Abstract
Background
Trials of deep brain stimulation (DBS) to the subcallosal cingulate gyrus (SCG) for treatment-resistant depression (TRD) have yielded mixed results. While open-label studies suggest effectiveness, randomized controlled trials (RCTs) have not consistently supported these findings. The study compared the efficacy of active versus sham SCG stimulation for TRD.
Methods
Participants (n = 35) in a major depressive episode and treatment resistance completed a 6-month double blind, crossover RCT, with an 18-month open-label phase. A Balaam design was applied with participants randomized to 1 of 4 stimulation groups over two 3-month phases. The primary outcome was a change in Hamilton Depression Rating Scale (HDRS) score at 6 months, with response defined as ≥50% reduction in HDRS-17 scores.
Results
While all groups showed improvement at 3 and 6 months, no significant differences were found among them. The OFF–OFF group had a numerically lower HDRS-17 score compared to the ON–ON group at the end of the RCT. No unexpected adverse events occurred. During the open-label phase, participants showed sustained reduction in HDRS-17 scores at 12, 18, and 24 months post-implantation, with successive observed-case response rates of 65.7%, 69%, and 73.1%, respectively. Improvements in life functioning were also noted.
Conclusions
This trial represents the largest single-centre, sham-controlled study of SCG DBS for TRD in the literature. Although the RCT showed no significant group differences, most participants achieved response during the open-label phase. Safety outcomes aligned with previous trials. Future RCTs should integrate insights from the past decade of DBS for TRD research to optimize outcomes. Key considerations include selecting DBS contact locations that ensure engagement of critical white matter tracts, employing novel and sufficiently long clinical trial designs to account for the non-specific effects of the DBS procedure, as well as incorporating biomarkers to guide DBS programming.
Plain Language Summary Title
Deep Brain Stimulation for Treatment-Resistant Depression: Results of a Randomized Controlled Trial and Long-Term Follow-Up
Plain Language Summary
Deep Brain Stimulation (DBS) is an invasive neurosurgical procedure that involves the implantation of electrodes into the brain to provide continuous stimulation to discrete neuroanatomical targets. Although DBS has established efficacy in neurological conditions such as Parkinson's Disease, the impact of DBS on improving depressive symptom in those with treatment-resistant forms of Major Depressive Disorder has not yet been established. This study presents the results of the largest single-centre, sham-controlled study of DBS to the Subgenual Cingulate Gyrus (SCG) for depression. People with Treatment-Resistant Depression (TRD) were randomized to having DBS on or off for each of two successive 3 month blocks, creating 4 groups (ON-ON, ON-OFF, OFF-ON and OFF-OFF). Participants and the psychiatrists who assessed their clinical status were unaware of which of the 4 groups they were in. At the end of the 6 month randomization period, all participants had DBS turned on and evaluated up to 2 years post-surgery during an open-label phase.
While all groups showed improvement at 3 and 6 months, no significant differences in depressive symptoms severity were observed across the groups. During the open-label phase, participants showed a clinically significant and sustained reduction in their depression scores at 12-, 18-, and 24-months post-surgery, with approximately 65-75% response rates observed during this period of time. The surgery was safe and no serious side-effects were observed in the post-operative, randomization or open-label phases.
Introduction
The burden associated with major depressive disorder (MDD) is among the highest across disorders worldwide. 1 Despite treatment advances, remission occurs in fewer than two-thirds, 2 leaving approximately 30% of people with treatment-resistant depression (TRD). TRD is defined as failure to achieve clinical response after at least 2 adequate trials of antidepressant therapy during the current episode. 3 TRD compromises quality of life, increases suicide risk, and contributes to higher morbidity and mortality compared to conditions like diabetes and cardiovascular disease. 4
Deep brain stimulation (DBS) targeting the subcallosal cingulate gyrus (SCG) has showed promising results for treating TRD. 5 Subsequent open-label trials have affirmed its acute and sustained antidepressant effects up to 6 years.5–8 While the first randomized sham-controlled trial of SCG-DBS for TRD showed no between-group differences, 9 the open-label extension data up to 8 years demonstrated significant response and remission rates. 10 In a double-blind, randomized, crossover trial of short (90 μs) versus long-pulse width (210–450 μs) stimulation for 6 months, Ramasubbu and colleagues reported significant antidepressant effects with both groups. 19 Results of sham-controlled studies of SCG-DBS for TRD are presented in Supplemental Table 1.
Other neuroanatomical targets for DBS have also been investigated for TRD, including the internal capsule/ventral striatum, lateral habenula, nucleus accumbens, and orbitofrontal cortex. However, results from these small- to medium-sized and largely open trials yielded equivocal results, particularly when active and sham stimulation were compared,17–21 supporting the need for additional evaluations.
Notably, positron emission tomography imaging showed that SCG-DBS symptom improvement correlates with changes in cerebral blood flow or glucose uptake in limbic and cortical areas.5,6,11 This suggests that SCG-DBS may modulate aberrant cortico-limbic circuits in TRD. Stimulation location may also drive clinical outcomes,12–16 which emphasizes the importance of engaging specific local white matter tracts (i.e., cingulum bundle, corpus callosum, and uncinate fasciculus) connected to cortico-limbic regions.
The efficacy of DBS needs to be established in order to provide an alternative treatment for TRD. Here, we report on a 6-month, sham-controlled, randomized crossover trial of SCG-DBS with an open-label extension to 2 years. We also assess DBS contact location and distance to white matter tracts of interest in relation to clinical outcome.
Methods
Study Design
This was a single-site, randomized, sham-controlled, double-blind, 6-month crossover study of SCG-DBS for TRD (n = 35). Participants received active or sham stimulation for 3 months, with a potential switch in the following 3 months (Figure 1), using a Balaam design. 22 This design allowed for comparisons to estimate treatment effects, even with carry-over effects. Participants were referred to study psychiatrists (PG, RS, SHK) at University Health Network (UHN), who assessed their eligibility. The study was conducted at the Toronto Western Hospital and Toronto General Hospital sites at UHN, approved by the UHN Research Ethics Board and registered (ClinicalTrials.gov: NCT01801319).
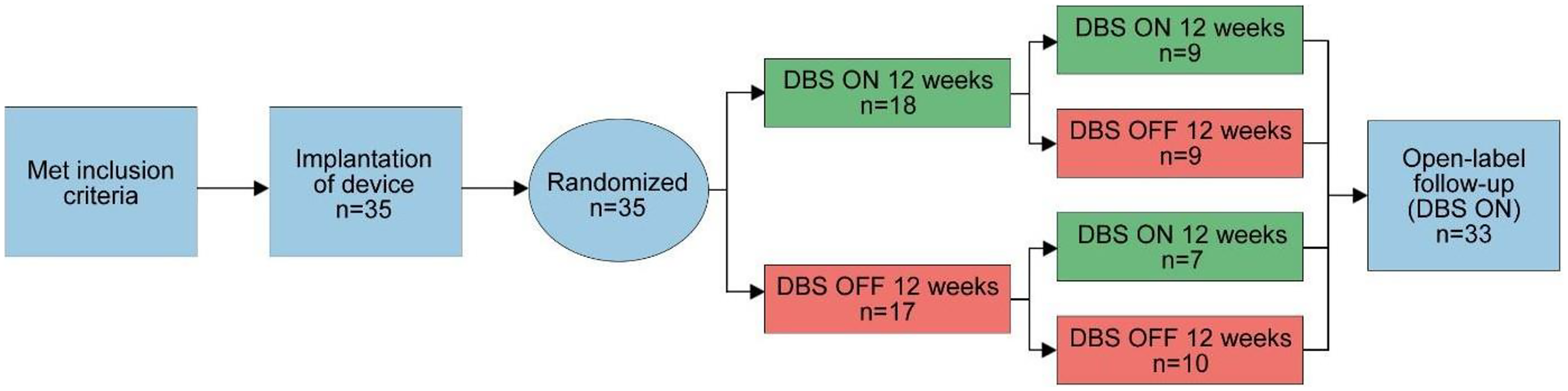
Study design.
Participant Sample
Eligible participants were between 21 and 70 years of age with a diagnosis of MDD according to DSM-IV-TR criteria, confirmed by the Mini International Neuropsychiatric Interview. 23 The first major depressive episode (MDE) must have occurred prior to age 45, with the current MDE ≥ 12 months. Participants were required to score ≥20 on the 17-item Hamilton Depression Rating Scale (HDRS-17) 24 across 3 pre-operative visits, meet criteria for treatment resistance (defined as failure to ≥4 adequate treatment trials from at least 3 different classes of pharmacotherapy or neurostimulation), have received lifetime psychotherapy, and have no changes in their antidepressant regimen 4 weeks before surgery.
Exclusion criteria were lifetime diagnosis of bipolar disorder or MDD with psychotic features, borderline/antisocial personality disorder or substance use disorder within the past 12 months, obsessive-compulsive disorder, post-traumatic stress disorder, panic disorder, or eating disorder within the last 6 months, generalized anxiety disorder as the primary diagnosis, imminent suicide risk, 3 or more suicide attempts within the last year, receipt of electroconvulsive therapy (ECT) 3 months prior to enrolment, severe medical disorders, contraindications to surgery, and pregnancy/lactation.
Procedures
Baseline visits
Following written informed consent, participants underwent 3 monthly baseline visits that involved structured psychiatric interviews, completion of clinician-administered and self-report scales on mood and life functioning, physical examination, and a review of current and previous antidepressant trials.
Surgical procedure
Pre-operative planning and electrode implantation have been previously described.5,25 Briefly, participants had a metal stereotactic frame placed on their head and received a 1.5 T magnetic resonance imaging (MRI) scan to guide surgical procedures. Participants received the Abbott Libra DBS system (n = 31) or the Medtronic Activa DBS system (n = 4). Two quadripolar DBS electrodes were inserted into the white matter tracts of the SCG under local anaesthesia. After implantation, intra-operative stimulation confirmed DBS contact placement within SCG white matter, followed by insertion of an implantable pulse generator (IPG) into the right subclavicular region under general anaesthesia. A post-operative MRI scan confirmed electrode placement. Participants were discharged with IPGs turned off.
Randomization
Two weeks after DBS implantation, participants were randomized to a stimulation sequence in a block size of 4: ON/ON, ON/OFF, OFF/ON, OFF/OFF. Clinical evaluations were completed by blinded psychiatrists (RS, SHK), and DBS programming was performed by an unblinded psychiatrist (PG). Everyone else on the research team was blinded. An assessment of blinding was completed by participants and evaluating psychiatrists following each treatment phase.
6-Month blinded phase
Participants had weekly visits during the first month and biweekly visits for the next 2 months. The subsequent 3-month period followed the same visit schedule. At each visit, participants completed self-report and clinician-administered questionnaires on mood and life functioning. Adverse events (AEs) were collected at each visit. At the 3-month crossover visit, IPGs were set to deliver either active or sham stimulation.
Medication use
Current antidepressant dosage could be decreased, but not increased, at the discretion of the evaluating psychiatrist. For insomnia, zopiclone (<7.5 mg), or trazodone (<100 mg) were permitted for a maximum of 14 nights. Lorazepam (<2 mg/day) was permitted for worsening anxiety for up to 14 days.
Adjustment of stimulation parameters
Selection and adjustment of stimulation parameters were carried out using an algorithm (Supplemental Table 2). Parameter permutations involved 2 pairs of contacts and 3 current levels (3.5, 5.0, and 6.5 mA). Treatment was initiated at the closest contact pair to the SCG (as per post-operative MRI) using 3.5 mA stimulation in the ON group. Pulse width and frequency were 90 ms and 130 Hz, respectively. Stimulation adjustment occurred at study visits in the ON condition if there was an HDRS-17 reduction ≤20% from baseline. To maintain the blind, the programming psychiatrist inspected IPG settings at each visit across all participants.
Open-label follow-up
After the randomized controlled trial (RCT), participants received open-label active stimulation and follow-up visits every 6 months. The same outcome measures as the RCT were collected at each visit. Results are presented 2 years post-implantation.
Outcome measures
The primary outcome measure was the HDRS-17. 24 Severity and change in depression were assessed using the Clinical Global Impression (CGI-S and CGI-I). Self-reported depression severity was measured using the Quick Inventory of Depression Symptoms-Self Report. 26 Life functioning was evaluated using the Sheehan Disability Scale (SDS) 27 and Short Form Health Survey-12.28,29 Anxiety was assessed using the Hamilton Anxiety Rating Scale. 30 Positive affect was evaluated using the 18-item Motivation and Energy Inventory (MEI), 31 8-item Oxford Happiness Scale 32 and Warwick Edinburgh Mental Wellbeing Scale (WEMWBS). 33 To assess DBS safety, we measured mania/hypomania using the Young Mania Rating Scale 34 and suicide risk with the Columbia-Suicide Severity Rating Scale (C-SSRS). 35
DBS electrode localization and determination of distance to white matter tracts
Lead localization was conducted with Lead-DBS software (https://www.lead-dbs.org/), as previously described.14,36 Pre- and post-operative structural MRI scans were inhomogeneity-corrected and rigidly co-registered before being nonlinearly transformed to standard Montreal Neurological Institute (MNI) space. Any brain shift was corrected using a linear transformation.37,38 Electrodes were manually localized on post-operative images and moved into MNI space, using the previously generated nonlinear warp fields. The MNI space coordinates of active electrode contacts (i.e., contacts selected for use, regardless of randomization) were identified at 3 and 6 months. The distance from active contacts to relevant white matter tracts was estimated. As previously reported,13,14,39 white matter tracts of interest were isolated from a whole-brain normative tractogram (http://www.humanconnectomeproject.org/). 40 The distance between active contacts and the nearest voxel overlapped by each normative tract was determined using ITK-SNAP (www.itksnap.org). 41
Statistical Analysis
Sample size justification
Study power was estimated using previous data 25 across simulated scenarios and incorporating the following factors: standard deviation uncertainties in HDRS-17 scores, unknown cross-over effect of stimulation after consecutive treatment periods, and 10% of subjects dropping out. The Balaam design provided sufficient power (79–95%) to detect a 5-point between-group difference in HDRS-17 scores with 40 participants.
RCT outcome data
A linear random-effects regression model was used to estimate the effect of treatment on HDRS-17 scores, controlling for baseline scores and period and accounting for variance from random effects due to participants and measurement random error. 42 The treatment effect estimate and 95% confidence interval (CI) were derived from the fitted model adjusting for a cross- over effect. A secondary analysis of the HDRS-17 included baseline demographic factors that differed between groups as a covariate. A random-effects linear regression model was used to analyse all secondary measures. Random effects generalized linear modelling was used to analyse data with binary dependent variables, where treatment effect estimates were derived with odds ratios. AEs and assessment of blinding are presented descriptively by group.
Electrode localization and white matter tract distance
Electrode location-related statistics were performed using non-parametric tests. Cohort-wise differences with respect to active contact location and active contact-to-white matter tract distance were evaluated using Kruskal–Wallis and Wilcoxon rank-sum tests. Potential differences in binary outcomes, namely the presence or absence of direct impingement of an active contact on a white matter tract (i.e., a distance of 0 mm between the active contact voxel and the nearest voxel belonging to the white matter tract), were examined with chi-square tests.
Open-label data
For each scale, a repeated-measures analysis of variance was used to evaluate the impact of time on scores, and paired-sample t-tests were used to evaluate differences in scores between follow-up timepoints. Rates of response (≥50% reduction on HDRS-17 from baseline) and remission (HDRS-17 ≤ 7) were calculated using intent-to-treat and observed-case methods.
Results
In total, 39 participants were enrolled in the study, of whom 35 underwent surgery and were randomized. Following unblinding, it was discovered that 3 participants received stimulation contrary to their randomization (n = 1 was randomized to ON–ON but received OFF–OFF; n = 1 was randomized to OFF–ON but received OFF–OFF; n = 1 was randomized to OFF–OFF but received OFF–ON). Analyses were based upon received stimulation. There were no significant group demographic differences (Table 1).
Demographic Data Across 4 Randomization Sequences.
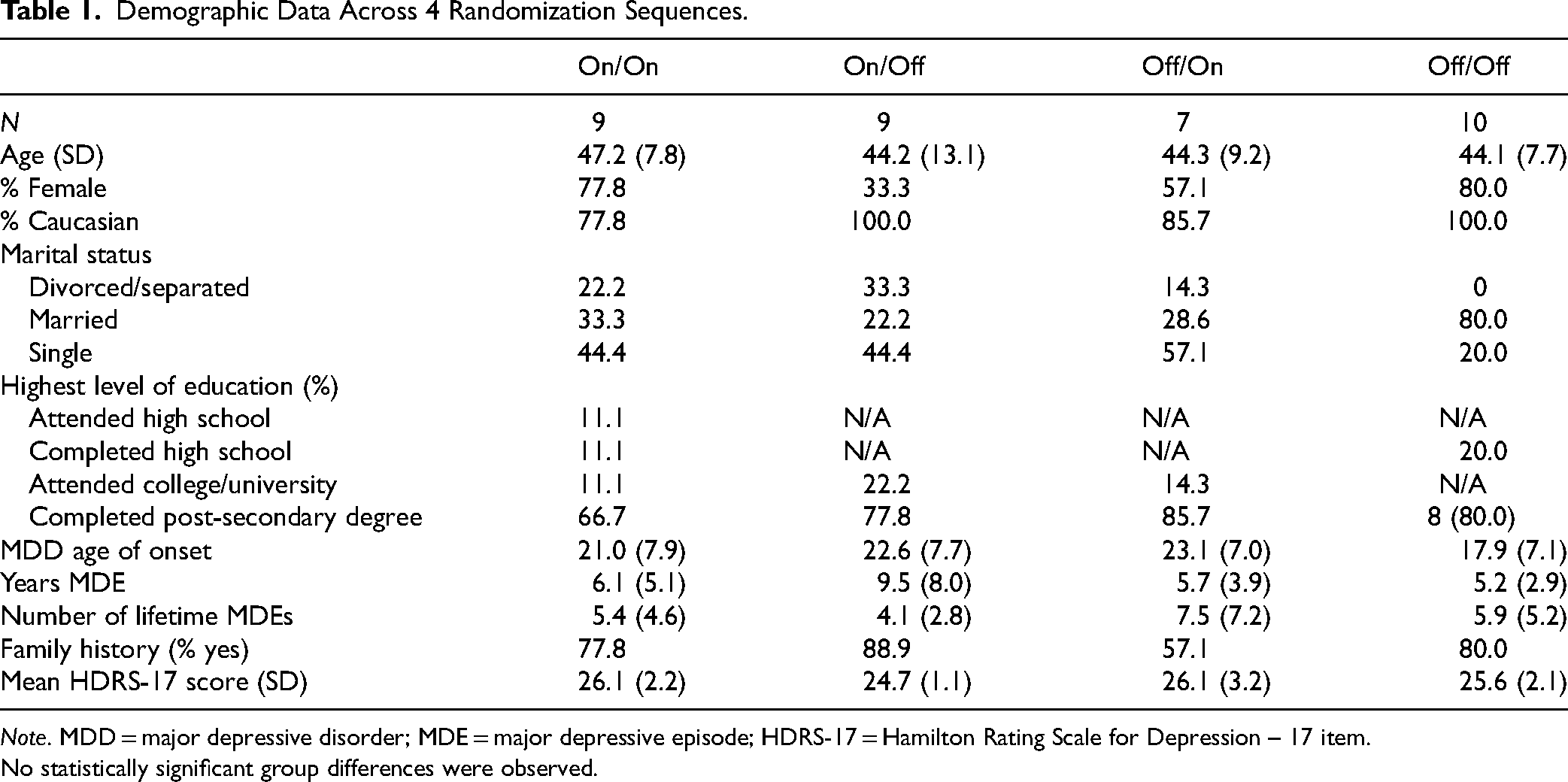
Note. MDD = major depressive disorder; MDE = major depressive episode; HDRS-17 = Hamilton Rating Scale for Depression – 17 item.
No statistically significant group differences were observed.
RCT Outcomes
Effect of DBS on HDRS-17 scores and response rate
HDRS-17 scores at 3 and 6 months are presented in Figure 2. Over treatment period 1, there was no significant difference between groups (t = 0.81, P = .42). Furthermore, there was no difference between groups in HDRS-17 change over visits (t = −0.02, P = .98). All participants reported a large drop in scores over visits (t = −7.86, P < .001).
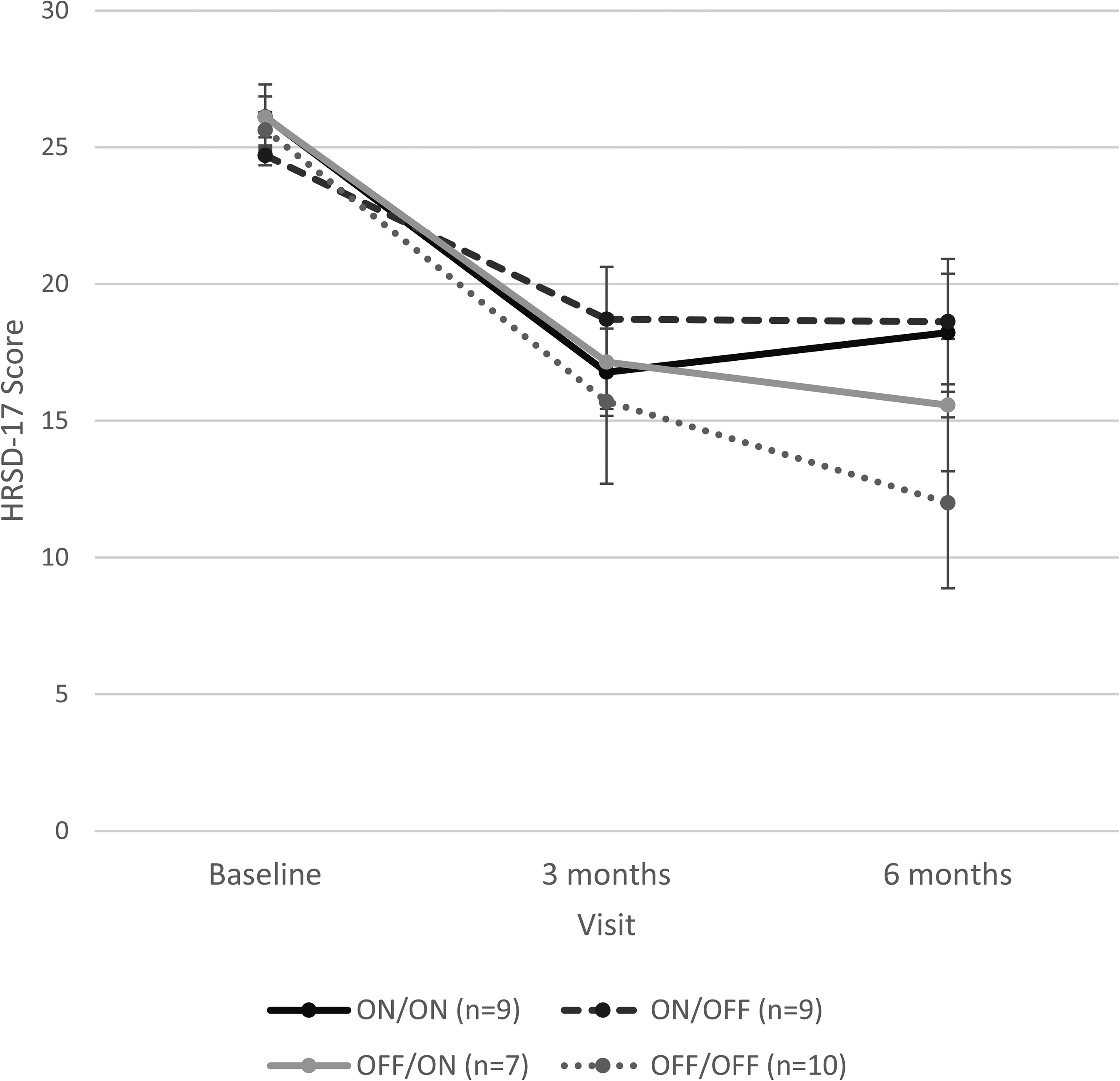
RCT phase outcomes based on the Hamilton Rating Scale for Depression – 17 item. Error bars represent standard error of the mean. RCT = randomized controlled trial.
A significant carry-over effect was observed [F(1,62) = 5.28, P = .03], justifying the use of the Balaam design. No significant treatment effect was found (t = 1.04, P = .30). Over 6 months, DBS activation led to an estimated 1.61-point increase in HDRS-17 scores compared to being off. The effect on the log odds of response was −0.8 (P = .36) with DBS on compared to off.
Including all follow-up timepoints, a significant treatment effect was observed, with a 1.55-point HDRS-17 increase when DBS was on compared to off (t = 2.17, P = .03), but no change in response odds (OR = 1.01, 95% CI, 0.49 to 2.09, P = .99). The response and remission rates at 3 and 6 months are presented in Table 2. Results from the secondary outcomes and assessment of RCT blinding are presented in the Supplemental Data.
Antidepressant Outcomes Duration Randomization Phases.

Active contact location and proximity to white matter tracts
Supplemental Table 3 summarizes the active contact locations in MNI space and distance to implicated white matter tracts for each group across timepoints. Many participants’ active contacts directly impinged on the forceps minor/rostrum of corpus callosum (3 month: 94.3%, 6 month: 94.1%), left cingulum bundle (3 month: 77.1%, 6 month: 82.4%), and right cingulum bundle (3 month: 31.4%, 6 month: 38.2%). By contrast, active contacts were rarely located within the right (3 month: 17.1%, 6 month: 17.6%) or left (3 month: 0.0%, 6 month: 2.9%) uncinate fasciculus. The lower middle contact of each electrode was used as the active contact in the majority of patients (3 month: 91.4%; 6 month: 85.7%).
At the 3-month timepoint, no significant differences were detected between the ON (n = 18) and OFF (n = 17) groups’ active contact coordinates. Right-sided active contact proximity to the right cingulum bundle was observed (z = 2.0, P = .04). Specifically, the OFF group's contacts (0.9 ± 1.4 mm) were closer to this tract compared to the ON group (1.7 ± 1.4 mm). A higher proportion of OFF (47.1%) than ON (16.7%) group active contacts directly impinged on the right cingulum bundle, although only marginally significant (P = .05).
At the 6-month evaluation, a difference in the anteroposterior position of the left-sided contact was identified among the 4 randomization groups (OFF/OFF, n = 10; OFF/ON, n = 7; ON/OFF, n = 8; ON/ON, n = 9). Post-hoc, Bonferroni-corrected Wilcoxon rank-sum tests indicated that the ON/OFF group's left-sided contacts (23.4 ± 2.1) were anterior to those of the ON/ON group (20.4 ± 1.7) (z = −2.6, P = .008). No group differences were seen with respect to active contact–white matter tract distance.
Open-Label Data to 2 Years
Effect of DBS on HDRS-17 scores
As there were no group effects in the RCT, we collapsed groups for the open-label analyses. There was a 13.8 point decrease on the HDRS-17 from baseline to year 1 (t(34) = 9.840, P < .001). This effect was sustained at 1.5 and 2 years (12–18 months: P = .141; 18–24 months: P = .771) (Figure 3). There were no differences in the observed case and intent to treat analysis. Response rates at 12, 18, and 24 months were 65.7% 69%, and 73.1%, respectively. The proportion of participants who responded and remitted to DBS was maintained over 2 years (Supplemental Figure 1).
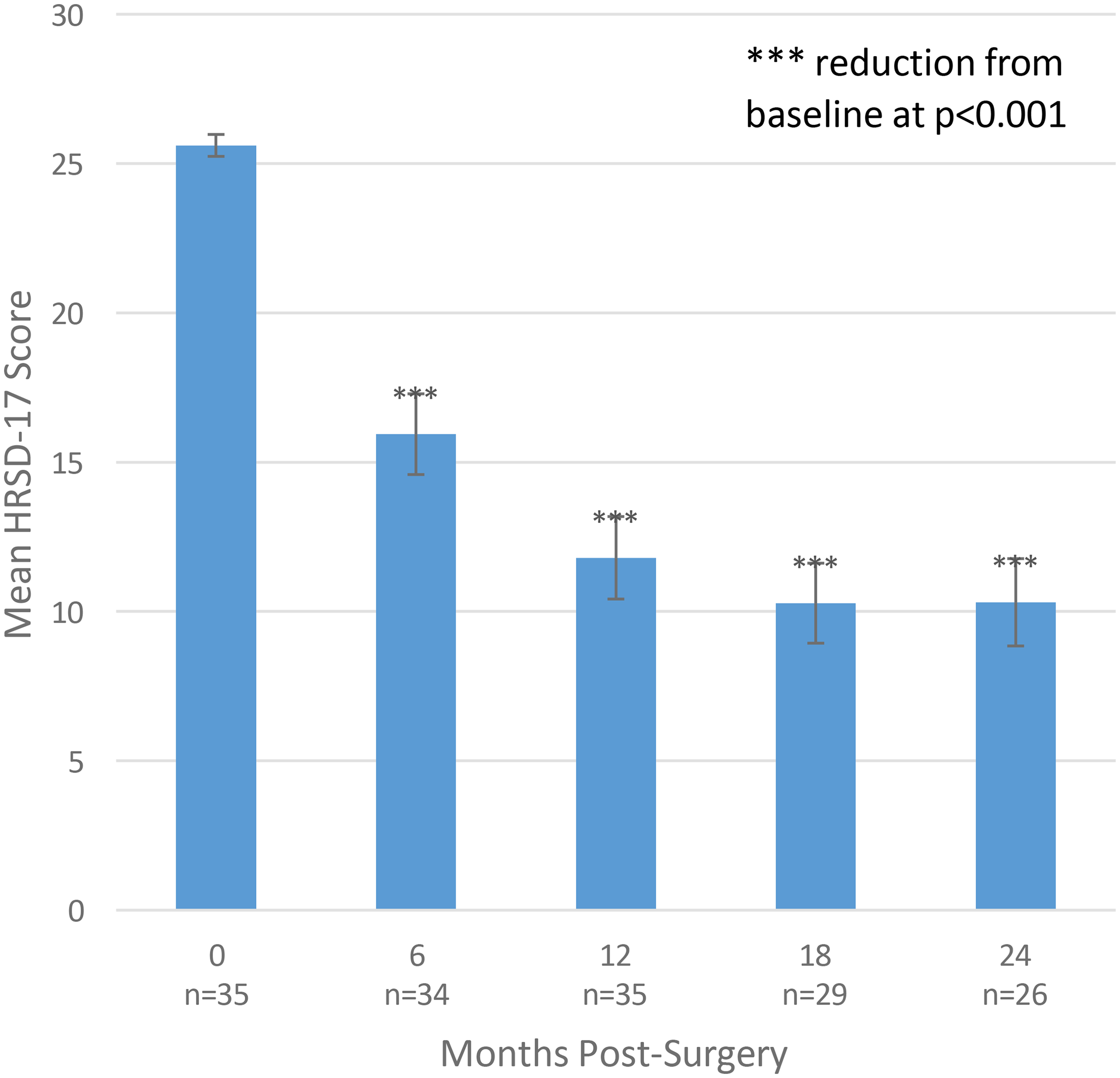
Open-label outcomes based on the Hamilton Rating Scale for Depression –17 item. Error bars represent standard error of the mean. HDRS-17: Hamilton Rating Scale for Depression – 17 item.
Secondary outcomes
The MEI improved by 19.6 points from baseline to year 1 (t(25) = −4.421, P < .001), with additional improvement from 12 to 18 months (t(18) = −2.566, P = .019), and maintenance to 2 years (18 to 24 months, t(15) =− 0.395, P = .698). Similarly, the WEMWBS improved over the 2-year period (F(2.185,32.780) = 8.228, P < .001): scores significantly increased from baseline to 1 year (t(27) = −4.069, P < .001), with no additional improvement from 12 to 18 months (t(20) = −1.844, P = .080) or 18 to 24 months (t(19) = 1.113, P = .280). Oxford Happiness Questionnaire (OHQ) scores did not improve over time (F(2.859,40.030) = 0.222, P = .872). Improvement in functioning based on the SDS was observed across the 2-year period (F(1.894,28.411) = 7.308, P = .003). The majority of this change occurred from baseline to 1 year (t(28) = 4.089, P < .001); no additional significant improvement from 1 to 2 years was observed (12 to 18 months: t(21) = 0.303, P = .765; 18 to 24 months: t(21) = 0.712, P = .485). Participants who were responders at 6 months based on HDRS-17 scores had significantly higher mental well-being scores at 18 months (WEMWBS; t(22) = 2.092, P = .048), and significantly lower SDS scores at 12 months (t(26) = −2.282, P = .031) and 18 months (t(23) = −3.137, P = .005).
Safety
Randomized controlled trial
No AEs were reported in 54.3% of participants. The number of AEs per participant across groups was similar: ON/ON: 1.2; ON/OFF: 1.1; OFF/ON: 1.0; OFF/OFF: 0.9. In total, 37 AEs were reported, of which 5 were serious adverse events (SAEs) (3 SAEs in the ON/OFF group and 2 in the OFF/OFF group). The common AEs possibly or definitely related to DBS were sleep disturbance (n = 2; 5.4%), headache (n = 1; 2.7%), pain at surgery incision site (n = 1; 2.7%), system dislodgment (n = 1; 2.7%), and hearing/visual disturbance (n = 1; 2.7%). No mania, sexual side effects, or suicidality occurred with DBS on. Suicidality based on the C-SSRS decreased over 1 year and this reduction was maintained at 2 years. Three SAEs were prior to randomization: 1 grand mal seizure post-surgery, 1 surgical revision of the DBS electrode placement, and 1 incision infection at the IPG site. The other 2 SAEs were in the OFF/OFF group and included hospitalization due to suicidality and kidney stones (pre-existing condition).
Open-label
In total, 13 AEs were classified as probably or definitely related to the DBS procedure or device: headache (n = 4; 30.1%), tightness in neck/IPG site (n = 2; 15.4%), IPG movement (n = 2; 15.4%), pulling sensation at lead extension site (n = 1; 7.7%), tingling sensation at lead (n = 1; 7.7%), arm pain (n = 1; 7.7%), battery failure (n = 1; 7.7%), and pruritus at clavicle scar site (n = 1; 7.7%). There were 5 SAEs unrelated to DBS: 4 for depression severity that were 12, 20, 21, and 25 months post-surgery, and 1 for blood clots 14 months post-surgery.
Discussion
This trial represents the largest single-centre, sham-controlled study of SCG-DBS for TRD in the literature. The results confirm the safety and tolerability of SCG-DBS in TRD patients. While depressive symptoms reduced during the RCT phase, no significant treatment effect was observed at 3 or 6 months. However, significant depression improvements were noted during the open-label phase, consistent with previous trials.9,10 The negative outcomes of the RCT could indicate that DBS lacks antidepressant benefits. However, evidence from studies of the mechanisms and clinical efficacy of SCG-DBS for TRD suggest design features of this RCT that potentially contributed to the outcomes.
First, the contacts selected to provide stimulation were not individualized based on the patient's anatomy. Evidence points to the importance of electrically modulating specific local white matter pathways for treatment success. Retrospective analyses of SCG-DBS for TRD identified common activation patterns in responders, particularly in the forceps minor, cingulum bundle, uncinate fasciculus, and midcingulate cortex. 15 Greater activation of white matter tracts have been linked to antidepressant response.13,19,39 In a prospective cohort, personalized DBS settings optimizing white matter engagement improved response (72.7%) and remission (54.5%) at 6 months. 16 Newer DBS devices with directional leads enhance targeted stimulation, 43 reinforcing the role of white matter engagement in DBS efficacy.
Second, the programming algorithm in this study may have been overly restrictive. Over the 6-month RCT, the algorithm allowed for 2 dose increases up to a maximum of 6.5 mA and 1 contact addition per electrode for the ON–ON group. However, Crowell et al. 10 reported positive long-term open-label SCG-DBS results with stimulation up to 9 mA. It is possible that the steady improvement in antidepressant response and remission, which reach those of other trials,9,10 may be due to the higher amplitudes of stimulation beyond the 6-month RCT. Furthermore, amplitude was the only stimulation parameter modified. Higher stimulation parameters may increase the volume of tissue activation, potentially contributing to the progressively better antidepressant outcomes observed in long-term cohorts.19,44
Third, electrode implantation alone, in both animal and clinical studies, can trigger biobehavioural changes that can persist months post-surgery. 45 Although the precise mechanisms of insertional effects remain unclear, they may involve peri-electrode edema, micro-haemorrhage, physical manipulation of brain tissue, or lingering effects of intra-operative test stimulation.46–48 In this context, 1 partial explanation for the OFF group's better-than-expected outcomes at 3 months could be the greater proximity of this group's electrodes to the right cingulum bundle.
Indeed, the high overall proportion of participants with electrode contacts positioned directly within the cingulum bundles and corpus callosum arguably increases the possibility that insertional effects may have played a role in the clinical outcomes observed in this trial. As having an extended period without active stimulation following an invasive procedure such as DBS may potentially raise concerns about withholding treatment to patients with severe and chronic forms of MDD, future RCTs can consider a pre-randomization period for symptom baseline return or an extended open-label optimization phase before double-blind continuation or discontinuation. 49
Fourth, the sham response rate was not negligible, as in other DBS studies.9,50 Meta-analysis of 50 RCTs on TRD treatments, including brain stimulation, found a large pooled placebo effect size (Hedges’ g = 1.05), comparable across modalities. 51 Frequent visits, necessary for DBS safety and adjustments, 52 may further elevate placebo rates. 53 Furthermore, meta- analytic data indicate placebo effects alter activity in the dorsolateral prefrontal cortex (DLPFC) and SCG, 54 which may obscure the effects of DBS on these regions in sham-controlled trials. Thus, traditional RCT designs may be suboptimal for DBS, necessitating alternative designs. Additionally, patient expectations undoubtedly played a role in the outcomes. Positive pre-surgery expectations of DBS to treat Parkinson's disease lead to better motor responses.55,56 In a large prospective cohort of patients with TRD receiving repetitive transcranial magnetic stimulation (rTMS), higher pre-treatment positive expectations improved remission rates. 57 Future RCTs of SCG-DBS should assess pre-treatment expectations on outcomes.
Fifth, recent studies suggest that DBS programming guided by electrophysiological biomarkers may improve treatment outcomes. In a proof-of-concept study, a closed-loop DBS system autonomously adjusting stimulation to a gamma power signature in the amygdala produced sustained antidepressant effects. 58 In SCG-DBS for TRD, Alagapan et al. 59 identified local field potentials in the SCG as a biomarker of depressive states that preceded clinical effects, and detected abnormalities before relapse. Biomarker-guided treatment led to a 90% response rate and 70% remission at 6 months, highlighting the potential of biomarker-driven DBS programming.
Conclusion
This double-blind RCT found no significant difference between active and sham SCG-DBS over 6 months but showed sustained improvements in the 2-year open-label phase. These persisting improvements, including motivation, wellbeing, and life functioning, are notable given the potential short-term trajectory of placebo responses, and a reduction in visit frequency to every 6 months. SCG-DBS was safe, with AEs consistent with previous trials. The Balaam crossover design used here was underpowered and the 3-month crossover phases may have been too brief to detect differences, as noted in previous DBS 9 and vagus nerve stimulation 60 trials. Future RCTs should optimize DBS targeting, trial design, and sham response reduction while integrating biomarkers to enhance precision and therapeutic effects.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251369226 - Supplemental material for Deep Brain Stimulation to the Subgenual Cingulate Gyrus for Treatment-Resistant Depression: A Randomized Controlled Trial and 2-Year Long-Term Follow-Up: Stimulation cérébrale profonde du gyrus cingulaire subgénual pour traiter la dépression résistante au traitement : Essai contrôlé à répartition aléatoire et suivi à long terme sur deux ans
Supplemental material, sj-docx-1-cpa-10.1177_07067437251369226 for Deep Brain Stimulation to the Subgenual Cingulate Gyrus for Treatment-Resistant Depression: A Randomized Controlled Trial and 2-Year Long-Term Follow-Up: Stimulation cérébrale profonde du gyrus cingulaire subgénual pour traiter la dépression résistante au traitement : Essai contrôlé à répartition aléatoire et suivi à long terme sur deux ans by Peter Giacobbe, MD, MSc, FRCPC, Sakina J. Rizvi, PhD, Amanda K. Ceniti, PhD, George Tomlinson, PhD, Gavin J.B. Elias, MD, PhD, Jürgen Germann, PhD, Rima Styra, MD, MSc, FRCPC, Andres M. Lozano, MD, PhD and Sidney H. Kennedy, MD, FRCPC in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
This study was partly sponsored by Abbott Laboratories and discretionary research funds from Sidney Kennedy (principal investigator). The role of the sponsor was to ensure regulatory standards for study conduct, data collection, and data monitoring. The statistical analysis was conducted by an independent biostatistician from the study team and sponsor (GT).
Declaration of Conflicting Interests
PG, AKC, GT, GJBE, JG and RS have no disclosures to report. SJR has received research funds or consulting fees from Allergan Canada, Pfizer Canada, Neurocrine Biosciences Inc., and Janssen Pharmaceuticals. She is the copyright holder for the Dimensional Anhedonia Rating Scale. AML is the co-founder of Functional Neuromodulation (a DBS-related company) and is a consultant for Boston Scientific, Medtronic, and Abbott (companies that produce DBS hardware). SHK has received honoraria or research funds from Abbott, Alkermes, Allergan, Boehringer Ingelheim, Brain Canada, Canadian Institutes of Health Research, Janssen, Lundbeck, Ontario Brain Institute, Ontario Research Fund, Otsuka, Pfizer, Servier, Sunovion, and Sun Pharmaceuticals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Abbott Laboratories.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
