Abstract
Background
Deep brain stimulation targeting the subcallosal cingulate (SCC-DBS) is a promising therapy for treatment-resistant depression. However, the lack of a consistent, rapid behavioural response to SCC-DBS complicates the selection of optimal stimulation settings following implantation, requiring a prolonged and burdensome trial-and-error process. Immediate biomarkers of effective stimulation could overcome this problem.
Methods
In this proof-of-concept study, three patients with SCC-DBS implants were scanned at 3 T using a block-design paradigm in which stimulation alternated between “ON” and “OFF” states in 30-s cycles during a single 6.5-min acquisition. Scans were performed using participants’ clinically optimized parameters. Blood-oxygen-level-dependent (BOLD) response maps were generated by contrasting DBS-ON and DBS-OFF conditions, and exploratory correlations with clinical outcome—indexed by percentage reduction in Hamilton Depression Rating Scale scores at 12 months—were also assessed.
Results
Contrasting stimulation settings enabled the identification of regional BOLD signal changes associated with DBS, revealing consistent hemodynamic changes in several brain regions during active stimulation. Specifically, the precuneus, posterior cingulate cortex, middle frontal gyrus, and frontal pole exhibited decreased BOLD responses during active DBS, while the occipital cortex, middle temporal gyrus, inferior parietal lobule, and superior frontal gyrus showed increased BOLD responses. Exploratory analysis further suggested a potential correlation between precuneus BOLD signal change and clinical improvement (R = −0.98, ppermute = 0.09).
Conclusion
These findings speak to the utility of block-design fMRI with cycling DBS stimulation as a tool to identify objective, brain-based biomarkers of effective SCC-DBS, potentially expediting stimulation parameter selection and therapeutic optimization.
Plain Language Summary Title
Tracking Rapid Brain Responses to Deep Brain Stimulation for Depression
Plain Language Summary
This proof-of-concept study explored whether real-time brain imaging could help identify immediate effects of deep brain stimulation (DBS) in patients with treatment-resistant depression. DBS targeting the subcallosal cingulate (SCC) is a promising therapy, but it currently lacks fast, reliable markers to guide optimal stimulation settings. As a result, programming DBS settings involves a slow, trial-and-error process. To address this, we tested a method called “cycling fMRI” in three patients who had previously received SCC-DBS. During a 6.5-minute brain scan, each patient’s stimulation settings were alternated between “on” and “off ” every 30 seconds. Using functional MRI (fMRI)—a technique that measures changes in blood oxygen levels as an indirect marker of neural activity—we compared brain responses between these alternating stimulation states. Results showed that switching the stimulation on caused immediate changes in several brain areas. The precuneus, posterior cingulate, and frontal regions showed reduced activity, while occipital and temporal areas became more active. These patterns suggest that DBS can rapidly influence brain networks involved in depression. Notably, the precuneus —a region associated with self-reflection and part of the brain’s default mode network —showed changes that may relate to clinical improvement. Exploratory analysis showed that stronger reductions in precuneus activity could be linked to better treatment outcomes. The study confirms that cycling fMRI can detect rapid brain changes during DBS and may help identify useful brain-based biomarkers. If validated in larger studies, this approach could speed up DBS programming and improve outcomes by personalizing treatment more efficiently. Overall, this research opens the door to using real-time brain imaging as a guide for optimizing psychiatric neuromodulation therapies.
Introduction
Major depressive disorder (MDD) is the leading non-fatal cause of disability globally. 1 While standard treatments—such as pharmacotherapy, psychotherapy, and electroconvulsive therapy—are effective for many, a significant proportion of individuals with MDD do not achieve adequate symptom relief. 2 For patients with severe, treatment-resistant depression (TRD), deep brain stimulation of the subcallosal cingulate (SCC-DBS) has emerged as a potential therapeutic intervention. Open-label trials report significant antidepressant effects, with an average of 60% of patients demonstrating meaningful and durable clinical improvement. 3
Despite these promising results, there is substantial variability in patient outcomes. 3 This variability is closely tied to challenges in optimizing stimulation parameters—including voltage and electrode contact configuration—in psychiatric applications. 4 Unlike DBS for movement disorders like Parkinson's disease, which benefits from immediate physiological feedback to stimulation (e.g., rapid alleviation of rigidity or tremor), SCC-DBS lacks real-time biomarkers with which to guide post-operative stimulation programming. In TRD, the therapeutic effects of DBS often gradually manifest over weeks or months, likely underpinned by unfolding neuroadaptive processes. 5 Consequently, stimulation programming can last up to a year, imposing significant logistical and financial burdens on patients and clinicians alike, and it can be difficult to thoroughly survey the parameter space so as to conclusively identify the “optimal” set of stimulation parameters. 6
Substantial recent research has focused on identifying functional brain-based biomarkers to optimize SCC-DBS programming and improve therapeutic outcomes. Several studies have employed electrophysiological techniques—such as EEG7–10 and local field potential (LFP)11–14 recordings—to identify neural oscillatory patterns, and PET imaging examining glucose metabolism,15,16 in order to identify markers associated with a favourable response to DBS in patients with depression. At least one study has taken an alternative tack, utilizing 3 Tesla functional MRI (fMRI) to map brain responses to SCC-DBS with high spatial resolution. Leveraging institutional expertise in the developed safety protocols,17–20 our group acquired resting-state fMRI (rsfMRI) runs in patients with fully implanted SCC-DBS devices both while their device was off (DBS-OFF) and while it was turned on (DBS-ON). By contrasting patients’ basal brain activity (as indexed by the amplitude of low-frequency fluctuations [ALFF] metric) between separate DBS-ON and DBS-OFF acquisitions, this analysis identified distinct stimulation-related changes within areas like the dorsal anterior cingulate cortex (dACC), posterior cingulate cortex (PCC), and precuneus. Changes in these areas, moreover, differed between clinically optimal and suboptimal stimulation settings and also correlated with long-term clinical improvement. 21
In parallel with this work, our group recently applied a different fMRI paradigm–one in which patients’ DBS devices were set to repeatedly cycle between DBS-ON and DBS-OFF during a single fMRI acquisition—to a sizable cohort of patients undergoing subthalamic nucleus DBS (STN-DBS) for Parkinson's disease. This “cycling” block design yielded blood-oxygen-level-dependent (BOLD) signal response maps that reflected changes in brain activity occurring within seconds of STN-DBS onset. Moreover, when paired with machine learning techniques, these BOLD response maps were able to differentiate clinically optimal and suboptimal stimulation parameters in individual patients. 22
In this proof-of-concept study, we aimed to advance the understanding of SCC-DBS's effects on brain-wide activity—particularly its immediate effects taking place within seconds of stimulation initiation—by extending the previously described “cycling” fMRI block design approach to this cohort. 22 By demonstrating the feasibility of this approach in SCC-DBS, we also hoped to lay the groundwork for future efforts to identify acute neuroimaging biomarkers that might streamline and enhance stimulation programming and ultimately improve patient outcomes.
Methods
Participants
After securing institutional approval (University Health Network #14-8255), patients who received SCC-DBS for depressive symptoms at Toronto Western Hospital were enrolled in this proof-of-principle study (Figure 1A). All potential participants had previously undergone surgical implantation and post-operative determination of stimulation parameters as previously described. 23 Eligibility for MRI scanning required patients to have MRI-compatible Medtronic DBS systems—consisting of 3387 quadripolar leads, a 37601 Activa PC or 37612 Activa RC implantable pulse generator, and either 37086 or 7482 extension wires. Long-term clinical outcomes were based on participants’ percentage reduction on the 17-item Hamilton Depression Rating Scale (HAMD-17) at 12 months post-surgery compared to pre-operative baseline scores.
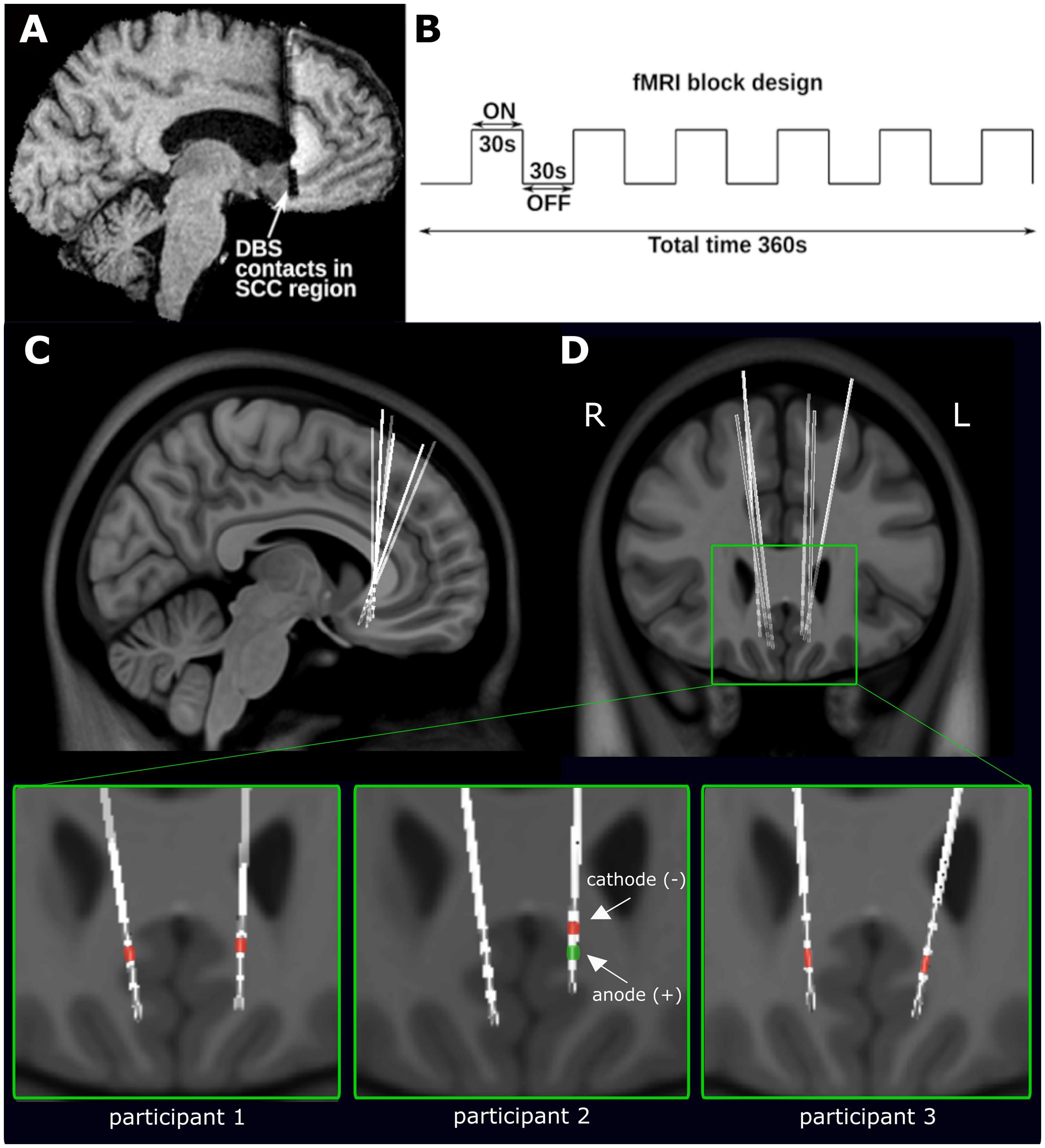
Experimental paradigm of 3 T fMRI imaging and DBS lead localization of TRD patients with SCC-DBS. (A) An exemplar sagittal T1-weighted image showing the implanted depth electrode with contacts located in the subcallosal cingulate area. (B) Functional MRI (fMRI) images were acquired using a block design in which each participant's DBS system was set to cycle between DBS-ON and DBS-OFF states every 30 s. Each fMRI scan was 6.5 min in duration; the first 30 s were discarded, leaving 6 min of usable data. (
MRI Safety Validation
MRI scanning of patients implanted with DBS devices has been systematically validated within our institution. Initial phantom studies characterized device heating, voltage fluctuations, and artifacts across multiple structural and functional pulse sequences under both ON and OFF stimulation conditions. Based on these safety data, subsequent clinical testing in a large cohort (>200 patients) demonstrated the safety of scanning at both 1.5 T and 3 T, with hardware-related artifacts thoroughly characterized and mitigated. These protocols were approved by our institutional research ethics board and have been consistently applied in studies involving implanted patients. It should be noted that these procedures were specific to scanners and protocols used at our institution, Toronto Western Hospital; institutions seeking to perform similar studies are encouraged to conduct their own phantom testing and safety validation.17–20
Imaging Acquisition
All participants were scanned with 3 Tesla MRI (General Electric Signa HDxt) following informed consent to participate in this study. In each case, anatomical images were first obtained using the T1-MPRAGE pulse sequence (FOV, 256 × 256 mm; flip angle, 20°; TR, 8 ms; TE, 3 ms; voxel size, 0.86 × 0.86 × 1 mm). Subsequently, functional MRI data were acquired using a multiphase 2D gradient echo-echo planar imaging (GRE-EPI) pulse sequence (FOV, 240 × 240 mm; slice thickness, 3 mm; flip angle, 76°; TR, 2151 ms; TE, 30 ms, 1 signal average; voxel size 3.75 × 3.75 × 3 mm).
Each participant completed a single fMRI session lasting 6.5 min, during which a 30-s DBS-ON/OFF cycling paradigm was implemented, as previously described (Figure 1B). 22 Stimulation was restricted to unilateral left-sided activation, delivered using each participant's clinically optimized parameters, and alternated between ON and OFF settings without a gradual ramp-up. The block design with sustained 30-s epochs was chosen to capture acute stimulation-related changes, allowing the hemodynamic response to stabilize during DBS-ON (task) periods and return toward baseline during DBS-OFF (rest) periods. This design provides high detection power for contrasting ON and OFF states while mitigating short-lived transition effects.24,25 Although some carry-over between conditions cannot be entirely excluded, averaging across multiple cycles further reduces such influences and emphasizes stable differences in brain activity. DBS cycling was manually synchronized with MRI acquisition to ensure precise timing.
Preprocessing and Exploratory Statistics
The first 30 s of each fMRI sequence were discarded to establish steady state and to allow participants to adapt to the scanning situation, yielding 6 min of usable scan time. Acquired fMRI data were then slice time corrected, motion corrected, rigidly registered to the T1-weighted image, non-linearly registered to a standard space Montreal Neurological Institute (MNI) brain, and spatially smoothed using a Gaussian kernel (6 mm full-width at half-maximum) using SPM12 (http://www.fil.ion.ucl.ac.uk) and MATLAB (Mathworks, Natick, MA, USA). Statistical parametric maps (t-maps) were estimated from the preprocessed fMRI data using the designed 30-s DBS-ON/OFF cycling stimulation paradigm. The hemodynamic response function was modelled using the canonical double gamma function. 26
To identify areas that showed consistent BOLD changes with active DBS, these individual BOLD response maps were then combined to generate a voxel-wise raw average map of t-statistics across participants. We corrected for multiple comparisons by first adjusting voxel values with threshold-free cluster enhancement (TFCE) 27 and then thresholding these adjusted values using the false discovery rate (pFDR < 0.01). 28 As an additional exploratory analysis, we looked into potential relationships between a specific region of BOLD change and clinical outcome. This was done by calculating an actual Pearson correlation coefficient and comparing this to a permuted null distribution (100 permutations using shuffled HAMD-17 reduction scores). R (R 4.0.4., https://www.r-project.org) and RMINC (https://github.com/Mouse-Imaging-Centre/RMINC) were used for statistical analysis.
To account for DBS-related artifacts on postoperative fMRI analyses, 17 regions affected by metallic susceptibility distortions from the leads and extension wires were manually segmented for each participant using MINC Display (https://github.com/BIC-MNI/minc-toolkit-v2). The resulting masks were subsequently registered into MNI152 standard space, and a binarized summation mask was generated. Voxels intersecting with this mask were excluded from all further analyses.
Results
Three participants who previously underwent SCC-DBS implantation for management of depressive symptoms were scanned with our DBS-ON/OFF cycling fMRI paradigm (age: 23–53, all female, HAMD-17 reduction from baseline: 0–69%). At the time of this study, these individuals had been actively using their stimulators for 5–14 years. DBS lead and active contact locations were reconstructed and visualized in MNI space using Lead-DBS (https://www.lead-dbs.org/), as previously described (Figure 1A and B). 21 Further information regarding patient demographics and individual clinically optimized stimulation parameters can be found in Supplemental Table 1.
Block design fMRI in which SCC-DBS stimulation was cycled on and off repeatedly within a single acquisition sequence detected immediate BOLD signal changes in several brain areas in a small cohort of patients, providing novel insight into the rapid distributed action of this intervention on brain function. Inspection of the group average t-map revealed several areas that showed significant changes in BOLD response with active SCC-DBS. The precuneus, posterior cingulate cortex, middle frontal gyrus, and frontal pole tended to exhibit decreased BOLD response during SCC-DBS stimulation, while areas such as occipital cortex, temporal pole/middle temporal gyrus, and inferior parietal lobule tended to show increased BOLD response (Figure 2).
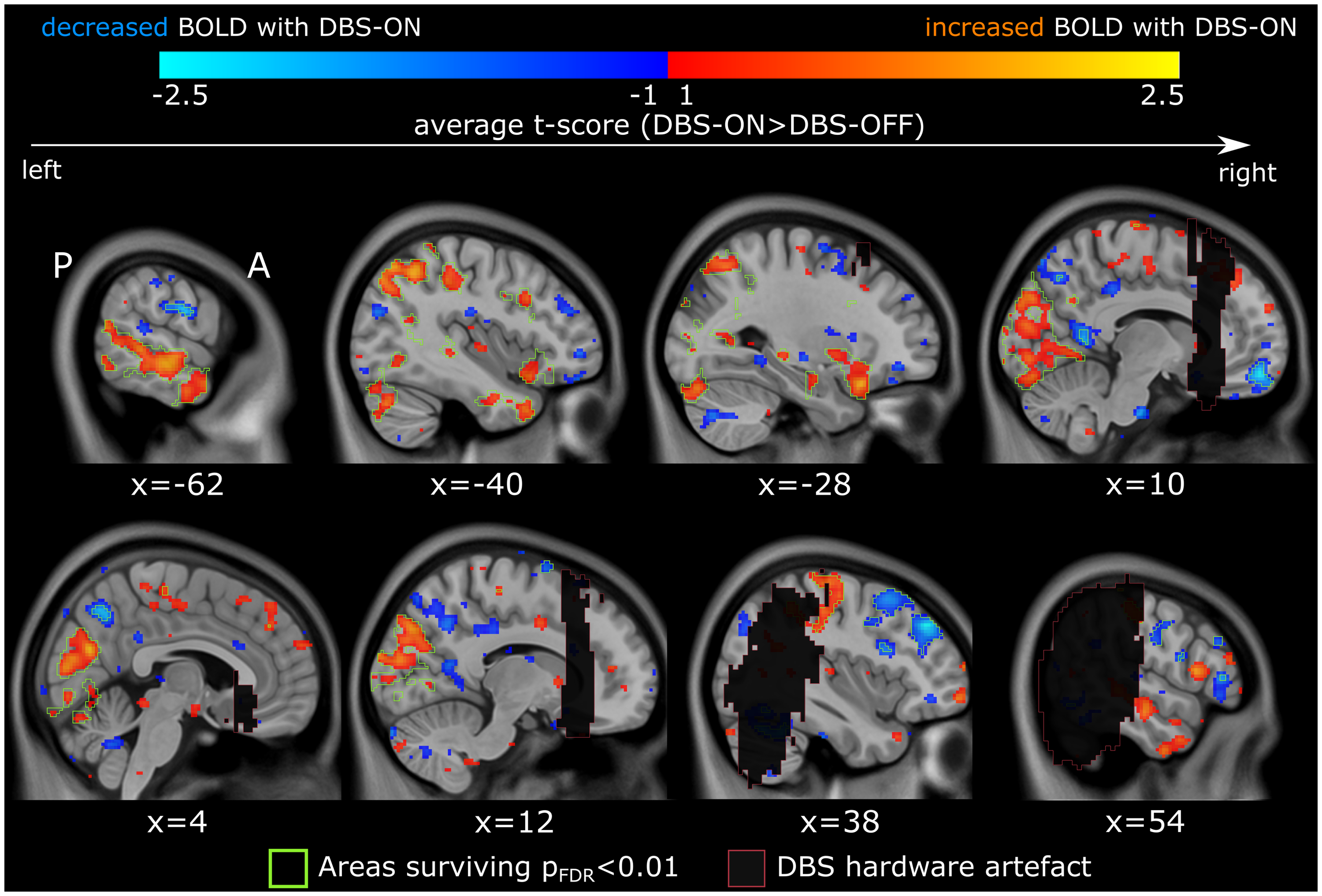
fMRI responses upon cycling of DBS-ON/DBS-OFF settings. Voxel-wise paired t-tests were conducted to contrast participant's (n = 3) DBS-ON state with their DBS-OFF state. A group average t-map showing areas of acute BOLD response to active SCC-DBS (contrast: DBS ON > OFF) is displayed in MNI standard space. Areas of BOLD response that survive multiple comparisons correction (pFDR < 0.01) are demarcated in green. DBS-related artifact regions, marked by red-outlined black boxes, were excluded from analysis. BOLD = blood-oxygen-level-dependent; DBS = deep brain stimulation; FDR = false discovery rate; MNI = Montreal Neurological Institute; SCC = subcallosal cingulate area.
Among these regions, the precuneus was selected for further analysis based on its prominent BOLD signal changes and its previously established involvement in SCC-DBS response. 21 Previous studies have reported that the precuneus, along with other key corticolimbic regions, exhibits decreased ALFF signal during active stimulation and contributes to predictive models of clinical outcome. Additionally, the precuneus is directly structurally connected to the SCC target region via the cingulum bundle, providing a strong neuroanatomical basis for its immediate response to stimulation (Figure 3A). 21 Given its consistent response to SCC-DBS, we examined whether the BOLD signal changes observed in this region during our stimulation paradigm were associated with clinical outcome, measured by HAMD-17 score improvement. Our exploratory analysis revealed a potential negative correlation between BOLD signal change in the right precuneus (x = 6, y = −62, z = 46) and clinical improvement (R = −0.98, Ppermute = 0.09) (Figure 3B and C), although larger sample sizes will be necessary to clarify the existence of this relationship.
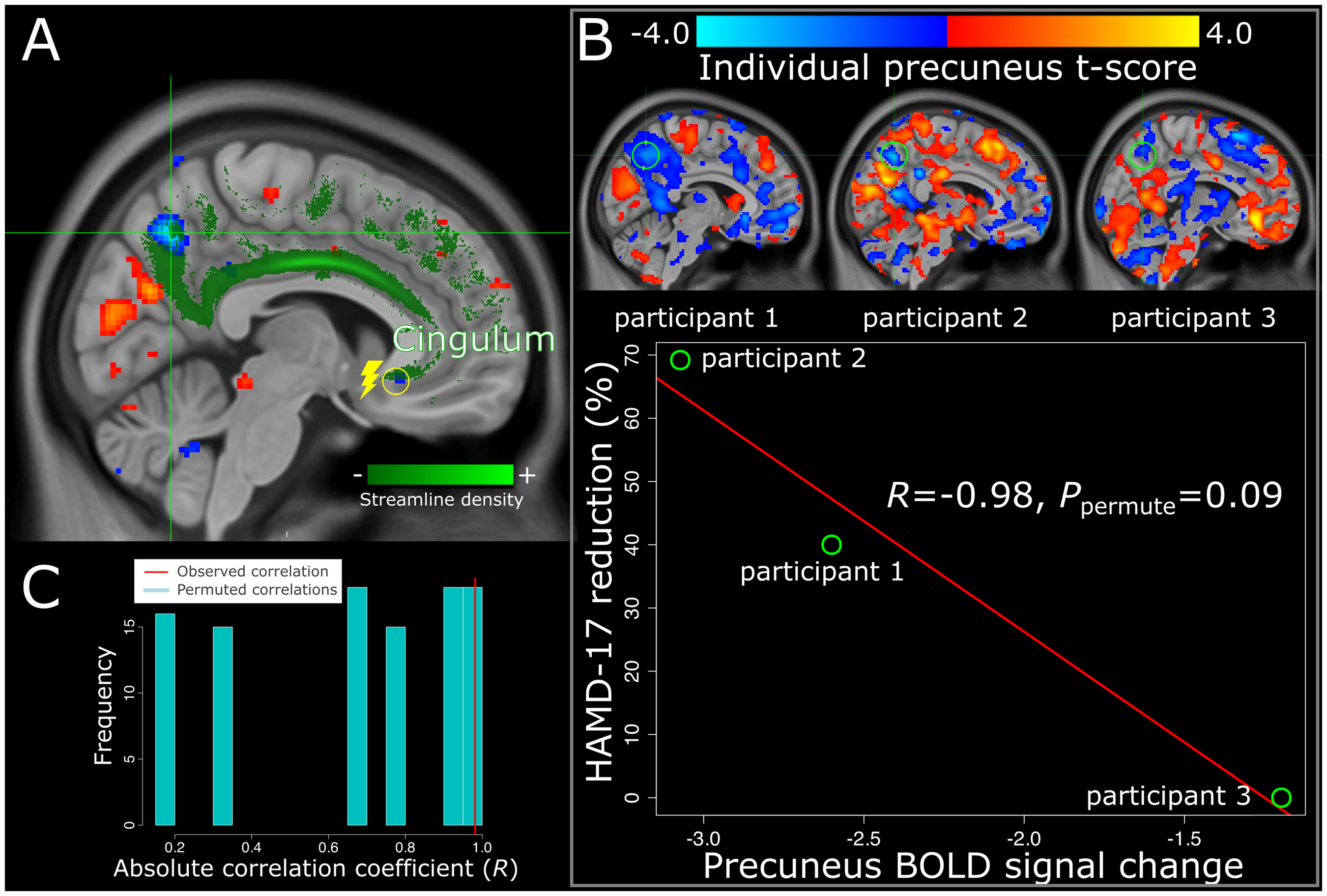
DBS-driven changes in the precuneus are associated with long-term clinical outcomes.
Discussion
We investigated the immediate neural effects of SCC-DBS in three patients with TRD using a “cycling” fMRI paradigm, where stimulation was systematically turned on and off. This method allowed us to capture real-time brain responses, revealing robust alterations in several regions within seconds of stimulation. Further, prior resting-state DBS work contrasting DBS-ON and DBS-OFF has reported convergent results when buffer periods were incorporated to permit the stimulation-associated BOLD signal to stabilize between conditions, providing precedent for the use of a cycling paradigm to assess acute stimulation effects. 21 In demonstrating that a cycling approach can track these acute neural changes, our proof-of-concept study supports its potential utility for biomarker identification. These findings align with prior fMRI research on the immediate effects of SCC-DBS, 21 further validating the use of real-time stimulation paradigms to assess rapid brain activity changes in response to psychiatric neuromodulation.
The absence of reliable real-time behavioural response to SCC-DBS complicates efforts to select and refine stimulation parameters, potentially attenuating the therapeutic benefits of this intervention. Building on prior rsfMRI by our group, 21 the current study sought to tackle this challenge by applying a cycling fMRI paradigm to systematically measure BOLD signal changes during active SCC-DBS. This approach identified significant brain activity alterations in several areas, particularly within key nodes of the default mode network (DMN). In line with prior fMRI SCC-DBS research, 21 we observed stimulation-related (likely cingulum bundle-mediated) decreases in activity of the precuneus and posterior cingulate cortex, key DMN hubs implicated in self-referential processing and negative valence rumination.29–31 Given that DMN hyperactivity in MDD is associated with these maladaptive cognitive patterns, 32 SCC-DBS may help to alleviate depressive symptoms in part by helping restore DMN network balance. Our exploratory analysis further pointed toward a potential correlation between precuneus activity and long-term clinical improvement, reinforcing prior findings that linked clinical outcomes to stimulation-induced changes in ALFF-indexed baseline activity within this area. 21 This consistency across two different fMRI methods highlights the precuneus as a potentially robust biomarker for treatment response and lends further credence to the cycling fMRI paradigm as an effective, time-efficient tool for assessing real-time neural responses during SCC-DBS.
Beyond the precuneus, our study identified acute SCC-DBS-induced changes in additional brain regions, including the middle frontal gyrus (encompassing the functionally defined dorsolateral prefrontal cortex [dlPFC]), frontal pole, temporal cortical areas, and occipital cortex. The dlPFC, a region subserving executive function, 33 showed reduced activity during stimulation, primarily in the right hemisphere. In depression, an interhemispheric imbalance is often observed, with relative hyperactivity on the right and hypoactivity on the left, contributing to impaired regulation of emotional processes.34–36 The right-sided reduction we observed may therefore represent a rebalancing of this system, consistent with the hypothesis that SCC-DBS helps restore dysregulated corticolimbic circuitry.35,37,38 The frontal pole, which may exhibit compensatory hyperactivity in depression as an attempt to override persistent negative mood, 37 displayed decreased BOLD signal during stimulation. Pre-operative hypometabolism in this region has been associated with SCC-DBS treatment response,39,40 suggesting that acute modulation may similarly serve as a biomarker of efficacy. The temporal pole, in contrast, exhibited increased BOLD signal, likely reflecting engagement of the uncinate fasciculus upon stimulation, a key white matter tract linking the SCC to the temporal pole. 40 Prior work from our group has shown that engagement of specific white matter pathways predicts SCC-DBS outcomes, 41 with metabolic analysis further linking uncinate fasciculus involvement to temporal pole metabolism39,40—both serving as potential predictors of treatment response. Lastly, we observed increased activity in the occipital lobe during SCC-DBS. Given that reduced occipital activity has been associated with cognitive dysfunction and attentional deficits in MDD, 42 its acute modulation during stimulation may help explain reports of improvements on some neuropsychological tests after SCC-DBS. 43
Certain limitations of this work should be acknowledged. For one, this is only a preliminary proof-of-principle study. The small sample size (n = 3) limits the generalizability of the findings and necessitates particular caution in interpreting the exploratory analysis on correlations between BOLD signal changes and clinical improvement. Validation of our results in larger, prospective cohorts is required to confirm their reliability and clinical applicability. Additionally, while standard preprocessing steps were applied, no rigorous denoising procedures were performed, which may limit the robustness of the observed effects; incorporating denoising in future work could further improve signal detection and reliability. Future work could directly compare the cycling fMRI paradigm described here with previously used rsfMRI approaches, further assessing the consistency of results yielded by the two methods and clarifying whether either offers meaningful advantages in time efficiency or sensitivity in terms of detecting clinically relevant stimulation-induced brain changes. Another important consideration is that all participants in this study had been receiving chronic stimulation at the time of scanning. While this limits our ability to capture the earliest neural changes following implantation, longitudinal application of this approach in SCC-DBS patients would provide valuable insight into how acute responses evolve over time and whether they may serve as early biomarkers of therapeutic outcome. 22 Prior work involving patients with Parkinson's disease demonstrated that cycling fMRI signatures elicited with “optimal” DBS were highly similar in both early and long-term patients, and that these signatures could be used to predict stimulation parameters in treatment-naïve individuals. This precedent suggests that the neural patterns observed here, despite being collected in chronically treated patients, may still carry predictive utility. The present study extends this methodology to SCC-DBS as a feasibility test, offering preliminary evidence of its potential utility and motivating future longitudinal trials. Other valuable work could involve incorporating machine learning techniques, as demonstrated in the Parkinson's disease cohort, 22 could help identify predictive markers of stimulation response.
Conclusion
This proof-of-principle study strengthens the case for using fMRI to identify objective, brain-based biomarkers of effective SCC-DBS, offering a potential pathway to streamline and personalize post-operative programming and improve therapeutic outcomes. While merely preliminary, these findings suggest cycling fMRI paradigms could hold promise—potentially as a complement to rsfMRI approaches—as a time-efficient way to map acute neural responses to this intervention.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251387573 - Supplemental material for Capturing Brain Response Patterns to Subcallosal Cingulate Deep Brain Stimulation Using Cycling fMRI: A Proof-of-Concept Study: Acquisition de schémas cérébraux de réponse à la stimulation cérébrale profonde ciblant le cortex cingulaire subgénual à l’aide de l’IRMf en cycles : étude de validation
Supplemental material, sj-docx-1-cpa-10.1177_07067437251387573 for Capturing Brain Response Patterns to Subcallosal Cingulate Deep Brain Stimulation Using Cycling fMRI: A Proof-of-Concept Study: Acquisition de schémas cérébraux de réponse à la stimulation cérébrale profonde ciblant le cortex cingulaire subgénual à l’aide de l’IRMf en cycles : étude de validation by Sarah A. Iskin, Gavin J.B. Elias, Asma Naheed, Jessica Pinto, Uyiosa Omere, Sidney H. Kennedy, Aaron Loh, Afis Ajala, Brendan Santyr, Alexandre Boutet, Andres M. Lozano and Jürgen Germann in The Canadian Journal of Psychiatry
Footnotes
Potential Conflicts of Interest
A.M.L. is the co-founder of Functional Neuromodulation (a DBS-related company) and is a consultant for Boston Scientific, Medtronic, and Abbott (companies that produce DBS hardware). S.H.K. is a consultant for Abbott. A.A. is a salaried employee of GE HealthCare. All other authors report no conflicts of interest.
Funding
This work was supported in part with funds from the Krembil Research Institute (Toronto, Canada) to JG, and by the National Institute of Neurological Disorders and Stroke of the National Institutes of Health, under Award R01NS133433 to AA.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
