Abstract
Methylene diphenyl diisocyanate (MDI) and toluene diisocyanate (TDI) are high production volume chemicals used for the manufacture of polyurethanes. For both substances, the most relevant adverse health effects after overexposure in the workplace are isocyanate-induced asthma, lung function decrement and, to a much lesser extent, skin effects. Over the last two decades many articles have addressed the reactivity of MDI and TDI in biological media and the associated biochemistry, which increased the understanding of their biochemical and physiological behavior. In this review, these new insights with respect to similarities and differences concerning the adsorption, distribution, metabolism, and excretion (ADME) of these two diisocyanates and the implications on their toxicities are summarized. Both TDI and MDI show very similar behavior in reactivity to biological macromolecules, distribution, metabolism, and excretion. Evidence suggests that the isocyanate (NCO) group is scavenged at the portal-of-entry and is not systemically available in unbound reactive form. This explains the lack of other than portal-of-entry toxicity observed in repeated-dose inhalation tests.
Keywords
Introduction
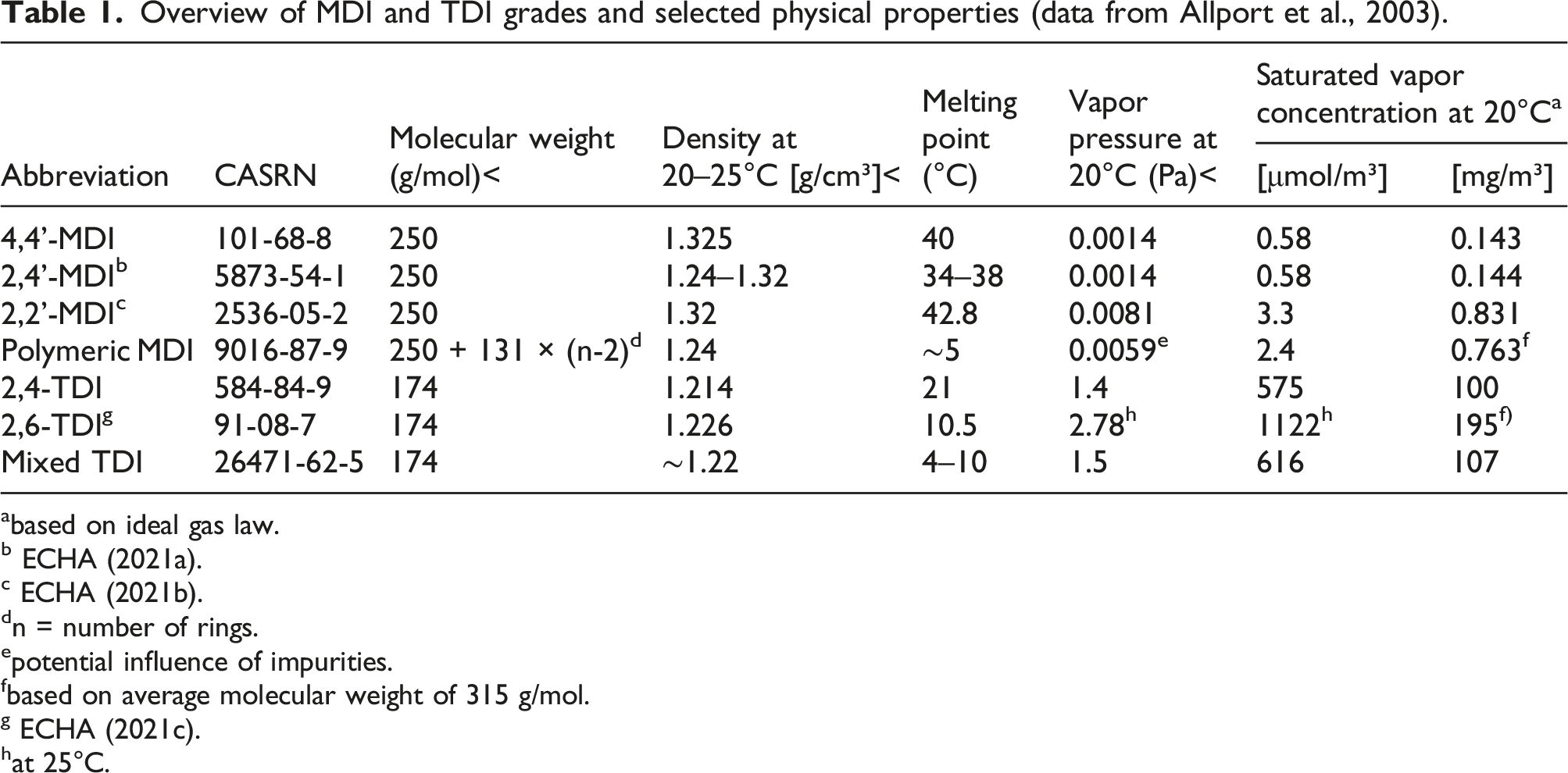
Methylene diphenyl diisocyanate (MDI) and toluene diisocyanate (TDI) are large-volume industrial chemicals used in the polyurethane industry for the production of flexible foam (mattresses, upholstered furniture), rigid foam for insulation (panels, refrigerators), integral skin foam (steering wheels, saddles), coatings, adhesives, sealants, and elastomers. Commercial TDI on the market is either the pure 2,4-TDI isomer (toluene-2,4-diisocyanate, CASRN 584-84-9), or a mixture of 2,4-TDI and 2,6-TDI (toluene-2,6-diisocyanate, CASRN 91-08-7) with a ratio of 80:20 to 65:35 (for the mixture, CASRN 26471-62-5). For monomeric MDI, the main grades on the market are pure 4,4’-MDI (>95% purity, CASRN 101-68-8) and mixtures of monomeric MDI (MMDI, CASRN 26447-40-5), composed of approximately 50% 4,4’-MDI, up to 50% 2,4’-MDI (CASRN 5873-54-1) and a few percent 2,2’-MDI (CASRN 2536-05-2). Since MDI is produced via phosgenation of a poly-addition product, from which monomeric MDI grades are obtained by subsequent distillation, an important part of the market volume is covered by the so-called “polymeric MDI,” which also contains higher molecular weight homologs like 3-ring MDI and 4-ring MDI. This group can be sub-categorized into “oligomeric” MDI (OMDI, CASRN 25686-28-6), which contains more than 50% (w/w) of MMDI, and “polymeric” MDI (PMDI, CASRN 9016-87-9), which contains less than 50% (w/w) of monomer. Chemical structures are shown in Figure 1 and an overview of the different types of MDI and TDI, their CASRN, and relevant physical properties is given in Table 1 (based on data from Allport et al., 2003). 2,4-TDI and 2,6-TDI (top row). MDI isomers (middle row—from left to right: 4,4’-MDI, 2,4’-MDI, 2,2’-MDI) and homologs (bottom row; for instance, 3-ring MDI (n = 1), 4-ring MDI (n = 2) and 5-ring MDI (n = 3)). Overview of MDI and TDI grades and selected physical properties (data from Allport et al., 2003). abased on ideal gas law. b ECHA (2021a). c ECHA (2021b). dn = number of rings. epotential influence of impurities. fbased on average molecular weight of 315 g/mol. g ECHA (2021c). hat 25°C.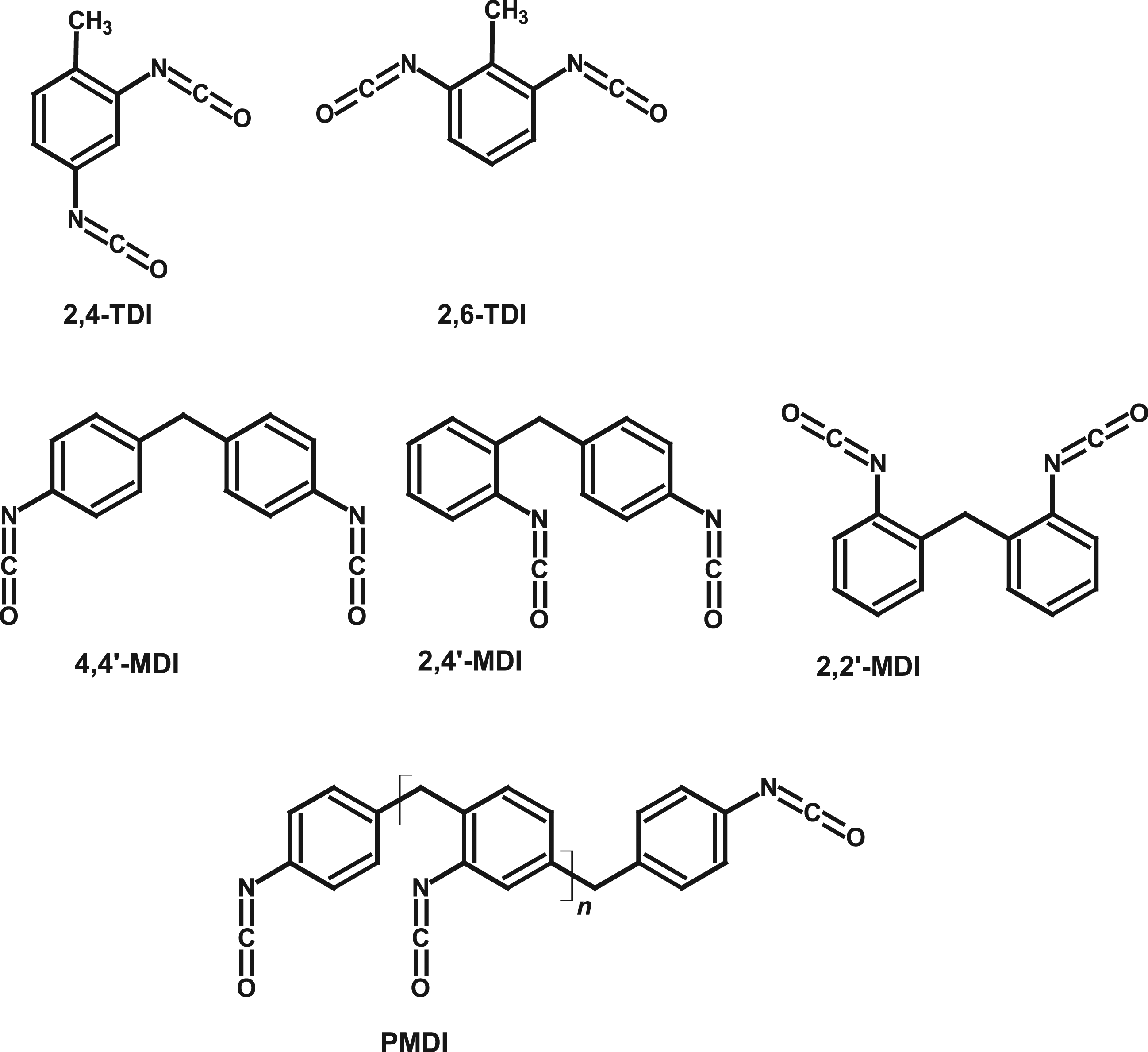
If MDI and TDI are to be compared with each other on the basis of inhalation studies it has to be noted that, due to differences in vapor pressure, animals in MDI studies are exposed primarily to aerosols whereas in TDI studies they are exposed to vapor.
The different grades of MDI and TDI exhibit similar chemical reactivity. Both substances are known to be capable of causing lung decrement and sensitization in overexposed workers as relevant adverse effects (ATSDR, 2018; MAK Commission, 2003, 2008).
Over the last two decades several papers have been published dealing with the biochemistry of TDI and MDI in vitro as well as in vivo. This review summarizes the current knowledge about the chemical reactivity, pre-systemic, and post-absorption biochemistry of MDI and TDI, and evaluates their similarity/dissimilarity.
We searched literature in TOXLINE 1 , using the following search terms:
(isocyanate)[TITLE] AND ((metabolism [Title/Abstract] OR distribution [Title/Abstract] OR absorption [Title/Abstract] OR excretion [Title/Abstract] OR disposition [Title/Abstract] OR biomonitoring [Title/Abstract] OR metabolites)[Title/Abstract]).
Setting the time frame from 1 January 1980 to 1 June 2021, we received 82 hits. These were selected for studies providing insight and understanding on the uptake, distribution, metabolism, and excretion of MDI and TDI, leaving 59 references. Further, we searched the internal library of the International Isocyanate Institute as well as citations included in the selected papers for relevant additional literature.
Chemical structures in the Figures were drawn in ACD/ChemSketch.
Results and discussion
Results are addressed in the following order. First, the reactivity of aromatic (di)isocyanates with various nucleophiles in aqueous medium is discussed. The purpose is to evaluate relative reaction rates, which in the end determine which reaction products are formed and which ones are not. Then, the reactions of MDI and TDI in simulated physiological solutions are reviewed: primary reactions leading to the formation of adducts with biomolecules, and secondary reactions that transform the adducts into more stable forms. In a third part, the findings are used as a basis to put into perspective the results of studies on adsorption, distribution, metabolism, and excretion of MDI and TDI, and to evaluate their biological availability against the results of repeated-dose toxicological studies.
General reactivity of aromatic (di)isocyanates with nucleophiles in aqueous solutions
The isocyanate (NCO) group is capable of addition reactions with functional groups containing active hydrogen, such as OH, NH2, and SH groups. Reactive OH is present in water molecules but also in threonine (Thr), tyrosine (Tyr) and serine (Ser) side chains of proteins. Reactive NH2 is present at the N-terminal amino acids and in lysine (Lys) side chains, and SH is present in the cysteine (Cys) side chains of glutathione (GSH) and proteins. The toxicological effects of the aromatic diisocyanates, TDI and MDI, are a direct consequence of the reactivity of the aromatic NCO group toward biomolecules. For understanding the effects of TDI and MDI in biological tissue, a thorough insight into their reactive chemistry is therefore indispensable.
The general reaction equation between aromatic NCO groups and nucleophilic molecules containing active (transferable) hydrogen atoms (H-X-R) is in essence simple (equation (1)). The relative order of reactivity is S > N >> O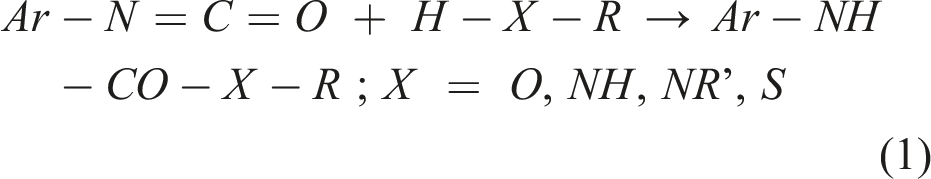
Inhaled isocyanates encounter the mucous membranes of the respiratory tract and may react with molecules in the epithelial lining fluid (ELF). The ELF is an aqueous solution containing 3–8 g/L albumin and 9 g/L total protein (Fröhlich et al., 2016; Hassoun et al., 2018). With a molecular weight of 67 kDa for serum albumin (serving as surrogate for the other proteins as well), this is in the range of 0.044–0.132 mM. With 56 Lys, 30 Cys, 33 Thr and 25 Ser (Isemura and Ikenaka, 1978; Jagodzinski et al., 1981; Peters and Peters, 1972), there would in theory be 2.46–7.39 mM NH2, 1.45–4.36 mM SH, and 2.55–7.66 mM OH groups. However, for the reaction with NCO groups, these X-H groups must be sterically accessible and present in a reactive form; for instance, Cys bound in disulfide bridges no longer provides a reactive SH group. Further, the ELF contains 0.43 m
Reaction with water
Aromatic NCO groups react with water releasing carbon dioxide; the transiently formed primary aromatic amine further reacts with remaining isocyanate groups to form urea bonds. This is illustrated in Figure 2. Schematic hydrolysis of an aromatic isocyanate and subsequent formation of a substituted urea.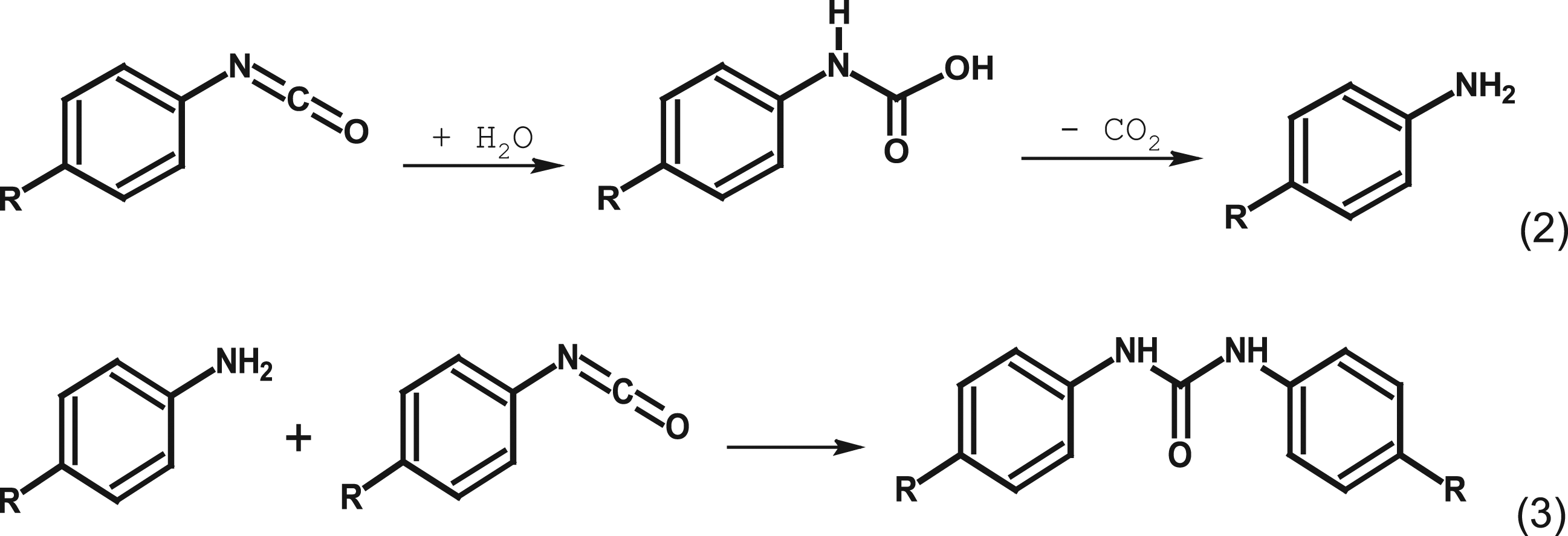
In a buffered and near-neutral medium, the decomposition of the intermediate carbamic acid is quick and does not require further attention in the given context: the pseudo-first order rate constant for the decomposition of the N-phenyl-carbamic acid (derived from aniline) in neutral aqueous buffers was k = 6 s−1 (half-life approximately 0.1 s) (Johnson and Morrison, 1972).
The reaction between NCO and water is rapid but not instantaneous. Kinetics for the model compound phenyl isocyanate (PhI) in aqueous media have been determined by Ekberg and Nilsson (1976) in water-dioxane mixtures, and by Castro et al. (1985) in LiCl solutions with 5% acetonitrile (ACN). The rate constants at 298 K in solutions with over 90% of water were 7.7 × 10−4 and 6.1 × 10−4 M−1 × s−1, respectively. These correspond to a half-life of 17–21 seconds. Recently, Neuland et al. (2021) performed experiments with 120 nM of 4,4’-MDI in water with 1% (v/v) ACN and found a reaction rate constant for the hydrolysis of MDI of 5.7 × 10−4 M−1 × s−1 per NCO group at 298 K. The corresponding half-life of MDI (two NCO groups) is approximately 11 seconds. In agreement with the results of Castro et al. (1985), no catalysis by H+ was observed, but hydrolysis was enhanced by ca. 50% when the pH was increased from 7 to 9.
Under the very dilute conditions investigated (Neuland et al., 2021), the occurrence of bimolecular reactions other than with water was excluded. Under such circumstances, the diamine was the sole ultimate reaction product and the evolution of the transient intermediate amino-isocyanate (one NCO group converted) could be followed in detail (Neuland et al., 2021). At higher concentrations (e.g., achieved by using more solvent) or under heterogeneous conditions (i.e., two-phase, for instance contact of aerosols with aqueous solutions), any amine formed preferentially reacts with the remaining NCO groups. With mono-isocyanate model compounds like PhI, typically investigated at 0.1–1 mM level combined with higher solvent concentrations, the formation of substituted ureas (reaction with the amine) was invariably observed (Castro et al., 1985; Ekberg and Nilsson, 1976; Naegeli et al., 1938a, 1938b). Under heterogeneous conditions and in the absence of nucleophiles more potent than water (see sections on reaction with N- and S-centered nucleophiles), substituted ureas were the main hydrolysis products of TDI and MDI and the corresponding diamines, TDA and MDA, respectively, are formed in minor yield only (<1%) (Ahn et al., 2013; Yakabe et al., 1999).
For the industrial manufacture of polyurethanes, tertiary amines are used as catalysts for the reaction between NCO and OH-bearing molecules including water (Szycher, 2013; Wen et al., 2014). Containing a free electron pair but no transferable H-atom, tertiary amines can activate the NCO and/or OH groups but cannot react themselves. Some aprotic polar solvents have similar structural features and can catalyze the reaction of NCO with water in a similar fashion. Examples are dimethyl formamide (DMF) (Chen et al., 2017), dimethyl acetamide (DMAc) (Pannone and Macosko, 1987), N-methyl pyrrolidone (NMP), and dimethyl sulfoxide (DMSO) (Barnard et al., 1972; Herbold et al., 1998). The result is an increased yield of primary aromatic diamine, which can confound the outcome of in vitro toxicity tests, for instance for genotoxicity screening. This was demonstrated by Herbold and colleagues (Gahlmann et al., 1993; Seel et al., 1998): changing the solvent from the catalytically active DMSO (increased formation of TDA or MDA) to the catalytically inactive ethylene-glycol-dimethyl-ether (no detectable formation of TDA or MDA) changed the outcome from the Ames mutagenicity screening test from positive (presence of diamine) to negative (solely diisocyanate and no diamine detected).
Reaction between isocyanate and O-centered nucleophiles
Reactions with alcohols have been used to investigate the relative reactivity of the various NCO groups in MDI and TDI. When 4,4’-MDI and 2,4-TDI react with ethanol, the second isocyanate group shows a somewhat slower reaction rate than the first isocyanate group (Ferstandig and Scherrer, 1959). The relative rates were 24.71 (first, 4-NCO of 2,4-TDI), 18.82 (first NCO of 4,4’-MDI), 5.88 (second NCO of 4,4’-MDI), and 1 (second, 2-NCO of 2,4-TDI), spanning about one order of magnitude. This is a narrow range compared to the relative reactivities of biological macromolecules versus water. In addition, it should be remembered that the relative reactivities of the biological nucleophiles (see equation (8)) are not expected to change very much with the electrophile (in casu, the type of aromatic NCO group). Based on this, and to the extent that the isocyanate can actually be solubilized, it can be expected that in biological media TDI and MDI form adducts with biomolecules in very similar proportions and to a large extent at similar rates.
Several amino acids (e.g., Ser, Tyr, Thr) and anti-oxidants (e.g., uric and ascorbic acid) contain OH groups that are potentially reactive toward NCO. Typical concentrations in biological tissues do not exceed mM levels. For example, a simulated lung lining fluid contains 1.22 × 10−4 M ascorbate and 1.29 × 10−4 M albumin (Hassoun et al., 2018); with two reactive OH groups in ascorbate, and 33 Thr, 25 Ser and 20 Tyr side chains per albumin (Peters and Peters, 1972), this results in 1.03 × 10−2 M alcoholic/phenolic OH functions in case all OH groups would be accessible. Under physiological conditions, however, these OH groups are not (or hardly) ionized. For example, for Tyr side chains in proteins, the pKa is 10.3 or more (Bashford et al., 1993), meaning that only approximately 0.1% of the Tyr side chains are deprotonated at a pH of 7.4. The overall reactivity of alcohols to NCO is approximately the same as that for water, and the reactivity of organic acids is significantly less than that of water (Sonnenschein, 2014). It can therefore be expected that, in aqueous environments, the rate of reaction with water (50 M) would always by far exceed that with other OH-bearing molecules (mM). This conclusion is supported by the slow alcoholysis observed by Mormann et al. (2006, 2008).
Buffer ions may contain reactive OH groups as well. Examples thereof are (bi)carbonate, phosphate, and borate buffers. Borate buffers have been found to catalyze the hydrolysis of isocyanates, in much the same way as amines do. The rate expression is (equation (2))
2
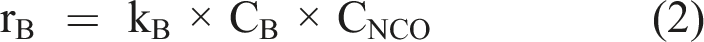
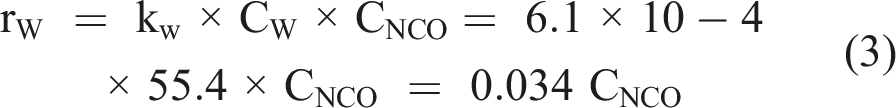
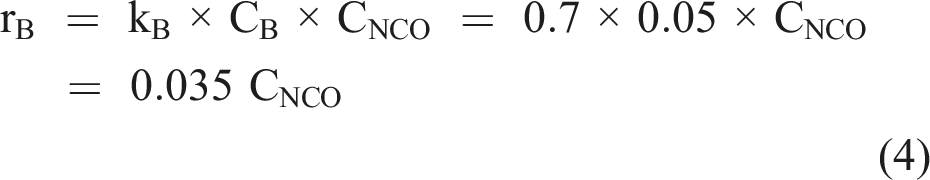
Phosphate does not exert a catalytic influence on isocyanate hydrolysis (Mader, 1968). However, phosphates are not inert and form carbamoyl phosphates (Cramer and Winter, 1959). Based on the rate constants determined by Mader (1968), the rate of reaction with a 50 m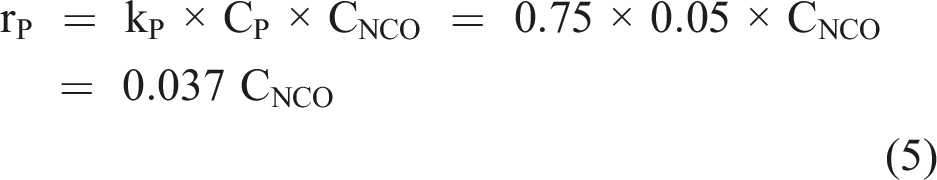
Reaction between NCO and N-centered nucleophiles
In water, the non-protonated amino group is a much more powerful nucleophile than the OH group. On Mayr’s nucleophilicity scale (Mayr, 2021), primary aromatic amines have an N-number around 13. Primary and secondary aliphatic amines have an N-number between 14 and18. The N-number is related to the second-order reaction rate constant k2 as (equation (6))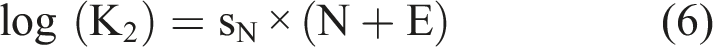
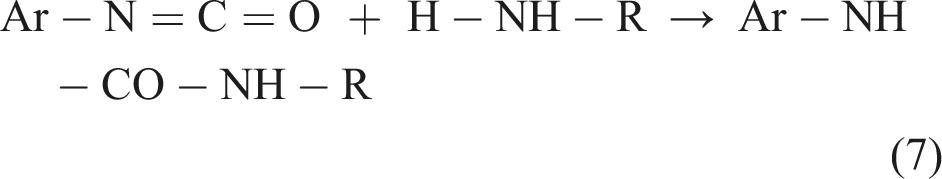
Physical chemical properties and aquatic toxicity of amino-terminated mono-ureas of MDI and TDI.

anominal loading of the respective urea. See also: https://www.diisocyanates.org/sites/dii/files/visual_select_file/properties_of_mdi-_and_tdi-ureas.pdf.
From a mechanistic point of view, the reaction between NCO and amines is assisted by water molecules to effectuate the proton transfer in (equation (7)) (Salvestrini et al., 2002). At 90% water (10% dioxane), Ekberg and Nilsson (1976) found the rate constant for urea formation to be 3.5 × 104 times greater than the one for hydrolysis. For the reaction between aromatic isocyanates and aliphatic primary or secondary amines, no publications were identified that report absolute rate constants. A study on relative reaction rates showed aliphatic amines reacting 10–20 times faster than primary aromatic amines (Davis and Ebersole, 1934).
Reaction between isocyanate and S-centered nucleophiles
In organic media or in non-ionized form, the reactivity of the SH group is less than that of the OH group (Dyer et al., 1961). In buffered aqueous media and depending on pH, however, the SH group can be deprotonated to form a thiolate (S−), which is an extremely potent nucleophile. Mayer and Ofial (2019) have determined the N-number (equation (6)) for the deprotonated form of GSH to be 20.97 on the Mayr scale. This makes GSH the most powerful scavenger of NCO in the body: the N-number indicates a nucleophilic reactivity that is at least 10 orders of magnitude higher than that of water, even without enzymatic assistance. The pKa of the thiol group in GSH lies between 8.5 (Shaked et al., 1980) and 8.75 (Poole, 2015). At the physiological pH of 7.4, about approximately 5% of GSH is present in the thiolate form
3
. Cantin et al. (1985) measured an average concentration of GSH in the ELF of 0.43 mM, and Bicer (2014) reported average levels of 0.32 m
On skin, the pH is approximately 5 (Luebberding et al., 2013). For amino-groups of proteins, a larger proportion will be protonated when compared to ELF. For GSH, a smaller proportion will be present as anion. However, skin has a high glutathione-S-transferase (GST) activity (Oesch et al., 2014). In addition, when TDI was added dropwise into aqueous solutions of N-acetyl cysteine (NAC), the corresponding bis-thiourethane was the main product and the yield of ca. 90% was hardly influenced by the pH of the buffered solution over the range of pH = 5.0 to 7.4 (Mormann et al., 2006).
Comparative view on the reaction rates between isocyanate and nucleophiles
The experimental data concerning the reactivity in aqueous solution of the aromatic isocyanate group in MDI and TDI toward molecules with active hydrogen are limited. However, Mayr parameters have been published for water, amines, alcohols, GSH, and the model substrate PhI. This allows for the calculation of rate constants using (equation (7)) (Mayer and Ofial, 2019; Mayr, 2021).
Estimate of relative reactivities at pH 7.4 and 25°C of the NCO-reactive functional group in PhI (E=−15.38 in equation (7)) with GSH as well as NH2- and OH- containing species present in airway epithelial lining fluid.

aParameters in water except: ethanolate in ethanol, water and ethanol in ACN. sN and N refer to equation (8).
bin lung lining fluid (Fröhlich et al., 2016; Hassoun et al., 2018).
cFraction in active form: FAF = 1/{1+10^(pKa-pH)}.
drelative rate = {(relative rate constant) x (theoretical concentration)/55} x FAF.
eγ-aminobutyric acid.
fper Lys residue.
In the case of inhalation, the concentrations of the NCO-reactive molecules are determined by the composition of the lining fluid of mucous membranes in the respiratory tract. For the low and solubility-limited concentrations of isocyanate, the concentration of the respective nucleophile may be regarded as approximately constant, and the different reaction products will be formed in proportion to the relative reaction rates, as shown in Table 3 for conversion of a single NCO group. For the NH2 group in Lys side chains in albumin, a pKa of 7.9 was assumed for the corresponding acid (Gerig and Reinheimer, 1975), and for the thiol group in GSH a pKa of 8.5 was used (Shaked et al., 1980).
The reaction rate relative to water given in Table 3 may be subject to some uncertainty since nucleophilicities were measured in different solvents and typically in absence of proteins and/or physiological solutions. However, the qualitative order is expected to be correct and is supported by data cited in the next sections.
Based on these considerations, and as long as GSH is not exhausted, inhaled aromatic diisocyanates will predominantly react with GSH, and subsequent reaction with amino-groups (e.g., Lys in proteins) is expected to be by far the most important pathway for isocyanate consumption (see next chapter). Under these circumstances, the textbook reaction (see Figure 2, equation (2)) is completely outpaced and the formation of diamine becomes negligible.
Reactivity of aromatic diisocyanates with biologically relevant nucleophiles
Formation and transformation of thiourethanes of methylene diphenyl diisocyanate and toluene diisocyanate
GSH is a tripeptide present in all mammalian tissues, and upon exposure, the aromatic diisocyanates MDI and TDI may react with this thiol at sites of deposition (e.g., skin, ELF). Based on the above reactivity considerations, it is not surprising that several studies have found that the prime reaction products of both MDI and TDI in aqueous solutions containing GSH are the corresponding thiourethanes (examples given in Figure 3), which are formed rapidly even without enzymatic assistance (Day et al., 1997; Wisnewski et al., 2011a, 2015, 2016a, 2019a). GSH adducts of 2,4-TDI and 4,4’-MDI reported in literature (Day et al., 1997; Wisnewski et al., 2011a, 2013, 2015, 2016a, 2019a).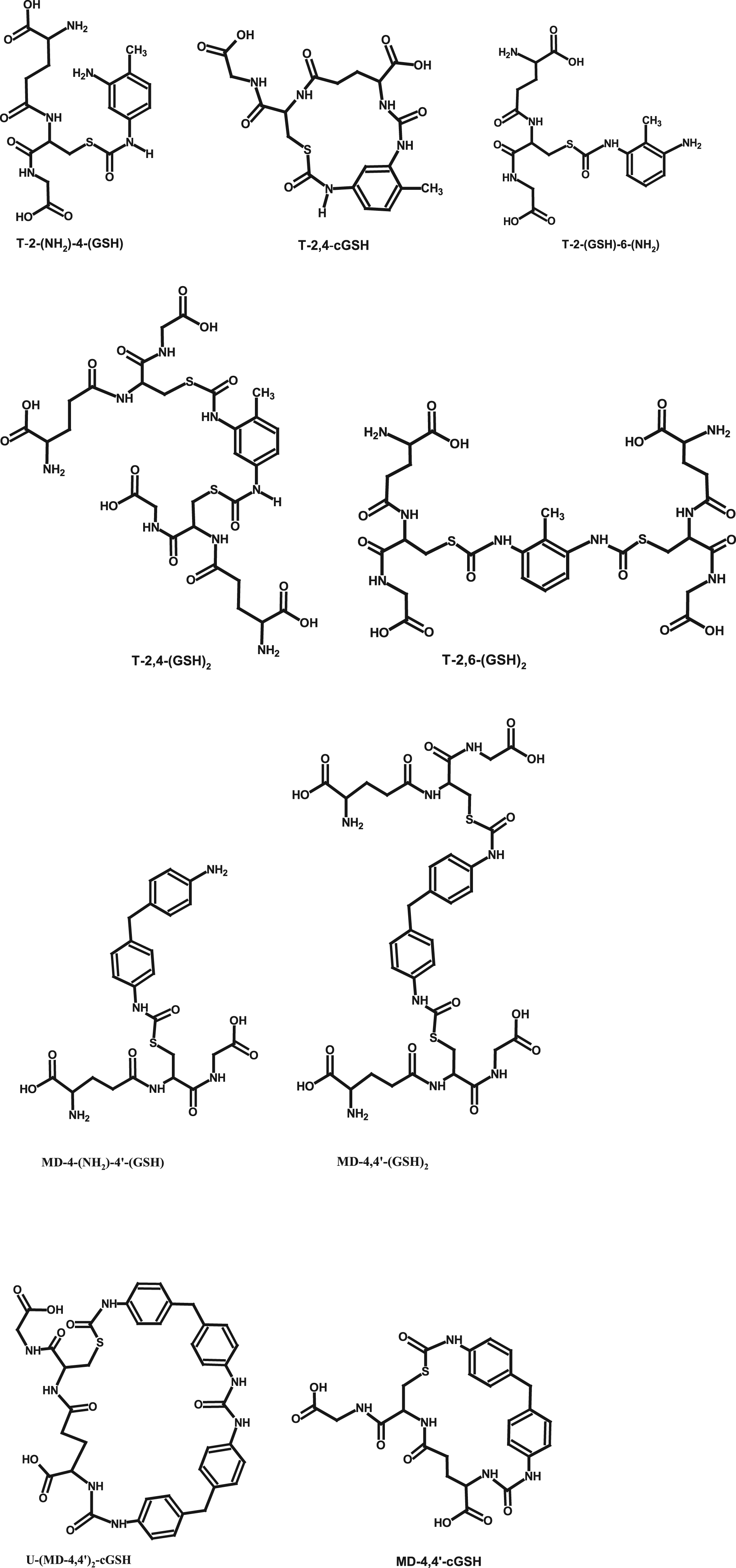
From the results of Day et al. (1997) and Wisnewski et al. (2019a), it follows that the reaction between GSH and TDI or MDI proceeds at the aqueous-organic interface. As long as GSH is available in excess, the main reaction product is the bis-GSH-adduct of the diisocyanate, and the hydrolytic polymerization of the GSH adducts is kinetically retarded, although not fully suppressed; this can be deduced from the experimental details given for the respective syntheses (Reisser et al., 2002; Wisnewski et al., 2011a, 2013, 2019a). Some amino-terminated mono-GSH-adduct is typically formed as well. Surrogates for GSH have been used as well to demonstrate the facile reaction between isocyanates and thiol-bearing molecules with similar yield patterns: cysteine methyl-ester (Chipinda et al., 2006), and N-acetyl cysteine (NAC) (Fleischel et al., 2009; Mormann et al., 2008). That yield pattern is consistent with the in vitro experiments performed by Wisnewski et al. (2019a) with GSH and MDI at physiological pH and temperature: for all three monomeric MDI isomers, the MDI-bis-GSH adducts were the main initial products, and MDA could not be detected at a 30 nM level, which corresponds to an MDA yield of less than 0.001%. In the course of the 2-h incubation period, a gradual re-arrangement of the S-based adducts toward N-based (incl. cyclic) adducts occurred (Wisnewski et al., 2019a). This transcarbamoylation process will be discussed under “Reactions of isocyanates and their thiourethanes with proteins.”
Thiourethanes (S-adducts) are stable in acidic environments, but slowly hydrolyze under neutral or mildly alkaline conditions, in particular in the absence of a sufficient level of thiol (Day et al., 1997; Mormann et al., 2008; Reisser et al., 2002). For the decay of the T-2,4-(GSH)2 in aqueous media 4 at pH 7.4 and 298 K, Day et al. (1997) determined a pseudo first-order rate constant for hydrolysis of 4.7 × 10−5 s−1 (corresponding half-life approx. 4 h). For the MDI-bis-GSH adduct, Reisser et al. (2002) determined a pseudo first-order rate constant for hydrolysis at pH 7.4 and 298 K of 2 × 10−4 s−1 (corresponding half-life approx. 1 h). The hydrolysis rate of the mono-GSH adduct formed was about one order of magnitude smaller. For comparison, Mormann et al. (2008) found a value of 2.8 × 10−4 s−1 for the hydrolysis of the MDI-bis-NAC adduct. The rate of hydrolysis of the MDI-adducts increased with increasing pH. The relative stability of the GSH-adducts leaves the opportunity for interaction with other biomolecules. For instance, the primary amino-group released by hydrolysis of the GSH adducts reacted with other thiourethane molecules and resulted in precipitates that were identified as oligo-ureas (Mormann et al., 2008; Reisser et al., 2002). Chipinda et al. (2006) determined a hydrolysis half-life of the TDI and MDI adducts with cysteine methyl-ester in the order of minutes. However, the use of 5,5’-dithio-bis(2-nitrobenzoic acid) (DTNB) as a scavenger for the released cysteine methyl-ester changed the reaction from an equilibrium to an irreversible one. Further, in a control-experiment, Chipinda et al. (2006) used a 100-fold excess of dimethylamine at pH 9 (at which dimethylamine is 98% protonated), and therewith had concurrently investigated the alkali- and borate-catalyzed hydrolysis as well as the aminolysis of the adducts. The two effects cannot be separated and, in view of the other results, are rather indicative of the already mentioned rapid reaction of the adducts with amines.
Reactions of methylene diphenyl diisocyanate and toluene diisocyanate and their thiourethanes with proteins
Overview of studies investigating the direct reaction of diisocyanates with amino acids or peptides and the transcarbamoylation reactions of S-based diisocyanate adducts.
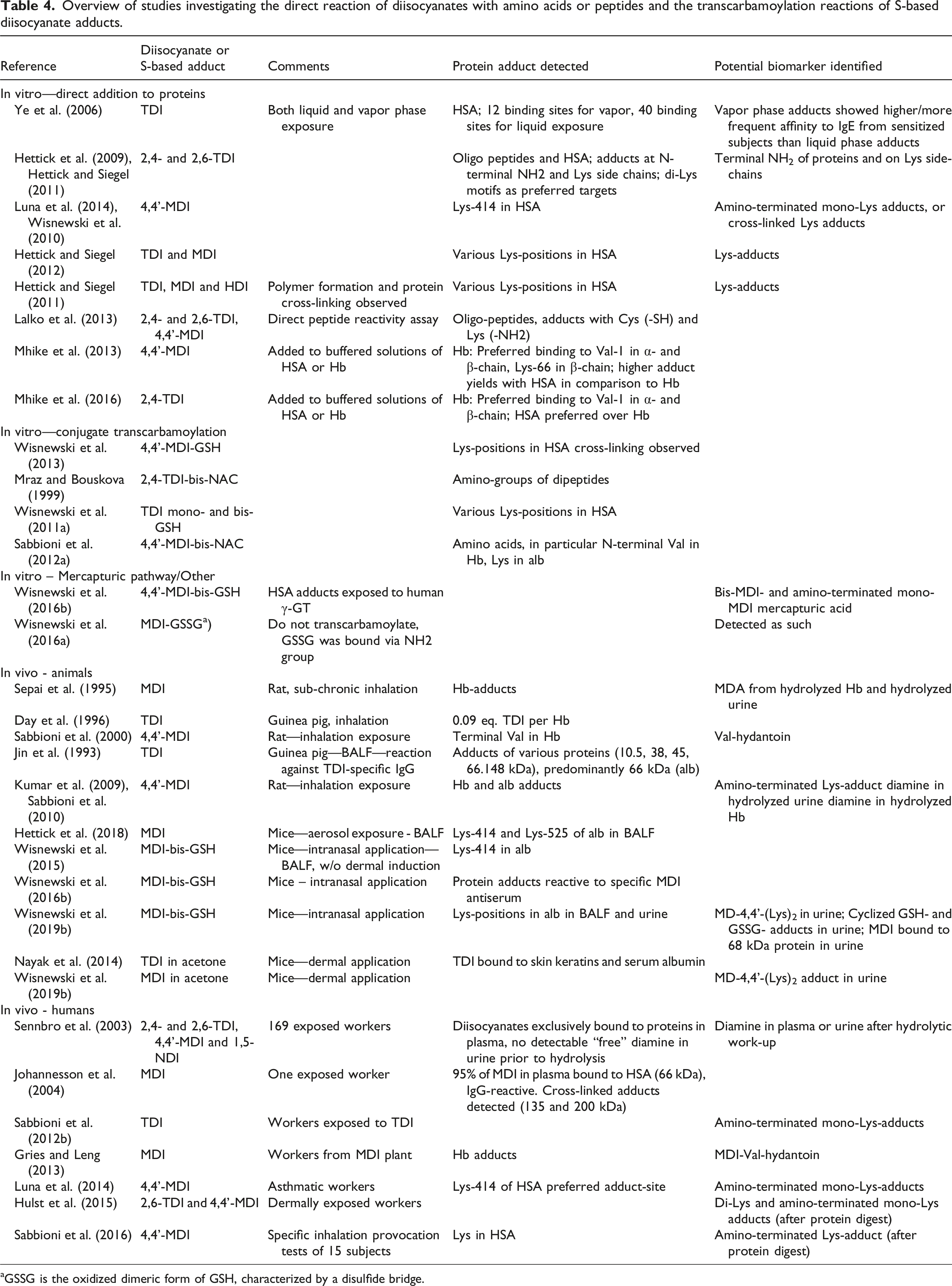
aGSSG is the oxidized dimeric form of GSH, characterized by a disulfide bridge.
The thiourethanes also readily transfer the TDI or MDI moiety onto amino groups of peptides or proteins by transcarbamoylation, forming urea bonds with the N-atom (Day et al., 1997; Mormann et al., 2008; Wisnewski et al., 2011a). As shown before, the reaction with amines is favored compared to the one with water (Table 3). Transcarbamoylation reactions have been investigated with a variety of thiourethanes and amino group bearing substances as reaction partners (Table 4). Transcarbamoylation is expected to occur when the GSH-adducts are transferred from a GSH-rich (ELF—161–430 μM) to a GSH-lean (plasma—1–3 μM) 5 environment (Bicer, 2014; Cantin et al., 1985), but has been shown to also take place at the portal-of-entry (Wisnewski et al., 2000). In aqueous solutions in vitro, the highest rate of transcarbamoylation was observed with other sulfhydryl groups, followed by amino groups, followed by hydrolysis (135 : 10: 1), whereby hydrolysis resulted in precipitation of oligomeric ureas (Mormann et al., 2008). The relative rates are consistent with those derived in Table 3.
Albumin adducts have been identified after TDI inhalation exposure in bronchoalveolar lavage fluid (BALF) of guinea pigs (Jin et al., 1993) and in BALF of mice exposed to MDI aerosol or following intranasal dosing with MD-4,4’-(GSH)2 (Hettick et al., 2018; Wisnewski et al., 2015; Wisnewski and Liu, 2016). After dermal exposure to MDI in dry acetone, or intranasal exposure to MD-4,4’-(GSH)2 for up to 5 days, 24 h urine samples of mice were collected and analyzed for MD-4,4’-(Lys)2 (Wisnewski et al., 2019b). In BALF of the intranasally exposed mice, MDI was bound to Lys, and levels increased over repeated exposures. Both exposure routes resulted in dose-dependent, detectable amounts of MD-4,4’-(Lys)2 in urine, declining after cessation of exposure. The urine of exposed mice also contained MDI bound to albumin. Cyclized adducts of MDI with GSH and GSSG found in the urine may indicate a kind of a “detoxification” reaction for MDI. TDI and MDI were shown to form adducts to dermal proteins in human skin callus; after protein isolation and tryptic digest, mono- and di-Lys adducts of MDI and TDI were identified (Figure 4) (Hulst et al., 2015). Dermal application of TDI in acetone to mice resulted in adducts to keratins and albumin, detectable in the stratum corneum, hair follicles, sebaceous glands, and draining lymph nodes (Nayak et al., 2014). Amino acid adduct derived biomarkers of MDI and TDI (Gries and Leng, 2013; Hettick and Siegel, 2012; Kumar et al., 2009; Luna et al., 2014; Sabbioni et al., 2000, 2001, 2010, 2012a, 2016; Wisnewski et al., 2019b).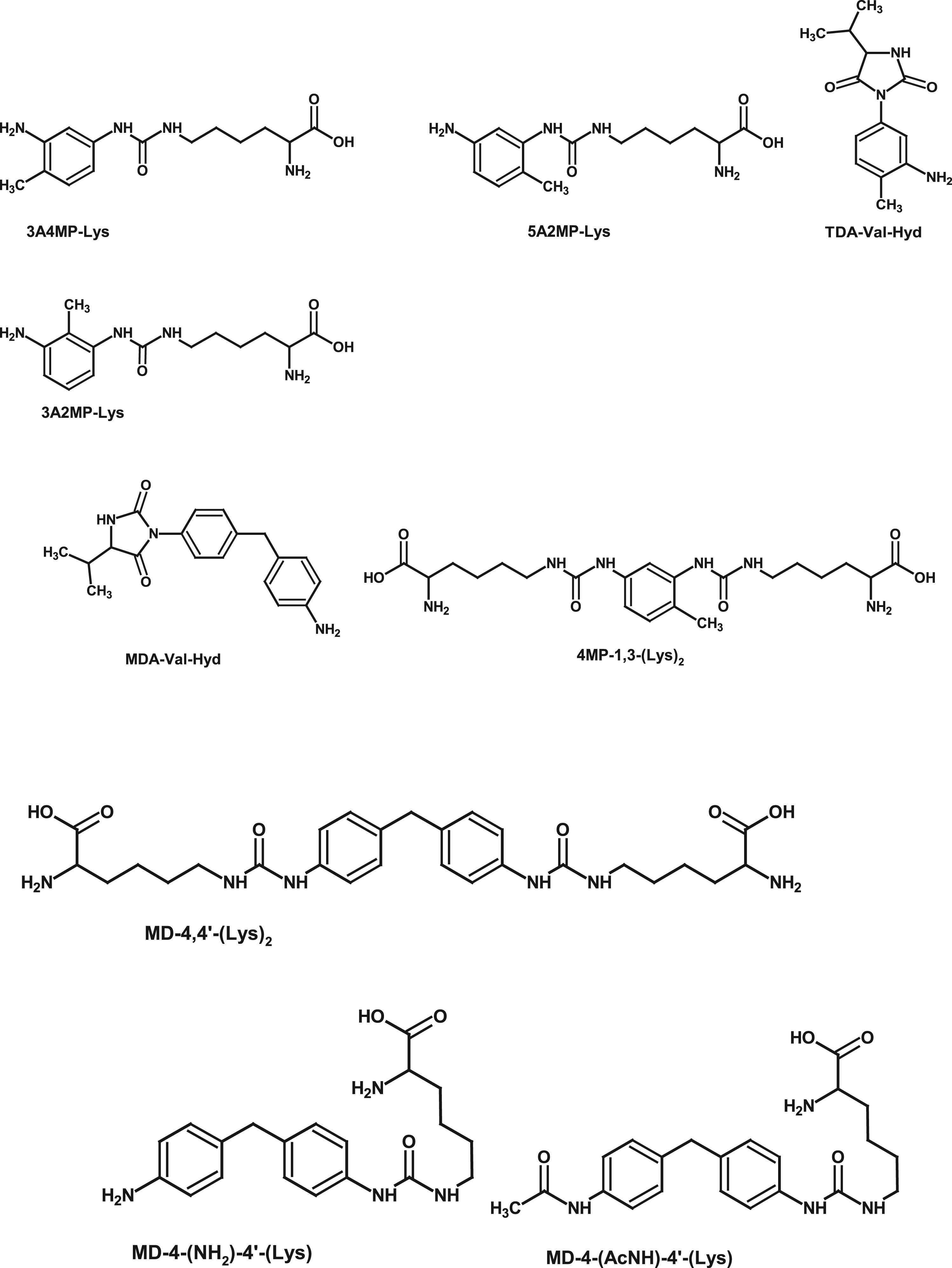
In vivo in humans, the transfer of the isocyanate moiety to plasma proteins could be demonstrated as well. In plasma, isocyanates were exclusively bound to proteins. No diamine was detected in any unhydrolyzed urine or blood sample (Lindberg et al., 2011; Sennbro et al., 2003). Johannesson et al. (2004) found at least 96% of the NCO to be bound to plasma proteins, 95% of which was bound to HSA.
Lys adducts of TDI were analyzed in blood from 10 accidentally exposed workers 26 days after exposure (Sabbioni et al., 2012b). Albumin was isolated, subjected to enzymatic digest and analyzed by HPLC-MS. This work shows that albumin adducts are a kind of “historic” memory for exposure. The half-lives were 19.6 days for 3A2MP-Lys, 21.7 days for 3A4MP-Lys, and 40.3 days for 5A2MP-Lys (for structures of these compounds, see Figure 4). These values coincide with the typical half-life for HSA, which is 20–25 days (Levitt and Levitt, 2016). H2N-4,4’-Lys could be quantified after enzymatic digestion of HSA from 8 out of 15 asthmatic workers (Luna et al., 2014). Based on these investigations, Lys-414 seems to be the primary site for MDI adduct formation on HSA.
As was shown for hexamethylene-1,6-diisocyanate (HDI), keratins, which are rich in Cys, are the preferred binding sites in biopsy samples from human lung and skin, whereas in BALF, HDI is primarily bound to serum albumin (Wisnewski et al., 2000). A similar behavior could be demonstrated for MDI (Wisnewski et al., 2011b) and TDI (Nayak et al., 2014). Once bound to albumin in the respiratory tract, a passive transport into the blood is to be expected (Folkesson et al., 1992), and this transport is increased in cases of lung injury (Folkesson et al., 1998). This demonstrates that the transcarbamoylation process already takes place in the airway lining fluid and does not necessitate prior transfer of the GSH-adducts into the blood stream.
The N-terminal Val in Hb is another binding site for MDI and TDI in rats and humans (Sabbioni et al., 2000, 2001). In a sub-chronic inhalation study with female rats, Hb-adduct levels and MDA in hydrolyzed urine were dose-dependent but did not increase between 3- and 12-month exposure to 0, 0.26, 0.7, or 2.06 mg/m³ 4,4’-MDI for 17 h/day and 5 days/week (Sepai et al., 1995).
On the basis of Hb adducts and MDA in hydrolyzed urine, dermal exposure cannot be discriminated from inhalation exposure, as was demonstrated with rats (Pauluhn and Lewalter, 2002); however, the Ac-MDA to MDA ratio was higher in Hb adducts after skin contact compared to inhalation. MDA in hydrolyzed urine achieved approximately two orders of magnitude higher levels than (Ac-)MDA from Hb adducts. From the Hb adducts, MDI-based Val-hydantoin could be released by acidic work-up and identified as an isocyanate specific biomarker in rats (Sabbioni et al., 2000) and exposed workers (Gries and Leng, 2013; Sabbioni et al., 2016).
The studies listed in Table 4 consistently show preferential transfer of the isocyanate to either the N-terminal Val residue in Hb or to various Lys residues in HSA. Several of these adducts may serve as a basis for biomarkers in TDI or MDI exposure studies (Hettick et al., 2018; Johannesson et al., 2004; Kumar et al., 2009; Luna et al., 2014; Mhike et al., 2013; Pauluhn et al., 2006; Sabbioni et al., 2000, 2001, 2010, 2012b, 2016; Wisnewski et al., 2011b, 2019b).
In in vitro studies, the amino group of Lys side chains in serum albumin presents the dominant reaction partner for NCO groups. This is consistent with the greater reactivity of Lys residues in albumin compared to the mostly protonated Val in Hb (Table 3). With respect to Hb adducts, when MDI dissolved in acetone was added to either HSA or Hb in buffered aqueous solutions, HSA bound 18 times more MDI than Hb at a MDI: protein molar ratio of 1:1. At a ratio of 40:1, HSA bound only 2.1 times more MDI than Hb (Mhike et al., 2013). A similar study with TDI revealed the same qualitative reaction pattern (Mhike et al., 2016): in Hb, Val-1 was first to react with TDI, followed by Lys-66 at increasing concentrations. The ratio of HSA-TDI to Hb-TDI, however, remained in the range of 2–4, showing less dependence on the NCO to protein ratio compared to MDI. As a result, a sort of “titration effect” can be noted: with increasing NCO to protein ratio, more and more side chains of Lys in HSA or Hb were occupied. Increasing MDI:albumin ratios resulted in increasing cross-linking of albumin (Hettick and Siegel, 2011; Mhike et al., 2013). A link between total HSA load and diisocyanate asthma is discussed by Sabbioni et al. (2017). Supplemental Table 1 (Supplemental Information–Section 2) provides an overview of the NCO binding locations in HSA. In the absence of solvents, when TDI interacted via the gas-phase, or when MDI was supplied as MD-4,4’-(GSH)2, there were 8 binding locations for TDI and 7 or 10 for MDI. At low NCO:HSA ratio, Lys-199, Lys-414, and Lys-525 were the most common binding sites for both diisocyanates.
The conjugation of MDI and TDI to proteins as a basis for antibody production and sensitization is discussed in several papers, and an increased reactivity of these adducts against IgG and IgE from asthmatic workers could be demonstrated (see e.g., Johannesson et al., 2004; Lummus et al., 2011; Luna et al., 2014; Wisnewski et al., 1999, 2010, 2013; Ye et al., 2006): the adduction level of HSA in asthmatic workers was much higher than in other, symptom-free workers with exposure to MDI (Sabbioni et al., 2017).
There has been significant debate in the literature about the mechanism of the transcarbamoylation reaction and the associated question of whether or not it requires the presence (“release”) of the NCO group as a free reactive intermediate. A summary is given in Supplemental Information–Section 1. That evidence suggests that the NCO is not freely available during the transcarbamoylation reaction. As mentioned before, once attached to a protein via a urea bond, the NCO group can be considered permanently bound for toxicological considerations.
This completes the detailed survey of the reactivity of aromatic (di)isocyanates with various nucleophiles in aqueous medium. The main concepts from this chapter that the reader should keep in mind as we proceed to the discussion of the implications for diisocyanate toxicology are: • MDI and TDI react rapidly and preferentially with thiols like GSH in aqueous solutions that contain biologically relevant nucleophiles. • The resulting thiourethanes readily react with primary amino groups in proteins. • Hydrolysis of larger loads of thiourethanes in the airways can result in the formation of insoluble oligo-ureas when the binding capacities of proteins in ELF are exhausted. • In serum, TDI or MDI seem to be exclusively bound to proteins: mainly albumin, and to a lesser extent, hemoglobin. In skin and BALF, TDI and MDI seem to be preferentially bound to keratins and albumin. Lys side chains are the main reaction partners. After digestion of the proteins, mono- and bis-Lys adducts of TDI and MDI are detected as biomarkers of exposure. • Increasing levels of isocyanate can titrate the Lys residues in HSA and Hb. HSA has nearly identical binding capacities for MDI and TDI, and at low NCO:HSA ratios, both isocyanates show affinity for the same binding sites. • Once reacted with amino groups of HSA, the NCO group remains bound as a stable substituted urea and is ultimately excreted in urine, either as a HSA adduct or as Lys-adducts. • The elimination rate of the isocyanate-albumin adducts matches the physiological turnover rate of serum albumin.
Effects on toxicokinetics and metabolism
The results of the identified studies addressing the toxicokinetics, distribution, and excretion of TDI and MDI after oral, dermal, and inhalation exposure are reviewed here. The findings are consistent with, and can be interpreted and explained based on, the chemical reactivity of TDI and MDI as described in the previous sections.
Oral route
Excretion and tissue distribution (% of administered dose) of radioactivity after oral gavage of 14 C-TDI in male rats.
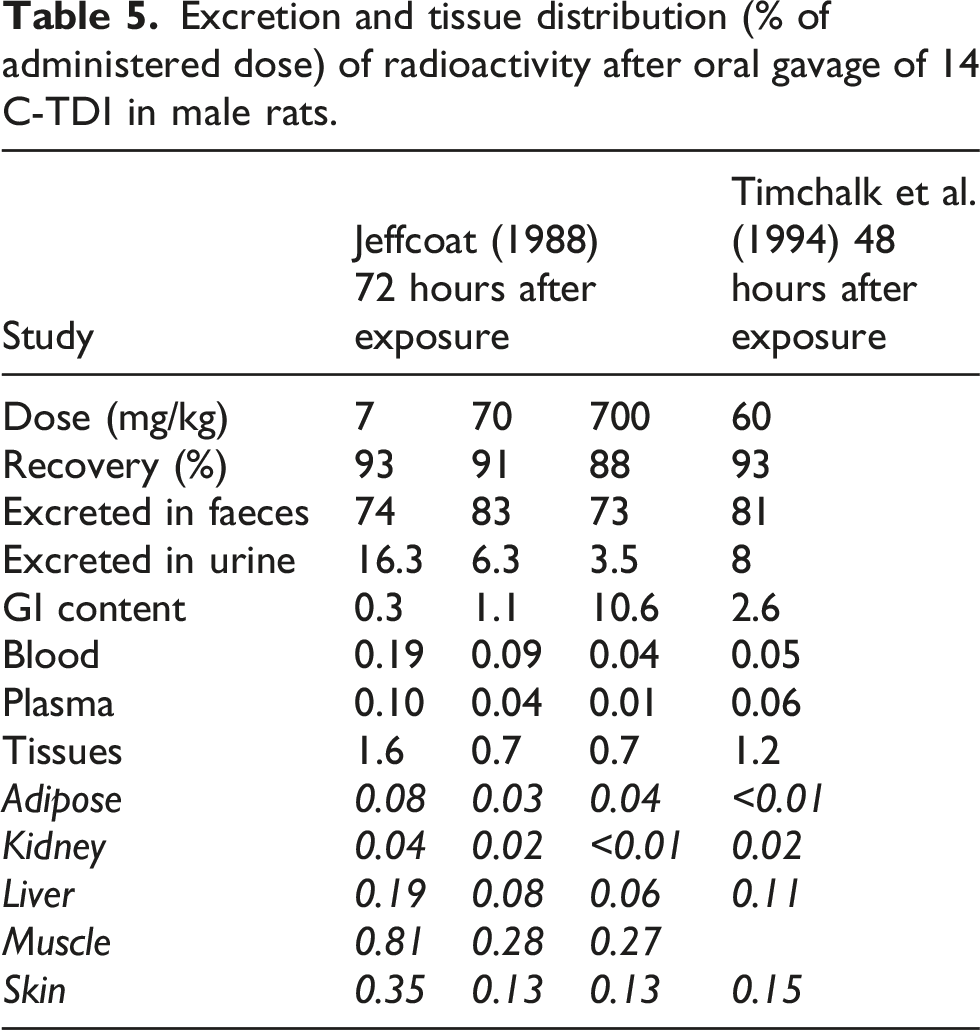
For MDI, no ADME study after oral dosing could be identified. In one study, however, grooming after dermal exposure obviously resulted in oral uptake. Radiolabeled MDI in dry acetone was applied on the shaved back of two female rats. After 24 h, radioactivity was mainly found in the epidermis, and some minor amounts on the tongue, in the blood, lung, liver and kidneys. In a second experiment, collecting samples over 48 h, the first results were confirmed. Further, approximately 1% of the radioactivity was excreted in urine and 20.3% in faeces. The comparatively large amount of radioactivity found in faeces was attributed to grooming behavior of the rats (Vock and Lutz, 1997).
Dermal route
ADME studies are available for both TDI and MDI (Bello et al., 2006; Hoffmann et al., 2010; Hulst et al., 2015) in rodents. One dermal absorption study with MDI on human volunteers was identified (Hamada et al., 2018). They have in common that penetration into the skin is limited and that systemic absorption through the skin is less than 1% of the administered dose.
After application of 320 mg/kg radiolabeled 2,4-TDI on the shaved skin of male rats (10 mg/cm2) for up to 8 h (Hoffmann et al., 2010), only 0.9% of the dose was systemically absorbed and 0.33% excreted in urine. Most of the material was recovered in the protective covering, with lesser quantities being recovered by skin-wash or remaining on the skin at and around the application site. Skin irritation was observed as a local effect (Hoffmann et al., 2010). In humans, the development of skin symptoms due to workplace exposure is rather rare (Allport et al., 2003: 163–165).
Bello et al. (2006) investigated the in vitro absorption of various isocyanates in previously frozen guinea pig skin via FT-IR spectroscopy. For MDI at an area dose of 0.21 μmol NCO per cm2, the NCO-absorption band decreased to approximately 40% after 5 minutes, 28% after 10 minutes, and 15% after 30 minutes. For a 10 times greater area dose, the decrease was much slower, and after 1 h, 43% of the initial intensity was observable. Even though the IR beam ranged only approximately 1 μm into the skin of 1 mm thickness, the authors concluded that there was most likely a saturable transport process rather than a chemical reaction of the NCO groups. That conclusion was based on the assumption that, as the NCO vibration at ca. 2270 cm−1 disappeared by reaction, the absorption of the aromatic ring vibration of MDI at 1523 cm−1 should have been stable instead of disappearing at the same rate. However, the reaction of the NCO group does not leave the ring-vibration unaffected (Badri et al., 2010). For example, as shown in Supplemental Figures 3 and 4 (Supplemental Information–Section 3), the ring vibration changes by the reaction of the NCO groups: in solid 4,4’-MDI, the aromatic ring vibration is at 1517 cm−1, whereas in the mono-urea, the ring vibration is shifted to 1591 cm−1. Therefore, skin absorption cannot conclusively be discriminated from reaction, but formation of the urea or of urea bonds with proteins would be consistent with the expected reaction pattern and other in vitro observations (Hulst et al., 2015).
In an in vivo study with rats, 15 mg/kg or 165 mg/kg radiolabeled 4,4’-MDI was applied to rat skin for 8 h under occlusive conditions. After 8 h exposure, the cover was removed and the skin was washed. Analysis was performed at the end of study (8 h), after 24 h and after 120 h (Hoffmann et al., 2010). The area dosages were 3.2 and 32 μmol NCO per cm2. Between 0.55 and 7.14% of radioactivity penetrated into the skin at the high dose, and 0.88% of radioactivity was systemically absorbed over the 120-h period (excreta, gut content). At the low dose, penetration into the skin was between 1.37 and 3.17%, and 0.69% was systemically absorbed over the 120 h period. After dermal administration, radioactivity in organs was below the detection limit of 1 μg/g tissue. Reaction with skin proteins was brought forward as an explanation for the low absorption. This would be consistent with the key initiating event for dermal sensitization (Kimber et al., 2018) and is also consistent with the results of Hulst et al. (2015), who showed that MDI reacts with skin proteins in vitro and that transport of these adducts via the lymphatic system is the likely mechanism for the systemic uptake through the skin. Via the lymphatic system, the adducts may finally end up in the liver and whence in biliary and/or urinary excretion and detection (Henriks-Eckerman et al., 2015; Yeh et al., 2008). When MDI dissolved in corn oil was injected intradermally, 26% of the radioactivity was absorbed and excreted mainly via faeces (Hoffmann et al., 2010). Other than in faeces, urine, and content of the GI tract, radioactivity was recovered in decreasing quantities in liver (0.32%), blood and plasma (0.23%), stomach and gut (0.11%), kidneys (0.07%), and other tissues (totaling 0.11%) (Hoffmann et al., 2010).
When MDI dissolved in dry acetone was dosed to the skin of mice, the di-lysine adduct of MDI (MD-4,4’-(Lys)2, see Figure 4) was detectable in urine of the mice within 24 h after dosing (Wisnewski et al., 2019b).
The low dermal penetration of 4,4’-MDI observed in animal studies is consistent with the results of a study in human volunteers. Dermal dosages between 10 and 50 mg per person, but at a constant area dose of 0.8 mg/cm2 MDI in petrolatum, were applied for 8 h, and plasma and urine was sampled for the following 48 h (Hamada et al., 2018). MDA concentrations in hydrolyzed plasma were between 0.1 and 0.2 ng/mL at 10 h after dosing and were nearly constant over the observation period. Between 0.01 and 0.20% of the applied dose was systemically absorbed and found in hydrolyzed plasma and urine. The authors concluded that the formation of MDA through hydrolysis must have been very limited, since otherwise MDA would have been readily absorbed. The reaction of NCO with skin proteins and absorption of the mobile adducts was named as the most likely absorption pathway.
Inhalation route
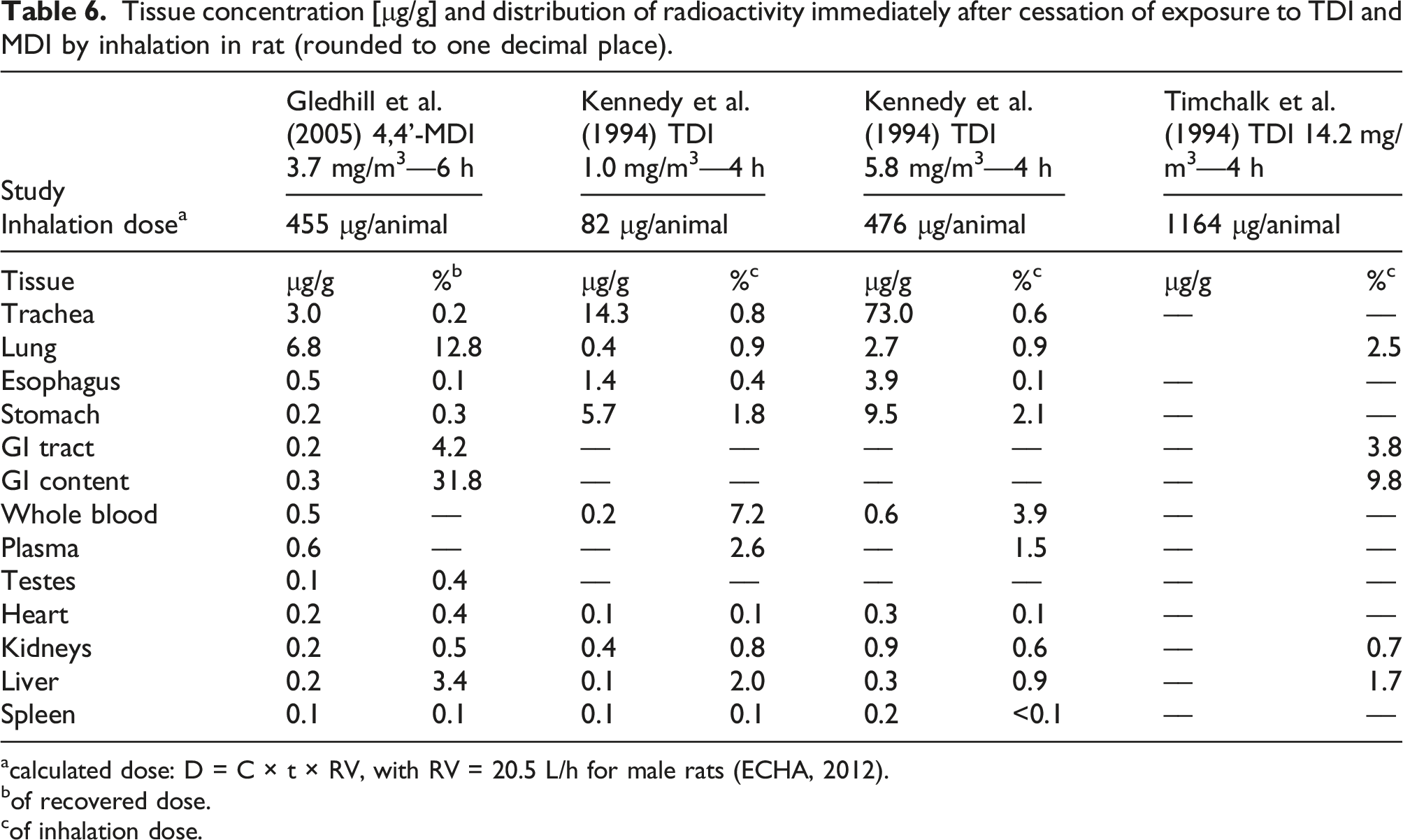
Tissue concentration [μg/g] and distribution of radioactivity immediately after cessation of exposure to TDI and MDI by inhalation in rat (rounded to one decimal place).
acalculated dose: D = C × t × RV, with RV = 20.5 L/h for male rats (ECHA, 2012).
bof recovered dose.
cof inhalation dose.
Male rats were exposed head-only to 0.15, 1.0, or 5.8 mg/m3 of radiolabeled 2,4-TDI for 4 h via inhalation, which corresponded to inhalation (not necessarily absorbed) doses of 12, 82, or 476 μg TDI per animal 6 (Kennedy et al., 1994). Directly after cessation of exposure, the animals were sacrificed and tissues analyzed. In the whole blood, the radioactivity increased with dose and was equivalent to 10.7, 7.3, and 3.9% of the low, medium, and high total tissue dose, respectively. In the airways, the tissue concentration increased almost proportionally to the exposure concentration. In other tissues, there was a clear but less than proportional dose-dependent increase in tissue load. These results are similar to prior work with guinea pigs (Kennedy et al., 1989). In the plasma, 97–100% of the radioactivity was bound to proteins with a molecular weight greater than 10 kDa. The presence of material in the esophagus and stomach was hypothesized to be the result of gasping during exposure and/or muco-ciliary lung clearance. Together with the sub-proportional dose-response in other tissues than the airways, this is in concordance with the expectation that at higher concentrations a greater percentage of the dosed TDI will end up in polyurea formation in the airways. A follow-up investigation with perfused lungs of guinea pigs revealed that after ventilation with 0.2 or 0.7 ppm radiolabeled TDI for 45 min, 25–35% of the estimated dose was retained in lung and perfusate (Kennedy et al., 1995).
In another study, rats were exposed head-only to 14.2 mg/m3 of radiolabeled TDI for 4 h via inhalation. Tissues were analyzed directly after exposure and 48 h thereafter (Timchalk et al., 1994). Over 48 h, ca. 15% of the radioactivity was excreted via urine, about 47% via faeces, 3.7% could be recovered as cage-wash, and 34% remained in the carcass. The half-time of urinary excretion was 20 h after inhalation exposure to TDI.
One study is available with MDI, whereby rats were exposed head-only for 6 h to 3.7 mg/m³ of respirable 4,4’-MDI aerosol, which corresponds to an inhalation dose of 455 μg per animal (Gledhill et al., 2005). Animals were sacrificed immediately, 8 h, 24 h, or 168 h after cessation of exposure. A satellite group was bile cannulated. In the bile cannulated rats after 48 h, 14% of the dose was excreted via bile, 34% via faeces, 12% in urine, and 24% was still in the gastro-intestinal tract. For the other animals, after 168 h, 84% of the radioactivity was excreted: 79% via faeces, and 5% via urine. The results in tissue distribution immediately after exposure are presented against those reported for TDI in Table 6.
Exposure to 5.8 mg/m³ TDI for 4 h results in a comparable total dose as 3.7 mg/m³ MDI for 6 h (476 μg against 455 μg). We hypothesize that the differences in tissue concentration between 4,4’-MDI and 2,4-TDI after inhalation exposure as shown in Table 6 can be explained by the physical properties of the two diisocyanates. With 3.7 mg/m³, the saturated vapor concentration of MDI was exceeded and exposure was to respirable aerosol. Aerosol particles follow the air stream and settle preferentially at low flow velocities, which occurs in bronchioles and alveoli. 2,4-TDI at 5.8 mg/m³ is in the vapor phase, which allows the molecules to interact with all regions of the respiratory epithelium much more readily than is the case for aerosol particles. Since aerosol particles are partly converted into high-molecular weight poly-ureas, a higher concentration of radioactivity was found in the lung compared to the trachea for MDI, whereas for TDI the situation is reversed. Muco-ciliary transport from the bronchi results in a relatively larger amount of TDI and its products to be found in the esophagus and in the stomach as compared to MDI. Concerning radioactivity in whole blood and the heart, an inhalation dose of 455 μg MDI (Gledhill et al., 2005) achieved a similar level of tissue residues as an inhalation dose of 476 μg TDI (Kennedy et al., 1994). For TDI, following Kennedy et al. (1994) approximately 3% of the inhaled TDI was found in blood, split between serum (2.4%) and cell pellet (0.6%). 7 MDI, too, showed higher binding ratios to serum albumin than to Hb. Biomonitoring studies with rats following inhalation exposure to MDI have shown that albumin adduct levels were 38 times higher than Hb adduct levels (Kumar et al., 2009; Sabbioni et al., 2000). Another study found albumin adducts to be at least 20 times higher than Hb adduct levels (Pauluhn et al., 2006). Both observations are consistent with the greater reactivity of Lys versus Val (see Table 3). For the kidneys, TDI achieved a higher tissue concentration than MDI. For the liver, TDI resulted in slightly lower tissue concentrations than MDI (Table 6). These observations seem to indicate slight differences in route or rate of excretion for the two diisocyanates, which is in line with the observation that, compared to TDI, a greater percentage of the MDI inhalation dose is excreted via faeces (muco-ciliary transport of polyurea and biliary excretion of MDI metabolites) rather than via urine.
Other than protein adducts, metabolites detected after inhalation exposure to TDI in 12 h urine samples were mono- and di-acetylated TDA (Timchalk et al., 1994). Contrary to oral TDI exposure or exposure to TDA, no free TDA could be detected in unhydrolyzed urine after inhalation exposure to TDI. In another subacute inhalation study with TDI in rats, all TDI was found to be bound to protein (Liu et al., 2013). Metabolites identified in rats after inhalation exposure to MDI were N,N’-diacetyl-4,4’-diaminobenzhydrol (OH-MDA-4,4’-(Ac)2) and N,N’-diacetyl-4,4’-diaminodiphenylmethane (MDA-4,4’-(Ac)2) in urine
8
and bile, N-acetyl-4,4’-diaminodiphenylmethane (MDA-4,4’-(Ac)) and N,N’-diacetyl 4,4’-diaminobenzophenone in urine (O=MDA-4,4’-(Ac)2), see Figure 5 (Gledhill et al., 2005). No unacetylated MDA was detected (Gledhill et al., 2005), and MDA was not considered to be a major metabolite of MDI inhalation (Sabbioni et al., 2000). Urinary and bile metabolites of MDI after inhalation exposure in the rat (Gledhill et al., 2005).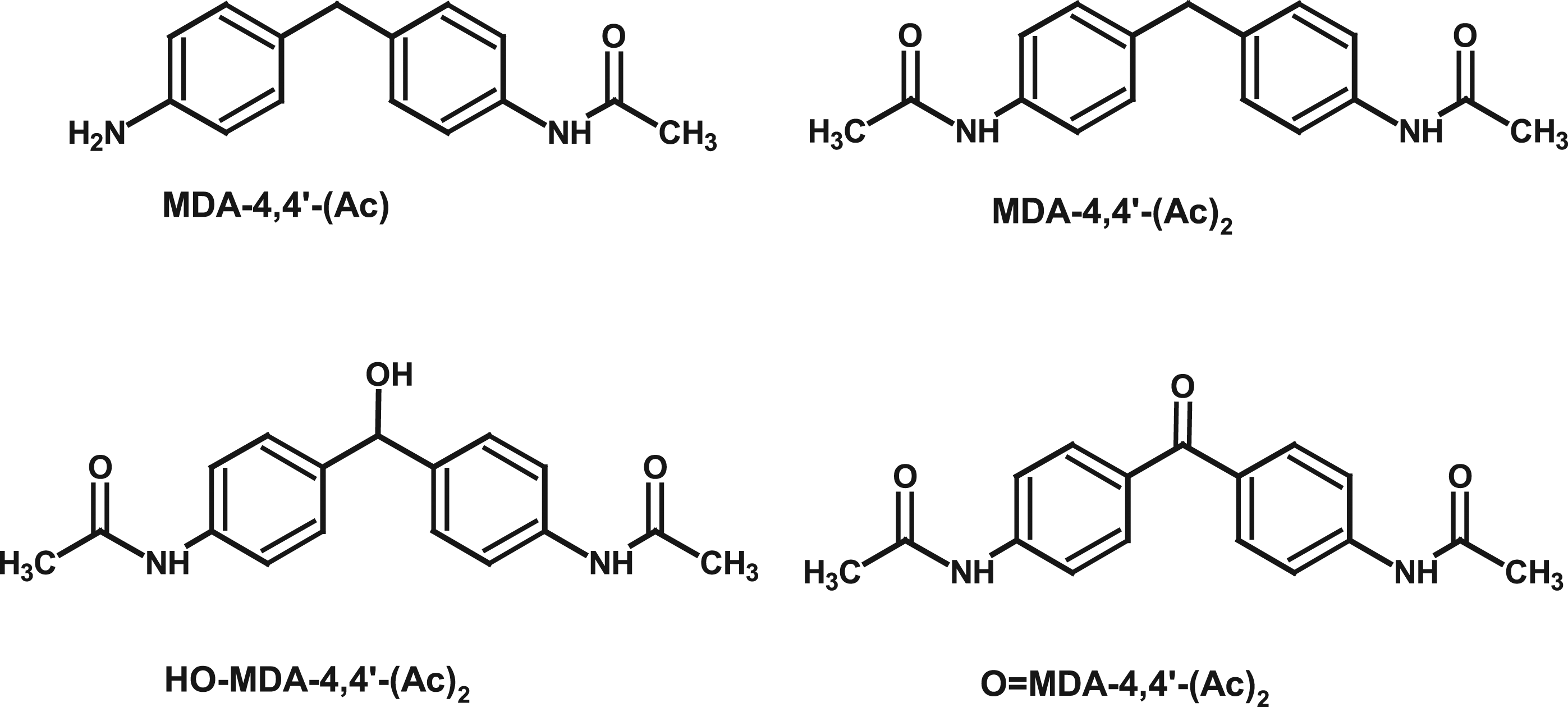
Compared to the MDI inhalation study (Gledhill et al., 2005), the same pattern of metabolites in urine could be found when rats received intratracheal dosages of the MDI-bis-glutathione adduct, MD-4,4’-(GSH)2, and when dogs were exposed to PMDI aerosol (Pauluhn et al., 2006). Urine was collected over 24 h after dosing; 0.2–0.4% of the dose could be recovered as MDA in hydrolyzed urine and 0.01% as Hb-adducts. From comparing metabolite profiles between the slow-acetylating dog and the fast-acetylating rat, the authors concluded that the metabolism of MDI is unlikely to involve free MDA.
The MDI-di-lysine adduct, MD-4,4’-(Lys)2, was detectable in urine of mice dosed with the MDI-bis-glutathione adduct intranasally (Wisnewski et al., 2019b); repeated dosages over five days resulted in increased urine levels.
All these observations for both TDI and MDI are consistent with the chemical reaction patterns described in the earlier sections.
(Human) biomonitoring
Standard biomonitoring of urine samples makes use of strong hydrolysis conditions to convert as much as possible of the adducts into the corresponding diamines for further analysis (Brorson et al., 1991; Lind et al., 1996; Marand et al., 2004; Rosenberg and Savolainen, 1986; Skarping et al., 1991). Caution in interpretation is warranted, since S-bound adducts are labile under mildly alkaline sample preparation steps at room temperature (Leinweber, 2011; Sabbioni et al., 1997), which may create the appearance of unbound diamines being present (Sepai et al., 1995).
Both the NCO-GSH adducts and NCO-protein adducts are subject to specific elimination processes, the end products of which can be analyzed as biomarkers. For GSH-adducts, this is the mercapturic acid pathway (Hanna and Anders, 2019), and cleavage of MD-4,4’ (GSH)2 by human γ-GT was demonstrated in vitro (Wisnewski et al., 2016b). Dermal dosage of MDI in acetone and intranasal application of MD-4,4’-(GSH)2 resulted in detectable levels of the MDI-di-lysine adduct in urine of mice (Wisnewski et al., 2019b).
The two pathways have different time constants, so that—similar to rodents—elimination from the human body appears to be at least bi-phasic: • Five persons received a controlled air exposure to 40 μg/m³ TDI (80 : 20); due to the greater volatility of the 2,6-TDI, the personal dose was close to 50 : 50 and reached approximately 60 μg for each of the 2,4- and 2,6-TDI isomers (Skarping et al., 1991). Urinary excretion and plasma levels were followed up to 15 h after exposure, and biological samples were analyzed as TDA after strong acidic or alkaline hydrolytic work-up. The urinary half-life was bi-phasic, and the initial half-lives were 1.9 h for 2,4-TDI and 1.6 h for 2,6-TDI. In other studies with human subjects, TDI elimination times were 2–3 h (Rosenberg and Savolainen, 1986), or biphasic with 2–5 h and 6 days (Brorson et al., 1991; Skarping et al., 1991). • In a biomonitoring study with strong hydrolysis work-up of urine, the half-life of urinary excretion of 2,4- and 2,6-TDA after airborne TDI exposure could be established as 5–8 days for 2,4-TDA and 9 days for 2,6-TDA (Lind et al., 1996). The plasma half-life ranged from 14–34 days (median 21 days) for 2,4-TDI and 16–26 days (median 21 days) for 2,6-TDI. These values are in good agreement with half-life of HSA (Levitt and Levitt, 2016; Sleep et al., 2013). • In specific challenge tests with TDI and MDI, the urinary elimination was investigated (Budnik et al., 2011). TDA from hydrolyzed urine achieved a peak between 4.1 and 4.8 h after exposure. After 6 h, no more TDA could be detected in hydrolyzed urine, and 15–20% was excreted after 8 h via urine. For MDA in hydrolyzed urine after MDI exposure, the urinary concentration had a peak at 13–15 h after exposure, and after 24 h, elimination was not complete. From the data provided, an elimination of 3–5% after 24 h in urine can be estimated for MDI. The slower elimination of MDI (5–12 h) compared to TDI (4–5 h) was also observed in another study (Yu et al., 2005); urinary half-lives of 10 h (median) to 15 h (mean) for MDI were reported in a biomonitoring study in the PU industry (Robert et al., 2007). • After specific inhalation challenge tests in workers, MDI-lysine adducts could be released from HSA by enzymatic digest. Approximately 0.6% of the MDI dose was calculated to be bound to albumin in humans. In rats, 0.24% of the inhalation dose of MDI was bound to serum albumin (Sabbioni et al., 2016). This lower level might be a result of the greater turn-over rate of RSA, which, at a plasma concentration of 26–32 mg/mL has a half-life of 2.5–3 days (Kumar et al., 2009; Peters and Peters, 1972). • In a publication concerning albumin adducts of TDI in exposed workers and then rat, Sabbioni et al. (2012b) estimated that ca. 0.2% of the inhalation dose is bound to RSA; in a study with rats, approximately 1.3% of the inhaled radiolabeled TDI was found in blood (Kennedy et al., 1994).
More detailed investigations have led to the identification of other biomarkers that are consistent with the putative adducts described in the chapter on reactions of NCO with nucleophiles: • In vitro, γ-glutamyltransferase hydrolyzed the MDI-bis-GSH adduct to the bis- and mono-MDI-mercapturic acid (Wisnewski et al., 2016b). • GSH-based adducts that cannot be transformed into mercapturates (e.g., N-bound cyclic GSH or GSSG adducts) were detected in vivo in the urine of exposed mice (Wisnewski et al., 2019b) or after in vitro incubation with murine liver S9 (Wisnewski et al., 2016b). • Hydantoin derivatives of the addition of NCO to the N-terminal Val residue in Hb were detected in vivo (Day et al., 1997; Gries and Leng, 2013; Kumar et al., 2009; Sabbioni et al., 2000). • Most frequently, the mono- and bis-Lys adducts could be detected after digestion of the proteins in case of exposure to TDI (Hulst et al., 2015; Luna et al., 2014; Sabbioni et al., 2012b) and as such in urine (Wisnewski et al., 2019b).
At present, the available biomarkers cannot differentiate between the dermal and inhalation routes of exposure. The detected adducts and biomarkers are all consistent with the previously described reaction pathways whereby the NCO group covalently binds with proteins either directly or via transcarbamoylation.
The toxicity of methylene diphenyl diisocyanate and toluene diisocyanate with respect to their biochemical activity
Repeated dose study results for MDI and TDI with inhalation exposure.
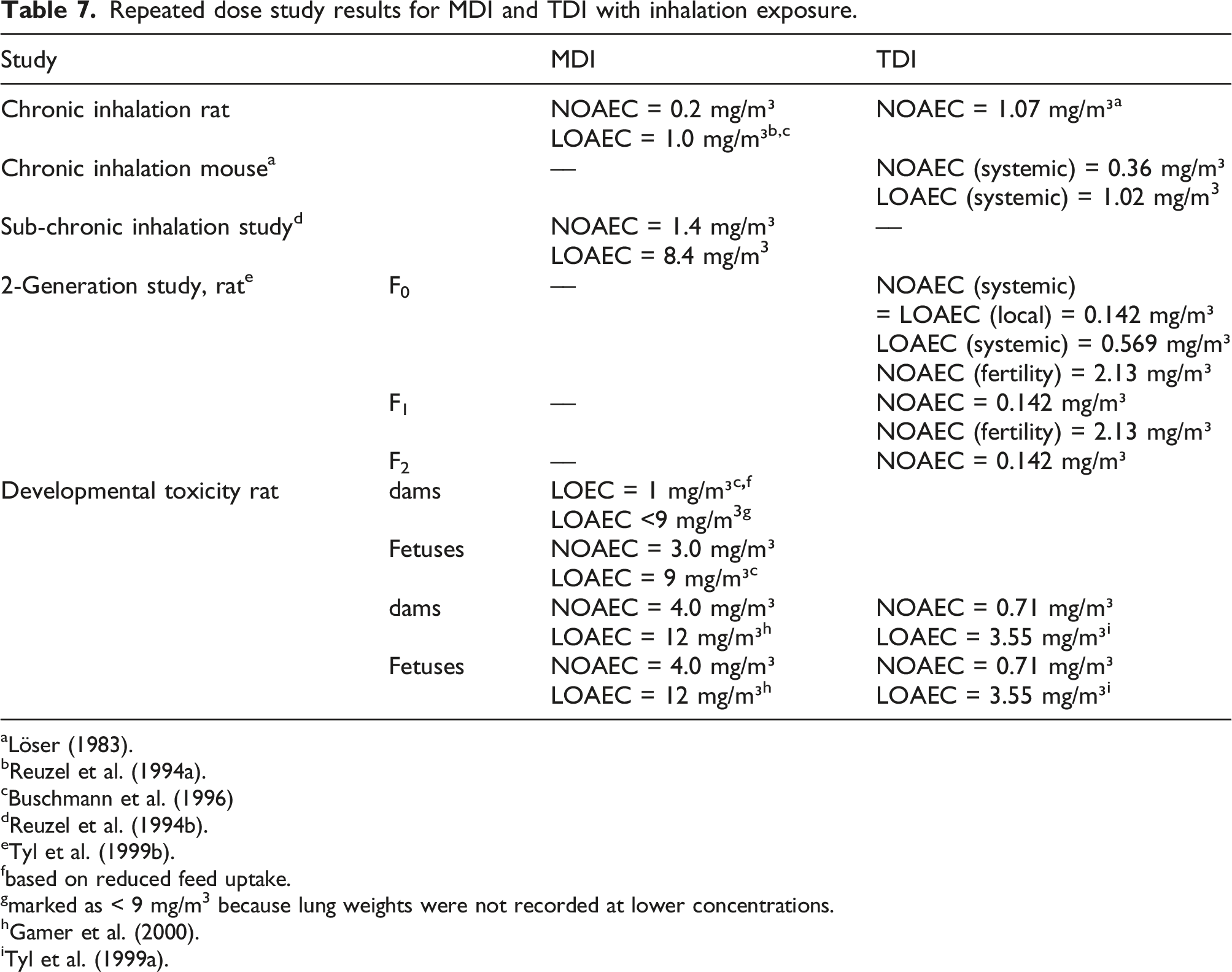
aLöser (1983).
fbased on reduced feed uptake.
gmarked as < 9 mg/m3 because lung weights were not recorded at lower concentrations.
A review of the toxicity of MDI and TDI was published in 2018 by the US ATSDR (2018). Under certain circumstances, for example, in the acidic environment of the stomach or when dissolved in organic solvents and then dispersed in water, MDI, and TDI may hydrolyze and form detectable amounts of MDA and TDA, known mutagens and carcinogens. The carcinogenicity detected in a chronic oral gavage studies of TDI with mice and rats exhibited a pattern of tumor sites and incidences that could be explained by the release of TDA, and the relevance for human exposure was questioned (Dieter et al., 1990, 1991; Doe, 1991; Sielken et al., 2012). The difficulty in assessing the genotoxic potential of MDI and TDI, and the relevance of released MDA and TDA is addressed in literature (Bolognesi et al., 2001; Prueitt et al., 2013) and in expert panel reviews (ATSDR, 2018; MAK Commission, 2003, 2008).
The primary acute and chronic effects of air exposure to MDI and TDI at the workplace are respiratory inflammation, respiratory sensitization, and lung function decrement; for dermal exposure, skin irritation and skin sensitization are the observable adverse effects (ATSDR, 2018; MAK Commission, 2003, 2008).
Besides respiratory inflammation and sensitization as a result of respiratory exposure, tumorigenesis was reported in chronic inhalation experiments with rats for MDI (Hoymann et al., 1995; Reuzel et al., 1994a), whereas TDI was not tumorigenic in rats and mice in chronic inhalation studies (Löser, 1983). For both MDI studies, lung tumors occurred towards the end of the studies at respiratory irritating concentrations (Feron et al., 2001). The German MAK Commission classified MDI as MAK-Cat.4 9 carcinogen and allocated an occupational exposure limit of 50 μg/m³ (MAK Commission, 2008). In these chronic rat inhalation studies, the NOAEC for tumor formation was 1.07 mg/m3 for TDI (Löser, 1983) and 0.2 mg/m3 for MDI; the LOAEC for MDI was 1.0 mg/m3 (Feron et al., 2001; Hoymann et al., 1995; Reuzel et al., 1994a). Concerning TDI, mice turned out to be more sensitive than rats; at the lowest dosage, severe effects on the respiratory epithelia were already observed, and reduced body weight gain and increased mortality was observed in the high dose group. The low dose could be described as a borderline NOAEC at 0.36 mg/m3, and 1.02 mg/m3 was adverse (Löser, 1983). For both MDI and TDI, observable adverse effects were confined to the respiratory tract. No other organ showed any histopathological changes. For MDI, particle loaded macrophages were found in the lung and the lung-associated lymph nodes. This finding indicates that the MDI aerosol droplets end up in particles, which can be expected to be polyurea based on the reactivity of isocyanates in aqueous solutions reported as described in the earlier chapters of this work.
In a sub-chronic inhalation study with MDI in rats, 8.4 mg/m³ was the LOAEC that caused respiratory distress, reduced body weight, and increased lung weights (Reuzel et al., 1994b); there were no other histopathological findings in other organs. Clinical chemical parameters and urine analysis did not show remarkable deviations from controls, demonstrating that albumin homeostasis was not affected.
Rats were exposed to 0.142, 0.569, or 2.13 mg/m³ TDI for 6 h/day and 5 days/week in a two-generation study (Tyl et al., 1999b). All concentrations caused rhinitis in exposed rats. The middle concentration caused a transient reduction in pup weight in the F2 generation, the high concentration resulted in a permanent pup weight reduction in the F1 and F2 generation. Parental body weight gain was depressed at the middle and high concentration. A systemic NOAEC of 0.142 mg/m³ could be derived for the F0 generation, and a NOAEC of 2.13 mg/m³ for fertility and 0.569 mg/m³ for postnatal development. Reproductive organs, liver, kidney, and pituitary gland did not show any effects.
Pregnant rats were exposed to 0.142, 0.71, or 3.55 mg/m³ TDI for 6 h/d during gestation days 6–15 (Tyl et al., 1999a). All concentrations caused red nasal discharge, which was rated as not adverse. At the highest concentration, a minimally reduced ossification and clinical signs of toxicity, reduced feed uptake, reduced body weight gain, and reduced body weight in the dams were noted. Fetotoxic or teratogenic effects were not observed. For pups and dams, the (systemic) NOAEC was 0.71 mg/m³, the LOAEC 3.55 mg/m³.
In a developmental toxicity study, pregnant rats were exposed to 1, 3 or 9 mg/m³ 4,4’-MDI for 6 h/d during gestation days 6–15 (Buschmann et al., 1996). In all treatment groups, there was a dose-dependent reduction in food consumption. The lung weights of the dams in the high dose group were significantly increased. A slight but significant increase in fetuses with asymmetric sternebrae was observed in the high dose group but remained within the range of historical control. In another developmental toxicity study with pregnant rats, the exposure regime was maintained, but the concentrations were 1, 4, and 12 mg/m³ (Gamer et al., 2000). At 12 mg/m³, 2 out of 12 dams died, all dams showed significantly reduced body weight gain, body weight, reduced liver, and increased lung weights. At the top dose, placental and fetal body weight was reduced, and the incidence of skeletal variations and skeletal retardation was increased. Therefore, for dams and pups the NOAEC was 4, the LOAEC 12 mg/m³.
For MDI, no study is available that explicitly addresses fertility. However, as MDI and TDI both show very similar behavior in reactivity, metabolism, distribution, excretion, and toxic effects, including the absence of histopathological effects in reproductive organs (Blackburn et al., 2015), it is deemed highly likely that MDI, as demonstrated for TDI, would not be toxic to fertility either.
It can therefore be confirmed that effects for both diisocyanates are confined to the portal-of-entry and that systemic toxicity is not observed below certain thresholds.
Summary and conclusion
This review summarizes the current knowledge about the (bio)chemical reactivity of MDI and TDI. Against this background, toxicity data generated to date for both diisocyanates allow to draw a conclusive picture.
Observations from all the in vivo repeated dose inhalation toxicity studies cited demonstrate that MDI and TDI both can cause adverse effects at the portal-of-entry. Ultimately, sensitization of the respiratory tract and the skin is also a result of “local” contact. Due to their chemistry, both isocyanates react with biological macromolecules at the site of impact.
As long as GSH is not exhausted, the reaction to GSH-adducts (thiourethanes) is kinetically favored, and the adducts are stable as long as GSH is present in excess. At declining GSH levels, the thiourethanes readily transfer the isocyanate moiety to primary amine functions to form the thermodynamically more stable N-substituted ureas that prevent further reaction of the NCO group. The transcarbamoylation predominantly takes place with certain Lys side chains in albumin, and to a lesser extent with Val in Hb. In the absence of GSH, hydrolysis of MDI, TDI, or their GSH-adducts leads to the formation of polyureas of MDI or TDI, and this polymer formation becomes the more important the higher the dose.
Albumin adducts are hypothesized to play a crucial role in the acquisition of sensitization to MDI or TDI. Albumin adducts synthesized by TDI vapor phase exposure, or by reaction between MDI-bis-GSH and albumin, bind IgG and IgE antibodies in serum of exposed workers (Johannesson et al., 2004; Sabbioni et al., 2017; Wisnewski et al., 2004, 2010, 2013, 2016a). Although dermal penetration of MDI and TDI is limited, protein adducts were identified after skin exposure (Hulst et al., 2015; Nayak et al., 2014; Wisnewski et al., 2011b); their uptake and processing in Langerhans cells (tissue-resident macrophages in the skin) may be a basis for the understanding of the role of skin contact for the acquisition of isocyanate hypersensitivity (Bello et al., 2007; Kimber et al., 2018; Nayak et al., 2014; Pauluhn, 2014; Wisnewski et al., 2011b). Concerning occupational exposure limits to prevent the acquisition of isocyanate asthma and other respiratory effects, the role of total cumulative dose versus peak exposure has been discussed (Daniels, 2018; Plehiers et al., 2020a, 2020b). The biochemistry and physiology of the isocyanate-albumin interaction may shed some more light on this debate. Although an adduct level threshold for albumin-isocyanate has not yet been identified, it is tempting to assume that only after a minimum adduct-level is reached will the immune system respond (Sabbioni et al., 2017). At what point a certain—yet unknown—critical adduct level is achieved, is ultimately a function of the adduct-formation rate, and the albumin-turnover rate.
At approximately similar inhalation dosages, metabolism studies have revealed a higher trachea versus lung load for TDI (Kennedy et al., 1994) than for MDI (Gledhill et al., 2005). We interpret these findings being attributable to differences in the physical form of MDI and TDI in inhalation exposure studies. Respirable MDI aerosol reaches the bronchioles and alveoli where it is expected to be mostly cleared by macrophages, partly drained by the lymph and partly cleared by the muco-ciliary escalator of the respiratory tract. A comparatively larger portion of the inhaled TDI vapor seems to react in the trachea of the rodents, already, where muco-ciliary clearance is the most important clearance mechanism at elevated loadings.
With the exception of sensitization, the albumin bound isocyanate is unlikely to pose a risk anymore, since the isocyanates are covalently bound as chemically stable substituted ureas. Albumin binding is known to reduce systemic availability and toxicity of drugs, and covalent binding of cytostatic drugs, for example, increased the maximum tolerable dose by a factor of 4 and more (Sleep et al., 2013).
In the form of albumin adducts, both diisocyanates can be excreted in urine, but endogenous digestion of albumin with release of Lys-adducts occurs as well, as these adducts are found as metabolites in urine. The breakdown products are obviously not systemically toxic at the concentrations that can be achieved after inhalation exposure: no histopathological damage in liver, kidneys, or organs other than the lung were observed in repeated dose inhalation toxicity studies.
Overall, MDI and TDI show a very similar pattern of NCO-adduct formation, adsorption, distribution, metabolism and excretion, target organs, and biological effects.
Supplemental Material
sj-pdf-1-tih-10.1177_07482337211060133 – Supplemental Material for Absorption, distribution, metabolism, and excretion of methylene diphenyl diisocyanate and toluene diisocyanate: Many similarities and few differences
Supplemental Material, sj-pdf-1-tih-10.1177_07482337211060133 for Absorption, distribution, metabolism, and excretion of methylene diphenyl diisocyanate and toluene diisocyanate: Many similarities and few differences by Thomas Schupp and Patrick M Plehiers in Toxicology and Industrial Health
Footnotes
Acknowledgements
Declaration of conflicting interests
Thomas Schupp is consultant to the International Isocyanate Institute, Inc., BASF Polyurethane GmbH (Germany) and the European Flexible Foam Producer Association (EUROPUR). Patrick M. Plehiers is employed by the International Isocyanate Institute, Inc. The Institute is funded by producers of MDI and TDI.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded with regular membership dues of the International Isocyanate Institute, Inc. There is no other funding to report.
Supplemental material
Supplemental material for this article is available online
Disclaimer
The opinions expressed in this paper are those of the authors, not necessarily of the International Isocyanate Institute, Inc. or its member companies.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
