Abstract
Background
Emotional processing deficits and frontolimbic dysfunction have been observed in patients with obsessive-compulsive disorder (OCD), with inconsistent evidence possibly due to symptom heterogeneity. We compared the functional activation and connectivity patterns of the frontolimbic structures during symptom provocation between patients with distinct symptom profiles of OCD.
Methods
Thirty-seven symptomatic OCD subjects were recruited and categorized based on predominant symptom profiles to contamination/washing symptom group (OCD-C, n = 19) and taboo thoughts group (OCD-T, n = 18), along with 17 healthy controls (HCs). All subjects were evaluated with comprehensive clinical assessments and functional magnetic resonance imaging while appraising personalized disorder-specific stimuli with contrasting neutral stimuli as part of an individualized symptom provocation task. Region of interest analyses and task-dependent seed-to-voxel connectivity of the frontolimbic circuit were compared between the groups, with correction employed for multiple comparisons.
Results
OCD-C subjects had decreased task-dependent mean activation of the left amygdala (adjusted mean difference = 13.48, p= 0.03) and right hippocampus (adjusted mean difference = 13.48, p = 0.04) compared to HC. Task-modulated functional connectivity analyses revealed that OCD-C had decreased connectivity of the right hippocampus with bilateral supplementary motor cortex and anterior cingulate gyrus (T = -5.11, p = 0.04); right insula with left cerebellum (T = -5.47, p = 0.02); and left insula with inferior temporal gyrus (T = -6.27, p = 0.03) than HC. OCD-T subjects had greater connectivity of right insula with left cerebellum (T = 6.64, p < 0.001) than OCD-C and increased connectivity of medial frontal cortex with right lateral occipital cortex (T = 5.08, p < 0.001) than HC.
Conclusions
Contamination-related symptoms were associated with decreased activation and connectivity of amygdala and hippocampus during symptom provocation, while the taboo thoughts were associated with increased connectivity of the insular cortex and medial frontal cortex. These findings suggest that distinct neurobiological markers may underlie the clinical heterogeneity of OCD.
Introduction
Despite its current nosological status as a unitary entity, obsessive-compulsive disorder (OCD) shows considerable heterogeneity in clinical profile, course of illness, and treatment response. 1 Factor analytic studies have found temporally and cross-culturally stable sets of symptom dimensions, including symmetry/arranging, aggression, forbidden/taboo thoughts, contamination/cleaning, and hoarding.2–5 These symptom profiles are correlated with distinctive clinical manifestations in terms of age at onset, comorbidities, degree of insight, and neuropsychological deficits, supporting their validity.6–8 Recent neuroimaging studies have shown that the standard cortico-striato-thalamo-cortical (CSTC) model alone may be inadequate to explain this heterogeneity 9 and indicated evidence for the involvement of wider cortical and subcortical structures beyond the CSTC circuit.10,11 It has been postulated that the diverse clinical presentations and the associated neurocognitive deficits may be explained by the varied involvement of the dorsal cognitive, ventral cognitive, frontolimbic, sensorimotor, and frontoparietal networks.12,13
Dysregulated fear, anxiety, or distress is often seen in OCD patients with an attentional bias toward stimuli with negative valence.14–18 Frontolimbic circuit comprising of amygdala, ventromedial prefrontal cortex, ventral anterior cingulate cortex, and hippocampus is implicated in the generation and evaluation of emotional responses, with a top-down control exerted by dorsal prefrontal cortex to regulate the generated emotions.12,13 Symptom provocation tasks designed using disorder-specific stimuli (using generic, symptom dimension-related or individualized stimuli) and contrasting non-specific (general aversive or neutral) stimuli are commonly employed in functional magnetic resonance imaging (fMRI) studies to study emotional processing deficits. 19 OCD patients have shown hyperactivity in bilateral amygdala, right putamen, anterior cingulate cortex, right insula, ventromedial prefrontal cortex, and right caudate extending to temporal and occipital cortices during symptom provocation.20,21
Few studies have compared the neural activation patterns during symptom provocation between OCD patients with different symptom profiles. Patients with predominant contamination symptoms, when presented with standardized OCD-related triggers, had altered activity of amygdala extending to frontal and subcortical structures, and patients with doubts/checking symptoms had increased hippocampal activation.22–24 The severity of taboo thought-related symptoms is found to correlate with hyperactivity in limbic/paralimbic structures, including amygdala, anterior insula, anterior cingulate cortex, and lateral prefrontal cortex in a moral dilemma situation, which could be attributed to the heightened moral sensitivity experienced by these patients. 25 It can be thus surmised that taboo thought-related OCD may be associated with a hyperactive limbic system.
A majority of the OCD symptom induction-based fMRI studies employed standardized triggers, which could have affected the efficacy of provocation, as the content of symptoms greatly varies among individual patients. Individualized symptom provocation (ISP) pictures are noted to be associated with stronger activation of OCD-relevant regions, with some symptom-specific activation.26,27 Further, the inclusion of OCD patients with mixed symptom profiles may have confounded the results, leading to conflicting findings. Studying patients presenting with distinctive symptom profiles using ISP task may throw more light on the distinctive neural correlates of symptom profiles of OCD.
The current study attempts to compare the neural correlates of emotional provocation in OCD patients with predominantly contamination and taboo thought symptoms. These symptom profiles have been chosen as they have been consistently found to be distinctive in most factor or cluster analysis studies with distinct clinical manifestation, underlying cognitive biases, and preliminary evidence for differential neuroimaging correlates.3,21,22 We aimed to compare the neural activation and connectivity patterns of frontolimbic circuit encompassing dorsomedial prefrontal cortex, ventromedial prefrontal cortex, anterior cingulate cortex, anterior insula, amygdala, and hippocampus between OCD patients with these two distinct symptom profiles and healthy individuals while visualizing personalized disorder-specific triggers.
Materials and Methods
Participants
Fifty-four subjects, including 37 OCD patients and 17 healthy controls (HCs), participated in the study. Patients were recruited from the specialty OCD clinic of the National Institute of Mental Health and Neuro Sciences (NIMHANS), Bengaluru, India. All recruited patients were symptomatic and had a diagnosis of primary OCD without other comorbid major psychiatric disorders. HCs were recruited from the community by word of mouth and comprised of students/staff of the institute or healthy caregivers of patients. All participants were right-handed, had at least 12 years of education with the ability to read/write English, and had no medical or neurological comorbidities or contraindications to undergo magnetic resonance imaging. The Institutional Ethics Committee approved the study, and written informed consent was obtained from all participants. A detailed description of research design and participant details is given in the Supplement.
Clinical and Diagnostic Assessments
The diagnosis and comorbidities were confirmed with the Mini International Neuro-Psychiatric Interview (MINI 7.0.2). 28 For the OCD subjects, the severity of OCD and symptom profiling were evaluated using the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS), Yale-Brown Obsessive-Compulsive Scale-Symptom Checklist (Y-BOCS-SC) and the Dimensional Yale-Brown Obsessive-Compulsive Severity Scale (D-YBOCS).29–31 OCD patients were categorized into either contamination obsessions/washing compulsions (OCD-C) (n = 19) or taboo thoughts, i.e. sexual or religious obsessions and related compulsions (OCD-T) (n = 18) groups based on the predominant symptom dimension. We defined the predominant symptom dimension as the clinical profile reported as the principal current and lifetime symptom in the Y-BOCS-SC, is present from the onset of illness, and is reported as most time-consuming and impairing. OCD patients with concomitant principal symptoms in any other dimensions in the Y-BOCS-SC or D-YBOCS were excluded. The severity of anxiety and depressive symptoms were evaluated in all participants (OCD and HC groups) using the Hamilton Anxiety Rating Scale (HAM-A) and Hamilton Depression Rating Scale (HAM-D), respectively.32,33
Functional Magnetic Resonance Imaging Paradigm
Stimuli Selection
All participants were exposed to a set of personalized OCD and neutral stimuli during MRI employing ISP task. A detailed symptom hierarchy worksheet was created for individual OCD subjects during the clinical interview, enumerating situations or triggers for the principal obsessive-compulsive symptoms. Pictures simulating the triggering situations were collected either from the subjects’ home environment or other open sources as suggested by the participant. These disorder-specific stimuli were rated by patients on a scale of subjective units of distress (SUD) ranging from 0 to 10. To ensure effective symptom provocation for OCD patients, stimuli with SUD between 5 and 9 were included in the final ISP task. The personalized disorder-specific pictures collected for OCD subjects were pooled and used for HC while preserving the confidentiality of patients. Similar to the OCD subjects, HC group was also evaluated for the associated distress for these pooled personalized OCD stimuli with SUD scale (Table S3). It was ensured that the HC group had equal exposure to both disorder-specific triggers (6 stimuli from each sub-group per trial) to avoid symptom dimension-specific effects.
A set of neutral stimuli (with SUD < 5) was identified through piloting from an independent sample of OCD patients. Contrasting neutral triggers were chosen from this Neutral set for all the current study participants undergoing imaging after confirming whether the SUD satisfies the pre-specified criteria. All the images were formatted to ensure uniform contrast and saturation (within and across conditions) before generating the task.
Individualized Symptom Provocation Task
The task consisted of three trials with eight blocks per trial. Each trial consisted of 12 OCD and 12 neutral stimuli presented randomly in four blocks of three images. Each stimulus was presented for 10 s, with an inter-block interval of 2200 msec. The second and third trials were the repetition of the first trial (Figure 1b). All participants were instructed to actively imagine themselves in the situation while seeing the images, appraise the situation and their thoughts/emotions, and not to divert their attention or perform mental compulsions. During the scan, participants confirmed the presence of distress (SUD >5) for each stimulus through a button press recorded as a binary response (yes or no). After the fMRI data acquisition, SUDs were re-evaluated to confirm persistent provocation. Further details on the selection and standardization of OCD/neutral stimuli are described in the Supplement.
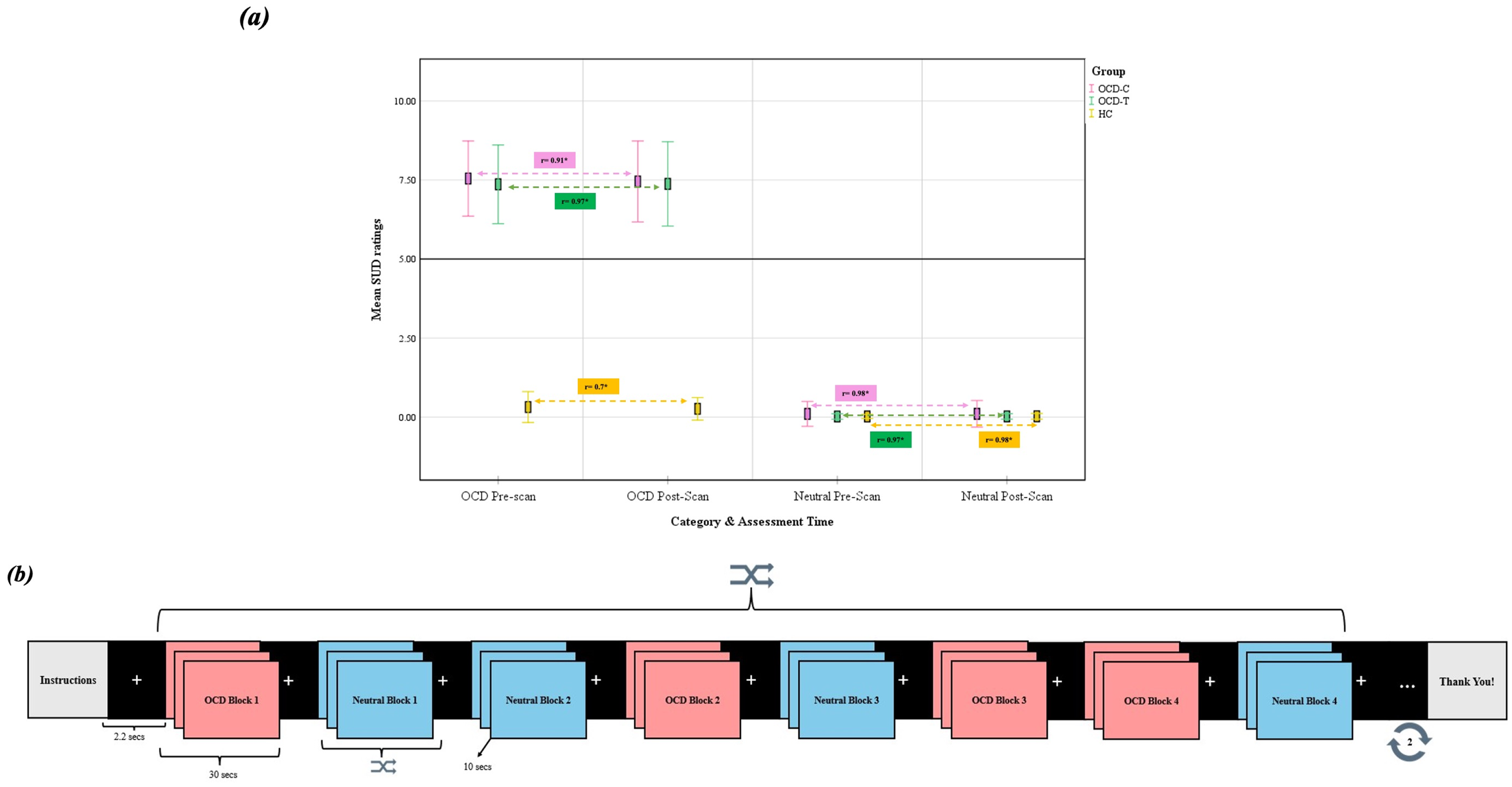
Individualized symptom provocation (ISP) task. (a) Distress ratings for the stimulus conditions before and after scan with correlation scores. (b) fMRI paradigm design. fMRI=functional magnetic resonance imaging.
Functional Magnetic Resonance Imaging Data Acquisition
Magnetic resonance imaging data were acquired using a 3.0 Tesla scanner (Philips, Ingenia). High-resolution T1-weighted single-shot 3D turbo field echo (TFE) images were obtained with a repetition time (TR) of 2500 ms, echo time (TE) of 2.9 ms, 9° flip angle, field of vision (FOV) of 256 × 256 × 192 mm, slice thickness of 1 mm without inter-slice gap, 192 slices, matrix of 256 × 256, and voxel size of 1 × 1 × 1 mm. The blood oxygen level-dependent (BOLD) sensitive echo-planar sequences were acquired while subjects were performing the task with TR of 2200 ms, TE of 28 ms, 80° flip angle, FOV of 211 × 211 mm, 3 mm slice thickness with inter-slice gap of 0.3 mm, 44 slices, matrix of 64 × 62 × 64, and voxel size of 3.0 mm. The sequence lasted for 10 min, and 300 dynamic scans were obtained.
Clinical and Behavioral Data Analyses
Sociodemographic and clinical variables across three groups were compared using analysis of variance (ANOVA). The group-level comparisons of the mean distress (SUD) scores associated with OCD and neutral conditions before and after scans were performed by ANOVA with post-hoc analyses. The percentage of stimuli causing distress/anxiety of SUD >5 was calculated for both conditions and compared between groups using ANOVA. Group-wise Pearson correlation coefficient (r) was calculated to assess the linear association between mean SUD scores for each stimulus before and after the scan (Figure 1a).
Imaging Analyses
Functional Magnetic Resonance Imaging Data Preprocessing and Denoising
The fMRI data was preprocessed using fMRIPrep through ENIGMA HALFpipe.34,35 Preprocessing involved head motion estimation and correction, susceptibility distortion correction employing phase difference field maps, and outlier detection followed by coregistration and spatial normalization. Realignment of the functional data was performed where all dynamic scans were coregistered to the first scan of the session as a reference image employing a six-parameter rigid body transformation, and scans were resampled using b-spline interpolation to correct for motion and magnetic susceptibility interactions. Potential outliers were identified with framewise displacement above 0.5 mm or global BOLD signal changes above five standard deviations. Functional and anatomical data were normalized into standard MNI space, segmented into gray matter, white matter, and CSF tissue classes, and resampled to 2 mm isotropic voxels following a direct normalization procedure. Final preprocessed functional images were obtained after applying smoothing with a 6 mm full-width half maximum kernel and grand mean scaling of 10000.
Noise reduction was performed by implementing an anatomical component-based noise correction (aCompCor) procedure by regression of potential confounding effects from cerebral white matter, cerebrospinal fluid, head-motion parameters, outlier scans, and task effects. From the number of noise terms included in this denoising strategy, the effective degrees of freedom of the BOLD signal after denoising were estimated to range from 61.9 to 116 (average 100.5) across all subjects. Quality assessments to check for normalization, registration, and motion artifacts were performed on the final preprocessed functional images prior to first-level and group-level analyses.
Region of Interest Analyses
Anatomical masks were created on the MNI152NLin2009cAsym template with Harvard-Oxford Atlas parcellation (templateflow, Version 0.1.9, May 2019) 36 bilaterally for ventromedial prefrontal cortex, dorsomedial prefrontal cortex, amygdala, hippocampus, anterior cingulate cortex, and anterior insula. A 5 mm sphere was created around the reference voxel, and these selected voxels within the sphere were binarized (Figure S4).
Confirmatory analyses were conducted on FSL fMRI expert analysis tool (FEAT). 37 Thresholded statistical images were generated with contrast OCD > Neutral for each participant. Average beta aggregates from these anatomical seeds were interrogated from the statistical images of individual participants. Group-level comparisons of the mean activity across frontolimbic structures were performed employing an analysis of covariance model after controlling for the age and sex distribution, homogeneity of the groups was evaluated with Levene's test for equality of variances, and post-hoc analyses were conducted with Bonferroni correction. The uncorrected p-values obtained were controlled for multiple comparisons using Bonferroni correction with a p-value set to <0.05.
Task-Modulated Functional Connectivity Analyses
The task-dependent functional connectivity between the hypothesized region of interest to every voxel was measured employing the generalized psychophysiological interference model on CONN functional connectivity toolbox version 22a (CONN functional connectivity toolbox: RRID SCR_009550, release 22. Hilbert Press. doi:10.56441/hilbertpress.2246.5840).38,39 The physiological regressor employed was the averaged BOLD time series extracted from the above-mentioned 11 Harvard-Oxford atlas ROIs included as seed regions. The psychological regressor was the task effect calculated for the contrast of OCD > Neutral convolved with the hemodynamic response function. Functional connectivity changes across conditions were characterized by the multivariate regression coefficient of the psychophysiological interaction terms in each model.
Group-level comparisons were done using a general linear model with each voxel as a dependent variable and groups of subjects as the independent variable after controlling for the effect of age and sex. Voxel-level hypotheses were evaluated using multivariate parametric statistics with random effects across subjects and sample covariance estimation across multiple measurements. Inferences were performed at the level of individual clusters (groups of contiguous voxels). Cluster-level inferences were based on parametric statistics from Gaussian Random Field theory. Results were thresholded with a combination of uncorrected voxel-level p < 0.001 and cluster-size FDR corrected (p-FDR) < 0.05. 40 Cluster-size p-thresholds were further FDR corrected to control for multiple comparisons. Additional sensitivity analyses are elaborated on in the Supplement (Tables S5 & S6).
Results
Demographic and Clinical Measures
The subjects across three groups were comparable in all sociodemographic domains except for sex (Table 1). The OCD-T group had higher anxiety and depression symptom scores than the other two groups, although none of the participants had more than mild severity of symptoms (Figure S3).
Sociodemographic and Clinical Characteristics of the Sample.
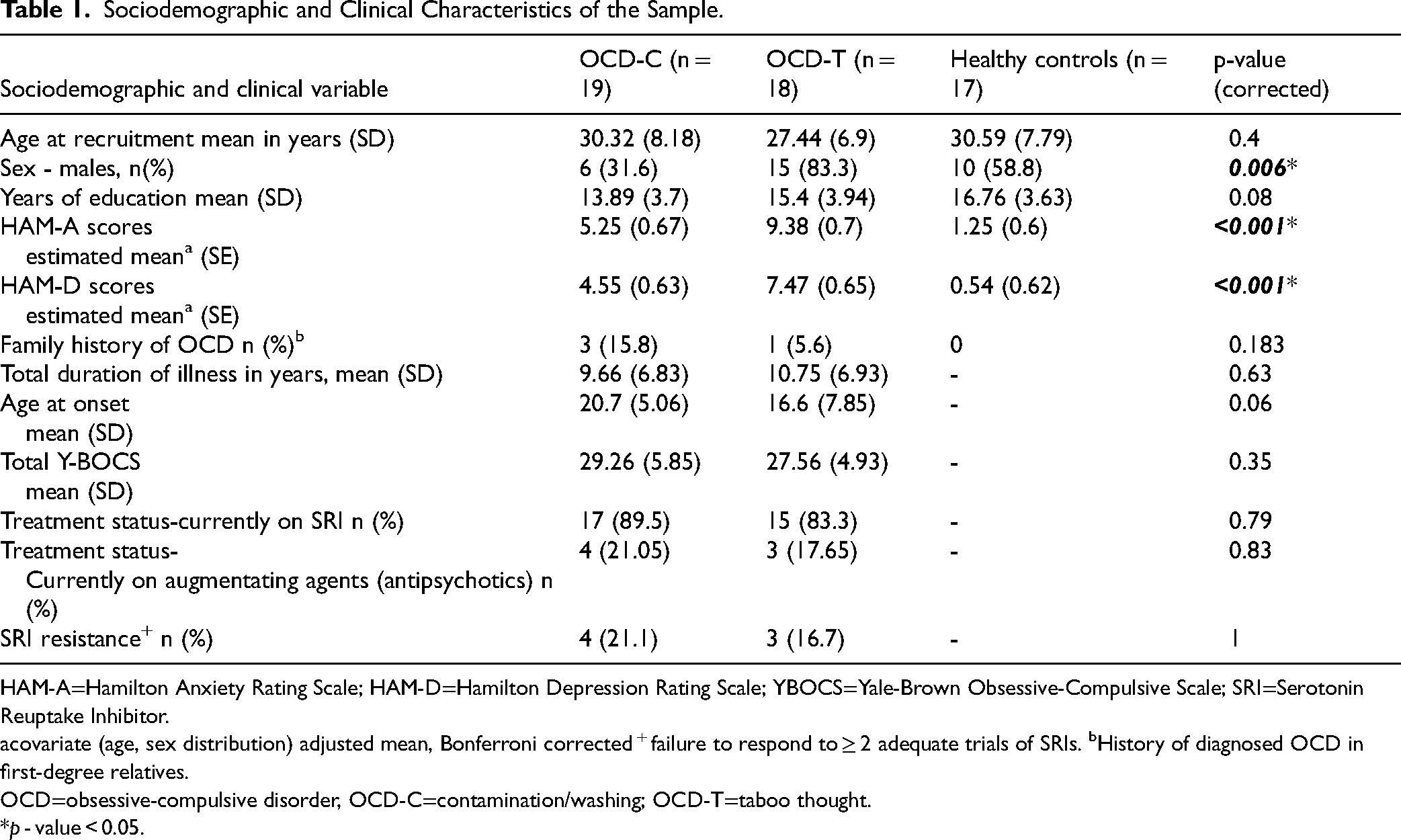
HAM-A=Hamilton Anxiety Rating Scale; HAM-D=Hamilton Depression Rating Scale; YBOCS=Yale-Brown Obsessive-Compulsive Scale; SRI=Serotonin Reuptake Inhibitor.
covariate (age, sex distribution) adjusted mean, Bonferroni corrected + failure to respond to ≥ 2 adequate trials of SRIs. bHistory of diagnosed OCD in first-degree relatives.
OCD=obsessive-compulsive disorder, OCD-C=contamination/washing; OCD-T=taboo thought.
*p - value < 0.05.
Effectiveness of Symptom Provocation
The mean distress reported for OCD stimuli pre- and post-scan were comparable between patient sub-groups. HCs experienced minimal provocation with disorder-specific stimuli before and after scan (Figure 1(a) and Table S3). Out of 36 OCD stimuli visualized during the scan, SUD >5 was reported by the OCD-C group on an average of 98.54% (SD: 2.98) and the OCD-T group for 97.9% (SD: 3.41). A significant linear correlation was noted between the SUD scores of both the conditions, pre- and post-scans (Figure 1(a)).
Task-Based Activation
The between-group difference for the task contrast of OCD > Neutral is illustrated in Table S4. OCD subjects with contamination symptoms were noted to have significantly reduced activity in the left amygdala during symptom provocation than those with taboo thought symptoms (adjusted mean difference = 15.68, p(Bonferroni corrected)= 0.01), and HCs (adjusted mean difference = 13.48, p(Bonferroni corrected)= 0.03). The contamination group was also noted to have reduced right hippocampus mean activity as compared to healthy individuals (adjusted mean difference = 17.05, p (Bonferroni corrected)= 0.04) while visualizing disorder-specific triggers over neutral triggers (Figure 2). However, the significant group comparisons did not survive FDR corrections.
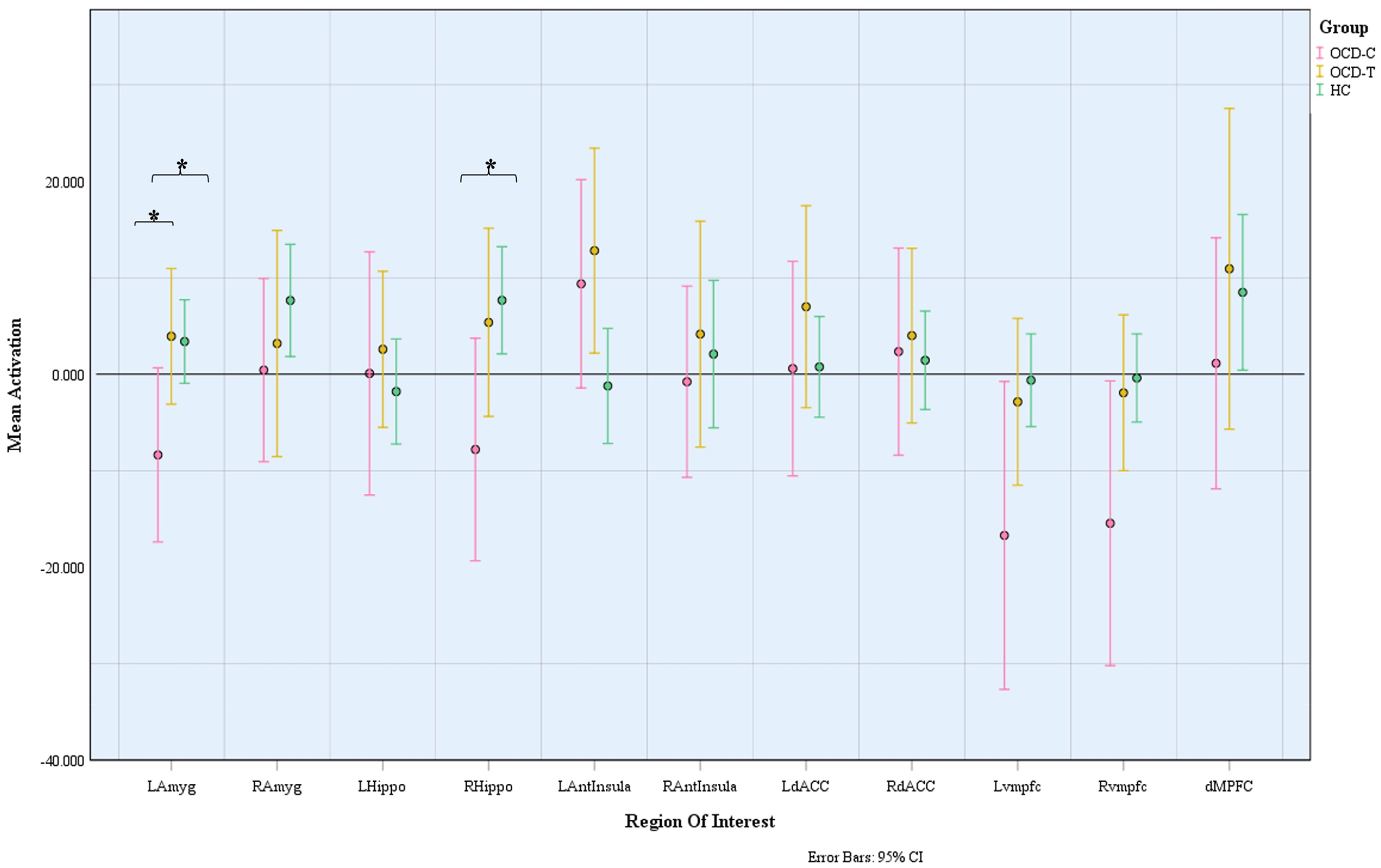
Task-based estimated mean activation during symptom provocation state (OCD > Neutral). OCD=obsessive-compulsive disorder. LAmyg=left amygdala; RAmyg=right amygdala; LHippo=left hippocampus; RHippo=right hippocampus; LAntInsula=left anterior insula; RAntInsula=right anterior insula; LdACC=left dorsal anterior cingulate cortex; RdACC=right anterior cingulate cortex; Lvmpfc=left ventromedial prefrontal cortex; Rvmpfc=right ventromedial prefrontal cortex; dMPFC=dorsomedial prefrontal cortex.
Task-Modulated Functional Connectivity
The significant group-level comparisons of the whole-brain task-based connectivity changes are depicted in Table 2. The OCD-T subjects had greater connectivity between right insula and left cerebellum than OCD-C group (T = 6.64, cluster size p-FDR = <0.001) and increased connectivity between medial prefrontal cortex and superior division of right lateral occipital cortex than HCs (T = 5.08, cluster size p-FDR = <0.001) while visualizing disorder-specific over neutral stimuli. Healthy subjects had greater connectivity of the right hippocampus with bilateral supplementary motor area and anterior cingulate gyrus (T = 5.11, cluster size p-FDR = 0.04), and bilateral insular cortex with left middle temporal cortex, left cerebellum, and bilateral intracalcarine cortex while appraising disorder-specific triggers than subjects with contamination symptoms (right insula: T = 5.47, and cluster size p-FDR = 0.02, T = 4.55, cluster size p-FDR = 0.03; left insula: T = 6.27, cluster size p-FDR = 0.03). The connectivity changes in OCD-T subjects remained significant after multiple comparison correction with corrected p < 0.05 (Figure 3).
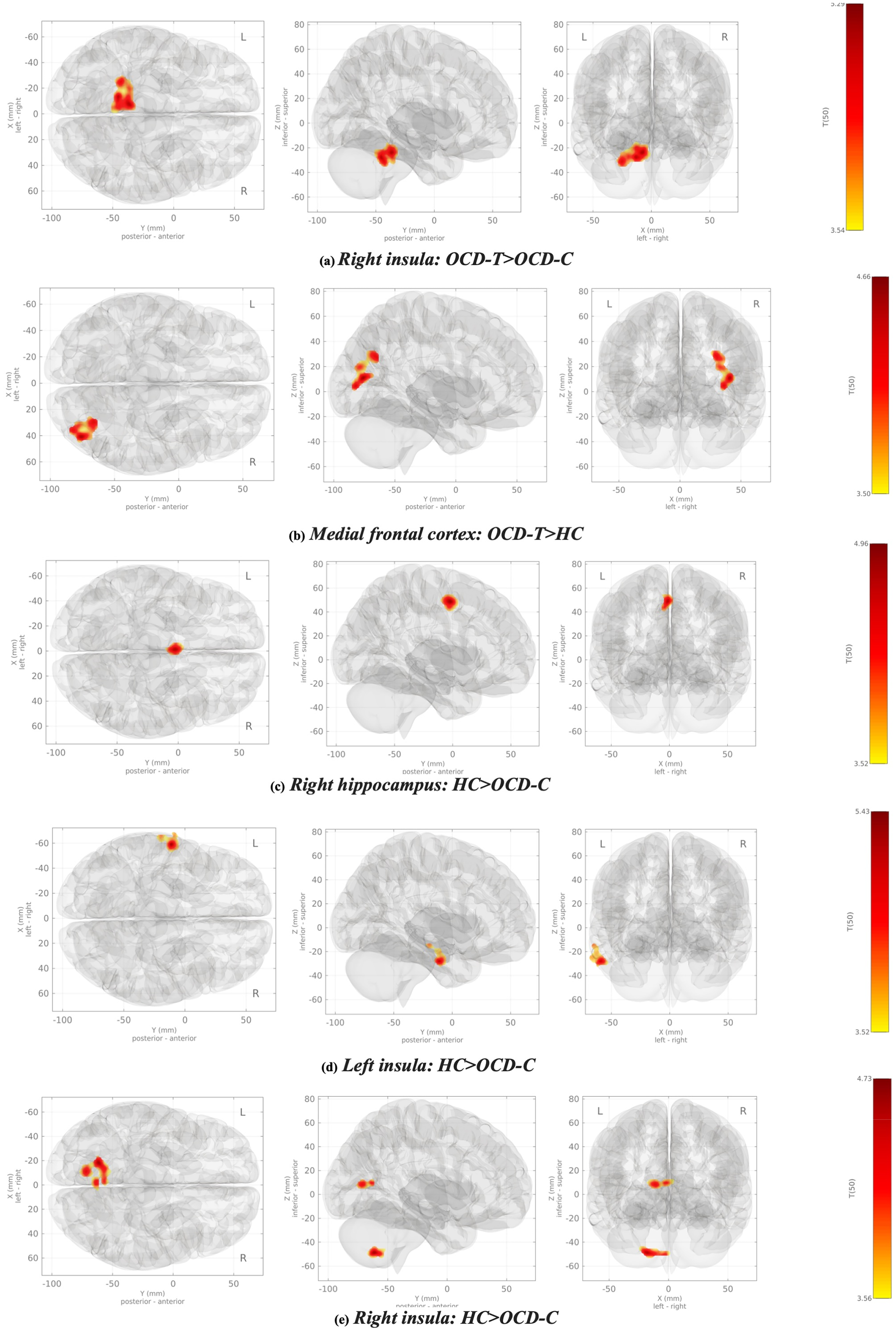
Network maps depicting the task-based functional connectivity of frontolimbic structures during emotionally provoked condition (OCD > Neutral). (a) Right insula: OCD-T > OCD-C; (b) medial frontal cortex: OCD-T > HC; (c) right hippocampus: HC > OCD-C; (d) left insula: HC > OCD-C; (e) right insula: HC > OCD-C. OCD=obsessive-compulsive disorder; HC=healthy control.
Task-Modulated (OCD > Neutral) Connectivity of Frontolimbic Circuit.

p-FDR: controlled for comparison across 11 seed regions.
OCD=obsessive-compulsive disorder; OCD-C=contamination/washing; OCD-T=taboo thought; HC=healthy control.
*p-value < 0.05.
Discussion
In this study, we found that the OCD-C had decreased activation of the left amygdala compared to both HC and OCD-T groups during the processing of personalized symptom provocation images. The OCD-C group also had decreased activation of the right hippocampus compared to the HC group. Task-modulated functional connectivity analyses revealed that OCD-C had decreased connectivity of the right hippocampus with bilateral supplementary motor cortex and anterior cingulate gyrus, right insula with left cerebellum, and left insula with inferior temporal gyrus than HC. OCD-T subjects had greater connectivity of right insula with left cerebellum than OCD-C and increased connectivity of medial frontal cortex with right lateral occipital cortex. Thus, we found evidence for aberrant activity and connectivity in emotional processing circuits, which differentiated the contamination and taboo thoughts groups.
As these aberrations were found during a personalized symptom provocation task, it could be explained by dysfunction in emotional processing and emotional memory, which are modulated by the amygdala and hippocampus. 41 The left amygdala has an established role in the rapid processing of image- or language-related affective information, and amygdala activation is hypothesized to facilitate representation and retrieval of emotional memory by hippocampus.42–44 Reduced activation of these structures in OCD-C subjects can thus indicate an inadequate emotional evaluation and registration of the provocative stimuli by the subjects with contamination dimension. Although previous studies have shown heterogeneous results with respect to amygdala activation, a meta-analysis found evidence for amygdala hyperactivation during emotional processing, which was modulated by other factors including psychotropic use, concomitant anxiety, and depressive symptoms. 20 In contrast to the above findings, we found decreased amygdala activity in the contamination group compared to the other two groups. Symptom profile may explain the heterogeneous neuroimaging findings observed in previous studies. 45
Aberrant connectivity between hippocampus and SMA found in the OCD-C group can indicate inefficient utilization of retrieved emotional memory, leading to abnormal motor planning with abnormalities in execution of complex behaviors based on the inputs from the environment.11–13 This can potentially explain the underlying neural mechanisms of the distinct elaborate motor compulsions performed by patients with contamination-related obsessions as compared to patients with prominent unacceptable thoughts.
We also found altered hippocampal-anterior cingulate gyrus connectivity in the OCD-C group, which can be related to abnormal error monitoring, cognitive inflexibility, and emotional regulation deficits, frequently noted in OCD patients.46–48 Recent work has suggested that the fronto-hippocampal interactions, including that from the SMA and dACC, are involved in retrieval stopping, i.e. inhibition of unpleasant emotional memories, 49 which appears to be dysfunctional in the contamination group. Overall, an altered interaction between these components of frontolimbic and sensorimotor circuits could potentially mediate the influence of abnormal emotional processing, leading to disrupted cognitive control of the prepotent automated motor responses and the emergence of compulsive behaviors. 11
Further, those with contamination symptoms had reduced connectivity of left insula with the middle and inferior temporal gyrus (MTG/ITG). The latter regions are reported to facilitate semantic memory processing, visual perception, and integration of multimodal sensory inputs.50–52 Reduced activity of the left MTG among OCD subjects was reported to be associated with poor insight, reflecting probable deficits in memory encoding and retrieval, 53 and patients with contamination symptom profiles were reported to have significantly impaired visuospatial recognition memory and working memory than other symptom dimensions.7,54 The contamination group were also noted to have altered connectivity of right insula with left cerebellar regions compared to the other two groups. Cerebellum has a pivotal role in emotional processing learning.55,56 Altered cerebellar activity has been observed in OCD patients, which is hypothesized to be a compensatory phenomenon secondary to cortico-striatal dysconnectivity in OCD.57,58
Patients with taboo thoughts had enhanced connectivity of the medial frontal cortex with the right occipital cortex. OCD patients experience behavioral inflexibility, impaired cognitive control, and abnormal error monitoring due to aberrant functioning of dorsal and ventral MFC.59–61 The MFC, right lateral occipital gyrus, and other visual association areas are involved in self-referential processing, and hyperactivation of cortical midline structures is associated with self-referential tasks in OCD patients.27,62,63 The increased connectivity of MFC with the visual association areas reflects heightened visual processing of stimuli with increased involvement of self-referential thinking during emotional provocation, presumably due to associated anxiety.
These findings must be interpreted cautiously, as the sample size was limited, which could have reduced the statistical power to identify other important findings and a possible explanation for why the task-based activation findings did not survive multiple comparison correction. Secondly, as most of the OCD subjects were on psychotropics, thus we could not control for the potential effects of medications on neural activation patterns. Further, we employed disorder-specific stimuli from the personal environment of OCD subjects to compare neural activation in healthy individuals. Thus, the effect of novelty with these stimuli in HCs could not be controlled considering the self-referential significance that OCD subjects hold for these stimuli. However, the distinctive reduced connectivity observed in the contamination group alone and not in the taboo thoughts group in contrast to healthy individuals hints that these findings might be related to symptom provocation rather than familiarity.
The addition of general aversive stimuli to our provocation paradigm could have helped understand whether the responses are specific to OCD triggers or a generalized reaction to any aversive stimulus. We focused on the front-limbic circuit alone and did not assess other brain regions implicated in OCD, such as the striatum.12,13 Further, heterogeneity within a given symptom dimension cannot be disregarded. Contamination/washing symptoms can manifest secondary to fear, perfectionism, disgust sensitivity, or sensory phenomena. Taboo thoughts also have heterogeneous content.
Overall, the current study demonstrates that OCD patients with different symptoms may have distinctive neurobiological profiles. This is supported by the different neurobiological responses to similar tasks in the current and previous studies evaluating patients with distinct symptoms. 45 Deep clinical and cognitive phenotyping of symptom dimensions on larger samples might help understand the heterogeneity of this complex disorder and foster research on symptom-specific treatment.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251328368 - Supplemental material for Differential Functional Connectivity of Frontolimbic Circuit During Symptom Provocation in Distinct Symptom Profiles of Obsessive-Compulsive Disorder: Connectivité fonctionnelle différentielle du circuit frontolimbique durant la provocation de symptômes dans des profils symptomatiques distincts du trouble obsessionnel-compulsif
Supplemental material, sj-docx-1-cpa-10.1177_07067437251328368 for Differential Functional Connectivity of Frontolimbic Circuit During Symptom Provocation in Distinct Symptom Profiles of Obsessive-Compulsive Disorder: Connectivité fonctionnelle différentielle du circuit frontolimbique durant la provocation de symptômes dans des profils symptomatiques distincts du trouble obsessionnel-compulsif by Navya Spurthi Thatikonda, Janardhanan C. Narayanaswamy, Ganesan Venkatasubramanian, Y. C. Janardhan Reddy and Shyam Sundar Arumugham in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
NST acknowledges the salary support received from the Department of Biotechnology, Ministry of Science and Technology, Government of India funded project “Accelerator Program for Discovery in Brain disorders using Stem cells” (ADBS) (BT/PR17316/MED/31/326/2015). SSA has received a research grant from DBT/Wellcome Trust India Alliance (IA/CPHI/18/1/503931).
Author Contributions
NST was responsible for conceptualization, subject recruitment, data collection, data analysis, interpretation of results, manuscript preparation; SSA was involved in conceptualization, planning of the study, interpretation of results, manuscript preparation and editing; JCN, VG, and YCJR were involved in conceptualization, planning of the study, interpretation of results and manuscript editing. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was carried out as a doctoral thesis work of NST funded by the Indian Council of Medical Research (ICMR) and a research bursary from the TSS foundation (TVSB/002/208/2020/01378).
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
