Abstract
Polymeric polyisocyanate prepolymer substances are reactive intermediates used in the manufacture of various polyurethane products. Knowledge of their occupational and environmental hazard properties is essential for product stewardship and industrial hygiene purposes. This work reports on the systematic design of a program to explore how structural features (i.e., types of polymeric polyol and diisocyanate reactants, functionality) and physical–chemical properties (i.e., octanol–water partition coefficient [log Kow], viscosity, molecular weight) of a group of 10 toluene diisocyanate (TDI)- and methylene diphenyl diisocyanate (MDI)-based monomer-depleted prepolymer substances can be related to their exposure and hazard potentials. The revelation of trends or thresholds in such relationships could form a basis for regulatory screening of existing or new prepolymer substances, while also informing the design of substances having reduced exposure and/or hazard profiles. As a first step, the aquatic exposure and hazard potentials of these 10 substances were investigated. The results of this investigation showed that yields of dissolved reaction products (derived from non-purgeable organic carbon measurements and carbon contents of the parent prepolymers) were inversely correlated with the calculated log Kow of the substances. For prepolymer loading rates of both 100 and 1000 mg/L in water, the average dissolved reaction product yields ranged from ≤1% to 32% and from ≤0.1% to 25%, respectively, over calculated log Kow values ranging from −4.8 to 45. For both loading rates, dissolved reaction products were not quantifiable where the calculated log Kow value was >10. Yet, none of the 10 prepolymers and tested loading rates exhibited acute adverse effects on the aquatic invertebrate, Daphnia magna, in the 48-h acute immobilization test. From a product stewardship perspective, polymeric prepolymers of TDI and MDI within the investigated domain and concentration range are not expected to be hazardous in the aquatic environment.
Keywords
Introduction
Aromatic diisocyanates (DII) are versatile and reactive species. Many polyurethane (PU) applications make use of the monomers toluene diisocyanate (TDI) or methylene diphenyl diisocyanate (MDI), for which the hazardous properties and corresponding industrial hygiene measures for safe handling and product stewardship programs are well-established. Of the most relevance in this context is how to handle effects of incidental emissions into the (mainly aquatic) environment and how to protect workers and users from potential adverse health effects. Regarding human health, the main concern is the potential for sensitization. When diisocyanates contact water, the main reaction products are oligomeric or polymeric substituted ureas, and only minor amounts of the corresponding diamine are formed (Allport et al., 2003). The substituted mono-ureas of TDI and MDI have been synthesized to serve as the lowest-molecular-weight representatives and were found to be non-toxic to aquatic species in standardized tests (Loddenkemper et al., 2017, 2019; Neuhahn et al., 2020). 1 Hence, it appears that the aquatic toxicity of TDI and MDI is mainly driven by the trace amount of toluene dianiline or methylene dianiline (TDA or MDA, respectively) that is formed (Allport et al., 2003). Toxicity of TDI and MDI to bacteria, algae, and freshwater fish was generally low with median effective loading (EL50) values >100 mg/L. Invertebrates, represented by Daphnia magna (D. magna), were consistently identified as the species most sensitive to the primary aromatic amines, which can occur as DII hydrolysis products, with EL50 values in the range of 3–20 mg/L (Allport et al., 2003; Chen et al., 2007; ECHA, 2022).
In many other PU applications, the monomeric diisocyanates are substituted by prepolymers or other oligomers (e.g., isocyanurates, biurets in coatings) for the purpose of controlling processing or product characteristics, or to reduce exposure potential (e.g., reduced vapor pressure) (Szycher, 2017). Polymeric (polyisocyanate) prepolymers are produced by capping a polymeric polyol (i.e., a polyol that satisfies the OECD definition of a polymer [OECD, 1993]) with an aromatic diisocyanate, often followed by removal of the excess diisocyanate (Allport et al., 2003) (e.g., by distillation) to <0.1% (Sonnenschein, 2014). Having at least two terminal isocyanate groups (-N=C=O or NCO), polymeric prepolymers remain reactive substances. Under major global legislation, substances containing one or more NCO groups are classified as sensitizers and hazardous to the environment by default, regardless of other substance properties such as molecular weight or hydrophobicity (ECCC (Environment and Climate Change Canada), 2021; Ge, 2021; EPA (United States Environmental Protection Agency) (1997)). While a precautionary approach for potential human and aquatic effects is understandable, it also lessens the incentive to develop innovative products with reduced hazard potential.
The introduction of prepolymer innovations requires the assessment of their potential hazards to comply with chemical control legislation (e.g., EPA, 2010). Since these substances typically have proprietary compositions, and because testing methods for evaluating their exposure and hazard properties are not always consistently applied across substances, predicting exposure and hazard properties of prepolymers from their structural and physical–chemical characteristics has, until now, not been feasible. In the present work, a comprehensive program was designed to synthesize, characterize, and test a set of 10 generic polymeric prepolymers with a targeted residual aromatic diisocyanate monomer content below 500 mg/kg (0.05%) for properties relevant for product stewardship and industrial hygiene purposes. This paper reports on the design of the experimental program and the investigation of hazards to the aquatic environment. In a companion paper, the dermal sensitization potency of a broadened set of polymeric prepolymers produced from both aromatic and aliphatic diisocyanates was investigated and reported (West et al., 2022).
Materials and methods
Experimental design
Ten polymeric prepolymer substances were designed according to a screening-level design of experiments (DOE) wherein design factors such as DII monomer type, polyol type and terminal OH groups, molecular weight, NCO functionality,
2
and calculated octanol–water partition coefficient (log Kow) were varied between lower, mid-point, and upper limits to represent a considerable breadth of existing and potential future commercial polymeric prepolymer products. The boundaries for this DOE were based on input of several industry experts in isocyanate prepolymer chemistry (see Acknowledgments) with the goal of covering key structural features and properties exhibited across a majority of commercially available polymeric prepolymers of TDI and MDI. The testing of commercial prepolymer products was avoided for reasons of confidentiality and to enable increased flexibility in designing the test program. A computer-optimized fractional-factorial DOE was not utilized since incorporation of some optimized design factors (and combinations thereof) into the prepolymer substances was not possible using commercially available polyol reactants. Figure 1 illustrates the structural and property domains covered by the 10 prepolymer test substances, generic structures of which are shown in Figure 2. The triangular shape of the experimental domain as illustrated in Figure 1 relates to some key boundaries imposed on the design of the polymeric prepolymer test substances. For example, the left apex is defined by the large contribution by the DII monomer moieties to the calculated log Kow of the polymeric prepolymers (MDI ≈ 5.2, TDI ≈ 3.7) and the requirement that the lowest molecular weight polymeric prepolymers are made from the reaction of at least two DII equivalents with a polyol that meets the OECD polymer definition (OECD, 1993). The slope of the upper face is determined by the maximum contribution to log Kow of the most hydrophobic polymeric polyol repeat units (e.g., caprolactone ≈ 1.5; TMEG
3
≈ 0.7 per unit), while the base of the triangle in a similar fashion is determined by the minimum (most negative) contribution to log Kow associated with the hydrophilic ethoxylate repeat unit (≈−0.27 per unit). An upper limit to the number-average molecular weight (Mn ≈ 4500 g/mol) is indicated by the vertical leg of the triangle, where it was determined that prepolymers with greater Mn are diminished in both commercial importance and regulatory concern. Likewise, unique polymeric prepolymers based on aromatic-, polycarbonate-, and amino-ether-based polyols as well as polyamines were excluded to minimize complexity of this screening-level evaluation. Only three MDI-based prepolymers of relatively high Mn were included in the design since the low vapor pressure of MDI makes it increasingly difficult to remove the monomer from prepolymers of decreasing molecular weight. Illustration of the structural and parametric domain covered by the 10 generic aromatic polymeric polyisocyanate prepolymer test substances, showing calculated octanol–water partition coefficients (log Kow) and number-average molecular weights (Mn) for MDI- (●) and TDI-(■) based difunctional and trifunctional ( Generic structures of the polymeric polyisocyanate prepolymer test substances prepared by reaction of 2,4:2,6-toluene diisocyanate (TDI; 65:35) and 4,4’-methylenediphenyl diisocyanate (MDI) with various polyethylene glycol (PEG), polypropylene glycol (PPG), polyethylene glycol–polypropylene glycol–polyethylene glycol (PPG/PEG tri-block), glycerin propoxylate (GLY/PPG), and diethylene glycol polycaprolactone (DEG/PCL) polyol substances. Structures were drawn using PubChem Sketcher V.2.4.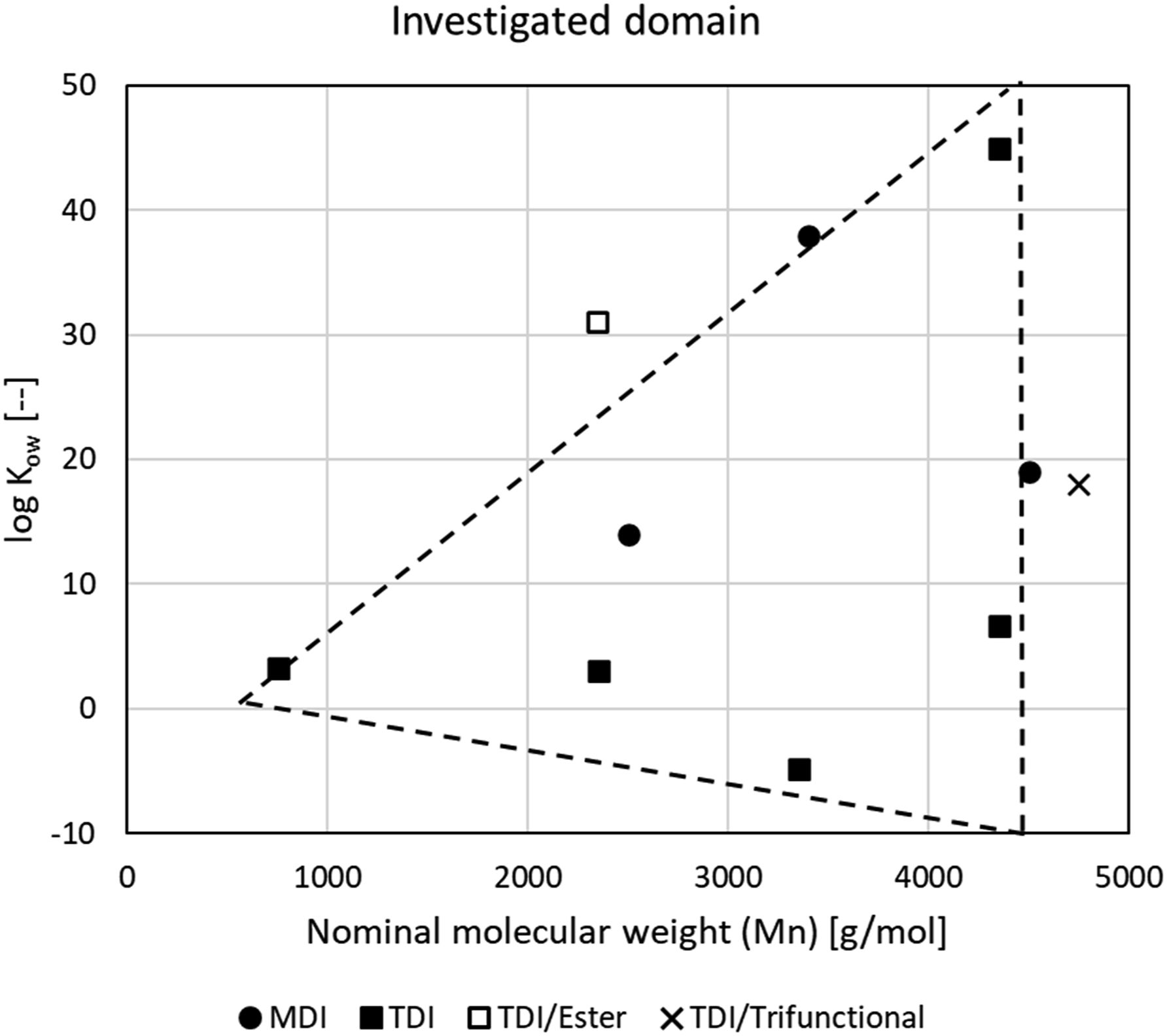
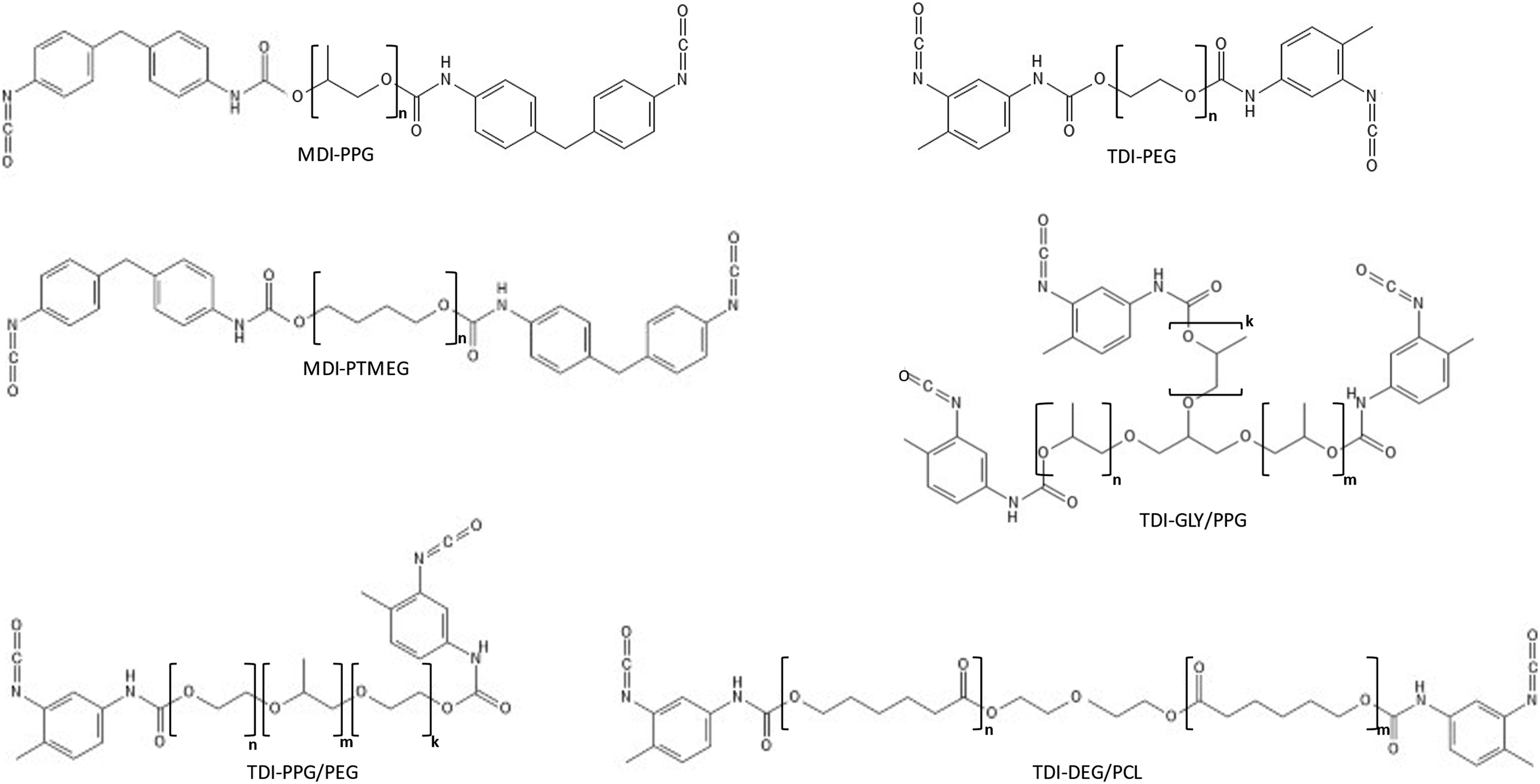
Synthesis of test substances
The polymeric prepolymer test substances were synthesized from commercially available TDI (MONDUR TD-65; Covestro LLC, Pittsburgh, PA), 4,4′-MDI (COSMONATE-PH; Kumho Mitsui Chemicals, Seoul, Korea) and polyol substances (Table S2 in Supplemental Information) at Anderson Development Company (Adrian, Michigan, USA) as described in the Supplemental Information. The polyols were selected from commercially available products with the narrowest available distributions in molecular weight and functionality for a given targeted polyol type and molecular weight. These selection criteria served to ensure that results could be interpreted based on nominal prepolymer features (e.g., functionality, Mn) and were not biased or confounded by constituents having higher molecular weight or lesser functionality than the targeted substances. Water content of each polyol was verified by Karl-Fischer titration and was reduced where necessary to <300 mg/kg by heating under vacuum.
The potential (hydrolytic) degradation of the finished prepolymer substances was minimized by immediate transfer into several 250 mL brown polypropylene bottles that were sealed under dry N2 gas with air-tight screw caps wrapped with vinyl tape. These containers were not opened until immediately prior to use in the testing and were immediately re-sealed under dry N2 gas atmosphere.
Characterization of test substances
Determination of NCO content, Mn, and polydispersity
The polymeric prepolymer test substances were characterized for NCO content (%NCO) by titration with dibutylamine (ISO 14896:2019). Viscosities were measured at temperatures of 60, 80, and 100°C (ASTM D4889-21, 2021). The number-average molecular weight (Mn) of each prepolymer was determined from the %NCO titration as follows:
Mn (g/mol) = (Functionality * 42 g/molNCO) * 100/%NCO
Each of the polymeric prepolymer test substances was also qualitatively analyzed by gel permeation chromatography (GPC) to verify the expected narrow molecular weight distributions and low quantities of adducts with multiple polyol units (data not shown).
Determination of residual monomer content
The residual DII monomer content of the test substances was determined by a catalyzed derivatization with methanol in anhydrous acetonitrile (ACN) and subsequent quantitation by HPLC-MS/MS. Preliminary method development work had revealed that the conventional derivatization with dibutylamine (e.g., ISO 17734-1:2013) resulted in partial aminolysis of the urethane bonds between the DII and the polyol moieties for some of the test substances that was sufficient to distort the measurements. A “softer” method of derivatization with methanol was used in this study, wherein a 1-g sample of the prepolymer substance was dissolved into 25 mL ACN, to which 4 mL of methanol (HPLC-grade) and two drops of dibutyl tin dilaurate catalyst were subsequently added. The resulting solution was brought to 50 mL with additional ACN, then mixed and placed in an oven at 55°C for 30 min. The samples were allowed to sit overnight, then diluted 1/10 in ACN, filtered (0.45 μm nylon syringe filters), and analyzed by HPLC. Chemicals used as well as calibration and analytical details are described in the Supplemental Information.
Calculation of log Kow
Log Kow values were calculated using the EPI-Suite KOWWIN module (v1.68; US EPA, Washington DC, USA) by input of SMILES (Simplified Molecular Input Line Entry) codes for a representative structure of each prepolymer substance having a formula weight approximating the Mn determined as above from %NCO.
Aquatic exposure potential
Test medium
A modified depleted mineral medium (DMM) was used in evaluating the aqueous solution/extraction behavior of the prepolymer substances, and subsequently, the acute aquatic toxicity of the water-accommodated fractions (WAF) to Daphnia magna (D. magna). This mineral medium was used as an alternative to the formulated Elendt M4 medium recommended in OECD Guideline 202 (OECD, 2004) because the latter contains constituents (e.g., phosphates, EDTA, 4 vitamins) that could potentially react with the NCO-bearing test substances. The DMM composition was based on the alternative “ISO test water” recommended in the OECD Guideline 202 with the amounts of calcium chloride and magnesium sulfate reduced by 10% to achieve a final hardness of ≤250 mg/L as CaCO3 as required by OECD Guideline 202 (OECD, 2004). The DMM was prepared from calcium chloride dihydrate (264.42 mg/L), magnesium sulfate heptahydrate (110.97 mg/L), potassium chloride (5.8 mg/L), and sodium bicarbonate (64.8 mg/L) in Millipore water and had a hardness of approximately 230 mg/L (as CaCO3) and a pH of approximately 8.0. Preliminary testing demonstrated the acceptable health and survival of D. magna adults and neonates in the DMM, which was also demonstrated in all control exposures associated with the prepolymer substance testing.
Water extractability test
The solution/extraction behavior of the prepolymer substances in water was evaluated using a procedure based on OECD Guideline 120 (EC, 2008; OECD, 2000) but with some key modifications. Although the guideline specifies testing of polymers at a loading of 10 g/L in water, prior experience with such testing of DII monomer and non-polymeric prepolymers had indicated that the yield of water-soluble hydrolysis products (as percentage of the initial polymer loading) generally increases with decreasing loading rate (Yakabe et al., 1999). Alternative loading rates of 100 and 1000 mg/L, corresponding to the highest (limit) concentrations typically associated with aquatic toxicity testing and associated aquatic hazard classifications, were used to define this inverse relationship with yield of dissolved hydrolysis products. Additionally, the solution/extraction behavior was determined at 24-h intervals over a total of 96 h, as opposed to the standard 24-h mixing time, with the intent to determine the stirring time necessary to achieve maximum dissolved exposure concentrations in the prepolymer WAFs prepared for the D. magna acute immobilization test. The water extractability of each prepolymer substance was tested using the same equipment, procedures, and conditions, while minimizing the extent to which differences in their physical forms (ranging from viscous liquids to dense waxy solids) affected the degree of dispersion into the DMM. The test was conducted similarly to the method employed by Yakabe et al. (1999). For better reproducibility, weighed 100- or 1000-mg portions of test substance were placed against the inside bottom corners of 1 L glass beakers (in triplicate) and cautiously overlaid with 1 L of the DMM. The beakers were covered with aluminum foil and the mixtures were stirred using PTFE-coated magnetic stir bars at 60 rpm for 96 h while avoiding contact of the stir bar with the test substance. Evaporated water was replaced with Millipore water at regular intervals, and aliquots (60 mL) were taken after 0, 24, 48, 72, and 96 h for analysis of dissolved organic carbon (DOC). Results of the DOC analyses were used to determine the stirring times to be used in preparation of WAF solutions for each prepolymer substance in the subsequent D. magna immobilization tests. The results also gave an indication of the reproducibility of DOC formation across the replicates, which cannot be easily achieved without careful construction and stirring of the mixtures. A description of the method used for DOC analysis is included in Supplemental Information.
Polyurea analyses
The polyurea solids formed from hydrolysis of each prepolymer substance (at 1000 mg/L loading) over 96 h were collected and dried to constant weight, and equal weight portions were introduced into anhydrous tetrahydrofuran (THF) at 10 g/L and vigorously shaken. The extraction mixtures were then centrifuged, the supernatant was collected in a separate vial, and the remaining pellet was dried to constant weight. The mass of polyurea dissolved into THF was determined gravimetrically, and the THF extraction solution and associated solids were analyzed for presence of unreacted isocyanate groups using Surface SWYPE colorimetric indicating pads for aromatic isocyanates (Part No 769-1022; SKC Inc., Eighty Four, PA, USA). A 1-mL aliquot of the THF extraction solution was applied to the SWYPE absorbent pad and allowed to develop for 3 min. A 1-g portion of the extracted polyurea solid was placed onto a glass plate, covered with 1 mL of mineral oil, and overlain with an SWYPE absorbent pad. The SWYPE pad was then used to compress and wipe away the wetted polyurea sample from the glass plate and its color was allowed to develop for 3 min. The degree of orange–red color development in these SWYPE pads was compared to that from a series of SWYPE pads to which 1-mL portions of 2,4-TDI solutions (1–1500 mg/L) in THF were applied (Figure 3). From these comparisons, the approximate concentrations of THF-extractable (LOQ ∼ 400 μg/kg) and unextractable isocyanate (LOD ∼ 4 mg/kg) associated with the polyurea solids could be determined. Calibration of surface SWYPE indicator pads with 2,4-TDI (left). Example of test results with TDI-PPG/PEG 2350 (right): the part of the precipitated ureas insoluble in THF (top) and the part soluble in THF (bottom) both show absence of residual unreacted isocyanate groups compared to calibration pads. PEG = polyethylene glycol; PPG = polypropylene glycol; TDI = toluene diisocyanate; THF = tetrahydrofuran.
Acute aquatic toxicity test
Test organism
The freshwater planktonic crustacean D. magna was used to evaluate the acute aquatic hazard potential of the polymeric prepolymer substances and their associated water-extractable hydrolysis products. This organism was chosen because of its sensitivity to primary aromatic amine substances (Chen et al., 2007) since any water-soluble hydrolysis products of the test substances would be expected to bear terminal primary aromatic amine groups. A population of parthenogenetic D. magna females of synchronized age structure has been maintained within the testing laboratory (Currenta GmbH & Co. OHG, Leverkusen, Germany) for more than 20 years under constant temperature (20 ± 2°C) and lighting (16:8 h light:dark, intensity <20 μE x m−2 x s−1) in formulated Elendt M4 medium that was renewed once a week. The Daphnia were exclusively fed unicellular green algae (Desmodesmus subspicatus) ad libitum, and neonates (<24 h post-hatch) were separated from their parents by filtration prior to use in the immobilization test.
Acute aquatic toxicity test
The assessment of acute aquatic toxic effects was conducted using static limit tests according to OECD Guideline No. 202 (OECD, 2004) and the recommendations of the OECD Guidance Document No. 23 on aqueous phase aquatic toxicity testing of difficult test chemicals (OECD, 2019). The Daphnia were exposed to WAFs derived from 100 and 1000 mg/L loadings of each test substance with no renewal over 48 h. The WAF were prepared by stirring the prepolymer substances in the modified DMM (in duplicate) for 48 h if DOC was less than the limit of quantitation (LOQ) of 2 mg/L over the full 96 h of the water extractability test. Otherwise, the WAFs were prepared using the stirring time (24–96 h) that produced the highest DOC concentration in the preceding water extractability test. After these defined stirring times, 200-mL portions of each replicate mixture were combined and filtered (Chromafil PET-45/25, 25 mm × 0.45 μm PET membrane syringe filter; Machery-Nagel GmbH & Co. KG, Dueren, Germany) with the first several mL of filtrate discarded and the remainder retained as the WAF. Portions (50 mL) of each WAF were divided among four 100 mL glass beakers, five Daphnid neonates were impartially added to each, and the beakers were covered with watch glasses. Control exposure solutions consisting of DMM without added test substance were stirred, filtered, and divided among replicate exposure vessels with Daphnids added in exactly the same manner as the corresponding WAFs. The Daphnid exposures were incubated at approximately 20 ± 1°C under a 16:8 h light:dark photoperiod with no feeding. Temperature, pH, and dissolved oxygen were recorded in a single replicate for each Control and WAF at the beginning and end of the exposures. Aliquots of each Control and WAF were also collected at the beginning and end of the exposures for determinations of dissolved organic carbon (by the non-purgeable organic carbon [NPOC] method as described in Supplemental Information) and of TDA or MDA.
The criterion used in this study for determination of adverse effects was the alteration of normal mobility behavior of the neonates as observed at 24- and 48-h exposure. The prevalence of these and any other abnormal behavior or appearance of the Daphnids was compared to that in the corresponding Control exposures.
Determination of dissolved toluenediamine and methylenedianiline
The concentrations of 2,4- and 2,6-toluenediamine (TDA) and of 2,4’- and 4,4’-methylenedianiline (MDA) in the Daphnid exposure solutions were determined by HPLC-MS/MS using an ABSciex API 5500 triple quadrupole mass spectrometer interfaced to an Agilent 1290 HPLC system as described and validated previously by Neuland et al. (2021) (also, see Supplemental Information). The LOQ in the Daphnid exposure solutions was 16.8 and 4.0 μg/L, respectively, for 2,4- and 2,6-TDA and 0.2 μg/L for both MDA isomers.
Results
Characteristics of the test substances
Nomenclature and properties of the generic polymeric prepolymer substances investigated. The nomenclature reflects the diisocyanate (MDI or TDI), the polyol type, and the nominal molecular weight of the polymeric prepolymer. Test substances are tabulated by monomer type in increasing order of nominal molecular weight.
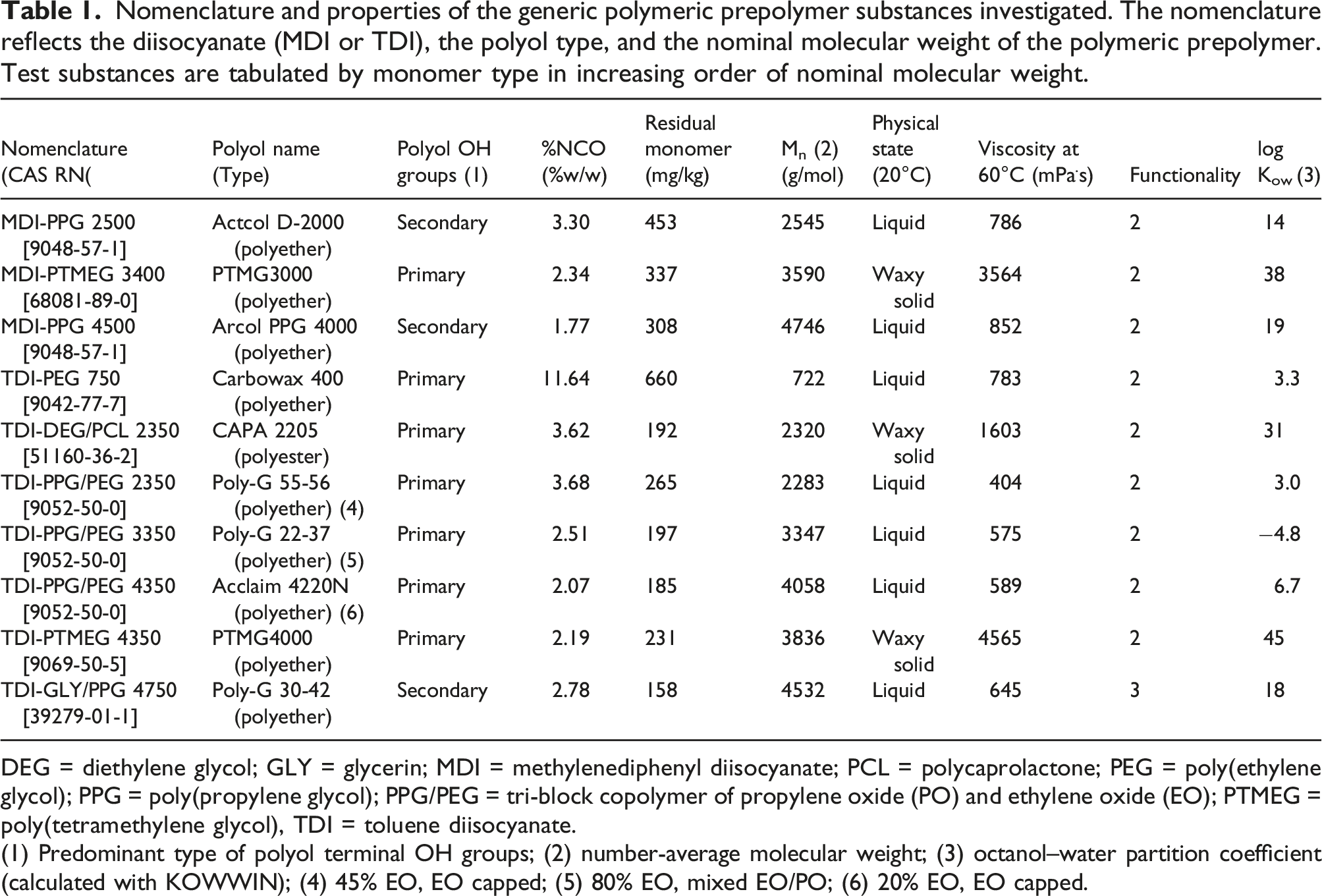
DEG = diethylene glycol; GLY = glycerin; MDI = methylenediphenyl diisocyanate; PCL = polycaprolactone; PEG = poly(ethylene glycol); PPG = poly(propylene glycol); PPG/PEG = tri-block copolymer of propylene oxide (PO) and ethylene oxide (EO); PTMEG =poly(tetramethylene glycol), TDI = toluene diisocyanate.
(1) Predominant type of polyol terminal OH groups; (2) number-average molecular weight; (3) octanol–water partition coefficient (calculated with KOWWIN); (4) 45% EO, EO capped; (5) 80% EO, mixed EO/PO; (6) 20% EO, EO capped.
Aquatic exposure potential
Water extractability test
Summary of individual and mean dissolved organic carbon concentrations measured in triplicate (R1–R3) water extractability tests at 100 mg/L and 1000 mg/L nominal loadings of the test substances after 96 h stirring. All Control mixtures were <LOQ of 2 mg/L. The nomenclature reflects the diisocyanate (MDI or TDI), the polyol type, and the nominal molecular weight of the polymeric prepolymer. Test substances are tabulated in order of decreasing octanol–water partition coefficient (calculated using KOWWIN). Values greater than LOQ are highlighted in bold text.
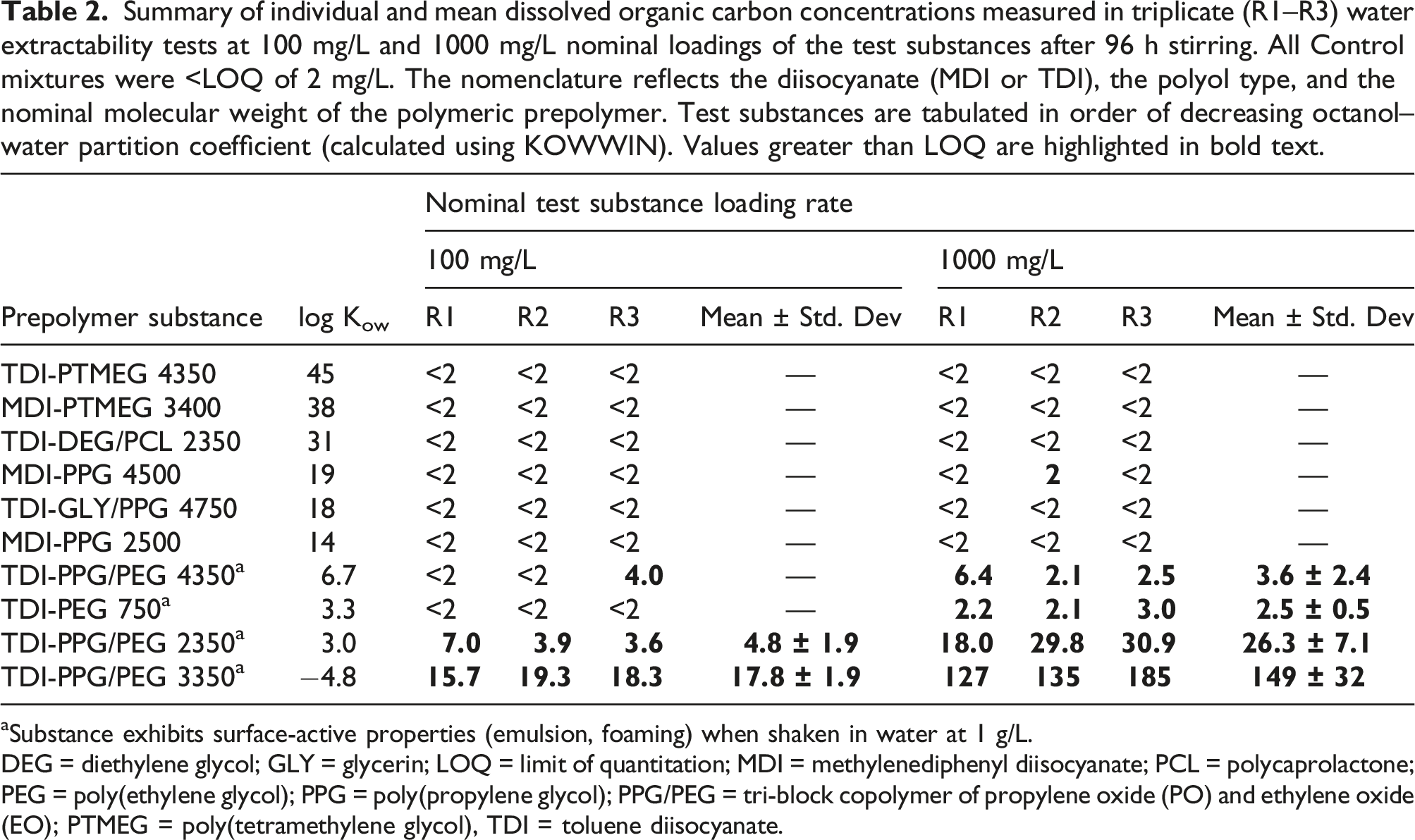
aSubstance exhibits surface-active properties (emulsion, foaming) when shaken in water at 1 g/L.
DEG = diethylene glycol; GLY = glycerin; LOQ = limit of quantitation; MDI = methylenediphenyl diisocyanate; PCL = polycaprolactone; PEG = poly(ethylene glycol); PPG = poly(propylene glycol); PPG/PEG = tri-block copolymer of propylene oxide (PO) and ethylene oxide (EO); PTMEG = poly(tetramethylene glycol), TDI = toluene diisocyanate.
This general picture was confirmed by the more sensitive NPOC measurements (LOQ of 0.5 mg/L) made on the WAF of the acute toxicity tests (Figure 4). For polymeric prepolymers with calculated log Kow <10, the NPOC values resulting from the water extractability testing correlated inversely and with high significance (R2 ≥ 0.96) with the calculated log Kow of the test substances. For test substances with calculated log Kow > 10, the measured NPOC values did not exceed the upper 95%-confidence limit of blank samples (approximately 0.5 mg/L); hence, these substances showed no significant contribution to dissolved organic material[4]. One test for which the blank contained 1.65 mg/L of NPOC was identified as a statistical outlier (grey markers in Figure 4). Relationship between water-extractable fraction (expressed as non-purgeable organic carbon, NPOC) and calculated octanol–water partition coefficient (log Kow). Open markers: loading rate 100 mg/L; filled markers: loading rate 1000 mg/L. Grey markers represent a statistical outlier (blank sample showed NPOC=1.65 mg/L). Horizontal dashed lines (from top to bottom): 11 mg/L (2% of 1000 mg/L prepolymer* loading), 1.1 mg/L (2% of 100 mg/L prepolymer* loading), and upper boundary of 95% confidence interval of blank values (ca. 0.5 mg/L). *: considering minimum 56% of C by weight in the prepolymers.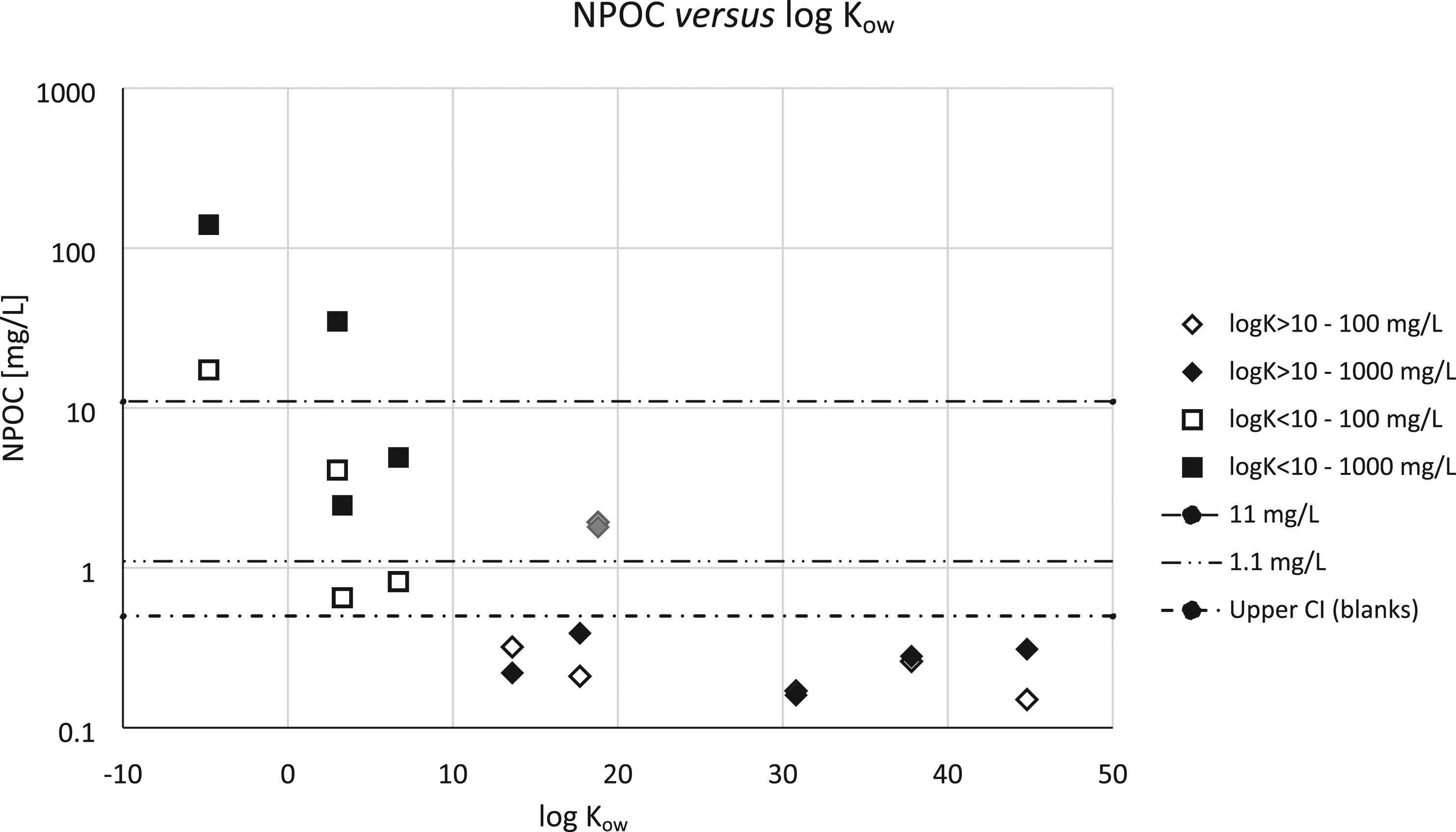
Determination of unreacted NCO groups in water-insoluble residue
The tests with colorimetric SWYPE detection pads were negative for the presence of unreacted isocyanate groups in both THF-soluble and insoluble fractions of the polyurea solids formed after 96 h of stirring for all 10 test substances. Figure 3 provides a typical visual comparison with the coloration obtained with the dilution series prepared with TDI.
Acute toxicity to Daphnia magna
Acute aquatic toxicity test
Aggregate number of D. magna (four replicates with five D. each) immobilized (IMM), showing abnormal swimming behavior (ABN) or showing abnormal coloration (COL) in acute toxicity testing of polymeric prepolymers of TDI and MDI. The nomenclature reflects the diisocyanate (MDI or TDI), the polyol type, and the nominal molecular weight of the polymeric prepolymer. Test substances are tabulated in order of decreasing octanol–water partition coefficient (calculated using KOWWIN).
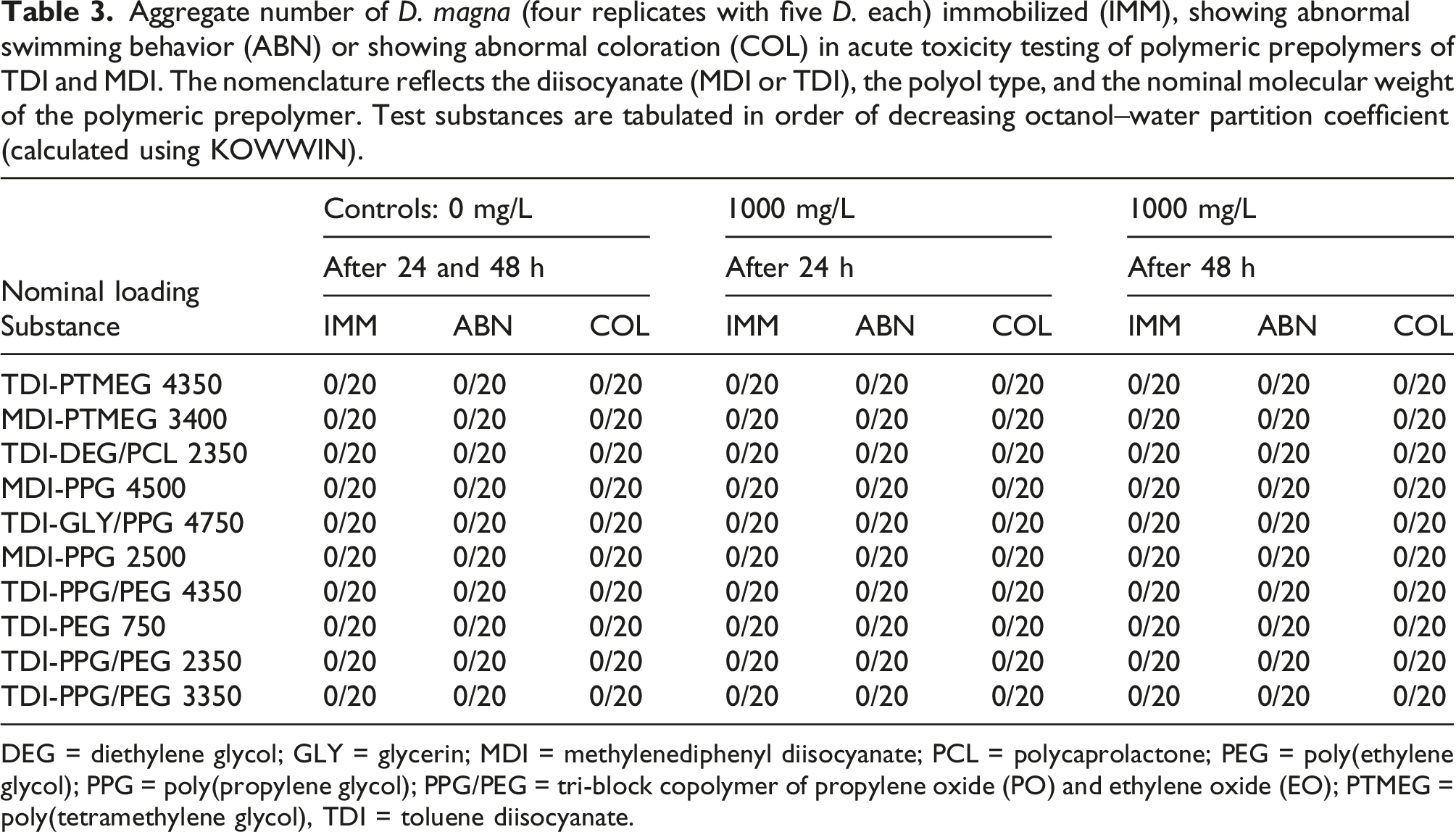
DEG = diethylene glycol; GLY = glycerin; MDI = methylenediphenyl diisocyanate; PCL = polycaprolactone; PEG = poly(ethylene glycol); PPG = poly(propylene glycol); PPG/PEG = tri-block copolymer of propylene oxide (PO) and ethylene oxide (EO); PTMEG =poly(tetramethylene glycol), TDI = toluene diisocyanate.
Determination of toluenediamine or methylenedianiline formed
For the seven TDI-based prepolymer substances, 2,4- and 2,6-TDA were not detected (LOQ = 16.8 and 4 μg/L, respectively) in the Daphnid exposure solutions derived from their 100- and 1000-mg/L loadings to the DMM. Likewise, for the three MDI-based prepolymers no 2,4’- or 4,4’-MDA was detected (LOQ = 0.2 μg/L). In cases where measurable DOC was found in exposure solutions derived from these prepolymer substances, very little or none was attributable to dissolved TDA or MDA.
Discussion
When stirred into water, the major part of the test substances resulted in a non-soluble residue in which NCO groups could no longer be detected (Figure 3). This was an expected observation, since in the organic phase, initially, NCO-terminated ureas are formed, of which the terminal groups hydrolyze to form amino-terminated ureas (Ahn et al., 2013; Herbold et al., 1998; Yakabe et al., 1999). At ambient conditions, the substituted ureas of TDI and MDI are stable under a broad range of pH values (Sendijarevič et al., 2004). The urea-NH groups have pKa values <1 (for R-NH2+ ←→ R-NH + H+) or >13 (for R-NH ←→ R-N- + H+) and, hence, are not ionized under typical environmental exposure conditions (Loddenkemper et al., 2019; Neuhahn et al., 2020). In addition, no other cationic N-species were formed. As amino-groups bound to an aromatic system, the terminal amino-groups have typical pKa values ≤5 (see Spence and Plehiers, 2022), so that little ionization is to be expected at typical test pH values (pH 6.0–9.0).
A second observation was that the TDA or MDA concentrations did not exceed the respective LOQ. Any diamine would typically originate from monomeric TDI or MDI in the test substance. Given that these were present at concentrations <500 mg/kg, the result was not surprising. However, the data also demonstrated that the urethane bond between the aromatic DII and the polyol was stable and did not hydrolyze at an appreciable rate under the investigated conditions. This was in line with the work of Christenson (1964), who determined kinetics for the hydrolysis of urethane model compounds and found the reaction to be very slow at pH values <9.
The constituents of the DOC are likely the aromatic amine-terminated polymer species formed from hydrolysis of the test substances, to the extent those are soluble in water. Contrary to other aromatic amines (Schultz et al., 1991), however, these amine-terminated polymer species did not exhibit acute toxicity to Daphnia magna, the species found to be most sensitive to aromatic diisocyanates and their associated diamine hydrolysis products (Chen et al., 2007). The absence of toxicity was also observed for those substances that become more solubilized because of their high ethoxylate content and/or surfactant nature. As indicated in Table 2, the prepolymer substances that were synthesized from the hydrophilic PEG and PEG–PPG–PEG tri-block polyols exhibited foaming and/or emulsification when their 0.1% mixtures with water were shaken. The representative structures of these four substances have calculated log Kow values of 3.0, 6.7, −4.8, and 3.3, and the calculated log Kow values of their corresponding amine-terminated (hydrolyzed) structures are approximately −1, 2.6, −9, and −0.7, respectively. These substances exhibited the largest extractability in water, yet no apparent toxicity to D. magna. Thus, the hydrolysis products formed from the 10 prepolymer substances tested were either too hydrophobic for aquatic exposure to occur, or too hydrophilic to exhibit toxicity by an assumed non-polar narcosis mode of action.
Summary and conclusion
To conclude, a screening level design-of-experiments approach was used to develop a test program for determining properties of generic monomer-depleted polymeric prepolymers of aromatic diisocyanates relevant for product stewardship and industrial hygiene purposes. In the investigated field of interest, it was found that for polymeric prepolymers with log Kow >10, NPOC was <0.5 mg/L at nominal loadings of 100 and 1000 mg/L (<1 or 0.1% of prepolymer loading, respectively). At the same 100- and 1000-mg/L nominal loading rates, none of the test substances exhibited acute toxicity to the species that was expected to be most sensitive to DII and the primary aromatic amine-terminated hydrolysis products (D. magna). From a product stewardship perspective, the polymeric prepolymers of TDI and MDI within the investigated domain and concentration range do not produce adverse effects to the aquatic environment.
Supplemental Material
Supplemental Material - Exploring structure/property relationships to health and environmental hazards of polymeric polyisocyanate prepolymer substances-1. Design of experiments, aquatic exposure, and acute aquatic toxicity
Supplemental Material for Exploring structure/property relationships to health and environmental hazards of polymeric polyisocyanate prepolymer substances-1. Design of experiments, aquatic exposure, and acute aquatic toxicity by Robert J West, Lisa Euskirchen, Astrid Neuhahn, Steve Seneker, and Mark W Spence in Toxicology and Industrial Health.
Footnotes
Acknowledgements
The authors are grateful to (in alphabetical order) Dr. R. Czeiszperger (Anderson Development Company), Mr. S. Franyutti (BASF Corp.), Mr. S. Grace and Mr. J. Miller (Covestro LLC), Dr. C. Phanopoulos (Huntsman Corp.), Dr. T. Willumstad (Dow), and Dr. T. Yu (Wanhua Chemical Group) for providing insights on the prepolymer chemistry design of experiments, polyol starting materials, and analytical characterizations, and to Dr. M. Ehlers (Currenta GmbH & Co OHG) for coordination and execution of laboratory testing.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S Seneker is retired from Anderson Development Company, a producer of polyurethane systems. RJ West and MW Spence are employed by the International Isocyanate Institute, Inc. The Institute is funded by producers of TDI and MDI. L Euskirchen and A Neuhahn report no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work reported herein was funded by the International Isocyanate Institute, Inc. Any opinions expressed are those of the authors, not necessarily of Anderson Development Company, Currenta, the International Isocyanate Institute, or its member companies.
Supplemental Material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
