Abstract
Various aspects of mammalian endocrine physiology show a time-of-day variation with a period of 24 h, which represents an adaptation to the daily environmental fluctuations resulting from the rotation of the earth. These 24-h rhythms in hormone abundance and consequently hormone function may rely on rhythmic signals produced by the master circadian clock, which resides in the suprachiasmatic nucleus and is thought to chiefly dictate the pattern of rest and activity in mammals in conjunction with the light/dark (LD) cycle. However, it is likely that clocks intrinsic to elements of the endocrine axes also contribute to the 24-h rhythms in hormone function. Here we review the evidence for rhythm generation in the endocrine master gland, the pituitary, and its physiological significance in the context of endocrine axes regulation and function.
We now know that in addition to the brain’s suprachiasmatic nucleus (SCN), most mammalian tissues exhibit circadian clock gene expression in a manner consistent with intrinsic circadian clock function. Indeed, for a number of these tissues, self-sustained circadian rhythm generation has been demonstrated by monitoring clock gene expression in tissue explants (Abe et al., 2002; Chu et al., 2013; Hughes et al., 2011; Yoo et al., 2004). Among the non-SCN tissues that showed the most robust rhythm-generating capacity is the pituitary, the “master gland” of the body (Abe et al., 2002; Yoo et al., 2004). It sits below the base of the brain and is anatomically connected with the mediobasal hypothalamus via a tissue stalk, also known as the infundibulum, and the median eminence, a circumventricular organ. The pituitary, or hypophysis, is functionally composed of distinct endocrine cell populations, which secrete hormones involved in the regulation of a wide range of physiological processes throughout the body, including growth, metabolism, sexual development, and reproduction (Figure 1; Nussey and Whitehead, 2001). This endocrine function is controlled, to a large extent, by releasing or release-inhibiting hormones produced by distinct neuronal populations in the hypothalamus and secreted into the portal vessels of the median eminence, which transport them to the hypophysis (Nussey and Whitehead, 2001). In response to these signals, pituitary hormones are then released into the circulation to reach their target sites, including organs that are by themselves humorally active, such as the thyroid gland, the adrenal gland, and the gonads (Nussey and Whitehead, 2001).
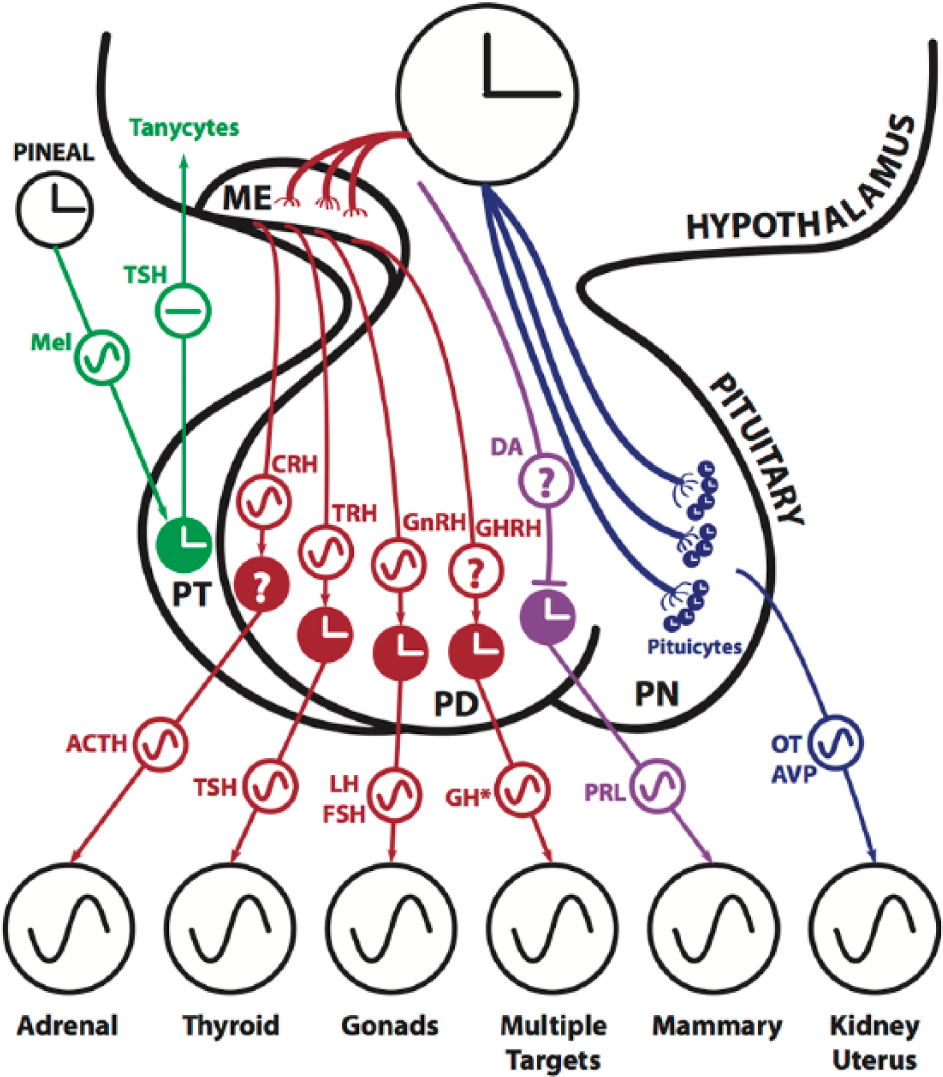
Circadian rhythmicity within the hypothalamic pituitary axes. Clock hands indicate intrinsic circadian clock function based on in vivo and/or cell culture evidence. ~, factor shows 24-h circadian fluctuations; *, GH shows pulsatile release or diurnal variation but lacks circadian rhythmicity. PRL production is suppressed by dopamine (DA) released from hypothalamic DA neurons. ACTH, adrenocorticotropic hormone; AVP, arginine vasopressin; CRH, corticotropin-releasing hormone; FSH, follicle-stimulating hormone; GH, growth hormone; GHRH, growth hormone–releasing hormone; GnRH, gonadotropin-releasing hormone; LH, luteinizing hormone; ME, median eminence; Mel, melatonin; OT, oxytocin; PD, pars distalis; PN, pars nervosa; PRL, prolactin; PT, pars tuberalis; TRH, thyrotropin-releasing hormone; TSH, thyroid-stimulating hormone.
In this review, we focus on the role of circadian rhythms within the pituitary gland and their involvement in the regulation of pituitary axes activity and corresponding cellular functions. We address the evidence for endogenous circadian clock function in the pituitary as a whole but also in individual hypophyseal endocrine cell types. Due to the vital role of the hypothalamus on the secretory activities of the pituitary gland, we also briefly discuss the circadian influence exerted on hypophyseal functions by hypothalamic neurons. We then address the evidence describing daily rhythms in pituitary hormone levels and specifically review the possible implications of intrinsic pituitary clock function within this context. As the pituitary represents the central relay station for hypothalamic neuroendocrine signals affecting processes with time-of-day–dependent activity, it seems plausible that in addition to the master SCN pacemaker, circadian clock function within the pituitary contributes to the normal execution of these processes. We assess the evidence in support of this notion. We also briefly touch on the evidence on the role of hypophyseal clock function in mediating photoperiodic responses in relevance to the timing of seasonal physiology. For in-depth reviews on the topic of seasonal timing and circannual rhythms, see Dardente et al. (2014), Follett (2014), and Wood and Loudon (2014).
Pituitary Anatomy and Axes
Based on developmental origins, the pituitary gland is divided into the adenohypophysis (anterior pituitary) and the neurohypophysis (posterior pituitary; Nussey and Whitehead, 2001). The adenohypophysis is further subdivided into 3 regions: the pars distalis (PD), the pars intermedia, and the pars tuberalis (Nussey and Whitehead, 2001).
The pars distalis harbors 5 types of endocrine cells: the somatotropes, corticotropes, thyrotropes, lactotropes, and gonadotropes, which secrete into the circulation growth hormone (GH), adrenocorticotropic hormone (ACTH), thyroid-stimulating hormone (TSH), prolactin (PRL), and luteinizing hormone (LH) along with follicle-stimulating hormone (FSH), both released by the gonadotropes (Nussey and Whitehead, 2001). Each of these pituitary hormones is an integral component of a specific neuroendocrine axis that exerts discrete physiological roles (Figure 1).
The pars intermedia separates the pars distalis from the neurohypophysis, but the physiological significance of this structure is not known in mammals (Tong and Pelletier, 1990). In humans, the pars intermedia regresses during childhood and becomes indistinct by adulthood (Nussey and Whitehead, 2001) and will therefore not be the focus of this review.
The pars tuberalis anatomically links the pars distalis to the hypothalamus. Studies have identified the presence of melatonin receptors in this region and have established strong links between the pars tuberalis and photoperiodic information processing. Therefore, the pars tuberalis seems to exert a key role in the regulation of seasonal rhythms in physiology and behavior (Dardente et al., 2014; Follett, 2014; Wood and Loudon, 2014).
The neurohypophysis, also known as the pars nervosa (PN; Figure 1), anatomically represents a direct continuation of the hypothalamus (Nussey and Whitehead, 2001). It largely consists of axons and terminals of neurons projecting to this location from the hypothalamus via median eminence and the infundibulum (Nussey and Whitehead, 2001). Two neuropeptides are released from the neurohypophyseal terminals into the circulation: arginine vasopressin (AVP, also known as antidiuretic hormone, ADH) and oxytocin (OT; Figure 1). A major target of the secreted AVP is the kidney, where the peptide regulates water conservation (Nussey and Whitehead, 2001). OT is known to stimulate milk ejection and uterine contractions in females and plays a role in ejaculation in males (Nussey and Whitehead, 2001).
Via the tropic cells of the pars distalis, the pituitary forms the nexus of 3 important endocrine axes—namely, the hypothalamic-pituitary-adrenal (HPA) axis, the hypothalamic-pituitary-thyroid (HPT) axis, and the hypothalamic-pituitary-gonadal (HPG) axis (Nussey and Whitehead, 2001). As such, it represents a critical relay station of outflowing (neuro)endocrine signals and is perfectly situated to receive circulating feedback. It is therefore a prime candidate to serve as a key integrator of circadian timing information into the endocrine system. We discuss the evidence for this with specific emphasis on the role of intrinsic clock function in the tropic cell classes of the pituitary. However, we first examine the evidence for rhythm generation in the pituitary as a whole.
Clock Gene Expression and Molecular Rhythm Generation in the Pituitary
The molecular mechanism of circadian timekeeping relies to a great extent on transcriptional-translational feedback loops. At its core, a transcription factor heterodimer formed by BMAL1 and CLOCK or NPAS2 drives transcription of the Period (Per) and Cryptochrome (Cry) genes via E-box promoter elements. Upon translation, PERs and CRYs first accumulate in the cytoplasm and then translocate back to the nucleus to repress their own transcription. As PER and CRY levels decrease, the cycle begins anew. In addition to this core feedback loop, auxiliary loops contribute to clock function and clock output transmission, including one involving the nuclear receptors Rev-erbα and β, which act as transcriptional suppressors at the Bmal1 promoter (Dibner et al., 2010; Mohawk et al., 2012).
Convincing evidence for intrinsic and self-sustaining circadian clock function in the pituitary has been obtained by culturing hypophyseal tissue explants from rodents that express luciferase under the control of clock gene promoters (Abe et al., 2002; Yoo et al., 2004). By monitoring bioluminescence emanating from the explants of the Per1-Luc rat, where luciferase expression is driven by the Period 1 (Per1) gene promoter, pituitary cultures were found to exhibit circadian rhythmicity that persisted for multiple days (Abe et al., 2002). Interestingly, separation of the anterior and posterior pituitary lobes revealed that both portions expressed similar rhythms when cultured in isolation, with Per1-luc expression peaking in the early subjective night (ZT14), in line with various extra-SCN brain regions that were simultaneously studied (Abe et al., 2002). By employing a PERIOD2::LUCIFERASE (PER2::LUC) fusion protein-based reporter mouse line, Yoo et al. (2004) confirmed these results by demonstrating that pituitary explants exhibited particularly robust circadian oscillations of PER2 and that this rhythm was not abolished by SCN lesion in vivo. Intrinsic rhythm generation in the pituitary was also reported for explants from Bmal1-Luc transgenic mice where luciferase expression is controlled by the promoter region of the essential clock component Bmal1 showing bioluminescence rhythms that lasted for +5 days (Chu et al., 2013). Pituitary explants from PER2::LUC mice were also examined by bioluminescence imaging employing an ultrasensitive CCD camera (Chu et al., 2013). Hourly imaging of whole pituitaries over multiple days revealed rhythmic photon emission throughout the gland, suggesting that most, if not all, pituitary cells express luciferase rhythmically and thus likely exhibit intrinsic circadian clock function (Figure 2). Interestingly, while bioluminescence rhythm amplitude and dampening rate were maintained in pituitary explants in the absence of vasoactive intestinal peptide type 2 receptors (Vipr2, also known as VPAC2R), the rhythm phase in such explants did not show a consistent relation to animal cull time (Hughes et al., 2011), which is in contrast to wild-type explants. Similar results were obtained with SCN explants from these knockout animals. Thus, VPAC2R may have a shared circadian functionality in both tissues reflected by the resistance to phase shifts triggered by external cues.
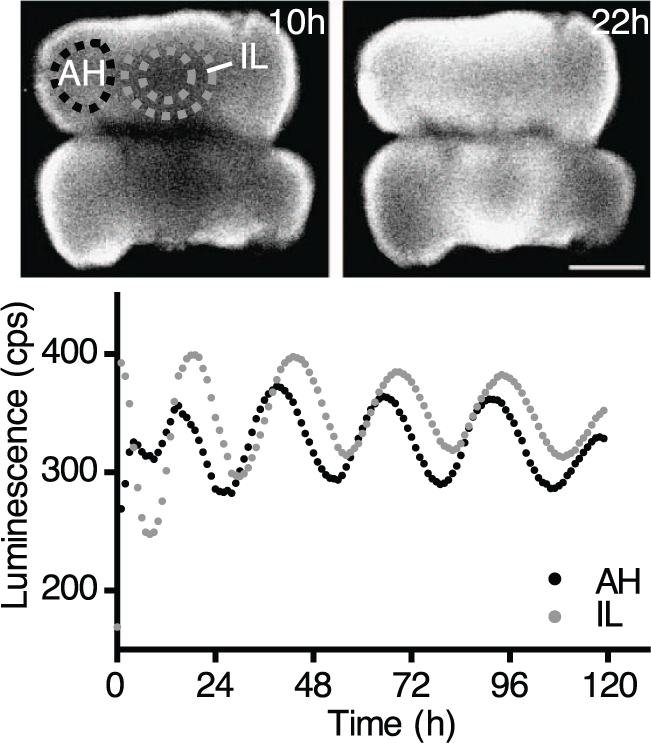
Bioluminescence imaging of circadian PER2-LUC rhythms in whole female pituitary explants from PER2::LUC reporter mice (adapted from Chu et al., 2013). Top, image of whole pituitaries at 10 and 22 h after explantation at ZT 4 to 5. Bottom, traces of bioluminescence averaged across the pituitary regions demarcated by dotted lines (upper image). Time of explantation is ZT 0. AH, adenohypophysis; IL, intermediate lobe.
To assess the extent of circadian control on the pituitary transcriptome, gene expression was examined on a genome-wide scale using gene chip technology with pituitary tissues harvested at 1-h time intervals across 2 days in constant darkness (DD) (Hughes et al., 2007). The authors identified more than 250 genes that oscillated with a 24-h period length by employing COSOPT and Fisher’s G-test at a false discovery rate of less than 5% (Hughes et al., 2007). Later on, using a more reliable and efficient algorithm called JTK_CYCLE also at a false discovery rate of 5%, the same group identified almost 400 genes in the pituitary that showed significant circadian oscillations in transcript abundance (Hughes et al., 2010). Another DNA microarray study based on a single time point (ZT 12) rather than a time course of tissue collection found roughly 250 genes that were differentially expressed in pituitaries from Bmal1–/– knockout versus wild-type mice (Guillaumond et al., 2012). Of these, however, only ~50 overlapped with the rhythmic gene set identified by the high-resolution time course analysis (Hughes et al., 2010). In addition to known clock genes, Guillaumond et al. (2012) also found, among the differentially expressed set, an overrepresentation of genes associated with gene ontology terms hormone metabolic process and response to hormone stimulus, implicating a role of BMAL1 and thus likely intrinsic clocks in the endocrine functions of the pituitary. In rhesus macaque, various components of the circadian clock, including Bmal1, Clock, Per1/2, and Cry1, were expressed in the pituitary glands of adult males. Using semi-quantitative reverse transcriptase polymerase chain reaction (RT-PCR), Bmal1 and Per2 expression were both found to be higher at 1300 h compared to 0100 h (Sitzmann et al., 2010).
Bur et al. (2010) employed quantitative RT-PCR (qPCR) to specifically study hypophyseal clock gene expression in response to changes in photoperiod or daily food access. Interestingly, the phases of peak abundances of clock gene transcripts were largely unaffected by photoperiodic changes when referenced to the time point of “lights off.” This is in line with the suggested mechanism of circadian clock involvement in the seasonal, pars tuberalis–mediated regulation of reproduction (see below). The authors further found that limiting food access to daily light period shifted the phase of the liver clock machinery as expected (Stokkan et al., 2001), but resulted in a profound dampening of rhythms in hypophyseal clock gene expression. These dampened rhythms exhibited only a slight shift in phase, indicating that the pituitary is not strongly responsive to a switch to daytime food access, which is in stark contrast to other peripheral oscillators tested (Damiola et al., 2000; Stokkan et al., 2001). The pituitary, therefore, seems to behave much like the SCN pacemaker here, which is generally unaffected by changes in feeding time (Damiola et al., 2000; Stokkan et al., 2001). Interestingly, adrenalectomy had no effect on the transcriptional response of hypophyseal clock genes to daytime feeding, which indicates that, in the pituitary, glucocorticoids do not hinder phase shifts in clock gene expression in response to restricted food access, contrary to the case of the liver clock (Le Minh et al., 2001). Hypophyseal clock gene expression appears to vary along the estrous cycle (Chu et al., 2013); qPCR analysis revealed that Rev-Erbα and Dbp transcripts show reductions in rhythm amplitude on the day of proestrus, suggesting that pituitary clock gene expression is influenced by cycle stage, as was reported for the uterus and the ovaries (Nakamura et al., 2010). In contrast to observations made in rodent and rhesus monkey pituitaries, clock gene transcripts did not show a 24-h variation in human postmortem pituitaries with the exception of Per1 when measured by qPCR (Wunderer et al., 2013). Clock gene protein products PER1, CRY1, and CLOCK also showed no daily variations in abundance. However, a time-of-day dependence on subcellular location of PER1 and CRY1 was observed, thus providing at least some indication of circadian clock function within the human pituitary at the molecular level.
Circadian Rhythms in the Adenohypophysis and Their Physiological Implications
Hypothalamic-Pituitary-Adrenal Axis
The HPA axis plays a critical role in the regulation of stress responses along with various other biological processes, including digestion, energy storage, mood, and emotions (Smith and Vale, 2006). Moreover, the HPA axis exhibits a characteristic daily rhythm: the synthesis and release of glucocorticoid (GC) from the adrenal cortex increase in anticipation of the daily wake phase. Consistently, the surge in circulating GC levels is oppositely phased in humans (morning) and laboratory rodents (evening) (Dallman et al., 1993). Since GCs facilitate energy utilization by stimulating gluconeogenesis in the liver and fat breakdown in adipose tissue, it is thought that the rise in circulating GCs prepares the organism for the impending active phase, which is associated with increased energy demands (Kalsbeek et al., 2012). For a review specifically addressing clock function in the adrenal gland, see Leliavski et al. (2015).
Corticotrophin-releasing hormone (CRH) is synthesized by neurons that reside in the medial parvocellular region of the hypothalamic paraventricular nucleus (PVN). The axons of these neurons project to the median eminence to release CRH into the portal vessels. CRH stimulates ACTH release from the corticotropes into the circulation. ACTH in turn targets the adrenal cortex to regulate the release of steroid hormones, including glucocorticoids and mineralocorticoids (Kalsbeek et al., 2012). The SCN has efferent projections that contact interneurons in and around the PVN, which are thought to transmit timing information to the CRH-containing neurons (Buijs et al., 1993). The SCN may convey this information to the HPA axis at least in part via AVP, as its release from the SCN has an inhibitory effect on the secretion of steroids from the adrenal cortex (Kalsbeek et al., 1992).
In rodents, Crh shows a clear circadian rhythm of messenger RNA (mRNA) abundance in the PVN, peaking around the time of lights-on and with a trough at lights-off (Girotti et al., 2009). This timing is compatible with the peptide’s role as a driver of ACTH rhythms, which peak much later during the day (~ZT 10), and this, in turn, is consistent with ACTH’s role as a regulator of adrenal GCs as their serum levels reach a maximum at around lights-off (Girotti et al., 2009; Oster et al., 2006). The rhythm in circulating ACTH could be solely driven by the SCN pacemaker via rhythmic CRH release, or it may also rely on intrinsic PVN clock function as clock genes appear to be rhythmically expressed in this hypothalamic nucleus (Girotti et al., 2009). The daily fluctuations in ACTH may also or alternatively depend on intrinsic clock function within the hypophyseal corticotropes. Interestingly, ablation of the SCN abolishes the time-of-day dependence of the corticosterone response to stress, while SCN lesion did not affect the ACTH response to stress (Sage et al., 2001). This suggests that an ACTH-independent mechanism of adrenal regulation may be involved in corticosterone secretion in addition to the classical HPA axis. One such possibility is direct neuronal innervation. In support of this hypothesis, transneuronal virus tracing from the adrenal reveals second-order labeling in neurons of the autonomic division of PVN and third-order labeling in the SCN itself (Buijs et al., 1999). In light of this observation, it was proposed that the autonomic nervous system (ANS) may be involved. Ishida and colleagues (2005) corroborated this suggestion by demonstrating that light can induce a plasma corticosterone surge via the SCN-sympathetic nervous system without direct activation of the HPA axis through ACTH stimulation. More recently, Wotus and colleagues (2013) further explored the contributions of humoral versus neuronal pathways in the daily rhythms of corticosterone levels. They employed a rat model based on a forced desynchrony protocol entailing exposure to an 11:11 LD cycle. Under these conditions, locomotor activity splits into 2 rhythmic components, with one following the 22-h period of the imposed LD cycle, while the other free-runs with a period of ~25 h (de la Iglesia et al., 2004). This behavioral split is associated with also a split of molecular rhythmicity within the SCN with the ventrolateral SCN (vlSCN) neurons following the period of the 22-h LD cycle while the dorsomedial SCN (dmSCN) neurons tick with a 25-h period corresponding to the free-running locomotor component. Upon transection of the thoracic splanchnic nerve that innervates the adrenal gland, these desynchronized rats showed a reduction and phase advancement of the peak of the corticosterone rhythm, which was dissociated from the rhythms of ACTH (Wotus et al., 2013). This suggests that the SCN also regulates plasma corticosterone independent of either sympathetic nervous system or ACTH-mediated input (Wotus et al., 2013). Therefore, a third putative mechanism, in addition to the previously described humoral and neuronal pathways, must be employed by the SCN in controlling the daily rise in plasma glucocorticoids. While the observed rhythm in circulating ACTH may solely depend on CRH input, it is conceivable that it also depends on clock function intrinsic to the corticotropes. However, a possible contribution of corticotropic clock function to HPA axis regulation has yet to be addressed experimentally.
Hypothalamic-Pituitary-Thyroid Axis
The tyrosine-based thyroid hormones (THs), triiodothyronine (T3) and thyroxine (T4), play an integral role in vertebrate growth, differentiation, development, and metabolism (Song et al., 2011). Consequently, circulating TH levels are highly regulated by the HPT axis, which is central to their function. At the top of the axis, a subset of PVN neurons, which have been shown to receive input from the hypothalamic arcuate nucleus (ARH) energy balance circuits as well as monoaminergic neurons of the brainstem, produce and release thyroid-releasing hormone (TRH) into the portal vessel system at the median eminence (Hollenberg, 2008). TRH then stimulates the pituitary thyrotropes to produce and secrete TSH, which in turn regulates the synthesis and secretion of THs by targeting the thyroid gland. Maintaining the intricate balance of plasma THs, which is arguably the prominent function of the HPT axis, requires sensitivity to T3 at both the levels of the pituitary and the hypothalamus, enabling necessary feedback regulation (Yen, 2001).
TSH, T3, and T4 all show daily variations in serum levels (Kalsbeek et al., 2000; Van Reeth et al., 1994), but the involvement of an endogenous circadian clock within the thyroid gland has been unclear. Recently, intrinsic clock function has been demonstrated for human thyrocytes (Philippe and Dibner, 2014). Primary human thyrocytes transfected with a Bmal1-luciferase lentiviral reporter showed high-amplitude circadian oscillations in reporter activity, and qPCR of clock gene transcripts further confirmed intrinsic clock function (Mannic et al., 2013). In the serum, higher abundance of TSH, T3, and T4 levels was observed during the light period in nocturnal rodents (Kalsbeek et al., 2000; Van Reeth et al., 1994), whereas TRH, TSH, and T3 showed a clear nocturnal peak during the late night/early morning in human subjects. Thermic ablation of the SCN resulted in a significant flattening of the diurnal T3 and T4 serum profiles under LD conditions (Kalsbeek et al., 2000). SCN lesion also resulted in a blunting of TSH rhythmicity, suggesting that the effects of the SCN pacemaker on the THs were at least in part mediated via pituitary thyrotropes (Kalsbeek et al., 2000). However, there was a more pronounced effect of SCN ablation on THs compared to TSH serum levels, which indicates that, as it appears to be the case for the HPA axis, the SCN also exerts its control on the thyroid via a neuronal pathway. Tracing of thyroid gland afferents by retrograde tracer injection revealed second-order labeling of TRH-containing cells in the PVN and third-order labeling in several hypothalamic cell groups, including the SCN (Kalsbeek et al., 2000). Thus, it is proposed that the SCN also has a dual-control mechanism on circulating THs by both the classical neuroendocrine HPA route as well as an ANS pathway. Interestingly, about two-thirds of the T3 found in cortical cell nuclei is produced locally (Crantz et al., 1982) by the conversion of T4 to T3 via 5′ iodothyronine deiodinase enzyme 2 (DIO2) found in astrocytes and hypothalamic tanycytes (Bianco et al., 2002), providing an additional pathway of HPT axis auto-regulation. Of note, T3 signaling in the hypothalamus seems to be critically involved in the seasonal regulation of reproduction (see below).
In addition to the proposed direct SCN control, circadian hypophyseal TSH secretion is regulated through feedback by T3 action on nuclear thyroid hormone receptors (TRs) found on pituitary thyrotropes (Richter et al., 2011). TR activation by T3 involves both coactivator binding as well as release of nuclear corepressor 1 (NCOR1), while recruitment of NCOR1 to the T3-TR complex leads to transcriptional repression (Astapova et al., 2011; Ortiga-Carvalho et al., 2005). To elucidate the mechanism that produces daily transcriptional rhythms of the TR-targeted gene Tshb, which is produced by pituitary thyrotropes, Aninye and colleagues (2014) studied TshB regulation in a thyrotrope cell line. Using chromatin immmunoprecipitation (ChIP), the group could show that NCOR1 enrichment did not occur at the TR binding site on the Tshb promoter but rather at a different site and this is unaffected by T3 treatment. Because it was shown previously that the core clock component REV-ERBα (also known as NR1D1) interacts with NCOR1 (You et al., 2010), the authors postulated that NR1D1, in conjunction with NCOR1, confers circadian control over Tshb expression by suppressing Tshb transcription. Following synchronization of the thyrotrope cells in culture, Tshb and Nr1d1 were shown to oscillate in antiphase, and ChIP analysis revealed that NR1D1 was enriched at the same Tshb promoter site as NCOR1, further supporting a suppressor role of NR1D1 at the Tshb gene locus (Aninye et al., 2014). Together, the data support the idea that an intrinsic thyrotrope clock drives rhythms in Tshb expression via the rhythmic suppressive action of NR1D1 on the Tshb promotor.
Hypothalamic-Pituitary-Gonadal Axis
The HPG axis is predominantly involved in sexual maturation and reproduction (Nussey and Whitehead, 2001). At the apex of the axis hierarchy sits gonadotropin-releasing hormone (GnRH), which is produced by a small number of neurons mostly residing in the medial septum and medial preoptic area (mPOA) (Skynner et al., 1999). The mPOA GnRH neurons send axons into the median eminence where GnRH is secreted into the portal vessel system and transported to the pituitary. There, GnRH binds to its cognate receptor, GnRHR, which is selectively expressed by gonadotropes. GnRH binding raises intracellular calcium levels, stimulating the release of LH and FSH from the pituitary gonadotropes into the circulation (Naor, 2009; Pawson and McNeilly, 2005).
An involvement of the circadian timing system in gonadotrope function has been suggested early on by pioneering work from Everett and Sawyer (1950), which provided the first line of evidence that a 24-h timing mechanism contributes to the generation of the preovulatory LH surge in rats. Treatment with central nervous system (CNS) depressants, such as barbiturate, in a 2-h window on the afternoon preceding the proestrus LH (and primary FSH) surge did not simply delay the surge until depressant action had subsided, as expected for a simple hourglass process, but surprisingly shifted the LH surge by exactly 1 day (Everett and Sawyer, 1950). This suggested that ovulation is triggered by a daily neuronal signal generated by the CNS and whose generation/propagation can be blocked by depressant treatment. Another key piece of evidence linking circadian rhythms and the reproductive axis was provided using ovariectomized (OVX) rats with implants yielding surge-permissive estradiol (E2). These animals exhibited a daily surge in LH serum levels, even in DD, strongly arguing that the LH surge is under tight circadian control, despite the fact that surging is suppressed for most of the estrous cycle due to the absence of surge-permissive levels of E2 on non-proestrous days (Christian et al., 2005; Legan et al., 1975). Critically, SCN lesions not only abolish circadian oscillations in locomotor activity but also prevent LH surge generation in E2-primed animals (Gray et al., 1978; Schwartz and Zimmerman, 1991). Moreover, transplantation of SCN grafts into SCN-lesioned females restored rhythms in locomotor activity but failed to rescue the LH surge, suggesting that SCN neurons need to establish appropriate synaptic connections to transmit the timing signal for LH surge generation (Meyer-Bernstein et al., 1999). The importance of the SCN within the reproductive axis was further corroborated with behaviorally “split” OVX + E2 hamsters housed under constant light conditions. These hamsters experience 2 bouts of locomotor activity that are 12 h apart, reflecting antiphase oscillations of the left and right sides of the bilateral SCN (de la Iglesia et al., 2000). Since both activity bouts are preceded by an LH surge, this suggests that surge timing is directly controlled by the SCN (de la Iglesia et al., 2003). The effects of genetic disruption of circadian clock function have also been examined in the context of reproductive physiology. ClockΔ19/Δ19 females, which show long-period circadian oscillations that frequently become arrhythmic in DD, exhibit irregular estrous cyclicity, high rates of embryonic abortion, and parturition failure and lack the proestrous LH surge (Miller et al., 2004). Bmal1–/– females, on the other hand, are completely infertile, which has been shown to result from embryo implantation failure due to deficient progesterone synthesis by the corpus luteum, into which the follicle transforms following oocyte release (Ratajczak et al., 2009). Interestingly, this reproductive deficit could be rescued by a single wild-type ovary, corroborating that implantation requires ovarian Bmal1 expression (Liu et al., 2014). For an in-depth review on the role of ovarian clocks in reproduction, see Sellix (2015). Together, the above-described results argue that the master circadian pacemaker is a crucial regulator of the LH surge and thus HPG axis function.
In addition to the SCN, clock proteins BMAL1 and PER2 have been found to oscillate with a 24-h period in GnRH neurons in vivo (Hickok and Tischkau, 2010), suggesting that an intrinsic circadian timer in GnRH neurons may also contribute to the central control of the LH surge. GnRH surge regulation has been suggested to involve SCN-born AVP and vasoactive intestinal peptide (VIP) as well as kisspeptin produced by neurons of the anteroventral periventricular nucleus (AVPV) and gonadotropin-inhibitory hormone (GnIH), which is expressed by a small group of cells in the dorsomedial hypothalamus. For a detailed account of the evidence, see Beltramo et al. (2014) and Williams and Kriegsfeld (2012).
In addition to clocks in the SCN, GnRH, and perhaps kisspeptin and GnIH neurons, local clocks in the peripheral components of the HPG axis, such as the gonadotropes of the adenohypophysis, may also be involved. IBuserelin, a GnRH analog, was shown to induce Per1 expression in immortalized gonadotropes in culture, and this induction relies on protein kinase C (PKC) and p42/44 mitogen-activated protein kinase (MAPK) signaling pathways (Olcese et al., 2006). It was further demonstrated that early growth receptor 1 (EGR-1) binding to the Per1 promotor mediates GnRH-stimulated transactivation (Resuehr et al., 2009). These data argue that the clock in hypophyseal gonadotropes is entrainable by GnRH. In further support of the existence of a functional clock within gonadotropes, the same authors showed that CLOCK/BMAL1 heterodimer complexes directly bind to E-box sequences located upstream of the GnRHR gene, to regulate its transcription (Resuehr et al., 2007). To test if gonadotrope clocks indeed contribute to in vivo reproductive physiology, mice that lacked BMAL1 selectively in gonadotropes (GBmal1KO) were generated using a Cre/loxP approach involving a “floxed” Bmal1 allele in conjunction with a GnRHR-Cre mouse line (Chu et al., 2013; Figure 3). Vaginal cytology in GBmal1KO females revealed that although the proportion of time spent in each estrous stage was normal, estrous cycle length variance was increased compared to wild-type littermates. To assess LH surge function in GBmal1KO mice, blood samples were collected for 5 to 7 consecutive days at ZT 11 as this time point corresponds to the time of the proestrous day when LH normally surges. GBmal1KO mice showed proestrous LH surge activation, but surge levels were observed to be 2- to 3-fold higher compared to wild-type females. Because the average LH baseline levels at all cycle stages were similarly elevated, the resulting proestrous LH surge-to-baseline ratio did not differ between the genotypes (i.e., GBmal1KO mice showed a general offset of the LH serum profile to elevated levels), but there was no selective change in the amplitude of the LH surge (Figure 3). Similarly, FSH serum concentrations were also elevated in the GBmal1KO females compared with wild-types. Other reproductive parameters, including puberty onset, time to impregnation, gestation time, or litter size, did not differ between the genotypes (Chu et al., 2013). Thus, it appears that local clock function in pituitary gonadotropes plays only a limited role in reproductive physiology, at least under the conditions tested. Furthermore, the general elevation of serum LH across all time points in the conditional knockout model is consistent with a rather noncircadian role of gonadotrope BMAL1 in LH physiology. Since Bmal1–/– mice, which are globally clock deficient, completely lack a proestrus LH surge (Chu et al., 2013), surge induction must involve circadian clocks elsewhere, possibly in the SCN.
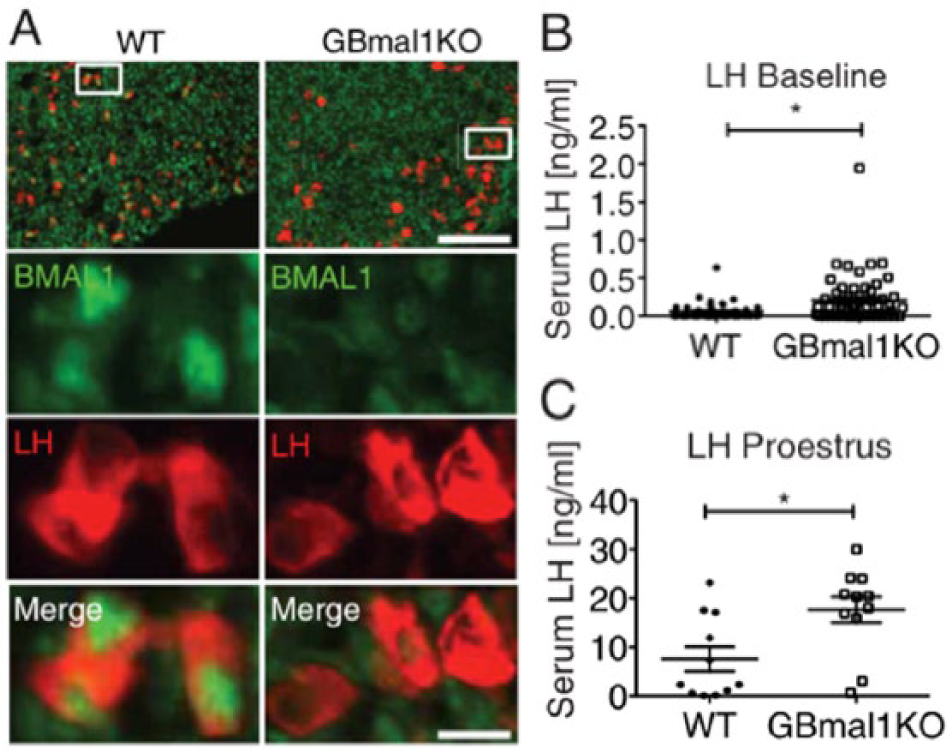
Loss of BMAL1 in gonadotrope cells and serum luteinizing hormone (LH) in GBmal1KO mice (adapted from Chu et al., 2013). (A) Double immunolabeling for BMAL1 and LH of anterior pituitary sections from wild-type (WT) and GBmal1KO females. Boxed areas are shown enlarged underneath. Baseline LH levels (B) and proestrus LH surge levels at ZT 11 (C). Dots indicate LH concentrations at individual, nonsurge (B) or surge (C) time points. Student t test: *P < .003 (B) or < .02 (C).
Somatotropes and Lactotropes
Growth hormone (GH), also known as somatotropin, is a peptide hormone secreted from the somatotropes in the adenohypophysis of mammals and plays a critical role in tissue growth and regeneration (Nussey and Whitehead, 2001). GH secretion is stimulated by GH-releasing hormone (GHRH) and inhibited by somatostatin (SS), both secreted from the hypothalamus into the median eminence and transported to the adenohypophysis via the portal system (Haus, 2007). In humans, serum GH levels exhibit a clear diurnal rhythm under LD conditions but are only secreted in a pulsatile pattern under DD conditions (Czeisler and Klerman, 1999). SS neurons have been localized to the SCN, and SS mRNA levels exhibit daily variations with higher levels during the day under both LD and DD conditions (Nishiwaki et al., 1995). Gh transcript abundance was shown to vary in the circadian range in a somatolactotrope cell line (Becquet et al., 2014). Expression of a dominant negative mutant form of BMAL1 led to an abrogation of rhythmicity in thyrotrophic embryonic factor (Tef), Contactin 1 (Cntn1), and Dio2 transcripts, confirming the presence of intrinsic clock function in this cell line (Becquet et al., 2014). Based on these data, it seems conceivable that circadian clocks contribute to GH rhythms under entrained LD conditions but are incapable of driving those rhythms in the absence of external (light) cues. Further research is clearly needed to determine if and how circadian clocks are involved in GH regulation.
Prolactin (PRL) is a protein hormone that is secreted from lactotropes, and while its physiological effects are pleiotrophic and species specific, it is prominently associated with lactation across species, stimulating milk production by the mammary gland (Grattan and Kokay, 2008). As opposed to other physiotropes, PRL does not appear to be under the control of a hypothalamus-born releasing hormone. Instead, PRL production is negatively regulated by dopamine (DA), which is released from DA neurons in the ARH and suppresses both Prl expression and lactotrope proliferation (Ben-Jonathan and Hnasko, 2001). In adult male rats, serum PRL levels showed a 24-h rhythmicity, peaking in the late afternoon (Dunn et al., 1972). Prl also showed daily transcriptional fluctuations in the rat pituitary somatolactotrope cell line (Becquet et al., 2014), and circadian Prl promotor activation was detected in anterior pituitary cells isolated from lactating rats (Leclerc and Boockfor, 2005). Furthermore, a putative E-box element was identified in the Prl promoter that interacted with BMAL1 and CLOCK in cultured cells, arguing that Prl expression is under direct control of an intrinsic lactotrope clock (Leclerc and Boockfor, 2005). Recently, however, using another somatolactotrope cell line, evidence was provided that the circadian Prl promotor activation may not rely on direct CLOCK/BMAL1 binding but instead depends on the interaction between pituitary-specific transcription factor 1 (PIT-1) and helicase-like transcription factor (HLTF; Guillaumond et al., 2011). These factors were shown to bind the Prl promoter via the putative, noncanonical E-box but exhibited no rhythmicity in their protein abundance. Intriguingly, HLTF associates with non-POU domain-containing octamer-binding protein (NONO) and splicing factor, proline- and glutamine-rich protein (SFPQ), both of which fluctuate in a circadian pattern. As their overexpression attenuates Prl promoter activity, these rhythmic factors are thought to negatively regulate Prl expression (Guillaumond et al., 2011). Interestingly, NONO has also been shown to interact with PER1, thereby antagonizing the circadian feedback suppression of PER proteins (Brown et al., 2005). While these data suggest that a local lactotrope clock has physiological implications, it has yet to be demonstrated in vivo.
Circadian Rhythms in the Neurohypophysis
The axons that extend into the neurohypophysis emanate from 2 specific hypothalamic areas: the PVN and the supraoptic nucleus (SON). The PVN and the SON contain magnocellular neurons that secrete either AVP or OT from their terminals into the neurohypophysis (Hatton, 1988). The plasma concentrations of AVP and OT have been reported to exhibit 24-h rhythmicity in rats under LD with both hormones peaking in the early dark phase (Noto et al., 1983; Windle et al., 1992). For AVP, a rhythm in protein abundance was also detected in the SON and PVN, with levels peaking at the end of the dark phase based on radioimmunoassays on protein extracts from tissue punches (Noto et al., 1983). In contrast, a more recent study found no evidence for Avp rhythms in LD in the rat PVN and the SON based on in situ hybridization (Jac et al., 2000). Interestingly, SCN-lesioned rhesus monkeys showed persistent OT rhythms in the cerebrospinal fluid even under constant light conditions, suggesting an extra-SCN circadian clock driving OT oscillations (Reppert et al., 1984).
While it appears that the neurohypophysis serves only as a secretory site for AVP and OT, where these peptides are released from nerve terminals of neurons residing in the hypothalamus, the pars nervosa is not a cell-free compartment. In fact, a specific glia-like cell type, the pituicytes, is found to be distributed throughout the neurohypophysis (Hatton, 1988). Under basal conditions, these pituicytes surround axons and axon terminals within the neurohypophysis, thereby impeding AVP and OT secretion into the circulation (Hatton, 1988). However, under conditions requiring elevated circulating levels of these hormones, pituicytes can retract from the neural processes, thus allowing for increased hormone secretion into the perivascular space, where the hormones enter the circulation via fenestrated capillaries (Hatton, 1988). Key to this regulation is the pituicytes’ ability to reversibly change their morphology from a flat amorphous or fusiform shape under basal conditions to a more stellate, astrocyte-like shape under conditions requiring increased hormone release (Hatton, 1988; Rosso et al., 2004). Interestingly, both AVP and OT stimulate stellate shape reversal in pituicytes by activating pituicytic vasopressin receptor 1a (Rosso et al., 2004). This cell type also displays a distinct diurnal pattern in nuclear volume (Lach et al., 1980). Together, these findings suggest that pituicytes may contribute to the generation of the daily rhythm in circulating AVP and OT. Whether exerting this function requires a circadian timer intrinsic to pituicytes, however, remains to be explored. The finding that posterior lobe explants from Per-luc1 rats also showed bioluminescence rhythms after separation from the anterior lobe, however, argues that pituicytes are indeed clock cells (Abe et al., 2002).
Circadian Rhythms in the Pars Tuberalis and their Role in Seasonal Physiology
As part of the adenohypophysis, the pars tuberalis (PT) represents the dorsal extent of the pars distalis (PD) enwrapping the infundibulum and ultimately abutting the median eminence ventrally (Nussey and Whitehead, 2001). The physiological significance of the PT remained largely obscure until the discovery of melatonin receptor expression in PT cells (Morgan et al., 1994). In mammals, melatonin is secreted from the pineal gland, and its circulating levels are highly elevated during the night (Brzezinski, 1997). As the daily profile of circulating levels of this “darkness” hormone directly depends on day length, it is an ideal candidate to translate changes in photoperiodicity into physiological responses. Indeed, removal of the pineal gland renders seasonal animals unresponsive to photoperiodic changes (Goldman, 2001). Binding of melatonin to the melatonin 1 (MT1) receptors in the PT selectively suppresses the expression of TSH in the PT, while thyrotropes within the PD remain unresponsive, as they are not involved in seasonal timing (Dardente et al., 2003). These TSH-expressing PT cells do not express T3 receptors, and hence they are not regulated via the classical HPT axis–mediated feedback loop (Bockmann et al., 1997). Instead, the PT-TSH cells appear to be pivotal to the seasonal regulation of reproduction.
When Soay sheep are exposed to a long photoperiod (i.e., an LD cycle of 16 h/8 h), melatonin-responsive PT-TSH cells upregulate their expression of TSH, which in turn acts on tanycytes, a glia cell type that is exclusively found in the ventral lining of the third ventricle at the level of the mediobasal hypothalamus. There, TSH induces the expression of DIO2, which—as already alluded to—converts T4 into the TR-ligand T3 (Bechtold et al., 2010; Hanon et al., 2008; Wood and Loudon, 2014). Recently, critical insights were gained into the mechanism by which PT-TSH cells, in conjunction with the circadian clock, translate changes in day length into changes in reproductive physiology (Dardente et al., 2010; Masumoto et al., 2010). It was revealed that photoperiodic induction of TSH in sheep and melatonin-producing mice is mediated by the de-repression of the transcriptional activator eyes absent 3 homolog (EYA3). Eya3 is acutely induced in the PT upon photoperiod lengthening beyond 12 h of light, hinting at an upstream role in the photoperiodic response pathway (Nakao et al., 2008). Analysis of the Eya3 promoter region identified canonical E-box elements as well as a D-box element that binds TEF (Dardente et al., 2010; Masumoto et al., 2010). Consistently, Eya3 expression shows circadian rhythmicity, invariably peaking at around 12 h after lights-off (Dardente et al., 2010). If, however, the animal is still exposed to darkness at the time of Eya3 peak expression, as is the case under short-day conditions, Eya3 expression is suppressed, thereby preventing transcriptional activation of Tshb. Once the photoperiod has sufficiently lengthened, however, the CLOCK/BMAL1 transcriptional activation of Eya3 can take place, which in turn leads to the induction of TSHb and triggering a physiological response (i.e., a change in the activity of the reproductive axis), which is thought to be mediated by the action of tanycyte-born T3 (Dardente, 2012). Together, these findings constitute a breakthrough in seasonal physiological research as they provide a molecular mechanism for the photoperiod-mediated switch in reproductive state. The discovery also demonstrated the critical role of the circadian clock in enabling the measurement of day length. To determine whether the photoperiodic switch depends on intrinsic circadian clock function within the PT-TSHb cells, as is suggested by the data, or by additional clocks elsewhere, elimination of clock function in a tissue-selective manner (i.e., limited to PT-TSHb cells) would be required.
Outlook
Ample evidence demonstrates that the major cell types and subdivisions of the pituitary harbor circadian clocks. Given the circadian variations observed in the biological processes in which the pituitary hormones are involved, it seems likely that pituitary clocks play a role in these processes. While cell culture–based studies support this notion, selective in vivo manipulation of clock function within distinct pituitary cell populations is needed to address the physiological significance of these clocks. In the case of the gonadotropes, such an approach has already been taken (Chu et al., 2013). However, selective elimination of clock function from this cell population does not appear to affect the circadian aspects of reproductive axis physiology. GBmal1KO mice showed only an overall increase in circulating LH levels but did not exhibit a change in amplitude of the LH/FSH surges (Figure 3). Consistently, these mice exhibited only mild phenotypes in other aspects of reproductive physiology (Chu et al., 2013). The absence of a substantial phenotypic change indicates that intrinsic gonadotrope clock function contributes little to reproductive physiology. However, this outcome could alternatively be due to developmental compensation for the genetic manipulation. To address this issue, a possible next step would be to disrupt local clock function in the adult animal by employing a transgene expressing a Cre-estrogen receptor fusion protein (CreER), which allows for temporal control of recombinase activity. The lack of a phenotype could possibly also result from redundancy within the endocrine axis (i.e., a profound effect may only be seen upon clock disruption in more than one axis element). Accordingly, one could attempt to additionally eliminate BMAL1 from GnRH neurons. Finally, given the significant effects of light on the circadian clock, GBmal1KO mice should be tested for HPG axis deficits in DD. Such strategies may as well be needed to uncover the in vivo roles of intrinsic clocks in other hypophyseal cell types, which have yet to be addressed directly. In conclusion, circadian clocks may have evolved to tick at every level of the hypothalamo-pituitary axes to ensure proper alignment of bodily physiology with the solar day, even under challenging health or environmental conditions.
Footnotes
Acknowledgements
This work was supported by an operating grant of the Canadian Institutes of Health Research (CIHR); K.-F.S. holds a New Investigator Award from CIHR.
