptive to adjust, for instance, the activity of SERCA in cardiac myocytes to daily rhythms in heart rate. In fact, although the correlation between acrophases of LA and DHA was only a tendency (r = 0.38, p = 0.10), the mean nocturnal decrease in phospholipid LA content observed here (Fig. 4) largely mirrors daily rhythms in heart rate and was almost 180° out of phase with the cycle of DHA. These rhythmic changes in membrane PUFA content may well contribute to the daily cycle in pathological cardiovascular events and sudden death (Guo and Stein, 2003; Scheer et al., 2010).
Another FA that has beneficial health effects is OA. The presence of OA in the lipid bilayer of smooth muscle cells induces a decrease in the surface packing of phospholipid headgroups, which favors the docking of peripheral signaling proteins involved in the control of blood pressure (e.g., G proteins and protein kinase C) (Terés et al., 2008). In fact, this hypotensive action of OA is thought be the mechanism behind the significantly lowered incidence of cardiovascular disease in Mediterranean areas, which are characterized by a high dietary uptake of OA (Terés et al., 2008). Therefore, it is interesting that the OA cycle was the only yearly rhythm in our dataset that was significantly synchronous among subjects and peaked in early winter, that is, close to the time when there is also a maximum in yearly cycles of blood pressure (e.g., Goodwin et al., 2001). On a daily basis, OA peaked in late morning (Fig. 4), the time of the circadian surge in blood pressure in humans (Goodwin et al., 2001; Giles, 2006). Thus, both yearly and daily cycles in OA may attenuate surges in blood pressure, provided that changes in phospholipid composition as observed here also occur in other tissues, such as smooth muscle cells.
Cycles of mitochondrial membrane remodeling, in contrast, may adjust the activity of cytochrome c oxidase and other proteins to daily (or seasonal) rhythms in energy turnover and metabolic rate. However, the relationship between membrane FA cycles and metabolism could also be the inverse: Pumps such as Na+/K+-ATPase and Ca2+-ATPase significantly contribute to basal oxygen consumption (Rolfe and Brown, 1997). Consequently, rhythms in phospholipid FA composition surrounding these pumps may be one of the underlying mechanisms that cause daily rhythms in metabolic rate. If this was the case, it would be interesting to see whether metabolic rate and hence energy expenditure of humans can be altered by manipulating the FA composition of their diets.
The examples above represent, of course, only a handful of possible targets of cyclic membrane remodeling. The plasma membrane and membranes of the endoplasmatic reticulum, the Golgi apparatus, the nucleus, lysosomes, and mitochondria are associated with hundreds of proteins and involved in a multitude of functions, including ion conductivity, cell signaling, endocytosis and exocytosis, protein and lipid synthesis, and cellular respiration. Potentially, each of these functions may be affected by rhythmic alterations of membrane composition.
