Abstract
Objective:
There is increasing interest in the use of deep brain stimulation as a treatment for psychiatric disorders. In this review, we consider the evidence for the effectiveness of deep brain stimulation for psychiatric indications, with a primary focus on obsessive compulsive disorder and major depressive disorder.
Methods:
Case reports, case series and clinical trials where deep brain stimulation was primarily utilised in the treatment of a psychiatric disorder, including obsessive compulsive disorder, major depressive disorder, anorexia nervosa or an addictive disorder were identified. The evidence for the effectiveness of deep brain stimulation in the treatment of obsessive compulsive disorder and major depressive disorder was reviewed with studies clustered by the site of implantation.
Results:
The majority of identified manuscripts report small case series or single cases. A limited number of studies have reported some form of randomised or blinded stimulation comparison. All of these comparative reports have included small samples of subjects (less than 20 per study in total) compromising the feasibility of making statistical comparison between outcomes in the comparison phases. The two exceptions to this have been industry-sponsored studies conducted in the treatment of major depressive disorder. However, both were stopped prematurely due to concerns about poor efficacy.
Conclusions:
There is insufficient evidence at this point in time to support the use of deep brain stimulation as a clinical treatment for any psychiatric disorder outside of research and programmes where formal outcome data are being systematically collated. While some promising initial data exist to support its potential efficacy for a number of psychiatric conditions, further research is required to establish optimal implantation targets, patient characteristics associated with positive therapeutic outcomes and optimal deep brain stimulation parameters and parameter-programming methods.
Keywords
Deep brain stimulation
Deep brain stimulation (DBS) is a procedure that involves the implantation of stimulation electrodes in specific highly localised brain regions with the aim of modifying local and connected brain activity (Greenberg, 2002). DBS has been extensively used in neurological disorders such as Parkinson’s disease, intractable tremor and dystonia and is less commonly used or under investigation for other disorders such as epilepsy and Tourette’s syndrome (Benabid, 2003; Katayama et al., 2003; Kupsch et al., 2003; Levy, 2003; Rosenow et al., 2004; Visser-Vandewalle et al., 2004). The indication for the use of DBS determines the placement of the individual stimulating electrodes. For many indications, there are a number of neuroanatomical targets that provide therapeutic relief. For example, three DBS implantation sites have been shown to be clinically efficacious for Parkinson’s disease: the globus pallidus, the subthalamic nucleus (STN) and a subdivision of the thalamus referred to as Vim (Rosenow et al., 2004), presumably because they can all influence activity of the motor control circuits relevant to the pathophysiology of Parkinson’s disease.
The actual DBS procedure involves several stages. Initially, the stimulating electrodes are inserted during a stereotactic neurosurgical procedure and a pacemaker-like device is connected to these and placed in the chest below the clavicle. Once the patient has recovered from surgery, the stimulator is turned on and various stimulation parameters (including voltage, pulse width and frequency) are adjusted to achieve optimal response of symptoms. Standard stimulating systems have four small electrodes on the end of the stimulation wire and one of the goals of this initial programming period is to identify the optimal stimulating electrodes for the patient in question. Once optimal stimulation parameters are set, stimulation is essentially ongoing, although changes in these parameters can occur as needed and the device may be turned off to preserve battery life, for example, overnight.
The mechanism of action of DBS remains the subject of speculation (McIntyre et al., 2004a, 2004b). Although it was in part developed from a model of the disruption of brain regions (i.e. invasive psychosurgery), it has also been demonstrated that the high frequency stimulation alters brain activity in a functional way, rather than just disrupting one limited brain region. The effect of stimulation is likely to vary with stimulation frequency; with frequencies in the range of 50–60Hz associated with stimulatory activity and stimulation at >100 Hz associated with the induction of inhibition (Benabid, 2003). It is possible that chronic stimulation induces adaptive plastic brain responses, such as altered synaptic activity in relevant circuits (McIntyre et al., 2004a, 2004b).
DBS for psychiatric disorders
A number of lines of clinical activity have led to interest in the potential use of DBS for the treatment of psychiatric disorders. First, there is a history of other surgical approaches to the treatment of psychiatric disorders, especially lesioning procedures such as anterior capsulotomy, subcaudate tractomy and limbic leucotomy (Fins, 2003). These aimed to disrupt connections between cortical and subcortical brain regions involved in the proposed symptoms of the disorders being treated.
Second, there has been a demonstration of mood-relevant effects when DBS has been used in the treatment of neurological conditions such as Parkinson’s disease (Kulisevsky et al., 2002; Okun et al., 2009). Third, there has been a progressive recognition that psychiatric disorders such as obsessive compulsive disorder (OCD) and depression involve disruption of complex brain networks involving multiple dysfunctional brain regions (Drevets et al., 2008; Pauls et al., 2014). This view has come to supplant previous ideas about the pathophysiology of these disorders, which were predominantly focused on abnormalities in specific monoamine neurotransmitters. A network understanding of the pathophysiology of disorders like OCD and depression lends itself to the development of therapeutic approaches which target specific nodes in this network or critical white matter pathways responsible for the connections between network notes.
Major depressive disorder, OCD and treatment resistance
Major depression is currently the leading cause of non-fatal disability in Australia and the fifth leading cause world-wide (Murray and Lopez, 2013). Depression is an extremely common psychiatric illness, with a 15–20% lifetime prevalence rate globally (BeyondBlue, 2006). Approximately one million Australian adults live with this condition each year (BeyondBlue, 2006). Approximately 30% of patients with depression do not respond to standard medication and psychological therapies (Fava and Davidson, 1996; Rush, 2007). Treatment-resistant depression results in considerable suffering for individuals, as well as increased burden of care for families. The economic burden is also considerable with the treatment costs for treatment non-responsive patients up to 19 times greater than those for patients with depression who respond to treatment (Crown et al., 2002).
The management of treatment-resistant depression includes repeated trials of medication, medication combinations, psychotherapy and forms of brain stimulation (transcranial magnetic stimulation and electroconvulsive therapy). However, there is a significant subgroup of patients (10–20%) who remain chronically treatment non-responsive, disabled and suffer greatly. Even for those patients who respond to the most potent antidepressant electroconvulsive therapy, relapse rates remain high (Tew et al., 2007) and many patients develop therapeutic ‘resistance’ over time. These patients currently have no effective treatment options.
OCD is not as prevalent as depression, but is still a relatively common mental illness with a lifetime prevalence of 2–3% (Karno et al., 1988; Ruscio et al., 2010; Weissman et al., 1994). It is characterised by recurrent, intrusive anxious thoughts (obsessions) accompanied by repetitive ritualised behaviours or mental routines (compulsions) that are frequently performed in an effort to reduce distress caused by obsessions. The intrusive distressing nature of obsessions and the substantial time spent executing compulsive behaviours and rituals can cause substantial impairment in interpersonal relationships, social and occupational functioning and the ability to carry out basic activities of daily living (Koran et al., 1996; Richter et al., 2003). Individuals with persistent or severe OCD report extremely low quality of life and have an elevated risk of attempting suicide, with between 10% and 27% of patients making an attempt during their lifetime (Alonso et al., 2010; Huppert et al., 2009). Comorbidity with other mental illnesses, most commonly major depression, is high.
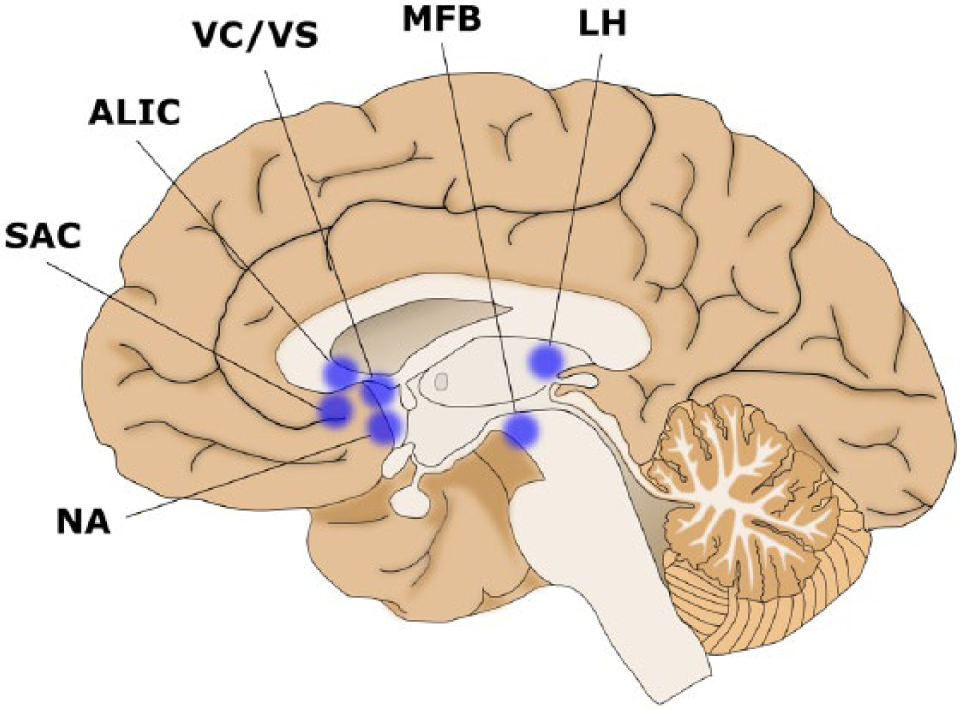
Deep brain stimulation for treatment-resistant depression: Implantation targets.
Treatment for OCD typically involves pharmacotherapy (e.g. selective serotonin reuptake inhibitors) which is often combined with psychotherapy (e.g. cognitive behavioural therapy/exposure and response prevention). However, OCD is a notoriously difficult condition to treat and up to 60% of patients do not obtain adequate benefit with standard treatment approaches (Kellner, 2010; Pallanti and Quercioli, 2006; Simpson et al., 2008). Approximately 10% of OCD sufferers remain densely treatment resistant to all known therapies and chronically afflicted with severe symptoms (Denys, 2006). Compared with psychiatric illness like depression, there are comparatively few treatments indicated for patients with OCD, leaving those with severe treatment-resistant illness with extremely limited therapeutic options and minimal hope for recovery.
The frequent occurrence of treatment resistance and an increasing body of research identifying abnormal brain circuits in depression and OCD has led to an interest in the use of DBS as a potential therapy for these conditions.
A review of DBS for psychiatric disorders
Below we discuss the rationale for DBS for psychiatric indications and summarise the outcomes of investigations conducted to date. The evidence described was identified via a search of relevant academic literature published using the search engines PubMed, Scopus, OVID Medline and Google Scholar using combinations of the following key words: deep brain stimulation, psychosurgery, psychiatry, depression, major depressive disorder, obsessive compulsive disorder, anorexia, addiction, substance dependence and substance abuse.
Where review articles were identified, their reference lists were also searched for additional relevant publications. Where multiple publications describing outcomes from the same patient cohorts were identified, only the most recent publication was included. When publications were identified that included a subset of patients described in a prior publication as well as a number of new patients, both publications were included. The latter of these studies was marked with a symbolic identifier (^, #, †) in tabulated presentation of study outcomes, and the overlap between the patient samples outlined beneath the table in question. Studies concerned with surgical outcomes and not reporting clinical data were excluded.
DBS in major depressive disorder
Potential anatomical targets for DBS applications in depression have been proposed based on (a) extrapolation from sites targeted in lesional psychosurgical procedures and (b) from the results of neuroimaging experiments. The majority of research to date has focused on DBS implantation in the white matter adjacent to the subgenual anterior cingulate (SAC) and on stimulation of the anterior limb of the internal capsule (ALIC) and the associated ventral striatal structures including the nucleus accumbens (NA; Table 2).
SAC
The SAC, a brain region used in early psychosurgery, was identified as a DBS target based on a series of positron emission tomography studies demonstrating changes in this brain region with successful antidepressant medication treatment of depression (Mayberg et al., 1999; Seminowicz et al., 2004). An overlapping series of clinical trials has been conducted evaluating the efficacy of DBS applied to the SAC in either single site or multisite clinical protocols. The initial report included the outcomes of six patients treated in an open-label fashion and followed for 6 months. This reported a 50% remission rate (Mayberg et al., 2005). Data from the same six patients were included in two reports of a larger sample of 20 patients followed for 12 months (Lozano et al., 2008) or greater than 3 years (Kennedy et al., 2011). Remission rates of 33% at 12 months and 43% at 3 years or last follow-up (up to 6 years) were reported. The most recent report from this series also documented improvements in physical health and social functioning and found that work participation rates had increased from 10% of patient’s pre-DBS to 65% at 6 years post-implantation (Kennedy et al., 2011).
Two further clinical trials have been reported by the same group. Holtzheimer et al. (2012) described outcomes for 17 patients (10 with major depressive disorder and 7 with bipolar affective disorder) who were followed for 2 years post-surgery. This trial incorporated a sham stimulation lead in phase and a subsequent period of stimulation discontinuation. The latter had to be removed from the protocol due to safety concerns with patients’ mental state deteriorating following stimulation discontinuation. The remission rate was 36% at 1-year and 92% at 2-year follow-up. A progressive improvement in social, occupation and psychological functioning was noted, with the average Global Assessment of Functioning scores increased by 44% from baseline at last follow-up. The second clinical trial was conducted in 21 patients across three trial sites in Canada (Lozano et al., 2012). This was an open-label study with 12-month follow-up. Response rates were 48% at 6 months and 29% at 12 months (Figure 2).
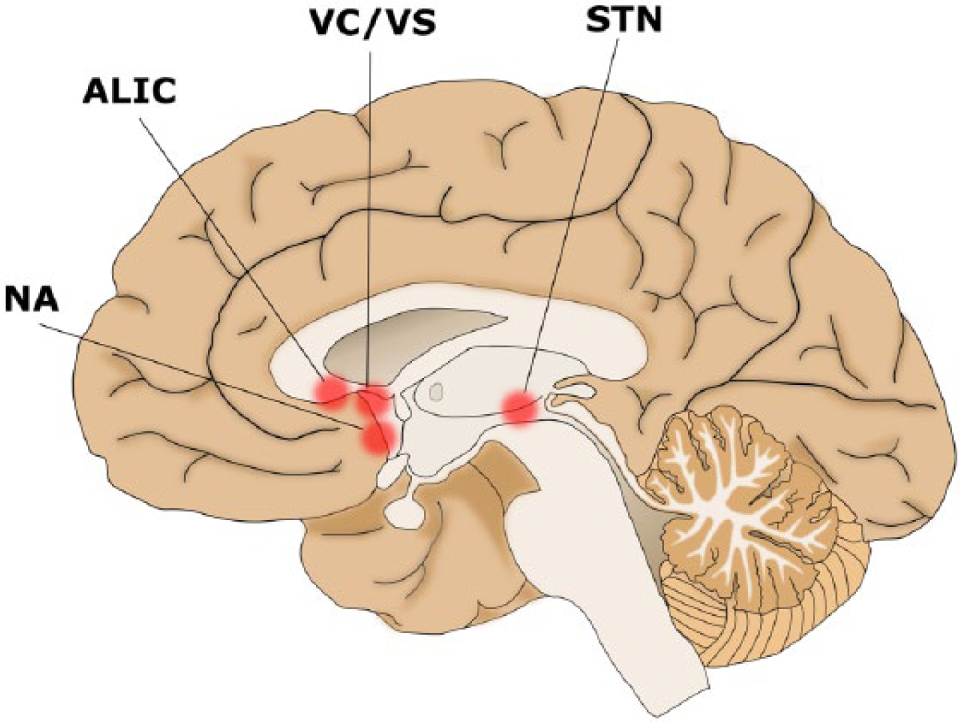
Deep brain stimulation for obsessive compulsive disorder: Implantation targets.
Only one publication independent of the originators of this approach has described the treatment of more than one patient with SAC DBS. In this Spanish study, 8 patients received DBS and were followed for 1 year (Puigdemont et al., 2011). The remission rate at this time was 50% and improved psychosocial functioning was described for a majority of patients, including taking up of leisure activities, initiation of social relationships and decreased need for assistance with personal care.
An attempt to commercialise the DBS SAC treatment approach has been made by St Jude Medical. The company commenced a multisite clinical trial which was expanded to 20 sites in 2011 and ended up recruiting at least 75 subjects. However, this clinical trial was halted in 2013 after a failed futility analysis (interim analysis of the data which suggested that it was unlikely that a trial would show clinical benefit; see the discussion in Morishita et al., 2014).
ALIC and the ventral striatum/ventral capsule
A second significant DBS target is the white matter of the ALIC and the associated ventral striatal structures including the NA. The ALIC was initially the target for psychosurgical treatment of OCD. It was upon this basis that DBS to this region was subsequently investigated for OCD. These studies noted a prominent antidepressant effect, often in excess of the benefits obtained in the core symptoms of OCD (personal communication Dr Greenberg and Dr Malone, May 2004). This led to interest in the conduct of studies of DBS at the ALIC specifically for the treatment of treatment-resistant depression.
There has been one published study that specifically targeted the ALIC: Malone et al. described the effects of DBS to the ALIC in 17 depressed patients. In the first publication, they described outcomes at 12 months and last follow-up (up to 3 years; Malone et al., 2009). In a second publication, the sample was extended to 17 patients, and the follow-up to up to 67 months (mean of 37.4 months; Malone, 2010). In this latter report, the 12-month remission rate was 41% (and 35% at last follow-up).
One multisite trial targeting the ventral capsule/ventral stratum has recently been published. In this study, 30 subjects were implanted and randomised to receive active or sham stimulation during a 16-week double-blind period (Dougherty et al., 2014). The majority of patients then received ongoing open-label stimulation. No significant differences in response rates were seen during the double-blind phase (20% in the active group and 14.3% in the sham group). Of the patients, 23.3% were responders at 2-year open-label follow-up.
NA
An approach related to ALIC stimulation has been to directly target the grey matter of the NA, which sits at the ventral end of the ALIC. It is a promising target for DBS as stimulation in this region could potentially disrupt or augment the amygdala – basal ganglia – prefrontal circuitry that is abnormally active in mood and anxiety disorders. The shell region has been thought to potentially be a ‘bottle-neck’ for information flow from the amygdala to the basal ganglia, and hence to the prefrontal cortex (Sturm et al., 2003). Blocking this in the grey matter of the NA would require considerably less electrical charge than blocking activity in the white matter tracts such as the internal capsule. This would have considerable advantage in that it might be possible to use smaller electrodes and less charge producing a considerably longer life time for the battery in the stimulation unit.
Only one group has directly targeted the NA to date. Following 12 months of persistent stimulation, 5 out 10 patients had achieved antidepressant response, 3 were in remission and anxiety was significantly reduced in all. All five patients who responded were still classified as responders (i.e. had no worsening of symptoms) at 2- and 4-year post-implantation follow-up (Bewernick et al., 2012). This group initially reported on the outcomes in three patients (Schlaepfer et al., 2008) who were subsequently included in a larger group of 10 followed up for 12 months (Bewernick et al., 2010). The sample was then extended to 11 patients who were followed for between 2 and 4 years (Bewernick et al., 2012). The 12-month remission rate was 30% but the 2-year remission rate had fallen to 9% (45% met response criteria persistently).
Other implantation targets
A recent study described outcomes of seven patients treated with DBS targeting the supero-lateral branch of the medial forebrain bundle (Schlaepfer et al., 2013). After experiencing a rapid onset of antidepressant symptoms, four patients were remitters at last follow-up (between 12 and 33 weeks). Two case reports have described DBS at other targets in the treatment of depression: one of these described DBS of the inferior thalamic peduncle (ITP; Jimenez et al., 2005) and one of the lateral habenula (Sartorius et al., 2010). These described remission of depression at 24 and 12 months, respectively.
DBS in OCD
A lengthy series of neuroimaging studies have explored the neurobiology of OCD and consistently identified the dysfunctional involvement of a series of cortical and subcortical brain regions and specifically dysfunction in cortico-striatal-thalamo-cortical circuitry in the pathophysiology of OCD (Graybiel and Rauch, 2000; Nakao et al., 2014). More specifically, in patients with OCD, there is abnormal (predominately increased) metabolic activity in the orbitofrontal cortex (OFC), the anterior cingulate cortex (ACC), medial prefrontal cortex and the caudate nucleus, particularly its ventral division (see Nakao et al., 2014). Studies have shown that OFC and caudate hyperactivity are directly correlated with symptom severity and these changes partially normalise with successful treatment. However, while neuroimaging investigations have contributed much to our understanding of the network of brain regions involved in the pathophysiology of OCD, the identification of DBS targets for OCD has been based on a combination of experience from lesional psychosurgery procedures, following observations of response to surgery for other conditions, such as Parkinson’s disease (as is the case with the STN) as well as gradual target refinement following ongoing evaluation of clinical outcomes in relation to lead location. The neuroanatomical targets used for DBS in the treatment of OCD have included the anterior limb of the ALIC, NA, ventral capsule/ventral striatum (VC/VS), STN and the ITP (Table 1).
DBS for OCD: Studies with a blinded component.
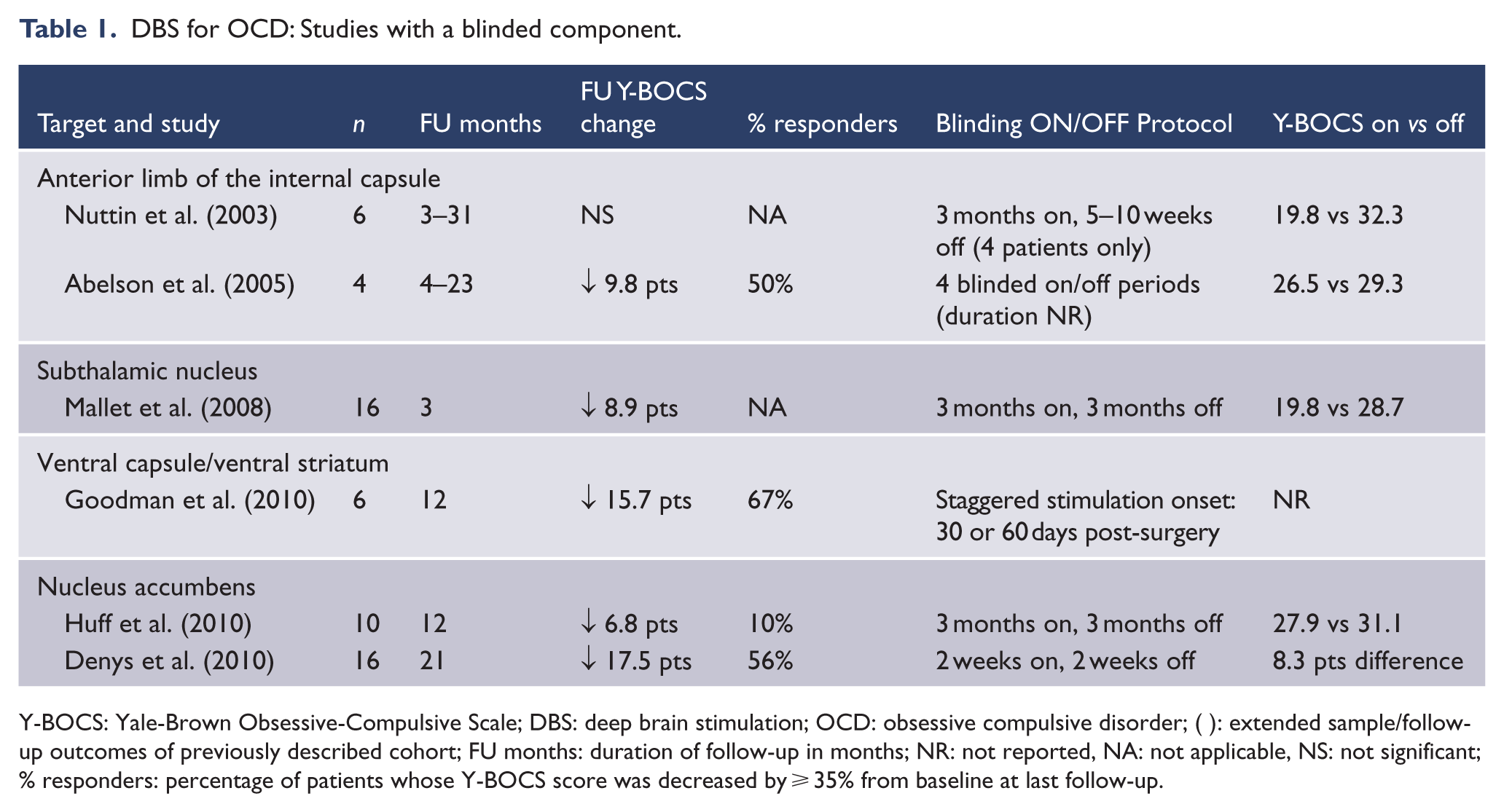
Y-BOCS: Yale-Brown Obsessive-Compulsive Scale; DBS: deep brain stimulation; OCD: obsessive compulsive disorder; ( ): extended sample/follow-up outcomes of previously described cohort; FU months: duration of follow-up in months; NR: not reported, NA: not applicable, NS: not significant; % responders: percentage of patients whose Y-BOCS score was decreased by ⩾ 35% from baseline at last follow-up.
ALIC and the ventral striatum/ventral capsule
One open-label case study and two small studies have described the efficacy of DBS applied to the ALIC in a total of 11 patients. The case study reported a marked reduction in OCD symptoms and improved psychosocial functioning 10 months post-stimulation onset (Anderson and Ahmed, 2003). The two subsequent studies included some periods of blinded ‘on – off’ stimulation. Limited detail on outcomes is provided in one report of six patients (Nuttin et al., 2003) but two of the four patients described in Abelson et al. (2005) were reported to be responders to treatment.
A larger number of reports have presented the results of stimulation at sites described as within the VC or VS. There is a significant overlap between the stimulation site in these studies and those ascribing implantation in the ALIC, with the localisation of stimulation typically several millimetres anterior to the anterior commissure and with a similar lateral and ventral localisation. There have been four open-label case series describing a total of 19 patients with response rates ranging from 40% to 100%. The largest study reported a 40% response rate in 10 patients followed for 3 years (Greenberg et al., 2006). Goodman et al. (2010) reported four out of six responders in a partially blinded study where the onset of stimulation was staggered in a blinded fashion. Data from a number of these studies were combined in a report in 2010 which describe the outcomes of a total of 26 patients implanted at the ALIC or VC/VS and followed for between 3 and 36 months (Greenberg et al., 2010). The overall response rate was 62%. In addition to a reduction in OCD severity, there was a substantial global reduction in severity of depression and generalised anxiety.
Importantly, as well as an improvement in core symptoms of psychopathology, there was a significant improvement in overall functioning in these patients. At last follow-up, 80% had demonstrated marked improvements in social and occupational functioning and ability to perform activities of daily living, with performance in each of these areas assessed as ‘fair to good’. No patients were reported to have deteriorated as a result of participation in DBS treatment and only 4 out of 26 were unchanged. Despite these impressive outcomes, it is notable that a significant proportion of patients continued to experience moderately severe symptoms of OCD in spite of DBS treatment, with only 38% achieving symptom scores considered consistent with remission. When it was performed, interruption of DBS stimulation was typically associated with a worsening of symptoms (Table 2).
DBS for OCD: open-label case reports and case series.
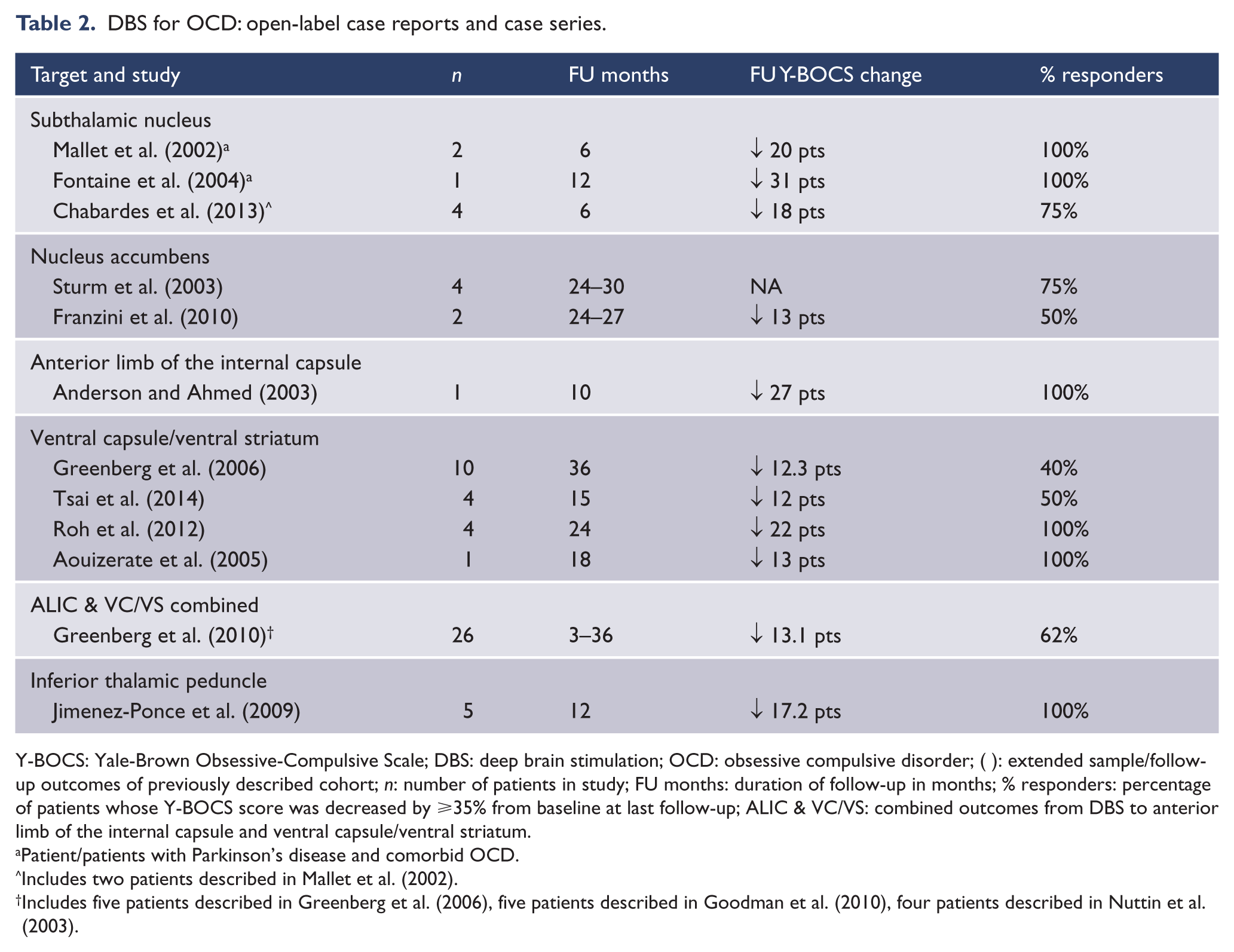
Y-BOCS: Yale-Brown Obsessive-Compulsive Scale; DBS: deep brain stimulation; OCD: obsessive compulsive disorder; ( ): extended sample/follow-up outcomes of previously described cohort; n: number of patients in study; FU months: duration of follow-up in months; % responders: percentage of patients whose Y-BOCS score was decreased by ⩾35% from baseline at last follow-up; ALIC & VC/VS: combined outcomes from DBS to anterior limb of the internal capsule and ventral capsule/ventral striatum.
Patient/patients with Parkinson’s disease and comorbid OCD.
Includes two patients described in Mallet et al. (2002).
Includes five patients described in Greenberg et al. (2006), five patients described in Goodman et al. (2010), four patients described in Nuttin et al. (2003).
A number of important further observations were made in this study. Specifically, it was notable that overall outcomes improved to a greater degree in patients implanted at later dates. This reflected a change in the site of DBS implantation to an area more posteriorly in the internal capsule. Patients implanted at this more posterior site were also noted to require substantially lower stimulation voltages to achieve therapeutic effects.
NA
A small number of reports have specifically targeted the NA rather than the more dorsal striatal or white matter regions. Sturm et al. (2003) described three out of four responders and Franzini et al. (2010) one out of two responders with open-label DBS. In addition, there have been two studies that were slightly larger numbers and blind periods of stimulation. In one of these studies, 10 patients received 3 months of active stimulation and 3 months of sham stimulation in a double-blind crossover fashion with subsequent open-label treatment and follow-up for 12 months (Huff et al., 2010). Despite this provision of double-blind stimulation, a direct quantitative analysis of outcomes across the double-blind phase was not provided. Five patients met partial response criteria by 12 months and significant improvements in quality of life and global functioning were reported.
The second study (Denys et al., 2010) involved 16 patients who received 8 months of open-label treatment followed by 2 weeks’ blocks of blinded active or sham stimulation. Nine patients responded in the open-label phase. Fourteen patients participated in the double-blind phase during which there was a significantly greater reduction in OCD symptoms with active stimulation. Small but significant functional improvements in work, social and family life were observed following the initial open-label phase and benefit in each of these areas had increased further at last follow-up.
STN
Interest in the STN as a potential target for DBS in OCD was sparked by observations of diminished compulsiveness in patients with Parkinson’s disease after undergoing implantation at this site. Mallet (2002) and Fontaine (2004) initially described promising results with open-label stimulation in a total of three patients with OCD and Parkinson’s disease, all of whom were responders.
Subsequently, in 2008 Mallet et al. reported outcomes of a multisite sham-controlled, double-blind, crossover trial of STN DBS in 16 patients with refractory OCD. Three months after surgery and following individualised determination of optimal stimulation parameters, patients were randomised to 3 months of active or sham (i.e. off) stimulation. Following a 1-month wash-out period (i.e. stimulation off), they crossed over to receive 3 months of the other condition. Under active stimulation, the severity of OCD symptoms was reduced and global functioning improved relative to sham. At the conclusion of 3 months of active stimulation, 75% of patients met response criteria in contrast to 38% following 3 months of sham stimulation. Neuropsychological measures, depression and anxiety were not modified by stimulation. This study reported a relatively high incidence of severe adverse effects, with one intracerebral haemorrhage (resulting in persistent contralateral hand palsy) and two hardware infections that necessitated explanation of the stimulators.
Most recently, Chabardes et al. (2013) described positive outcomes for two additional patients who experienced a 78% and 34% reduction in OCD severity following 6 months of persistent stimulation.
DBS in other psychiatric indications
Anorexia nervosa
A number of small recent studies have described the possible use of DBS in the treatment of anorexia nervosa. A case report of the treatment of a 56-year-old woman with comorbid depression and anorexia, where DBS stimulation occurred in the SAC (Israel et al., 2010), had shown improvement in symptoms of anorexia which appeared at least partially independent of improvement in depression. A subsequent report has described outcomes for six patients stimulated at the same site (Lipsman et al., 2013). Four of the six patients had substantially improved functional symptoms at 9 months, including in mood, with three appearing to have had a substantial improvement in core symptoms of anorexia with associated improvement in weight. Patients whose symptoms of anorexia improved also reported a greatly increased quality of life.
Another report described the outcomes of four patients treated with DBS applied to the NA (Wu et al., 2013). All four patients appeared to achieve a substantial improvement in symptoms of anorexia with an average follow-up period of over 3 years (Tables 3 and 4).
DBS for TRD: Studies with a blinded component.
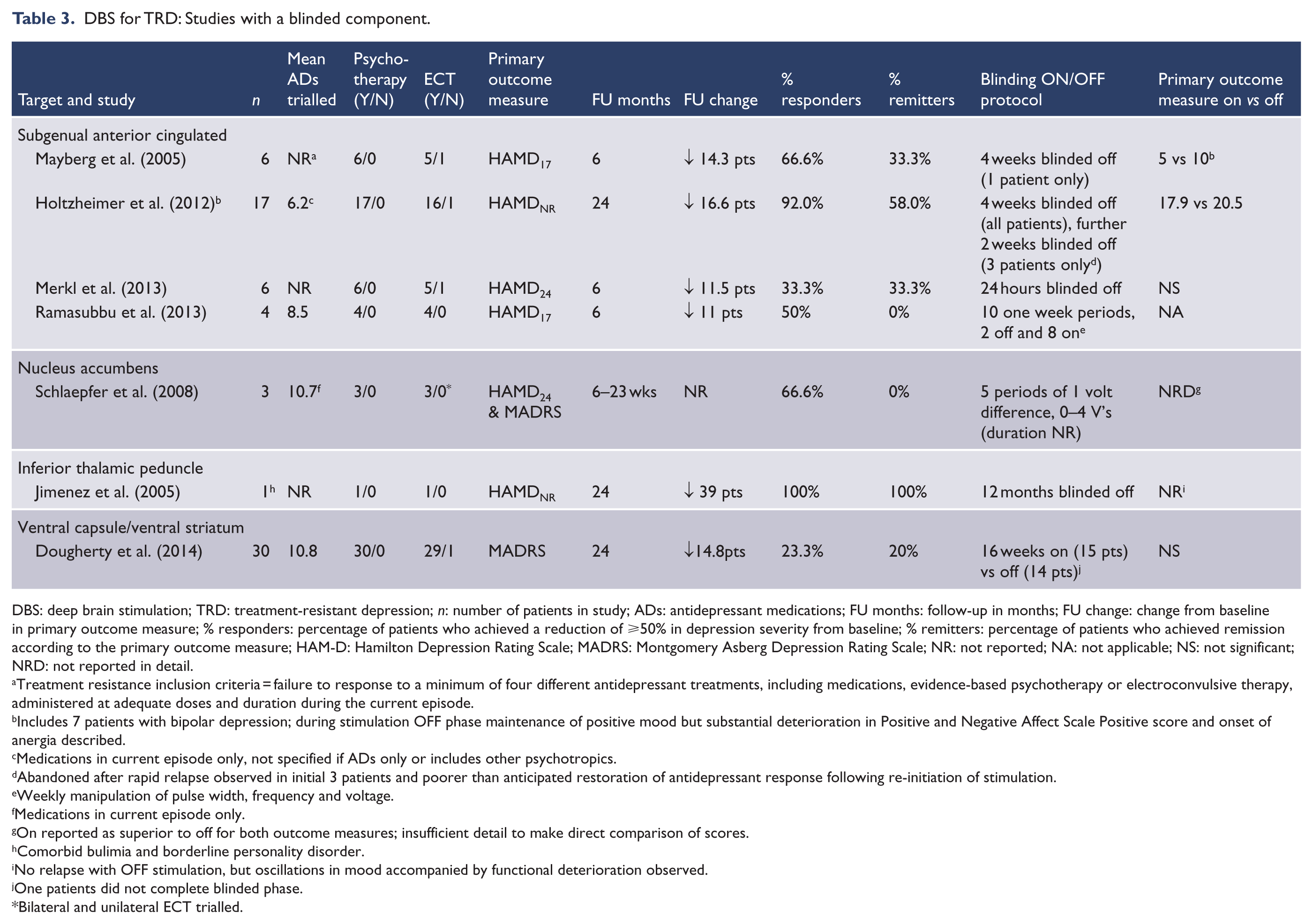
DBS: deep brain stimulation; TRD: treatment-resistant depression; n: number of patients in study; ADs: antidepressant medications; FU months: follow-up in months; FU change: change from baseline in primary outcome measure; % responders: percentage of patients who achieved a reduction of ⩾50% in depression severity from baseline; % remitters: percentage of patients who achieved remission according to the primary outcome measure; HAM-D: Hamilton Depression Rating Scale; MADRS: Montgomery Asberg Depression Rating Scale; NR: not reported; NA: not applicable; NS: not significant; NRD: not reported in detail.
Treatment resistance inclusion criteria = failure to response to a minimum of four different antidepressant treatments, including medications, evidence-based psychotherapy or electroconvulsive therapy, administered at adequate doses and duration during the current episode.
Includes 7 patients with bipolar depression; during stimulation OFF phase maintenance of positive mood but substantial deterioration in Positive and Negative Affect Scale Positive score and onset of anergia described.
Medications in current episode only, not specified if ADs only or includes other psychotropics.
Abandoned after rapid relapse observed in initial 3 patients and poorer than anticipated restoration of antidepressant response following re-initiation of stimulation.
Weekly manipulation of pulse width, frequency and voltage.
Medications in current episode only.
On reported as superior to off for both outcome measures; insufficient detail to make direct comparison of scores.
Comorbid bulimia and borderline personality disorder.
No relapse with OFF stimulation, but oscillations in mood accompanied by functional deterioration observed.
One patients did not complete blinded phase.
Bilateral and unilateral ECT trialled.
DBS for TRD: open-label reports and case-series.
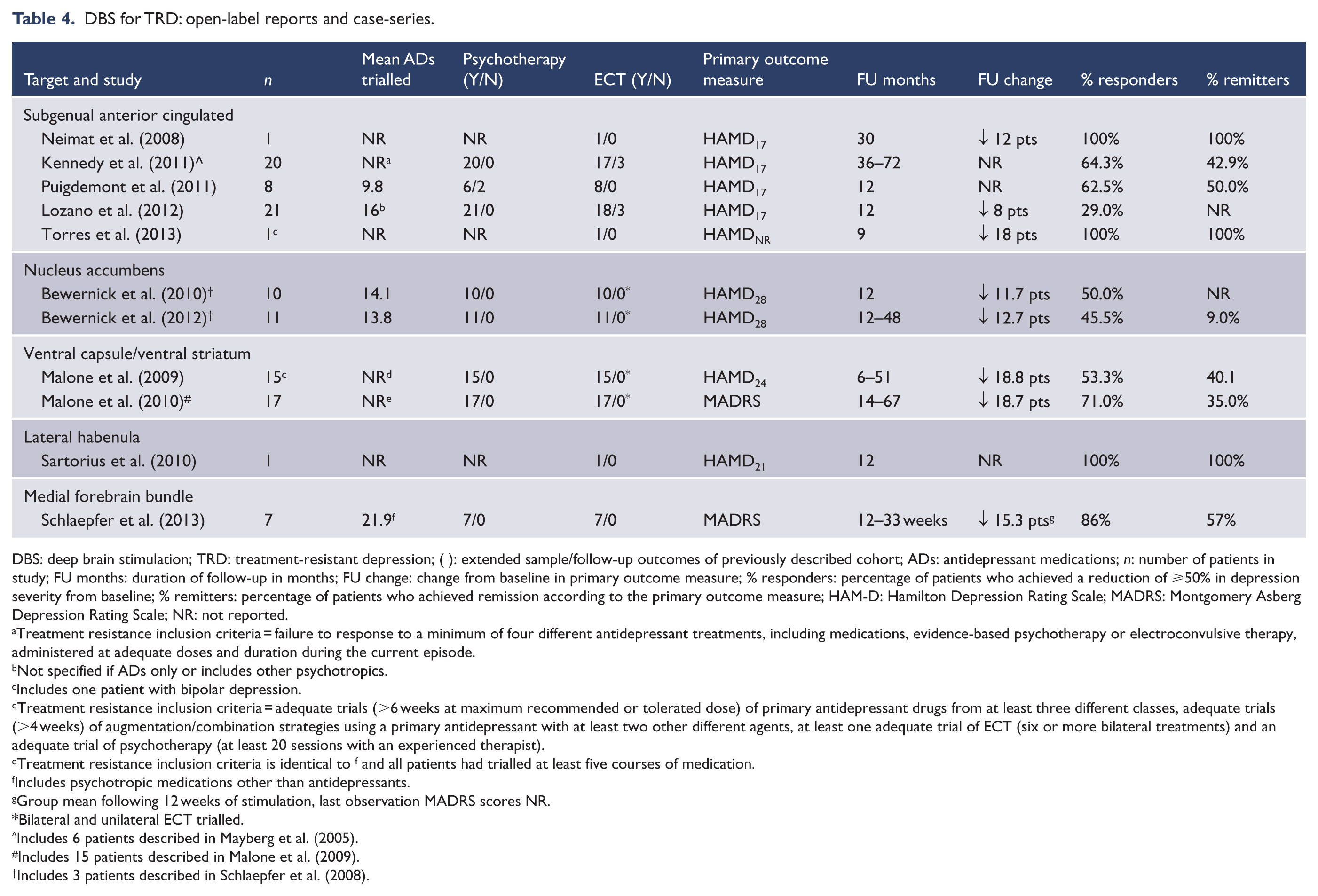
DBS: deep brain stimulation; TRD: treatment-resistant depression; ( ): extended sample/follow-up outcomes of previously described cohort; ADs: antidepressant medications; n: number of patients in study; FU months: duration of follow-up in months; FU change: change from baseline in primary outcome measure; % responders: percentage of patients who achieved a reduction of ⩾50% in depression severity from baseline; % remitters: percentage of patients who achieved remission according to the primary outcome measure; HAM-D: Hamilton Depression Rating Scale; MADRS: Montgomery Asberg Depression Rating Scale; NR: not reported.
Treatment resistance inclusion criteria = failure to response to a minimum of four different antidepressant treatments, including medications, evidence-based psychotherapy or electroconvulsive therapy, administered at adequate doses and duration during the current episode.
Not specified if ADs only or includes other psychotropics.
Includes one patient with bipolar depression.
Treatment resistance inclusion criteria = adequate trials (>6 weeks at maximum recommended or tolerated dose) of primary antidepressant drugs from at least three different classes, adequate trials (>4 weeks) of augmentation/combination strategies using a primary antidepressant with at least two other different agents, at least one adequate trial of ECT (six or more bilateral treatments) and an adequate trial of psychotherapy (at least 20 sessions with an experienced therapist).
Treatment resistance inclusion criteria is identical to f and all patients had trialled at least five courses of medication.
Includes psychotropic medications other than antidepressants.
Group mean following 12 weeks of stimulation, last observation MADRS scores NR.
Bilateral and unilateral ECT trialled.
Includes 6 patients described in Mayberg et al. (2005).
Includes 15 patients described in Malone et al. (2009).
Includes 3 patients described in Schlaepfer et al. (2008).
Addiction
A small literature has also explored the potential use of DBS for the treatment of addictive disorders, primarily with a focus on stimulation of the NA reward system. There have been a number of individual case reports suggesting remission of symptoms in the treatment of alcohol and heroin addiction (Kuhn et al., 2007; Valencia-Alfonso et al., 2012) and a small case series (Muller et al., 2009). In this latter report, Muller et al. (2009) described the successful treatment of three patients with alcohol dependence. Stimulation of the NA resulted in a dramatic reduction in alcohol craving that resulted in abstinence in two patients, and a significant reduction of use in a third.
A further study has reported on rates of nicotine dependence in patients receiving NA stimulation for other disorders including Tourette’s syndrome, OCD or anxiety disorders (Kuhn et al., 2009). This study reported slightly higher rates of successful smoking sensation compared to what would typically be expected across the same time points.
Safety and adverse effects
There are two major types of safety concerns associated with administration of DBS, those related directly to the neurosurgical procedure and implanted device hardware, and adverse effects arising from the stimulation itself.
Procedure and device-related adverse events
Concerns directly related to the neurosurgical procedure are similar when DBS is being applied for a psychiatric indication as to when it is being used for neurological disorder. Procedure-related adverse effects include haemorrhage (1–2% of procedures), seizure induction (<1%, usually in the first 24 hours following implantation), infection (2–3%, usually superficial) and other general surgical or anaesthetic complications (Benabid, 2003; Greenberg and Rezai, 2003; Hariz, 2002). It is worth noting that different target sites in the brain do require different DBS implantation trajectories and it is possible that higher rates of haemorrhage could result from implantation sites in less accessible brain regions, for example, where there are a greater number of blood vessels in the immediate region. However, there is no evidence to date that this impinges on the overall safety of DBS at the major targets used to treat psychiatric disorders.
In general, the procedure-related complication rate is dependent on the competency and experience of the surgical team and the rate of DBS complications are typically low and usually mild and reversible (Benabid, 2003). In one case series of 60 patients who underwent DBS for Parkinson’s disease, only one patient was left with any deficit associated with a surgical complication, a mild aphasia resulting from a small haemorrhage (Lagrange et al., 2002).
Device-related complications can occur and include lead fracture and malfunction of the neurostimulator. Both have been reported in relation to DBS for psychiatric indications (Bewernick et al., 2010; Malone et al., 2009; Okun et al., 2007), but with advances in device technology these are likely to be increasingly uncommon and there is no evidence to suggest that occurrence rates differ from DBS for neurological indications.
Stimulation-related adverse events
Adverse effects arising directly from stimulation itself are common and strongly linked to the neuroanatomical site of stimulation. They most often occur at contacts and stimulation parameters that are not optimal for therapeutic benefit and are elicited during the ‘trial-and-error’ style search for optimal stimulation settings. Stimulation-induced side effects are frequently transient and rapidly reversible with adjustment or cessation of stimulation.
Transient induction of hypomania and agitation/anxiety are two of the most commonly reported stimulation-related side effects and both have been induced via stimulation at a number of brain regions (Denys et al., 2010; Dougherty et al., 2014; Goodman et al., 2010; Greenberg et al., 2006; Huff et al., 2010; Malone et al., 2009). Insomnia is common following voltage increases and can necessitate temporary increases in sedative medications (Goodman et al., 2010). In our experience, standard hypnotic medications including benzodiazepines are frequently ineffective under these circumstances: more potent sedatives such as quetiapine may be required. Temporary worsening of other psychiatric symptoms such as further reduced mood, suicidal ideation, increased irritability and obsessionality can also occur and patients should be monitored closely for these (Dougherty et al., 2014; Greenberg et al., 2010; Lozano et al., 2008; Malone et al., 2009).
Sensory disturbances such as tingling/pin-and-needles, hot and cold flushes, metallic tastes and other gustatory and olfactory experiences can occur. Autonomic symptoms such as nausea, dizziness, sweating and changes in blood pressure have also been described following stimulation at numerous implantation sites (Bewernick et al., 2012; Greenberg et al., 2010; Jimenez-Ponce et al., 2009; Okun et al., 2007). Motor effects include oculomotor disturbance, orofacial muscle contractions and temporary motor slowing (Greenberg et al., 2010; Schlaepfer et al., 2013).
With respect to cognition, transient confusion, memory disturbance and verbal perseveration have been described in response to certain DBS parameters. These have typically been temporary and there is no evidence of persistent cognitive impairment or related functional decline (Greenberg et al., 2010; Grubert et al., 2011; McNeely et al., 2008; Malone et al., 2009). There are, however, relatively few dedicated studies that have examined neuropsychological outcomes in detail.
Stimulation OFF effects have also been described – this refers to the sudden return of psychiatric symptoms if the device is turned off or malfunctions (Greenberg and Rezai, 2003; Goodman et al., 2010). This is a common occurrence in patients who obtained therapeutic benefit from DBS when their stimulator battery becomes depleted. Early DBS devices had a battery life of approximately 18–24 months (dependent on individual stimulation parameters) before replacement via surgical day procedure was required. Recently re-chargeable batteries have been developed. These allow patients to charge their battery using an external re-charger and device manufacturers estimate these models will run for up to 10 years before replacement is required.
Suicide risk following DBS
There have been a number of suicide attempts and completed suicides following DBS for psychiatric indications (Bewernick et al., 2012; Dougherty et al., 2014; Greenberg et al., 2010; Kennedy et al., 2011). Without exception, all patients undergoing psychiatric DBS suffer from chronic severe mental illness and are at substantially elevated risk of suicide prior to surgery. The suicidality observed in these individuals’ post-DBS was not associated with acute alterations in stimulation parameters and is not thought to be related to the procedure itself, but rather a consequence of severe psychiatric disease that has not responded to what is seen as a treatment of last resort (Bewernick et al., 2010; Dougherty et al., 2014; Kennedy et al., 2011).
Discussion
The development of DBS as a psychiatric treatment poses a number of very unique challenges and requires careful consideration of a range of clinical and ethical factors, some of which we will discuss here.
In regard to clinical efficacy, this review reveals that the literature is almost exclusively a series of case reports and open-label series. Several multisite (negative) industry-sponsored studies have been conducted in depression but the results of these have not been published. The published reports are mostly positive with response rates most typically of ~50% for both depression and OCD.
One important factor when considering the efficacy of DBS for psychiatric indications is implantation site, especially given that several have been targeted for the conditions described above. Clearly, caution is warranted when considering the relationship between clinical outcomes for a particular illness and implantation at different anatomical targets, as currently there is insufficient evidence to ascertain whether DBS at one site is clinically superior to another. The variance in reported efficacy between individual studies and disparate brain regions likely reflects the influence of the small number of patients in each sample, heterogeneity of patient characteristics, differing levels of treatment resistance, widely divergent follow-up durations and differences in research design and outcome measures. Until specifically designed head-to-head clinical comparator trials of DBS at different implantation targets are conducted, the superiority of one site over another cannot not be confidently determined for any indication. In addition, as with DBS for movement disorders, it may be that certain implantation sites are more efficacious for patients with specific types of symptoms. However, confirmation of this possibility awaits future dedicated research trials.
There are several substantive gaps in the DBS evidence base. There is an absence of published substantive medium to long-term double-blind, randomised controlled trials. Given the complexity of conducting this type of research, it is unlikely that a significant number of these trials will be conducted in the near future and conclusions will need to be drawn from mostly small studies and possibly one or two large multicentre research efforts. There are a number of specific areas in which further research is required beyond the basic establishment of efficacy:
Patient characteristics. Little information is currently available as to patient characteristics that may predict successful response to DBS treatment. Studies have included insufficient samples to allow an exploration as to the differential effectiveness of DBS in illness subtypes. In the context of depression, research should investigate the relationship between melancholia and treatment response. In OCD, future studies are required to explore whether subtypes of disorder, for example hoarding, are differentially more or less responsive to treatment.
Level of treatment resistance. All patients included in OCD and depression trials to date have been considered to have some degree of treatment resistance: that is, they have failed to respond to other treatment modalities. However, the degree of treatment resistance varies substantially across studies. For example, some studies require patients to have exhausted an extensive range of antidepressant strategies. Others have not required patients to have previously tried treatments with established efficacy, such as electroconvulsive therapy.
Stimulator programming and stimulation parameters. There is marked heterogeneity in the procedures used to determine optimal DBS stimulation settings. In some studies, these are set by a nonflexible fixed protocol. In other studies, stimulation levels are determined based upon an initial interrogation of multiple stimulation settings and adjusted frequently across time. It is possible that different protocols will suit different clinical applications and may be associated with different trajectories of clinical response. It is clear that programming of the DBS device should be undertaken by a clinician with a comprehensive understanding of the technical aspects of stimulation parameters and clinical expertise in the disorder in question. The optimal timing of programming changes is also completely unclear (and may vary by implantation site and disorder). Although short-term mood effects can be produced with DBS stimulation changes, it is not clear if these predict longer term outcomes. The optimal duration of time between stimulation adjustments could possibly vary between minutes and weeks/months.
The complexity of programming also relates to the complexity of conducting double-blind trials of DBS procedures. If fixed settings are used in a double-blind (ON or OFF) manner, this may undermine the benefit of the procedure by not allowing the best parameters to be established. If a full interrogation of parameters occurs prior to randomisation to establish the optimal settings, patients become accustomed to the experience of DBS making subsequent blinding problematic. Many patients will also be unblinded in a randomised withdrawal design. As we develop an enhanced understanding of the time course of therapeutic responses to DBS, some of these problems may be letter addressed. The challenge will be in designing trials that are feasible but allow sufficient time for neuroplastic responses to stimulation.
It is notable that DBS has been reported to be effective when utilised by highly experienced researchers who have pioneered this technique. The multisite studies that have failed to replicate these effects may have been impacted by methodological inconsistencies during the rollout of this technique resulting in lower response rates. If this is the case, however, the difficulties of technique translation must be considered in weighing up how this approach could be successfully transitioned into clinical practice.
Clinical management of patients undergoing DBS for a psychiatric indication can be an extremely complex and lengthy process. Expertise from numerous clinical disciplines is required to both monitor for adverse events and maximise potential for therapeutic benefit. There is a general consensus among many of the larger and more experienced research groups in this area that optimal care is provided by multidisciplinary treating teams that include expert input from psychiatry, neurosurgery, clinical psychology and neuropsychology (Nuttin et al., 2014; Ooms and Denys, 2014).
It also appears important that follow-up is provided for a protracted period of time following surgical implantation. Adjustment of DBS parameters can take very long periods of time (months to years) to optimise. Patients need to be monitored to ensure that maximal clinical outcomes are achieved and to monitor for the emergence of stimulation-related adverse events. It also appears likely that optimal outcomes are obtained when post-operative management includes recovery-oriented psychological therapy in order to translate reductions in symptom severity into functional improvement in day-to-day life (Holtzheimer and Mayberg, 2010). In our experience, patients, sometimes those who have the greatest therapeutic response, encounter significant problems re-adapting to daily life and require considerable psychological support to facilitate this process. This can include family based or relationship therapy to help families adjust to the wellness of an individual and their desire to re-establish previously relinquished roles.
Finally, advancing the use of DBS in the treatment of psychiatric conditions is likely to require a better understanding of its fundamental mechanism of action and how this can be best adapted to the treatment of relevant disorders. An approach which just extrapolates from the use of DBS in conditions like Parkinson’s disease is likely to be insufficiently sophisticated to produce optimal outcomes. Although generally the field of DBS is moving from an approach which considers it a ‘reversible lesion’ to consider DBS stimulation in some way ‘functional’, our understanding of this remains limited. Given the increasing recognition of the importance of oscillatory activity in certain frequencies underlying specific cognitive functions, we await models that can integrate this information to better construct DBS protocols that are likely to have the greatest therapeutic impact.
Conclusions
DBS is a promising and emerging form of treatment for a range of neurological and psychiatric disorders. There is no evidence that the use of DBS in psychiatric disorders is associated with greater risks than when this treatment is used for the treatment of neurological conditions. However, insufficient studies have been conducted to date to establish its effectiveness or to comprehensively characterise its safety. Therefore, the use of DBS treatment cannot be recommended at this stage in the clinical management of any psychiatric disorder. The provision of DBS for psychiatric indications should only occur for patients who are enrolled in a clinical trial. The conduct of this type of research should only be done by adequately experienced and trained multidisciplinary clinical research teams. At a minimum, this should include individuals with expertise in neurosurgery, psychiatry and neuropsychology. DBS implantation should be done by a neurosurgeon with substantive expertise in stereotactic DBS procedures, and who is currently performing these operations for other indications on a regular basis. The programming of DBS device should be conducted under the close supervision of a psychiatrist with substantial experience in the clinical management of patients with the disorder being treated.
Given that DBS is not a proven treatment at this stage, clearly the provision of DBS should only be provided to individuals who are able to fully consent to undertaking the procedure and are cognisant of the relative risk/benefit ratio. It is our opinion that the consent of these patients and their individual suitability for DBS treatment should be reviewed by an independent body prior to commencement of any DBS procedure. This review process should consider the psychiatric appropriateness of the proposed research-based treatment, the neurosurgical expertise of the individual performing the DBS procedure, the quality of the team providing long-term care for the patient and the patient’s capacity to provide informed consent to and engage in the treatment process. This review should be of each individual patient undergoing the procedure and being in addition to formal ethics review of the research protocol in which DBS is being performed.
Footnotes
Acknowledgements
The authors are grateful to Rodney Anderson for constructing the two DBS implantation target figures.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Funding
P.B.F is supported by a NHMRC Practitioner Fellowship (606907). P.B.F. has received equipment for research from MagVenture A/S, Medtronic Ltd, Cervel Neurotech and Brainsway Ltd and funding for research from Cervel Neurotech. R.A.S. is supported by a NHMRC Early Career Researcher Fellowship (1036201). Support for the evidence review in this paper was provided by the NSW Ministry for Health through the Sax Institute and by a NHMRC project grant (APP1077859).
