Abstract
In insects, the role of circadian clocks in the temporal regulation of adult emergence rhythm under natural conditions has not previously been reported. Here we present the results of a study aimed at examining the time course and waveform of emergence rhythm in the fruit fly Drosophila melanogaster under seminatural condition (SN). We studied this rhythm in wild-type and clock mutant flies under SN in parallel with laboratory condition (LAB) to examine (1) how the rhythm differs between SN and LAB, (2) what roles the circadian clock protein PERIOD and the circadian photoreceptor CRYPTOCHROME (CRY) play in the regulation of emergence rhythm under SN, and (3) whether there is seasonality in the rhythm. Under SN, wild-type flies displayed tightly gated emergence, peaking at “dawn” and gradually tapering down toward the evening, with little or no emergence by night, while in LAB, flies emerged throughout the light phase of light-dark (LD) cycles. The period loss-of-function mutant (per 0 ) flies were arrhythmic in LAB but displayed weak rhythmic emergence under SN. Under SN, cry mutants displayed less robust rhythm with wider gates, greater variance in peak timing, and enhanced nighttime emergence compared to controls. Furthermore, flies showed seasonal variation in emergence rhythm, coupled either to light or to humidity/temperature depending on the severity of environmental conditions. These results suggest that adult emergence rhythm of Drosophila is more robust in nature, is coupled to environmental cycles, and shows seasonal variations.
Temporal regulation of rhythmic behaviors by environmental cycles is a fundamental property of most organisms that enables them to maximally exploit resources in their environment and to minimize the effects of adverse conditions (Pittendrigh, 1993; Saunders, 2002). In the fruit fly Drosophila melanogaster, the act of adult emergence is clock controlled and entrainable to daily cycles of light and temperature (Saunders, 2002). Emergence is largely restricted to daytime with a sharp peak around dawn. One popular hypothesis regarding the circadian regulation of emergence at dawn stresses the importance of temperature and humidity as key factors (Pittendrigh, 1993). Although the notion of humidity and temperature gating emergence rhythm is strong, it has not been empirically tested. Also light, which is known to be the primary zeitgeber for circadian clocks, has not been examined rigorously for its role in the entrainment of emergence rhythm, particularly when presented in conjunction with other time cues such as humidity and temperature.
Most previous studies aimed at understanding circadian regulation of emergence rhythm in Drosophila used well-defined laboratory conditions where flies were subjected to rectangular light-dark (LD) or high-low temperature cycles or, in very few cases, simultaneously cycling light and temperature (Saunders, 2002). Since the cycles used were rectangular, they do not represent natural conditions where a wide variety of time cues are present simultaneously, levels of which change gradually by large magnitude, and hence do not provide clues on how circadian regulation of emergence rhythm occurs in nature. The richness, strength, and stochasticity of time cues are also much higher in nature compared to any standard laboratory protocol. Keeping these details in mind, the present study was designed to examine how emergence rhythm of Drosophila entrains to environmental cycles in nature. We also wished to examine whether and how seasonal variations in the environment modulate the time course and waveform of emergence rhythm. We refer to our experimental condition as seminatural (SN) because of unavoidable manipulations that were necessary to carry out the experiments.
Materials and Methods
Fly Strains Used
We used 2 control strains, Canton-S (CS) and white-eye (w 1118 ), which previously have been shown to have wild-type circadian phenotypes, to simultaneously study adult emergence rhythm under SN and laboratory (LAB) conditions. To understand the role of circadian clocks in the regulation of emergence rhythm under SN, we used the loss-of-function period mutants (per 0 ). To examine the role of light and temperature in the regulation of emergence rhythm under SN, we used 1 hypomorphic (cry b ) (Stanewsky et al., 1998) and 2 loss-of-function mutant strains of cryptochrome (cry 01 and cry 02 ) (Dolezelova et al., 2007; Kaushik et al., 2007). All 4 mutants (per 0 , cry 01 , cry 02 , and cry b ) were on w genetic background. The w 1118 strain used was the same one used to generate cry 01 and cry 02 strains (Dolezelova et al., 2007).
Fly Stock Maintenance
Flies were maintained in an incubator under LD cycles at constant temperature (~25°C) and relative humidity (~70%) on cornmeal medium. Food in the fly vials was changed every alternate day. Freshly emerging adults were collected in Plexiglas cages (25 × 20 × 15 cm3), and to start a new generation, yeasted food plates were placed in these cages ~6 h prior to egg collection. Dim red light was used to handle flies during night. For the assays, glass vials containing eggs were placed in aluminum vial racks and transferred immediately from laboratory to shelves in the outdoor enclosure/incubator.
Assay Conditions
Seminatural condition (SN)
The assays were done within JNCASR, Bangalore campus (12°59′N 77°35″E), inside an enclosure constructed under a canopy (Suppl. Fig. S1). The enclosure was an iron cage (122 × 122 × 122 cm3) with grids (6 × 6 cm2) allowing free flow of air and covered only on the top with a sloping translucent plastic sheet.
Laboratory condition (LAB)
The laboratory assay condition was LD cycles (lights-on at 1000 h and lights-off at 2200 h) at constant temperature (25 ± 0.5°C; mean ± SD), and relative humidity (70% ± 5%), inside an incubator.
Emergence Rhythm Assay
For the assays, eggs laid over a period of ~6 h were collected and dispensed at high density (~300 eggs per vial) into glass vials (18 cm height × 2.4 cm diameter) containing ~6 mL of cornmeal food. Twenty vials of each strain were used for the assay (10 each for SN and LAB). Eggs were collected in vials, transferred immediately into assay regimes, and monitored daily for darkened pupae and thereafter every 2 h; emerging adults were counted. The daily profiles of light, temperature, and humidity were also monitored simultaneously using DEnM (Trikinetics, Waltham, MA, USA). The profiles of emergence and environmental variables shown in the figures are averages across 4 days. The SN experiment was repeated with 1 of the control strains (w 1118 ) in 3 separate months marked by moderate to large changes in light, temperature, and humidity (Suppl. Table S1) to study whether the rhythm varied with seasons. To obtain a clearer idea of correlation between emergence and environmental factors, particularly during dawn (between 0400 h and 1000 h), we carried out a separate study in April 2012, in which emergence was sampled along with environmental factors every 15 min.
Analyses of Emergence Data
The emergence profiles of each strain were plotted by averaging daily profiles of 10 replicate vials over 4 successive days. Correlation analysis between number of flies emerging in 2 h bins and corresponding light, temperature, and humidity was done using Spearman test on raw data collected over 4 days. To compare emergence rhythm across strains, regimes, and months, we quantified 3 measures of robustness: gate width, variance in emergence peak, and nighttime emergence. Gate width was estimated as the time interval between start and end of emergence in 1 complete cycle (using 5% of total emergence in that cycle as cutoff). First a spline was drawn on emergence data, and then the 2 time points in every cycle at which emergence levels reached 5% of total were noted. The gate width of every cycle was then estimated as the time difference between these two 5% cutoff phases. Peaks of emergence were determined using analysis of variance (ANOVA) with time point as fixed factor, followed by post hoc multiple comparisons using the Tukey test. Variance in peak timing was estimated as day-to-day variation in timing of emergence peak in each vial, averaged over replicate vials. Nighttime emergence under SN was calculated by defining night as the duration when light intensity was below 10 lux. The gate width, variance, and nighttime emergence data were subjected to ANOVA with strain, environmental regime, and assay month as fixed factors. Error bars shown in the figures are standard errors of mean (SEM). All statistical analyses were implemented using STATISTICA for windows.
Results
Emergence Was Correlated with Environmental Factors
To examine whether there is any dependence of emergence rhythm on environmental factors, we estimated correlation between emergence and light, temperature, and humidity using data collected in 2 separate months marked by harsh (April 2011) and moderate (July 2011) environmental conditions (Suppl. Table S1). Under harsh conditions, emergence in control (CS and w 1118 ) and cry b strains was correlated positively with humidity and negatively with temperature but was not correlated with light (Fig. 1, Suppl. Table S2). Emergence in per 0 flies was negatively correlated with light and temperature but not correlated with humidity. Under moderate conditions, emergence in control (w 1118 ) and cry b strains was positively correlated with light but not with humidity and temperature (Fig. 1, Suppl. Table S2), while emergence in cry null flies showed no correlation with light but was positively correlated with humidity and negatively with temperature (Fig. 1, Suppl. Table S2). These results suggest that under SN, emergence is coupled to light only when environmental conditions are not too harsh, while under extreme conditions it is coupled to humidity and temperature but not with light, and that such associations are circadian clock and CRY dependent.
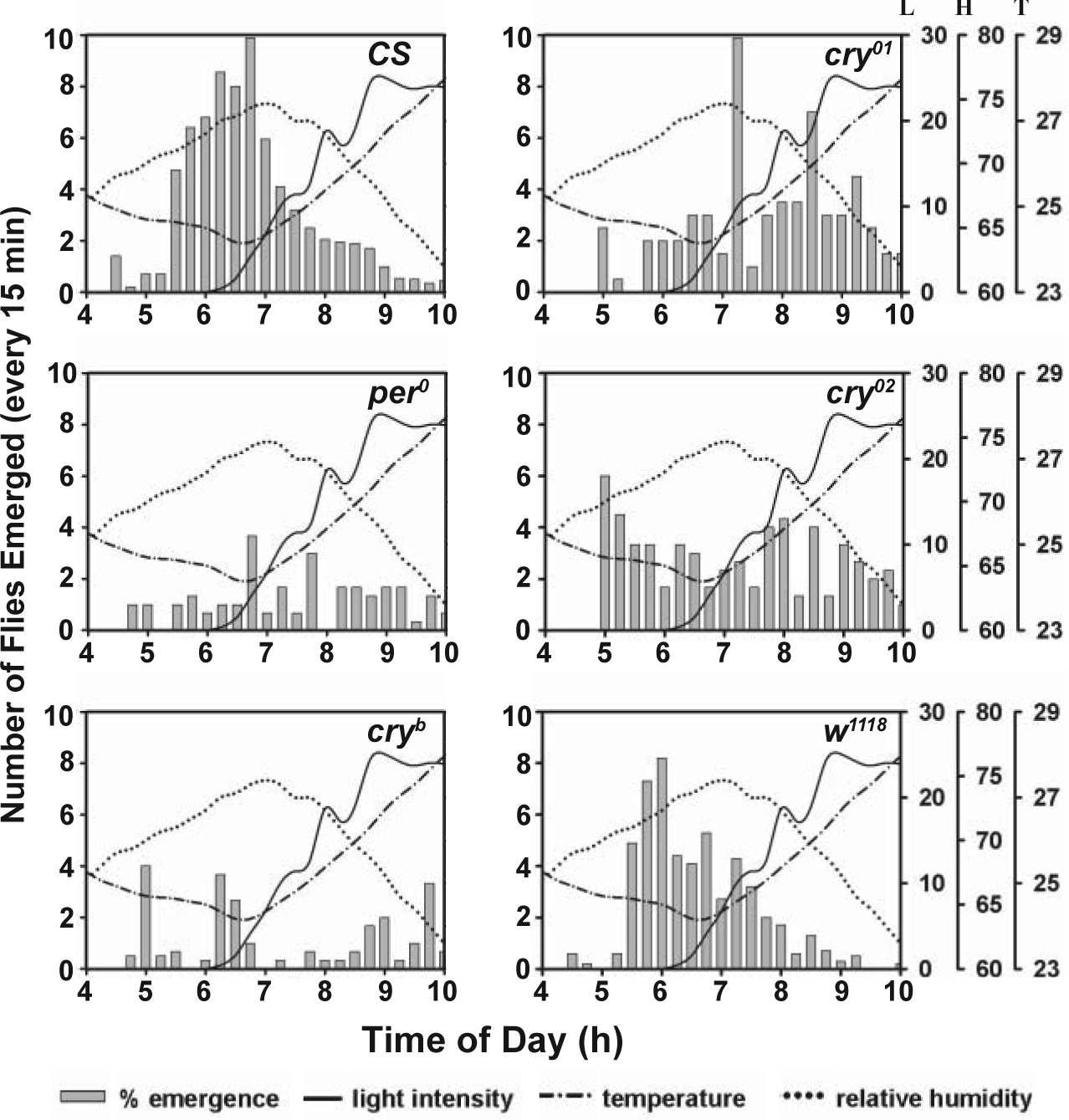
Emergence waveforms of control (CS, n = 1512; w 1118 , n = 1060 [April], n = 1762 [July]) and mutant (per 0 , n = 1202; cry b , n = 2360 [April], n = 2529 [July]; cry 01 , n = 1982; cry 02 , n = 1088) flies under seminatural (SN) conditions. All data shown in this figure were collected in the months of April 2011 (left panels) and July 2011 (right panels). Three separate axes on the right of the panels are for these environmental factors: L = light intensity (in lux), H = relative humidity (%), and T = temperature (°C). Error bars indicate SEM.
Emergence in control (CS and w 1118 ) flies was correlated negatively with light and temperature and positively with humidity in the morning, while that in clock mutants (per 0 /cry b /cry 01 /cry 02 ) showed no correlation with any of the 3 environmental factors (Fig. 2, Suppl. Table S2). This suggests that during dawn, emergence is coupled to all 3 environmental factors.
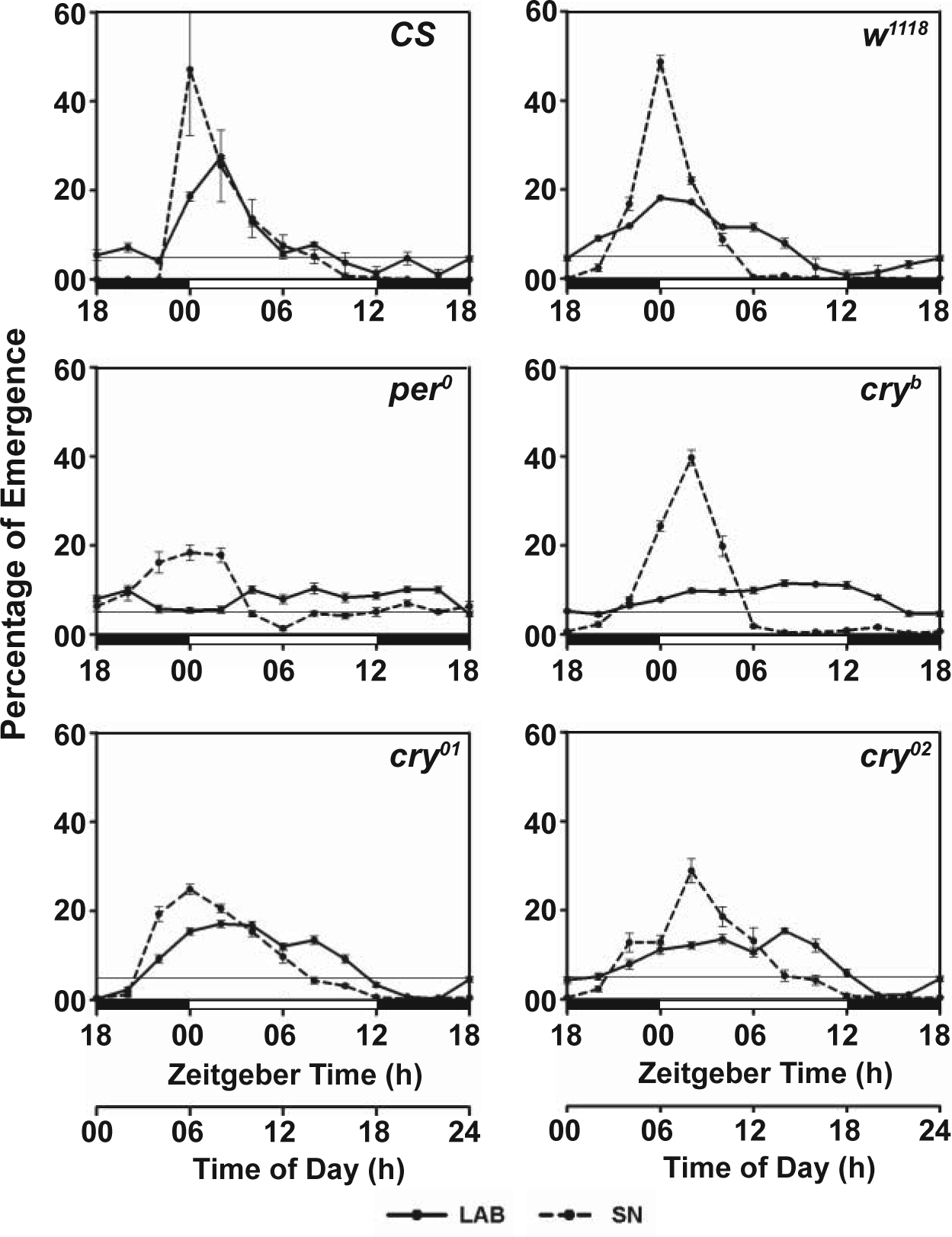
Emergence waveforms of control and mutant flies during early morning (0400-1000 h) based on data collected in 15 min bins under SN conditions in April 2012. Three separate axes on the right of the panels are for these environmental factors: L = light intensity (W/m2), H = relative humidity (%), and T = temperature (°C).
Emergence Rhythm Is More Robust under SN than LAB
The common trend (with few exceptions) that we observed in our study was that emergence rhythm is more robust under SN than LAB (Table 1). ANOVA followed by post hoc multiple comparisons using the Tukey test revealed that gate width and nighttime emergence (except cry 01 /cry 02 ) were significantly lower in SN than LAB (Fig. 3, Table 1, Suppl. Table S3). A careful examination of the emergence data suggests that shortening of gate width in SN compared to LAB was much greater for w 1118 flies than any other strain.
Summary of gate width, nighttime emergence, and variance of flies under laboratory (LAB) and seminatural (SN) conditions in all strains.
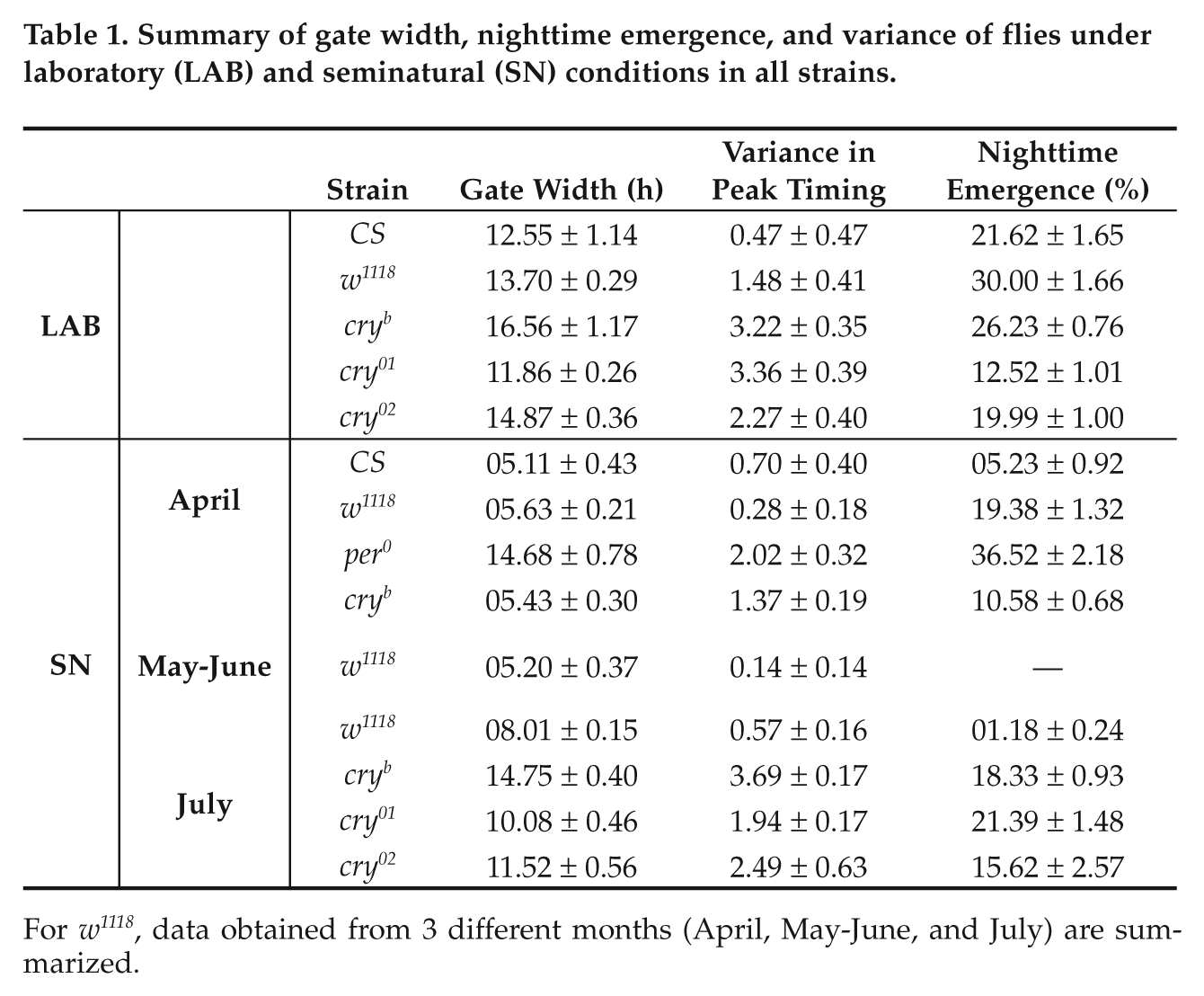
For w 1118 , data obtained from 3 different months (April, May-June, and July) are summarized.
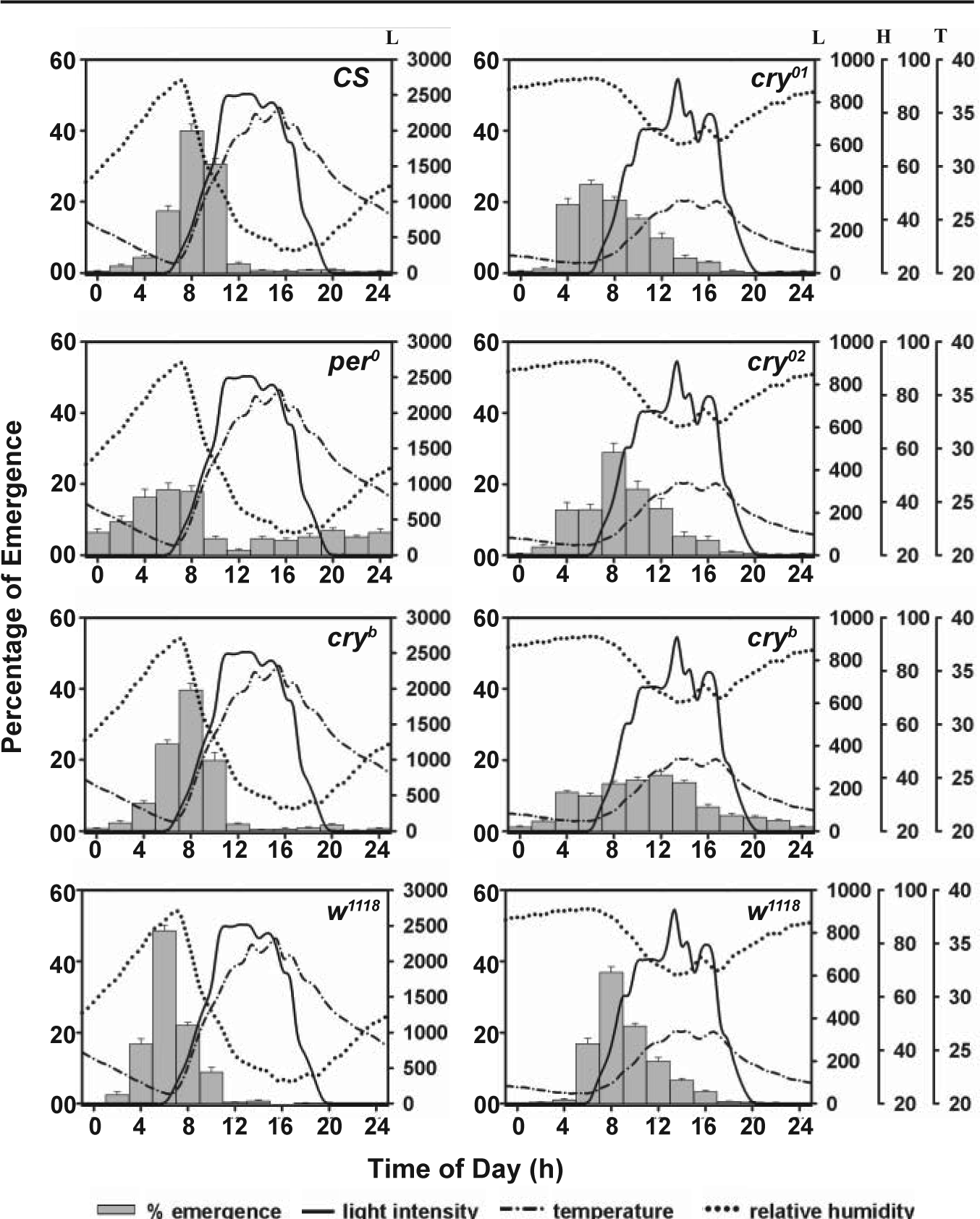
Emergence waveform of control and mutant flies under laboratory (LAB) and seminatural condition (SN). In all strains, gate width was greater under LAB compared to SN condition. Gray horizontal line shows the 5% cutoff used to calculate gate width.
per 0 Flies Showed Rhythmic Emergence under SN But Not in LAB
The per 0 flies were rhythmic under SN but not in LAB; emergence in these flies between 0400 h and 0800 h was significantly higher than at all other time points, which implies daily rhythmicity of emergence (Figs. 1, 3). However, under SN, emergence in per 0 flies was less robust than w 1118 controls. ANOVA followed by post hoc comparisons revealed that the gate width, variance and nighttime emergence were significantly greater in per 0 flies compared to w 1118 controls (Fig. 1, Table 1). These results suggest that SN makes emergence of even per 0 flies rhythmic; however, in wild-type flies functional circadian clocks make the rhythm more robust.
Presence of CRY Improves Robustness of Emergence Rhythm under SN
Under SN, cry mutants (cry b /cry 01 /cry 02 ) displayed less robust emergence rhythm compared to w 1118 controls. Based on statistical analyses of daily emergence it appears that cry mutants cycle with a near-24 h pattern. ANOVA followed by post hoc comparisons revealed that under SN, gate width, variance (except cry 01 ), and nighttime emergence of cry mutants was significantly greater than w 1118 flies (Fig. 1, Table 1, Suppl. Table S3), which suggests that presence of CRY improves the robustness of emergence rhythm under SN. In the LAB, only gate width (except cry 01 /cry 02 ) and variance (except cry 02 ), were significantly greater than w 1118 flies, whereas nighttime emergence was reduced compared to w 1118 (Fig. 3, Table1). While emergence in most strains of flies anticipated rise in light in LAB and of light/temperature in SN, increased nighttime emergence of w 1118 flies in the LAB was primarily because of greater anticipation compared to cry mutants. While robustness of emergence rhythm was poor in all cry mutants, cry b was the worst, which may probably be due to differences in their genetic background. Although we assumed that circadian mutants differ from their genetic controls in terms of impaired ability of clocks alone, it is associated with potential caveats, such as diminished amplitudes of emergence waveform possibly contributing to these observed defects.
Possible Evidence for Seasonality in Emergence Rhythm
To examine whether there is seasonality in emergence rhythm, we assayed the rhythm in w 1118 flies during 3 separate months marked by moderate (July), intermediate (May-June), and large (April) changes in light intensity, humidity, and temperature (Suppl. Table S1). We observed small change in photoperiod (~0.50 h) but significant change in the shape of humidity profile between the 3 months. For example, in July, maximum humidity and minimum temperature remained at reasonably high and low levels, respectively, for much longer during the day compared to April. Emergence profile of w 1118 flies was significantly different between these months (Fig. 1, two bottom panels; May-June profile not shown). ANOVA followed by post hoc comparisons revealed that gate width of w 1118 flies was greater in July compared to April and May-June, and variance was greater in July than May-June, while their nighttime emergence was higher in April than July (Fig. 1, Table 1, Suppl. Table S3). Since hardly any flies emerged during nighttime in May-June, we could not estimate nighttime emergence. Taken together, the results suggest that robustness of emergence rhythm varies across months, implying seasonality.
Discussion
The results of our study suggest that robustness of emergence rhythm was increased under SN compared to LAB (Fig. 3, Table 1), which could possibly be due to the presence of multitude of zeitgebers in nature in relatively higher contrast than laboratory, where the only time cue present was in the form of presence and absence of relatively low intensity light (~100 lux). Natural light cycles seem to improve robustness of emergence rhythm by enhancing its peak and reducing nighttime emergence, as was observed in a study in April 2012, where we monitored the rhythm under SN, in presence or absence of light (data not shown). Interestingly, per 0 flies, which showed arrhythmic emergence in LAB, became rhythmic when subjected to SN. This is consistent with the findings of previous studies which reported that per 0 flies display residual activity/rest rhythm in laboratory (Dowse and Ringo, 1987; Helfrich and Engelmann, 1987) and a more robust rhythm in nature (Vanin et al., 2012). The per 0 flies also showed significantly higher emergence during the wettest and coolest part of the day, quite similar to wild-type flies, however, their emergence was less tightly gated than controls, which suggests that in nature, circadian clocks inhibit emergence at unfavorable times of the day.
Correlations between Emergence and Environmental Variables: What Do They Tell Us?
When environmental conditions were harsh, we found positive correlation between emergence and humidity and negative correlation with temperature, which supports the notion that emergence occurs mostly during the wettest and coolest time of the day (Pittendrigh, 1993). Lack of correlation between emergence and light suggests that under harsh environmental conditions, emergence follows environmental changes in humidity and temperature more than such changes in light. These results support the idea that light may not always dominate over temperature (Vanin et al., 2012). However, in our study, when environmental conditions were moderate, emergence was correlated with light and not with humidity and temperature. This was consistently seen in most wild-type flies assayed under moderate environmental conditions (data not shown). Moreover, under moderate conditions we found less robust correlation of emergence with light in cry b and no correlation with light in cry null flies (Suppl. Table S2). Given that cry mutants showed less robust emergence rhythm compared to controls, these results suggest that presence of CRY and/or light improves stability of emergence rhythm under SN.
Adult emergence is confined to morning when environmental factors vary the most. To test whether emergence during this period is associated with environmental factors, we examined correlation of emergence with environmental factors. Interestingly, emergence in 2 control strains (CS/w 1118 ) of flies was correlated positively with humidity and negatively with light and temperature, while that in per 0 and cry mutants showed no correlation with any of the environmental factors (Fig. 3, Suppl. Table S2). The negative correlation with light is suggestive of the fact that emergence is enhanced during dawn, followed by subsequent decline concurrent with increase in light intensity, rather than the idea that light suppresses emergence. This suggests that under SN, time course and waveform of emergence rhythm are determined by a complex interaction of a multitude of environmental variables, acting in a hierarchical manner, the nature of which is determined by ecological conditions where the species has evolved. While humidity and temperature are probably high up in the hierarchy, light may not be far behind. An important caveat in our conclusions based on the correlations of emergence with humidity is that while light and temperature data that we recorded from environment are more likely to reflect the actual values for these factors inside fly vials, this may not be the case for humidity, because fly vials contain semisolid food that may locally alter humidity and therefore the actual values recorded might vary to some extent from what flies experience in the vial. Taken together, these results suggest that an extensive study of emergence rhythm under natural conditions along with simulated studies in the laboratory is likely to provide a realistic picture of circadian entrainment and of adaptive significance of rhythmic processes. Without such experiments, some of the correlations that we report here do not lead to strong and clear conclusions.
Is There Seasonality in Emergence Rhythm?
We compared gate width of emergence, variance, and nighttime emergence of w 1118 flies from data collected during the months of April, May-June, and July. The gate width and variance increased in July, which suggests that when the environment was wetter and cooler, emergence rhythm became relatively less robust in contrast to April and May-June, when afternoons were warmer and drier. This is probably because in July, flies could afford to emerge even in the afternoon, because high humidity and low temperature conditions persisted for much longer. We also observed high nighttime emergence in April compared to July probably because in April wetter and cooler conditions prevailed only during night.
Under harsh environmental conditions, emergence was correlated with humidity and temperature but not with light, and when conditions were moderate, it was correlated with light but not with humidity and temperature. However, emergence in the morning was correlated with all 3 environmental factors. Since circadian clocks are synchronized by light, emergence rhythm is likely to be correlated with it, and perhaps an increase in humidity enhances robustness of the rhythm by timing it to occur during late night/early morning and an increase in temperature inhibits emergence later in the day resulting in a sharper waveform. Natural conditions enhance robustness of emergence rhythm, and even flies lacking functional clocks showed rhythmic emergence. Furthermore, with variations in environmental conditions, flies show changes in gate width, variance, and nighttime emergence, and in the way they couple to environmental conditions.
Footnotes
Acknowledgements
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
