Abstract
Many nonvisual functions are regulated by light through a photoreceptive system involving melanopsin-expressing retinal ganglion cells that are maximally sensitive to blue light. Several studies have suggested that the ability of light to modulate circadian entrainment and to induce acute effects on melatonin secretion, subjective alertness, and gene expression decreases during aging, particularly for blue light. This could contribute to the documented changes in sleep and circadian regulatory processes with aging. However, age-related modification in the impact of light on steady-state pupil constriction, which regulates the amount of light reaching the retina, is not demonstrated. We measured pupil size in 16 young (22.8 ± 4 years) and 14 older (61 ± 4.4 years) healthy subjects during 45-second exposures to blue (480 nm) and green (550 nm) monochromatic lights at low (7 × 1012 photons/cm2/s), medium (3 × 1013 photons/cm2/s), and high (1014 photons/cm2/s) irradiance levels. Results showed that young subjects had consistently larger pupils than older subjects for dark adaptation and during all light exposures. Steady-state pupil constriction was greater under blue than green light exposure in both age groups and increased with increasing irradiance. Surprisingly, when expressed in relation to baseline pupil size, no significant age-related differences were observed in pupil constriction. The observed reduction in pupil size in older individuals, both in darkness and during light exposure, may reduce retinal illumination and consequently affect nonvisual responses to light. The absence of a significant difference between age groups for relative steady-state pupil constriction suggests that other factors such as tonic, sympathetic control of pupil dilation, rather than light sensitivity per se, account for the observed age difference in pupil size regulation. Compared to other nonvisual functions, the light sensitivity of steady-state pupil constriction appears to remain relatively intact and is not profoundly altered by age.
Keywords
Light is perceived by the visual system to allow conscious vision but also by a photoreceptive system based on light-sensitive, melanopsin-expressing intrinsically photosensitive ganglion cells (ipRGCs) optimized to detect changes in light irradiance rather than to participate in image formation (Brown et al., 2010; Hatori and Panda, 2010; Schmidt et al., 2011). This non–image-forming (NIF) photoreceptive system is responsible for circadian entrainment but also acutely regulates many other nonvisual functions such as melatonin secretion, sleep, alertness, performance, cognitive brain functions, and pupillary constriction (Brainard and Hanifin, 2005; Chellappa et al., 2011; Hatori and Panda, 2010; Schmidt et al., 2011; Vandewalle et al., 2009). The NIF system shows a peak sensitivity at around 460 to 480 nm (blue light) (Brainard and Hanifin, 2005; Mure et al., 2009) so that the impact of light on nonvisual functions is greater under shorter wavelength blue light compared to lights of longer wavelength such as green light (Cajochen et al., 2005; Lockley et al., 2003; Lockley et al., 2006; Mure et al., 2009; Mure et al., 2007; Vandewalle et al., 2011; Vandewalle et al., 2009). This maximal sensitivity to short wavelength light is primarily due to the recruitment of melanopsin ipRGCs, which also receive modulatory inputs from rods and cones (Berson, 2003; Hatori and Panda, 2010; Provencio et al., 2000; Schmidt et al., 2011). ipRGCs project directly to numerous brain regions including the suprachiasmatic nucleus (SCN), site of the master circadian clock, the ventrolateral preoptic nucleus (VLPO), which contains sleep-active neurons, and the olivary pretectal nucleus (OPN), crucial for pupillary constriction (Berson et al., 2010; Hatori and Panda, 2010; Schmidt et al., 2011).
Aging is associated with changes in functions affected by the nonvisual photoreception system including sleep and circadian entrainment (Turner et al., 2010). For instance, studies have demonstrated that healthy aging is associated with advanced sleep timing, more wakefulness during the sleep episode, and increased rate of napping (Buysse et al., 1992; Carrier et al., 2001; Carrier et al., 1997). Age-related changes in the circadian timing system such as phase advance, reduced amplitude of circadian rhythms, and problems to adapt to circadian challenges (e.g., shift work, jet lag) have also been reported (Carrier et al., 1996; Duffy et al., 1998; Kawinska et al., 2005; Monk et al., 1995). These age-related modifications may be triggered in part by a decreased sensitivity to light or by the decreased ability for light to drive nonvisual functions. Indeed, the impact of light on the circadian phase, and therefore on circadian entrainment, seems to diminish with aging (significant impact found in Duffy et al. [2007] but not in Sletten et al. [2009]). Recent evidence also suggests that acute effects of light such as melatonin suppression, subjective alertness enhancement, and induction of
Major modifications occur with aging at the level of the eye such as a decrease in scotopic and photopic sensitivity as well as in the number of retinal photoreceptors (Freund et al., 2011; Hebert et al., 2004; Sturr et al., 1997). Due to progressive opacification and yellowing of the lens, light transmission decreases with age, especially for shorter wavelength (blue) light (Kessel et al., 2010). In addition, aging is associated with a substantial decrease in pupil size (senile miosis) in darkness and under various light irradiance levels (Bitsios et al., 1996; Winn et al., 1994). PLR is characterized by an initial, transient, and robust phasic constriction at light onset followed by a sustained steady-state tonic response at a lower constriction level (McDougal and Gamlin, 2010; Mure et al., 2009). Age-related changes in the phasic response have been observed (Bitsios et al., 1996). Yet, how aging affects the wavelength sensitivity of steady-state pupil constriction and its response as a function of changes in irradiance is unknown.
In the present study, we first assessed how the amount of light reaching the retina changes with age by measuring pupil size under green (550 nm) and blue (480 nm) light exposures at different irradiance levels in healthy young and older individuals. We then assessed whether age-related changes in pupil size affected steady-state PLR, defined here as the relative change in pupil size under these light conditions, and whether the ability of light to trigger this nonvisual response changes with aging. We anticipated that, in older compared to young subjects, a smaller pupil size would be observed under all light conditions, accompanied with a reduction in steady-state PLR, particularly for blue light.
Materials and Methods
Subjects
Healthy subjects, 16 young (22.8 ± 4 years; 10 females) and 14 older (61 ± 4.4 years; 10 females), participated in the present study. Recruitment interviews established the absence of medical, traumatic, psychiatric, or sleep disorders. Questionnaires were used to exclude candidates with extreme chronotype (Morningness-Eveningness Questionnaire [MEQ] ≤30 and ≥70) (Horne and Ostberg, 1976), poor sleep quality (Pittsburg Sleep Quality Index [PSQI] ≥7) (Buysse et al., 1989), high anxiety (Beck Anxiety Inventory [BAI] ≥11) (Beck et al., 1988), or depression (Beck Depression Inventory II [BDI-II] ≥11) (Steer et al., 1997) scores (Table 1). Candidates with a body mass index >27 were excluded. None had worked on night shifts during the preceding year or traveled across more than one time zone during the last 2 months. All participants were nonsmokers, moderate caffeine and alcohol consumers, and were not using medication, except for hormonal contraceptives in 9 of 10 young women. Prior to participation, subjects went through an extensive professional ocular examination to confirm normal color vision and the absence of ocular problems. Lens opacification as well as the absence of cataracts was assessed subjectively using the Lens Opacities Classification System III (LOCS-III) from 1 (clear lens) to 5 (cataract) (Chylack et al., 1993). All young subjects had clear lenses (level 1), and all older subjects’ lenses were classified below level 4. This experiment was performed in accordance with institutional guidelines and received the necessary ethical approvals. Written informed consent was obtained from each subject.
Subjects’ characteristics (number of subjects or mean ± standard deviation).
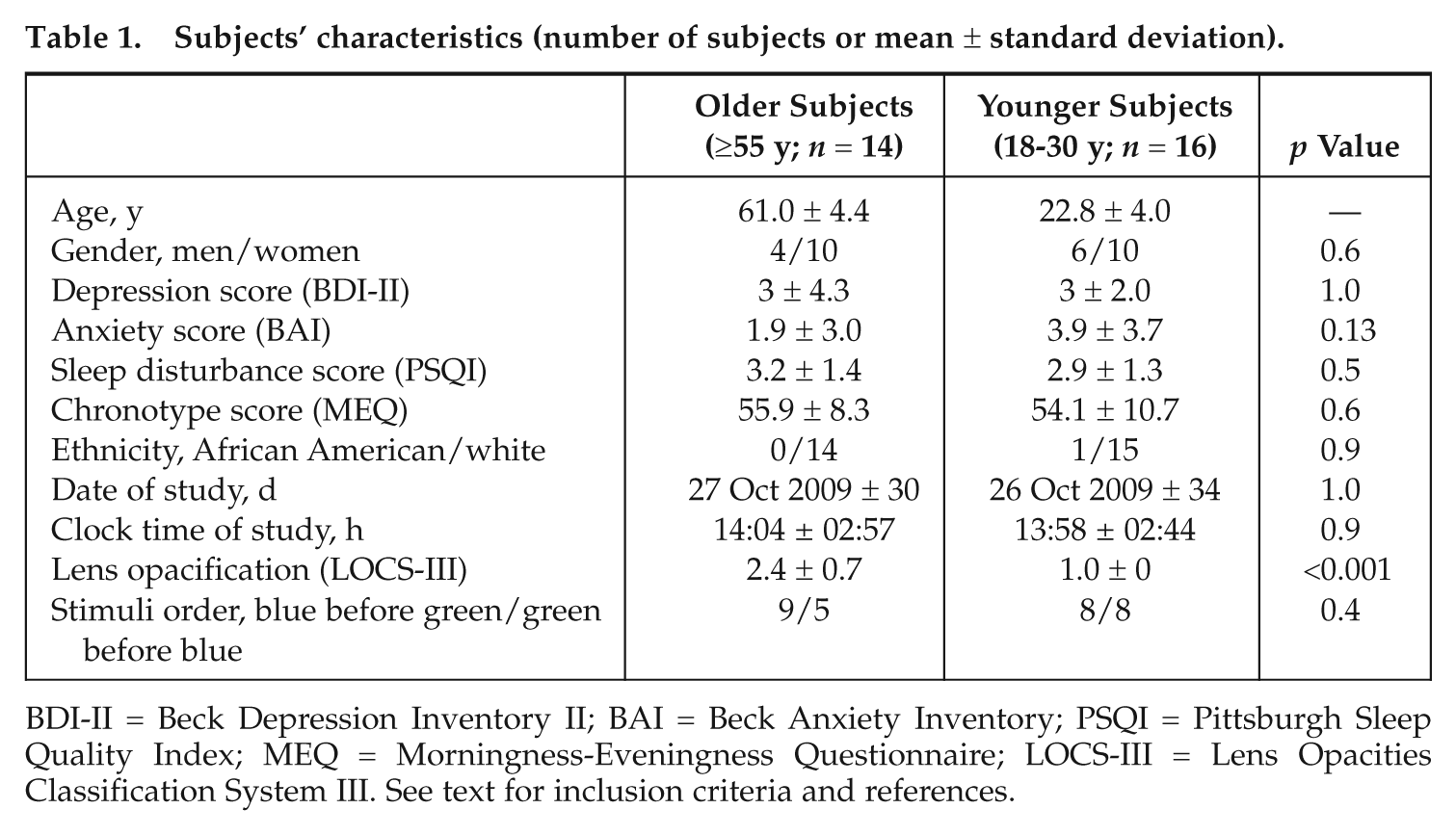
BDI-II = Beck Depression Inventory II; BAI = Beck Anxiety Inventory; PSQI = Pittsburgh Sleep Quality Index; MEQ = Morningness-Eveningness Questionnaire; LOCS-III = Lens Opacities Classification System III. See text for inclusion criteria and references.
Protocol
Upon arrival, subjects were dark-adapted for 15 minutes (0 lux), and baseline pupil size was assessed at the end of this adaptation period (Fig. 1). Pupil size was then measured under blue (480 nm; full width at half maximum, FWHM = 10 nm) and green (550 nm; FWHM = 10 nm) monochromatic light exposures presented at low (7 × 1012 photons/cm2/s), medium (3 × 1013 photons/cm2/s), and high (1014 photons/cm2/s) irradiance levels. Light exposures lasted 45 seconds, during which subjects were asked to maintain a fixed gaze, and were separated by 2 minutes of darkness (0 lux; free gaze or eyes closed). Three light exposures of each wavelength were administered for the 3 irradiance levels (pseudorandom order; high irradiance was never administered first). The order of blue and green monochromatic lights was counterbalanced between subjects and groups. Pre-exposure pupil size was assessed at the end of each 2-minute period in darkness. Note that we verified that pseudorandomization prevented an exposure to significantly affect the subsequent exposure (supplementary material). Data were collected during the day between 1000 h and 2000 h. Average clock time of testing did not differ between the 2 groups, and all experiments were carried out in the fall season.
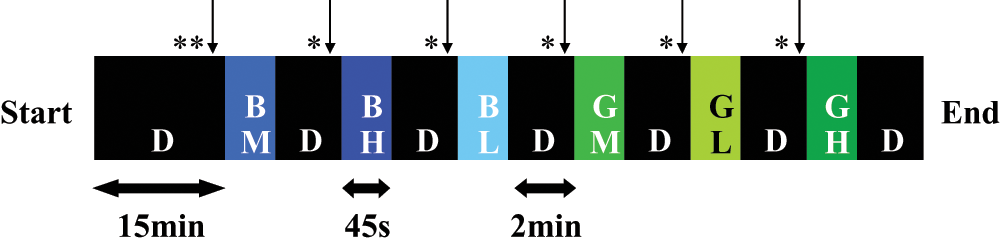
Experimental protocol. Example for one subject. Wavelength and irradiance were counterbalanced between subjects and groups. *↓ = baseline pupil value; **↓ = pre-exposure pupil value; D = darkness; BL, BM, and BH = blue light of low, medium, and high irradiances, respectively; GL, GM, and GH = green light of low, medium, and high irradiances, respectively.
Light Settings and Data Acquisition
Narrow interference band-pass filters (Edmund Optics, Barrington, NJ) were used to produce blue and green monochromatic lights using a white light source (PL950, Dolan-Jenner Industries, Boxborough, MA). Switch between filters was achieved using a filter wheel (AB301-T, Spectral Products, Putnam, CT) inserted between the light source and an optic fiber (Dolan-Jenner Industries). The latter brought light to a diffusing glass placed 2 cm in front of the subject’s left eye. The light source and the filter wheel were computer-controlled and synchronized with pupil data acquisition using COGENT2000 (http://www.vislab.ucl.ac.uk/Cogent/) under MatLab 7.0.4 (MathWorks Inc., Natick, MA). Light spectra were assessed at the level of the diffuser (Lightspex, GretagMacbeth, Munich, Germany) and confirmed to peak at 480 nm and 550 nm. Irradiance levels were verified using a calibrated radiometer (PM100D, Thorlabs Inc., Newton, NJ).
Pupil size was recorded using an eye-tracking device (EyeFrame Scene systems, Arrington Research Inc., Scottsdale, AZ) consisting of an infrared camera mounted on eyeglass frames maintained at the level of the nasium. As PLR is a consensual mechanism (i.e., light in one eye constricts the pupil in the other eye), light was administered to the subject’s left eye while pupil size was captured from the right eye. Pupils were not pharmacologically dilated because the aim of the present study was to investigate whether changes in pupil size affected steady-state PLR. In addition, it was shown that pupils of older individuals remain smaller relative to that of younger individuals even when pupil constriction is pharmacologically blocked (Kergoat et al., 2001). Pupil width and height (arbitrary units relative to the frame of the display window on the computer screen) were acquired at a sampling rate of 60 Hz, and the display frame was constant for all acquisitions to ensure equivalence of the unit size.
Data Analysis
Data were analyzed using MatLab 7.10 (MathWorks Inc.). Artifacts in pupil measures, including eye movements and blinks, were excluded using a circularity coefficient exclusion criterion (ratio between width and height <0.70 or >1.30). Following published procedures (Canver et al., 2010; Wyatt, 2010), pupil data were smoothed using a nonparametric locally weighted linear regression (smoothing type: rlowess; span: 0.8). In our experimental setting, the transition between lights-on and lights-off was not sharply regulated (~500 milliseconds for full illumination or extinction), therefore not allowing a reliable analysis of phasic PLR. As a consequence, we excluded the first 6 seconds of each illumination to make sure that we only included a stable response and averaged pupil size during the remaining 39 seconds of each light exposure (representative data are displayed in Suppl. Fig. S1). Baseline pupil size and pre-exposure pupil size were estimated by averaging the last second (60 data points) of the 15 or 2 minutes of darkness prior to illumination, respectively. Before analyses, pupil size was inferred by computing the ellipsoidal surface obtained with width and height arbitrary units (p × ½ width × ½ height). Steady-state PLR was estimated by normalizing mean pupil size during the illumination according to baseline pupil size or pre-exposure pupil size. Statistical analyses were computed in SPSS 17.0 (SPSS Inc., Chicago, IL) using a 2-tailed Student
Results
Analysis of baseline (dark-adapted) pupil size (i.e., following 15 minutes in darkness) showed that older subjects (arbitrary units [a.u.], mean ± SEM = 0.08 ± 0.009) had a smaller pupil size than young subjects (0.11 ± 0.008) (
The repeated-measures 3-way ANOVA on absolute pupil size during illumination (no normalization) revealed a significant interaction between age groups and irradiance (
We then normalized sustained pupil size during light exposure according to baseline pupil size to analyze PLR (Fig. 2). The repeated-measures 3-way ANOVA of normalized pupil values revealed that blue light induced significantly more constriction (66.67% ± 2.10%) than green light (64.47% ± 2.25%) (
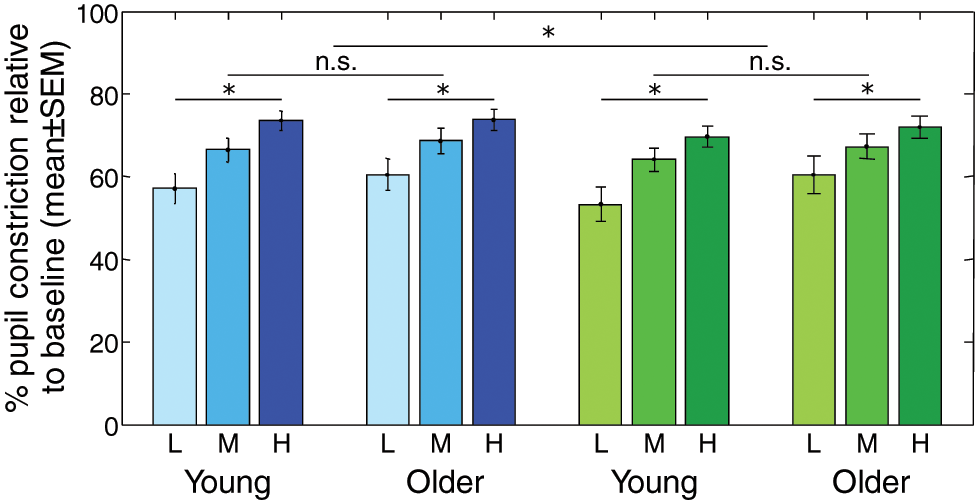
Steady-state pupil constriction in young and older individuals. Mean pupillary constriction relative to baseline ± SEM in each age group. Blue bars: blue light at low (L), medium (M), and high (H) irradiances. Green bars: green light at low (L), medium (M), and high (H) irradiances. Effects of wavelength and irradiance levels were significant (*
We also computed supplementary analyses in which only the last 5 seconds of each illumination were included (40-45 seconds). The statistical results obtained are identical to those obtained previously, both with absolute and normalized data (supplementary material).
Discussion
This study investigated the effects of age on pupil size and steady-state PLR using shorter (blue = 480 nm) and longer (green = 550 nm) wavelength monochromatic light exposures at 3 different irradiance levels. Our results confirm that older subjects have a smaller pupil size than young subjects (Bitsios et al., 1996; Winn et al., 1994). This difference was observed both after prolonged dark adaptation and during blue and green light exposures at the 3 irradiance levels. The observed interaction between irradiance level and age groups is in agreement with previous results that reported larger age-related decreases in pupil size under lower compared to higher irradiance levels (Winn et al., 1994). Our results also support previous reports showing that PLR is greater under blue than green light exposure and for higher irradiances (Mure et al., 2009; West et al., 2011). However, despite the fact that irradiance levels exerted a stronger impact on the absolute pupil size of younger compared to older individuals, steady-state PLR did not differ between the 2 age groups for either blue or green light exposures at the 3 irradiance levels.
A previous study by Bitsios et al. (1996) showed no age-related differences under white light for the latency of the phasic PLR, while other parameters of the phasic response were affected by age (amplitude, maximum constriction velocity, maximum constriction acceleration), although steady-state pupil constriction was not investigated. At the eye level, senile miosis is one of the primary functional mechanisms reducing the amount of light reaching the retina (Winn et al., 1994). In our sample, the observed age-related reduction in pupil size (~30%) is comparable with previous reports in young and older populations (Bitsios et al., 1996). Senile miosis may constitute a contributing factor to the reduced impact of light on nonvisual functions in older subjects (Herljevic et al., 2005; Jud et al., 2009; Sletten et al., 2009). Indeed, studies have reported a correlation between pupil size and light-induced melatonin suppression in young subjects (Gaddy et al., 1993; Higuchi et al., 2008). In our study, despite the 30% reduction in pupil size and the increase in lens opacification (for a professional assessment, see LOCS-III [Steer et al., 1997] score in Table 1), older subjects showed a steady-state PLR similar to that of younger subjects. Future studies should investigate the functional impact of age-related reduction in pupil size on other nonvisual responses to light.
One explanation for the lack of difference could be that compensatory mechanisms may allow a normal steady-state PLR despite the age-related decrease in the amount of light reaching the retina. For instance, an increased sensitivity to light may develop with age to compensate for chronic exposure to lower levels of light due to lens opacification. Several studies in young individuals have shown that, following prolonged exposure to low light levels, exposure to light induces stronger suppression and phase shift of melatonin secretion (Chang et al., 2011; Hebert et al., 2002; Jasser et al., 2006). An alternative explanation for the smaller pupil size in the elderly, despite the lack of a difference in relative pupil constriction, is a loss of tonic control of pupil dilation, previously suggested in a study of nonhuman primates (Clarke et al., 2003). The authors of the study reported that pharmacological blockade of the sympathetic pupillodilator pathway reduces pupil size but does not affect PLR dynamics to different broadband white light intensities. The idea of a loss of autonomic control in the aged is consistent with the results in our study that showed smaller absolute pupil areas in the older subjects but no difference in steady-state PLR under blue or green wavelength light exposures.
A statistical power analysis on steady-state PLR indicated a small effect size for the main effect of age (0.13) that would require 230 subjects per group to reach a power of 0.80 (a = 0.05), supporting the notion that age did not affect the PLR response in the present study. However, interactions between group and irradiance and between group and wavelength showed medium effect sizes (0.24 and 0.30, respectively) that would require 42 and 45 subjects per group to reach a power of 0.80 (a = 0.05). Studies with larger samples are required to gain a better understanding of age-related interactions according to wavelength and irradiance light levels. Furthermore, future studies should consider extending the duration of light exposure.
In general agreement with previous hypotheses (Revell and Skene, 2010), our data suggest that, although aging alters several nonvisual functions regulated by light, the degree to which the impact of light on these functions is reduced may vary considerably. It is worth noting that our sample was slightly younger than that of previous studies showing age-related differences in nonvisual effects of light (61 years on average in our sample v. 65.8 years [Jud et al., 2009; Sletten et al., 2009] or 68.3 years [Duffy et al., 2007]). However, such small age differences appear unlikely to fully explain the very limited effect of aging on steady-state PLR in our data compared to that of other studies. Interestingly, another recent investigation did not find a significant impact of age on the ability of light to phase-shift circadian rhythms (Sletten et al., 2009), contrary to a previous investigation (Duffy et al., 2007).
The lack of age-related differences in the steady-state PLR response compared to that reported in other nonvisual responses may be related to the involvement of different melanopsin ganglion cell types and their target brain structures or the output pathways involved. For instance, recent studies in rodents have found that light sensitivity is not equivalent for all nonvisual functions regulated by light. The sensitivity of the circadian system for entrainment and phase shift was greater than that of PLR or masking (Butler and Silver, 2011; Hut et al., 2008). There is evidence that different populations of melanopsin-expressing ipRGCs may mediate these light sensitivity differences. Five types of these ipRGCs (M1-M5) have been identified to date, their projections overlapping only partially (Berson et al., 2010; Ecker et al., 2010). In addition, 2 subtypes of M1 cells have been identified, with one subtype innervating mainly the SCN while the other projects to all other known brain targets of ipRGCs including the OPN (Chen et al., 2011). In this respect, it appears plausible that the impact of aging varies for different nonvisual functions regulated by light, such as circadian entrainment, melatonin secretion, and PLR, which present different light sensitivities and are mediated, at least in part, by different populations of ipRGCs.
Footnotes
Acknowledgements
We thank Jean Paquet for his support on statistical analyses as well as CARMS members for their critical comments on the article. This work was funded by the IRSC, CRSNG, FRSQ, Fonds France-Canada pour la recherche, Fonds
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
