Abstract
Objective:
Deep brain stimulation is an experimental intervention for treatment-resistant depression. Open trials have shown a sustained response to chronic stimulation in many subjects. However, two recent randomised, double-blind, placebo-controlled trials failed to replicate these results. This article is a conceptual paper examining potential explanations for these discrepant findings.
Method:
We conducted a systematic review of the published studies obtained from PubMed and PsycINFO. Studies were selected if they directly examined the impact of deep brain stimulation on depressive symptoms. We excluded case reports and papers re-describing the same cohort of patients. We compared them with data from the placebo-controlled trials, available from Clinicaltrials.gov and abstracts of the American Society for Stereotactic and Functional Neurosurgery. We supplemented our investigation by reviewing additional publications by the major groups undertaking deep brain stimulation for mood disorders.
Results:
We selected 10 open studies reporting on eight cohorts of patients using four different operative targets. All published studies reported positive results. This was not replicated in data available from the randomised, placebo-controlled trials. Many studies reported suicide or suicide attempts in the postoperative period.
Conclusion:
We consider the placebo effect, the pattern of network activation, surgical candidacy and design of a blinded trial including the length of a crossover period. We suggest a greater focus on selecting patients with melancholia. We anticipate that methodological refinements may facilitate further investigation of this technology for intractable depression. We conclude by noting the psychiatric adverse events that have been reported in the literature to date, as these will also influence the design of future trials of deep brain stimulation for depression.
Introduction
Major depression is a disorder of mood, motivation, cognition and vegetative functioning that runs an episodic, recurrent course across the lifespan. In Australia, the 12-month prevalence of major depression ranges from 4% to 10%, depending on the assessment instrument chosen (Australian Bureau of Statistics, 2008; Goldney et al., 2010). Of those individuals with an affective episode in the previous year, half are classified as severe (Slade et al., 2009). In North America, major depression is the fifth leading source of disability-adjusted life years (DALYs) (Ferrari et al., 2013), even when excluding deaths attributable to suicide. By 2020, depression is expected to be second only to cardiovascular disease in global burden (Murray and Lopez, 1997).
Treatment-resistant depression (TRD) refers to cases in which physical treatments, such as antidepressant medication or electroconvulsive therapy (ECT), fail to produce complete remission. The fact that residual symptoms are associated with heightened risk of relapse (Judd et al., 1998) and poorer social functioning (Paykel, 2002) has prompted clinicians to trial alternative strategies to restore patients to their premorbid level of functioning.
TRD is variously defined (Fava, 2003), based on the number, duration and intensity of medication trials; the use of antidepressants from different classes; antidepressant augmentation with antipsychotic medication; and the use of ECT or other neuromodulatory therapies. In a naturalistic setting, over 30% of patients fail to remit despite sequential treatment with combination drug therapy (Rush et al., 2006). ECT leads to remission in as many as 75% of depressed individuals (Husain et al., 2004). However, side effects (most notably retrograde amnesia; Donahue, 2000; Lisanby et al., 2000) and social disapproval limit its use.
Deep brain stimulation
Deep brain stimulation (DBS) is a minimally invasive method of brain manipulation, with a long history of successful use in neurological conditions such as epilepsy, chronic pain and Parkinson’s disease (PD) (Benabid et al., 1994; Gildenberg, 2005). Using the principles of stereotactic functional neurosurgery, electrodes are precisely sited bilaterally in a deep brain nucleus and connected to an implantable pulse generator (IPG) that delivers continuous high-frequency stimulation at the surgical target. Stimulation parameters can be adjusted in the postoperative period to vary the size and shape of the stimulation field, the intensity of the stimulation, as well as the frequency and width of the stimulation pulses.
Electrodes are termed quadripolar because they possess four contact points, each of which can be programmed as the active (cathodal) contact. Spacing between contacts may vary between lead specifications. If the IPG case is designated as the anode (monopolar stimulation), a spherical electrical field is created around the stimulating contact. In bipolar stimulation, a second contact point is used as the anode, and this creates a smaller, more focussed electrical field. This may be used to avoid diffusion of current into neighbouring neural elements, a process that is also influenced by the characteristics (impedance) of the neural tissue itself.
At the level of the neuron and neurocircuit, the mode of action of DBS is complex and not fully understood. Negatively charged ions are injected into brain tissue at the active contact and move in the extracellular space. These can have a variable effect on the membrane potential of individual neurons depending on their position and orientation with the stimulation field. Direct axonal activation may excite neurons or inhibit activity through activation of inhibitory afferents. Early assumptions that DBS creates a reversible lesion, or introduces a ‘correct’ pattern of neuronal discharges, have given way to the more sophisticated theory that DBS modulates pathological network activity such as the burst firing of neurons seen in animal models of PD (Vila et al., 2000).
DBS for depression
The successful use of DBS in neurological disorders has raised the prospect of the use of DBS in the treatment of intractable psychiatric conditions. This has been driven not only by an underlying biological, neuroscientific, circuit-based model of severe mental disorder (Haber and Knutson, 2010) but also by the history of functional neurosurgery for psychiatric disorders. Prior to DBS, stereotactic ablative neurosurgery was a ‘last resort’ treatment for selected cases of intractable depression. Using diathermy, cryoablation or radioactive seeding, lesions were sited in the anterior limb of the internal capsule (anterior capsulotomy), the cingulate gyrus (anterior cingulotomy) or the region posterior to the orbitofrontal cortex (OFC) (subcaudate tractotomy). These targets were developed empirically, but the early evidence supporting them has been criticised, particularly a reporting bias in favour of positive outcomes (Matthews and Eljamel, 2003).
The analysis of later cohorts of patients treated with ablative neurosurgery was more promising (Christmas et al., 2011) and was supported by the finding that major depression is associated with neurobiological abnormalities in these surgical sites or their key projection areas. Accordingly, DBS for depression is now based on a model of cortico-striatal and cortico-limbic network dysfunction. Evidence accrues from the incidence of depression in neurological conditions such as PD, Huntington’s disease (HD) and cerebrovascular disease (CVD) affecting fronto-striatal regions, pointing to a common antecedent neural pathway in these ‘secondary’ mood disorders that is activated by various pathobiologic mechanisms (Cummings, 1993; Mayberg, 1994). In patients with ‘primary’ depressive disorders, positron emission topography (PET) identifies discrete areas of hypometabolism in the cingulate and prefrontal cortices (Bench et al., 1992; Drevets et al., 1997). Subgenual cingulate hypermetabolism has also been observed (Mayberg et al., 1999). Disturbed metabolism normalises with successful treatment of the mood disorder (Mayberg et al., 2000, 2005). Modern tractographic imaging identifies common overlapping connectivities between the surgical sites, suggesting the existence of a shared affective system (Schoene-Bake et al., 2010) that can be targeted with neuromodulation. DBS for PD has demonstrated that neuromodulation of a single subcortical site can induce disseminated network changes (Benabid et al., 2002).
The advantages of DBS over lesion surgery are significant. DBS is adjustable, non-destructive and reversible. Once implanted, the device can be turned on and off, permitting a crossover study of its effects within an individual patient. This approach has been used to demonstrate the efficacy of DBS for treatment-resistant obsessive–compulsive disorder (OCD) (Denys et al., 2010).
Objective
Our aim of this paper was to systematically review the data on DBS for TRD in the light of two recent randomised, double-blind, placebo-controlled trials (RCTs) that failed to differentiate active from sham treatment. Extending on existing published analyses, we wished to generate hypotheses for the failure of these trials. Our goal was to suggest refinements in patient selection and protocol design that might increase the likelihood of detecting an effect of active treatment. This is therefore a conceptual paper and not merely a restatement of data already available in the literature. We do not believe it is premature to speculate in this manner prior to the publication of these results in the peer-reviewed literature, although clearly this places limitations on the precision of any conclusions that can be drawn. Publication bias against negative results from clinical trials often delays or even prevents full publication from occurring (Johnson and Dickersin, 2007).
We also sought to document the adverse events associated with DBS for depression, with a focus on psychiatric issues. These also bear on the design of future clinical trials and should be considered by investigators.
Method
We searched the PubMed and PsycINFO databases using the following search terms: ‘depression’ AND ‘deep brain stimulation’ AND ‘treatment resistant’. We returned 135 and 175 results, respectively. Only those studies that directly examined the impact of DBS on depressive symptoms were selected for in-depth review. We excluded case reports as our interest was in larger series at academic centres. Some papers re-presented data pertaining to the same cohort of patients. As our aim was not to produce an exhaustive summary of every article, we opted to consolidate our selection to the one or two most significant, comprehensive or recent reports per cohort. We included 10 articles reporting on eight patient cohorts (Bewernick et al., 2012; Holtzheimer et al., 2012; Kennedy et al., 2011; Lozano et al., 2008, 2012; Malone, 2010; Malone et al., 2009; Merkl et al., 2013; Puigdemont et al., 2012; Schlaepfer et al., 2013). For an un-distilled systematic review that includes case reports, the reader is directed to a recent paper by Morishita et al. (2014). We did not perform a meta-analysis as the small number of studies and limited sample size make this premature. Two RCTs of DBS for TRD were identified by searching the US National Institutes of Health clinical trials registry (ClinicalTrials.gov). Available data from these trials were sourced from published conference abstracts of the American Society for Stereotactic and Functional Neurosurgery (Mayberg, 2014; Rezai, 2012). A more comprehensive report of one of these trials is now in press (Dougherty et al., 2015). We identified one further placebo-controlled, double-blind study that was not included in the main analysis (Schlaepfer et al., 2008). This was a small (three patients) pilot study of the immediate and short-term effects of varying stimulation intensity on depressive symptoms.
We also reviewed additional publications by the groups undertaking DBS for mood disorders that were returned in the initial database search. We selected three such articles to support our analysis (Johansen-Berg et al., 2008; Lujan et al., 2013; Riva-Posse et al., 2014).
Results
Scientific issues
Open trials
The current literature reports on eight cohorts of depressed patients who have undergone DBS as part of an open trial. The characteristics of these trials are summarised in Table 1. There are four operative targets described:
The subgenual cingulate white matter or subcallosal cingulate gyrus (Cg25WM) (Holtzheimer et al., 2012; Kennedy et al., 2011; Lozano et al., 2008, 2012; Mayberg et al., 2005; Merkl et al., 2013; Puigdemont et al., 2012);
The anterior limb of the internal capsule (ALIC) or ventral capsule/ventral striatum (VC/VS) (Malone, 2010; Malone et al., 2009);
The nucleus accumbens (NAcc) (Bewernick et al., 2012) (ventral contacts similar to ‘2.’, but dorsal contacts differ due to shorter distance between contacts);
The superolateral branch of the medial forebrain bundle (slMFB) (Schlaepfer et al., 2013).
Summary of the results from published open trials of DBS for TRD.
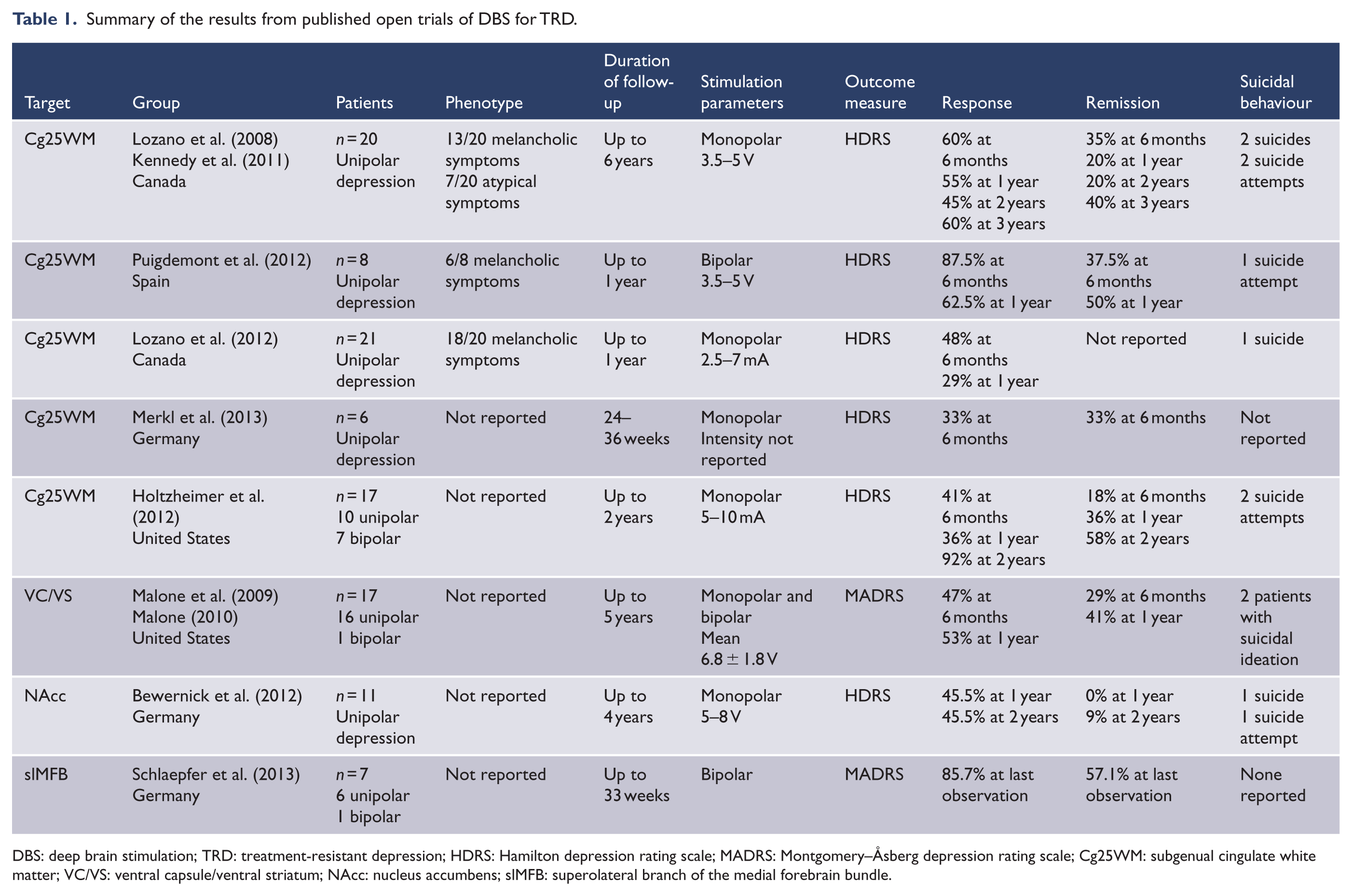
DBS: deep brain stimulation; TRD: treatment-resistant depression; HDRS: Hamilton depression rating scale; MADRS: Montgomery–Åsberg depression rating scale; Cg25WM: subgenual cingulate white matter; VC/VS: ventral capsule/ventral striatum; NAcc: nucleus accumbens; slMFB: superolateral branch of the medial forebrain bundle.
All trials only included patients with TRD. Patients had a chronic depressive disorder or an illness with multiple relapses, and in most, the current episode had lasted greater than 12 months. Patients also met minimum criteria for the severity of the current episode, assessed using a depression rating scale such as the Hamilton (HDRS) or Montgomery–Åsberg (MADRS).
Some studies reported the clinical features of the depressive phenotype (melancholic vs atypical). Two studies included patients with bipolar depression (Holtzheimer et al., 2012; Schlaepfer et al., 2013). Patients were excluded if they had non-affective psychosis, a significant personality disorder, a substance use disorder (except tobacco) and had made numerous or recent suicide attempts.
All published trials reported positive findings. Intraoperative responses to electrical stimulation at the target site included ‘connectedness’ (Mayberg et al., 2005) and ‘increased appetitive motivation’ (Schlaepfer et al., 2013), although these responses were not predictive of long-term response. At 6 months postoperation, approximately half the patients showed a positive response to DBS (measured by a >50% reduction in score on the HDRS or MADRS). Fewer were in remission (defined by scoring in the non-depressed range on these scales). The onset of treatment response was relatively slow, but once established appeared stable, with the response remaining constant or even increasing over greater lengths of follow-up (see Table 1). The exception was slMFB DBS, in which a clinically significant response was seen after 1 week (Schlaepfer et al., 2013). This rapid response was hypothesised to reflect the connectivity of this fibre bundle within the affective circuit. Overall, the observed response-rate and stability are remarkable, given the refractoriness of depression in the sample and given that relapse is the rule among non-resistant depressed patients who have been treated with conventional therapies.
Some studies contained elements of blinding or sham stimulation. Mayberg et al. (2005) blinded patients during initial stimulation optimisation. After 6 months, one remitter then entered a single-blind crossover phase OFF–ON stimulation. Holtzheimer et al. (2012) included a sham lead-in phase in 11 patients and a single-blind discontinuation phase in 3 patients. Merkl et al. (2013) used a randomised, placebo-controlled method to select the optimal electrode contacts. Patients and raters were later blinded to stimulation parameters in the follow-up phase, while two patients in the cohort were assigned to a delayed onset of stimulation.
Randomised, placebo-controlled, double-blind trials
Both major companies in the neuromodulation field have undertaken RCTs. Participants were randomised to either active or sham stimulation before entering an open phase in which all received active treatment.
In the Reclaim trial (Medtronic, ClinicalTrials.gov identifier NCT00837486), 29 participants were treated with VC/VS DBS and completed a randomised, double-blind, 4-month phase of active or sham treatment. At this point, 3/15 (20%) responded to active stimulation and 2/14 (14.3%) responded to sham stimulation. The mean (standard deviation [SD]) reduction in MADRS scores was 19.6% (34.9%) for active and 24.6% (28.8%) for sham stimulation (Dougherty et al., 2015; Rezai, 2012). At 12 months, by which time all participants had received at least 8 months of open-label stimulation, six met response criteria, of whom four met remission criteria. The mean reduction in MADRS was 24.4% (33.8%). At 24 months, seven participants met response criteria, of whom six were in remission. The mean reduction in MADRS was 40.2% (34.4%). In contrast to the open trials, the authors report that no participant who met responder criteria maintained this for the duration of the follow-up period. Only five patients held responder status for a 6-month period. Based on a power analysis, the investigators initially planned to enrol 208 patients. However, they report that enrolment was ceased following the results from this initial cohort of patients. The authors note that their study is consequently underpowered to detect a significant difference between active and sham treatment. The results of this trial are summarised in Table 2.
Summary of the results from the published RCT of DBS for TRD.

RCT: randomised, double-blind, placebo-controlled trial; DBS: deep brain stimulation; TRD: treatment-resistant depression; MADRS: Montgomery–Åsberg depression rating scale; VC/VS: ventral capsule/ventral striatum.
In the Broaden trial (St. Jude Medical, ClinicalTrials.gov identifier NCT01801319), participants were treated with Cg25WM DBS and underwent a 6-month blinded crossover phase including two periods of active and two periods of sham stimulation. A futility analysis conducted when 75 patients had reached the 6-month follow-up estimated the probability of a successful outcome to be no greater than 17.2% (Mayberg, 2014; Morishita et al., 2014). The results of this study are yet to be comprehensively reported in the peer-reviewed literature.
The early reports suggest that the results of these RCTs are much less positive than those from the open studies. How can we account for this difference?
Placebo
It is conceivable that the positive treatment outcomes reported in open trials reflect a non-specific placebo effect, known to occur in studies of surgical interventions (Wartolowska et al., 2014), including those using sham neurosurgical procedures (Freed et al., 2001). The placebo effect in depression is firmly established (Enserink, 1999) and may cause regionally specific metabolic changes in the brain comparable to those induced by active treatment (including in area Cg25; Mayberg et al., 2002). A significant effect of sham stimulation was noted during the lead-in phase of an earlier trial of Cg25WM DBS (Holtzheimer et al., 2012). It is notable that in the Reclaim trial (Dougherty et al., 2015), physician programmers followed a careful titration protocol to avoid breaking the blind, reducing the likelihood of those in the active group becoming aware of their status (although as discussed below, this may have had unintended consequences).
However, by definition the patients in these studies demonstrated resistance to other physical treatments, including ECT. This suggests that a significant and enduring placebo effect is unlikely to have been in operation until treatment with DBS. Additionally, the size of the response to sham stimulation in the Reclaim study (14.3%) was considerably smaller than the observed response to active treatment in open trials. Even once the study entered the open phase, response rates did not rise to levels comparable with the open trials. Limited data from blinded pilot studies also suggest an effect of active stimulation (Schlaepfer et al., 2008). Moreover, pooled data from placebo-controlled, double-blind trials in OCD have demonstrated a significant treatment effect for DBS (Kisely et al., 2014), indicating its value in other treatment-resistant neuropsychiatric conditions.
Targeting and stimulation
Both Reclaim and Broaden were overseen by clinicians with considerable expertise in the field, using methods for operative targeting that have been refined through global surgical experience with ablative therapies and later with DBS for psychiatric indications (Greenberg et al., 2010). However, these methods rely on the use of anatomical landmarks and may lose specificity when applied to an individual patient’s neuroanatomy. Accurate electrode placement is a necessary condition for successful DBS. Tractography activation models (combining diffusion-weighted imaging and electrical field modelling) have demonstrated the importance of both accurate contact selection and adequate stimulation at the Cg25WM site (Lujan et al., 2013). Stimulation at an alternative contact separated by only millimetres, or sub-therapeutic stimulation at the optimal contact, produces a different pattern of cortical and subcortical network activation. Distinct patterns of connectivity in the perigenual anterior cingulate cortex (ACC) may account for this phenomenon (Johansen-Berg et al., 2008). The subgenual region is strongly linked to the NAcc and OFC, whereas contacts situated in the nearby pregenual ACC are less likely to activate these pathways. It is also conceivable that the recruitment of additional centres in these multisite trials entailed the involvement of clinicians whose expertise was primarily in DBS for movement rather than psychiatric disorders. It is noteworthy that the leading surgical centres offering DBS for OCD have described an evolution in surgical targeting of the VC/VS region, which they consider to be the major factor accounting for improvements in treatment efficacy over a decade (Greenberg et al., 2010).
We suspect that the pattern of network activation is a crucial factor in successful DBS. Uniform positioning of electrodes in stereotactic space may not be sufficient, as inter-individual variability in neural architecture and pathway distribution may cause differential patterns of network activation (Riva-Posse et al., 2014). Targeting in future trials may employ tractography to identify sites that promote activation of key white matter tracts, rather than relying on anatomical coordinates. Indeed, this technique was employed by Schlaepfer et al. (2013) to visualise the slMFB in their successful open trial. Postoperatively, the tractography activation maps of non-responders can be compared with the characteristic pattern noted in responders to Cg25WM DBS (Riva-Posse et al., 2014). This offers a potential biomarker to guide DBS programming and ensure that the key prefrontal, cingulate and striatal regions are activated.
Information regarding stimulation optimisation is available from the Reclaim trial (Dougherty et al., 2015). The investigators wished to maintain the blind, avoiding the induction of any stimulation-dependent sensations during dose titration. At baseline, they tried to delineate programming settings that produced adverse sensations, so as not to mistakenly elicit them during the blinded period. However, active contacts were selected based on acute changes in mood, anxiety and energy during this initial programming session, and all participants were stimulated at least overnight to assess long-term tolerability. This means that all participants were in fact exposed to active stimulation prior to blinding. Therefore, participants would be unblinded based on whether they subsequently did or did not experience similar sensations during the study. Furthermore, some patients in the active treatment arm may not have recovered these subjective changes outside of this initial programming session – possibly because stimulation intensity during the treatment phase was insufficient or because, based on preliminary findings among the open trials, it appears that acute sensations are not necessarily predictive of long-term responses. These patients may erroneously believe that they were in the sham treatment arm, forming negative expectations of their treatment.
It is also possible that the blinded treatment phase was of insufficient duration to modulate pathological network activity, given that in the open trials response rates were generally reported after 6 months and increased with longer periods of chronic stimulation. A delayed response may also reflect the need to optimise stimulation parameters in individual patients, meaning that a treatment effect will be underestimated if a crossover phase commences too early after DBS device activation. However, in the case of the Reclaim trial, response rates did not reach the levels seen in open trials even after 24 months.
Candidacy
In PD, clinicians can predict the likely benefits of DBS from the patient’s response to levodopa (Charles et al., 2002). There is currently no equivalent of a levodopa challenge in DBS for depression. Moreover, depression as defined in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5) is a heterogeneous condition that may include several disorders with different endophenotypes along with extreme dimensional variations of ‘normal’ behaviour (Parker, 2007b). This broad approach to diagnosis results from a ‘top-down’, symptom-based, consensus approach that is not guaranteed to correlate with ‘bottom-up’ pathobiologic mechanisms. For example, other co-morbid DSM-5 diagnoses such as cluster B and C personality disorders may contribute to persistently depressed mood. By their very nature these are enduring disorders whose neural correlates are poorly characterised. Treatment resistance is regarded as necessary from an ethical standpoint to achieve candidacy for DBS because at present this remains an experimental procedure that is considerably more invasive than existing therapies and may carry additional unknown risks that have hitherto remained unidentified due to its novel status. Yet treatment resistance heuristically conceptualised as a homeostatic failure in a depressed population may well also be a surrogate of diagnostic inaccuracy where significant comorbidity exists.
There are various approaches to patient selection for DBS. All research groups exclude those with significant co-morbid personality vulnerabilities, but treatment-resistant psychiatric disorders are likely to have adverse effects on personality. It therefore seems unreasonable to expect ‘pure’ cases of TRD. Personality is considerably less malleable than symptoms of a mood disorder, and patients may continue to be functionally impaired even if some aspects of their depression respond to DBS. The latency of response observed in the open trials and the tendency of benefit to accrue as the duration of follow-up increases could in part reflect a gradual reorganisation of these pathoplastic personality changes. For the purposes of a placebo-controlled trial where it is not feasible to measure outcomes at 5 years, it may be more appropriate to assess specific aspects of the depression construct that approximate more ‘biological’ endophenotypes, such as emotional signal detection in a neuropsychological task (Hyett et al., 2014). These behavioural measures could themselves be measured more sensitively than clinician-rated scaled items, increasing the prospect of detecting a benefit from active treatment.
It may also be possible to include a narrower range of phenotypes or a subset of depressed patients who are more likely to respond to DBS. Melancholia, previously termed endogenous depression, is one such candidate. It is a heritable form of mood disorder with strong biological underpinnings (Parker et al., 2010). Melancholia is associated with homeostatic dysregulation, such as hypercortisolaemia (Mitchell, 1996), and a preferential response to biological treatments over social interventions or placebo (Brown, 2007). Melancholic patients may therefore respond better to DBS. However, diagnosing melancholia on symptom profile alone is challenging. The DSM-5 includes a melancholic symptom specifier, but this has not proven useful in distinguishing a melancholic from a non-melancholic presentation (Parker, 2007a). Additional features that may support the presence of a melancholic syndrome are psychomotor changes (Parker et al., 1994), the absence of major antecedent life events (Wakefield, 2012) and derangements in the levels of certain inflammatory markers (Spanemberg et al., 2014), although some of these measures have not been consistently replicated. Although some DBS trials include patients with melancholic features, it is unclear how this was defined by the investigators and thus how reliably melancholic was their clinical presentation.
Melancholic depressives are more likely to respond to ECT (Petrides et al., 2001). However, most patients in DBS studies have failed to respond to this treatment – which may constitute an inadvertent selection bias against both melancholic depression and a response to intracranial neuromodulation. Only 34% of patients in the Reclaim trial had shown a response to ECT (Dougherty et al., 2015). An alternative method for recruiting to trials of DBS would be to include patients who do show a response to ECT, but who continue to relapse during maintenance treatment with pharmacotherapy, psychotherapy or other physical therapies such as transcranial magnetic stimulation.
Another means of refining patient selection is to identify biomarkers that predict a positive response to treatment. Functional imaging studies suggest that increased activity in the pre- and subgenual cingulate cortex predicts a positive response to treatment with antidepressant medication or psychological therapy (Fu et al., 2013). Likewise, increased activity in the right striatum and right insula cortex predicts a poorer treatment response. However, findings are discrepant between populations and across methodologies, with different patterns of activation engendered by different cognitive or affective tasks. Variability in the pattern of hyper- and hypometabolism may also reflect the variable symptom profile of the depressive illness (e.g. ruminations vs apathy) or compensatory strategies employed by the network to maintain residual functioning. Finding a neural signature of likely response to DBS is therefore not likely to be a viable strategy at the present time.
Designation of responders
When response is defined as a 50% reduction in self-reported or clinician-rated depression, non-response is common. However, it is unclear how meaningful this is at the individual level. In a chronic and disabling illness such as TRD, a 40% reduction in symptoms is technically a ‘non-response’ but may nonetheless be a meaningful improvement in quality of life. An analysis of the experience and quality of life in non-responders has not been reported, although functional recovery is correlated with greater symptom reduction (Kennedy et al., 2011; Malone et al., 2009). The authors of the Reclaim study report that 26 patients elected to continue with DBS after 24 months despite only 24% of the patients in the cohort being responders at this time (Dougherty et al., 2015).
Adverse events
We have discussed methodological issues in the design of RCTs of DBS for depression, in the anticipation that further studies will be undertaken, given the early promise of this therapy in a severe and intractable condition. But what should clinicians know about the risks of this treatment and how should that influence the design of future trials? The surgical risks of this procedure are relatively well understood and have been derived from the widespread use of DBS in neurological disorders. Only one patient in the DBS for depression cohorts suffered a major surgical adverse event, an intraoperative bleed that led to a temporary hemiparesis, from which he made a full recovery (Schlaepfer et al., 2013). However, from a psychiatric perspective, the nascent nature of this field means that there may be unknown risks that are currently not well delineated.
One issue is the abrupt return of depressive symptoms when stimulation is ceased in a patient who has hitherto responded to therapy. Compared to DBS for PD and other movement disorders, current use of DBS for TRD necessitates greater stimulation intensity and leads to early battery depletion. In Cg25WM DBS, battery replacement occurred after a mean (SD) of 72 (11) weeks of treatment (Holtzheimer et al., 2012). Inadvertent cessation of stimulation due to battery failure or even inadvertent deactivation may be a serious event leading to a depressive relapse (Malone et al., 2009; Merkl et al., 2013). Cessation of active stimulation in three patients during a blinded discontinuation phase was also associated with a return of depression (Holtzheimer et al., 2012). This element was then withdrawn from the study protocol on ethical grounds. Therefore, in any crossover trial in which the patient responds to active treatment, criteria must be specified for unblinding to prevent significant patient deterioration during inactive treatment. This may be especially important in future placebo-controlled trials, in which longer periods of blinded treatment may be necessary to assess efficacy of active over sham stimulation. In DBS for OCD, some candidates refused to enter a blinded phase once they had benefitted from active treatment (Denys et al., 2010). Indeed, sudden cessation of stimulation in this group led to an obsessive and affective ‘storm’ of symptoms that were more severe than pre-treatment levels (Ooms et al., 2014). Crossover studies of this type may thus be ethically problematic when compared to trials only involving a delayed onset of stimulation.
The psychological adjustment to device implantation and chronic stimulation also requires study. Experience with PD suggests that patients and their families must adapt to changes in body image, re-negotiate social and occupational roles after a period of chronic disability and integrate subtle postoperative changes in personality, values and commitments that may be linked to stimulation (Lewis et al., 2014; Schupbach et al., 2006). This process can be experienced as authentic or alienating, depending on the psychological orientation and preparedness of the patient (Kraemer, 2013; Witt et al., 2013). One patient in an early RCT of DBS for OCD committed suicide during the open maintenance phase of the study (Abelson et al., 2005). This patient had responded to DBS but suffered an exacerbation of depressive symptoms after an unsuccessful return to work. This highlights the challenges faced by those in recovery from a longstanding chronic illness, even after successful treatment. Long-term psychiatric care is necessary for those facing the ‘burden of normality’ (Wilson et al., 2001).
The iatrogenic induction of elevated mood can occur with subthalamic stimulation for PD (Appleby et al., 2007; Daniele et al., 2003). This may disrupt the period of postoperative adjustment by coarsening personality and generating impulsive behaviour. The induction of impulsivity in severely depressed patients may also increase the risk of suicidal behaviour (see below) (Hawton et al., 2005). Among depressed patients, agitation was reported in DBS of Cg25WM (Lozano et al., 2012) and the NAcc (Bewernick et al., 2012), transient mood elevation in NAcc DBS (Bewernick et al., 2012) and hypomania in VC/VS DBS in a patient with bipolar disorder (Malone et al., 2009). It seems a reasonable hypothesis that patients with bipolar disorder would be more susceptible to this phenomenon.
Could DBS be a factor in postoperative suicide? Suicide after DBS is a complex phenomenon with many potential determinants (see Figure 1). All but three of the studies reported completed suicides or suicide attempts in the postoperative period (see Tables 1 and 2). These were not necessarily linked to non-response (Holtzheimer et al., 2012). Of the remaining three (Malone et al., 2009; Merkl et al., 2013; Schlaepfer et al., 2013), one cohort described several cases of postoperative suicidal ideation (Malone et al., 2009). The other studies were unlikely to detect an increase in suicidal events because the cohorts were very small and there was a short duration of follow-up.
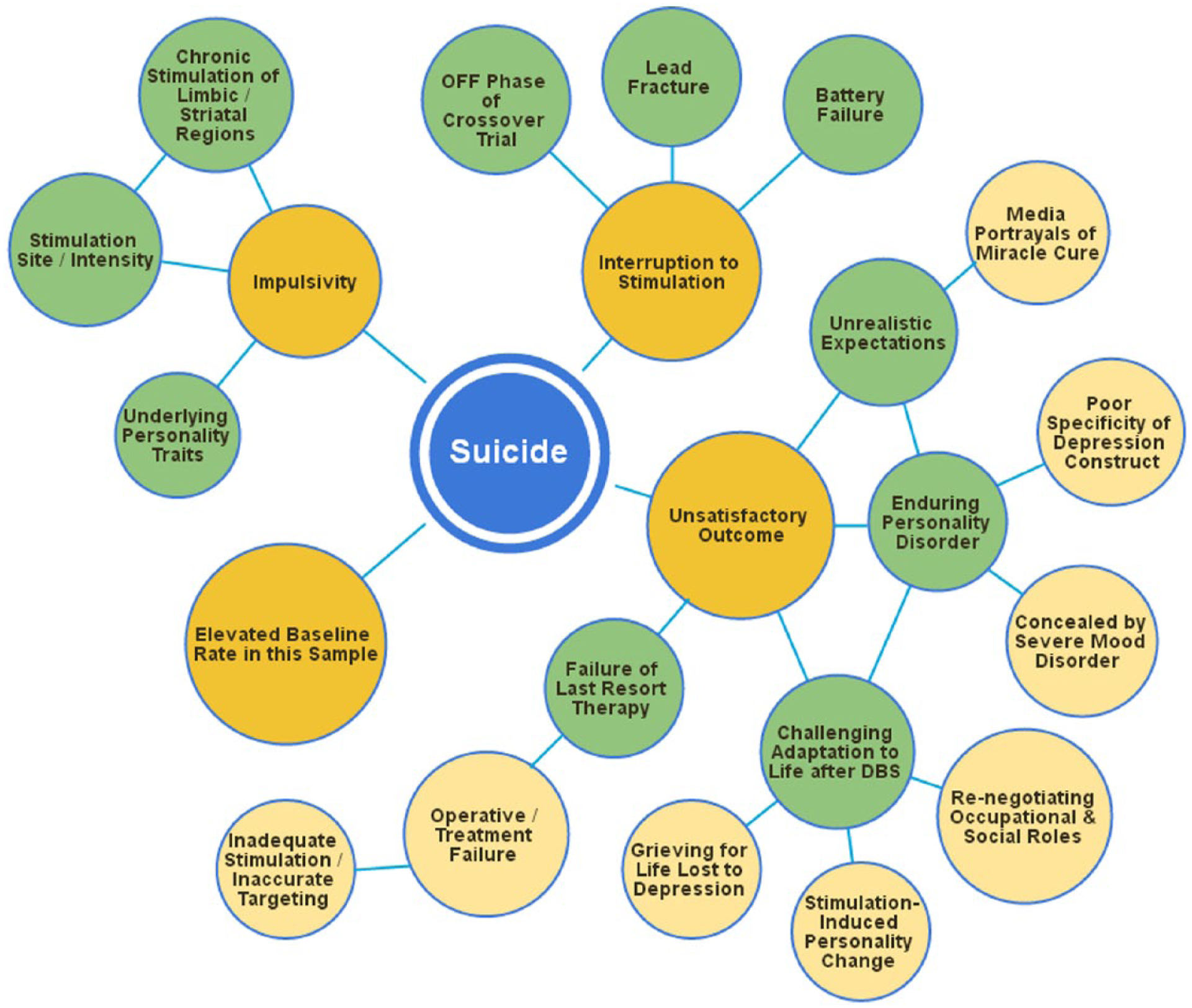
The constellation of factors that may contribute to post-DBS suicide.
The mortality risk of suicide associated with depression is elevated substantially above general population norms (Harris and Barraclough, 1998). The lifetime risk of suicide among severely depressed patients hospitalised with suicidal ideation has been estimated at 8.6% (Bostwick and Pankratz, 2000). Therefore, to be a sufferer of TRD is already to be at considerable risk of death by suicide. It is unclear whether the suicides seen after DBS reflect the base rate in this population or an elevated rate.
Most groups stated that suicide was not linked to DBS. This seems most likely in cases where death occurred at an interval distal to device implantation or stimulation adjustment. However, given that the surgical targets in the affective network overlap with a putative reward system (i.e. the ventral striatum, cingulate cortex and their connections with the prefrontal cortex), it is conceivable that chronic stimulation may produce a bias in decision-making that increases the risk of suicidal behaviour. Impulsivity is an established biological risk factor for suicide (Hawton et al., 2005) with an origin in aberrant functioning of the prefrontal, striatal and limbic network (Mann, 2003). Neuropsychological correlates of impulsivity increase after DBS of the subthalamic nucleus for PD, including impaired response inhibition (Witt et al., 2004) and increased speed of decision-making under situations of cognitive conflict (Frank et al., 2007). A retrospective case series found suicide to be the major cause of mortality in the first year after DBS for PD, with suicide attempts far more likely to be lethal than in the general population (Voon et al., 2008). The neuropsychological sequelae of DBS for depression have not been examined in detail, although the Reclaim study reported a detrimental effect of DBS on mental flexibility in the context of response inhibition (Dougherty et al., 2015). Further investigation is necessary.
Non-specific effects of treatment could also be a factor in post-DBS suicidality. Failure to respond to a ‘last resort’ treatment like DBS may be profoundly demoralising. Sufferers may also enter therapy with unrealistic hopes of dramatic improvement, fuelled by misleading media portrayals of recovery (Racine et al., 2007). Part of the preoperative consent process must involve negotiating a mutuality of realistic expectations about treatment outcome between the patient and clinical team.
Conclusion
Many individuals suffer from residual depressive symptoms that fail to remit with existing biological or psychological therapies. DBS may be an option for those sufferers with the most severe forms of intractable depression. In this article, we have speculated on the reasons behind the disappointing outcome of recent RCTs in this field. We have justified our speculation on the grounds that it is unclear when these negative results will be fully available in the public domain and that attention to methodological factors may assist other groups in the design of successful trials.
We have highlighted the importance of a careful approach to patient selection, electrode targeting and the design of the crossover or blinded protocol. Novel strategies are necessary to define the target population because standard psychiatric diagnostic criteria may not usefully identify these individuals. We discuss melancholia, response to ECT and neural biomarkers, noting that there is limited evidence to support these strategies. On the other hand, established techniques for target localisation may benefit from the use of tractography, and this has been successfully employed in an open trial. Finally, if the benefits of stimulation are realised in the order of years rather than months, lengthy follow-up studies will be needed to demonstrate efficacy of DBS over placebo, or investigators will need to develop targeted and objective assessment criteria that can be evaluated with sufficient statistical power. We anticipate that with these methodological refinements, DBS may yet prove to be a treatment option for a subset of individuals who are profoundly disabled by their mood disorders.
Footnotes
Acknowledgements
The authors would like to thank Professor Wayne Hall for helpful comments on the manuscript. This paper was presented at the 21st Congress of the European Society for Stereotactic and Functional Neurosurgery, Maastricht, the Netherlands, 17–20 September 2014.
Declaration of interest
Philip Mosley and Rodney Marsh have participated in an educational activity sponsored by Medtronic. They are investigators in a clinical trial of DBS for OCD supported by Medtronic. Adrian Carter reports no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
