Abstract
The water extractability and acute aquatic toxicity of seven aliphatic diisocyanate-based prepolymer substances were investigated to determine if lesser reactivity of the aliphatic isocyanate groups, as well as increased ionization potential of the expected (aliphatic amine-terminated) polymeric hydrolysis products, would influence their aquatic behavior compared to that of previously investigated aromatic diisocyanate-based prepolymers. At loading rates of 100 and 1,000 mg/L, only the substances having log Kow ≤9 exhibited more than 1% extractability in water, and a maximum of 66% water extractability was determined for a prepolymer having log Kow = 2.2. For the more hydrophobic prepolymer substances (log Kow values from 18–37), water extractability was negligible. High-resolution mass spectrometric analyses were performed on the water-accommodated fractions (WAF) of the prepolymers, which indicated the occurrence of primary aliphatic amine-terminated polymer species having backbones and functional group equivalent weights aligned to those of the parent prepolymers. Measurements of reduced surface tension and presence of suspended micelles in the WAFs further supported the occurrence of these surface-active cationic polymer species as hydrolysis products of the prepolymers. Despite these characteristics, the water-extractable hydrolysis products were practically non-toxic to Daphnia magna. All of the substances tested exhibited 48-h EL50 values of >1,000 mg/L, with one exception of EL50 = 157 mg/L. The results from this investigation support a grouping of the aliphatic diisocyanate-based prepolymers as a class of water-reactive polymer substances having predictable aquatic exposure and a uniformly low hazard potential, consistent with that previously demonstrated for the aromatic diisocyanate-based prepolymers.
Keywords
Introduction
Assessments of potential risks to human health and the environment are necessary to comply with various global chemical control regulations, as well as to advance sustainable development within the chemicals industry. In the past, such assessments have not been of high priority for synthetic polymer substances, as the high molecular weight of polymers is generally associated with lesser hazards, exposures, and risks compared to their monomeric precursors and to other non-polymeric chemicals. Today, there is heightened awareness of the occurrence and potential impacts of plastic polymer wastes in the environment, which, to a large extent, has raised concern for the pollution potential of all synthetic polymers in the environment—including those that cannot be visibly seen as pollutants. The European Commission has recently completed a study evaluating potential approaches for selecting and grouping polymers that may be subject to future registration and evaluation under EU-REACh (European Commission, 2020). That study pointed to the need for assessing polymers based on grouping or read-across approaches since; (1) polymers are by definition complex mixtures of homologues/isomers, (2) there is wide variety in composition within each polymer type, and (3) there are a large number of polymer types in commerce. The cost, time, and complexity associated with such assessments becomes insurmountable if each polymer (e.g., by unique CAS Registry Number) is to be assessed on an individual basis. Among the polymer types prioritized for registration and evaluation under the EU-REACh polymer grouping approach are the cationic polymers, and polymers having reactive functional groups. These same polymer types are among those that currently cannot be exempted from regulatory evaluations as so-called Exempt Polymers, Reduced Regulatory Requirement (RRR) polymers, or Polymers of Low Concern (PLC) by authorities such as the United States Environmental Protection Agency (US EPA, 1997; US EPA, 2010), Environment and Climate Change Canada (ECCC, 2022), and the Chinese Ministry of Environment (Ge, 2021). The polymer class known commercially as the diisocyanate prepolymers is included among the “polymers with reactive functional groups” type, and when emitted to the environment, these may form hydrolysis products that are identified as “potentially cationic polymers” (US EPA, 1997). In light of these polymer characteristics and the large number, variety, and volume of their commercial variants produced, the diisocyanate-based prepolymers present a commercially important and technically challenging case for assessing exposure, hazard, and risk using a polymer grouping approach.
Whereas the aromatic diisocyanate-based prepolymers studied previously (West et al., 2022a) are used in applications requiring rapid reactivity, durability, and high resistance to thermal and chemical degradation, the aliphatic prepolymers are used where colorfastness and resistance to photolytic degradation are necessary in addition to these other durability traits. Examples of applications for which these properties are important include protective coatings for metals, non-yellowing floor coatings, waterproofing membranes, and weather-resistant elastomeric sealants and adhesives (see https://www.americanchemistry.com/industry-groups/aliphatic-diisocyanates-adi/applications). The aliphatic polyisocyanate prepolymer chemistry has key roles in enabling alternative energy advancements—some of the many technologies they enable include use as sealants in LED lighting and solar panel assemblies, and as finish coatings for wind turbine blades. The manufacture, transport, use, and end-of-life stages for these and other aliphatic diisocyanate prepolymer applications could potentially (though unlikely) result in emission to the aquatic environment. Thus, it is important for both regulatory and product stewardship purposes to assess the aquatic exposure and hazard properties of these polymers.
The preceding articles in this series have described an approach for experimental design, synthesis, characterization, and testing of diisocyanate-based prepolymers to explore relationships of structure and physical-chemical properties to aquatic exposure and hazard (West et al., 2022a) and to skin sensitization potency (West et al., 2022b). The seven aliphatic isocyanate-based prepolymer substances prepared and evaluated in the latter study were evaluated here with respect to their aquatic exposure (extractability in water) and acute aquatic toxicity (immobilization of Daphnia magna) potentials. The present work builds on the results of the prior study (West et al., 2022a) by including analytics that further defined the composition and properties of the water-extractable fractions of the aliphatic prepolymer substances. These analyses included the following: - High-resolution mass spectrometric analyses to elucidate the structural features of the water-extractable species. - Measurements of surface tension of the water-extractable fractions, to determine if surface-active properties may enhance the extraction, suspension, and dissolution of the prepolymer hydrolysis products in water. - Presence of suspended particles (i.e., micelles or micro-emulsions) that could contribute to D. magna immobilization by the prepolymer substances via physical toxicity mechanisms.
The aquatic exposure and hazard properties of the aliphatic diisocyanate monomers, hexamethylene diisocyanate (HDI), and isophorone diisocyanate (IPDI), which were used to synthesize the subject prepolymers, have been thoroughly tested and summarized previously (ECHA, 2023a; 2023b). Both the water solubility and acute toxicity of water-extractable fractions for these water-reactive aliphatic diisocyanate monomers are attributed to the aliphatic diamines (i.e., hexamethylenediamine, HDA; isophorone diamine, IPDA) formed from their hydrolysis. The reported results from acute aquatic toxicity tests with freshwater fish, algae, and invertebrates indicated that the invertebrate (Daphnia magna) is most sensitive of the tested species to HDI, IPDI, HDA, and IPDA (ECHA. 2023a-d). Considering this, and for consistency with the prior evaluation of aromatic diisocyanate-based prepolymers, Daphnia magna was selected as the sensitive test organism to evaluate acute aquatic hazard potential in the present study.
The aliphatic diisocyanate-based prepolymers are excluded from polymer of low concern (PLC) and similar polymer exemption considerations under various chemical control laws owing to their expected hydrolysis to form amine-terminated polymer species, which can occur in protonated forms in the environment (ECCC, 2022; Ge, 2021; US EPA, 1997). Formal characterizations of these envisioned hydrolysis products, which are expected to have essentially the same molecular weight, functionality, and polymer backbone features as their parent prepolymers, have not been investigated or reported previously. In addition, polymers having the expected structure/property features of these hydrolysis species are not commercially available, and therefore have not been evaluated for physical/chemical and toxicological properties. Thus, the QSAR tools (e.g., EPI Suite; US EPA, 2012), which are routinely used by regulatory authorities to evaluate new polymer substances, may not be sufficiently “trained” to accurately predict the aquatic exposure and hazard potentials of the isocyanate prepolymers and their associated hydrolysis products. In particular, the physical-chemical properties (i.e., MW, log Kow) of these polymers may extend well beyond the applicable domains of the existing QSAR tools. For the more broadly defined category of cationic polymer substances, the aquatic hazard properties and associated mechanisms of toxicity have been recently reviewed (Connors et al., 2023; ECCC, 2020; Hansen et al., 2023). However, none of the substances included within these assessments (i.e., quaternary ammonium functionalized polymers) have direct structural analogy to those of interest to this study (i.e., protonated aliphatic amines). Therefore, this study was designed to provide insight into the aquatic hazard properties of a unique and commercially significant class of polymer substances and their expected environmental transformation products.
Materials and methods
Experimental design
Identity and key properties of the aliphatic diisocyanate-based prepolymer test substances. The nomenclature indicates the diisocyanate type, polyol backbone construct, and nominal molecular weight of the polymeric prepolymers.
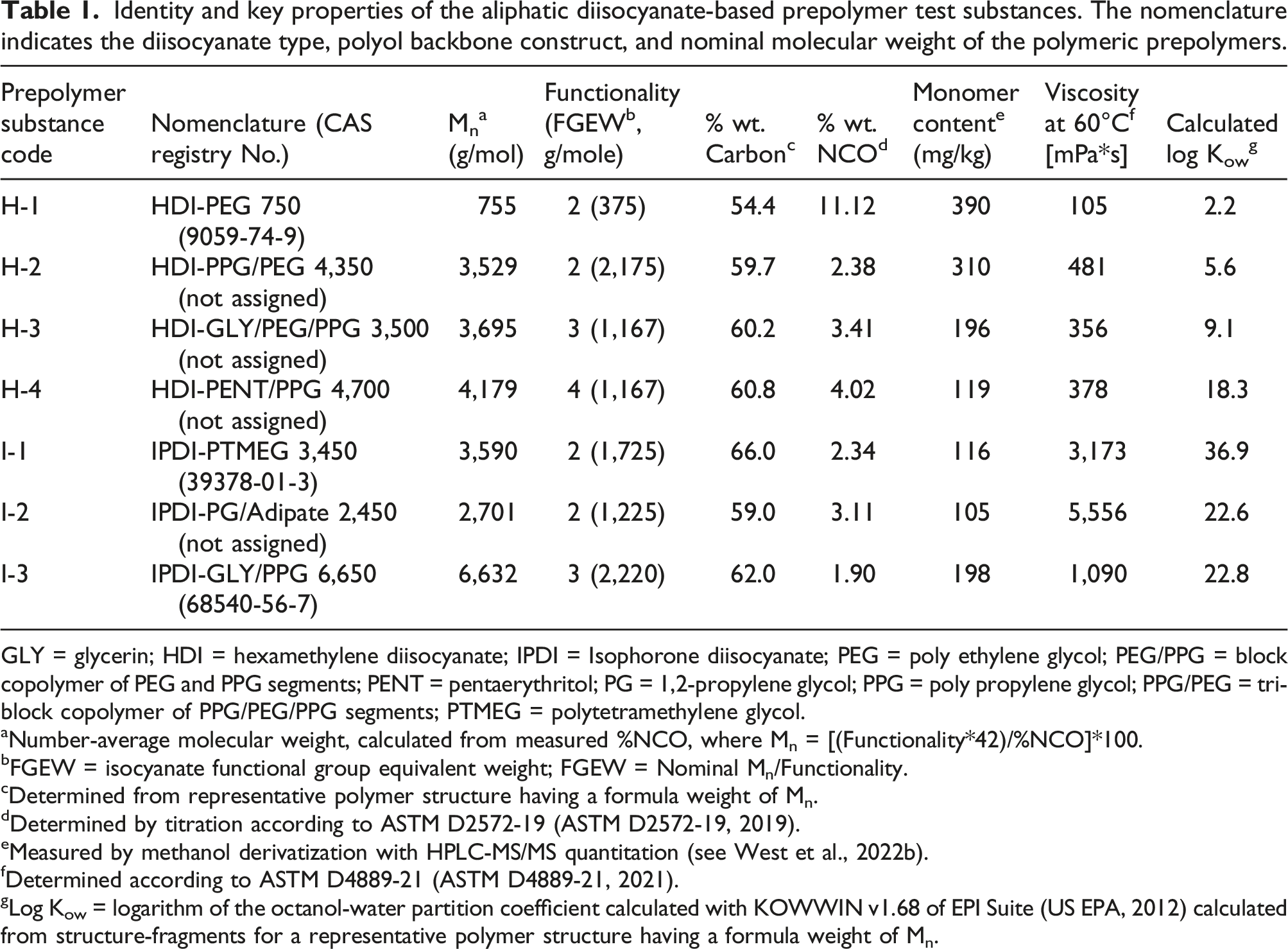
GLY = glycerin; HDI = hexamethylene diisocyanate; IPDI = Isophorone diisocyanate; PEG = poly ethylene glycol; PEG/PPG = block copolymer of PEG and PPG segments; PENT = pentaerythritol; PG = 1,2-propylene glycol; PPG = poly propylene glycol; PPG/PEG = tri-block copolymer of PPG/PEG/PPG segments; PTMEG = polytetramethylene glycol.
aNumber-average molecular weight, calculated from measured %NCO, where Mn = [(Functionality*42)/%NCO]*100.
bFGEW = isocyanate functional group equivalent weight; FGEW = Nominal Mn/Functionality.
cDetermined from representative polymer structure having a formula weight of Mn.
dDetermined by titration according to ASTM D2572-19 (ASTM D2572-19, 2019).
eMeasured by methanol derivatization with HPLC-MS/MS quantitation (see West et al., 2022b).
fDetermined according to ASTM D4889-21 (ASTM D4889-21, 2021).
gLog Kow = logarithm of the octanol-water partition coefficient calculated with KOWWIN v1.68 of EPI Suite (US EPA, 2012) calculated from structure-fragments for a representative polymer structure having a formula weight of Mn.
As indicated previously (West et al., 2022b), the residual monomer contents of these substances were reduced by vacuum stripping (to within 0.01%–0.04% wt.) to minimize potential influence of the monomeric HDI and IPDI and associated hydrolysis products (i.e., HDA and IPDA) on the measured exposure and hazard properties. Generic structures of the seven test substances are shown in Figure 1, and relevant physical-chemical properties are summarized in Table 1. Representative generic structures of the aliphatic diisocyanate-based prepolymer test substances. GLY = glycerol; HDI = hexamethylene diisocyanate, IPDI = isophorone diisocyanate, PEG = polyethylene glycol; PEG/PPG = di-block copolymer of polyethylene glycol and polypropylene glycol segments; PENT = pentaerythritol; PG = 1,2-propylene glycol; PPG = polypropyleneglycol; PPG/PEG = tri-block copolymer of polyethylene glycol/polypropylene glycol/polyethylene glycol segments; PTMEG = polytetramethylene glycol. Structures were drawn by the authors using ACD Labs ChemSketch v2020.2.0 software.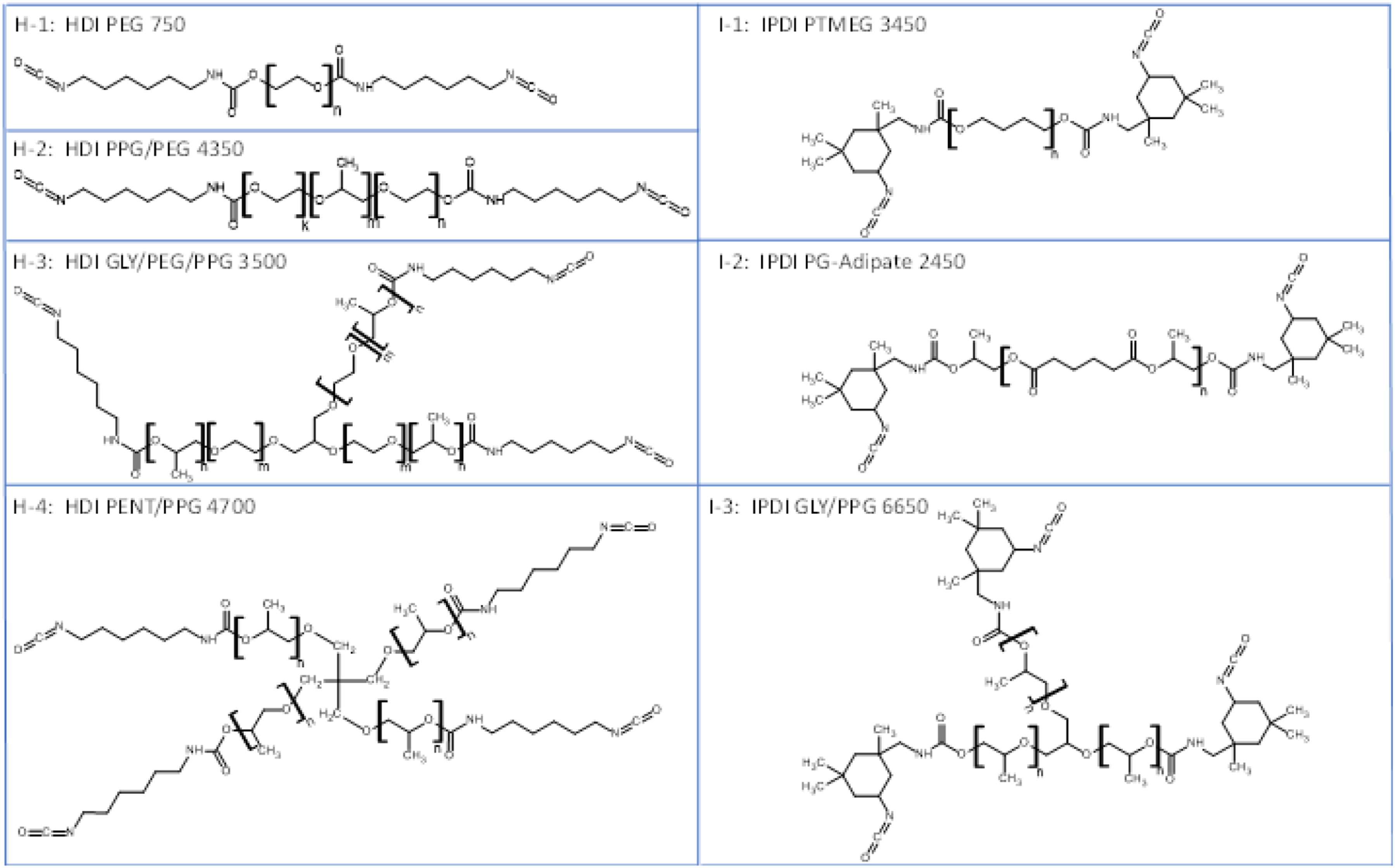
Synthesis of test substances
The prepolymer test substances were prepared by reaction of HDI (Desmodur® H; purity ≥99.5%; Covestro LLC, Pittsburgh, PA, USA) or IPDI (VESTANAT® IPDI; purity ≥99.5%; Evonik Corporation, Parsippany, NJ, USA) with selected polymeric polyols at Anderson Development Company (Adrian, Michigan, USA). Six of the polyol substances were obtained from commercial sources with documented number-average molecular weight (Mn), hydroxyl number, and water content. These same properties were determined for the seventh polyol (i.e., pentaerythritol propoxylate, Mn = 4,000), which was custom synthesized (Covestro LLC, Pittsburgh, PA, USA) to enable testing of a quadri-functional prepolymer at a targeted molecular weight that could not be obtained commercially. The polyols were selected to provide the narrowest practical distributions in molecular weight and functionality for a given targeted polyol type and molecular weight range. These selection criteria served to ensure that results could be interpreted based on nominal prepolymer features (e.g., functionality, Mn) without substantial bias introduced by constituents having greater molecular weight and/or lesser functionality than the nominal targeted values.
The potential (hydrolytic) degradation of the finished prepolymer test substances during storage was minimized by immediate transfer into several 250 mL brown polypropylene bottles that were sealed under dry N2 gas with air-tight screw caps wrapped with vinyl tape. These containers were not opened until immediately prior to use in the testing and were immediately re-sealed under a dry N2 gas atmosphere. Similar prepolymer substances have been shown to be stable for several years in the unopened container when such precautions were taken during their preparation and packaging.
Characterization of test substances
Measurements of prepolymer properties
Selected chemical and physical-chemical properties of the prepolymer test substances were characterized as previously described (West et al., 2022a; 2022b). The parameters measured and their associated methods are summarized as follows: • Isocyanate content (%wt) by ISO 14896:2018, titration with dibutyl amine (ISO, 2019) • Isocyanate monomer content (mg/kg) by derivatization with methanol, quantification by HPLC-MS/MS (see West et al., 2022b) • Dynamic viscosity @ 60, 80, and 100 C by ASTM D4889-21 (ASTM, 2021)
The number-average molecular weight (Mn) of each prepolymer was determined from the % NCO titration results as follows:
To confirm the expected narrow molecular weight distributions of the prepolymer test substances, the weight-average (Mw) and number-average (Mn) molecular weights and polydispersity (Mw/Mn) of each substance were also qualitatively evaluated using gel permeation chromatography (GPC). The experimental conditions and results of the GPC analyses are summarized in the Supplemental Information.
Estimations of prepolymer properties
The octanol-water partition coefficient (log Kow) for each prepolymer substance was estimated as previously described (West et al., 2022a). For each of the test substances, a representative polymer structure was created having formula weight approximately equal to the number-average molecular weight (Table 1) using ACD/Labs ChemSketch software (v2020.2.0, Advanced Chemistry Development, Inc., Toronto, Ontario, Canada). From these structures, corresponding simplified molecular input line entry system (SMILES) codes were generated for input into US EPA EPI Suite (v4.12; US EPA, 2012) running the KOWWIN v1.68 and ECOSAR v1.11 prediction software. In cases where the molecular weight of an input structure was too high to be computed by the software, representative structures of lesser molecular weight homologues (i.e., fewer polyether repeat units) were computed to determine the polyol repeat unit contributions to log Kow. These values for the missing repeat units of the target polymer were added to that of the computed lesser molecular weight polymer structure to calculate/estimate the log Kow.
The acid dissociation constants (pKa) and octanol-water distribution coefficient (log D, at pH 7) of the proposed amine-terminated polymeric hydrolysis products of the prepolymers were similarly estimated using the Playground v1.2 online property prediction interface (ChemAxon, 2023). The representative structures were drawn using ChemSketch software and imported as an MDL Molfile directly into the ChemAxon web portal.
Aquatic exposure potential
Test medium
The extractability of the prepolymer substances in water was evaluated according to a modification of OECD Guideline 120 (OECD, 2000) as described previously (West et al., 2022a). As in the prior work, a formulated mineral medium was used for the water-extractability measurements so that the undiluted water-extractable fractions could be used directly in the subsequent acute Daphna magna exposures. To the extent possible, the contents of the mineral medium were minimized with respect to components (aside from water) which could directly react with the isocyanate substances, while still supporting suitable survival of the Daphnid neonates. The minimized mineral medium (MMM) was prepared by combining equal volumes of the modified depleted mineral medium (DMM) described in the prior study (West et al., 2022a) with the Elendt M4 medium described in OECD Guideline 202 (OECD, 2004), with each being prepared in reverse osmosis water. The MMM therefore contained the same concentration of macro-nutrients (CaCl2, MgSO4, NaHCO3, and KCl), and one-half the concentration of vitamins (Thiamine, B12, Biotin) and trace elements of the standard Elendt M4 medium. The MMM was prepared in separate batches just prior to use and had measured hardness ranging from 219 to 268 mg/L (as CaCO3) and pH ranging from 7.0 to 7.9.
Water-extractability test
The water extractability of each prepolymer substance was evaluated in triplicate experiments at gravimetric loadings of 100 and 1,000 mg/L in the MMM. Precisely weighed portions of 100 or 1,000 mg of the prepolymer substances were applied to the lower inside walls of 1 L glass beakers. The MMM (1 L) was carefully added to each beaker while minimizing dispersion of the prepolymers, and magnetic PTFE-coated stir bars were used to stir at approximately 100 r.p.m., producing a vortex of 2-3 mm depth in the center of the stirred MMM. Control mixtures without added test substances were similarly prepared in triplicate for each testing campaign using 2 L beakers and 2 L portions of the MMM, and these were stirred and sampled in the same manner. The beakers were covered with Parafilm® M (MilliPore Sigma, Merck Life Science UK Ltd.), and their initial liquid levels were marked to allow tracking and replacement of any evaporated water with RO water. Homogeneous samples (∼45 mL) of the stirred reaction mixtures and controls were taken after 0, 24, 48, and 72 h of continuous stirring at 20 ± 1°C. These samples were filtered through 0.45 micron syringe filters (Pall Acrodisc™ with 25 mm Supor™ polyethersulfone membranes; Pall Europe, Hampshire, United Kingdom), with the first several mL of filtrate discarded to lessen any leachable filter contaminants and to saturate any adsorptive surfaces of the filter components. The remainder of sample filtrate was collected for analyses of non-purgeable organic carbon (NPOC). Portions (1 mL) of the stirred reaction mixtures were collected at 0 and 72 h to analyze for presence of unreacted aliphatic isocyanate groups using a colorimetric SWYPE™ test kit (described below). When results of the NPOC analyses confirmed that dissolved organic carbon concentrations had reached their maxima, 150–300 mL portions of the reaction mixtures and controls were collected and filtered through a series of 0.45 and 0.2 µm syringe filters (Pall Acrodisc™ with 25 mm Supor™ polyethersulfone membranes). Care was taken to avoid passage of air through the filters while the first several mL of filtrate were discarded and the remaining filtrate was collected. From the filtered portions of each reaction mixture and control, a sample was taken for NPOC analysis and pH measurement. Equal portions of the remaining filtrate from each replicate were pooled to become the water-accommodated fractions (WAFs) used in the D. magna limit test exposures. Samples of these pooled exposure solutions were also collected for analyses of NPOC, pH, turbidity, and surface tension as described below.
Polyurea analyses
Stirring of the remaining portions of the original 1,000 mg/L reaction mixtures was continued for a total of 96h. The contents of each beaker, along with any adhered solids that could be dislodged from the glass, were vacuum filtered through Whatman GF/A filter paper (55 mm). The SWYPE™ testing pads were firmly pressed against portions (∼1 g) the wet solids collected on the filter papers to determine if any unreacted isocyanate groups could be detected on the surface of the solids. Weighed portions of these solids were then combined with tetrahydrofuran (THF, 1 mL per 10 mg solids) in glass vials and vigorously shaken. Portions of the THF extract (1 mL) and the extracted polyurea solids were separately analyzed for unreacted isocyanate groups using the colorimetric SWYPE™ pads.
Acute aquatic toxicity tests
Test organisms
The test organisms were obtained from a continuously maintained Daphnia magna culture, which was originally purchased from MicroBioTests Inc. (Ghent, Belgium). Cultures of the organism were started from neonates (<24 h post-hatch, 15-20 per vessel) and maintained in 1000-mL glass beakers containing 1000 mL of the MMM. The cultures were fed daily with a concentrated suspension of green microalgae (Chlorella vulgaris). The hardness of the MMM ranged from 219 to 268 mg/L (as CaCO3) over the entire culturing period, and preliminary testing showed the MMM to support acceptable health and survival of D. magna adults and neonates. The cultures were maintained for up to 35 days, and media in each culture was renewed twice per week. With exception of the culture medium used, all other parameters associated with maintenance of the cultures, and selection/placement of the neonates within exposure solutions were as previously described (West et al., 2022a) and in accordance with OECD Guideline 202 (OECD, 2004).
Limit tests
The filtered and pooled WAFs obtained from the water-extractability testing were evaluated for their potential to cause immobilization or sublethal effects of Daphnia magna, using a static limit test procedure based on the OECD Guideline 202: Daphnia sp. Acute immobilization test (OECD, 2004) and the recommendations of the OECD Guidance Document No. 23 on aqueous phase aquatic toxicity testing of difficult test chemicals (OECD, 2019).
Portions (50 mL) of each WAF were divided among four 100 mL glass beakers, five Daphnid neonates were impartially added to each, and the beakers were covered with glass petri dishes. Control exposure solutions were divided among replicate exposure vessels with Daphnids added in exactly the same manner as for the corresponding WAFs.
Definitive dose-response test
A definitive dose-response test was performed for one of the seven test substances (prepolymer H-2; Table 1), which exhibited >20% immobilization in the 48 h D. magna immobilization limit test. Individual water-extractability mixtures were prepared at each of five gravimetric prepolymer loading rates (46.5, 102, 207, 454, and 1000 mg/L) and a control using 1-L beakers and 1-L portions of continuously stirred MMM as described above. After 72 h stirring, the mixtures and control were filtered as previously described, and the resulting WAFs were divided among four replicate 100-mL beakers to which five D. magna neonates were impartially added. Samples of the WAF and control solutions were collected for analyses of NPOC, surface tension, and turbidity at the beginning and end of the exposures as described below. The median effective loading rate (EL50) was determined from the number of immobilized Daphnids at each loading rate and control at 24 and 48 h. A statistical determination of the EL50 value was conducted using a Spearman-Kärber test and CETIS statistical software (v1.8.6.8; Tidepool Scientific Software LLC, McKinleyville, CA, USA). A corresponding median effective concentration (EC50) was estimated by interpolation of the measured NPOC concentration associated with the EL50 value and converting this to a polymer concentration based on % carbon of the proposed hydrolysis product of prepolymer H-2.
Both the limit test and definitive dose-response Daphnid exposures were incubated at 20 ± 1°C under a 16:8 h light:dark photoperiod with no feeding. Temperature, pH, and dissolved oxygen were recorded in a single replicate for each Control and WAF at the beginning and end of the exposures. The Daphnids in each replicate exposure vessel were observed at 0, 24, and 48 h to determine counts of immobile individuals and to record any evidence of sublethal effects (e.g., discoloration, floating, etc.) compared with that in the corresponding Control exposures. Daphnids were counted as immobile if they did not show swimming behavior within 15 s of gentle agitation of the exposure solution.
Analytical methods
Non-purgeable organic carbon (NPOC)
The dissolved NPOC concentrations in filtered samples of the water-extractability reaction mixtures and D. magna exposure solutions were measured using a Shimadzu Model TOC-LCPH/CPN Carbon Analyzer (Shimadzu UK Limited, Milton Keynes, Buckinghamshire, U.K.) interfaced with an autosampler. The operation and calibration of the NPOC analyses were carried out as previously described (West et al., 2022a). The NPOC attributable to the water-extractable components of the test substance and hydrolysis products was derived by subtraction of the NPOC values determined in control solutions (background value) from NPOC measured in the respective test substance exposure samples. The LOQ (limit of quantification) for the measurement was defined as the NPOC concentration in the corresponding control sample determined at each analysis time point. These LOQ values ranged from 0.8 to 4.3 mg/L for all control solutions and associated sampling time points.
The control-corrected NPOC values were normalized to exact prepolymer loading rates of 100 and 1,000 mg/L to enable a direct comparison of water extractability across replicates and between test substances. These normalized NPOC values were divided by the theoretical carbon content of the respective prepolymer (computed for a representative polymer molecule having formula weight approximating the measured Mn) to determine the water extractability (%).
Unreacted isocyanate analyses
The presence of unreacted aliphatic isocyanate groups in the stirred aqueous test substance reaction mixtures, and on the recovered polyurea solids (after 96 h stirring) before and after extraction in tetrahydrofuran (THF), was evaluated using a colorimetric surface wipe test as previously described (West et al., 2022a) and which in this study was specific for detection of aliphatic isocyanates (SKC Inc. SWYPE™, Cat. No. 769-1023; 84, PA, USA). For liquid samples (water or THF extracts), a 1-mL sample was applied directly to the SWYPE™ indicating pad. For solid (polyurea) samples, the SWYPE™ pad was pressed and held firmly against a ∼1-g portion of the wet polyurea solids prior to and after extracting with THF. Any resulting color change of the SWYPE™ pad was compared to a range of colors developed from application of 1-mL portions of calibration standards prepared by dissolving each of the prepolymer test substances in anhydrous acetonitrile (1–10 mg/kg). The approximate limits of detection (LOD) for these analyses of the liquid and solid sample matrices were equivalent to 2 mg/kg as prepolymer test substance for the H-1, H-2, H-3, H-4, and I-1 prepolymers, and 1 mg/kg for the I-2 and I-3 prepolymers.
Turbidity
The Tyndall Effect (https://en.wikipedia.org/wiki/Tyndall_effect) was employed to measure turbidity in the Daphnid exposure solutions, which could indicate presence of suspended polyurea particles and/or dispersed surfactant polymer micelles. A Hach model 2100N turbidity meter (Hach Lange Ltd., Manchester, United Kingdom) was used, which is designed for measurement of turbidity from 0.1 to 4000 NTU (Nephelometric Turbidity Units). A set of Gelex secondary turbidity standards (Hach Lange Ltd.) was used for calibration of the instrument, to convert the percentage of visible light transmittance through the sample to the corresponding NTU output.
Turbidity measurements were performed on the filtered (0.45 and 0.2 µm) and pooled WAFs prepared from the 1000-mg/L loadings (limit tests) for each test substance and on corresponding controls. The measurements were also performed on portions of the WAFs prepared from five loading rates of the prepolymer H-2 (HDI PPG/PEG4350) substance and the corresponding control solution of the definitive EL50 dose-response test. The turbidity in a given test substance WAF sample was compared to that in the corresponding control to determine any contribution to turbidity associated with presence of the test substance/hydrolysis products. Prior to each analysis, the samples were gently agitated to re-suspend any settled solids but to avoid entrainment of air bubbles that could interfere with the measurements.
Surface tension
Measurements of surface tension were performed on the WAF solutions derived from the 1,000-mg/L loadings (limit test) of each test substance and for the WAF derived from the 1,000-mg/L loading rate of the dose-response testing of prepolymer H-2. Measurements were also performed for the corresponding control solutions to determine the contribution of water-extractable test substance/hydrolysis products to surface tension of the WAF solutions. The measurement method followed that described in OECD Guideline 115: Surface Tension-Ring Method (OECD, 1995) and used a surface tension (torsion) balance (White Electrical Instrument Co., Malvern, England) employing the ring method. The instrument was calibrated using distilled water to determine a calibration factor, F, which was derived as follows: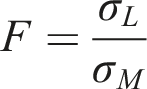
σL = the value given in the literature for the surface tension of water (mN/m) at 20°C and
σM = the measured value of the surface tension of water (mN/m) at 20°C
High-resolution mass spectrometry
Samples of the WAFs derived from 1000-mg/L test substance loadings of the limit tests, and from the 1000-mg/L loading of the definitive D. magna dose-response test for prepolymer H-2, were analyzed by direct infusion time-of-flight mass spectrometry (DI-TOF/MS). These analyses were performed with the intent of elucidating structural information (i.e., molecular weight, polymer backbone structure, end-group functionality) associated with the water-extractable components and/or reaction products of the prepolymer substances. Samples of each WAF and corresponding control solution were diluted 1:1 (v:v) with acetone, gently mixed, and directly infused into an ABSciex TripleTOF 5600+ mass spectrometer (SCIEX UK, Macclesfield, Cheshire, U.K.). Infusions of the relevant control samples were used to identify and exclude any background or artefact ion signals present in both the control and test substance-associated samples. Instrument ionization parameters were optimized to maximize detection of the suspected test substance-associated signals. Details of the instrumentation and analysis parameters associated with the DI-TOF/MS analyses are provided in the Supplemental Information.
Temperature, pH, dissolved oxygen
Temperatures of the stirred test substance reaction mixtures and control solutions, as well as of the Daphnid exposure solutions, were recorded using a digital min/max thermometer, which was placed in a surrogate vessel containing water and incubated alongside the test/exposure vessels. The pH of the water-extractability reaction mixtures and Daphnid exposure solutions were determined using a digital pH meter connected to a combination pH electrode. Dissolved oxygen in the Daphnid exposure vessels was recorded at 0, 24, and 48 h using a digital polarographic dissolved oxygen meter, which outputed the direct dissolved oxygen concentration (in mg/L) and the percentage of the dissolved oxygen air saturation value (% A.S.V.) at the measured sample temperature.
Microscopic examinations
Both mobile and immobile Daphnids were recovered from the WAF exposure solutions of the definitive dose-response test for prepolymer H-2 and corresponding control exposures after 48 h exposure. These were examined using a Lynx EVO stereo zoom microscope (Vision Engineering Ltd. Working Surrey, U.K.) at 20× magnification to record any visible evidence of exposure-related lethal or sublethal effects on the test organisms.
Results
Prepolymer characterizations
The characterizations of molecular weight (number-average; Mn), isocyanate % weight, residual monomer content, viscosity, and calculated octanol-water partition coefficient (log Kow) for the seven prepolymer test substances were reported previously (West et al., 2022b). The domain of these test substance properties covered by the prepolymer test substances is summarized as follows:
Mn: 755–6,632 g/mol
Isocyanate Functionality: 2–4
% wt. NCO: 1.9–11.1
Monomer Content: 105–390 ppm (0.0105%–0.039% wt.)
Viscosity: 105–5,556 MPa*s @ 60°C
Log Kow: 2.2–36.9
Analyses of the prepolymers by GPC used molecular weight calibrations based on polystyrene standards (266–66,000 g/mol), which may not accurately represent the theoretical number-average molecular weight (Mn) values derived from those of the polyol and diisocyanate reactants and/or the actual values derived from the isocyanate titration of the diisocyanate-based prepolymers. The GPC chromatograms indicated a prominent peak corresponding to the expected molecular weight of each prepolymer, for which a corresponding polydispersity value (i.e., Mw/Mn) was obtained. For all seven prepolymer substances, the percentage of eluted oligomeric peak area associated with polystyrene molecular weight cut-offs of 1,000 and 500 g/mol were ≤9% and ≤7%, respectively (see Supplemental Information). Therefore, all of the prepolymers could be characterized as having “low oligomeric content” according to regulatory criteria for reduced risk or exempt polymers (ECCC, 2022; USEPA, 2013).
As described previously (West et al., 2022b) the residual isocyanate monomer content of the prepolymers were precisely measured using a methanol derivatization technique and quantified using HPLC-MS/MS. Conversion of the isocyanate monomer contents of the prepolymer test substances to equivalent diamine concentrations resulted in maximum possible concentrations across all of the 1,000-mg/L test substance loadings (assuming 100% conversion) of 0.27 mg/L as hexamethylene diamine (HDA) and 0.09 mg/L as isophorone diamine (IPDA). Because these concentrations were at least 70-fold less than the reported 48-h EC50 values for D. magna immobilization of HDA (19.8 mg/L; ECHA 2023c) and IPDA (23 mg/L; ECHA, 2023d), residual unreacted diisocyanate monomers were not expected to impact results of the NPOC measurements or the acute toxicity assays, and HDA and IPDA concentrations were not measured.
Aquatic exposure potential
Water extractability
Figure 2 shows the time course of NPOC concentrations versus stirring time for the 1,000-mg/L reaction mixtures (mean ± 1SD, n = 3). The measurements of NPOC in the test substance reaction mixtures at 0, 24, 48, and 72 h indicated that measurable amounts of water-extractable reaction products could be detected within 24 h and attained a maximum concentration within 72 h stirring in the MMM (Figure 2). The mean NPOC concentrations in the prepolymer I-2 and I-3 reaction mixtures showed an unexpected slight decrease at 72-h stirring. Therefore, the WAFs used in the Daphnid immobilization tests were collected, filtered, and pooled after 72 h stirring for all seven test substances, and for the definitive dose-response test with prepolymer H-2. The mean control-corrected NPOC values were divided by the theoretical carbon content (%wt., as determined for polymer formula weight approximating Mn) of their respective prepolymer test substances to give the percentage of original polymer loading that was extractable into water. These mean maximum water-extractability values ranged from 0.2%–66.1% and from 0.02%–59.0% of the 100- and 1,000-mg/L prepolymer loadings, respectively (Table S2, Supplemental Information). Similar to what was observed with the aromatic prepolymer substances, extractability of the aliphatic prepolymer substances into water was inversely correlated with their calculated log Kow values and inversely proportional to prepolymer loading rate. Only the three prepolymers having log Kow ≤9.1 exhibited water extractability >1% of the initial prepolymer loadings. For the remaining four test substances, having calculated log Kow values of 18, 23, 23, and 37, the maximum water extractability ranged from 0.2% to 0.8% and from 0.02 to 0.6% for the 100- and 1,000-mg/L loadings, respectively. Plot of control-corrected non-purgeable organic carbon (NPOC) concentrations (mean ±1 SD, n = 3) evolved from 1,000-mg/L loadings of HDI (solid line) and IPDI (dashed line) based prepolymers when stirred in water over 72 h.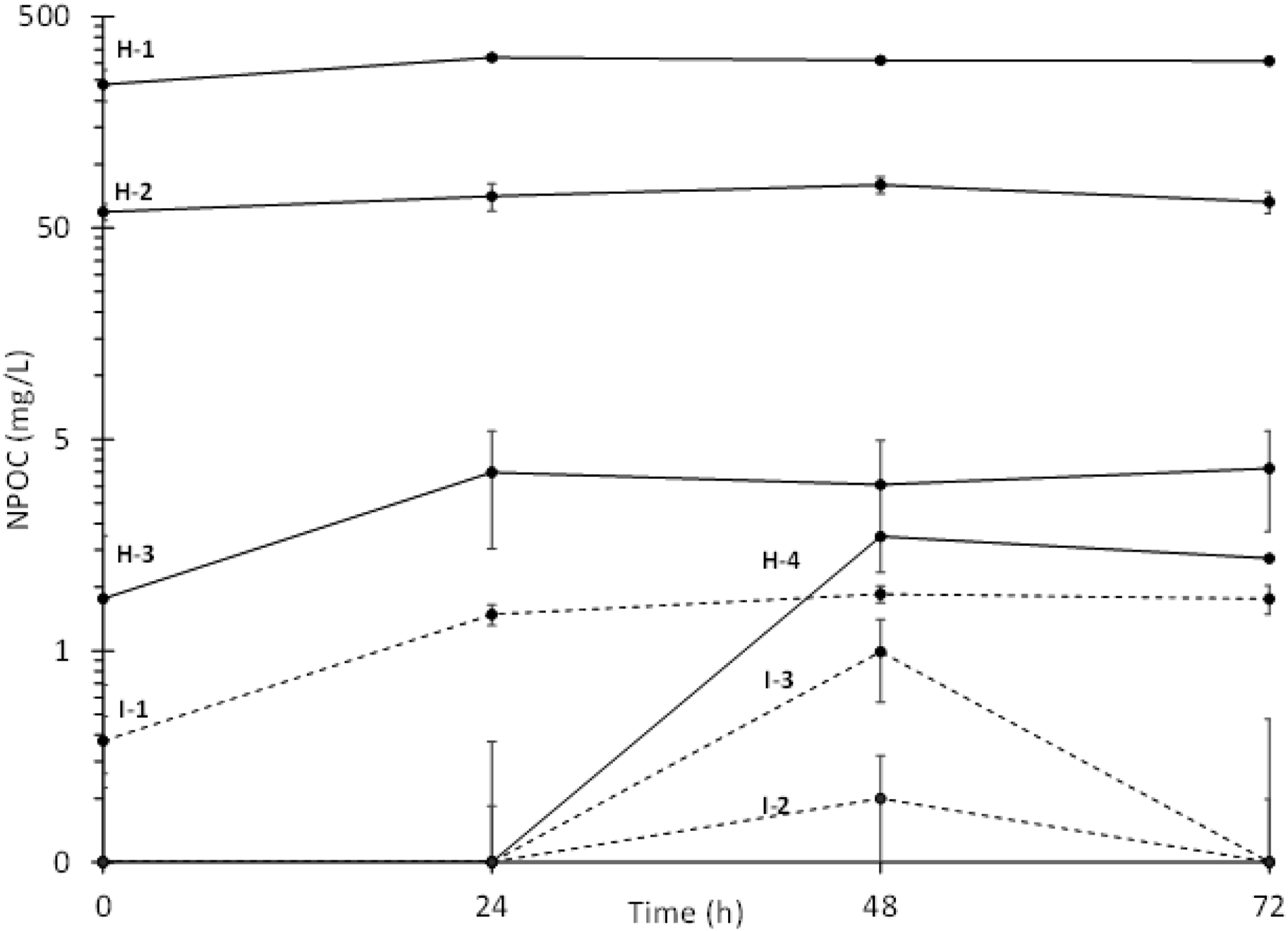
Measurements of NPOC were also performed for the control and WAF exposure solutions prepared at five loading rates (72-h stirring) for the D. magna immobilization dose-response testing of prepolymer H-2. The relationship of NPOC concentration versus loading rate for prepolymer H-2 is shown in Figure 3, which indicates a saturation of extractable hydrolysis product concentrations at/above the 454-mg/L loading rate. Plot of percent Daphnia magna immobilization (vertical bars) plotted on the Y-axis, and non-purgeable organic carbon (NPOC, ꟷ●ꟷ), turbidity (NTU*100, •••+•••) and surface tension (mN*M, - -x- -) plotted on the secondary Y-axis as a function of loading rate (mg/L) for limit tests of each prepolymer and for the dose-response test of prepolymer H-2.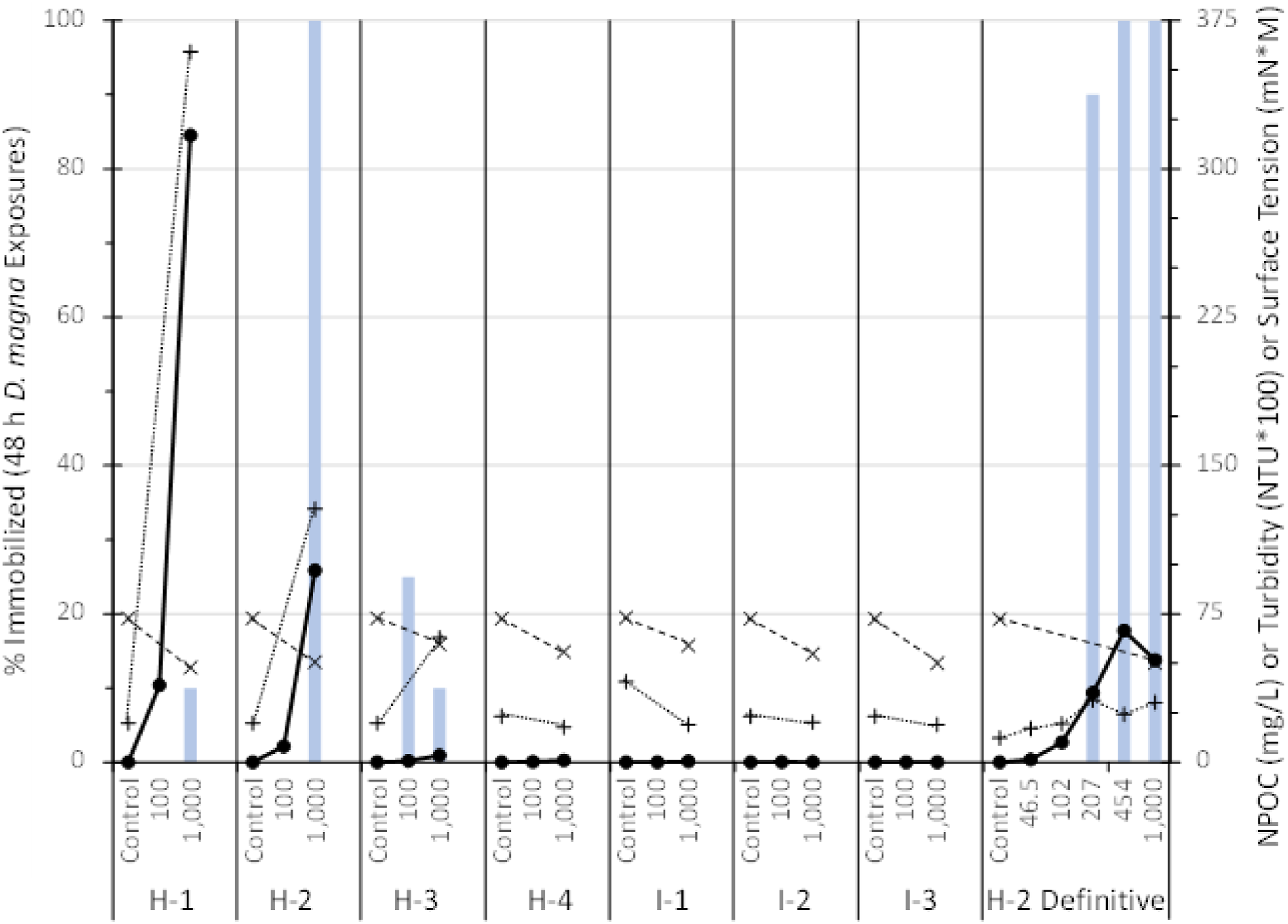
Visual observations of the prepolymer substances behavior when stirred in water were consistent with their measured extractability in water (as measured NPOC concentrations) and calculated log Kow values. While not measured as part of this study, the bulk density of isocyanate prepolymers typically falls within 1.2 ± 0.2 g/cm3, and these substances tend to readily sink in static water. Three of the HDI-based prepolymers (H-1, -2, -4) formed hazy dispersions when stirred in the aqueous MMM, while the reaction mixtures of the remaining four prepolymers maintained clear bulk aqueous phases with globules of prepolymer coalesced and adhered to the beaker walls and stir bars and/or floating at the surface. The pH of the stirred reaction mixtures were negligibly changed (<0.5 units) from that of their initial values over a range of loading rates after 72 h stirring.
Determination of unreacted NCO groups
The colorimetric SWYPE™ analyses provided a convenient semi-quantitative means of determining the relative rates of aliphatic isocyanate end-group reactivity in water across the seven prepolymer substances. The SWYPE™ analyses of stirred/homogenous reaction mixtures at 0- and 72-h stirring indicated that unreacted isocyanate groups were not detected at levels equivalent to >2 mg/kg of any parent test substance. Two of the three IPDI-based prepolymers (I-2, I-3) showed detectable unreacted isocyanate groups in their polyurea solids (before and after extraction with THF) after being stirred in water for 96 h, while neither the third IPDI-based nor all of the HDI-based prepolymers contained detectable unreacted isocyanate groups in their polyurea solids. Full results of these analyses are included in the Supplemental Information.
Acute toxicity to Daphnia magna
Acute aquatic toxicity test
The results of the acute immobilization tests demonstrated that the MMM was suitable for maintenance and growth of the Daphnid neonates, as there were no immobile or lethargic Daphnids found in any of the 48 h Control exposures across all limit and definitive dose-response tests. The pH, dissolved oxygen, and temperature across all of the Daphnid assays were maintained within the ranges recommended in the OECD Guideline 202 (OECD, 2004). To summarize, pH of the WAF exposure solutions was not adjusted during or after the 72-h stirring phase, or before or during the exposure phase, and it remained within pH range of 7.0–7.9, while the Control exposure solutions had pH between 7.0 and 7.8. The dissolved oxygen values associated with all Daphnid exposures were maintained well above the required minimum 3 mg/L and ranged from 8.3 to 9.57 (94%–101% air saturation value). Temperatures recorded within designated WAF and Control exposure vessels ranged from 18.6°C to 20.2°C, while the temperature within the incubator room ranged from 19.4°C to 21.0°C. Thus, the exposure temperature was maintained within the required range of 18°C–22°C, and all other OECD-specified criteria for validation of the OECD 202 test were fully met.
For WAFs associated with four of the seven prepolymers (H-4, I-1, I-2, I-3), no immobilization or sublethal effects were observed in limit tests over 48-h exposures to the WAFs derived from 100- and 1,000-mg/L loadings and their corresponding Control solutions. For these substances, the 24- and 48-h median effective loading rate (EL50) and no observed effect loading rate (NOELR) values were determined as >1,000 mg/L and ≥1,000 mg/L, respectively.
The WAFs associated with the remaining three prepolymers (H-1, H-2, H-3) showed varying degrees of immobilization, as shown in Figure 3. The Daphnids exposed to the 1,000-mg/L WAF of prepolymer H-1 (HDI PEG 750) exhibited 10% immobilization (i.e., two of 20 exposed) after 48 h, where no immobilization or sublethal effects were observed for this same WAF at 24 h or in the WAF derived from 100-mg/L loading after 48 h. Although these exposures had the highest concentrations of dissolved NPOC among all seven substances, the effects on Daphnia magna can be considered as negligible. Thus, for prepolymer H-1, the 24- and 48-h EL50 and NOELR values were determined as >1,000 mg/L and ≥1,000 mg/L, respectively, considering that in aquatic toxicity testing (especially of chronic effects) the concentration associated with effects to 10% or 20% of exposed organisms (i.e., EC10 and EC20) is often used as a surrogate of the no observed effect concentration (NOEC; Beasley et al., 2015).
The Daphnid exposures to WAFs derived from both 100- and 1,000-mg/L loadings of prepolymer H-3 (HDI GLY/PEG/PPG 3500) resulted in extents of immobilization that did not follow a typical dose-response trend. Exposures to the 1,000-mg/L WAF resulted in 0% and 10% immobilization and no sublethal effects after 24 and 48 h, respectively. However, exposures to the 100 mg/L WAF resulted in 5 and 25% immobilization after 24 and 48 h, despite having a mean control-corrected NPOC concentration (0.7 mg/L), which was five-fold less than that in the 1,000-mg/L WAF (3.5 mg/L). Considering this, the immobilizations of ≤25% were considered random/sporadic and not wholly attributed to exposure to dissolved hydrolysis products of the prepolymer. Thus, the EL50 and NOELR values determined for both 24- and 48-h exposures were determined as >1,000 mg/L and ≥1,000 mg/L, respectively.
Daphnid exposures to the WAF derived from a 1,000-mg/L loading of prepolymer H-2 (HDI PPG/PEG 4350) resulted in 100% immobilization after 24 h, whereas no immobilization resulted from exposures to the WAF derived from the 100-mg/L loading. The mean control-corrected NPOC exposure concentrations associated with these WAFs were 8.2 and 97 mg/L, respectively, which would indicate an expected dose-response relationship occurs between the exposure concentrations and percent immobilization. Considering these results from the limit tests, a definitive dose-response test was performed for prepolymer H-2 to more precisely determine the EL50 and NOELR values. The five independently prepared WAFs derived from this substance produced increasing NPOC exposure concentrations with increased loading rates, except that from the 1,000-mg/L loading, which had a slightly lower mean NPOC concentration than that from the 454-mg/L loading (Figure 3). Only the three highest loading rates were associated with immobilization or sublethal effects on the Daphnids, and the percent immobilization correlated with the prepolymer loading rate, although the measured NPOC concentrations appeared to reach saturation at/above the 454-mg/L loading (Figure 3). The Spearman-Kärber test was used to determine a 24-h EL50 value of 423 mg/L with 95% confidence limits of 317 to 564 mg/L, and a 48 h EL50 value of 157 mg/L (95% c.i. = 142–173 mg/L). The 24- and 48-h NOELR were determined empirically to be 207 and 102 mg/L, respectively.
Post-exposure examinations
Immediately following the 48-h exposure interval, both mobile and immobile (dead) Daphnids were retrieved from the prepolymer H-2 WAF and Control exposure solutions and examined under a dissecting stereo microscope. The Daphnids exposed to the Control and 46.5-mg/L WAF appeared completely normal in activity and appearance with no food residues or discoloration visible within the digestive tract. Conversely, the Daphnids recovered from the WAFs of the highest two loading rates, which were 100% immobile at 48 h, exhibited burst carapaces and deformed abdomens with yellow-colored digestive tracts. The immobile Daphnids recovered from the 207-mg/L WAF showed similar signs, while the two remaining live individuals exhibited sublethal effects (lethargy). Daphnids recovered from the 102-mg/L WAF exposure were all similarly mobile compared to those in the Control exposure. However, these examined individuals had accumulations of an off-white colored solid on their antennae.
Surface tension
The 1,000-mg/L WAFs prepared from each of the seven prepolymers had measured surface tensions that were less than those of their corresponding Controls (Figure 3). These surface tension values measured for all seven WAFs ranged from 47.9 to 59.7 mN/m at 20°C, while those in the Control exposure solutions ranged from 72.3 to 73.1 nN/m, and that of the reverse osmosis water used in their preparation was 73.2 mN/m. Although their NPOC concentrations varied from non-detectable to 317 mg/L, all of the WAFs would appear to have contained components that decreased the surface tension of water. In a newly released “Guidance Document for the Notification and Testing of New Chemicals and Polymers,” Environment and Climate Change Canada have defined a “surface-active” substance as one that decreases the surface tension of water to <60 mN/m at 20°C (ECCC, 2022). Therefore, the WAF prepared from each of the seven prepolymer substances contained surface-active substances, which apparently formed upon introduction, mixing, and reaction (hydrolysis) of the prepolymers in water.
Turbidity
Turbidity in the WAF solutions and corresponding Controls was measured to verify that any suspended particles or emulsion formed during stirring of the prepolymers in water were efficiently removed during the filtration step. The turbidity in all Control exposure solutions ranged from 0.12 to 0.41 NTU, and that in the 1,000-mg/L WAFs ranged from 0.18 to 3.6 NTU without added Daphnids (Figure 3). The three prepolymer substances (H-1, H-2, H-3) that produced the highest extractability into water also exhibited the highest turbidity, both before and after preparation of their associated WAFs.
Turbidity measurements were also performed for the five WAFs and Control exposure solution prepared in the definitive dose-response testing of prepolymer H-2. These measurements were performed on samples that did not contain Daphnids, and on the WAFs after 48 h exposure to the Daphnids. These measurements showed increased turbidity with increased loading rates from 46.5 to 207 mg/L and a leveling in turbidity values for the 207-, 454-, and 1,000-mg/L WAFs. As could be expected, turbidity was also slightly increased across all of the WAFs and Control solutions following exposure to the Daphnids, which could be attributed to depuration and/or microbial growth.
Mass spectrometry
The direct infusion of the 1,000-mg/L WAFs and associated Controls into a high-resolution (TOF) mass spectrometer produced qualitative evidence for the molecular weight, functionality, and structural features of the dissolved constituents and/or hydrolysis products of the seven prepolymers. Spectral signals that were observed in the Control solutions (Figure 4(a)) were automatically discounted from the spectral signals found in corresponding WAF solutions. For all seven prepolymers, signals were obtained that were consistent with multiply-charged aliphatic amine-terminated polymer species having molecular weights and polyol backbone repeat units corresponding to those of their parent prepolymers. It should be noted that none of the obtained spectral signals were particularly intense, due to the relatively low concentration of dissolved constituents (as NPOC) and the distribution of these polymeric constituents across a range of molecular weight. No meaningful/interpretable signals were identified in the spectral scans collected under the negative polarity mode. The DI-TOF/MS scans performed in positive polarity mode revealed interpretable signals for components of the WAFs derived from all seven prepolymer substances, which can be summarized as follows: • Signals (m/z) associated with multiply-charged polymer species, with charge (z) corresponding to the nominal isocyanate functionality of the parent prepolymer. • Signals (m/z) corresponding to molecular mass (m) of approximately 40% or more of the parent prepolymer Mn values. • A homologous series of signals having mass/charge (m/z) differentials indicative of the propylene oxide (m/z = 58), tetramethylene oxide (m/z = 72), and adipate (m/z = 79) repeat units of the respective prepolymer backbones. Representative mass spectra obtained from direct infusion time-of-flight mass spectrometric (DI-TOF/MS) analyses of a control exposure solution (a) and the water-accommodated fraction (WAF) associated with 1,000-mg/L loading of prepolymer H-2 (b) and a daughter ion spectrum of the detected constituent having m/z = 427 (c).
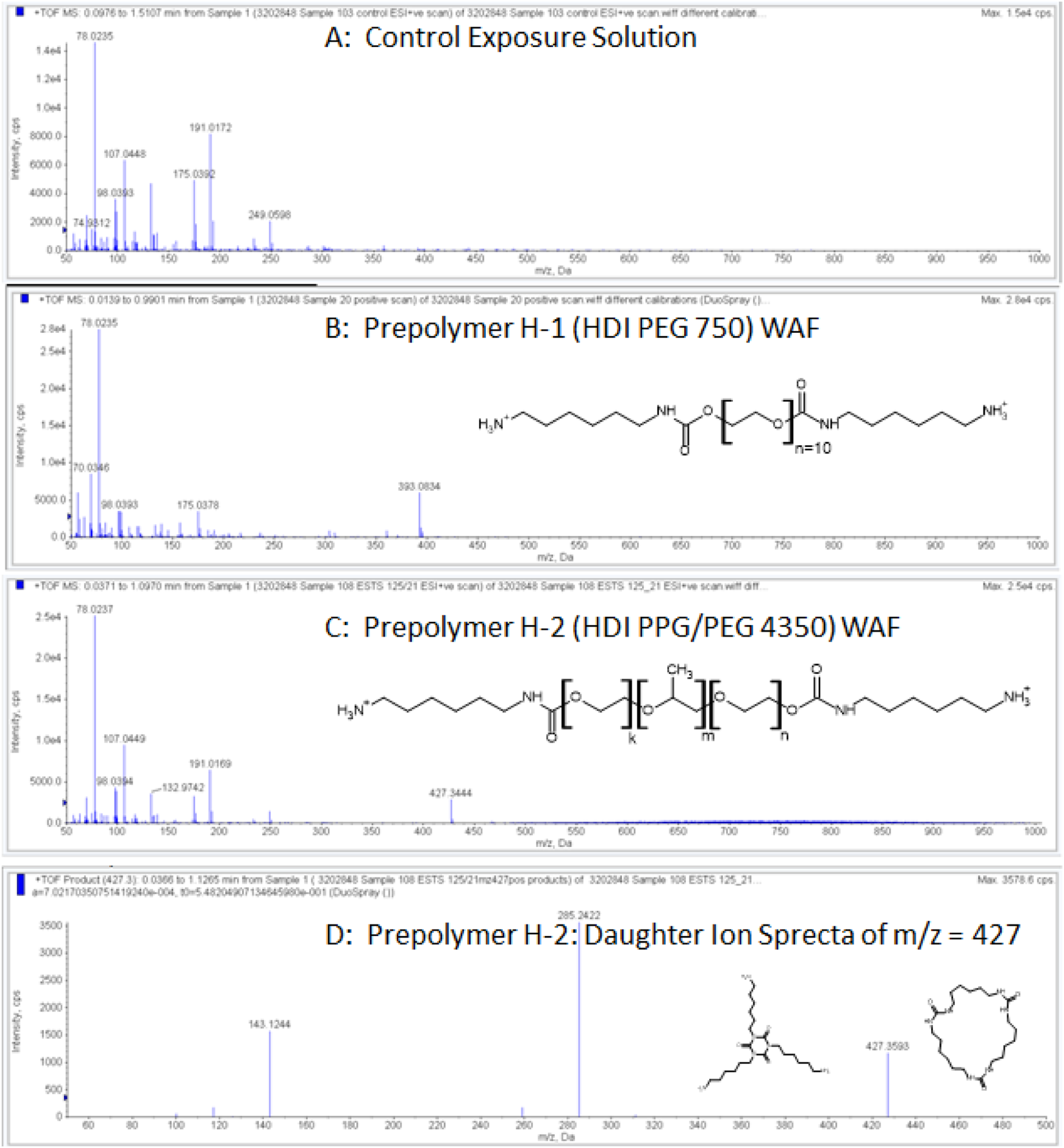
Further elaboration and illustrations of the DI-TOF/MS information obtained can be found in the Supplemental Information. Since only the WAFs derived from prepolymer H-2 (HDI PPG/PEG 4350) were associated with significant immobilization of the Daphnids, the DI-TOF/MS signals associated with this prepolymer are further elaborated as follows.
A diffuse band of signals was observed, which spanned the m/z range of ∼480–990 Da (Figure 4(b)). These signals indicated doubly charged (diamine) polymer species, and they represented a distribution of polymer homologues ranging in nominal mass from ∼970 to 1,880 Da. Polymer constituents of this MW range would be associated with the lower end of the molecular weight distribution of bis-(6-aminohexyl carbamate) derivatives of the PPG/PEG 4000 polyol. A second signal of interest was found at m/z = 427.3, indicative of a singly charged [M+H]+ species. This molecular species could be further fragmented in the MS source to yield the daughter ion spectrum shown in Figure 4(c), wherein the molecular ion and fragment signals could be attributed to one (or both) of the two plausible isomeric structures shown (Figure 4(c)). The first being the tri-amine hydrolysis product of the isocyanurate trimer of HDI (structure shown at left of Figure 4(c)). The second isomer is a cyclic urea, which could be formed from three molecules of an HDI amino-isocyanate species that could theoretically occur as an intermediate product of HDI hydrolysis.
The obtained spectral information, albeit associated with rather weak signals, collectively supports the conclusion that the water-extractable hydrolysis products of HDI- and IPDI-based prepolymer substances are the aliphatic amine-terminated analogs of their parent isocyanate-terminated polymers, for which the molecular weight, functionality, and original polymer backbones are conserved.
Discussion
The findings of this study provide valuable information for assessing the aquatic fate, exposure, and potential effects of the aliphatic diisocyanate-based prepolymers and can extend the understanding of these properties already reported for the related aromatic diisocyanate prepolymers.
Aquatic hazard potential
Most importantly, the results of this study indicated that the water-extractable reaction products of the aliphatic prepolymers are practically non-toxic to Daphnids on an acute exposure basis, as the 48-h EL50 were >1,000 mg/L with the one exception of EL50 = 157 mg/L. According to the current globally harmonized system (GHS) for hazard classification and labeling of chemicals, acute aquatic hazards are not classified when measured median effective concentrations (EC50, EL50) exceed 100 mg/L (UN, 2023). It would therefore appear that the regulatory concern for potential aquatic hazard of both aromatic and aliphatic diisocyanate-based prepolymers, owing to their potential to become cationic polymers in the environment (ECCC, 2022; Ge, 2021; US EPA, 1997; US EPA, 2013), is not warranted.
In the few instances where immobilization of Daphnids was observed in this study, the extents of immobilization could not be directly linked to the molecular weight, structure, or charge density of their purported hydrolysis products. While charge density of polycationic prepolymers was suggested to be the key driver of their aquatic hazard potential (Boethling and Nabholz, 1997), the expected polymeric hydrolysis product of prepolymer H-2 has the lowest charge density (0.7% amine-N) and highest degree of Daphnid immobilization of the three prepolymers exhibiting immobilization in the Daphnid limit tests. Conversely, the expected polymeric hydrolysis product of prepolymer H-1 had the highest charge density (4% amine-N) and only 10% immobilization occurred in the 1,000-mg/L limit test. Whereas the WAF of each prepolymer was shown to be surface-active, this property alone cannot be linked to the observed immobilization of the Daphnids, as WAFs that did not cause Daphnid immobilization had surface tensions that were essentially the same as those that did cause immobilization (Figure 3). Roberts (2000) showed that aquatic toxicity of surfactants was most strongly correlated with structure/properties of the surfactant molecule (e.g., log Kow) and not to the lowered surface tension of water occurring in their presence. It could be expected that the polymeric hydrolysis products of prepolymer H-2 are more toxic than those of H-1, as the former has a higher calculated log Kow (2.8) than that of the latter (−0.66). The expected hydrolysis product of prepolymer H-3 has a relatively large calculated log Kow of 6.3, which may indicate that toxicity to the Daphnids was limited by its low water solubility.
Summary of predicted properties of the proposed amine-terminated hydrolysis products of the aliphatic diisocyanate-based prepolymer substances.
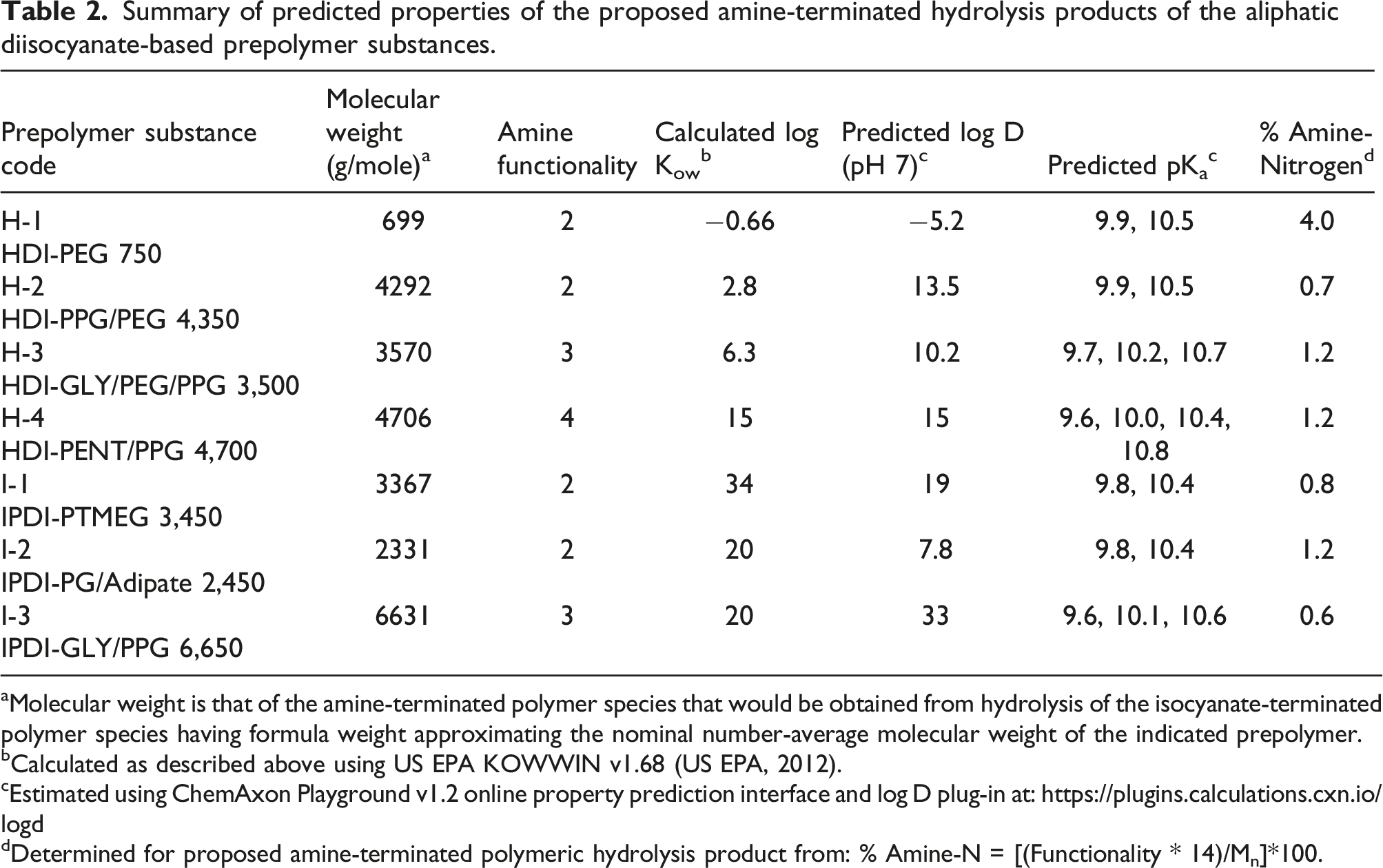
aMolecular weight is that of the amine-terminated polymer species that would be obtained from hydrolysis of the isocyanate-terminated polymer species having formula weight approximating the nominal number-average molecular weight of the indicated prepolymer.
bCalculated as described above using US EPA KOWWIN v1.68 (US EPA, 2012).
cEstimated using ChemAxon Playground v1.2 online property prediction interface and log D plug-in at: https://plugins.calculations.cxn.io/logd
dDetermined for proposed amine-terminated polymeric hydrolysis product from: % Amine-N = [(Functionality * 14)/Mn]*100.
Aquatic exposure
The results obtained from stirring these seven prepolymers in water were indicative of a so-called “reactive dissolution” process, which is expected to occur when a bulk diisocyanate (organic) phase is dispersed into water (Yakabe et al., 1999). When the terminal isocyanate groups of the diisocyanate molecules occurring at the surface of the organic diisocyanate phase are hydrolyzed, the polar amine-terminated molecules that form would tend to be pulled into solution within the stirred bulk aqueous phase where the remaining isocyanate group can be further hydrolyzed to form the diamine. If the amino-isocyanate intermediate is not sufficiently soluble in water, and/or if the aqueous phase is not vigorously mixed, the initially formed amino group will be reacted with a neighboring isocyanate group to form a urea linkage within the bulk diisocyanate phase. In the presence of excess water, these concurrent and competing hydrolysis and urea formation reactions are repeated until all isocyanate groups are reacted, and ultimately form a mixture of dissolved diamine species and an insoluble polyurea. The relative yields of these hydrolysis end-products depend on many factors, including reactivity, hydrophobicity, and viscosity of the parent diisocyanate, the starting mass ratio of diisocyanate and water, and the degree of mixing (sheer). The same general reactions and controlling factors apply for reactive dissolution of diisocyanate-based prepolymers as well. Since most polyisocyanate substances, whether monomers or polymers, are highly hydrophobic the predominant products resulting from their heterogeneous contact with water are the insoluble polyureas (Yakabe et al., 1999). Only when the polyisocyanate is fully dissolved in the aqueous phase can a quantitative yield of the corresponding polyamine be achieved (Neuland et al., 2022).
The isocyanate group functionality (f) and corresponding % amine-N content of the proposed polymeric hydrolysis products (Table 2) had little influence on extractability of the prepolymers in water. For example, the prepolymer I-2 (f = 2) and I-3 (f = 3) have essentially the same calculated log Kow value of ∼23 and very similar water extractability (0.2% and 0.3%) despite having different theoretical % amine-N values of 1.0 and 0.6, respectively. Compared with prepolymers I-2 and I-3, prepolymer H-4 (f = 4) has a slightly lower log Kow value of 18 and % amine-N value of 1.3, yet its water extractability (0.3%) is only slightly higher than that of prepolymer I-1, which has a similar % amine-N of 0.8 and a much higher log Kow of 37. Thus, the % amine-N of the envisioned hydrolysis products of these aliphatic diisocyanate-based prepolymers did not play a significant role in differentiating their extractability in water.
The aliphatic diisocyanate-based prepolymers showed increased extractabilities in water compared with those reported by West et al. (2022a) for the aromatic diisocyanate-based prepolymers. For example, prepolymer pairs H-1/T-6, H-2/T-4, and I-1/M-1 were synthesized from the same polyols and had similar calculated log Kow values (see West et al., 2022b). As shown in Figure 5, the water extractabilities of the aliphatic based prepolymers (H-1, H-2, and I-1) were notably larger than those of the corresponding aromatic prepolymer. This can be explained on the basis of the proportionately higher water solubilities (or conversely, smaller log D values) of aliphatic amine-terminated polymers compared with those of hypothetical aromatic amine-terminated polymer hydrolysis products. The conversion of the isocyanate group to the corresponding amine results in about the same reduction in the calculated log Kow value regardless of whether the isocyanate group is aromatic (TDI, −2.1; MDI, −1.5) or aliphatic (HDI and IPDI, −1.43). However, the aromatic amine groups have pKa of ≤5 (Spence and Plehiers, 2022), while the primary aliphatic amine groups are estimated to have pKa > 9.6 (Table 2). Thus, the aliphatic amine-bearing hydrolysis products will occur predominantly in the highly soluble protonated (–NH3+) form while the aromatic amine-bearing hydrolysis products will occur in the predominantly neutral form at pH ∼ 7 of the water-extractability experiments. Since the protonated amine forms cannot react with neighboring isocyanate groups and are also highly water-soluble, it could be expected that urea-forming reactions (i.e., amine + isocyanate => urea) are less prevalent within and/or at the surface of the isocyanate prepolymer phase during heterogeneous mixing of the aliphatic diisocyanate-based prepolymers in water. Plot of maximum water extractability (%) versus calculated log Kow for aliphatic (circles) and aromatic (triangles, West et al., 2022a) diisocyanate based prepolymer substances occurring over 96-h stirring at 100-mg/L (●, ▲) and 1,000-mg/L (o, Δ) initial loadings. Prepolymer pairs H-1/T-6, H-2/T-4, and I-1/M-1 were prepared from the same polyol backbones. Dashed line represents 2% water-extractability threshold (ECCC, 2022).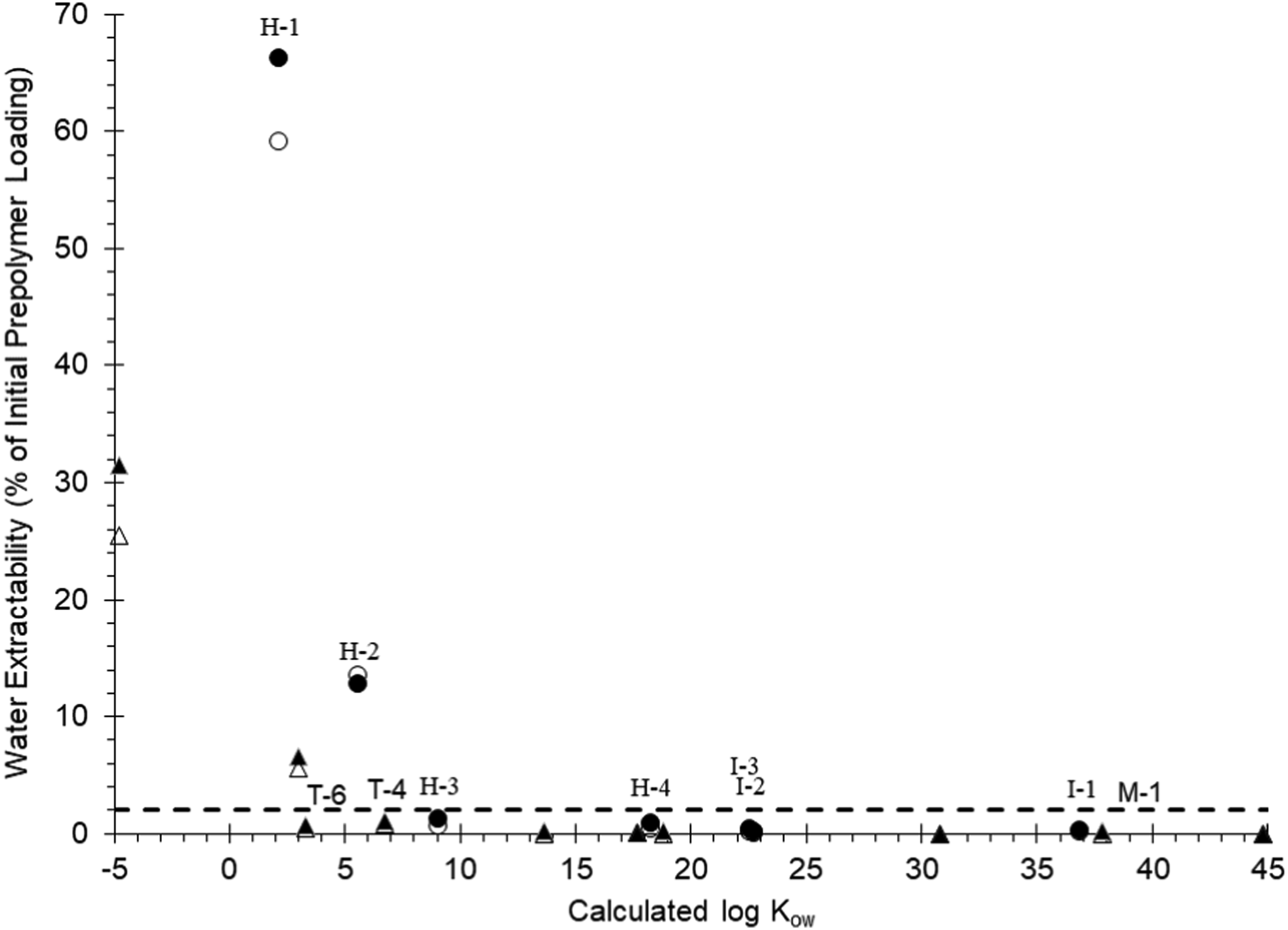
Using the colorimetric SWYPE™ test kit, the isocyanate functional groups of the prepolymers were shown to be essentially fully reacted in the aqueous phase and were mostly reacted away in the organic (solid) phase after 96-h stirring. The undissolved prepolymer constituents and hydrolysis products of prepolymers I-2 and I-3, which remained associated with the reaction mixture solids, contained a small amount of unreacted isocyanate groups even after 96 h of stirring in water. While not directly investigated here, the presence of suspended particles or micelles having unreacted isocyanate groups could contribute to physical toxicity as speculated above. If micelles are formed from dispersion of the prepolymers in water, the reactive isocyanate groups could remain within the core of the micelles and thus be protected from rapid reaction with water and with the colorimetric indicator of the SWYPE™ pads. This principle is the basis for water-based polyurethane dispersion (PUD) formulations that form a cured film/coating upon evaporation of the water solvent (Tennebroek et al., 2018). The occurrence of unreacted isocyanate groups associated with the solids isolated from two of the IPDI-based prepolymers, and not with any of the HDI-based prepolymers, could be explained by the lesser reactivity of the secondary –NCO group of IPDI compared with that of the primary –NCO group of HDI (Spence and Plehiers, 2022). The rate of water diffusion into the organic (prepolymer) phase could also play a role in determining the rate of isocyanate hydrolysis in the organic phase, as indicated for polymeric methylenediphenyl diisocyanate (pMDI) in the work of Yakabe et al. (1999). The viscosity of the prepolymer in this case plays a role in governing the rate of water infusion into the bulk prepolymer phase, and effusion of prepolymer into the aqueous boundary layer. As shown in Table 1, the IPDI-based prepolymers had much larger values of dynamic viscosity (1,090–5,556 MPa*s) than those of the HDI-based prepolymers (105–481 MPa*s), so viscosity likely also played some role in differentiating the extents of isocyanate conversion among the HDI- and IPDI-based prepolymers.
Prepolymer hydrolysis products
Evidence from the present study demonstrated that polyureas were indeed the predominant hydrolysis end product for the highly hydrophobic (e.g., log Kow ≥9.1) aliphatic diisocyanate-based prepolymers. By analogy to the aromatic polyureas formed from hydrolysis of aromatic diisocyanates (Sendijarevič et al., 2004), these aliphatic polyureas were expected to be resistant to hydrolytic and mechanical degradation and would occur as recalcitrant components of soil or sediments if the prepolymers were to be released into the environment. For the most hydrophilic of the tested prepolymers (H-1, log Kow = 2.2), about 66% of the original prepolymer mass could be converted into the presumed polymeric diamine hydrolysis product. It is important to note that in the present study (Figure 5), as well as in prior work of West et al. (2022a) and Yakabe et al. (1999), the yield of water-soluble reaction products was inversely proportional to the loading rate in water. Therefore, while testing of these prepolymer substances at higher loading rates (>1,000 mg/L) could yield higher concentrations of soluble reaction products, the yield as a fraction of polymer loading rate (i.e., the water extractability) would be lower.
The analyses of the WAF exposure solutions by DI-TOF/MS constitute the first known/reported attempt to identify the structure/identity of the water-soluble hydrolysis products formed from the aliphatic diisocyanate-based prepolymers. It has been long asserted that hydrolysis of isocyanate-based polymers has the potential to form amine-terminated and potentially cationic polymers in the environment (US EPA, 1997). Although the spectral signals obtained did not provide robust evidence for occurrence of amine-terminated polymer species over the full molecular weight range of each parent prepolymers, this was likely due to the low concentration of polymer present in the sample and the expected decreasing water solubility of these species with increasing molecular weight. Alternative mass spectrometric techniques, such as MALDI-TOF could perhaps provide more sensitive and comprehensive evidence to confirm the presence of these high molecular weight species in the WAFs of these prepolymers. It is not unexpected that contact of the prepolymers with water over 96 h would result in hydrolysis of the terminal isocyanate groups while leaving the polyol backbones intact. It has been shown that the polyether polyols are resistant to hydrolysis, and only the lower molecular weight homopolymers (i.e., PEG and PPG) are easily biodegraded (Schupp et al., 2017; Tisler et al., 2021). Hydrolytic degradation of the polyester polyol backbones (e.g., 1,2-propanediol-adipate) would be expected only under elevated pH (≥9) and temperature, and would also be limited by their low water solubility.
The structure associated with the signal m/z = 427.3 detected in the WAF of prepolymer H-2 (Figure 4(c)) is most likely to be the tri-amine hydrolysis product of the isocyanurate trimer formed from HDI, as depicted by the left structure in Figure 4(c). The second (right) structure could occur if an amino-isocyanate intermediate of HDI hydrolysis were formed in sufficient yield, with three of these intermediate molecules reacting with each other to form the cyclic urea product. Since the H-2 prepolymer had an unreacted monomer content of only 310 ppm (and theoretical maximum of 30 mg/L in the 1,000-mg/L WAF), the formation of this cyclic urea product at sufficient yield to be detected by DI-TOF/MS seems unlikely. Conversely, the HDI isocyanurate trimer species (i.e., CASRN 28182-81-2) could have been formed under the elevated temperature associated with the final vacuum stripping of excess HDI during synthesis of the prepolymer (Parodi, 1989). The acute 48-h D. magna EL0 and EL50 values of 25 and 127 mg/L were reported for the HDI trimer (ECHA, 2023e) using testing procedures similar to those described here. Since the EL0 and EL50 values for prepolymer H-2 determined in this study were 102 and 157 mg/L, respectively, the HDI trimer species would have to be present at very high concentrations to account for the immobilization exhibited in the WAF of prepolymer H-2.
Regulatory aspects
Aquatic hazard assessment
The results of this study have potential implications for how the diisocyanate-based prepolymer substances can be evaluated within regulatory frameworks in the future. As stated above, none of the diisocyanate-based prepolymers evaluated in this and in the preceding study (West et al., 2022a) would be classified under GHS on the basis of their acute EL50 values. However, the question remains about how to properly evaluate or predict the acute aquatic toxicity of their proposed aliphatic amine-terminated polymer hydrolysis products. Diisocyanate substances (including prepolymers) have been presumed to be highly hazardous because of their potential to form cationic (amine-terminated) polymers in the aquatic environment (ECCC, 2022; Ge, 2021; US EPA, 1997). The US EPA defines these “polycationic polymers” of concern as any polymer molecule having multiple positive charges, being soluble or dispersible in water, with molecular weight >300 g/mol, and amine functional group equivalent weight (FGEW) >400 g/mol (US EPA, 2010). The US EPA guidance for assessing these polymers broadly defines them as those possessing permanently cationic functional groups (e.g., phosphonium, sulfonium, and quaternary ammonium cations), and those that are reasonably anticipated to become cationic (e.g., all primary, secondary, and tertiary amines). This latter group of “potentially cationic polymers” are those for which their occurrence as cationic functional groups is pH-dependent (i.e., are not permanently cationic) and is stated to include the expected hydrolysis products of all isocyanate substances (US EPA, 1997). Aromatic amine-terminated polymers are excluded from this group of potentially cationic polymers of concern (Boethling and Nabholz, 1997), presumably because of the much lower pKa values and associated potential to become ionized in the environment. The assessment of these aliphatic amine-terminated polymers is complicated by the several different quantitative structure-activity relationships (QSAR) that have been developed and recommended for predicting acute and chronic aquatic hazards to fish, invertebrates, and algae from the structure and/or properties of broadly defined class of “cationic polymers.” For example, the US EPA ECOSAR software (v1.11, US EPA, 2012) includes substance class-specific QSAR equations for aliphatic amines and for polycationic polymers. The carbamate esters QSAR of the ECOSAR software is also applicable, but a much lesser contribution of this functional group of the prepolymer hydrolysis products to acute aquatic toxicity is indicated than for the aliphatic amine and polycationic polymer attributes. A fourth QSAR for cationic surfactants is also available but can only be implemented for cationic surfactants having varied numbers of methylene carbon units within a presumed unsaturated hydrocarbon backbone. The US EPA’s interpretive assistance document for assessment of polymers recommends use of the “polycationic polymer” QSAR equations for predicting aquatic toxicity of polycationic polymers as a function of polymer backbone (carbon-, silicon-, or natural-based) and % amine-N content of the polymer (US EPA, 2013). Although it is stated that toxicity of the polycationic polymers has little dependence on whether the cationic functional group is permanent (i.e., quaternary ammonium) or pH-dependent, pendant or occurring within the polymer backbone, the dataset used to develop these QSARs for “polycationic polymers” does not include results for carbon-based polymers having pendant primary aliphatic amine groups (Boethling and Nabholz, 1997). Rather, much of the data used to develop these QSAR equations are derived from tests with quaternary ammonium- and secondary amine-bearing polymers having much higher molecular weights and charge densities (% amine-N) than those of the aliphatic amine-terminated polymers of interest to this study.
The proposed polymeric hydrolysis products of the aliphatic diisocyanate prepolymers can be described generally as linear or branched, neutral and non-ionizable polymer chains that are terminated with polar/hydrophilic primary amine groups, and for which the polyol type and molecular weight of the polymer chains impart varying degrees of hydrophobic character. These amphiphilic structural characteristics are analogous to those of aliphatic “fatty” primary amines and diamines, and of the polyether polyamine substances (Huntsman Corporation, 2023), where hydrophobic character (i.e., calculated log Kow values) similar to that of the prepolymer hydrolysis products can be imparted by varied length of their aliphatic hydrocarbon or polyether chains. The predicted EC50 values for 48-h D. magna immobilization from the alkyl amines and polycationic polymers QSARs were compared to reported measured values for several alkylamine, alkyl diamine, and polyether polyamine substances approximating the log Kow and % amine-N values of the proposed amine-terminated polymer hydrolysis products of this study (see Supplemental Information). From this comparison, it was found that the polycationic polymer QSAR, which is based on correlation of % amine-N with acute EC50 values for (mostly) quaternary ammonium compounds, consistently predicts EC50 values that are lower (more toxic) than the measured values. One notable exception is where this polycationic polymer QSAR predicts the hydrolysis product of prepolymer H-2 to be less toxic (EC50 = 115) than the measured EC50 = 41 mg/L (derived from the interpolated NPOC concentration at EL50 = 157 mg/L). The polycationic prepolymer QSAR also generates a predicted EC50 value that is over 1,000-fold lower for the hydrolysis products of prepolymer H-1 (EC50 = 0.1 mg/L) than that of H-2 (EC50 = 115 mg/L), whereas results from the Daphnid immobilization limit tests of this study would indicate the hydrolysis product of prepolymer H-2 to be about 10-fold more toxic than that of prepolymer H-1. Based on this limited comparison, the polycationic polymer QSAR (Boethling and Nabholz, 1997; US EPA, 2013) should not be applied in predicting the acute aquatic toxicity of the hydrolysis products of the aliphatic diisocyanate-based prepolymers.
An ion-trapping mechanism has been proposed to explain the pH-dependent aquatic toxicity of alkyl amine substances (Baumer et al., 2017; Finlay and Callow, 1997), whereby the neutral amine species is more rapidly transported across cell membranes than is the cationic form, and the neutral amine becomes positively charged and “trapped” within the lower pH environment of the cytosol. Since such a preferential uptake mechanism would not occur for the “permanently cationic” polymer species, it would seem mechanistically inappropriate to assess aquatic hazard of the pH-dependent and permanently cationic polymers as a single substance class. The aliphatic amine QSAR appears to generate predicted EC50 values, and the expected decrease in EC50 with increasing calculated log Kow values, which better matches the measured EC50 values and correlation with log Kow for aliphatic amine-terminated polymer substances. The “polyquaternium” cationic polymers reviewed by Boethling and Nabholz (1997), Connors et al. (2023), Hansen et al. (2023), and Environment and Climate Change Canada (ECCC, 2020) can have molecular weights in excess of 10,000 g/mol and have acute aquatic EC50 values much lower than those indicated for the amine-terminated polymeric hydrolysis products of the diisocyanate-based prepolymers. Based on the evidence described above, the aquatic hazard potential of hydrolysis products from aliphatic diisocyanate-based prepolymers should not be assessed in the same way as polymers bearing the permanently cationic quaternary ammonium functional group. For example, Environment and Climate Change Canada has assessed the potential health and environmental risks of aliphatic amines (ECCC, 2021) and the quaternary ammonium polyamines (ECCC, 2020) as separate chemical classes as well. The results of the present study, and from reported aquatic hazards of closer structural analogs (such as summarized in Supplemental Information) may be useful in refining QSARs that are better suited to the diisocyanate-based prepolymer substances and their associated hydrolysis products.
Aquatic exposure assessment
The extent to which polymers can be dissolved or dispersed in water can be used as a screening criterion upon which regulatory decisions about the need for additional testing of aquatic fate and hazard properties can be made. For example, polymers having water extractability >2% are required to be tested for hydrolysis as a function of pH, ready biodegradability, and acute aquatic toxicity (to algae, or to the most sensitive among fish, Daphnia, or algae) under new polymer notification requirements (Schedules 10, 11) of Environment and Climate Change Canda (ECCC, 2022). The testing of water extractability and of these other environmental fate and aquatic hazard parameters could be made unnecessary and avoidable for the diisocyanate-based prepolymers if a polymer property or structural descriptor were identified at which water extractability of below 2% could be conservatively assured for this polymer class. In the previous evaluation of the aromatic diisocyanate-based prepolymers (West et al., 2022a), it was found that negligible extractability in water (<1% of prepolymer loading) occurred for those having log Kow >10. For the aliphatic diisocyanate-based prepolymers of this study, this same threshold of log Kow >10 is supported even though the polymeric hydrolysis products of the aliphatic prepolymers appeared more water-soluble than those of their aromatic counterparts (Figure 5). The prepolymer H-3, having a calculated log Kow of 9.1, showed a maximum water extractability of 1.3% while the water extractabilities of all other prepolymers having higher calculated log Kow were <1%. Correlations of water extractability with other prepolymer parameters, such as the predicted logarithm of the octanol-water distribution coefficient (log D; ChemAxon, 2023) and calculated log Kow values of the amine-terminated polymer hydrolysis products, and calculated log Kow values of the hypothesized amino-isocyanate hydrolysis intermediates, did not reveal better-defined trends or thresholds to delineate the 2% water extractability cut-off for either the aliphatic prepolymers or the combined aliphatic and aromatic prepolymer datasets (data not shown). Although such parameters may be better linked to the mechanism of reactive dissolution occurring when diisocyanate-based prepolymers are mixed with water, the available methods for estimating log D of ionizable substances are not yet implemented within regulatory assessment frameworks, at least to the same extent to which the calculation of log Kow via the KOWWIN software (US EPA, 2012) is applied and accepted. According to the US EPA ECOSAR QSAR definition document for aliphatic amines, the water solubility of aliphatic amine substances having calculated log Kow >8 is expected to be below the concentration at which acute or chronic toxicity can typically occur (US EPA, 2012). Therefore, both aquatic exposure and hazard are expected to be negligible for aliphatic diisocyanate prepolymers having calculated log Kow >10.
Conclusion
Both the aliphatic and aromatic diisocyanate-based prepolymers having log Kow >10 exhibit negligible extractability in water, and all of these substances evaluated in the present and prior study are practically non-toxic to Daphnia magna regardless of variation in their structural and physical-chemical properties. Aside from some apparent potential to contribute to physical toxicity, the water-extractable hydrolysis products for the evaluated set of aliphatic diisocyanate-based prepolymers exhibit low potential hazard with the aquatic invertebrate, Daphnia magna. Considering the diversity in structural and physical-chemical properties occurring across these seven prepolymers, and of the ten aromatic-based prepolymers evaluated previously (West et al., 2022a), it can be concluded that this combined grouping of water-reactive isocyanate prepolymers collectively presents a very low hazard to the aquatic environment.
From a scientific testing and assessment perspective, the present study and its results illustrate the necessity for attention to test substance properties and adjustment of standardized test methods to suit them. The OECD “Guidance Document on Aqueous Phase Aquatic Toxicity Testing of Difficult Test Substances” (OECD, 2019) describes many such adjustments to standardized testing procedures (e.g., OECD 120 and OECD 202) which were implemented here. The careful preparation of WAF exposure solutions and associated control solutions, along with supplemental analyses of NPOC, turbidity, surface tension, and DI-TOF/MS enabled a very precise evaluation and comparison of aquatic hazard potentials across these unique, reactive polymer substances. Yet even with the precautions taken, the influence of physically mediated toxicity could not be excluded. The recently released ECCC guidance on new substance notification for chemicals and polymers (ECCC, 2022) also includes very detailed and specific guidance for evaluation of water-reactive polymers. Such guidance should be consulted carefully when designing and implementing testing strategies for assessing environmental exposure and hazard properties of polymer substance categories. The observed minimal effects of these substances upon acute exposures to D. magna, along with analysis of applicable QSAR prediction methods for the expected aliphatic amine-terminated polymer hydrolysis products, indicated that the high concern for aquatic hazard of isocyanate-based polymers is not warranted. It is now apparent that such concerns for permanently cationic polymer substances should not also be attributed to aliphatic amine-terminated polymers for which cationic character is pH-dependent.
From a product stewardship perspective, the results of this and prior work could provide a basis for grouping of aliphatic and aromatic diisocyanate-based polymeric prepolymers into a single class of water-reactive polymer substances, for which regulatory evaluations of environmental fate, exposure, and hazard can be completed more efficiently. The findings from this study can also aid in the future design and commercialization of new diisocyanate-based prepolymers having minimized impacts on the environment. Considering the wide variety of substances evaluated in the present and previous work (West et al., 2022a), a clear weight of evidence is now available to demonstrate low aquatic hazard potential and predictable aquatic exposure potential for this polymer substance category.
Supplemental Material
Supplemental Material - Exploring structure/property relationships to health and environmental hazards of polymeric polyisocyanate prepolymer substances-3. Aquatic exposure and hazard of aliphatic diisocyanate-based prepolymers
Supplemental Material for Exploring structure/property relationships to health and environmental hazards of polymeric polyisocyanate prepolymer substances-3. Aquatic exposure and hazard of aliphatic diisocyanate-based prepolymers by Robert J West, Rebecca Jarrom, Mitch Kelly, Glenn S Simon and Stephanie J Snyder in Toxicology and Industrial Health.
Footnotes
Acknowledgments
The authors are grateful to (in alphabetical order): W. Dayoub (Vencorex US Inc.), S. Grace (Covestro LLC), C. King (Evonik Corp.), J. Miller (Covestro LLC), and T. Yu (Wanhua) for providing insight on the prepolymer chemistry design of experiments, polyol starting materials, and analytical characterizations, and to S. Seneker (retired from Anderson Development Company, Adrian, MI, USA) for synthesizing the test substances. Dr. P. Plehiers and Mr. M. Spence of the International Isocyanate Institute, Inc. are acknowledged for helpful cooperation in coordination of this study with prior work, and for helpful review and editing of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M Kelly and R Jarrom have no known or stated conflicts of interest. G Simon is a consultant for Vencorex US, Inc., a producer of HDI and IPDI. S Snyder is employed by Covestro LLC, a producer of TDI, MDI, HDI, and IPDI. RJ West is a private consultant for the American Chemistry Council ADI Panel.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work reported herein was funded by the American Chemistry Council (ACC) Aliphatic Diisocyanates (ADI) Panel. Any opinions expressed are those of the authors, and not necessarily of ECsolved LLC, CSB Compliance, Smithers ERS Ltd, Simon Toxicology LLC, Covestro LLC, the American Chemistry Council and its member companies. The ACC ADI Panel is funded by producers of HDI and IPDI.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
