Abstract
A gradual adaptation to a shifted light-dark (LD) cycle is a key element of the circadian clock system and believed to be controlled by the central circadian pacemaker, the suprachiasmatic nucleus (SCN). Endocrine factors have a strong influence on the regulation of the circadian clock network and alter acute photic responses of the SCN clock. In females, endocrine function depends on the stage of the ovarian cycle. So far, however, little is known about the effect of the estrous cycle on behavioral and molecular responses to shifts in the LD rhythm. Based on this, we investigated whether estrous state affects the kinetics of phase shift during jetlag in behavior, physiology, and molecular clock rhythms in the SCN and in peripheral tissues. Female mice exposed to an advanced LD phase at proestrous or metestrous showed different phase-shift kinetics, with proestrous females displaying accelerated adaptation in behavior and physiology. Constant darkness release experiments suggest that these fast phase shifts do not reflect resetting of the SCN pacemaker. Explant experiments on SCN, adrenal gland, and uterus confirmed this finding with proestrous females showing significantly faster clock phase shifts in peripheral tissues compared with the SCN. Together, these findings provide strong evidence for an accelerated adaptation of proestrous compared with metestrous females to new LD conditions that is accompanied by rapid behavioral, physiological, and molecular rhythm resetting. Not only do these findings open up a new avenue to understand the effect of estrous cycle on the clock network under changing environmental conditions but also imply a greater susceptibility in proestrous females.
Entrainment of the mammalian circadian system to the light-dark (LD) cycle is regulated via a central pacemaker, the hypothalamic suprachiasmatic nucleus (SCN). This structure synchronizes behavioral, physiological, and molecular rhythms to external time by coordinating peripheral clocks found in all tissues and cells (Pilorz et al., 2018). At the molecular level, circadian clocks are based on transcriptional-translational feedback loops (TTFLs) involving several rhythmically expressed clock genes and their proteins (Buhr and Takahashi, 2013; Partch et al., 2014; Reppert, 2000; Shearman et al., 2000). The circadian clock network, however, can be disturbed in response to shift work or jetlag that commonly impose irregular schedules of sleep-wake patterns and mealtimes. Such irregularities of circadian rhythms have been linked to health disorders such as metabolic and cardiovascular dysfunctions, increased risks of breast cancer (Evans and Davidson, 2013), and reduced fertility caused by miscarriage (Gamble et al., 2013; Mahoney, 2010; Mong et al., 2011).
Animal studies use two principal approaches to illustrate the consequences of circadian rhythm disturbances. On one hand, genetic mutations or deletions in core clock genes such as Bmal1 or Clock have revealed disturbances in metabolism and cardiovascular function (Marcheva et al., 2010; Rudic et al., 2004; Shimba et al., 2005; Turek, 2005; Yamaguchi et al., 2013). On the other hand, similar effects have been shown after manipulation of circadian rhythms through misalignment with the external LD cycle mimicking jetlag or shiftwork (Baron and Reid, 2014; West et al., 2017). Interestingly, such external misalignment also affects internal circadian clock coupling (Xie et al., 2019). Yamazaki et al. (2000) demonstrated different rates of re-entrainment of the central and peripheral oscillators under simulated jetlag conditions. Moreover, there are indications that at the level of single-tissue clocks, some clock genes recover their rhythms faster than others suggesting a certain degree of plasticity within the TTFL itself (Kiessling et al., 2010; Stokkan et al., 2001; Yamazaki et al., 2000). Because of the difference in the kinetics of phase shifts between the SCN and peripheral oscillators, the whole organism is in a state of internal as well as external desynchrony for several days (Yamaguchi et al., 2013) that is reflected by the gradual re-entrainment of locomotor activity.
Already under stably entrained conditions, the phase relationship between the central clock and clocks in reproductive and non-reproductive organs varies across the estrous cycle (Nakamura et al., 2005, 2010). For instance, while reproductive organs such as the uterus show advanced Per1 and Per2 expression during proestrous (PE) as well as after estradiol (E2) treatment, non-reproductive organs such as liver and kidney show a phase delay of their clocks (Nakamura et al., 2005). This tissue specificity in the effect of estrous cycle stages on circadian clock gene expression has been explained by fluctuating estrogen concentrations and differentially distributed estrogen receptors—ERα and ERβ—in different target tissues and cell types (Azcoitia et al., 1999; Chakraborty et al., 2005; Hileman et al., 1999; Kruijver and Swaab, 2002; Naugle et al., 2014; Shughrue et al., 1997; Vida et al., 2008; Chung et al., 2007). Hence, the physiological effect of estrogens may depend on whether a cell expresses ERα, ERβ, or both. ER distribution is also species dependent. In the SCN of female mice, ERs are mainly expressed in the shell with ERβ being predominantly present in non-Arginine vasopressin expressing neurons, where ERα is modestly expressed (Vida et al., 2008; Gundlah et al., 2000; Hileman et al., 1999; Kuiper et al., 1997; Shughrue et al., 1997a, 1997b). In humans, the female SCN exhibits stronger nuclear ERα expression compared with males whereas ERβ expression does not differ between both sexes (Kruijver et al., 2003). Neonatal SCN neurons in rats, however, show equal presence of ERα and ERβ (Guo et al., 2001; Su et al., 2001). Differences in ER distribution and expression in different species, brain regions, and developmental ages may account for structural, functional, and developmental changes (Royston et al., 2016; Su et al., 2001). On the basis of these findings, it can be inferred that estrogens may have a direct effect on the SCN and may influence non-photic information due to ER’s presence in the shell. There is indeed a large body of evidence showing that gonadal estrogens affect behavioral circadian rhythms (Blattner and Mahoney, 2013) leading to a modulation of diurnal activity patterns along the estrous cycle and in response to estrogen treatment (Brockman et al., 2011; Gentry and Wade, 1976; Morin et al., 1977; Nakamura et al., 2010; Ogawa et al., 2003; Wollnik and Turek, 1988) with phase advances and higher activity during the (high-estrogen) PE compared with metestrous (ME) and diestrous (DE) stages (Albers et al., 1981; Albers, 1981; Morin et al., 1977; Wollnik and Turek, 1988). In line with this, estrogen replacement in ovariectomized female rodents advances the onset of activity compared with non-treated animals (Albers, 1981). Similar effects of estrogen administration on circadian rhythms of locomotor activity can also be achieved in aging and old female rodents (Yin et al., 2019). Hence, the SCN acts as a prominent target of estrogenic effects (Shughrue et al., 1997a, 1997b; Vida et al., 2008). This also suggests that estrogens may modulate responses of the master clock to changes in the environment. In addition to direct effects of estrogens on the SCN, they may influence circadian function at other brain nuclei such as medial preoptic area (MPO), anteroventral paraventricular nucleus (AVPV), lateral geniculate nucleus (IGL), dorsal and medial raphe, as well as arcuate nucleus (ARC) (Horvath et al., 1998; Merchenthaler et al., 2004; Sheng et al., 2004; Shughrue et al., 1997a, 1997b) indicating that estrogen may act through multiple neuronal regions to regulate circadian function.
Given that the phase relationship between central and peripheral clocks as well as activity patterns vary across the estrous cycle, it is likely that the female circadian system may differentially respond to external perturbation depending on estrous cycle stage. Our current knowledge about the characteristics of photic entrainment under jetlag conditions derives mostly from studies on male animals. However, little is known about clock re-entrainment and its association with estrous cycle stage in females. In the present project, we sought to investigate whether an acute phase change of the LD cycle causes different kinetics of phase shifting of central and peripheral clocks during PE versus ME. For this, we quantified running-wheel activity in response to a 6-h phase advance of the LD cycle. Females in PE exhibited accelerated phase shifts in locomotor activity compared with females in ME, which behaved more similar to males. Intriguingly, this accelerated phase shift in PE females seems not to correlate with fast resetting of the SCN. These findings are corroborated by experiments with PER2::LUC explants of SCN and peripheral tissues, suggesting an estrous cycle dependent phase shifting that may involve a crosstalk between central and peripheral processes regulating behavioral and physiological adaptation in female mice at PE. The SCN appears not to play a dominant role in this process.
Materials and Methods
Animals
All animal experiments were conducted in accordance with the German law of animal welfare and ethically assessed and licensed by the Ministry of Agriculture, Environment and Rural Areas (MELUR) of the state of Schleswig-Holstein. The 2- to 3-month-old female and male wild-type and heterozygous PER2::LUC reporter mice (B6.129S6-Per2tm1Jt/J) on a C57BL/6J background (Yoo et al., 2004) were used throughout the study. Mice in all experimental procedures were singly housed under a 12-h:12-h LD cycle with white light (250 lux when measured at the bottom of the cage), complete darkness and food and water provided ad libitum. In addition, three experimental groups, consisting of PE, ME, and males, were kept separately in three opaque cabinets with frequent air exchange and a constant ambient temperature of 22 °C to prevent mutual interference of behavior and to ensure consistency of the estrogen cycle and behavior within the same group.
Wheel-running Activity
Wheel-running activity of PE, ME females, and males was recorded using ClockLab software (Actimetrics). Wheel data were collected in 1-min bins. Females were subjected to 6-h LD phase advances at ME and PE, respectively. The phase advance was induced in both females and males by advancing the onset of dark phase by 6 h resulting in shortening of light phase on the first day of phase shift. Both estrous stages, ME and PE, were induced by adding a palmful of bedding from a cage of a single male mouse containing urine and feces “male bedding” to the female’s cage (Suppl. Fig. S1) (Jemiolo et al., 1986). This non-invasive method was used to prevent estrous irregularities or pseudopregnancy caused by repeated vaginal stimulation (Cora et al., 2015; Goldman et al., 2007). To confirm the success of PE or ME induction by addition of male bedding prior to the experiment, female mice at ZT4 (zeitgeber) were examined for estrous stage in two consecutive cycles using vaginal smears starting on the fourth and sixth day after addition of male bedding. A sterile inoculating loop (VWR International) coated with isotonic NaCl 0.9% saline solution was carefully inserted into the vagina and the smear was transferred into a small (100 µL) NaCl 0.9% saline drop that was placed on a glass slide. Subsequently, vaginal smears were stained as per standard procedures (Cora et al., 2015). The smears were inspected for cell types under a microscope (Nikon) using 10× magnification. PE was identified by a presence of small round nucleated cells with a uniform shape whereas ME was characterized by a mixture of neutrophils, cornified, and few nucleated cells (Cora et al., 2015). In addition, mice were scored for having regular estrous cycles if they exhibited 4- to 5-day cycles. If mice showed more than 3 days of ME and DE or more than 2 days PE or estrous (E) or did not follow the standard order of the estrous states, they were evaluated as having irregular estrous cycles. After confirming the expected estrous stages and the regularity of the estrous cycle, the females were reintroduced to the males’ bedding to induce PE or ME for an experiment on phase shifting. On the day of the 6-h phase shift, the estrogen stage was confirmed by taking a single vaginal smear at ZT4 before the 6-h phase shift. The chosen time (ZT4) is suitable for the assessment of estrous stages (Nelson et al., 1982). Females which did not show the expected estrous stage were excluded from the experiment. Since the stage of the estrous cycle was only checked once, we cannot exclude that some mice may have had an irregular estrous cycle.
Indirect Calorimetry: Energy Expenditure, and Food and Water Consumption
For collection of metabolic data, six female mice were individually housed in PhenoMaster cages (TSE Systems, Bad Homburg, Germany) containing weighing sensors recording food intake and water consumption at 20-min intervals. Simultaneously, a gas calorimetry system measured oxygen consumption (VO2). Mice were kept at room temperature (about 22 °C) and fed a standard chow diet (Altromin No. 1314, containing 14% fat, 59% carbohydrates, 27% protein). Prior to data collection, mice were kept for 1 week under LD conditions for acclimatization to the new environment. This was followed by a LD cycle for a week and a 6-h LD phase advance at PE, at which the mice remained for another 6 days. Body mass was measured at the beginning and at the end of the experiment.
General Activity and Automated Food Intake Monitoring
A BioDAQ food intake monitoring system (Research Diets, New Brunswick, MJ) was used to detect long-term food intake at 10-min intervals. General activity was recorded by custom-made passive infrared detectors at 1-min intervals and analyzed using ClockLab software (Actimetrics, Evanston, IL). Six females were kept singly in cages located in an opaque cabinet with a light intensity of 250 lux (when measured at the bottom of the cage), air exchange, a constant ambient temperature of 22 °C, and had access to food ad libitum through a food hopper placed on a scale (Stengel et al., 2012). Prior to data collection, mice were habituated for 1 week to the single housing in the BioDAQ apparatus. On the day of ME, initiated by placing male bedding 6 days earlier, females underwent a 6-h phase advance in the LD cycle. Prior to the 6-h phase advance, the estrous stage was confirmed by vaginal smear collection at ZT4.
Explant Tissue Cultures
PER2::LUC females at PE or ME and males were subjected to a 6-h phase advance. On the second day post-shift, mice were culled at ZT11 of the new LD cycle, and two 300 µm thick SCN, adrenal gland, and ovary slices were collected from each animal and cultivated as described (Landgraf et al., 2015; Nakamura et al., 2005; Pilorz et al., 2014; Yamazaki and Takahashi, 2005; Yoo et al., 2004). Only one sample from each tissue that showed more than four stable cycles of PER2::LUC was considered for data analysis. The dissection lasted in total 15 min and was conducted as follows: After cervical dislocation, brain and peripheral tissues were instantly removed and stored for a very short time in chilled Hank’s buffered salt solution (HBSS) (Invitrogen). Next, tissues were sliced with a vibratome (Leica), placed on Millicel culture membranes (PICM, Millipore) in 35-mm petri dishes containing 1.5 mL recording medium (DMEM low-glucose [Sigma Aldrich]), 10 mM D-glucose (SigmaAldrich), 3 mM 7.5% Sodiumbicarbonate (Roth), 10 mM Hepes (pH 7.2) (Thermo Fisher Scientific), 1% penicillin/streptomycin (10,000 U) (Thermo Fisher Scientific), 2% B-27 (Thermo Fisher Scientific), 0.1 mM luciferin (Promega), and stored in the incubator at 32 °C until placing all sealed tissue dishes into the LumiCycle luminometer (Actimetrics, Wilette, IL) for data collection. Bioluminescence was recorded for several days. The LumiCycle software (Actimetrics Inc., Wilmette, IL) was used for data analysis. The incubation temperature was held at 32 °C throughout the recording. The circadian period was estimated using a periodogram analysis over three consecutive cycles. Phase was measured as the time of the first peak in luminescence after 24 h in culture. The peak was estimated as the highest point of smoothed data in the interval between 12 and 36 h in culture. By plotting the peaks of each tissue, a phase map was drawn (Yamazaki and Takahashi, 2005). A parallel set of females and males was not subjected to the LD phase shift. The unshifted PE and ME females served as controls for the respective group shifted at PE or at ME. The unshifted male mice represented a control group for shifted males only.
Pharmacological Treatment
The 6-h phase advance experiment was carried out on 15-month-old (n = 8) (ovary-intact) C57Bl/6 female mice. Before subjecting the females to the phase shift, the mice were singly housed in a cage equipped with a running-wheel and kept under a normal LD cycle, the light intensity was 250 lux when measured at the bottom of the cage. On day of the 6-h phase shift, mice were either injected intraperitoneally with 50 µg/kg 17-beta-estradiol (E2) (Sigma Aldrich) dissolved in sesame oil (Sigma Aldrich) or vehicle (sesame oil). The dose of E2 was chosen based on previous work showing physiological circulating estradiol concentration (Ciana et al., 2003; Eid et al., 2020). The injection was conducted at ZT4 to simulate increased estrogen concentrations in the middle of the day phase.
Data Analysis
Results are presented as mean ± SEM and analyzed using SPSS Statistics 25. All data were tested for normal distribution using Kolmogorov-Smirnov test. Non-normally distributed data were log10 transformed. Statistical significance of group comparisons was tested with one-way or Mann-Whitney U test. Two-way analysis of variance (ANOVA) was used to test two grouping factors. Wilcoxon test or Tukey’s post hoc analysis were used to estimate between-subject differences. Bonferroni correction was applied when several dependent and independent statistical tests were performed. A repeated-measures adjustment was used to test phase differences during adaptation to the new LD cycle. To estimate onsets of events obtained from TSE as well as from BioDAQ, ActogramJ (Schmid et al., 2011) was used. The prediction of the SCN phase shift in PE females on the last LD cycle was assessed using backward extrapolation from the DD rhythm at cycles 3 to 7 (after new LD). The onset of the daily activity bout was defined as the first of three consecutive 10-min bins of activity that were not separated by more than two 10-min bins of inactivity before the next recorded active bout (Royston et al., 2016). Average onsets during jetlag were modeled by a sigmoidal dose response curve using Prism 8 software (Graphpad, La Jolla, CA). PS50 is defined as the time at which half of the phase shift was completed. To determine PS50, a sigmoid dose response curve with variable slope was fitted to onset time points of locomotor activity, feeding behavior, and oxygen consumption (Kiessling et al., 2010). The recovery from the phase shift and, thus, full entrainment of activity to the new LD cycle was defined on the time when the activity onset occurred following the dark phase. This phase was determined in the first 10 days. Entrainment to the new LD cycle was considered as completed when activity onset was within 20 min during 10-day period before the 6-h phase advance (Valentinuzzi et al., 1997). Period length of the activity was estimated with chi-square periodogram using MATLAB R2014a.
Results
Accelerated Phase Resetting to Light in Females at PE
Before subjecting the female PER2::LUC reporter mice to a single 6-h phase advance of the LD cycle at either their PE or ME stage, we evaluated the regularity of the estrous cycle after adding male bedding into the female’s cage. All females showed variability in the stages of the estrous cycle during two cycles (Suppl. Fig. S1 A-B), but in the first cycle, all females showed regular estrous cyclicity that started with PE after 4 days of adding male bedding and after 6 days all females were in ME (Suppl. Fig. S1 A-B). In the second cycle, fewer females were showing regular estrous cycle (Suppl. Fig. S1 B). Since all females showed regularity in estrous cycle in the first cycle, 4 and 6 days after male bedding adding, we subjected female PER2::LUC reporter mice to a single 6-h phase advance of the LD cycle at either PE or ME. In parallel, a cohort of male animals were phase shifted for comparison. Females as well as males showed a stable entrainment to the LD cycle before phase shift (Suppl. Fig. S2 and Figure 1) and a progressive adaptation of activity rhythms in response to the phase shift (Figure 1a and 1b). However, PE females showed an accelerated phase shift to a new LD cycle compared with ME females and males (Figure 1b and 1c). In PE, 50% of the phase shift (PS50) was reached at 0.77 ± 0.14 days compared with ME females (PS50 = 4.20 ± 0.33 days) and males (PS50 = 3.76 ± 0.12 days); one-way ANOVA between groups: F2,20 = 14.711, p < 0.001; post hoc PE versus ME females p < 0.001, PE females versus males p = 0.011, ME females versus males p = 0.220.
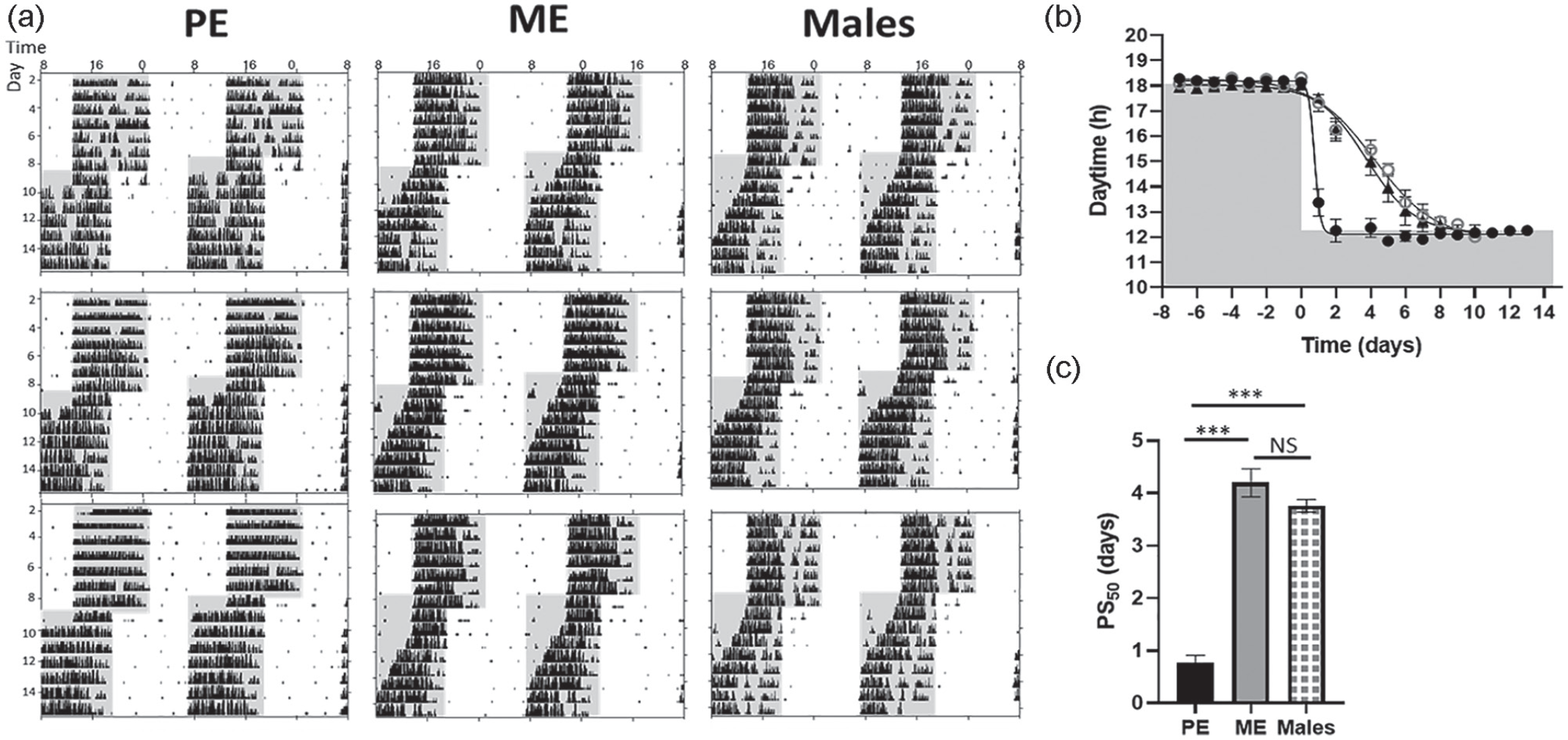
Accelerated phase shift of wheel-running activity to a new LD cycle in females during PE. (a-c) wild type females and males were entrained to a 12-h:12-h LD cycle for >6 days before being subjected to a 6-h phase advance (ME females n = 6, PE females n = 12, males n = 6). (a) Representative double-plotted wheel-running actograms and (b) Average activity onset kinetics using sigmoidal curve fitting with variable slope; data shown are PS50 defined as half-time of the completed phase shift. Shading indicates lights off. (c) PS50 values (means ± SEMs days) of wheel-running activity in PE females, ME females, and males; black bar represents PE females, gray bar ME females, and chequered bar males; differences between the groups were assessed using one-way ANOVA. Abbreviations: LD = light-dark; ME = metestrous; PE = proestrous; PS = phase shift; NS = not significant; ANOVA = analysis of variance.
In an independent experiment without running-wheel access, PE females also showed a more rapid adaptation to a shifted LD cycle in general activity, feeding behavior, and oxygen consumption rhythms (Figure 2 and Suppl. Figs. S3 and S4). All three parameters revealed comparable 50% phase shifts for PE females (activity: 0.9 ± 0.10 days, feeding: 1.0 ± 0.10 days, VO2: 1.0 ± 0.10 days; Wilcoxon test: activity vs. feeding Z = −1.782, p = 0.15; activity vs. VO2: Z = −2.201, p = 0.09, VO2 vs. feeding: Z = −0.319, p = 0.753) and for ME females (PS50 activity: 4.02 ± 0.10 days, PS50 feeding: 4.01 ± 0.13 days; U test: PE vs. ME activity: Z = −3.07, p = 0.001, PE vs. ME feeding: Z = −2.88, p = 0.002). However, shifting was significantly slower in ME females (U test: activity PE vs. ME Z = −2.882, p = 0.02; feeding PE vs. ME: Z = −2.882, p = 0.02). Of note, total activity, 24-h food intake, and 24-h VO2 consumption remained unchanged across the whole experiment and for both estrous groups (Suppl. Fig. S5; rep. ANOVA: general activity: F1,10 = 0.009, p = 0.926; feeding behavior: F1,10 = 2.186, p = 0.170; VO2: F1,10 = 0.176, p = 0.684). Finally, the wheel-running did not reinforce the accelerated phase advance as PS50 values estimated from general activity did not differ significantly from those in wheel-running activity (Figures 1c and 2c, U test general activity vs. wheel-running in PE: Z = −1.76, p = 0.09; ME: Z = −0.32, p = 0.82).
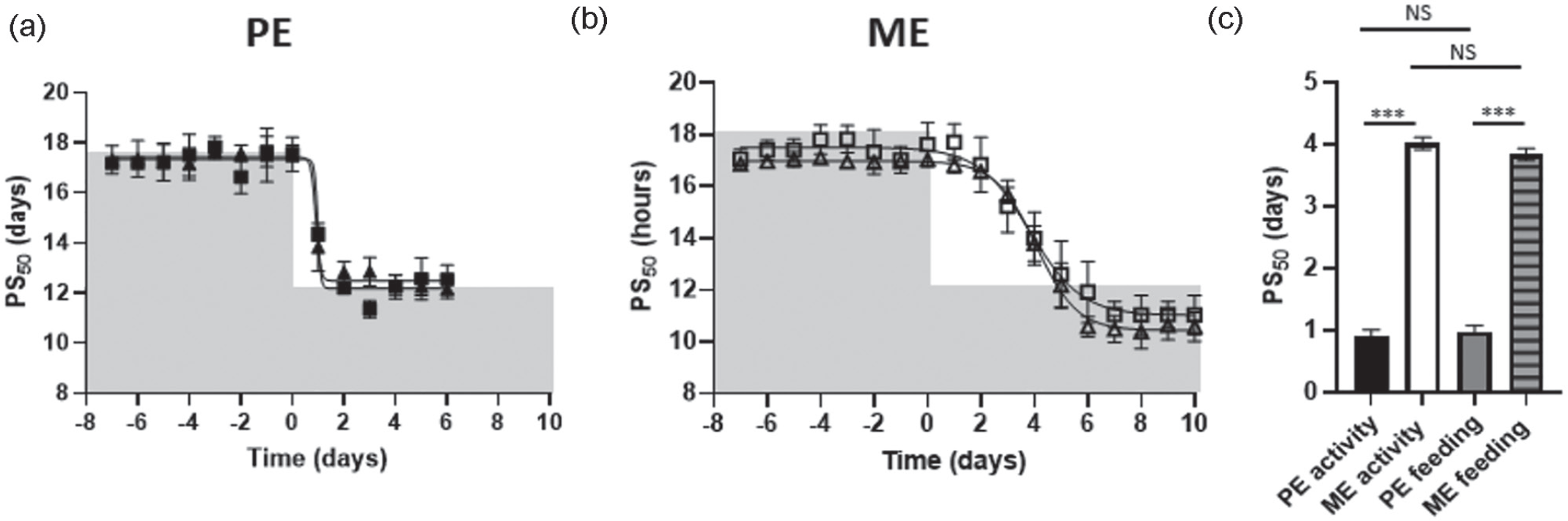
Rapid phase shift of general behavior and physiological rhythms to a LD phase shift in PE female mice. (a-c) In a separate experiment, no running-wheel was available, wild type PE and ME females were entrained on a 12-h:12-h LD cycle for >6 days before being subjected to a 6-h phase advance. (a-b) Average activity and feeding onsets of kinetics, represented by PS50 using sigmoidal curve fitting with variable slope in PE females (n = 6) and ME females (n = 6). Filled triangles: activity in PE mice; open triangles: activity in ME mice. Filled squares: feeding in PE mice; open squares feeding in ME mice; shaded area depicts lights off. (c) PS50 values (mean ± SEM days) of general activity and feeding in PE and ME females. Filled bar: PE activity, open bar: ME activity, gray bar: feeding PE, gray striped bar: feeding ME. Groups were compared using one-way ANOVA. Abbreviations: LD = light-dark; PE = proestrous; ME = metestrous; PS = phase shift; NS = not significant; ANOVA = analysis of variance.
To test to which extent the accelerated phase shift to the new LD cycle in PE is driven by the central clock in the SCN, PE and ME females were released into constant darkness (DD) on the third day after the LD shift. The phase of the SCN pacemaker was assessed by determining onsets on the first day in DD and comparing them with the onsets on the last day in LD (Figure 3). In PE mice, post-shift DD onsets were 1.66 ± 0.19 h delayed when compared with LD onsets on the previous day (Figure 3b, Wilcoxon test: onset on the first day of DD vs. onset second day of shift, Z = −2.201, p = 0.03). Contrary to that, ME females showed much smaller onset phase differences (first day DD vs. onset second day LD) upon release to DD (Figure 3b). Overall, onsets in the first day in DD were comparable between PE and ME mice (U test: Z = −1.29, p = 0.24) (Figure 3c) as well as the period length (Figure 3d). Strikingly, extrapolated onsets of activity on the second day LD in PE females, using extended fitting line evaluated in DD, did not differ from the onsets of activity in ME females (Figure 3e, U test: Z = −0.48, p = 0.70).Together, these data suggest that the kinetics adaptation to the new LD cycle observed in PE female mice differ between behavior and the SCN pacemaker.
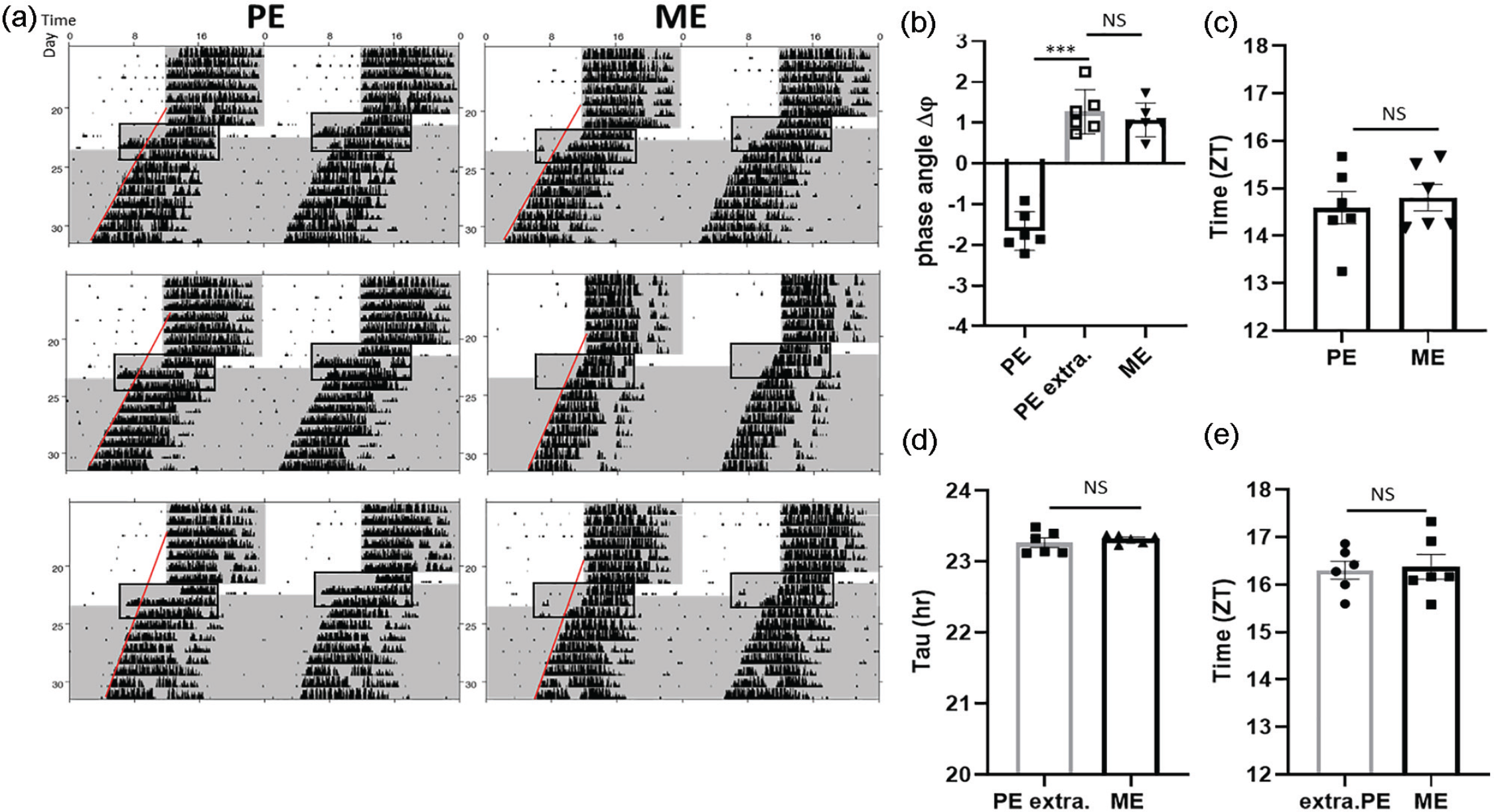
Accelerated phase shifting of wheel-running activity after an LD phase advance in PE females does not reflect central clock resetting. (a) Three exemplary actograms demonstrate wheel-running activity rhythm phase advances during the first 2 days after 6-h LD phase shift followed by DD in PE (n = 6) and ME (n = 6) females. (b) Phase difference between day 2 of the new LD and day 1 of DD in PE and ME females. Bar framed in gray represents extrapolated PE females. The predicted phase angle in extrapolated PE females is evaluated by backward extrapolation (line) from DD rhythm cycles (3-7 after the new LD). (c) Phase shift in PE and ME females relative to old LD cycle after extrapolation of the activity onset on day 2 in PE females. (d) Period length (tau) in PE and ME females estimated from the first day of DD including 6 consecutive DD days. (e) Extrapolated onset of activity in PE and ME females on the last day of LD cycle. Framed area highlights the region of interest consisting of last 2 days of new LD and first day of DD. Shading depicts lights off and DD. Data shown are means ± SEMs, Mann-Whitney U test. Abbreviations: LD = light-dark; PE = proestrous; DD = constant darkness; ME = metestrous; NS = not significant.
We next assessed whether the accelerated phase shift of PE females to the new LD cycle observed in behavior and physiology reflects enhanced phase adaptation of the SCN clock and peripheral tissue clocks to the new LD cycle. PER2::LUC females as well as males were subjected to a 6-h LD phase advance, sacrificed on the second day post-shift, and SCN, adrenal gland, and ovarian explant slice cultures were prepared for luminescence recording (Figure 4a and 4b). SCN explants from, both, PE and ME as well as males exhibited a significant phase advance in the luminescence rhythm compared with non-shifted animals (two-way ANOVA: F5,39 = 20.08 p < 0.000, jetlag vs. control: PE: p < 0.000, ME: p < 0.000, male: p < 0.000). In line with the DD behavior, although, shift magnitudes were similar between PE, ME, and males (Figure 4c post hoc: jetlag: PE vs. ME p = 0.48, PE vs. male: p = 0.18, ME vs. male: p = 0.75). In contrast, peripheral tissue clock rhythms in the adrenal gland and ovaries were shifted significantly faster in PE than in ME and male mice (adrenal gland: two-way ANOVA: F5,40 = 18.89, p < 0.000, post hoc PE vs. ME: p < 0.000, PE vs. male: p < 0.000; ovary: two-way ANOVA: F3,27 = 16.95, post hoc PE vs. ME: p < 0.000), whereas the control PE and ME showed comparable phase in both peripheral tissues (adrenal gland post hoc: ME vs. PE p = 0.82, post hoc PE control vs. ME control: p = 0.42) (Figure 4c). Finally, consistent with behavioral phase on the second day of the phase shift, ME and male mice revealed comparable clock rhythms phasing in the SCN as well as in adrenal gland (post hoc: SCN: p = 0.24, adrenal gland: p = 0.89). These results imply an activity-associated uncoupling of peripheral clock rhythms from the central pacemaker in PE, but not in ME females and males.
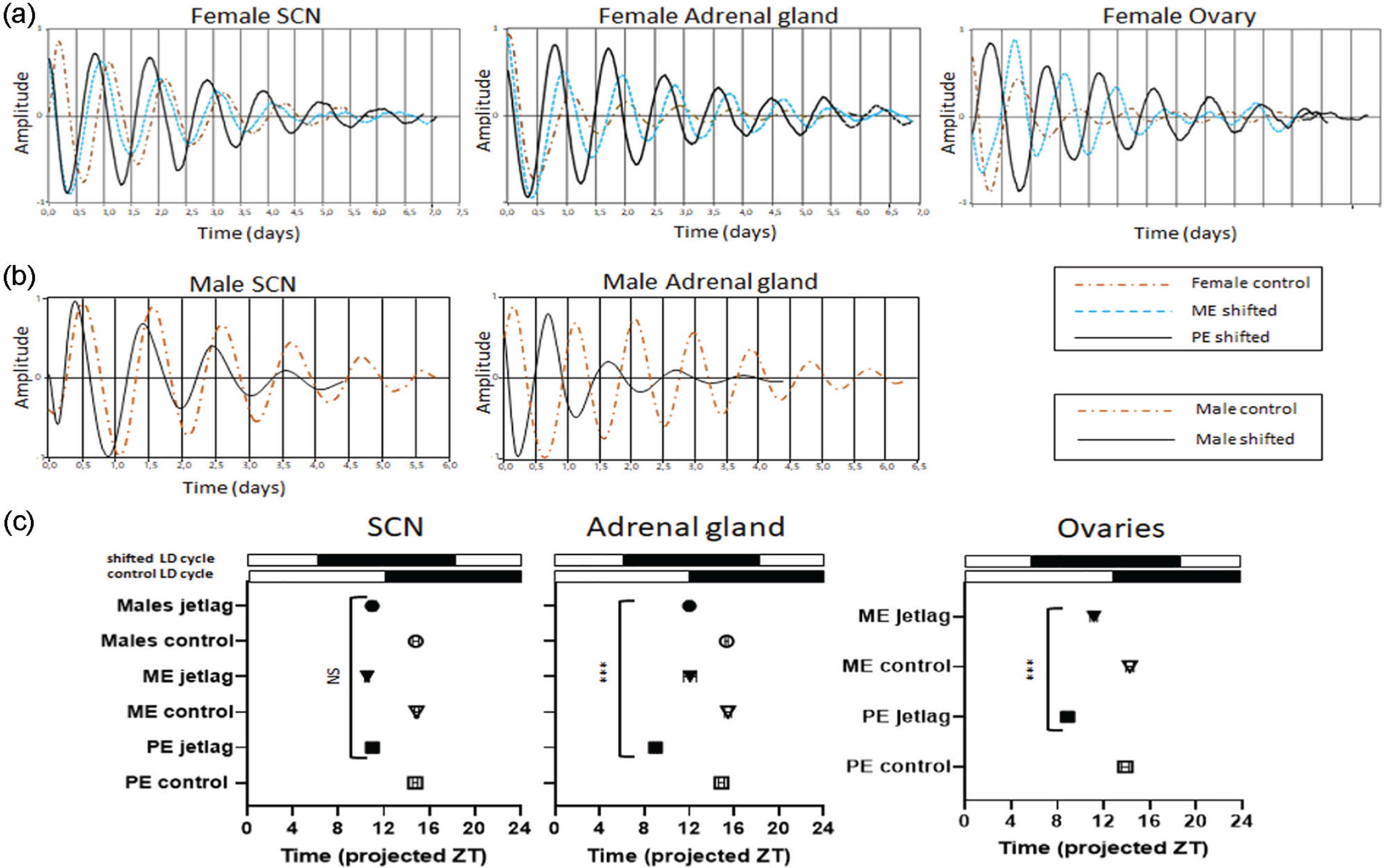
Accelerated phase shift of molecular rhythms to a single-phase shift in PE female mice. (a-b) In a separate experiment, PER2::LUC females in ME (n = 10) and PE (n = 10) as well as (n = 6) males were subjected to a 6-h phase advance LD cycle. In parallel, a control group consisting of PE, ME, and male mice did not undergo any phase shift and was kept under unchanged LD cycle. (a-b) Representative bioluminescence rhythms of tissue cultures (SCN, adrenal gland, and uterus) prepared on the second day of the new LD cycle at ZT11. (a) Plots in the same figure represent tissues obtained from single PE (solid line), ME (dotted line), and control (dot and dashed line) females. (b) Plots represent tissues (SCN and adrenal gland) obtained from males, which were 6-h phase shifted (solid line) and not shifted (dot and dashed line). (c) Phase map of peak PER2::LUC expression rhythms on the second day of phase shift and not shifted ME, PE, and male mice, which represent control. For the phase determination, the first peak of shifted animals was used and was set in relation to the time point of light-off of the new LD cycle with light-off = 12. Black and white bars above the phase maps depict (1) “control LD cycle”: an unshifted LD cycle; (2) “shifted LD cycle”: a new LD cycle phase advanced by 6 h; data are represented as means ± SEMs. Filled symbols illustrate 6-h phase-shifted mice, open symbols represent unshifted mice (control), group differences were tested using two-way ANOVA. Abbreviations: PE = proestrous; ME = metestrous; LD = light-dark; SCN = suprachiasmatic nucleus; ZT = zeitgeber; NS = not significant; ANOVA = analysis of variance.
There is a strong evidence from animal models that estradiol replacement in aging females mitigates many symptoms such as changes in metabolism and locomotor activity (Mauvais-Jarvis et al., 2013; Stuenkel et al., 2015; Yin et al., 2019). Although mice do not undergo a human equivalent menopause, they show acyclicity that is characterized by significant reduction of estrogen levels (Felicio et al., 1984; Frick, 2009; Nelson et al., 1982; Syed et al., 2010). To elicit whether estradiol application at low estrogen levels has a similar phase advancing effect as observed in young adult PE mice, 15-month-old C57Bl/6 female mice, which show stable entrainment under LD cycles (Suppl. Fig. S7) were subjected to a 6-h phase advance and were injected at ZT4 on the day of phase shift with E2 or vehicle, respectively. Intriguingly, in line with PE female mice, aging females injected with E2 showed significantly larger phase shifts compared with control females (Figure 5a-5c). In old females injected with E2, 50% of the phase shift was reached at (1.58 ± 0.28) days whereas in control females at (3.39 ± 0.27) days (Figure 5c, U test: Z = −2.32, p = 0.03).
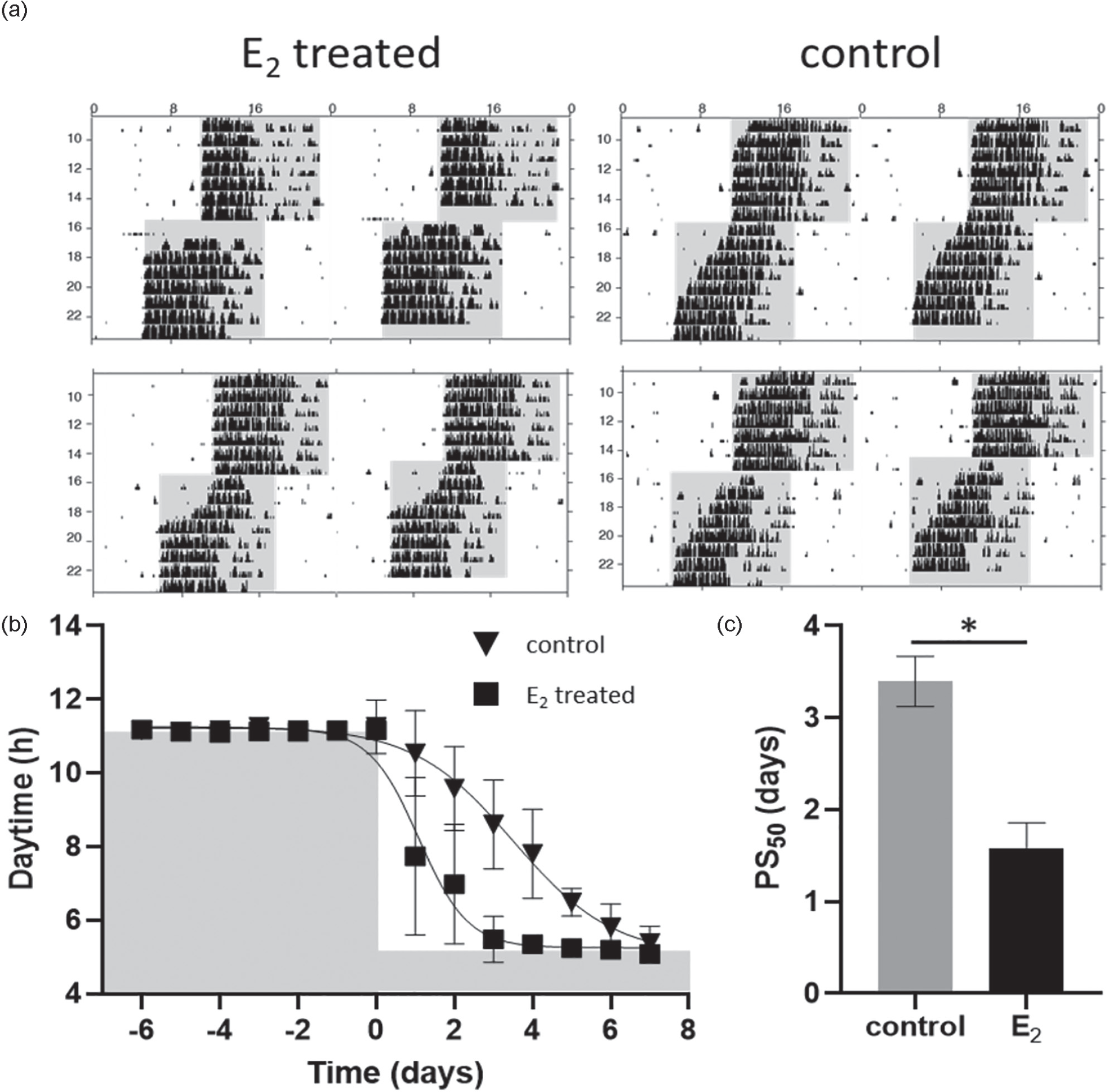
Accelerated phase shift of wheel-running activity to a new LD cycle in 15-month-old females after estrogen administration. (a-c) wild type females (n = 8) >1-year old were entrained to a 12-h:12-h LD cycle before being subjected to a 6-h phase advance. On the day of phase advance, day 0 females were divided into two groups, one group (n = 4) was injected intraperitoneally with 50 µg/kg E2, the second group (n = 4) represents a control group and received an intraperitoneal injection consisting of 100 µL sesame oil. (a) Representative double-plotted wheel-running actograms and (b) average activity onset kinetics using sigmoidal curve fitting with variable slope; data shown are PS50 defined as half-time of the completed phase shift. Shading indicates lights off, triangle depicts vehicle-treated (control), and square illustrates E2-treated female mice, data are represented as means ± SEMs (c) PS50 values (means ± SEMs days) of wheel-running activity in control and E2 treated females; gray bar represents control mice, black bar E2 treated females; differences between the groups were assessed using U test. Abbreviations: LD = light-dark; PE = proestrous; PS = phase shift; NS = not significant.
Discussion
Our findings demonstrate for the first time that the phase of the estrous cycle determines the kinetics of the phase shift of circadian rhythms to a shifted LD cycle. Females subjected to a 6-h phase advance in LD cycle at PE, phase shift their locomotor activity, and physiological and molecular rhythms significantly faster compared with ME females. Surprisingly, this accelerated phase shift during PE does not align with SCN resetting, but rather appears to be regulated further downstream. These findings invite the speculation that the different estrogen concentrations at both estrous cycle phases differently affect phase shifting. This is further supported by the fact that ME females—characterized by low circulating estrogen concentrations—show a more gradual phase shift to a new LD cycle, similar to that of male and aging female mice, which lasts about 7 days. Such transient shift of behavioral rhythms to a new LD cycle has already been well characterized in male mice and it has been shown that adaptation is a gradual process and requires multiple transient circadian cycles of 5 to 7 days following a 6- to 8-h phase advance of the LD cycle (Honma et al., 1985; Legates et al., 2009).
Such transient shifts to a new LD cycle are characterized by disturbed rest-activity rhythms caused by a weakened coupling between central and peripheral clocks (Nicholls et al., 2019; Ramkisoensing and Meijer, 2015; Takasu et al., 2015). In most setups, the phase shift to LD phase advances is rather slow in nocturnal animals requiring multiple circadian cycles for completion (Honma et al., 1985). In the present project, however, we observed that PE female mice shifted on average within 2 days to a phase-advanced LD cycle whereas ME females and males required on average 7 days to complete adaptation. Similar fast phase shifts were observed in E2 treated 15-month-old females, characterized by weak or absent E2 rhythmicity (Nelson et al., 1995; Lu et al., 1979).
The observed significantly larger phase shift in peripheral tissues compared with the central clock in PER2::LUC PE females in the present study indicates an internal clock dissociation. Phase differences in SCN and peripheral tissues may also be caused by tissue dissection itself in particular in tissues with increased phase dissociation (Leise et al., 2020). The fact that the phase dissociation between the peripheral clocks and the central clock was similarly lower in both ME females and males in response to a shifted LD cycle compared with PE females speaks against a dissection effect on phase change in ME and male mice.
There are two possible ways of synchronizing the activity and physiological rhythms to the new LD cycle in PE females: (1) via brain regions that highly respond to estrogens and are not reciprocally synchronized and (2) via peripheral oscillators.
The rhythms of the SCN are synchronized with the daily LD cycle through the phase-shifting effect of light on the SCN (Ramkisoensing and Meijer, 2015). The SCN in turn coordinates the internal circadian rhythms with the external LD cycle (Dibner et al., 2010). These main features of photic-phase shifting have been studied in detail in male rodents, but little is known about the phase shift to a new LD cycle in females. A number of studies have shown that female rats and hamsters display phase advances of locomotor activity rhythms and higher overall activity during PE and estrous stages relative to ME and DE under stable LD cycles (Walmer et al., 1992; Wollnik and Turek, 1988). This extended activity associates with high circulating estrogen concentrations in both PE and estrous phases (Albers et al., 1981; Morin et al., 1977) and is inhibited by progesterone application (Takahashi and Menaker, 1980). This implies a complex regulation of female locomotor behavior by steroid hormones. Such complexity has been confirmed by studies in aromatase knock-out (ArKO) and gonadectomized mice (Brockman et al., 2011). Comparison of circadian parameters between ArKO and gonadectomized males and females reveal that functioning P450 plays a substantial role in developmental effects of estrogens on biological rhythms including the amount of daily activity, period length, and light pulse responses. For instance, males exhibit high phase advances when exposed to light in the late night, whereas females respond with large phase delays to light during the early night (Brockman et al., 2011). Hence, differences in light effects on the magnitude in activity phase shifts may be associated with steroid hormones, which modify the molecular activity in the SCN directly.
Two independent research groups have shown that castrated male and female mice reveal reduced numbers of c-Fos in the SCN that can be reversed by sex hormones (Abizaid et al., 2004; Karatsoreos et al., 2007). Surprisingly in the present study we observed a greater phase shift in PE females as well as in E2 treated aging females compared with ME and males. One could speculate that the light pulse at ZT18 caused by the LD shift could have induced the accelerated phase shift in adult and aging females. This time point, however, lies within a “delay region” and therefore it causes phase delay under acute light exposure (Ouk et al., 2019; Pendergast et al., 2010). Furthermore, given that males exhibit greater phase shift to light pulses at the end of the dark phase compared with females (Brockman et al., 2011), we can reject the notion that the accelerated phase shift in PE and aging females is the result of a light pulse at ZT18.
The way of phase shift initiation may be an additional critical factor for the phase-shift kinetics. In the present study, the 6-h phase advance was induced by shortening the light phase that was followed by a dark pulse at ZT6. Dark pulses at ZT6 cause a robust phase advance regulated by arousal-promoting mechanisms activated by orexin neurons in the medial and lateral hypothalamus. This is accompanied by suppression of c-Fos (Marston et al., 2008) and reduction of clock gene expression in the SCN (Horikawa et al., 2000; Mendoza et al., 2008). The observed accelerated phase advance at PE could therefore be explained by an arousal-promoting stimulus as shown by Marston et al. (2008). In addition, estrogens increase orexin expression (Grafe and Bhatnagar, 2018). Hence, it can be postulated that a dark pulse on the first day of the LD shift as well as estrogen stage may have a decisive effect on the magnitude of the phase shift in females.
In general, the sex hormone estrogen reaches its highest level in PE and is associated with increased activity, glucocorticoid concentrations, and arousal in females. This effect of estrogens on locomotor activity was validated by administration of external estrogens to female rats (Blizard et al., 1975; Ogawa et al., 2003; Slonaker, 1924). Interestingly, this increased locomotor activity does not strictly dependent on a functional SCN, as SCN lesions in female rats do not abolish increased activity after estrogen treatment (Hansen et al., 1979; Palm et al., 1999; Raisman et al., 1977; Södersten, 1984; Wiegand et al., 1980). Thus, other estrogen receptive brain nuclei may regulate locomotor activity in female rodents. In line with this, the SCN in females is characterized by a lower ERβ expression compared with other estrogen receptive brain nuclei known to regulate preovulatory LH surge and locomotor activity (Fahrbach et al., 1986; de la Iglesia et al., 1999; Kriegsfeld, 2013; Smarr et al., 2013; Takeo and Sakuma, 1995). How these estrogen receptive brain regions—and with them activity—may become uncoupled from or weak coupled with the SCN at PE during an environmental change during the phase shift requires future investigations.
Locomotor activity itself has been shown to influence numerous aspects of circadian rhythms (Bobrzynska and Mrosovsky, 1998; Mrosovsky, 1996; Novak et al., 2012; Reebs and Mrosovsky, 1989). A comparison of phase-shift kinetics in PE females with and without running-wheel access shows that the accelerated re-entrainment is not influenced by the reinforcing effect of wheel-running. In addition, like activity, feeding behavior and oxygen consumption showed a rapid phase shift to the new LD cycle in PE females. This offers the possibility that feeding-related metabolites may have affected circadian rhythm resetting to a new LD cycle. Nutrient metabolites such as lipids and fatty acids have been suggested as direct regulators of clock gene expression resulting in modulations of energy metabolism (Gnocchi et al., 2015). Hence, although it is generally accepted that timing of events is SCN dependent, it is also likely that peripheral oscillators expressing ERs may play a role in the timing and stabilization of clocks and metabolic rhythms in the brain and the periphery (Frank et al., 2014; Rando and Wahli, 2011; Yan and Silver, 2015). Peripheral tissue rhythms, in turn, may modulate metabolically integrated hypothalamic circuits and, thus, behavioral adaptation to a new LD cycle. Adrenal gland derived corticosterone (CORT) is a suitable candidate for modulating metabolic as well as brain rhythms. It has already been shown that CORT plays a pivotal role in the synchronization of peripheral clocks as well as in resetting of the central clock after a phase shift (Sage et al., 2004; Kiessling et al., 2010). Importantly, the hypothalomo-pituitary-axis is characterized by sexual dimorphism (Goel et al., 2014). Plasma CORT shows daily rhythms in mice with peak at the end of the light phase. Interestingly, female mice show higher CORT conc. compared with males (Ahowesso et al., 2011) with the highest adenocorticotropic hormone (ACTH) and CORT levels at PE (Viau and Meaney, 1991). Hence, it can be assumed that the observed accelerated phase shift in behavioral, physiological, and molecular rhythms in PE females may associate with the elevated CORT secretion. We have not investigated the level of the CORT in phase-shifted animals; however, we observed greater phase shift in adrenal gland that is accompanied by accelerated phase shift in ovaries in PE females compared with ME and males. Importantly, these accelerated phase shifts in both peripheral clocks did not go together with advanced phase of the SCN. Kiessling et al. (2010) have shown that the activity rhythm is regulated via two ways: One involves the SCN that regulates the clock phase in adrenal gland and the second one comprises adrenal gland clock that feedbacks the SCN through the neuronal system resulting in stabilization of SCN rhythms. Since we do not observe an increased phase shift in the SCN from behavioral or from molecular angle, it is of interest to investigate whether other brain regions, high receptive to CORT, receive the adrenal clock feedback during a changed LD cycle resulting in accelerated behavioral re-adaptation.
Taken together, our study reveals that PE phase associated with elevated estrogen concentration facilitates accelerated phase shift in behavioral, physiological as well as molecular rhythms to a new LD cycle, which seems to be uncoupled from resetting of the SCN pacemaker. Since the accelerated phase shift of locomotor activity and peripheral rhythm on the second day in PE females was not in phase with the SCN and the SCN phase was comparable with that of ME females, we hesitate to call this rapid phase-shift entrainment. However, it is not surprising that already phase-advanced reproductive organs and the adrenal gland in PE females (Nakamura et al., 2010) show an accelerated phase shift in response to the photic-phase shift that could be initiated via signals from the autonomic nervous system (Buijs et al., 2003; Buijs et al., 2013; Ishida et al., 2005).
In addition, for the advanced phase of activity, feeding, and estrogen responsive peripheral oscillators, a selective pressure can be envisioned, as it would enable females to respond quickly to acute fluctuations in the environment such as timed food availability or predation during the critical reproductive period. Reproduction is energetically costly. Therefore, females have evolved strategies to optimize energy demand for self-maintenance and offspring production (Drent and Daan, 1980; Gittleman and Thompson, 1988). Rodents such as mice have limited ability to store energy and instead rely on the continuous acquisition of external resources during the reproductive phase (Houston et al., 2007; Rödel et al., 2016). Phase changes in environmental rhythms put an organism at risk of reducing its reproductive success. Hence, it is of great benefit for the ovulating female to adapt behavioral as well as physiological rhythms as quickly as possible. Based on our results, the SCN appears to be resistant to rapid external perturbation during PE, and therefore it resets slower compared with peripheral oscillators. This may buffer the circadian system against frequent phase shifts under high-estrogen conditions. Future experiments will aim to investigate the molecular targets through which estrogen alters the expression of circadian rhythms in the brain and periphery, for example, in ovariectomized female mice. Understanding the mechanism regulating the phase of circadian rhythms in females and how estrogen contributes to their modulation will provide important insight into the sexually dimorphic regulation of behavior and energy metabolism.
Supplemental Material
Pilorz_et_al_Supplemental_material_jetlag_resetting_of_behavioral_physiological_and_molecular_rhythms_in_pro-estrous_female_mice_03092020JBR – Supplemental material for Rapid Jetlag Resetting of Behavioral, Physiological, and Molecular Rhythms in Proestrous Female Mice
Supplemental material, Pilorz_et_al_Supplemental_material_jetlag_resetting_of_behavioral_physiological_and_molecular_rhythms_in_pro-estrous_female_mice_03092020JBR for Rapid Jetlag Resetting of Behavioral, Physiological, and Molecular Rhythms in Proestrous Female Mice by Violetta Pilorz, Beke Kolms and Henrik Oster in Journal of Biological Rhythms
Footnotes
Acknowledgements
This work was supported by a grant of the German Research Foundation (DFG; OS353-10/1) to H.O. We thank our animal care takers, Torsten Schröder and Kerstin Lünsmann, for their excellent support.
Author Contributions
V.P. performed research design and research project, analyzed data, and wrote the manuscript. B.K. performed and analyzed behavioral data. H.O. participated in project design, data interpretation, and drafting.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
