Abstract
The presence of an endogenous circadian clock within most mammalian cells is associated with the amazing observation that within a given tissue, these clocks are largely in synchrony with each other. Different tissues use a variety of systemic or environmental cues to precisely coordinate the phase of these clocks. The cornea is a unique tissue in that it is largely isolated from the direct blood supply that most tissues experience, it is transparent to visible light, and it is exposed directly to environmental light and temperature. Melatonin is a hormone that has been implicated in regulation of the cornea’s circadian clocks. Here, we analyze the ability of rhythmic melatonin to entrain corneas ex vivo, and analyze the phase of corneal circadian clocks in vivo both in light: dark cycles and in constant darkness. We find that the presence of a retina from a melatonin-proficient mouse strain, C3Sn, can photoentrain the circadian clocks of a co-cultured mouse cornea, but a retina from a melatonin-deficient strain, C57Bl/6, cannot. Furthermore, pharmacologic blockade of melatonin or use of a retina with advanced retinal degeneration, Pde6brd1, blocks the photoentraining effect. Corneal circadian clocks in vivo adopt an advanced phase in C3Sn mice compared with C57Bl/6, but the circadian clocks in the liver are unaffected. This observation is not attributable to a shorter endogenous period of the cornea or behavior between the strains. Some transcripts of circadian genes in the corneas of C3Sn mice also show an advanced phase of expression in a light: dark cycle, while the transcript of Per2 exhibits a light-dependent transient induction at the onset of darkness. We conclude that melatonin acts as a phase modifying factor in a rhythmic manner for the circadian clocks of the cornea.
Most mammalian tissues exhibit autonomous circadian clocks (Mohawk et al., 2012). The circadian oscillations from individual tissues can be measured by molecular assays (Balsalobre et al., 1998), observed using the expression of reporter genes (Kuhlman et al., 2000; Yamazaki et al., 2000; Yamaguchi et al., 2000), or simply observed as overt rhythmic physiology (Bunning, 1958). The circadian clocks of individual peripheral tissues must be synchronized, or entrained, to a correct phase by an external stimulus. The phases of most mammalian tissues are coordinated by output signals from the suprachiasmatic nucleus (SCN) in the hypothalamus, although several examples of tissue clocks which can deviate from the SCN-dictated phase have been found (Damiola et al., 2000; Buhr and Van Gelder, 2014; Buhr et al., 2019).
At the molecular level, the rhythms are produced by the rhythmic transcription and translation of genes of the core clock network. In this transcriptional/translational feedback loop, CLOCK and BMAL1 proteins initiate the transcription of target genes (Gekakis et al., 1998; Bunger et al., 2000; Hogenesch et al., 1998). Some of these CLOCK: BMAL1 targets, particularly members of the Cry and Per gene families, then form protein complexes, which repress further transcriptional activity of the CLOCK: BMAL1 dimer (Buhr and Takahashi, 2013). This core mechanism of a negative feedback loop is conserved among different tissues within an animal; however, some use unique proportionate contributions of constituent groups of clock genes (DeBruyne et al., 2007; Baggs et al., 2009; Ruan et al., 2012). One common feature within mammalian clocks is that Per genes act both as state variables and as immediate early genes responding to environmental changes such as light, temperature, or biochemical signals (Shigeyoshi et al., 1997; Kornmann et al., 2007; So et al., 2009; Brown et al., 2002). This feature makes the Per genes particularly valuable as reporters of the state of the clock and environmental changes (Yamazaki et al., 2000; Yoo et al., 2004).
The cornea is an example of a tissue with robust circadian expression of clock genes both in vivo and in a culture dish (Yoo et al., 2004; Baba et al., 2015). The mammalian cornea is a unique peripheral tissue in its lack of blood vessels, its extensive innervation from the trigeminal nerve, and its transparency to light in the visible range. At the tissue level, circadian and/or diurnal rhythms have been observed in the corneal epithelium mitosis rate (Doughty, 1990; Xue et al., 2017), corneal thickness (Kikkawa, 1973; Harper et al., 1996), and even corneal topography (Read et al., 2005). There has been some suggestion that these rhythmic changes in corneal physiology influence intraocular pressure, which would have important implications in the treatment of glaucoma (Kotecha et al., 2009). Using a bioluminescent circadian gene reporter, Tosini and colleagues have measured rhythmic Per2Luciferase activity in the murine corneal epithelium and endothelium (Baba et al., 2015). Using this same reporter, we have observed photoentrainment of the circadian clocks in cultured mouse cornea to short wavelength light, and this photoentrainment required the presence of the opsin OPN5 (Buhr et al., 2015). However, we could not detect diurnal photo-synchronization of the cornea in vivo, consistent with previous reports (Kofuji et al., 2016), and found that the expression of OPN5 was induced by the culturing procedure (Díaz et al., 2020).
Melatonin is a neuro-modulating hormone synthesized at its highest levels by the pineal gland, but it is also produced by other tissues such as the retina (Simonneaux and Ribelayga, 2003). In the retina, melatonin regulates other neuro-modulating transmitters such as dopamine (Dubocovich, 1983; Nowak et al., 1989; Zhang et al., 2018), which itself then has reciprocal interaction onto the expression of melatonin (Iuvone et al., 1987). In multiple species, melatonin regulates the dynamic structure and function of various retinal cells such as rods, cones, and horizontal cells (Pierce and Besharse, 1985; Wiechmann et al., 1988; White and Fisher, 1989). From a health perspective, topical administration of melatonin increased the healing rate of damaged corneas and may assist in alleviation of dry eye symptoms (Nayak et al., 2020; Hoyle et al., 2006). The Tosini laboratory has demonstrated that a single administration of melatonin or its agonists causes phase shifts to the cornea’s clocks ex vivo (Baba et al., 2015). We wished to further explore the impact of melatonin as a means to synchronize the corneal clocks to photic signals. Interestingly, the most commonly used mouse strain for laboratory research, C57Bl/6, does not produce melatonin (Goto et al., 1989; Ebihara et al., 1986). Here, we test whether rhythmic melatonin exposure from a biologic tissue can entrain cornea clocks ex vivo, and whether the absence of melatonin influences corneal clock phase in vivo.
Materials And Methods
Animals
All animal procedures were performed in compliance with the Institutional Animal Care and Use Committee at the University of Washington, Seattle, WA. C3Sn.BL1A-Pde6b+/DnJ (“C3Sn”; stock # 003648) mice, C57Bl/6J (“C57Bl/6”; stock # 000664), and C3H/HeJ (“C3H”; stock # 000659) were purchased from Jackson Laboratories, Bar Harbor, ME (www.jax.org). C3Sn mice are C3H mice in which the retinal degeneration mutation (Pde6brd1/rd1) has been restored to a non-degenerative allele (Pde6bwt/wt). Opn5–/–;Per2Luciferase mice were maintained on a background of C57Bl as first described in (Buhr et al., 2015). For experiments using C3Sn; Per2Luciferase tissue, the Per2Luciferase allele was outcrossed from a C57Bl/6;Per2Luciferase stock for 6 generations onto an inbred C3Sn background. All experimental mice were between 6 weeks and 1 year of age, and both male and female mice were used. All C3H mice were at least 4 months old before being included.
Genotyping of N6 C3Sn × C57Bl/6 Mice
To determine if the N6 backcrossed C3Sn mice contained the genetic signatures of the C3Sn lineage at the loci of the Aanat and Asmt genes, 3′ end mismatch polymerase chain reaction (PCR) was performed as described in Shimomura et al. (2010). Briefly, single nucleotide polymorphisms (SNP) were determined between C3Sn and C57Bl strains at the genomic loci of Aanat and Asmt. Primers were designed so that the 3′ nucleotide contained one or the other version of the SNP, and an artificial mismatch was also introduced at the third position from 3′ to make allelic determination more robust. Genomic DNA was then tested by end-point PCR using Amplitaq (Fisher) with a cycle of 95 °C for 10 min, and then 32 cycles of 95 ° for 1 min, 60 °C for 30 s, and 72 °C for 30 s. We also ran real-time PCR on genomic DNA with Power Up SYBR Green mix (Thermo Fisher) on an ABI 7500Fast (Applied Biosystems) machine using 95 °C for 10 min, and then 40 cycles of 95 °C for 10 s and 60 °C for 30 s (Suppl. Fig. 1A and 1B). Primers used: Aanat: C3Sn Forward: 5′- CCAGCATGACCCAGTCTCAC -3′ C57Bl6 Forward: 5′- CCAGCATGACCCAGTCTCAT- 3′ Aanat Reverse: 5′- CCCGAGCTGAGAGCTTTTTA -3′ Asmt: C3Sn Forward: 5′- CAACAAGGATTTCACCACTAC -3′ C57Bl6 Forward: 5′- CAACAAGGATTTCACCACTAA -3′ Asmt Reverse: 5′- TGGGCCCATAATAAGCAAAG -3′
Melatonin ELISA
Retinas of C3Sn, N6 C3Sn × C57Bl/6, and C3H mice were cultured in 1.2 mL of Neurobasal media in a 5% CO2 incubator at 36 °C. A light: dark cycle was produced from LEDs of 415 nm (2 × 1014 photons cm–2 s–1) and 475 nm (1.5 × 1014 photons cm–2 s–1). Total cell culture media was collected from one cohort 2 h after lights-on and another cohort 12 h later at 2 h after lights-off and was frozen at –80 °C. Competitive enzyme immunoassay (EIA) ELISA was run using a colorimetric assay at 450 nm (LSBio #LS-F25779) on total cell culture media.
Ex Vivo Organotypic Tissue Culture
Retinas of C57Bl/6, C3Sn, or C3H mice were quickly dissected in cold HBSS (Gibco) from mice euthanized by CO2 asphyxiation. The retinas were placed on cell culture inserts (Millipore, PICMORG50) which floated in Neurobasal A (Gibco) media containing B-27 Plus serum-free supplement (Life Technologies), 25 units/mL penicillin; 25 µg/mL streptomycin, and 2 mM Glutamax (Gibco). These were incubated at 36 °C in an environment of 5% CO2 overnight. The following day, corneas were dissected in cold HBSS from Opn5–/–;Per2Luciferase mice, which were euthanized by CO2 asphyxiation. An individual cornea was gently placed directly on top of each retina with the cornea epithelium facing downward so that the cornea’s epithelium was in direct contact with the ganglion cell layer of the retina. These co-cultures were then transferred to DMEM (Cellgro) media containing B-27 Plus serum-free supplement (Life Technologies), 25 units/mL penicillin; 25 µg/mL streptomycin, 2 mM Glutamax (Gibco), 10 mM HEPES (Life Tech), 352.5 µg/mL NaHCO3, and 0.1 mM D-Luciferin potassuim salt (Biosynth). These cultures were sealed with vacuum grease (Corning) and maintained at 36 °C.
Tissues from the right and left eyes of the same mouse pairs were cultured in separate culture dishes such that two cornea: retina co-culture dishes were generated from two mice. The two dishes of each pair were then placed on opposite sides of a clock device, which exposes the cultures to oppositely phased 9 h:15 h light: dark cycles as described in detail in Buhr and Van Gelder (2014). Light was administered from LEDs of 415 nm (2 × 1014 photons cm–2 s–1) and 475 nm (1.5 × 1014 photons cm–2 s–1) which were on continuously to avoid temperature changes. After 4 days of ex vivo light: dark cycles, the culture dishes were transferred to a Lumicycle photomultiplier tube apparatus which measures bioluminescence continuously for up to 32 independent cultures (Actimetrics). The Per2Luciferase luminescence rhythms were measured in darkness for at least 3 days. Phases and periods were measured using a best fit sine wave in the Lumicycle Analysis software package (Actimetrics). Peak Per2Luciferase phases were compared with the time of the last light to dark transition prior to transfer to constant darkness for analysis on Rayleigh plots. Rayleigh plots were generated for clustering analyses using Oriana software (Kovach Computing, UK).
Luzindole (Sigma Millipore # L2407) was dissolved in DMSO and added to DMEM for a final concentration of 50 µM (0.1% DMSO) or 10 µM (0.02% DMSO). Luzindole remained in the media throughout the experiment.
Tissue Collection for Quantitative Reverse-Transcription Polymerase Chain Reaction (RT-PCR)
C3Sn and C57Bl/6 mice were placed in light: dark cabinets equipped with LEDs of 415 nm (4 × 1014 photons cm–2 s–1), 475 nm (7 × 1014 photons cm–2 s–1), and 530 nm (4 × 1013 photons cm–2 s–1), which were on for 12 h daily. After 3 weeks of 12 h:12 h light: dark cycle exposure, the mice were placed into constant darkness. Four mice of each strain including both sexes were euthanized by cervical dislocation at 3-h intervals on the last day of the light: dark cycle and for 2 days of darkness. Eyes and liver were rapidly dissected, corneas and retinas were dissected in ice cold HBSS (Gibco), and all tissues were flash frozen in TRI-Reagent (Ambion/Thermo Fisher) at –80 °C. Total RNA was isolated using the manufacturer’s protocol using bromo-chloropropane for isolation of aqueous RNA fraction followed by isopropanol/ethanol precipitation. RNA was reverse transcribed into cDNA using High Capacity RNA to cDNA kit (Thermo Fisher) which includes both random hexamers and oligo-dT priming. QPCR was performed using PowerUp SYBR Green RT-PCR mix (Thermo Fisher) on an Applied Biosystems 7500Fast real-time PCR machine. Relative quantities of transcripts were quantified using the 2^-ΔΔCt method comparing the Ct of individual clock genes with β-actin from the same sample. Values were normalized to the lowest value of the gene set. Primers used were as follows: Per1: Forward, 5′—CCCAGCTTTACCTGCAGAAG Reverse, 5′—ATGGTCGAAAGGAAGCCTCT (91 bp), Per2: Forward, 5′—CCAACACAGACGACAGCATC- 3′ Reverse, 5′—TCTCGCAGTAAACACAGCCT (216 bp), Bmal1: Forward, 5′—GACATTTCCTCAACCATCAGCG Reverse, 5′—GCATTCTTGATCCTTCCTTGGT (200 bp), Dbp: Forward, 5′—CGAAGAACGTCATGATGCAG Reverse, 5′- GGTTCCCCAACATGCTAAGA (118 bp), β-actin: Forward, 5′—AGGTGACAGCATTGCTTCTG Reverse, 5′—GCTGCCTCAACACCTCAAC (188 bp).
CircWave 1.4 software (developed by R.A. Hut, www.euclock.org) was used to determine the strength of the circadian rhythm and the phase of the resulting rhythm. Briefly, the software fits a horizontal line at the mean of the whole data series and determines if individual time points significantly differ from that mean. Then, it uses harmonic regression to find the number of sine waves necessary to fit the data. A maximum of three sine waves were allowed to determine if a data series displayed a 24-h rhythm. A p value of <0.05 was also required from the wave fit data to raw data to conclude a circadian or diurnal rhythm was present. The software also determines a horizontal center of gravity, which correlates to a mean of data in the time dimension, which represents the phase of peak expression of the fit data curve. Two-way ANOVA with Tukey post hoc analyses was run on the raw qRT-PCR data comparing mouse strain and time of collection for each gene using Sigma Plot 11.0 software to detect differences between these groups. However, this does not directly reflect a measurement of difference of phase. For this, CircWave-generated center of gravity output data were compared with a two-tailed t-test between the strains for each gene using Sigma Plot 11.0 software.
Animal Behavior Measurements
C3Sn and C57Bl/6 mice of both sexes between 7 weeks and 1 year of age were housed individually in cages equipped with running wheels. The revolutions of the running wheels were detected by an electronic microswitch on the side of the cage and recorded by a computer. The principal period over 10 days of free-running behavioral activity in constant darkness was determined by ClockLab Analysis (Actimetrics).
Results
Entrainment of Corneas by C3Sn Retinas, but not C57Bl/6 Retinas
Our first goal was to allow for melatonin to be presented to cultured corneas in a physiologically relevant manner while avoiding unsealing and handling the sealed tissue culture dishes. Previous reports have shown that disturbing a cultured cornea can alone cause phase shifts (Baba et al., 2015). We took advantage of two previous findings: (1) Opn5–/– corneas do not photoentrain to light: dark cycles on their own (Buhr et al., 2015), and (2) certain strains of mouse retina secrete melatonin rhythmically in culture while others do not (Tosini and Menaker, 1998). Sealed organotypic cultures were made with a wild-type retina on a cell culture insert with a cornea from an Opn5–/–;Per2–Luciferase mouse on top, in direct contact with the retina (Figure 1a). Retinas were either from C57Bl/6 (melatonin-deficient) or C3Sn (melatonin-proficient) mice. Two cornea: retina co-cultures, from the right and left eyes of the same two mice, were then placed on opposite sides of a light: dark apparatus which provided anti-phasic light: dark cycles for 4 days (Figure 1a). The luminescence traces from the co-cultures (only the Opn5–/–;Per2Luciferase corneas were luminescent) were measured in constant darkness for the following 4 days. The Per2Luciferase luminescence traces from corneas cultured in the presence of a C3Sn retina adopted opposite phases to each other (from diametrically opposed positions, 0° and 180°, in the light: dark apparatus) after the light: dark cycle treatment, whereas the phases of corneas cultured with C57Bl/6 retinas were not different (Figure 1b and 1c). When analyzed on a Rayleigh plot, the phase distribution of cultures in the C3Sn group cluster to a common phase relative to the previous light: dark cycle (Figure 1d). Individual corneas co-cultured with C57Bl/6 retinas displayed a more dispersed phase distribution in relation to the previous light: dark cycle. Thus, the presence of melatonin-proficient C3Sn retinas caused a light-induced phase synchronization of the co-cultured Opn5–/– corneas, but the presence of melatonin-deficient C57Bl/6 retinas did not.
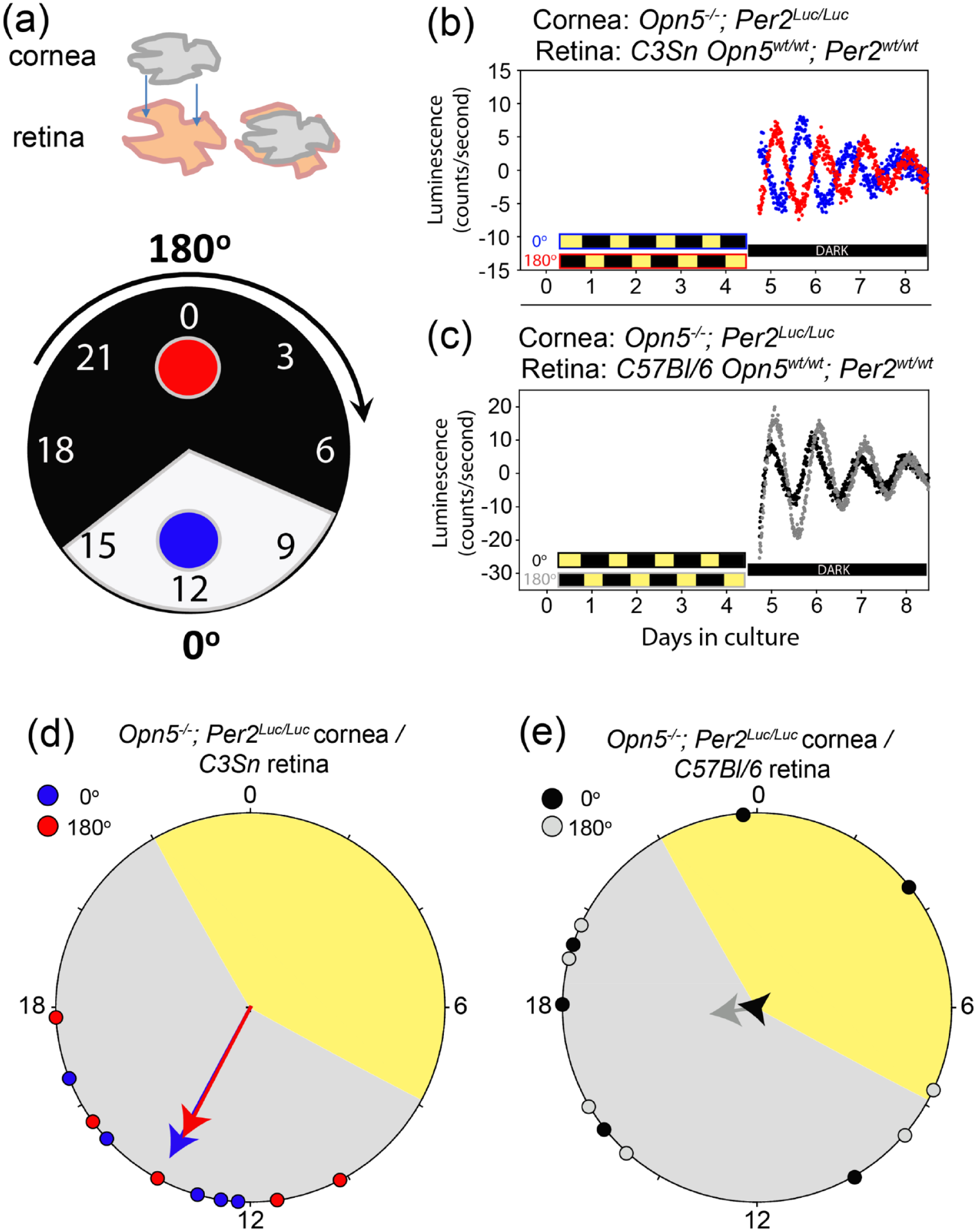
Corneas photoentrain when co-cultured with C3Sn retinas, but not with C57Bl/6 retinas. (a) Schematic of the co-culture arrangement with a single cornea placed directly on top of a single retina (upper). Pairs of co-cultured tissues are then placed in diametrically opposed positions of a light: dark apparatus in either the 0 ° (blue) or 180 ° (red) which exposes the two dishes to oppositely phased light: dark cycles. (b) Opn5–/–;Per2Luciferase cornea: C3Snwt/wt retina pairs were exposed to light: dark cycles for 4 days before being measured in darkness for the following 4 days. Red and blue traces show representative examples of Per2Luciferase rhythms of 0° (blue) or 180° (red) co-cultures. Traces in anti-phase represent a full photoentrainment, while traces at the same phase represent a lack of light influence. (c) The same as B, but with C57Bl/6wt/wt retinas with Opn5–/–;Per2Luciferase corneas in 0° (black) or 180° (gray). (d) Rayleigh plot of phases of individual cultures (n = 5 for each light position) as shown in (b). Vector arrows show the strength of clustering of phases with the center of circle representing a lack of clustering, and arrows reaching the outer circumference of the circle representing perfect clustering at one phase point. Red and blue points represent the phase of an individual co-culture dish after the light: dark cycle. (e) The same as (d) but quantifying the data from samples as shown in (c) (n = 5 for each light position). Note the lack of clustering when corneas are co-cultured with C57Bl/6 retinas.
Blocking Melatonin Abrogates Co-culture Entrainment
There are likely to be more genetic differences between the two inbred mouse strains beyond the two enzymes of the melatonin synthesis pathway mutant in C57Bl/6 (Ebihara et al., 1986). To further test the role of melatonin, we performed the same experiment with Opn5–/–;Per2Luciferase: C3Sn (cornea: retina) co-cultures in the presence of the melatonin antagonist luzindole. Luzindole blocks both melatonin receptors with a higher affinity for MT2 than MT1 (Dubocovich et al., 1998), and MT2 has been specifically implicated in the mouse corneal melatonin response (Baba et al., 2015). With chronic presence of luzindole in the media, Opn5–/–;Per2Luciferase corneas were no longer photoentrained by the C3Sn retinas (Figure 2b and 2c). Many of the co-culture pairs from oppositely phased light: dark cycles displayed similar phases to each other when returned to constant darkness, indicating that the anti-phasic light cycles did not influence their phase in the absence of melatonin signaling.
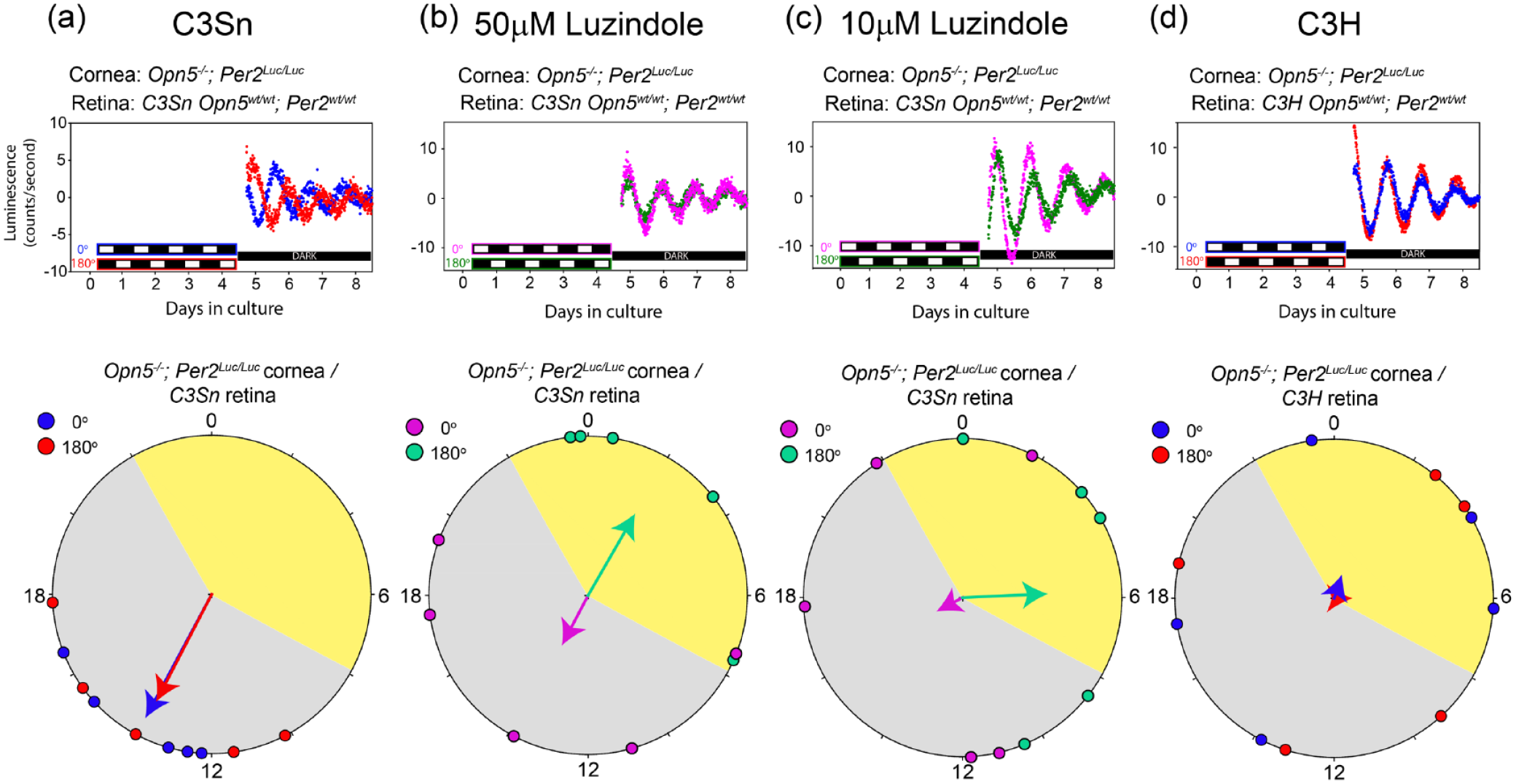
Melatonin signaling is required for cornea: retina co-culture photoentrainment. (a) A set of representative traces of cornea: retina co-culture pairs after ex vivo photoentrainment (upper), and the same Rayleigh plot as shown in Figure 1d (lower). (b and c) Representative traces of Opn5–/–;Per2Luciferase cornea: C3Snwt/wt retina pairs in the presence of luzindole throughout the experiment in the 0° (magenta) or 180° (green) positions of the clock apparatus (upper). Rayleigh plot of resulting phases of five independent replications of the 4-day light: dark experiment (lower). 50 µM (b) and 10 µM (c) luzindole did not result in tight clustering of phases to the dark period as in (a). (d) Representative traces of Opn5–/–;Per2Luciferase cornea: C3Hrd1/rd1 retina pairs. Rayleigh plot of the resulting phases of five independent replications of the 4-day light: dark experiment. Adult degenerated retinas did not cause phase clustering of corneal phases in light: dark cycles.
In the mammalian retina, melatonin is primarily synthesized by rods and cones in the outer photoreceptors layer (Bubenik et al., 1978; Wiechmann and Hollyfield, 1989). One of the most common melatonin-proficient mouse strains, C3H, contains a naturally occurring mutation in the Pde6b gene (rd1) which causes degeneration of the rods and cones (Keeler, 1926; Pittler and Baehr, 1991). Accordingly, without rods and cones, the degenerated retina ceases rhythmic and light-regulated production of melatonin (Tosini and Menaker, 1998). We next co-cultured Opn5–/–;Per2Luciferase corneas with retinas from C3H mice which were at least 4 months old, to allow for complete retinal degeneration of the outer photoreceptor layer. After exposing the pairs of co-cultured tissues to oppositely phased light dark cycles for 4 days, the resulting phases of the corneas were not clustered to a common phase of the light: dark cycles (Figure 2d). Similar to what was observed in the presence of luzindole, the phase of the free-running corneal clocks in constant dark conditions were similar to their biologic pair, but not influenced by the light: dark cycle. Because C3Sn mice retain their rods and cones in adulthood (Contreras et al., 2021) and C3H (rd1) do not, but are otherwise the same strain (Chang, 2015), this suggests that the rods and cones are necessary for the photoentrainment of corneas co-cultured with retinas. When considered with the lack of photoentrainment of the corneas co-cultured with C57Bl/6 or C3Sn retinas in the presence of luzindole, cornea cultures are capable of photoentrainment in the presence of retinas capable of rhythmically producing melatonin.
Phases of Corneal Clocks In Vivo From C3Sn and C57Bl/6 Mice
The experiments described above were all performed on tissue ex vivo. We wished to determine if differences existed between the melatonin-proficient and melatonin-deficient strains in vivo. Two cohorts of mice from each strain, C3Sn and C57Bl/6, were exposed to 12 h:12 h light: dark cycles for 3 weeks and were then transferred to constant darkness. Corneas and livers were collected from individual animals over 2 days beginning at circadian time 0 (CT0) of the first day of constant darkness (the animals were in darkness for 12 h prior to the first collection from zeitgeber times 12-24, with ZT12 being the onset of darkness in the light: dark cycle). Quantitative RT-PCR analyses of the clock gene transcripts of Per2, Per1, Bmal1, and Dbp were compared between the two strains. Using harmonic regression analyses, which allowed for a primary sine wave plus up to two harmonics, the rhythmicity of the data and the phase (center of gravity of the peak of data) was determined (CircWave 1.4 software, developed by R.A. Hut). Significant near-24-h rhythms were detected in all transcripts analyzed for the cornea and liver (Figure 3a and 3b). Interestingly, the center of gravity phases occurred significantly earlier in the cornea transcripts of C3Sn mice than in C57Bl/6. The phases were 7.0 ± 1.0 h earlier for Per2, 5.63 ± 1.1 h for Per1, 4.5 ± 1.0 h for Bmal1, and 5.8 ± 1.4 h for Dbp in C3Sn corneas compared with C57Bl/6, for an average phase angle difference of 5.7 h between the strains (p < 0.05 for data compared between strains two-way ANOVA, Tukey post hoc, and two-tailed t-test of center of gravity between strains for each gene; Figure 3a). In contrast, the phases determined by center of gravity analyses for transcripts in the livers of the same mice showed no difference between the C3Sn and C57Bl/6 mice (Figure 3b). This indicates that the advanced cornea phase is not indicative of all peripheral tissue clock phases.
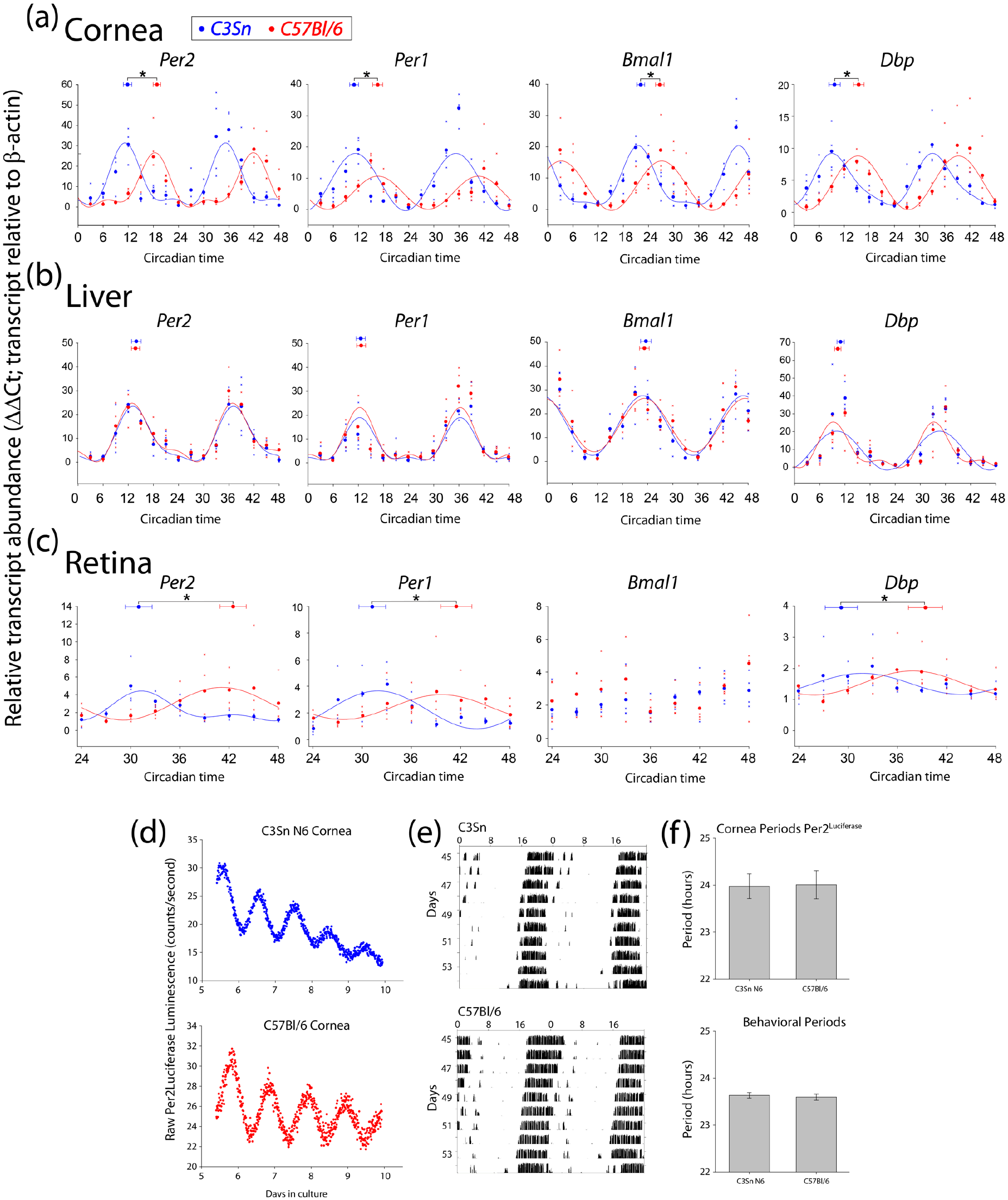
Corneas of C3Sn mice have an advanced phase angle compared with C57Bl/6. Relative transcript abundance of the indicated genes for (a) cornea, (b) liver, and (c) retina at the indicated circadian times. Each collection began after at least 12 h of darkness from the previous 12 h:12 h light: dark cycle (36 h of darkness for the retina). Large points along the curve represent the mean values of the individual data points represented by small “x” points. Curves represent harmonic regression sine wave fits, with no more than three harmonics. Points at the top of each panel with error bars represent the average center of gravity ± standard error of mean for the data of the same color: C3Sn in blue and C57Bl/6 in red. *p < 0.05 in two-tailed t-test of center of gravity between the strains considering biological replicate sets. n = 4 mice for each time point for each strain. All genes displayed an advanced phase in the corneas of melatonin-proficient, C3Sn mice compared with C57Bl. (d) Representative Per2Luciferase traces from C3Sn N6 corneas (blue) or C57Bl/6 corneas (red). (e) Double plotted free-running running wheel activity in constant darkness of C3Sn and C57Bl/6 mice. (f) Quantified period data from cornea Per2Luciferase rhythms (upper) and behavioral free-run rhythms (lower). Columns represent the mean ± standard error of mean for each (n = 9 for each genotype in luciferase rhythms and were measured over 4 days; n = 6 for each genotype for behavioral rhythms and were measured over 10 days). No period differences were detected in either behavioral or corneal free-running rhythms.
One potential cause of an advanced phase angle in the cornea during the first 2 days of darkness would be if the C3Sn mice had a shorter endogenous period of oscillation compared with C57Bl/6. To rule out this possibility, we measured the free-running periods of both the corneal clocks and the behavioral clocks of the two strains. C3Sn mice were crossed to the Per2Luciferase mouse line, and the Per2Luciferase allele was then backcrossed to C3Sn for six generations (N6). The periods of Per2Luciferase luminescence rhythms were not significantly different between the C3Sn N6 (23.97 ± 0.26 h) corneas and the C57Bl/6 (24.01 ± 0.30 h) corneas (Figure 3d and f). Similarly, when C3Sn mice (congenic) and C57Bl/6 mice were allowed to run in running wheels in constant darkness, their free-running behavioral rhythms were not significantly different (C3Sn = 23.63 ± 0.05 h, C57Bl/6 = 23.60 ± 0.07 h, Figure 3e and f). To confirm that the N6 backcrossed mice were melatonin proficient, we analyzed total cell culture media of cultured retinas in a light: dark cycle 2 h after lights-on and 2 h after lights-off using a competitive ELISA assay. The highest measurements of total melatonin concentration in the static culture dish were below the suggested concentration optimal for the ELISA system for all genotypes tested (~2 pg/mL measured vs. 7 pg/mL suggested lowest concentration). However, the melatonin detected in darkness was consistently greater than in light for both C3Sn congenic and N6 backcross, but was not significantly different in retinas of 4-month-old C3H mice in dark versus light (Suppl. Fig. 1C).
We also collected retinas from the mice on the second day of constant darkness. The amplitude of clock gene expression was lower in the retinas of both strains compared with the cornea and liver expression (Figure 3a-3c), but was generally in agreement with previously published retinal transcript oscillations (Ruan et al., 2006; Storch et al., 2007). Also in corroboration with previous reports of the retinas of melatonin-proficient mice displaying different phases than melatonin-deficient (Dinet et al., 2007; Dinet and Korf, 2007), we measured a difference of 11.5 ± 1.6 h for Per2, 10.8 ± 1.6 h for Per1, and 10.82 ± 2.0 h for Dbp between the peak phases of C3Sn and C57Bl/6 retinas (p < 0.05 in two-way ANOVA with Tukey post hoc, plus p < 0.05 comparing center of gravity phase for each gene with two-tailed t-tests; Figure 3c). A significant circadian oscillation was not detected in the expression of Bmal1 for either strain.
Finally, we collected RNA from the cornea, liver, and retina in another cohort of mice entrained to a 12 h:12 h light: dark cycle to measure diurnal expression of the same transcripts. The transcript expression of Per1, Bmal1, and Dbp in the cornea showed a similar, earlier phase of peak expression in C3Sn mice than in C57Bl/6 (Bmal1 = 3.4 ± 1.0 h difference, Dbp = 4.7 ± 1.1 h, Per1 = 2.37 ± 1.6, p < 0.05 comparison of center of gravity within each gene in two-tailed t-test, also p < 0.05 in two-way ANOVA considering all raw data; Figure 4a). The expression of Per2 in the cornea in a light: dark cycle displayed a unique pattern. At ZT 15, the expression of Per2 in the C3Sn cornea was significantly higher than the other points in the time-series of C3Sn corneas (p < 0.01, one-way ANOVA, Tukey post hoc). Interestingly, this phase, following the onset of darkness, is one at which the melatonin secretion is at its maximum (Tosini and Menaker, 1998), and is in agreement with the ex vivo observation of an induction in Per2Luciferase following melatonin administration (Baba et al., 2015). However, with the sharp increase of expression at this time point, four sine wave harmonics were required to fit the data, making it difficult to assign an appropriate phase to the data (CircWave 1.4). No significant differences were observed in the phase of any of the four clock genes in the liver between the C3Sn and C57Bl/6 strains in light: dark cycles (Figure 4b).
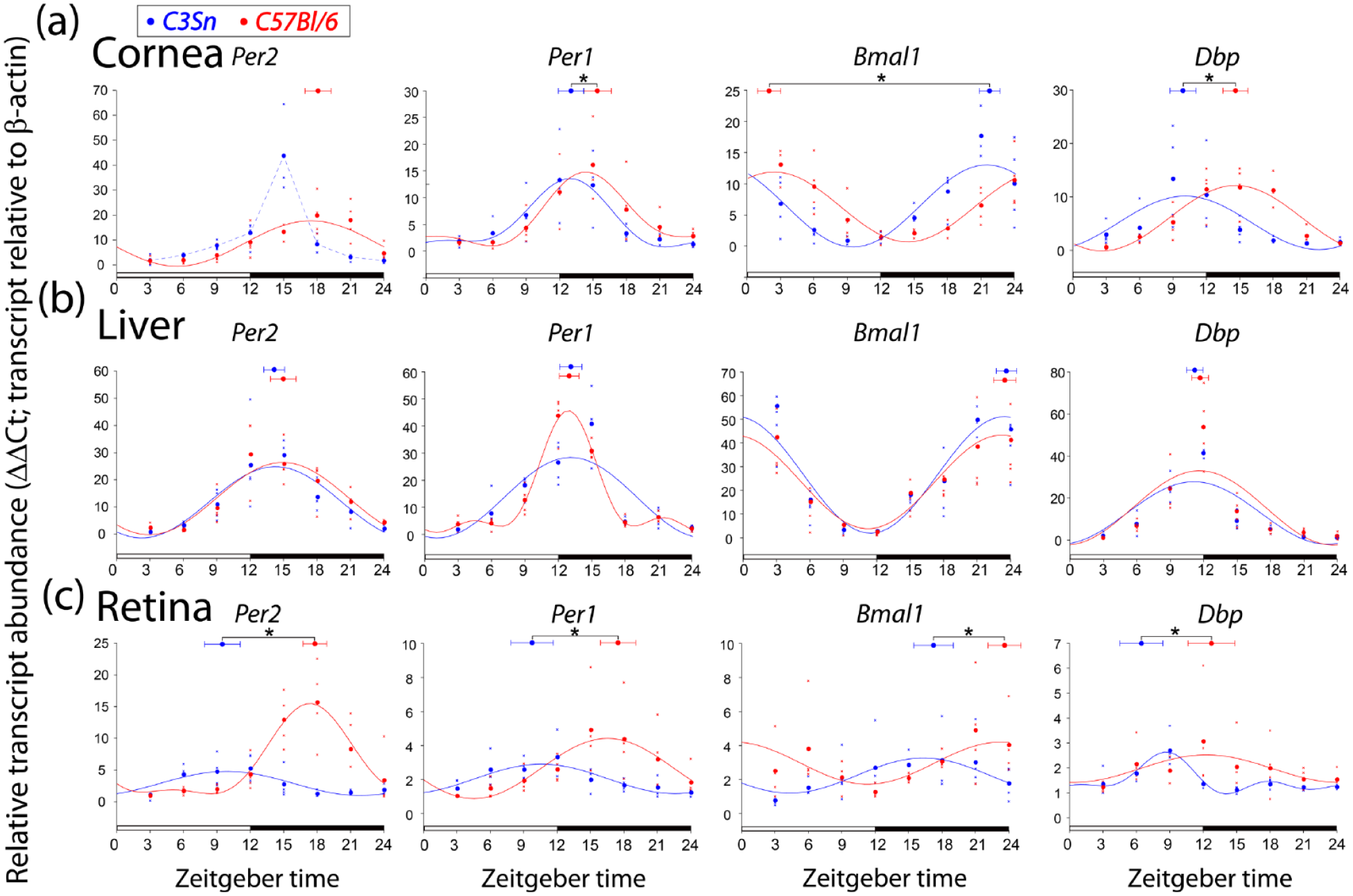
Light effects on the expression of clock transcripts in the cornea. Relative transcript abundance of the indicated genes for (a) cornea, (b) liver, and (c) retina at the indicated zeitgeber (external time) times. Each collection began after at least 3 weeks of 12 h:12 h light: dark cycle. Large points along the curve represent the mean values of the individual data points represented by small “x” points. Curves represent harmonic regression sine wave fits, with no more than three harmonics. Points at the top of each panel with error bars represent the average center of gravity ± standard error of mean for the data of the same color: C3Sn in blue and C57Bl/6 in red. *p < 0.05 in two-tailed t-test of center of gravity between the strains considering biological replicate sets. n = 4 mice for each time point for each strain. For Bmal1 center of gravity in the cornea (a), the statistics were run with the C57BL data as a double-plot value (CT + 24 h) so that values would be on the same portion of the cycle for analysis. For Per2 expression in the cornea of C3Sn mice (a), dashed line simply connects the mean points. Phase differences between strains were observed in the corneas and retinas of mice in light: dark cycles, but liver clock gene expression showed no difference.
In the retina, the profiles of the clock genes in light: dark cycles were similar in phase as to what was observed in constant darkness, with the C3Sn retinas leading the phase of C57Bl/6 retinas in all four genes (Figure 4c). In contrast to constant darkness, Bmal1 expression was detected as rhythmic in light: dark conditions with the center of gravity of C3Sn expression being 5.6 ± 1.8 h earlier than in C5Bl/6. The difference in phase was 8.3 ± 1.6 h for Per2, 7.7 ± 1.9 h in Per1, and 7.4 ± 1.9 h for Dbp (p < 0.05, two-tailed t-test on center of gravity).
Discussion
Here we demonstrate that the presence of a melatonin-proficient retina (C3Sn) can cause the photoentrainment of an Opn5–/– cornea ex vivo, but a melatonin-deficient retina (C57Bl/6) cannot. We further show that blocking melatonin signaling pharmacologically or using a retina with the rd1 mutation blocks this effect. We conclude that rhythmic melatonin signals from a biologic source can act as an entraining cue on the circadian clocks of the cornea. Alternatively, melatonin could also be acting as a synchronization signal within the retina, coordinating rhythmic release of another entraining signal. The peak values of melatonin detected in our ELISA of retina cultures are lower than previously reported levels (Tosini and Menaker, 1998; Tosini and Menaker, 1996). This likely results from (1) our static culture compared with flow-through conditions (Tosini and Menaker, 1998), (2) our single dark time point may not have represented the peak concentration compared with the regular collection frequency in flow-through systems, and (3) the volume we collected from one retina was 1.2 mL of static media which may not represent dynamic physiologic volumes, especially at the retina: cornea junction. However, we observed a clear difference in melatonin present in the media comparing dark and light in all but the degenerated adult retinas (Suppl. Fig. 1C).
When tested in vivo, we saw a difference in phase of the expression of four clock genes in the cornea of melatonin-proficient mice compared with melatonin-deficient mice, but no difference in another peripheral tissue, the liver. We also corroborate previous reports of melatonin-proficient retinas displaying a different circadian phase than in melatonin-deficient mice; although, here we analyzed RNA transcript, not protein abundance (Dinet et al., 2007; Dinet and Korf, 2007).
It is important to note that the source of melatonin in vivo that exerts the effect on the cornea is unclear. In fact, it may not be a solitary source. The primary source of systemic melatonin is the pineal gland, but there are a number of other sources of endogenous melatonin in mammals (Simonneaux and Ribelayga, 2003). The retina and the Harderian glands are also both sources of rhythmic melatonin, both show higher levels of melatonin in the dark, and both are in close proximity to the cornea (Bubenik et al., 1978). The lack of blood vessels in the majority of the cornea suggest that the tissue is unlikely to receive the melatonin signal directly from the blood supply. However, melatonin can readily permeate through the layers of the cornea (Bessone et al., 2020), and melatonin itself is a very diffusible molecule which penetrates cell membranes (Yu et al., 2016). Therefore, melatonin from the retina diffusing through the aqueous humor and vitreous, or melatonin from the Harderian gland present on the ocular surface are both possible sources.
It is clear that melatonin is not the sole synchronizing factor in the cornea. If this were the case, it might be expected that the phases of individual corneas from melatonin-deficient C57Bl/6 mice would display scattered phases in vivo (Figures 3 and 4). Rather, the phases of corneal circadian clocks in the absence of melatonin are tightly regulated, albeit to a unique phase. In addition, the Per2 phases of the corneas exposed to C3Sn retinas ex vivo adopt a phase (peaking slightly after the light to dark transition) which is different from the phase measured in vivo (peaking approximately at the light to dark transition). However, it should be noted that the phase of the protein translation reporter, Per2Luciferase, may be different than the transcriptional phase measured by RT-PCR. Other potential synchronizing factors of the healthy corneas in vivo include glucocorticoids and temperature (Pezük et al., 2012; Buhr et al., 2010). Light appears to only act as a synchronizing cue when the cornea is damaged (Díaz et al., 2020).
Finally, this work serves as another reminder that researchers always must be aware of the limitations of the model organism with which they work. The C57Bl6/J mouse model is likely the most commonly used mouse strain. While the lack of melatonin has not rendered this mouse strain overtly unhealthy or arrhythmic, our work highlights subtle differences between melatonin-deficient and melatonin-proficient strains.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304211032385 – Supplemental material for Melatonin Adjusts the Phase of Mouse Circadian Clocks in the Cornea Both Ex Vivo and In Vivo
Supplemental material, sj-pdf-1-jbr-10.1177_07487304211032385 for Melatonin Adjusts the Phase of Mouse Circadian Clocks in the Cornea Both Ex Vivo and In Vivo by Alex V. Huynh and Ethan D. Buhr in Journal of Biological Rhythms
Footnotes
Acknowledgements
This work was supported by NIH R01 GM124246 and R01 EY026921 to EDB, the Latham Vision Research Innovation Award, an unrestricted grant to the University of Washington Department of Ophthalmology from Research to Prevent Blindness. Much of this work was inspired by the work of Dr. Michael Menaker and is dedicated in his memory.
Conflict Of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
NOTE
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
