Abstract
Blue light sensitivity of melatonin suppression and subjective mood and alertness responses in humans is recognized as being melanopsin based. Observations that long-wavelength (red) light can potentiate responses to subsequent short-wavelength (blue) light have been attributed to the bistable nature of melanopsin whereby it forms stable associations with both
Ocular light exposure in mammals can generate a range of acute nonvisual physiological and behavioral responses, which are maximally sensitive to short-wavelength blue light (λmax ~ 480 nm). In humans, these include suppression of the nocturnal pineal hormone melatonin (Brainard et al., 2001; Thapan et al., 2001), circadian phase shifting (Lockley et al., 2003; Sletten et al., 2009), and enhancement of mood, alertness, and performance (Lockley et al., 2006; Revell et al., 2006). This blue light sensitivity in humans (λmax 440-480 nm) is attributed to the photopigment melanopsin expressed in the inner retina in a subset of intrinsically photosensitive retinal ganglion cells (ipRGCs) (Berson et al., 2002; Gooley et al., 2001), which innervate brain areas regulating these nonvisual responses (Gooley et al., 2003; Hattar et al., 2006). In addition, the ipRGCs receive photic input from the rod and cone visual photoreceptors, which contribute to nonvisual responses in a context-dependent manner (Altimus et al., 2010; Lall et al., 2010).
Melanopsin is an atypical vertebrate opsin, being most homologous to invertebrate rhabdomeric opsins (Provencio et al., 2000) and having a sluggish, yet sustained, response to light (Berson et al., 2002; Dacey et al., 2005). Furthermore, through studies of human melanopsin (Melyan et al., 2005) and the
In the real world, it is likely that the proposed photosensitivity and photoisomerase functions of melanopsin will be stimulated simultaneously by short- and long-wavelength light, under both broadband polychromatic white lights and natural lighting (400-700 nm). This is different from the consecutive presentation of short and long wavelengths of light used in the previous in vivo studies (Mure et al., 2009; Mure et al., 2007). If melanopsin bistability is to be of practical use, then it is essential that the response to concurrent blue and red light is assessed. Therefore, in the current study, we aimed to determine whether the acute suppression of melatonin and elevation of mood and alertness by short-wavelength blue light (λmax 479 nm) could be altered (enhanced or reduced) by simultaneous presentation of long-wavelength red (λmax 627 nm) light.
Materials and Methods
All procedures were approved by the University of Surrey Ethics Committee and were conducted in accordance with the Declaration of Helsinki and conformed to international ethical standards. Participants gave written consent.
Participants
Healthy men (
Prestudy measurements
The protocol used was similar to that previously described (Revell and Skene, 2007), with participants maintaining a regular, actigraphically (AWL, Cambridge Neurotechnology, UK) monitored sleep/wake schedule for at least 7 days before and throughout the in-laboratory sessions. For 72 h before each laboratory session and for its duration, participants refrained from caffeinated drinks, alcohol, excessive exercise, bright lights, and nonsteroidal anti-inflammatory drug intake. In addition, subjects performed a 48-h urine collection (sequential 4-h collection bins, 8 h overnight) for measurement, via radioimmunoassay, of 6-sulfatoxymelatonin, the major urinary metabolite of melatonin (Stockgrand Ltd., University of Surrey, Guildford, UK). The acrophase of 6-sulfatoxymelatonin production was determined by cosinor analysis, enabling light administration to be individually timed to occur on the rising phase of each participant’s endogenous melatonin rhythm.
In-laboratory protocol
Details of the protocol were previously described (Revell and Skene, 2007). A randomized, within-subject, crossover design was used. Participants were assigned to one of two study protocols that differed only in the light conditions administered (study A: 8 overnight sessions,
The participants’ pupils were fully dilated by a single drop of Minims Tropicamide (1.0%; Chauvin Pharmaceuticals, Romford, UK) placed in each eye 90 min before the light exposure. Blood samples were taken at regular 15-min intervals relative to the light exposure from 30 min before to up to 90 min after the start of the light pulse. The plasma fraction from each sample was stored at −20°C until melatonin analysis by radioimmunoassay. In addition, verbal ratings of subjective sleepiness (Karolinska Sleepiness Scale (Akerstedt and Gillberg, 1990) and mood and alertness (four 9-point scales: alertness, cheerfulness, depression, calmness) (Revell et al., 2006) were obtained at 15-min intervals from 15 min before until 45 min after the start of the light exposure.
Light Exposure
As previously described (Revell et al., 2010), the 30-min light pulse was administered using a specially constructed 45-cm diameter sphere (Apollo Lighting, Leeds, UK). The sphere was coated with white reflectance paint (WRC-680 Labsphere; Pro-Lite Technology, Bedfordshire, UK) and was illuminated via a fiber-optic cable connected to an ultra-high-pressure mercury lamp (Focus 100LS3, 100 W; Philips Lighting, Eindhoven, The Netherlands) to provide patternless illumination encompassing the entire visual field. Monochromatic light was produced using narrow-bandwidth interference filters (Coherent Ealing Europe Ltd., Watford, UK) with λmax 480 and 630 nm (half-maximal bandwidth λ0.5 10 nm). The actual in situ λmax at eye level was shifted to 479 and 627 nm, respectively, as measured by a calibrated spectrometer (Ocean Optics BV, Dunedin, FL). Light irradiances were adjusted using neutral density filters (Kodak, Hemel Hempsted, UK) and were verified at the subject’s eye level using a calibrated radiometer (R203; Macam Photometrics Ltd., Livingston, Scotland) before and after each light exposure. Head movements and gaze were restricted, and subjects were monitored by trained researchers throughout the light stimulus.
Short-wavelength blue (λmax 479 nm) and long-wavelength red (λmax 627 nm) monochromatic lights were administered alone or in combination at a range of photon densities (Table 1). The photon densities were selected on the basis of previously determined irradiance response curves to monochromatic light for melatonin suppression (Thapan et al., 2001) and the reported spectral responses of the human pupil light reflex (Mure et al., 2009), to be within the melanopsin operating range and to ensure that either a potentiation or a reduction in the response could be observed. The highest irradiance of red monochromatic light tested was matched to that previously shown to be effective at enhancing subsequent pupil constriction responses to blue light (Mure et al., 2009). In study A, 3 irradiances of 479 nm light (1013-1014 photons/cm2/sec) were administered with and without a fixed irradiance (5 × 1013 photons/cm2/sec) of 627 nm light (
Irradiances (photons/cm2/sec) of Monochromatic (λmax 479 or 627 nm) and Bichromatic (λmax 479 + 627 nm) Lights Administered in Study A (Fixed 627 nm Light Irradiance) and Study B (Fixed 479 nm Light Irradiance)
Analysis and statistics
Plasma was separated within 10 min of collection by refrigerated centrifugation (3000 rpm for 10 min) and stored at −20 14;°C until analysis. Plasma melatonin was assayed by radioimmunoassay (affinity-stripped plasma and antimelatonin antiserum G/S/704-8483, Stockgrand Ltd., University of Surrey, Guildford, UK; tritiated melatonin TRK-798, Amersham International Ltd., Buckinghamshire, UK) (Fraser et al., 1983). The limit of detection was 6 pg/mL, and the interassay coefficients of variation were 16% at 25 pg/mL, 8% at 89 pg/mL, and 8% at 112 pg/mL (
Results
The ability of blue (λmax 479 nm) and red (λmax 627 nm) monochromatic light, of varying photon densities presented alone and in combination, to influence plasma melatonin levels and subjective mood and alertness was quantified in healthy male participants.
Minimal suppression of melatonin range (−2.9% to 16.7%;

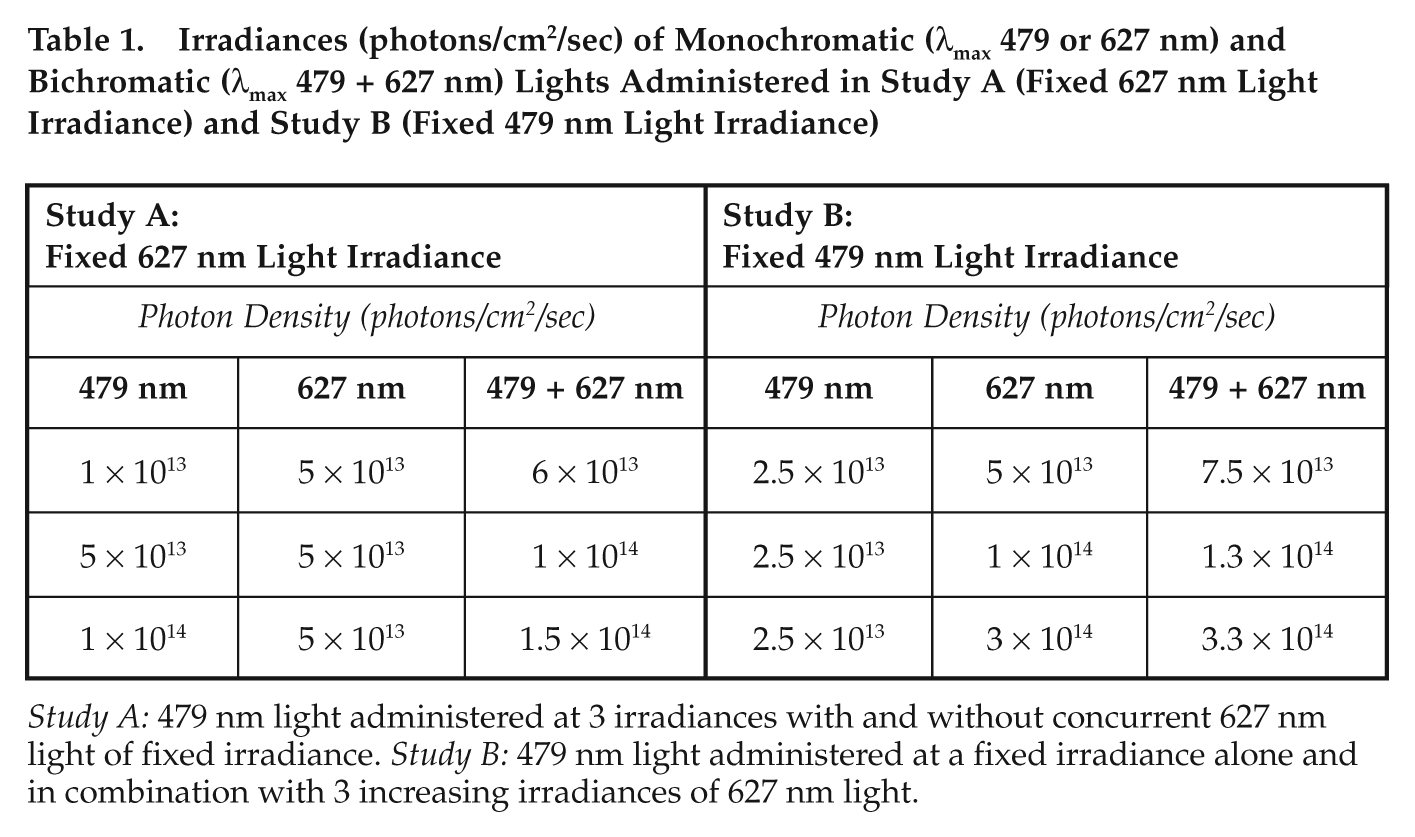
Plasma melatonin profiles (pg/mL) during and after a 30-min light stimulus (shaded area) in 3 participants under no-light control conditions, red (627 nm) monochromatic, blue (479 nm) monochromatic, and red and blue bichromatic (479 + 627 nm) light. (
Blue (479 nm) light produced significantly greater melatonin suppression than photon-matched red (627 nm) light as evidenced by within-subject comparisons at 5 × 1013 and 1 × 1014 photons/cm2/sec (Figure 2A,
Percentage melatonin suppression (mean ± SEM) during a 30-min nocturnal red (627 nm) or blue (479 nm) monochromatic light pulse (shaded area) in the same individuals. (
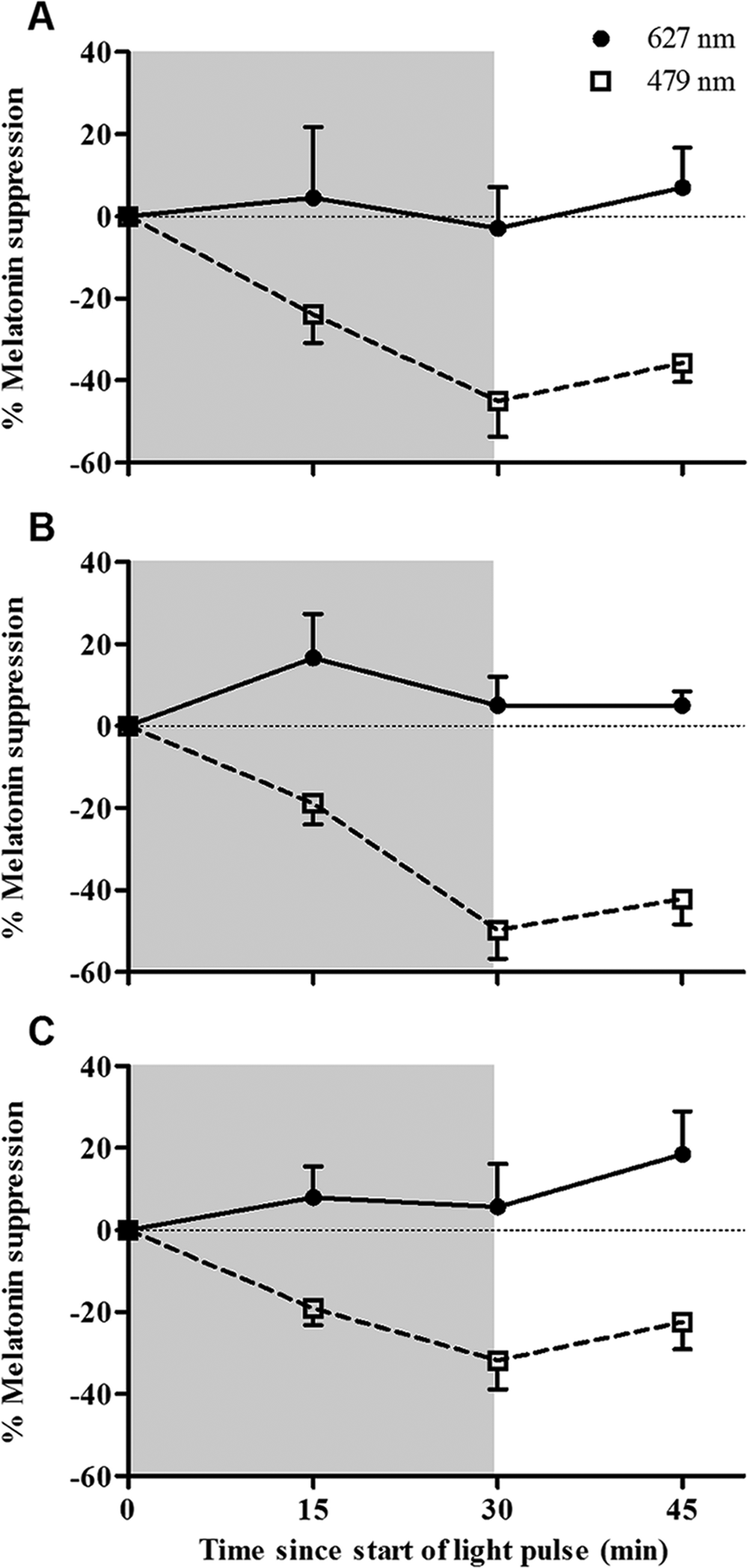
Melatonin suppression was calculated by comparing the plasma melatonin levels before (0 min) and at the end (30 min) of the light stimulus. For each study, the levels at 0 min did not differ significantly between light conditions (including the no-light control) (study A: 56.7-64.0 pg/mL, mean range; 59.4 ± 5.6 pg/mL, mean ± SEM;
Plasma melatonin levels (mean ± SEM;
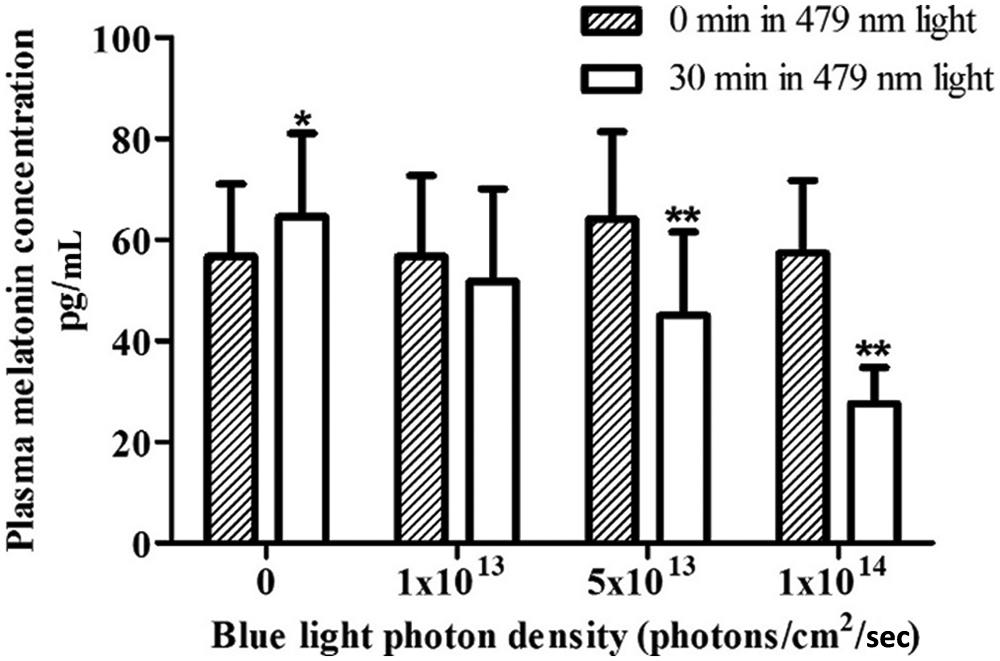
In study A, irradiance dependency of melatonin suppression was observed for both 479 nm light alone and 479 + 627 nm light pulses (Figure 4A) (
Percentage melatonin suppression (mean ± SEM) at the end of a 30-min nocturnal monochromatic blue light stimulus (λmax 479 nm) alone and in combination with long-wavelength red light (λmax 627 nm). (
At lower photon densities (5 × 1013 photons/cm2/sec), 479 nm light significantly (
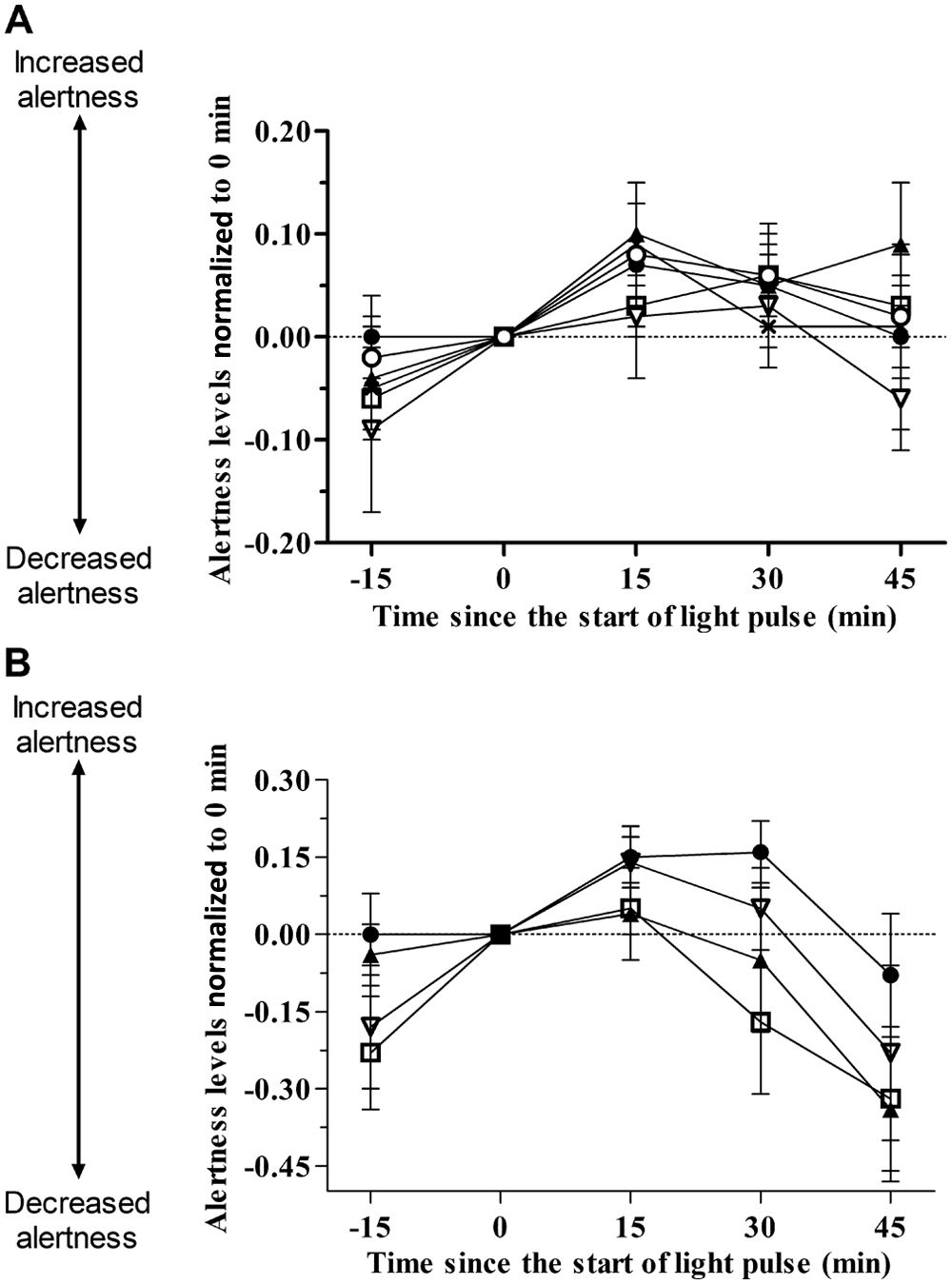
Subjective alertness levels (mean ± SEM) before, during, and after a nocturnal 30-min monochromatic or dichromatic light stimulus normalized to 0 min. (
Discussion
The current study aimed to test the hypothesis that acute melatonin and subjective measures of alertness and mood to short-wavelength blue light (λmax 479 nm) could be significantly altered by concurrent presentation of long-wavelength red light (λmax 627 nm).
In accordance with previous studies of light-induced melatonin suppression (Brainard et al., 2001; Thapan et al., 2001), we observed a clear irradiance dependency with blue monochromatic light. In addition, minimal melatonin suppression (non–control adjusted) was observed with red monochromatic light. This is in agreement with a previous study (Hanifin et al., 2006) where a 90-min red monochromatic light stimulus (1.9 × 1018 photons/cm2), over 6000 times brighter than our highest photon density red light, only induced 18% melatonin suppression (control adjusted) that did not significantly differ from the no-light control response (Hanifin et al., 2006).
The current study demonstrates for the first time that simultaneous presentation of red 627 nm light neither enhanced nor reduced the degree of melatonin suppression observed with blue 479 nm light alone. This was true when a fixed-irradiance red light pulse (5 × 1013 photons/cm2/sec) was added to increasing irradiances of blue light (study A) and when a fixed-irradiance blue light pulse (2.5 × 1013 photons/cm2/sec) was administered with increasing irradiances of red light (study B). At lower irradiances (5 × 1013 photons/cm2/s), 479 nm light was more effective at enhancing subjective alertness levels than photon-matched 627 nm light. The lack of this discrepancy at higher irradiances suggests that at these levels, either the irradiance of the light is more determinative than wavelength for alertness or the subjective scales lack sufficient resolution at higher photon densities. Similar to the melatonin suppression data, the addition of a fixed-irradiance red light did not enhance the alerting response to blue light alone. The observed photic enhancement of subjective mood and alertness at night, irrespective of light irradiance and composition, in dark-adapted individuals is in accordance with our previous data (Revell et al., 2010).
Previous studies in mice and humans (Mure et al., 2009; Mure et al., 2007) have attributed the ability of a long-wavelength red light pulse to enhance responses to subsequent blue light exposure to the bistable nature of melanopsin. It has been proposed that, through red light stimulation of the M state, the proportion of R state available for subsequent stimulation by blue light will be increased, resulting in an augmented response. By contrast, concurrent presentation of red and blue light, as used in the present study, has the theoretical potential to both increase and reduce responses observed with blue light alone. The action of red light on the pool of M state could either enhance the response by increasing the available proportion of “blue light–responsive” R state or reduce the response by simply attenuating the sustained firing of the M state under prolonged exposure to blue light. In addition, the presence of longer wavelength red light could have stimulated the M- and L-cone photoreceptors whose input could have also modulated the response to blue light alone. Indeed, a limitation of the current experimental design (and all previous human studies) is that the relative contribution of each photoreceptor class cannot be isolated; rather, the responses measured result from the aggregate input of all 3 classes of photoreceptors. Increasing evidence suggests that, under specific conditions, rods and cones might be important in regulating nonvisual responses to light (Lucas et al., 2003; Altimus et al., 2010; Lall et al., 2010). Studying patients with retinal disorders who lack specific classes of a classical photoreceptor may provide information into the response dynamics of melanopsin.
The lack of significant impact of the concurrent presentation of red and blue light in the current study compared to the observed enhancing effect of consecutive red and blue light for the pupil light reflex (PLR) (Mure et al., 2009) may be attributable to differences in the protocol such as time of day, light stimulus duration, periods of darkness, and dependent variables. First, the PLR was measured in the morning compared to our nocturnal assessments of melatonin suppression; circadian variation in photopigment contribution to nonvisual responses has been observed previously (Tsai et al., 2009) and could be pertinent here. Second, it is possible that, under concurrent red and blue light administration over a 30-min period, the red light simultaneously enhanced (regenerating R state) and repressed (attenuating M state firing) the response to blue light, resulting in no overall change in response. Third, as the relative sensitivities of the M and R states are unknown, it is possible that the irradiances of red light used in the current studies were insufficient to activate the M state under constant blue light illumination. Finally, it is possible that regeneration of the R state under constant illumination (current study) may be slower than under complete darkness (PLR study) such that we observed neither potentiation nor suppression of response. The dynamics of the photoisomerase (M to R state) function under both light and dark conditions remain to be determined. A protocol regime incorporating an extended period of darkness between the red and blue light stimuli may be necessary to resolve the photoisomerase function.
The bistability of melanopsin is an emerging area, and it is possible that, in the current study, the optimal long wavelength and/or irradiance light, to demonstrate photoisomerase activity, was not used. Although the in vivo studies of Mure and colleagues (2007, 2009) implicated wavelengths >600 nm (used in the current study), exposure to blue light in Amphioxus results in an only slightly red-shifted photoproduct (520 nm) that was then capable of photoregenerating the bleached isoform (Koyanagi et al., 2005). Further complicating the interpretation of the present and previous results is the proposed existence of a light-independent (dark) regeneration mechanism (Koyanagi et al., 2005) as well as the possibility of more than one meta-state (Ritter et al., 2004). Melanopsin in dark-adapted mouse retinas has been shown to be exclusively bound to 11-cis retinal (Walker et al., 2008), and this apparent photo-independent regeneration may have contributed to the results observed by Mure and colleagues (Mure et al., 2009; Mure et al., 2007). Thus, regeneration of the R state may be a highly complex process, and identification of photoisomerase activity may be protocol dependent. Finally, the existence of several subtypes of melanopsin ipRGCs that differentially innervate different brain areas involved in nonvisual responses (Baver et al., 2008; Ecker et al., 2010) and are themselves differentially reliant on rod/cone signaling (Schmidt and Kofuji, 2010) could also contribute to the response diversity observed between the PLR (Mure et al., 2009) and the melatonin suppression responses (current study).
Establishing whether concurrent administration of red light can significantly alter nonvisual responses to blue light is important as this experimental paradigm is likely to be more practical and applicable than the consecutive light pulses used in previous studies (Mure et al., 2009). Under the current experimental conditions, there was no evidence of simultaneous red light significantly altering nonvisual responses to blue light alone. Future optimization of lighting for health and well-being will require the relative contribution of each photoreceptor class to be deciphered as well as the complexities of the melanopsin states to be defined. Recent work in transgenic mice lacking rods and cones has demonstrated that the melanopsin photosensitivity function alone can predict circadian and pupil responses to a diverse range of poly-chromatic lights (Enezi et al., 2011). These data are in agreement with the current study and, although not precluding the existence of melanopsin bistability, suggest that the red long-wavelength content of a light stimulus may not be a significant factor contributing to nonvisual responses to polychromatic light.
Footnotes
Acknowledgements
This work was supported by the 6th Framework project EUCLOCK (018741).
The authors thank Philips Lighting, Eindhoven, The Netherlands, for providing the light sources, and Stockgrand Ltd. for providing radioimmunoassay reagents. They also thank Daniel Barrett, Dr. Shelagh Hampton, Dr. Benita Middleton, Ivonne Vogt, and the Surrey CRC clinical and medical teams for their assistance.
The author(s) have declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: V.L.R. acts as a scientific advisor to Lumie Ltd, Cambridge, UK. D.J.S. is a director of Stockgrand Ltd, Guildford, UK.
