Abstract
Following decades of research under controlled laboratory conditions, there has been growing interest in the recent past to study circadian rhythms in nature. Recent studies conducted under natural conditions have been fruitful in exploring several characteristics of circadian rhythms that remained cryptic and previously masked under standard laboratory conditions, reemphasizing that the complexity of circadian rhythms in nature increases multifold under the influence of multiple zeitgebers. However, our understanding of the contributions of different zeitgebers in shaping various rhythm characteristics still remains elusive. Previously, Vaze et al. reported that chronotype differences between the morning emerging (early) and evening emerging (late) populations of Drosophila melanogaster are considerably enhanced under natural conditions compared to standard laboratory conditions. In the present study, we assess the role of 2 primary zeitgebers in nature—light and temperature—individually and in unison in driving chronotype differences. We report that when provided independently, temperature cycles enhance divergence between the early and late chronotypes more strongly than light, but when together, light and temperature appear to act antagonistically and that appropriate phase difference between light and temperature cycles is essential to promote chronotype divergence. Thus, our study highlights the importance of light and temperature, as well as their interaction with circadian clocks in mediating early and late chronotypes in fruit flies D. melanogaster.
Rhythmic regulation of behavioral and physiological processes ensuring their occurrence at appropriate times of the day (or life stages), thus enhancing survival and ultimately fitness of organisms, is believed to have led to the evolution of circadian clocks (and other such biological timing systems) (Dunlap et al., 2004). Circadian clocks govern such rhythmic processes at both populational (e.g., adult emergence rhythm in insects) and individual (e.g., activity/rest rhythm) levels (Saunders, 2002; Dunlap et al., 2004).
The pattern of adult emergence in Drosophila melanogaster under 12:12-h laboratory light/dark cycles (LD12:12) assumes a shouldered unimodal waveform with a prominent primary peak around “lights-on” and a less prominent shoulder close to “lights-off” (Sheeba et al., 2001). Landmark studies by Pittendrigh and coworkers on the emergence rhythm in Drosophila and other insects (Pittendrigh, 1967, 1981; Pittendrigh and Minis, 1971; Skopik and Pittendrigh, 1967) have helped establish some of the conceptual foundations of circadian rhythms. Two core circadian clock genes, period and timeless, were also identified in mutagenesis screens for arrhythmic adult emergence phenotypes (Konopka and Benzer, 1971; Sehgal et al., 1994), thus highlighting the contribution of emergence rhythm in studying circadian clocks. Despite being a key readout, emergence rhythm has received less attention than the rest/activity rhythm in the domains of exploring molecular and neuronal mechanisms underlying circadian rhythms.
The nature of emergence waveforms varies across insect species. The primary peak of emergence occurs close to dawn in the yellow dung fly Scopeuma stercoraria (Lewis and Bletchley, 1943), Queensland fruit fly Dacus tryoni (Myers, 1952; Bateman, 1955), and in moths Pectinophora gossypiella (Pittendrigh and Minis, 1964) and Heliothis zea (Callahan, 1958). Flour moth Anagasta kuhniella primarily emerge in the post-afternoon and early evening hours (Bremer, 1926; Scott, 1936; Moriarty, 1959), while in some Chironomids, emergence is observed during the night (Palmen, 1955). Irrespective of the differences in the waveform of emergence across insect species, one of its noticeable aspects is the restriction of emergence to specific times of the day, a phenomenon referred to as “gating” (Skopik and Pittendrigh, 1967). Gating results in a specific duration becoming an “allowed zone” for emergence (gate width), during which a substantial proportion of flies emerge out of their pupae, and another interval becoming a “forbidden zone,” during which little or no emergence occurs (Pittendrigh, 1966). This gating is so stringent that flies that complete their development after the closure of emergence gate remain inside their puparium until the next gate opens on the following day (Skopik and Pittendrigh, 1967). Depending on the ecology, emergence at unfavorable hours of the day may be maladaptive to individuals due to the presence of predators, lack of food availability, or unfavorable environmental conditions like high temperature and low humidity resulting in desiccation, all of which reduce survivability of the newly emerged individuals. Thus, gating is likely to be an adaptation to the selection pressures imposed by various environmental factors, but the specific roles of different zeitgebers in nature in modulating gating remain poorly understood.
There have been previous attempts to use laboratory selection approach to study evolution of the circadian clocks’ phasing of adult emergence. Pittendrigh (1967) and Pittendrigh and Minis (1971) selected for “early” and “late” emerging strains of Drosophila pseudoobscura and moth Pectinophora gossypiella under LD12:12. Several years later, Pittendrigh and Takamura (1987) imposed a similar selection on Drosophila auraria but under a very short photoperiod of LD1:23. In all cases, the populations responded to selection with the early and late emerging populations exhibiting phase divergences (difference in mean emergence phase) of about 4 h in D. pseudoobscura, about 5 h in P. gossypiella, and about 6 h in D. auraria. All 3 species also showed correlated changes in their circadian clock periods albeit in different directions. In D. pseudoobscura and P. gossypiella, the early flies had longer clock periods and the late flies shorter clock periods, while the early and late flies of D. auraria had shorter and longer clock periods, respectively. Although Pittendrigh attributed this paradox to a slave oscillator (driven by a master oscillator) controlling emergence rhythm and to differential coupling between pacemaker and zeitgeber arising from the differences in light regimes used during selection, the selection protocols used in these studies were limited by a lack of (a) replication at the populational level within a selection regime, (b) information on population ancestry, and (c) precise details of selection protocols employed. Over the decades, the field of experimental evolution has demonstrated that even moderately small differences in the above-mentioned factors may considerably affect the response of selected populations (evolutionary trajectories of populations greatly differ depending on the standing genetic variation harbored by the baseline populations and microenvironmental differences), thus leading to misinterpretation of data (reviewed in Sharma and Joshi, 2002; Prasad and Joshi, 2003). Improvising on the methodological aspects of previous studies and incorporating the basic prerequisites of experimental evolution, we initiated a long-term laboratory selection study aimed at examining the evolutionary trajectory of circadian clocks in response to selection for different phases of adult emergence and subsequently their roles in mediating chronotype differences. The populations used in our study comprise 4 independent replicates of large random mating populations and therefore harbor sufficient genetic variation, thus providing an ideal platform for various kinds of evolutionary studies. This is in contrast to studies performed on inbred, so-called wild-type strains, which may provide an inaccurate picture of the underlying genetic architecture of the phenotypes under study.
Contrary to previous studies by Pittendrigh (1967) and Pittendrigh and Minis (1971), the early and late populations in our study evolved shorter and longer circadian clock periods (τ) (Kumar et al., 2007a). This ambiguity can be attributed to the possible limitations in the selection protocol employed in former studies (discussed earlier). Also, the early and late populations evolved dominant morning and evening oscillators (Kumar et al., 2007b), respectively, and divergent photic phase response curves (PRCs) for emergence rhythm (Kumar et al., 2007a), and under laboratory 12:12-h LD cycles, the 2 populations differentially used light in the morning and evening hours to exhibit their respective emergence chronotypes (Vaze et al., 2012b). Previous studies have reported correlation between circadian periods and chronotypes in humans (Duffy et al., 1999; Roenneberg et al., 2003). Also, morning and evening chronotypes in humans are correlated with the nature and duration of light exposure (Duffy et al., 2001; Roenneberg et al., 2003; Goulet et al., 2007). Taken together, these studies suggest that chronotypes of the early and late populations can be attributed to differences in the underlying circadian clocks and their interaction with environmental cycles.
Several recent studies under semi-natural conditions (SN) have explored the effects of multiple zeitgebers on the emergence and activity rhythms of Drosophila (De et al., 2012, 2013; Menegazzi et al., 2012, 2013; Vanin et al., 2012; Prabhakaran et al., 2013). Although these studies have revealed several interesting and unexplored aspects of circadian rhythms that remained masked under standard laboratory conditions, they have only managed to convince us about the complexity of the circadian clock-zeitgeber interaction, failing to provide further insights into the nature of such interactions. Vaze et al. (2012a) reported that environmental cycles in nature considerably enhanced chronotype differences between the morning (early) and evening (late) selected populations of D. melanogaster compared to standard laboratory condition. Adding to the enhanced chronotype differences, emergence rhythms of the early and late populations also exhibited significant reduction in gate widths in SN, suggesting a causal role of multiple zeitgebers in modulating the emergence waveform.
Following up on the reported observations of Vaze et al. (2012a), by simulating various zeitgeber profiles in the laboratory, we intended to test the following hypotheses: (a) stepwise change in light cycles mimicking twilight conditions in nature are sufficient and more effective than abruptly changing light cycles to promote phase divergence between early and late emergence chronotypes, (b) stepwise change in temperature cycles mimicking nature are sufficient and more effective than abruptly changing temperature cycles to promote phase divergence, (c) combined cycling of light and temperature promotes phase divergence better than light and temperature independently, and (d) the phase difference between light and temperature cycles observed in nature plays a role in promoting phase divergence.
Materials and Methods
Experimental Populations
Ancestry, maintenance, and selection protocols for the 3 sets of populations used in the present study are described in detail elsewhere (Kumar et al., 2007a). Briefly, 4 replicates of the earlyi and latej (i = j = 1..4) populations were initiated from 4 controlk (k = 1..4) populations such that the early and late populations with a given subscript share a common ancestry with that of the control population of the same subscript. In other words, the early1 and late1 populations were initiated from the control1 population and thus share a common ancestry. All fly populations were maintained as independent outbred units (no mating and thus no gene flow between them) in Plexiglas cages (25 × 20 × 15 cm3), each housing approximately 1200 flies (sex ratio = 1:1) and provided with ad libitum banana-jaggery (BJ) medium, replaced with fresh media every alternate day. The maintenance regime comprised 12:12-h light/dark cycles (LD12:12) with ~0.4 Wm−2 light during the light phase and dim red light (λ > 650 nm) during the dark phase, with 25 ± 0.5 °C (mean ± SD) temperature and 75% ± 5% relative humidity.
All populations were maintained on a 21-day discrete (nonoverlapping) generation cycle. The parent stocks were provided with yeast paste for 3 days prior to egg collection, following which ~300 eggs were collected and dispensed into culture vials containing ~6 mL BJ media. Preadult rearing conditions were the same as described above, and from the initiation of emergence, flies emerging in the morning between External Time (ExT) 03 to 07 h (external time 00 h represents the midpoint of the dark phase; Daan et al., 2002) were collected for 4 consecutive days and formed the early populations, while flies emerging in the evening between ExT13 and 17 for 4 consecutive days formed the late populations. The control populations were not subjected to any conscious selection pressure and comprised flies emerging throughout the day for 4 consecutive days.
To minimize the effects of nongenetic inheritance (Bonduriansky and Day, 2009) due to different selection regimes, all fly stocks were subjected to 1 generation of relaxed selection pressure and are called the “standardized populations.” All assays described here were performed on the standardized populations.
Adult Emergence Assay
For adult emergence assay, eggs collected from the standardized populations were dispensed into 10 vials (per population) containing BJ medium with ~300 eggs per vial, following which the vials were transferred to various experimental regimes (discussed later). These vials were monitored for the initiation of emergence and were henceforth subjected to 2-h interval checks to record the number of adult flies emerging from each vial, and the assay was continued for at least 5 consecutive days.
Details of different experimental regimes used in the study are provided in Table 1. Briefly, experimental regimes primarily comprised cycles of abrupt and stepwise changes in light and temperature either individually or in combination. Abruptly changing light cycles (ALC) employed rectangular waveforms of light with a sudden upshift from 0 Wm−2 to 0.4 Wm−2 marking lights-on (ExT06) and a sudden down-shift to 0 Wm−2 at lights-off (ExT18). Similarly, abruptly changing temperature cycles (ATC) employed rectangular waveforms of temperature cycling between 18 °C and 28 °C with sudden temperature upshift and downshift occurring at ExT06 (ExT00 indicates the midpoint of cryophase) and ExT18, respectively. Stepwise changing light cycles (SLC) also involved cycling of light between 0 and 0.4 Wm−2, but in gradual steps of 0.04 Wm−2 every 20 min, and therefore comprised 10 steps spanning 3 h 20 min in the morning and evening. Similarly, stepwise changing temperature cycles (STC) employed a rate of 1 °C change in temperature every 20 min, thus comprising 10 steps spanning 3 h 20 min in the morning and evening. In both SLC and STC, ExT06 marks the onset of stepwise light/temperature increase, and offset of stepwise light/temperature decrease in the evening is marked by ExT18. Furthermore, combined cycles of light and temperature comprised 2 types of regimes: abruptly and stepwise changing light + temperature in-phase (ALTC1 and SLTC1) and out-of-phase (ALTC2 and SLTC2). ALTC1 and SLTC1 employed the combined waveforms of light and temperature as discussed above (ALC and ATC for ALTC1; SLC and STC for SLTC1) with low temperature (18 °C) coinciding with the dark phase and high temperature (28 °C) with the light phase. Out-of-phase cycles of light and temperature (ALTC2 and SLTC2) also employed the combined waveforms of light and temperature as in ALTC1 and SLTC1, but in these cases, the temperature cycles lagged the light cycles by 4 h. The rate of increase and decrease of light and temperature cycles, as well as the 4-h phase difference between them, was set to resemble conditions in nature during the month of March 2011 in Bangalore, India (12°59′N 77°35′E), as reported in Vaze et al. (2012a), where a large difference in phase of emergence was first reported. Emergence assays were performed in Percival Drosophila chambers (Percival, Perry, IA) programmed to simulate the different experimental regimes discussed above.
Description of Experimental Regimes Used in the Study.
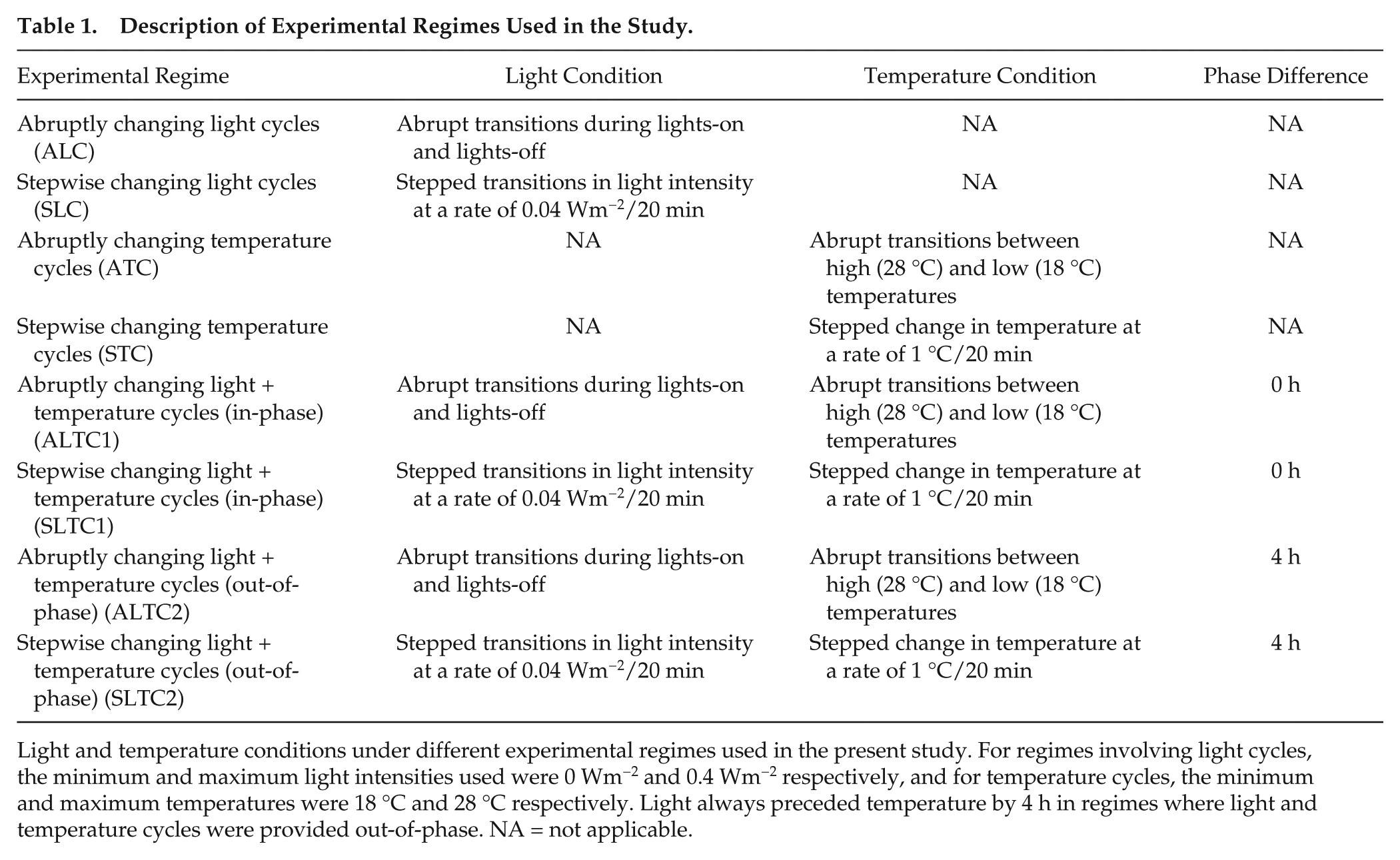
Light and temperature conditions under different experimental regimes used in the present study. For regimes involving light cycles, the minimum and maximum light intensities used were 0 Wm−2 and 0.4 Wm−2 respectively, and for temperature cycles, the minimum and maximum temperatures were 18 °C and 28 °C respectively. Light always preceded temperature by 4 h in regimes where light and temperature cycles were provided out-of-phase. NA = not applicable.
Analyses of Emergence Characteristics
Only vials with at least 3 consecutive cycles of emergence and with a minimum of 15 flies per cycle were used for data analyses. Emergence data were analyzed using directional statistics, with all tests implemented on the R statistical language platform (R Development Core Team, 2011) using “CircStats” (Jammalamadaka and SenGupta, 2001) and “Circular” packages (Agostinelli and Lund, 2013). The mean phase (time of the day measured in ExT h) of emergence and length of the mean polar vector were calculated as described in Zar (2009). On the circular time scale used for analysis, 00° refers to the external time 00 (ExT00), and 15° is equal to 1 h (Daan et al., 2002). While mean phase (θ) indicates mean time of the day around which emergence is concentrated, length (r) of the polar vector serves as a measure of coherence, and thus higher r values are indicative of greater coherence in emergence rhythm and vice versa. The gate width (gw) of emergence refers to the duration during which 90% of the total emergence occurs.
Under any given experimental regime, the Rayleigh test (α = 0.01) was used to test for the null hypothesis that emergence of a population is randomly distributed throughout the day and therefore has no mean direction (Batschelet, 1981; Jammalamadaka and SenGupta, 2001; Zar, 2009). Rejection of the null hypothesis favors the alternate hypothesis that there exists a mean direction of emergence, thus facilitating further statistical analyses. Employing parametric tests in directional statistics requires that the data approximately follow a “Von Mises” distribution (a circular equivalent of “Gaussian” distribution) with high values of the concentration parameter “κ” (Jammalamadaka and SenGupta, 2001). Furthermore, such tests require low scatter and unimodality in data to facilitate high statistical power. Since not all of these criteria were met under certain experimental regimes, we adopted nonparametric methods for all our statistical analyses unless specified.
The nonparametric test for dispersion (NPTD) (Batschelet, 1981) and Rao’s test for homogeneity were used to test if the mean phase of emergence (θ) between the populations under a given experimental regime differed significantly from each other (Jammalamadaka and SenGupta, 2001). All pairwise comparisons were carried out at α = 0.01 (99% confidence level) following Bonferroni corrections to ensure that the total family-wise error rate did not exceed α = 0.05 (95% confidence level).
Test for Phase Divergence between Populations
To quantify the extent of phase divergence between 2 populations, we used the measure “angular distance (β).” Angular distance is the shortest distance in degrees between 2 points located on a circular scale (Zar, 2009), and thus the difference between mean angles of emergence (θ) of any 2 populations would indicate the extent of phase divergence between them. The angles were later expressed as ExT in h by carrying out a simple linear transformation. Since our intention was to study the role of different zeitgebers in enhancing chronotype differences between the early and late populations to the extent observed under SN (Vaze et al., 2012a), we compared phase divergences between the populations under any given experimental regime to SN using the Wilcoxon rank-sum test or Wilcoxon-Mann-Whitney test (Zar, 2009) at α = 0.05 (95% confidence level). The regime being tested is considered to mimic SN in enhancing chronotype differences if the phase divergence between the populations in that regime is either equal to or greater than that observed under SN.
Analyses of Gate Width of Emergence
The population (block) means of gw values when subjected to the Shapiro-Wilks test for normality (α = 0.05) were found to be normally distributed and therefore were analyzed by the method of analysis of variance (ANOVA). Differences among the populations within an experimental regime were analyzed by a 2-way mixed-model ANOVA with population as fixed and block as a random factor, whereas a 3-way ANOVA model was used to test for differences among populations across experimental regimes with population and regime as fixed factors and block as a random factor. Post hoc multiple comparisons in both cases were performed using Tukey’s HSD method at α = 0.05. ANOVA and other linear statistical analysis were executed on Statistica for Windows, Release 5.0B (StatSoft, Tulsa, OK).
Results
The mean emergence phase (θ expressed as ExT) values for all populations were calculated only after subjecting the emergence data to Rayleigh test (α = 0.01), following which the null hypothesis that emergence is randomly distributed across the day was rejected. All tests for within-population differences for both mean emergence phase (θ) (NPTD and Rao’s test for homogeneity) and gate width (gw) (ANOVA) were performed as described in Materials and Methods, and unless specified, differences between populations under a given regime were significant.
In view of the hypothesis being tested, the results will primarily focus on (a) whether the extent of phase divergence under the regime being tested is equal to or greater than that observed under SN (tested by the Wilcoxon rank-sum test), and (b) gate width under a given regime being tested is equal to or lesser than that observed under SN (tested by ANOVA).
Emergence under Semi-Natural (SN) Environmental Cycles and Abruptly Changing Light Cycles (ALC)
For the ease of comparison across regimes, data from Vaze et al. (2012a) were reanalyzed by the method of circular statistics (see Materials and Methods for details of analyses). As reported in Vaze et al. (2012a), emergence of the early populations was restricted to early morning (θe = 4.46 h); the control populations emerged around 3 h later (θc = 7.47 h), and the late populations emerged significantly later (θl = 10.26 h, 3 h after control and 6 h after early populations; Figure 1a, top and middle panels; Table 2). The mean emergence phases under ALC were delayed compared to SN, with the early populations emerging at 8.72 h and the controls at 10.84 h (Figure 1b, top and middle panels; Table 2). The late populations, on the other hand, had a delayed mean emergence phase of 13.24 h with emergence extending into the dark phase (Figure 1b, top and middle panels; Table 2).
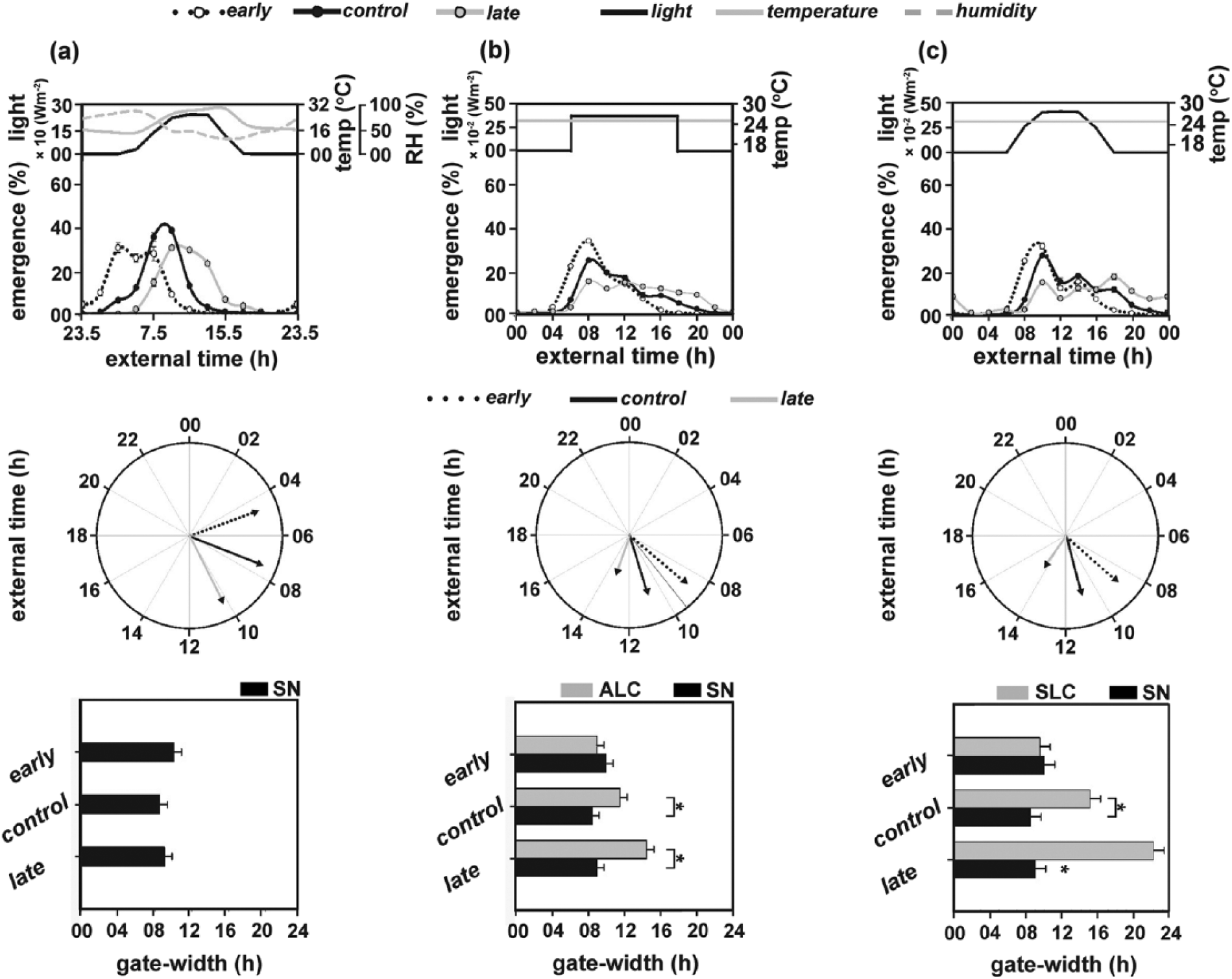
Emergence waveforms (top panel) and the respective polar plots (middle panel) (nonparametric test for dispersion and Rao’s test for homogeneity) of the early, control, and late populations under (a) semi-natural conditions (SN), (b) abruptly changing light cycles (ALC), and (c) stepwise changing light cycles (SLC). External time 00 (ExT00) (top and middle panels) indicates midpoint of the dark phase (Daan et al., 2002). Bottom panel: Gate width of emergence of the early, control, and late populations under (a) SN, (b) ALC in comparison with SN, and (c) SLC in comparison with SN. Asterisks indicate significant differences (analysis of variance followed by Tukey’s HSD). Error bars represent 95% confidence interval for visual hypothesis testing. Data for SN have been reproduced from Vaze et al. (2012a) and replotted for comparisons across regimes.
Emergence Parameters of the 3 Populations under Different Experimental Regimes.
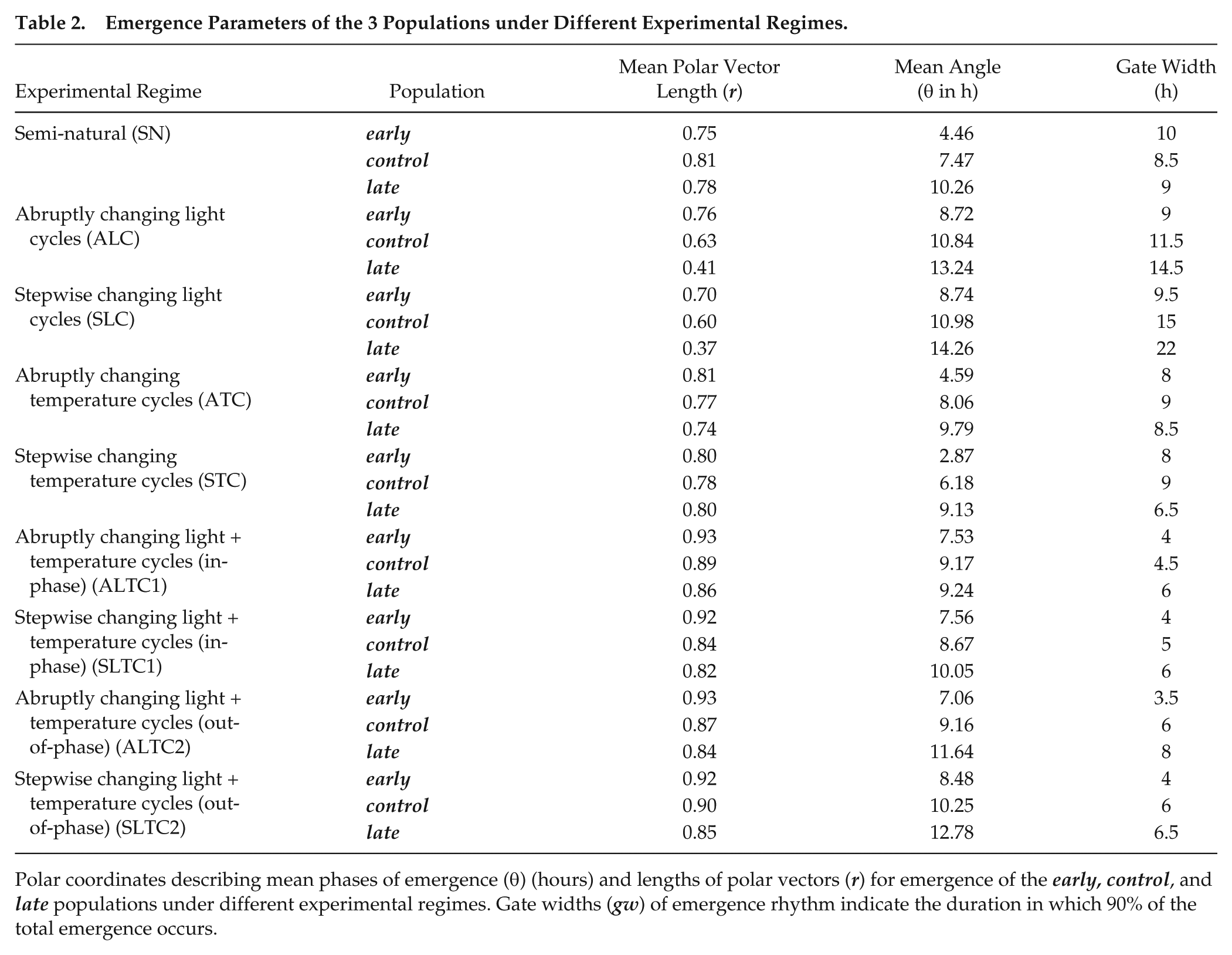
Polar coordinates describing mean phases of emergence (θ) (hours) and lengths of polar vectors (r) for emergence of the early, control, and late populations under different experimental regimes. Gate widths (gw) of emergence rhythm indicate the duration in which 90% of the total emergence occurs.
The phase-divergence between the early and late populations (βe-l) was significantly reduced from 5.76 h in SN to 4.51 h under ALC (Wilcoxon test, p < 0.05; Figure 1a,b, top and middle panels; Table 2). Similarly, phase divergence was reduced from 3.1 h in SN to 2.12 h under ALC between the early and control populations (βe-c) and from 2.75 h in SN to 2.4 h under ALC between the control and late populations (βc-l) (Wilcoxon test, p < 0.05; Figure 1a,b, top and middle panels; Table 2).
The gate widths of the control (gwc = 11.5 h) and late (gwl = 14.5 h) populations were significantly wider under ALC compared to SN (F2,6 = 25.8, p < 0.01), while that of the early populations (gwe = 9 h) did not differ between ALC and SN (Figure 1a,b, bottom panel; Table 2).
SN thus promotes greater divergence in chronotypes of the 3 populations by (a) increasing phase divergences (β) between the populations and (b) tightening the gate widths of emergence, thereby reducing the extent of overlap between their emergence waveforms (Figure 1a,b; Table 2).
Since the extent of phase divergence between the populations was less pronounced under ALC compared to that in SN, we tested if stepped cycles of light resembling gradual dawn and dusk transitions might improve the phase divergences.
Emergence under Stepwise Changing Light Cycles (SLC)
The mean emergence phases of the 3 populations under SLC (θe = 8.74 h, θc = 10.98 h, θl = 14.26 h) were similar to those in ALC (Figure 1b,c, top and middle panels; Tables 1, 2). While phase divergences between the early and late populations under SLC (βe-l = 5.51 h) were similar to those under SN (Wilcoxon test, p > 0.01), differences between the early and control (βe-c = 2.22 h) and control and late (βc-l = 3.3 h) populations were significantly smaller under SLC compared to SN (Wilcoxon test, p < 0.01; Figure 1c, top and middle panels; Table 2). In comparison to ALC, phase divergences between populations under SLC tended to be greater but not statistically significant (Table 2).
The early and late populations exhibited narrower (gwe = 9.5 h) and wider (gwl = 22 h) gate widths of emergence, respectively, compared to the controls (gwc = 15 h). In comparison with SN, gate widths of the control and late populations (F2,6 = 12.72, p < 0.01; Figure 1c, botom panel; Table 2) but not the early populations were significantly wider under SLC.
Therefore, SLC was not effective in reproducing the effects of SN either in terms of phase divergence or gate width reduction. But in comparison with ALC, phase divergences between the populations, even though not statistically different, were marginally greater under SLC.
Emergence under Abruptly Changing Temperature Cycles (ATC)
Having observed that neither abrupt nor gradual changes in light could reproduce the effects of SN, we assessed the effects of temperature, which is another robustly cycling zeitgeber in nature, by using abruptly cycling temperature cues (Table 1).
The mean emergence phases of the 3 populations (θe = 4.59 h, θc = 8.06 h, θl = 9.79 h) under ATC were advanced (compared to ALC and SLC) and were comparable to those observed under SN (Figure 2a, top and middle panels; Table 2). Also, phase divergences between the early and late populations (βe-l = 5.2 h) were significantly higher under ATC than SN (Wilcoxon test, p < 0.01; Figure 2a, top and middle panels). However, phase divergences between the early and control populations (βe-c = 3.48 h) or the control and late populations (βc-l = 2.93 h) under ATC did not differ significantly from those under SN (Wilcoxon test, p > 0.01; Figure 2a, top and middle panels; Table 2). Also, gate widths of the 3 populations (gwe = 8 h, gwc = 9 h, gwl = 8.5 h) under ATC did not significantly differ from those under SN (F2,6 = 2.28, p > 0.05; Figure 2a, bottom panel; Table 2).
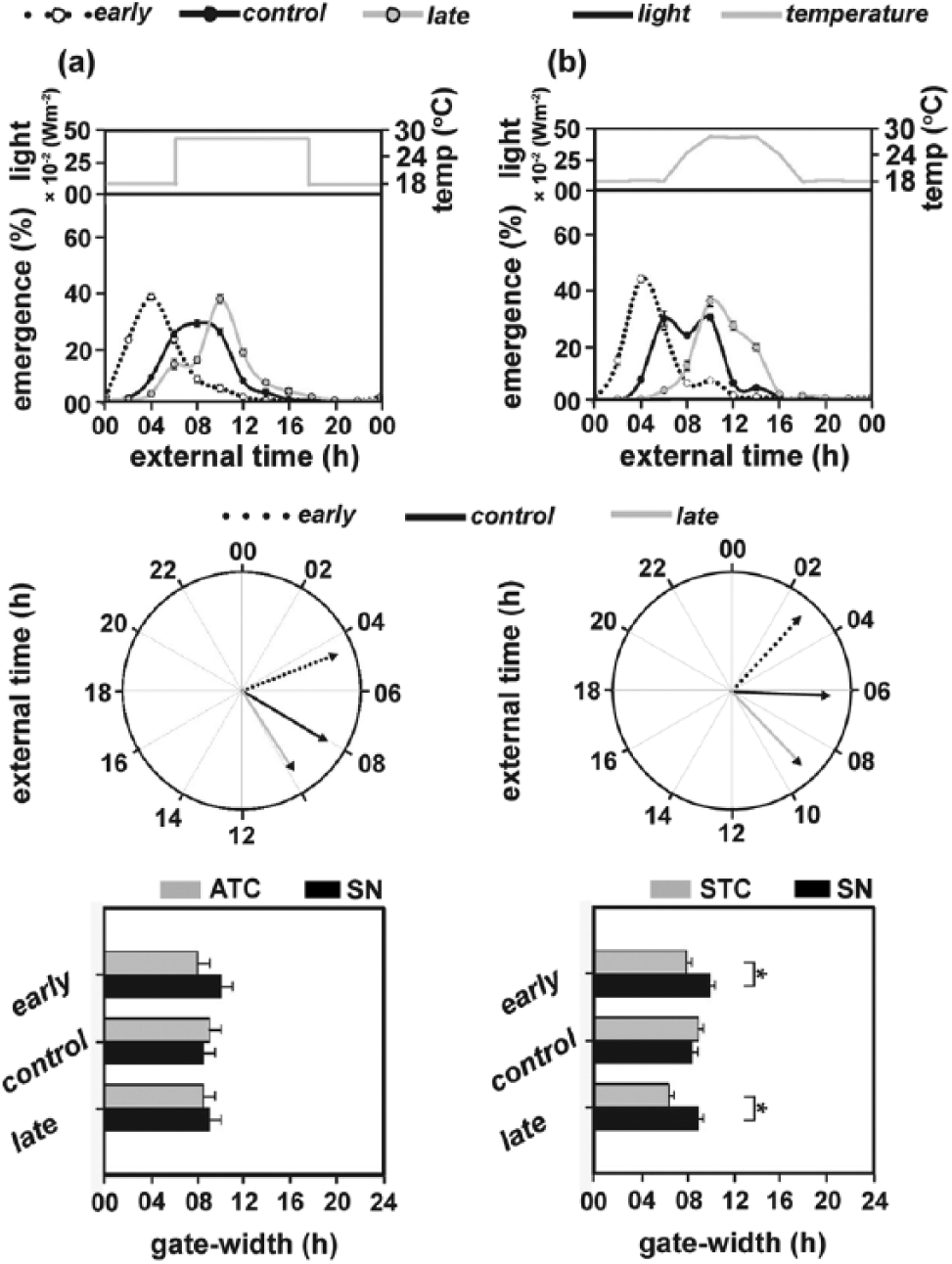
Emergence waveforms (top panel) and the respective polar plots (middle panel) (nonparametric test for dispersion and Rao’s test for homogeneity) of the early, control, and late populations under (a) abruptly changing temperature cycles (ATC) and (b) stepwise changing temperature cycles (STC). External time 00 (ExT00) (top and middle panels) indicates midpoint of the cryophase (Daan et al., 2002). Bottom panel: Gate width of emergence of the early, control, and late populations under (a) ATC and (b) STC regimes in comparison with semi-natural conditions (SN). All other details same as in Figure 1.
Thus, abruptly changing temperature cycles were highly effective in reproducing the emergence chronotypes of early and late populations as observed in SN in all aspects, including enhanced phase divergences between populations, reduction in gate widths, and also overall advancement in emergence waveforms. In addition, narrow gate widths of all 3 populations under ATC suggest that temperature plays a key role in tightening gate width of the adult emergence rhythm in the absence of light.
Emergence under Stepwise Changing Temperature Cycles (STC)
Even though ATC effectively reproduced the effects of SN, since abrupt transitions in temperature are not realistic in terms of what is observed in nature, we further tested if stepwise increase and decrease in temperature can also reproduce or further enhance phase divergences between the populations.
When assayed under stepwise changing temperature cycles (STC; Table 1), mean phases of emergence of the early and control populations but not the late populations were advanced compared to ATC (θe = 2.87 h, θc = 6.18 h, θl = 9.13 h; Figure 2b, top and middle panels; Table 2). Also, as in ATC, phase divergences of the early populations from both late and control populations (βe-l = 6.26 h, βe-c = 3.31 h) were significantly greater under STC than SN (Wilcoxon test, p < 0.05; Table 2), while those between the control and late populations were not different between STC and SN (Table 2).
All 3 populations exhibited narrower gate widths of emergence (gwe = 8 h, gwc = 9 h, gwl = 6.5 h) under STC compared to SN (Figure 2b, bottom panel; Table 2). While gate widths of the control populations were not significantly different between STC and SN, those of the early and late populations were significantly narrower under STC than SN (F2,6 = 18.6, p < 0.05; Figure 2b, bottom panel; Table 2).
Thus, stepwise changing temperature cycles also effectively enhanced chronotype differences between the 3 populations similar to and, in some cases, more than that observed in SN. Reduced gate widths of emergence under STC further strengthen the idea that temperature plays a key role in the reduction of gate widths. Additionally, phase divergences were further enhanced by small magnitude under STC, suggesting that, similar to light, stepwise increase and decrease in temperature also contribute to the enhancement of phase divergences.
Emergence under Abruptly Changing In-Phase Light and Temperature Cycles (ALTC1)
Furthermore, to test the combined effect of light and temperature cycles, as well as the role of phase difference between the two (as seen in nature), we assayed the emergence rhythm of the 3 populations under cycles of abruptly changing in-phase and out-of-phase light and temperature (ALTC1, ALTC2; Table 1).
While under ALTC1, the early populations emerged earlier (θe = 7.53 h) and the late populations emerged later than the controls (θl = 9.24 h, θc = 9.17 h), there was a marginal advancement in emergence of all 3 populations greater than that in ALC and SLC but not to the extent observed in ATC, STC, and SN (Figures 1, 2, 3a, top and middle panels; Table 2). Wilcoxon test revealed that phase divergences between the 3 populations (βe-l = 1.71 h, βe-c = 1.64 h, βc-l = 0.84 h) under ALTC1 were acutely disrupted and significantly lower compared to all other previous regimes, including SN (p < 0.05).
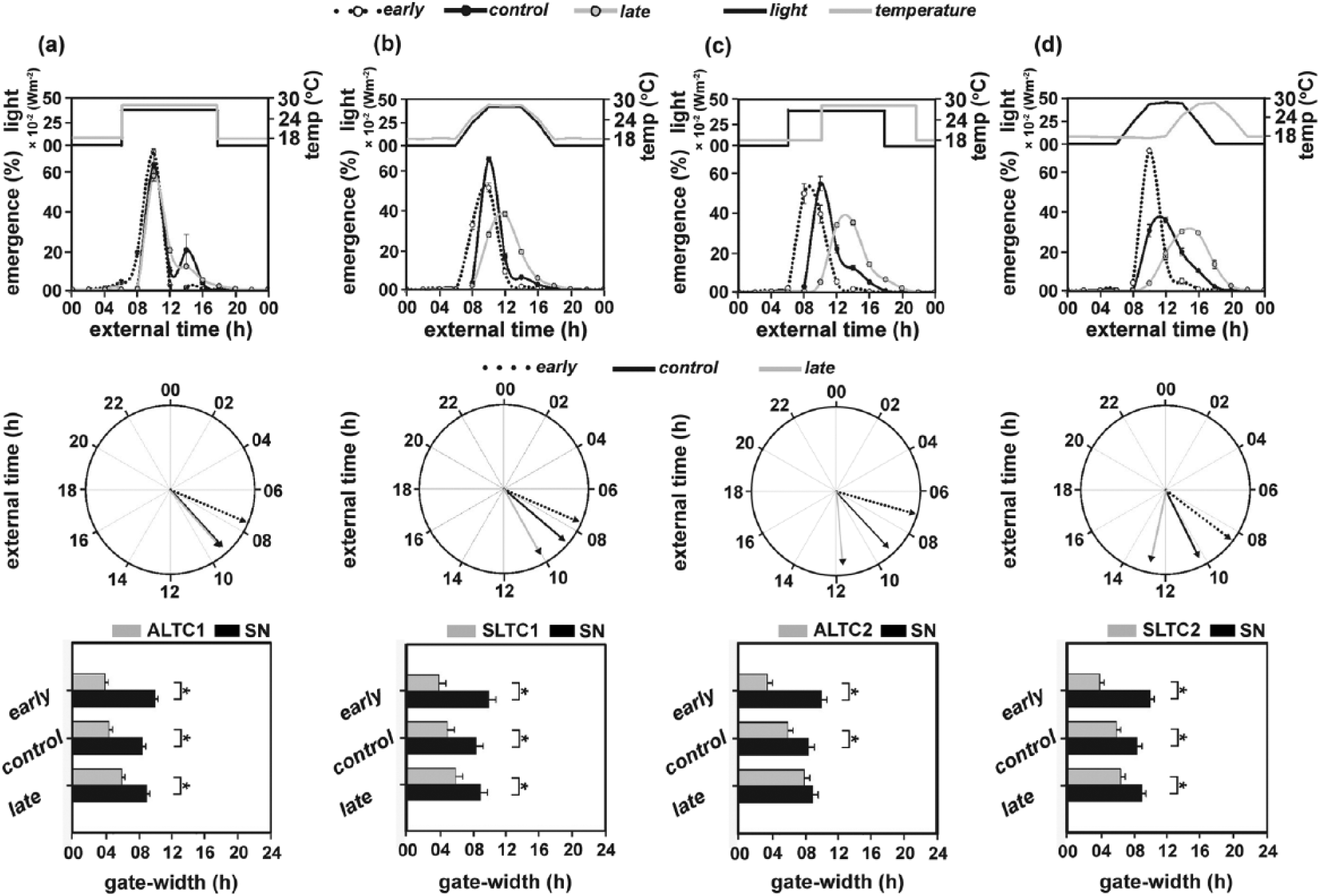
Emergence waveforms (top panel) and the respective polar plots (bottom panel) (nonparametric test for dispersion and Rao’s test for homogeneity) of the early, control, and late populations under in-phase (a) abruptly changing light and temperature cycles (ALTC1) and (b) stepwise changing light and temperature cycles (SLTC1), as well as out-of-phase (c) abruptly changing light and temperature cycles (ALTC2) and (d) stepwise changing light and temperature cycles (SLTC2). External time 00 (ExT00) (top and middle panels) indicates midpoint of the dark phase (Daan et al., 2002). Bottom panel: Gate width of emergence of the early, control, and late populations under (a) ALTC1, (b) SLTC1, (c) ALTC2, and (d) SLTC2 regimes in comparison with semi-natural conditions (SN). All other details same as in Figure 1.
Interestingly, gate widths of all 3 populations (gwe = 4 h, gwc = 4.5 h, gwl = 6 h) were significantly narrower under ALTC1 compared to SN (F2,6 = 21, p < 0.01; Figure 3a, bottom panel; Table 2).
While chronotype differences between the 3 populations were greatly attenuated and instead phase convergence was observed under ALTC1, it could only partially reproduce the effects of SN by considerably reducing the gate width of emergence rhythm.
Emergence under Stepwise Changing In-Phase Light and Temperature Cycles (SLTC1)
We assayed emergence under stepwise changing in-phase light and temperature cycles (SLTC1; Table 1) to test if such a regime would minimize or rescue the phase-converging effects brought about by ALTC1.
With mean phases (θe = 7.56 h, θc = 8.67 h, and θl = 10.05 h), emergence of all 3 populations was phase-advanced under SLTC1 compared to light cycles (ALC and SLC) but not to the extent observed under ATC, STC, and SN (Figures 1, 2, 3b, top and middle panels; Table 2). Phase divergences between the 3 populations (βe-l = 2.48 h, βe-c = 1.10 h, βc-l = 1.37 h) were also significantly smaller than those observed under SN (Wilcoxon test, p < 0.05; Table 2).
The gate widths of emergence (gwe = 4 h, gwc = 5 h, gwl = 6 h) under SLTC1 were reduced and significantly narrower than those under SN (F2,6 = 19.19, p < 0.01; Figure 3b, bottom panel; Table 2).
Thus, SLTC1 partially reproduced the effects of SN, failing to enhance chronotype differences between the 3 populations but significantly reducing gate widths. Contrary to our previous observations, we did not observe any further enhancement of phase divergences under SLTC1 in comparison with ALTC1. This indicates that the phase divergence promoting effect of stepwise cycles can be overridden when the 2 zeitgebers are in-phase and highlights the importance of phase difference between light and temperature cycles in nature.
Emergence under Abruptly Changing Out-of-Phase Light and Temperature Cycles (ALTC2)
With ALTC1 and SLTC1 highlighting the importance of the phase difference between light and temperature in the enhancement of phase divergences, we tested if abruptly changing out-of-phase (light cycles phase leading temperature by 4 h) cycles of light and temperature can mimic the effects of SN (ALTC2; Table 1).
With mean emergence phases of 7.06 h, 9.16 h, and 11.64 h for the early, control, and late populations, respectively, no phase advancement in emergence was observed under ALTC2 (Figure 3c, top and middle panels; Table 2). Comparisons by the Wilcoxon test revealed that phase divergence between the early and late populations (βe-l = 4.58 h) under ALTC2 did not differ from SN (Wilcoxon test, p > 0.05), but phase divergences between the early and control (βe-c = 2.09 h) and control and late populations (βe-c = 2.48 h) were significantly lower than those under SN (Wilcoxon test, p < 0.05).
Furthermore, gate width of the late (gwl = 8 h) populations under ALTC2 did not differ significantly from SN, whereas those of early (gwe = 3.5 h) and control (gwc = 6 h) populations were significantly narrower than under SN (F2,6 = 26.45, p < 0.01; Figure 3c, bottom panel; Table 2).
Therefore, with a marginal increase in phase divergences and narrower gate widths of emergence of all 3 populations, ALTC2 was even more effective than the in-phase cycles (ALTC1 and SLTC1) in enhancing phase divergences but only partially effective in reproducing the effects of ATC, STC, and SN.
Emergence under Stepwise Changing Out-of-Phase Light and Temperature Cycles (SLTC2)
Finally, we assayed emergence under stepwise changing out-of-phase light and temperature cycles (SLTC2), which resembled SN in terms of the zeitgeber profiles, with a gradual increase and decrease of light and temperature levels, as well as a phase difference of 4 h between the 2 cycles (Table 1).
With no observable phase advance, mean emergence phases of the 3 populations (θe = 8.48 h, θc = 10.25 h, θl = 12.78 h) under SLTC2 were found to be similar to those under ALTC2 (Figure 3d, top and middle panels; Table 2). Interestingly, analysis revealed that phase divergences between the 3 populations under SLTC2 (βe-l = 4.29 h, βe-c = 1.76 h, βc-l = 2.53 h) were lower than those under SN (Wilcoxon test, p < 0.05; Table 2).
The gate widths of all 3 populations (gwe = 4 h, gwc = 6 h, gwl = 6.5 h) under SLTC2 were significantly narrower than those under SN (F2,6 = 21, p < 0.01; Figure 3d, bottom panel; Table 2).
Thus, even though SLTC2 enhanced phase divergences between the 3 populations, surprisingly it failed to mimic the effects of SN. Similar to ATC, STC, and SN, the gate widths of emergence were significantly narrower under SLTC2.
Discussion
While recent studies have reported several interesting features of circadian rhythms in nature and attributed it to the cycling of multiple zeitgebers (De et al., 2012, 2013; Menegazzi et al., 2012, 2013; Vanin et al., 2012; Prabhakaran et al., 2013), what remains unclear is the relative contribution of different zeitgebers independently, and in unison, to the regulation of rhythmic behaviors. In addition to addressing the hypotheses (see introduction), the regimes we used also allowed us to test the role of (a) independent and combined effect of light and temperature, (b) gradual increase and decrease of zeitgeber levels simulating nature, and (c) phase difference between light and temperature profiles (as observed in nature) in enhancing phase divergence between the early and late emergence chronotypes, and in modulating gate width of the emergence rhythm.
Since the fly populations used in this study have been reared under 12:12-h LD cycles at constant temperature, and based on previous reports of differential temporal photosensitivity in these populations (Vaze et al., 2012b), we speculated that light might be a primary regulator of the early and late chronotypes. However, when assayed under conditions of abrupt light transitions (ALC), phase divergences of emergence were significantly lower and gate widths higher compared to those in SN (Figure 1a,b; Table 2). Even under stepwise cycling of light (SLC), the shape of emergence profiles and other characteristics of the rhythm did not match those seen in SN (Figure 1a,c; Table 2). Thus, cycles involving abrupt or stepwise changes in light could not reproduce the effects of SN, suggesting that light cannot independently promote phase divergence and might require the presence of other zeitgebers. Interestingly enough, we observed that phase divergences between the early and late populations under SLC were enhanced by an hour more than those in ALC (Table 2), indicating that stepwise increase and decrease in light, mimicking twilight transitions, contribute to the enhancement of phase divergence although only by a small magnitude. Such twilight transitions have been previously reported to enrich entrainment of circadian clocks in mammals (Boulos et al., 1996; Sharma et al., 1998; Boulos et al., 2002), although there have been contradicting claims by others (Comas and Hut, 2009).
Since temperature is another important zeitgeber that robustly cycles in nature, we tested the effects of abruptly (ATC) and stepwise (STC) changing temperature cycles. Interestingly, both ATC and STC were highly effective in enhancing phase divergences between the 3 populations and also in significantly reducing their gate widths to the extent observed in SN (Figures 1a, 2). Furthermore, similar to light cycles, phase divergence between the early and late populations under STC was enhanced by about an hour more than in ATC (Figure 2; Table 2), suggesting that stepwise changing temperature mimicking twilight conditions also contributes to promote phase divergence. These results suggest that most features of the emergence rhythm observed under SN are likely to be influenced by temperature cycles.
The cycling of light and temperature in nature is interdependent, and there often exists some phase difference between light and temperature with light preceding temperature in a season-dependent manner. Therefore, to study the role of combined cycles of the 2 zeitgebers and also the phase difference between them in promoting phase divergence, we assayed adult emergence of the 3 (early, control, and late) populations under cycles of both light and temperature in-phase (ALTC1 and SLTC1) and out-of-phase (ALTC2 and SLTC2). When in-phase, neither abrupt nor stepwise changing cycles of light and temperature were effective in enhancing phase divergences between the populations and instead reduced it acutely, leading to phase convergence (Figure 3a, b; Table 2), but the gate widths of all 3 populations were significantly reduced and in some cases even lower than those observed in SN. The phase divergences between the populations under ALTC2 and SLTC2 were greater than ALTC1 and SLTC1 but not comparable to SN (Figures 1a, 3; Table 2), and gate widths of emergence of all 3 populations were reduced to the extent observed in SN (Figure 3). Therefore, ALTC2 and SLTC2 were partially effective in reproducing the effects of SN. Nevertheless, both ALTC2 and SLTC2 conditions were more effective than ALTC1 and SLTC1 in the enhancement of phase divergences, thus highlighting the importance of phase difference between the zeitgebers in mediating early and late emergence chronotypes in nature.
Since SLTC2 resembles SN better than any other regimes in terms of zeitgeber profiles, we expected the features of emergence profiles observed in SN to be best expressed under SLTC2. Quite surprisingly, SLTC2 could not entirely reproduce the effects of SN. One possible reason for this discrepancy may be the restricted flexibility in programming of experimental fly chambers used, due to which the light and temperature profiles observed in nature could not be perfectly reproduced. In nature, light intensity changes in orders of 0.1 lux, but due to technical limitations, our study employed a rate of 10 lux (approximately 0.04 Wm−2) every 20 min, which was not identical to nature. Therefore, the first light that the pupae experienced was a direct upshift from 0 lux to 10 lux unlike that of 0.1 lux in SN. Adult emergence in D. melanogaster has been shown to be mediated by (a) circadian clocks that time emergence and (b) a direct masked response to lights-on (McNabb and Truman, 2008). Thus, we speculate that the first exposure to 10 lux when lights are turned on could initiate a masked response leading to a burst of emergence immediately following lights-on. This can be clearly seen in Figure 3 as a sudden increase in emergence in both the early and control populations within the first 2 hours following lights-on and might partially contribute to reduced phase divergences, thus highlighting the inadequacy of stepwise cycles of light and temperature employed to mimic their SN profiles. Additionally, we did not incorporate daily changes in the light quality (changes in composition of different wavelengths) and humidity, which are likely to promote phase divergences in nature, and thus, lack of these factors also partially explains the inability of SLTC2 to entirely reproduce the effects of SN.
We find maximal phase divergences observed under ATC and STC conditions to be interesting as they represent a completely novel environment for the populations that have evolved under LD12:12 and constant temperature for over 230 generations. The enhanced phase divergences under ATC and STC can be explained under the framework of the dual-oscillator model proposed by Pittendrigh (Pittendrigh et al., 1958), according to which the circadian clock comprises a pacemaking A oscillator, which is primarily light sensitive and temperature insensitive, and a B oscillator (coupled and driven by A but also feeds back onto A), which is primarily temperature sensitive and light insensitive. Under light cycles (ALC and SLC), the A oscillator dominates over B and uses light to phase the early and late emergence, whereas under ATC and STC in the absence of light, the A oscillator, being temperature insensitive, receives feedback from the temperature-sensitive B oscillator, which in turn drives enhanced phase divergences under these conditions. If this is true, it is reasonable to speculate that the evolved phase divergence between the early and late populations may be driven by differences in the B oscillator (however, this does not negate the possibility of differences in the A oscillator). This, in combination with the A oscillator, drives chronotype differences, the maximal expression of which is restricted by the dominance of the A oscillator under light cycles. When subjected to cycles of both light and temperature (ALTC2 and SLTC2), the combined effect results in chronotypes intermediate to those seen under light and temperature cycles alone (ATC and STC), suggesting antagonistic effects of light and temperature (Figure 4). Even though coupled oscillators have been postulated to control morning and evening activity in Drosophila (Grima et al., 2004; Stoleru et al., 2004), the above explanation remains largely speculative since there is no empirical evidence of differential sensitivity of the 2 oscillators to light and temperature. Another noticeable aspect of our finding is the phase advancement of the emergence waveform in the presence of temperature cycles. We speculate that such phase advances might be driven by thermosensitive splicing of period transcripts, which has earlier been shown to play an important role in timing the activity/rest rhythm in a temperature-dependent manner (Majercak et al., 2004, Low et al., 2012) but needs to be validated in future experiments.
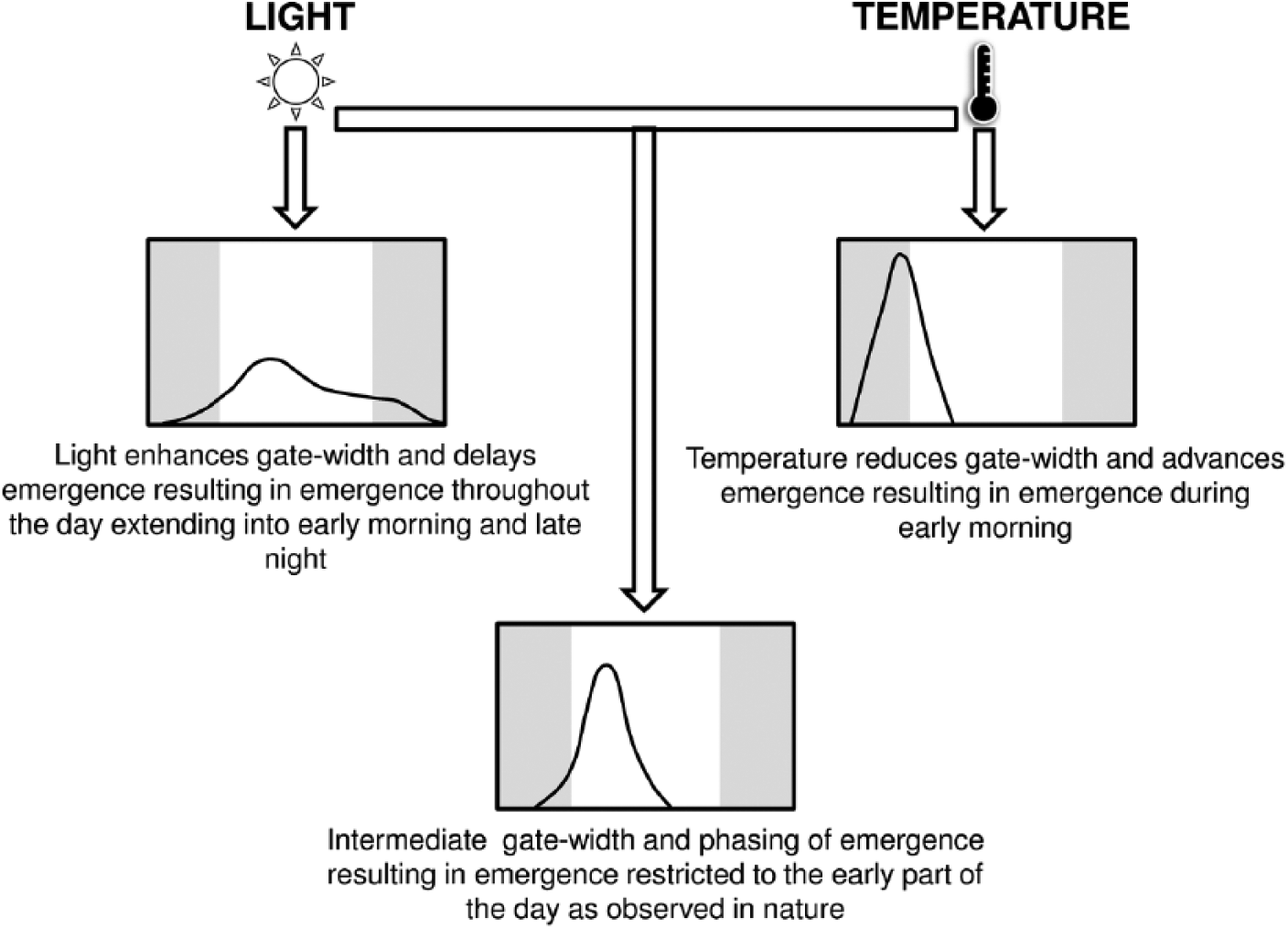
Schematic of the effects of light and temperature cycles on the timing of adult emergence rhythm in fruit flies Drosophila melanogaster.
Furthermore, enhanced gate widths of emergence under ALC and SLC indicate that light widens gate width of emergence rhythm, whereas comparisons with temperature regimes suggest that temperature has an opposing effect of light (Figures 1-4). In addition to significantly reducing gate widths, temperature cycles considerably phase-advanced emergence, thus causing it to occur during the early morning hours (Figures 2, 4). When in combination, the phase advancing effect of temperature is reduced by light and the gate-widening effect of light is reduced by temperature; as a consequence, emergence during early morning hours is phase-delayed and restricted to the early part of the day as observed in nature (De et al., 2012; Vaze et al., 2012a). Such antagonistic effects of light and temperature in mediating circadian rhythms have been reported earlier; for example, a recent study reported that CRYPTOCHROME, a photopigment that promotes light entrainment, also antagonizes entrainment of Drosophila circadian clocks to temperature cycles (Gentile et al., 2013). Additionally, evolution of gated emergence in Drosophila that restricts emergence to morning has been hypothesized to be an adaption to enhance survivability of newly emerged individuals (Pittendrigh, 1966). Therefore, it is plausible that such antagonistic interaction of light and temperature with circadian clocks is essential to restrict emergence during the day.
Results from in- and out-of-phase cycles suggest that the phase difference between light and temperature cycles also serves as an essential cue for promoting phase divergences between the 3 populations. Since coupling of PERIOD and TIMELESS oscillations in Drosophila clock neurons has been shown to vary across seasons (Menegazzi et al., 2013), these results suggest that information about the phase difference between light and temperature might be critical for circadian clocks in mediating chronotype differences across seasons. However, variation in chronotypes across seasons and the relative roles of light and temperature can only be tested by employing combined cycles of varying magnitude of phase differences between light and temperature.
Summarizing our results, we find that when provided independently, temperature is more effective than light in enhancing chronotype differences, but when together, the 2 zeitgebers interact antagonistically. Zeitgeber cycles mimicking twilight transitions of nature also contribute to enhanced phase divergence, albeit by a small magnitude. Also, phase difference between the 2 zeitgebers serves as an essential cue for circadian clocks to mediate chronotype differences. Thus, our study sheds light on the importance of clock-zeitgeber interaction and the role of different zeitgebers in nature to time behavior and mediate chronotype divergence.
Footnotes
Acknowledgements
The authors thank Abhilash Lakshman, Anuj Menon, Radhika Shindey, Manishi Srivastava, and Vishwanath Varma for their assistance with the experiments and also Sheeba Vasu for suggesting useful changes to the manuscript. We thank 2 anonymous reviewers for reading our manuscript and suggesting useful changes.
Author Contributions
KLN and VKS designed the experiments and prepared the manuscript. KLN, GG and KR performed the experiments. KLN analyzed and interpreted the data.
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
