Abstract
The circadian clock represents an anticipatory mechanism, well preserved in evolution. It has a critical impact on most aspects of the physiology of light-sensitive organisms. These rhythmic processes are governed by environmental cues (fluctuations in light intensity and temperature), an internal circadian timing system, and interactions between this timekeeping system and environmental signals. Endocrine body rhythms, including hypothalamic-pituitary-thyroid (HPT) axis rhythms, are tightly regulated by the circadian system. Although the circadian profiles of thyroid-releasing hormone (TRH), thyroid-stimulating hormone (TSH), thyroxine (T4), and triiodothyronine (T3) in blood have been well described, relatively few studies have analyzed molecular mechanisms governing the circadian regulation of HPT axis function. In this review, we will discuss the latest findings in the area of complex regulation of thyroid gland function by the circadian oscillator. We will also highlight the molecular makeup of the human thyroid oscillator as well as the potential link between thyroid malignant transformation and alterations in the clockwork.
Keywords
Most physiological processes in the body oscillate in a daily fashion. These include cerebral activity (sleep-wake cycles), metabolism and energy homeostasis, heart rate, blood pressure, body temperature, digestive and renal activity, and immune system function (Marcheva et al., 2013). Among other essential aspects of physiology, endocrine body rhythms are tightly regulated by the circadian system. Consequently, the hypothalamic-pituitary-thyroid (HPT) axis is under circadian oscillator control at multiple levels, from hypothalamic neurons to the thyroid gland (Fig. 1). In this review, we aim at highlighting 1) circadian aspects of the regulation of thyroid gland function, 2) properties of thyroid cellular clocks, and 3) alterations of the thyroid clockwork in follicular thyroid malignancies.
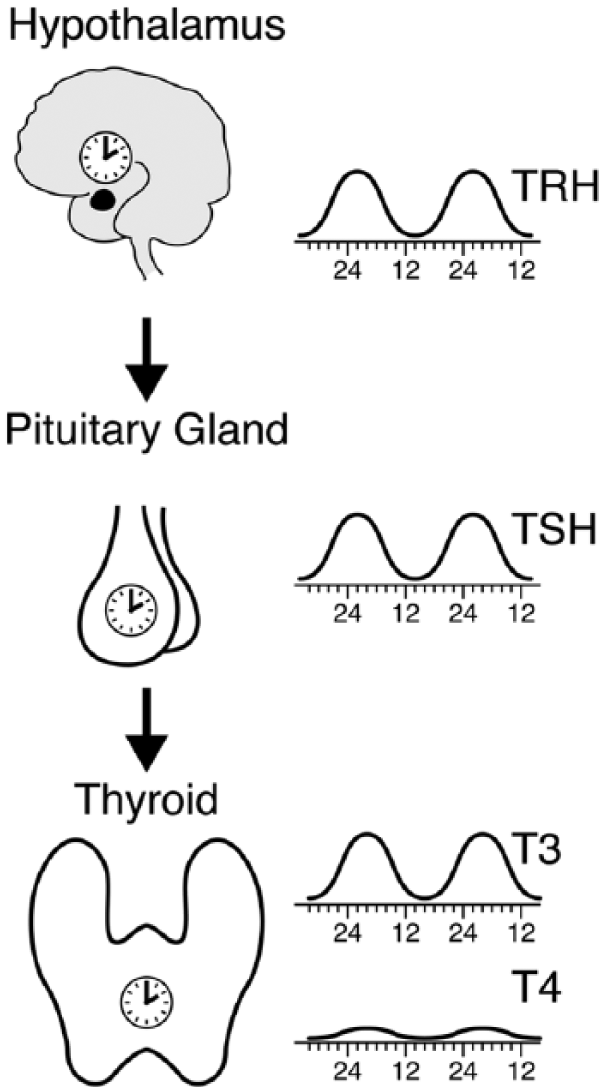
Circadian control of the hypothalamic-pituitary-thyroid (HPT) axis. The circadian system exerts control at all levels of the HPT axis: 1) thyroid-releasing hormone (TRH) mRNA exhibits circadian oscillations, probably due to direct control by the SCN, with a peak around 0200 h; 2) thyroid-stimulating hormone (TSH) has a strongly oscillatory profile in the blood, with a peak at 0200 h to 0400 h, and TSH mRNA may be under circadian regulation (see text); and 3) triiodothyronine (T3) peaks with a phase delay of about 1.5 h compared with the phase of TSH (see text for explanation), while thyroxine (T4) exhibits weaker circadian amplitude and no clear correlation with the phase of TSH (Russell et al., 2008).
Molecular Makeup of Central and Peripheral Clocks
Daily rhythms of behavior and physiology are not just acute responses to timing cues provided by the environment but are also driven by an endogenous circadian timing system that allows for the anticipation of geophysical time changes. The master pacemaker, located in the suprachiasmatic nucleus (SCN) in the ventral hypothalamus, coordinates all overt rhythms in the body through neuronal and humoral outputs. In humans, the SCN consists of 2 clusters, with about 100 000 neurons in the bilateral structure, with each neuron comprising a self-sustained, cell-autonomous molecular oscillator (Albrecht, 2012; Hofman and Swaab, 2006). Research conducted during the past years has shown that virtually all body cells possess their own clocks that, in terms of their molecular makeup, are indistinguishable from those operating in SCN neurons. While the oscillators of SCN neurons are synchronized primarily through photic signals received via the retinohypothalamic tract, feeding time, as driven by rest-activity rhythms, appears to be the dominant timing cue (Zeitgeber) for entrainment of peripheral clocks (Dibner et al., 2010). The current molecular model for the generation of circadian oscillations is based on interlocked negative feedback loops of gene expression. In mammals, the major loop comprises 2 PAS-domain helix-loop-helix transcriptional activators BMAL1 and CLOCK, forming a heterodimer that activates the transcription of the negative core-clock limb actors, Per and Cry, which accumulate and negatively feedback on their own transcription. This negative feedback loop generates cycles of about 24 h in gene expression. On top of the transcription-translation loop, posttranslational events such as the control of protein phosphorylation, sumoylation, acetylation, O-GlcNAcylation, degradation, and nuclear entry contribute critically to the generation of daily oscillations in clock gene products (Asher et al., 2008; Asher et al., 2010; Cardone et al., 2005; Diernfellner and Brunner, 2012; Durgan et al., 2011; Gallego and Virshup, 2007; Li et al., 2012; Vanselow and Kramer, 2007).
Htp Axis
In humans, the thyroid (from Greek thyreos, “shield,” and eidos, “form”) is a large endocrine gland (12–20 g in size) with 2 symmetric lobes, situated in the base of the neck (Longo et al., 2011). The thyroid gland produces 2 related hormones, thyroxine (T4) and triiodothyronine (T3), which act through nuclear receptors in target tissues (thyroid hormone receptor α (TRα), and TRβ; Evans and Mangelsdorf, 2014; Zhao et al., 2014). T3 and T4 play pivotal roles in cell differentiation during development. Throughout adult life, T3 and T4 maintain body temperature and metabolic homeostasis by their direct impact on key pathways that regulate energy storage and expenditure (Bonde et al., 2014; Mullur et al., 2014; Webb, 2010; Zhang et al., 2014). T4 is the predominant thyroid hormone form and is synthesized in the epithelial cells forming thyroid follicles. T4 is rapidly transformed to the more active T3 by the activating iodothyronin deiodinases, DIO1 and DIO2, with DIO2 being primarily responsible for the rapid increases of T3 in serum and in specific tissues (Maia et al., 2005; Longo et al., 2011). Deiodination of T4 by the inactivating enzyme DIO3 leads to the production of the inactive hormone, reverse T3 (rT3; Brent, 2012; Mullur et al., 2014). T4 is secreted from the thyroid gland into the circulation in an approximately 20-fold excess over T3, and it is much more stable with a half-life of about 7 d, in comparison to 0.75 d for T3. While 99.98% of T4 and 99.7% of T3 are bound to circulating plasma proteins, including thyroxine-binding globulin, transthyretin, and albumin, the homeostatic mechanisms regulating the thyroid axis are mainly directed toward maintenance of normal concentrations of free thyroid hormone fractions, FT4 and FT3 (Longo et al., 2011). The levels of circulating FT4 and FT3 represent essential clinical parameters for identification of thyroid-related pathologies. Therefore, considerable effort is being undertaken to improve the accuracy of FT4 and FT3 dosage to allow for personalized diagnosis of thyroid homeostasis (Dietrich et al., 2012).
The HPT axis is a classic example of an endocrine feedback loop. Thyroid-stimulating hormone (TSH), a polypeptide hormone secreted by the thyrotrope cells of the anterior pituitary, plays a pivotal role in controlling the HPT axis and serves as the most reliable physiologic marker of thyroid hormone action. TSH is modulated by the hypothalamic tripeptide thyrotropin-releasing hormone (TRH) and directly stimulates thyroid gland production of T4 and T3 via the TSH receptor on the thyroid follicle cell membrane (Fig. 1). In turn, T4 and T3 feedback to inhibit the hypothalamic production of TRH and pituitary production of TSH. Reduced levels of thyroid hormone increase basal TSH production and enhance the TRH-mediated stimulation of TSH, while high thyroid hormone levels rapidly and directly suppress TSH gene expression and inhibit the stimulation of TSH by TRH (Dietrich et al., 2012; Kalsbeek and Fliers, 2013; Mullur et al., 2014).
Circadian Regulation of Thyroid Function
Based on current understanding, the SCN exerts control over the endocrine system either through the sleep-wakefulness cycle or independently of sleep, through neural and endocrine roots (reviewed in Hastings et al., 2007; Kalsbeek and Fliers, 2013). Consequently, most hormones are reported to exhibit pronounced circadian profiles with variable amplitudes in blood, superimposed on ultradian and pulsatile rhythms over 24 h (Haus, 2007). While some hormone oscillations are remarkably sleep dependent (e.g., prolactin and growth hormone), others are sleep independent and persist in a constant routine protocol (e.g., TRH, cortisol, melatonin). The connection between circadian and endocrine systems is reciprocal, with melatonin, corticosteroids, estrogen, and androgens representing important synchronizers for the SCN or peripheral clocks (Haus, 2007).
The HPT axis is subject to complex time regulation at all levels, exhibiting pulsatile, ultradian, circadian (Fig. 1; Haus, 2007; Kalsbeek et al., 2000; Kalsbeek et al., 2005; Kalsbeek and Fliers, 2013), and circannual rhythms (Wood and Loudon, 2014; Yoshimura, 2013). TRH, TSH, FT3, and FT4 all exhibit circadian profiles in the circulation. In human subjects, TRH, TSH, and T3 show a clear nocturnal peak around 0200 h to 0400 h, while FT4 exhibits a much less evident circadian profile (Fig. 1; Haus, 2007; Kalsbeek and Fliers, 2013; Russell et al., 2008). Melatonin may modulate the response of the anterior pituitary to TRH and of thyroid to TSH (Mazzoccoli et al., 2004). The SCN directs neuronal outputs into the paraventricular nucleus of the hypothalamus and may be responsible for the circadian pattern of TRH mRNA (reviewed in Kalsbeek and Fliers, 2013). Numerous studies indicate that TSH has a pronounced circadian rhythm in the blood (Fig. 1). A recent study on diurnal variations of TSH levels in the circulation, performed with high temporal resolution in 117 human subjects, confirmed high-amplitude circadian variations of hormone levels with pronounced nocturnal peaks (Roelfsema et al., 2014). Moreover, it indicated that the amplitude of these oscillations was not influenced by sex, body mass index (BMI), or age, while the onset of the nocturnal surge was significantly phase delayed with increasing BMI and phase advanced with aging, which is in agreement with previously observed clock property changes upon aging (Pagani et al., 2011; Roelfsema et al., 2014). Of note, evaluation of the TSH circadian profile can be used in the clinic as an important parameter for the diagnosis of central hypothyroidism manifestations, often masked by other anterior pituitary hormone secretion deficiencies (Grunenwald and Caron, 2014). The relationship between the circadian profile of TSH and those of T3 and T4 has been established by cross-sectional study in healthy individuals, showing a strong correlation between TSH and T3 levels, with the T3 hormone peak lagging approximately 90 min behind the peak of TSH (Russell et al., 2008). Such correlation and phase delay for T3 as compared with TSH may suggest that TSH determines T3 levels through either stimulating T3 release from thyroid or by increasing peripheral conversion of T4 into T3. By contrast, no correlation was observed between T4 and TSH profiles (Russell et al., 2008).
While the circadian profile of TSH in the circulation has been well studied, the underlying mechanism for this phenomenon is not yet clear. Recent findings indicate that the Tshb transcript, coding for the β-subunit of TSH, might exhibit circadian oscillations antiphasic to those of Reverbα in a synchronized cultured mouse thyrotroph cell line (TaT1.1; Aninye et al, 2014). The authors proposed a plausible mechanism with NCOR1 co-interacting with Reverbα rather than with the thyroid receptor (TR), in order to regulate circadian TSH and T3 secretion independently of the TR (Aninye et al., 2014). In addition, pituitary expression of the TSH subunits GSUa and TSHb in red drum Sciaenops ocellatus exhibited strongly circadian profiles, with a 6- to 12-h phase shift as compared with the phase of T4 (Jones et al., 2013). Moreover, the pituitary expression pattern of the enzyme Dio3 was circadian and mirrored that of TSH subunits, further underscoring circadian regulation of Dio3 and TSH on transcriptional levels as well as the potential importance of Dio3 expression for TSH inactivation (Jones et al., 2013). Interestingly, a conserved D-element in the TSHb promoter was proposed to be directly bound by Thyrotroph Embryonic Factor (TEF) in Soay sheep, with TEF binding affinity being higher than that of DBP. Thus, the sensitivity of TSHb D-element to TEF might be decisive for photoperiod responsiveness in mammals (Dardente et al., 2010). Taken together, these findings provide the first body of evidence for the transcriptional regulation of TSH and thyroid hormones by the circadian clock. On the other hand, thyroid hormone receptor–associated protein-150 (TRAP150) has been demonstrated to affect critically core clock function. TRAP150 was identified as a selective co-activator of the BMAL1-CLOCK complex, which in turn oscillates under BMAL1-CLOCK transcriptional control. Down-regulation of TRAP150 led to low-amplitude, long period cycles, thus identifying TRAP50 as a crucial element of the positive feedback loop (Lande-Diner et al., 2013).
An additional level of circadian regulation of the HPT axis is the expression of thyroid hormone receptors. Importantly, Rev-erbα is encoded on the opposite strand of the Trα1 gene and its splice variant Trα2 in rodents and in humans. Moreover, Rev-erbα mRNA was demonstrated to inhibit the splicing reaction generating TRα2 in vitro (Chawla and Lazar, 1993; Munroe and Lazar, 1991). TRα is rhythmically expressed in mouse liver, where it was shown to be directly bound by BMAL1 (Rey et al., 2011) and REVERBα/β (Solt et al., 2012). Both TRα and TRβ exhibit pronounced circadian oscillations in mouse white adipose tissue and in brown adipose tissue (Yang et al., 2006). By contrast, the profile of TRα is constant over 24 h in skeletal muscle, and the same is true for TRβ in liver and skeletal muscle. These findings may suggest differential and complex roles for TRα and TRβ, in addition to other members of the nuclear receptor family, in orchestrating body metabolism by the circadian system (Yang et al., 2006; Zhao et al., 2014).
Cell-Autonomous Clocks are Functional in Human Primary Thyrocytes
Beyond the presence of clocks in most cell types in the body, this rhythm-generating circuitry has been revealed in cultured immortalized fibroblasts (Balsalobre et al., 1998; Nagoshi et al., 2004). These self-sustained, cell-autonomous clocks can be synchronized in vitro by a variety of stimuli including serum shock, dexamethasone, forskolin, insulin (Balsalobre et al., 2000), glucose (Hirota et al., 2002), or temperature cycles (Saini et al., 2012). Using this in vitro synchronization approach, molecular clocks have been revealed in a variety of tissue explants and primary cells from rodent (Marcheva et al., 2010; Yoo et al., 2004) and human (Brown et al., 2005; Pulimeno et al., 2013) origin. Recently, high-amplitude circadian oscillators were described in cultured human primary thyrocytes established from healthy thyroid tissue (Mannic et al., 2013). Primary human thyrocytes transduced with Bmal1-luciferase lentivectors exhibited pronounced circadian oscillations following dexamethasone synchronization (Fig. 2A, solid line). In line with the outcome of reporter experiments, endogenous clock gene expression measurements by quantitative polymerase chain reaction suggested that the core-clock transcripts exhibit circadian oscillatory patterns in synchronized thyrocytes. Both approaches suggested a circadian period of about 27 h for these cells, which is longer in comparison to primary human skin fibroblasts (24.5 h) or human pancreatic islets (23.6 h; Brown et al., 2005; Pulimeno et al., 2013). Of note, circadian oscillation period length varies significantly among different mouse organ explants cultured in vitro (Yoo et al., 2004), possibly reflecting differences in general transcriptional rates and cell sizes among these organs (Dibner et al., 2009; Schmidt and Schibler, 1995).
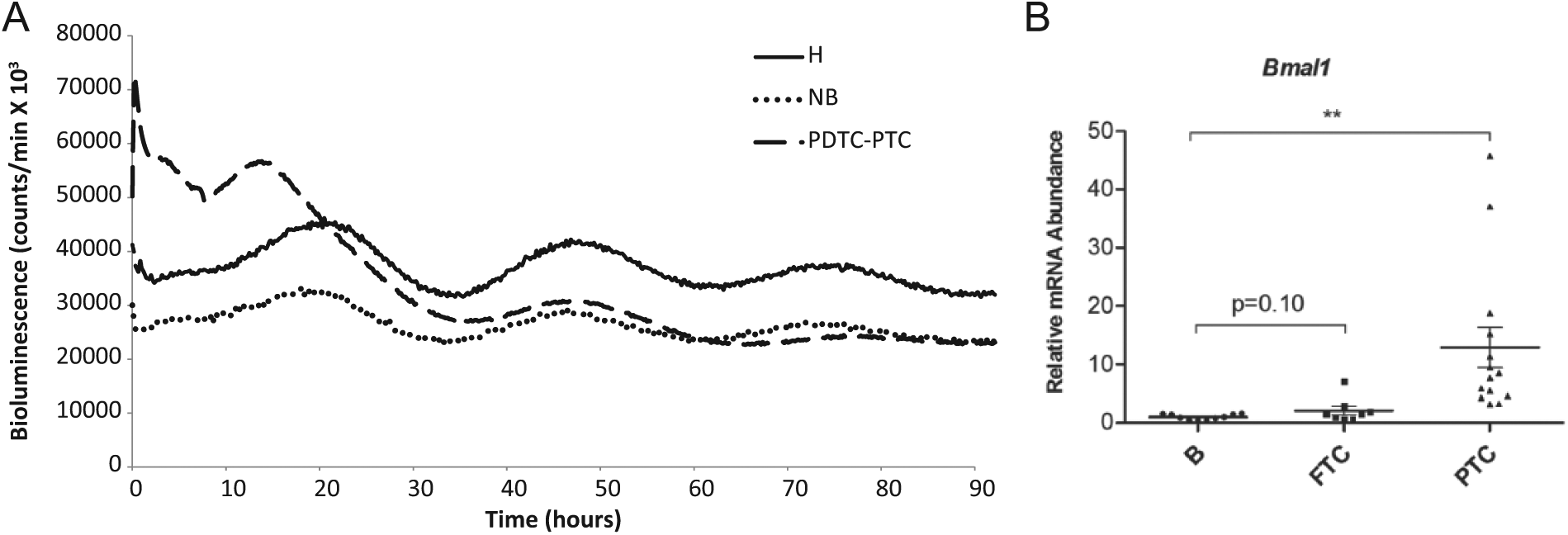
Thyroid cellular clocks in physiology and in follicular malignancies (adapted from Mannic et al., 2013). (A) Circadian oscillations of Bmal1-luciferase reporter in human primary thyrocytes. Bmal1-luciferase oscillations were recorded in in vitro synchronized human primary thyrocytes derived from healthy tissue, benign thyroid nodule, or malignant nodule with combined poorly differentiated thyroid carcinoma (PDTC) and papillary thyroid carcinoma (PTC) diagnostics, or PDTC-PTC (H, NB, and PDTC-PTC: solid, dotted and dashed lines, respectively). Cells were transduced with Bmal1-luciferase lentivectors and synchronized 4 d later with a dexamethasone pulse, and Bmal1-luciferase bioluminescence profiles were recorded from 2 parallel dishes per donor; n = 3 donors for thyrocytes derived from healthy tissue and benign nodules; n = 1 donor for PDTC-PTC. The curves represent an average of all the performed recordings for each cellular type. (B) Expression of Bmal1 in tissue biopsies of benign thyroid nodules, follicular thyroid carcinoma (FTC), and PTC. Quantitative polymerase chain reaction for Bmal1 was performed on cDNAs obtained from tissue samples from nodules with benign (n = 10), FTC (n = 8), or PTC (n = 14) postoperative diagnoses. The relative mRNA expression was normalized to the average of Gapdh and 9S and then reported to the mean value of the transcript levels in nodular benign tissues. Results were expressed as mean ± SEM. Mann-Whitney test was applied to assess the significance in the transcript expression level differences between FTC or PTC as compared with benign nodules (*p <0.05, **p < 0.01, ***p < 0.001).
Thyroid Follicular Malignancies and the Clock Connection
In addition to an intrinsic circadian oscillator, another fundamental attribute of a cell is its ability to divide and multiply. While the circadian clock is the body’s molecular timekeeping system, the cell division clock executes a precise temporal control mechanism with multiple checkpoints for proper cell division. Recent findings reveal that circadian and cell cycle clocks might be linked (Bieler et al., 2014; Feillet et al., 2014; Kowalska et al., 2013; Matsuo et al., 2003; Nagoshi et al., 2004). Furthermore, clock genes have been linked to the cell cycle, DNA damage, apoptosis control, and carcinogenesis (Altman et al., 2010; Fu et al., 2002; Hua et al., 2006).
Thyroid cancer represents the most common endocrine malignancy and accounts for 1% of all human malignancies. The incidence of differentiated thyroid carcinomas has increased in the past decades to the extent that now they are estimated to rank third in the rate of increase in the United States (Siegel et al., 2014). Of note, an association between insomnia and a higher incidence of thyroid cancer was suggested by an epidemiological study in postmenopausal nonobese women (Luo et al., 2013). The majority of thyroid tumors (85%) are well differentiated, whereas a small proportion (about 5%-7%) lose their features of cell origin and are classified as poorly differentiated or undifferentiated/anaplastic (Ibrahimpasic et al, 2013). At present, multiple ultrasonographic, immunohistochemical, or genetic features have been associated with malignancy, but none is sufficiently accurate to distinguish follicular adenoma from carcinoma with high probability when viewed independently (Sigstad et al., 2012). Therefore, surgery is required for all lesions diagnosed as a suspicious follicular neoplasm and for some of the lesions of undetermined significance (Cibas and Ali, 2009). The search for preoperative markers for thyroid malignancies is therefore of the utmost clinical importance.
Of note, alterations in clock characteristics were recently observed by us in thyroid follicular malignancies in in vitro synchronized primary thyrocytes (Mannic et al., 2013; compare dashed and dotted lines in Fig. 2A), as well as in tissue biopsies, with Bmal1 being up-regulated (Mannic et al., 2013; Fig. 2B) and Cry2 down-regulated in follicular thyroid carcinoma (FTC) and papillary thyroid carcinoma (PTC) nodules. Hence, while the transition from benign to malignant thyroid nodules does not completely ablate the functional thyroid clock, it is certainly associated with clock alterations, probably restricted to first cycle kinetics (Fig. 2A). Moreover, dramatic changes of Bmal1 levels in PTC nodules (Fig. 2B) further underscore the link between cancerous transformation and changes in the circadian clock or in the individual core-clock genes and may potentially be employed in clinics (Mannic et al., 2013).
Future Perspectives
Perturbation of circadian rhythms in animals and humans, such as shift work and social jetlag situations, has been well-documented to interfere with numerous aspects of health and to provoke serious conditions including metabolic diseases, cardiovascular disorders, and malignant transformations (Levi et al., 2010; Marcheva et al., 2013). Exploring the mechanisms of circadian regulation of thyroid function using primary thyroid explants and cell culture, as well as unravelling the role of the circadian system in thyroid malignant transformation, might be of importance for future initiatives. While the circadian regulation of the HPT axis has been well explored, surprisingly few studies have so far addressed the reciprocal connection (i.e., the effects of thyroid hormones on the body’s circadian clocks). Therefore, the focus on potential synchronization effects of TRH, TSH, and thyroid hormones on the circadian clockwork in different organs represents an interesting direction. In a broader context, exploring the impact of circadian misalignments on hormone secretion patterns, including those of TSH and T3, that can induce detrimental effects on body metabolism is of utmost scientific and clinical importance. Future studies are necessary for deciphering the mechanisms underlying molecular regulation of the endocrine system by clocks and finding novel mechanisms of how clock-controlled processes function in pathophysiological situations.
Footnotes
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
