Abstract
Objective
The effectiveness of ECT under naturalistic conditions has not been well-studied. The current study aimed to 1) characterize a naturalistic sample of ECT patients; and 2) examine the long-term outcomes of ECT on depressive symptoms (Beck Depression Inventory-II; BDI-II) and functional disability symptoms (WHO Disability Assessment Schedule 2.0) in this sample.
Methods
Participants were adults who received ECT for a major depressive episode at an ambulatory ECT clinic between September 2010 and November 2020. Clinical and cognitive assessments were completed at baseline (n = 100), mid-ECT (n = 94), 2–4 weeks post-ECT (n = 64), 6-months post-ECT (n = 34), and 12-months post-ECT (n = 19).
Results
At baseline, participants had severe levels of depressive symptoms (BDI-II: M = 41.0, SD = 9.4), and 62.9% screened positive for multiple psychiatric diagnoses on the MINI International Neuropsychiatric Interview. Depressive symptoms (F(4,49.1) = 49.92, P < 0.001) and disability symptoms (F(3,40.72) = 12.30, P < 0.001) improved significantly following ECT, and this was maintained at 12-months follow-up. Improvement in depressive symptoms trended towards significantly predicting reduction in disability symptoms from baseline to post-ECT, (F(1,56) = 3.67, P = 0.061). Although our clinical remission rate of 27% (BDI-II score
Conclusions
Participants were severely symptomatic and clinically complex. ECT was effective at reducing depressive symptoms and functional disability in this heterogeneous sample. Although a large amount of missing data may have distorted our calculated response/remission rates, it is also likely that clinical heterogeneity and severity contribute to lower-than-expected remission and response rates to ECT.
Electroconvulsive therapy (ECT) is a highly efficacious approach for severe unipolar depression1,2, with ECT outperforming pharmacotherapies, simulated or sham ECT, and placebo1,3–5 A meta-analysis by Dierckz and colleagues 6 reported a remission rate of 50.9% for patients with unipolar depression. However, observed rates vary considerably, with remission rates from controlled trials ranging from between 30% to 100% 4 and often reported between 80–90%7,8. In such controlled trials, ECT is administered under ideal circumstances with study subjects selected to minimize comorbidity.
ECT is becoming more commonly used as a first-line approach for treating depression7,9 and, in practice, is typically administered to severely ill and diagnostically complex patients who have failed to respond to several other treatment interventions. This increased utilization highlights the importance of investigating ECT's effectiveness under naturalistic conditions where the clinical, demographic, and treatment characteristics of patients are more heterogeneous than those in prospective clinical trials. Here, naturalistic study of ECT is defined as ECT administered under ‘real-world’ conditions with no research-related restrictions on the patients seen or the way ECT is administered.
Research on the effectiveness of naturalistic ECT, often in community settings, is growing. The remission rates reported in naturalistic settings tend to be lower, with one large, multisite community study reporting remission rates between 30.3%-46.7% in a mixed mood disorders sample 10 . Patients referred for ECT typically present with depression complicated by numerous psychiatric comorbidities (e.g., anxiety, personality disorders), trauma history, complex pharmacotherapies, pre-existing cognitive impairment, and diverse medical comorbidities, which have been found to negatively impact response to treatment for affective disorders.11–15 However, the extent to which this heterogeneity impacts the effectiveness of ECT remains unclear and understudied.
Examining the effects of ECT in a heterogeneous sample provides a more ecologically valid assessment of the effectiveness of ECT. Accordingly, the present study aims to: 1) characterize a naturalistic sample of ECT patients demographically, clinically, and cognitively; 2) evaluate the short- and long-term effectiveness of ECT in remediating depressive symptoms; and 3) examine the impact of ECT on disability symptoms and examine whether changes in depressive symptoms predict a reduction in disability symptoms. The data collected for this study were from a naturalistic, longitudinal study of treatment response in depressed adults receiving ECT in an ambulatory ECT clinic.
Methods
Participants
Participants (N = 100) were adults who received acute outpatient ECT at the ECT Clinic at St. Joseph's Healthcare, Hamilton (SJHH) between September 2010 and November 2020. The sample was a mixed mood disorders population (e.g., Major Depressive Disorder, Bipolar Spectrum Disorders). Patients were excluded for inability to provide informed consent, complete assessments, or communicate adequately in English. See Figure 1 for participant flow data and Results section for a detailed sample characterization. Ethics approval was received from the SJHH Research Ethics Board (REB) and the Hamilton Integrated REB.
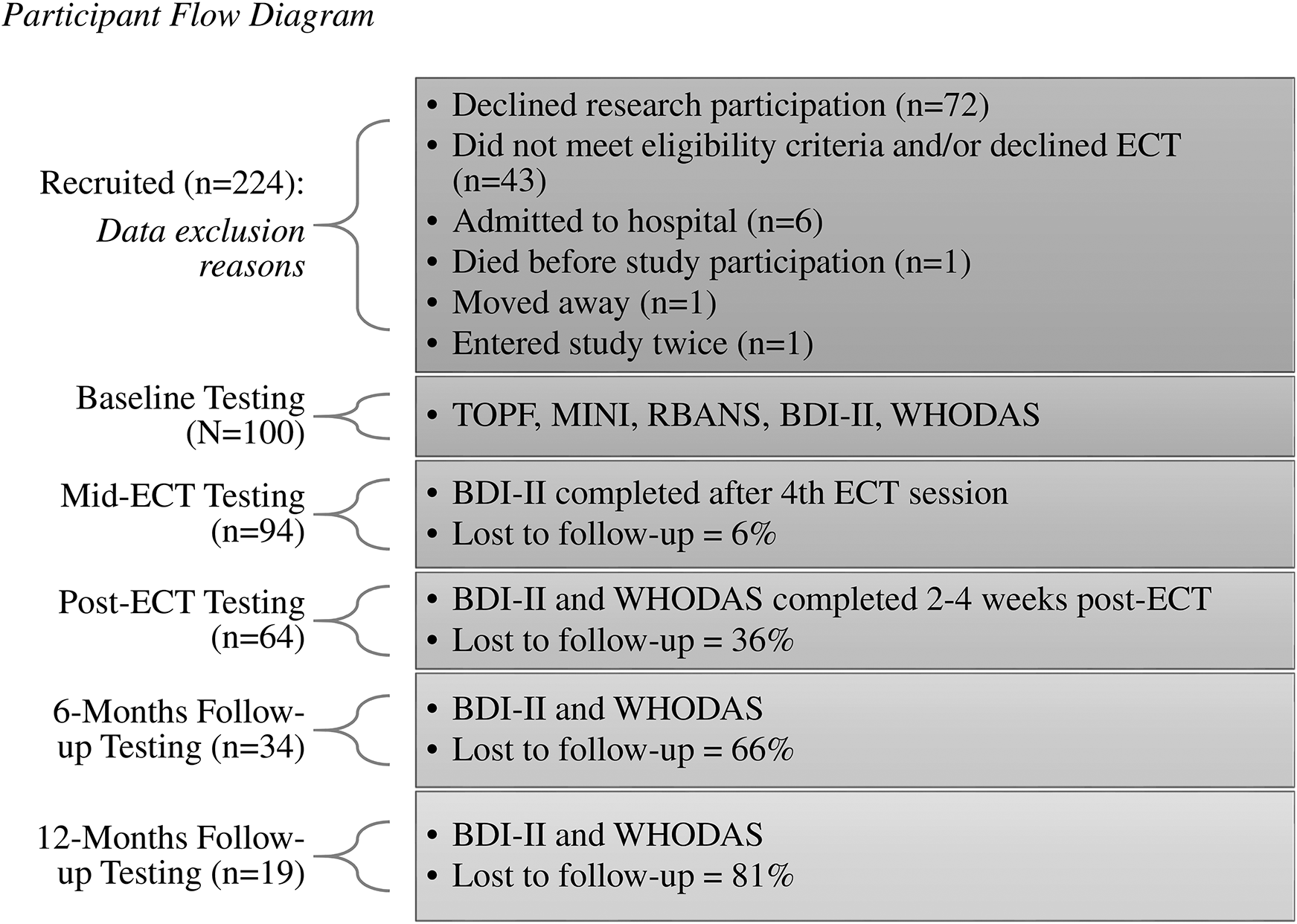
Participant flow diagram.
Assessments
Assessments were performed at the Clinical Neuropsychology Services and ECT Clinics at SJHH by trained nursing staff and a licensed clinical neuropsychologist and/or clinical and research staff supervised by a licensed clinical neuropsychologist. Additional measures administered for the larger project are not reviewed here.
The MINI International Neuropsychiatric Interview (MINI)16,17 version 7.0.0 screened for psychiatric diagnoses as per the Diagnostic and Statistical Manual for Mental Health Conditions–4th edition (DSM-IV-TR). The Personality Assessment Inventory (PAI)18,19, developed for use in clinical populations (e.g., severe depression), was used to capture additional psychological and emotional factors. PAI T-scores (standardized mean = 50) were used in subsequent analyses and comparison was made to the PAI's established reference ranges 20 , when relevant, to assess whether observed scores were elevated for clinical populations. The Beck Depression Inventory (BDI-II) 21 assessed depressive symptoms (severity ratings: 0–13 = minimal, 14–19 = mild, 20–28 = moderate, 29–63 = severe). The World Health Organization's Disability Assessment Schedule 2.0 (WHODAS) 22 measured daily health and disability functioning, with higher scores reflecting greater disability (score range: 0–48).
The Advanced Clinical Solutions Test of Premorbid Functioning (TOPF) 23 provided an estimate of premorbid intelligence. The Repeatable Battery for Assessment of Neuropsychological Status® (RBANS) 24 measured cognitive functioning in five domains/indices: immediate memory, delayed memory, visuospatial/constructional, language, and attention. Standardized scores (SS; mean = 100) were used for the TOPF and RBANS, with higher scores reflecting better cognitive performance.
Procedure
An ECT team psychiatrist and anesthesiologist evaluated patients to determine suitability for ECT before being approached about study participation. ECT was administered with a Thymatron ECT device using bitemporal (BIL) or right unilateral (RUL) electrode placement
25
based on discussion between the patient and prescribing physician. If there was no response after the sixth RUL ECT, BIL ECT was recommended. ECT Stimulus dosing was initially done using an age-based method
26
, and more recently by seizure threshold titration (i.e., 1.5X threshold for BIL ECT, 6X threshold RUL ECT). If the seizure duration was
Descriptive Statistics for ECT Variables.

Note. aRepresents the total number of patients who received a first, second, third, or fourth course (e.g., 100 participants completed a first acute course, of those 23 completed a second course, and so on). BIL = Bitemporal; BIL-RUL = started BIL and switched to RUL; ECT = Electroconvulsive Therapy; RUL = Right Unilateral; RUL-BIL = started RUL and switched to BIL.
Results
Analyses were conducted using IBM® SPSS® Statistics Version 27 with a 0.05 alpha level, two-tailed. Post-hoc analyses used Bonferroni correction. Missing data were excluded on an analysis-by-analysis basis. Relevant test assumptions were met unless otherwise specified.
Sample Characterization
Data are summarized in Table 2. At baseline, the sample (n = 100) had, on average, severe levels of depression (BDI-II: M = 41.0, SD = 9.4). On the MINI, 81.4% screened positive for Major Depressive Disorder, 18.9% for a Bipolar Spectrum Disorder, and 62.9% for more than one psychiatric diagnosis. Several baseline PAI scale T-scores were significantly elevated when compared to established guidelines 20 , including extremely elevated Depression (M = 88.0, SD = 11.1), significantly elevated Anxiety (M = 71.1, SD = 12.7), and moderately elevated Anxiety and Related Disorders–Traumatic Stress (M = 66.2, SD = 17.8).
Sociodemographic Characteristics of Participants at Baseline.
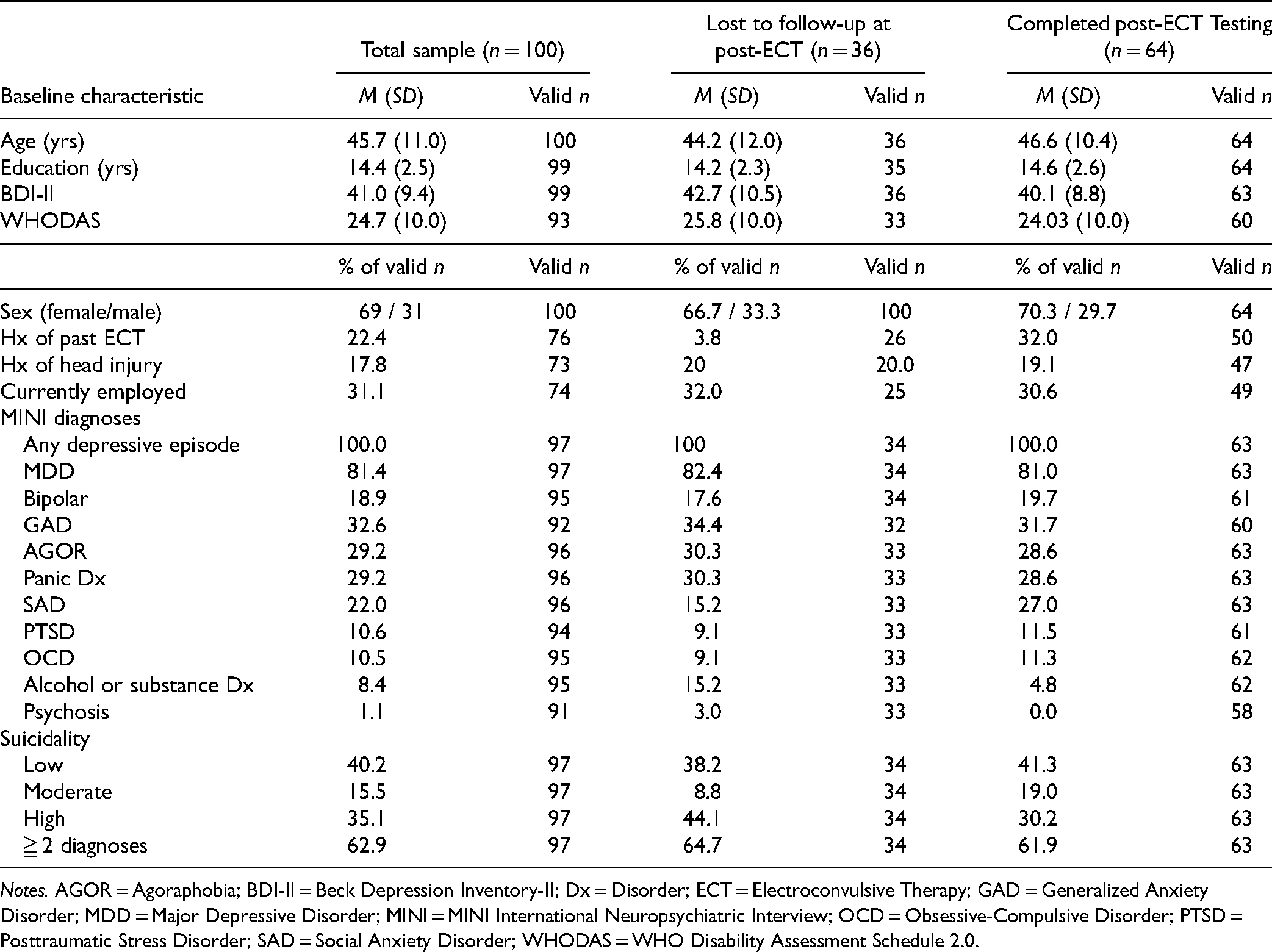
Notes. AGOR = Agoraphobia; BDI-II = Beck Depression Inventory-II; Dx = Disorder; ECT = Electroconvulsive Therapy; GAD = Generalized Anxiety Disorder; MDD = Major Depressive Disorder; MINI = MINI International Neuropsychiatric Interview; OCD = Obsessive-Compulsive Disorder; PTSD = Posttraumatic Stress Disorder; SAD = Social Anxiety Disorder; WHODAS = WHO Disability Assessment Schedule 2.0.
Mean TOPF SS and total RBANS SS fell within the average descriptor range (SS: 90–110) compared to standardized normative data. We examined whether patients’ current cognitive performance (RBANS) was comparable to their expected level of functioning based on a premorbid estimate (TOPF). Paired-samples t-tests revealed that baseline TOPF SS were significantly higher than baseline RBANS Attention, Immediate Memory, Delayed Memory, Visuospatial/Constructional, and Language indices, P < 0.001 (Table 3). Thus, our sample’s current cognitive performance, although not grossly impaired, was lower than expected compared to their own estimated premorbid functioning.
Cognitive Characteristics of Participants at Baseline.
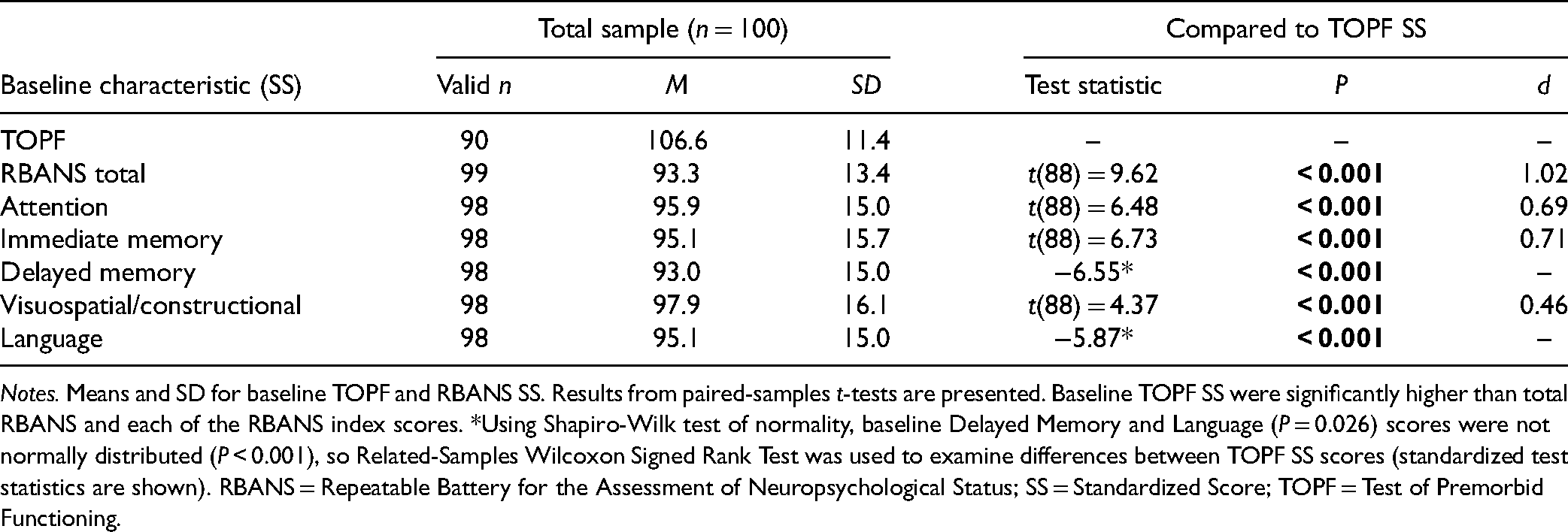
Notes. Means and SD for baseline TOPF and RBANS SS. Results from paired-samples t-tests are presented. Baseline TOPF SS were significantly higher than total RBANS and each of the RBANS index scores. *Using Shapiro-Wilk test of normality, baseline Delayed Memory and Language (P = 0.026) scores were not normally distributed (P < 0.001), so Related-Samples Wilcoxon Signed Rank Test was used to examine differences between TOPF SS scores (standardized test statistics are shown). RBANS = Repeatable Battery for the Assessment of Neuropsychological Status; SS = Standardized Score; TOPF = Test of Premorbid Functioning.
Differences Between Lost to Follow-up and Post-ECT Sample
To examine whether clinically meaningful differences existed between participants who completed the post-ECT assessment (n = 64) and those lost to follow-up (LTFU; n = 36), independent samples t-tests (continuous variables), Mann-Whitney U (non-normally distributed variables), and Pearson chi-square (categorical variables) were used. At baseline, those LTFU performed significantly worse on the RBANS Immediate Memory index, t(96) = −2.34, P = 0.022, d = −0.492, and the Delayed Memory index, Mann-Whitney U = 1,600.0, n1 = 35, n2 = 63, P < 0.001, than those who completed the post-ECT evaluation. Those LTFU were less likely to have received ECT in the past,
Clinical Effectiveness
Linear mixed modeling revealed a significant effect of time on BDI-II scores, F(4,49.1) = 49.92, P < 0.001. As expected, mean BDI-II scores were significantly lower at mid-ECT (M = 26.56, SE = 1.22, P < 0.001), post-ECT (M = 26.32, SE = 1.77, P < 0.001), 6-months post (M = 24.45, SE = 2.04, P < 0.001), and 12-months post (M = 28.26, SE = 2.99, P = 0.003), compared to baseline (M = 41.01, SE = 0.96).
Mid-ECT, 25 participants (26% of mid-ECT n=94, 25% of baseline N=100) had responded (
Post-ECT, 26 participants (41.3% of post-ECT n=64, 26% of baseline N=100) had responded, of which 17 (27%, 17% of N) had achieved remission. Thirty-seven participants (58.7%, 37% of N) were classified as non-responders. Post-ECT response was not calculated for 37% of participants (n = 36 LTFU; n = 1 missing baseline BDI-II data). Two participants who did not complete post-ECT testing returned for the 6-month post-ECT evaluation; one was classified as a responder, the other as a non-responder.
Differences Between Post-ECT Response Groups
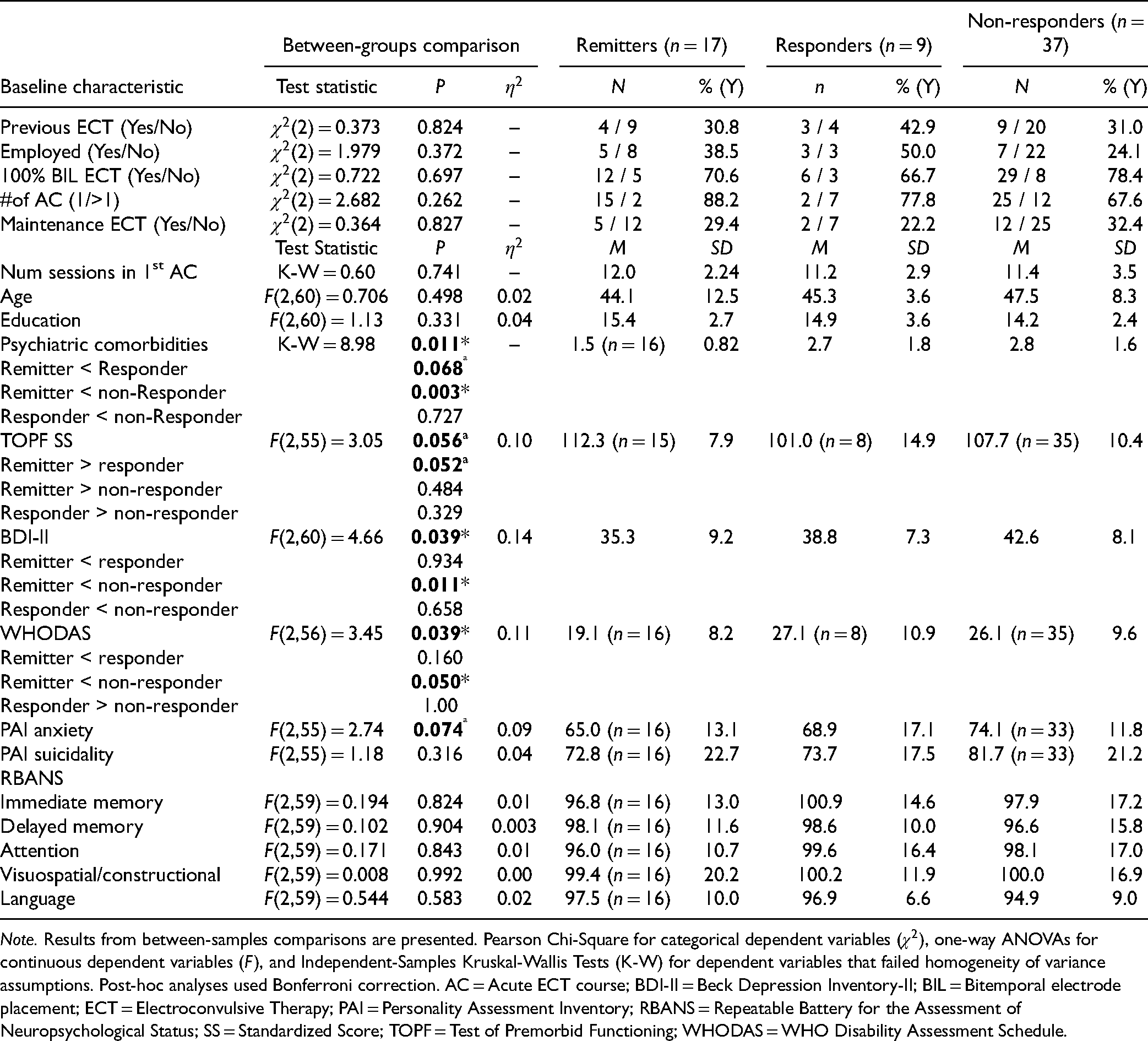
We examined baseline differences between post-ECT response groups using Pearson chi-square (categorical variables), one-way analysis of variance (continuous variables), or Kruskal-Wallis (non-normally distributed or failed the homogeneity of variance assumption) tests. (Table 4). Remitters, Responders (who benefitted from ECT but did not remit), and Non-Responders were treated as separate groups.
Between-groups Comparison of Post-ECT Response Groups.
Note. Results from between-samples comparisons are presented. Pearson Chi-Square for categorical dependent variables (
At baseline, a significant effect of Group was revealed for the number of psychiatric comorbidities (MINI), with Remitters having fewer comorbidities than Non-Responders (P = 0.003) and Responders (trending at P = 0.068). We further examined the response and remission rates in those with and without comorbidities. The necessary data were available for 52 participants. In patients without comorbidities (n = 24), fourteen responded (58.3%), of which eleven remitted (45.8%). In contrast, in patients with at least one comorbidity (n = 38), eleven responded (29.0%), of which only five (13.2%) remitted. Running the analyses including only anxiety and related disorders comorbidities returned the same results.
There was also a significant effect of Group for BDI-II and WHODAS scores. Remitters had lower BDI-II (P = 0.011) and WHODAS (P = 0.050) scores at baseline than Non-Responders. The effect of Group on TOPF SS trended towards significance, with Remitters performing better at baseline than Non-Responders, P = 0.052. No significant effect of Group (P > 0.05) was found for other variables listed in Table 4.
Disability Symptoms
A linear mixed model revealed a significant effect of time on WHODAS scores, F(3,40.72) = 12.30, P < 0.001. Compared to baseline (M = 24.75, SE = 1.05), mean WHODAS scores were significantly lower at post-ECT (M = 18.52, SE = 1.19, P < 0.001), 6-months post (M = 16.98, SE = 1.41, P < 0.001), and 12-months post (M = 18.35, SE = 2.08, P < 0.046).
Predicting Changes in Disability Symptoms
Absolute change from baseline to post-ECT was computed for BDI-II and WHODAS. Linear regression revealed that BDI-II change scores (M = −14.57, SD = 12.64) trended towards significantly predicting WHODAS changes scores (M = −5.93, SD = 10.15), F(1,56) = 3.67, P = 0.061, R2 = 0.061, such that greater reduction in depressive symptoms predicted greater reduction in disability symptoms. After removing one outlier (>3 SD from mean) from the WHODAS change variable, the linear regression analysis was significant, F(1,55) = 4.30, P = 0.043, R2 = 0.073.
Discussion
Although the efficacy of ECT in unipolar depression is well-established in controlled trials 1,2, the effectiveness of ECT with heterogeneous patient characteristics and treatment specifications is less well known. We aimed to characterize the clinical presentation of individuals undergoing ECT in a naturalistic setting and examine the broad effectiveness of ECT regarding treatment responsiveness, depressive symptoms severity, and functional outcomes. Detailed examination of the relationship between clinical response, anesthetic agent, and various ECT stimulus parameters is beyond the scope of this paper and will be described in a separate report.
Clinical Characterization
Overall, the patients referred to ECT represented a heterogeneous group of severely symptomatic and clinically complex individuals. Despite being referred to ECT for depression in the context of unipolar or bipolar depression, psychiatric comorbidities were common (62.9% with
Significant functional impairment was apparent, with high unemployment levels (68.9%) and elevated self-reported disability symptoms. This high unemployment rate is noteworthy considering this was a working-age sample with most having completed post-secondary education and scoring in the high end of average on a psychometric estimate of intellectual ability.
We observed lower levels of cognitive performance on a screening battery (RBANS) compared to what would be expected based on the sample's overall intellectual capacity and educational attainment. This finding is not surprising in a severely depressed sample and suggests that a notable degree of relative cognitive impairment secondary to depression was present before ECT. Specifically, our sample performed significantly below their estimated premorbid intellect on measures of immediate memory, delayed memory, attention, visuospatial construction, and language. Interestingly, baseline immediate and delayed memory performance also distinguished those who completed post-ECT testing from those who did not, with those lost to follow-up demonstrating significantly worse performance than those who returned.
Treatment Effectiveness
Encouragingly, depressive and disability symptoms improved significantly following ECT, and this improvement was maintained up to 12-months post-ECT. Additional improvement in depressive symptoms from mid-ECT to post-ECT was not statistically significant. Improvement in depressive symptoms trended towards significantly predicting reduced disability symptoms from baseline to post-ECT, suggesting that a reduction in depressive symptoms following ECT may transfer to improved daily functioning and reduced disability. Future research should examine whether these gains translate to meaningful functional changes, such as return-to-work.
Our clinical remission rate of 27% (of post-ECT n) and overall response rate of 41.3% (of post-ECT n) were considerably lower than the rates reported in the extant controlled clinical trials4,6 and somewhat lower than those reported in community settings 10 . For example, Prudic and colleagues reported a remission rate of 30.3% to 46.7%, depending on the criteria used, and a response rate of 63.7% in a mixed mood disorder sample across multiple community settings 10 . With only 27% reaching remission, 73% of our post-ECT sample still reported clinically significant levels of depression (on the BDI-II) after treatment.
Our assessment of outcomes is compromised as 36% of our intent-to-treat sample was lost to follow-up (did not complete post-ECT testing); therefore, their level of response to ECT is unknown as symptom measurement immediately post-ECT using the BDI-II was not part of standard care outside of this study. Attrition rates in the broad ECT literature vary considerably 5 or are not adequately reported. In Prudic and colleagues’ community ECT study, only 35 of their 398 subjects did not provide clinical outcome data (i.e., 8.8% lost to follow-up) 10 . The extent to which our higher attrition rates biased reported outcomes is unclear. Encouragingly, at mid-ECT—with 94% of participants retained—we found no significant differences in immediate response to ECT (absolute change in BDI-II scores from baseline to mid-ECT) between those lost to follow-up and those who returned. In the future, our team will attempt to better understand the factors that predict attrition by reviewing clinical notes to assess ECT response in those lost to research follow-up.
Our sample's clinical heterogeneity likely contributed to the lower remission and response rates. As many controlled and naturalistic studies with better outcomes took steps to reduce this heterogeneity (e.g., excluding those with clinical comorbidities), there is considerable converging evidence for this hypothesis. Further, the baseline differences we found between response groups provide preliminary empirical evidence for this hypothesis. Overall, remitters appeared less symptomatic and clinically complex at baseline than non-responders.
Specifically, the high rates of comorbid anxiety disorders and elevated anxiety symptoms observed in our sample likely contributed to the reduced effectiveness of ECT. At baseline, remitters presented with significantly fewer psychiatric comorbidities than non-responders. As anxiety disorders were the most frequent comorbidities, those who remitted were less likely to present to ECT with concurrent anxiety or related disorders (e.g., PTSD). Further, the response and remission rates in patients without these comorbidities were considerably higher (58.3% and 45.8%, respectively of post-ECT n) than those in patients with at least one comorbidity (29% and 13.2%, respectively of post-ECT n). These findings are congruent with previous research. Brus and colleagues 27 have reported reduced remission rates in those with a comorbid anxiety disorder than those without (33.3% vs. 47.5%, respectively). Similarly, high rates of diverse clinical comorbidities, as observed in our sample, may also lead to reduced remission rates. Previous studies exploring diagnostic factors related to remission have found that comorbid substance and alcohol use disorders in unipolar depression 27 , personality disorders in unipolar and bipolar depression27,28, and obsessive-compulsive disorder in bipolar depression 28 are associated with reduced remission to ECT.
We further observed significantly lower baseline depressive and disability symptom severity in remitters than non-responders; thus, it appears that those who remitted following ECT were less clinically severe before starting treatment than those who did not respond. The literature examining the predictive effects of depressive symptom severity on ECT outcomes is inconclusive5,29, with several studies finding greater response with more severe baseline symptoms 30 and others with milder symptoms 31 . We draw note to the fact that only outpatients receiving ECT were followed in this study and, as the most severely ill patients would likely have been hospitalized, the most seriously ill, (e.g., those with psychotic symptoms) were not included in our analyses. Here, we looked at baseline differences between response groups, but further examination into the predictive effects of baseline clinical severity on ECT outcomes is warranted.
There is a paucity of research examining the role of baseline cognition in ECT outcomes. There is mounting evidence that baseline cognitive impairments are associated with reduced treatment response to antidepressant medications (e.g., SSRIs), and psychotherapies (e.g., CBT)11,12,32. Our study found a trend suggesting that estimated premorbid intellectual functioning may differ between response groups, such that remitters present with higher premorbid intellectual functioning than responders; however, this finding should be re-examined in future research where factors such as motivation and cooperation with testing are considered. In addition, we found that patients who returned for post-ECT testing, compared to those lost to follow-up, performed significantly better on baseline measures of immediate and delayed memory. These may represent true differences or may be a function of differences in the subjects’ willingness or ability to fully cooperate with the testing procedures. Immediate change in depressive symptoms (mid-ECT) was comparable for those lost to follow-up and those who completed post-ECT evaluations. Failure to attend scheduled follow-up visits may be partly due to cognitive dysfunction. Thus, baseline cognitive functioning and willingness to cooperate with assessment protocols are necessary constructs to consider when evaluating potential engagement in and response to ECT treatment.
There was also extensive variability in patients’ ECT treatment length, and we made no attempts to control for variations in treatment delivery in our exclusion criteria or data analyses. While we feel this offers a needed perspective in the literature, we expected this treatment heterogeneity to impact our remission rates. Interestingly, we found no significant differences between response groups on previous ECT, electrode placement, number of acute courses, whether maintenance ECT was received, or the number of sessions in the first acute course. Still, it will be important to examine whether specific ECT stimulus parameters (e.g., pulse frequency, pulse width) are related to response in a clinically complex population.
A high proportion of our sample (82%) received bitemporal ECT, which may reduce the generalizability of the findings. The inclusion of unipolar and bipolar depressed patients, while intentional, poses a limitation for interpreting the findings. The use of the BDI-II as our primary outcome measure is relatively unique in the ECT literature; however, a lengthier clinician administered semi-structured interview (e.g., HAM-D) was not feasible in our clinic. Still, nuanced differences in the symptoms assessed across measures should be considered.
Several relevant variables, including medication data, duration of current depressive episode, number of past depressive episodes, and total number of patients seen in the ECT clinic were either not included in the research database or inconsistently reported. This lack of data represents a significant limitation, as these variables may have impacted treatment response to an unknown degree 33 .
Conclusions and Future Directions
Patients presenting for ECT were severely symptomatic and clinically complex. We found that depressive and disability symptoms improved significantly following ECT, with these improvements maintained at 12-months post-ECT. Our remission and overall response rates were considerably lower than the range reported in controlled studies and somewhat lower than those in naturalistic settings, with the caveat that our high attrition rate (36%) may have affected the accuracy of our analysis. Our sample's clinical heterogeneity and severity appeared to have contributed to the lower remission rate observed. Remitters presented to ECT with fewer psychiatric comorbidities, less severe depressive and disability symptoms, and higher levels of estimated premorbid functioning than non-responders.
There remains a paucity of literature examining ECT effectiveness in heterogeneous samples. As ECT is often proposed for seriously ill patients after most other approaches have proven unsuccessful, many seemingly poor candidates may receive ECT out of therapeutic desperation. Nonetheless, examining predictors of treatment response in ECT with clinically complex samples (e.g., psychiatric comorbidities, trauma history, personality disorders) will provide critical insight into who is most likely to benefit and can help guide the informed consent process (e.g., setting expectations).
Future research should examine how baseline cognitive functioning impacts patients’ ability to engage in and respond to ECT–an endeavour currently underway in our laboratory. It would be beneficial to examine how baseline cognition and subjective evaluations of cognitive functioning interact to impact outcomes and whether lower cognitive functioning is associated with reduced treatment adherence.
Footnotes
Data Access
Data for this project was collected at the St. Joseph's Healthcare, Hamilton (SJHH) Hospital under the approval of the SJHH Research Ethics Board and the Hamilton Integrated Research Ethics Board. Due to the clinically sensitive nature of the research and the conditions of our ethics approval, supporting data cannot be made openly available.
Acknowledgments
We thank the patients and their treating clinicians for their participation, without which this study would not be possible. We kindly acknowledge the support received from the staff at SJHH who have contributed to this research project since its inception: Donna Burnett, Catherine Dool, Helen Begin, Michele Crossan. We also acknowledge support from research students who have contributed to data collection/entry for this project: Katherine Jongsma, Joelle LaRoche, Grace Lee, Alexander Manta, Maaida Maqbool, Golda Osei-Bonsu, Vagisha Pruthi, Christina Puccinelli, Serena Sandhu, Muriel Tang.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported, in part, by a St. Joseph’s Healthcare Hamilton Professional Advisory Committee grant to HEM and by Canadian Institutes of Health Research grants to MCM. MCM is supported by the Homewood Chair in Mental Health and Trauma at McMaster University.
