Abstract
Background:
Data demonstrating the real-world, long-term effectiveness of vortioxetine in elderly patients with major depressive disorder (MDD) are clinically useful to confirm findings from randomized trials.
Methods:
RELIEVE was a multinational, 24-week, observational, prospective study in outpatients with MDD initiating vortioxetine treatment in routine care settings (NCT03555136). Here, we report data from a subgroup of 130 patients aged ⩾ 65 years. The primary study outcome was changed from baseline in patient functioning assessed using the Sheehan Disability Scale (SDS). Other clinical outcomes included depression severity (Patient Health Questionnaire-9 [PHQ-9] and Clinical Global Impressions–Severity [CGI-S]), cognitive performance (Digit Symbol Substitution Test [DSST]) and symptoms (Perceived Deficits Questionnaire – Depression-5 item [PDQ-D-5]), and health-related quality of life (HRQoL) (EuroQoL 5 Dimensions 5 Levels [EQ-5D-5L]).
Results:
Clinically meaningful and statistically significant improvements in patient functioning, depressive symptoms, cognitive function, and HRQoL were observed at week 24. Least squares mean SDS, PHQ-9, CGI-S, PDQ-D-5, DSST, and EQ-5D-5L scores improved from baseline by 6.5, 5.7, 1.2, 3.2, 4.4, and 0.11 points, respectively (p < 0.01 for all). Adverse events were observed in 23.1% of patients.
Conclusions:
Consistent with previous clinical studies of vortioxetine, this study supports the effectiveness and safety of vortioxetine in treating elderly patients with MDD in a real-world setting over a 6-month period. Patients showed clinically relevant and sustained improvements in psychosocial functioning, depressive symptoms, and cognitive function after receiving vortioxetine, which was generally well tolerated. Main study limitations include the open-label study design and lack of a placebo or comparator group.
Introduction
Depression is a psychiatric disorder experienced by an estimated 280 million people worldwide (World Health Organization, 2023). Major depressive disorder (MDD) is marked by significant impairments in psychosocial functioning, which can persist despite improvements in disease severity (IsHak et al., 2016). Thus, in addition to alleviating depressive symptoms, improving psychosocial functioning has become a critical treatment goal for patients with MDD. Elderly people are disproportionately affected by MDD, with a global prevalence of 13.3% (Abdoli et al., 2022) compared with a prevalence of approximately 5% in the general population (World Health Organization, 2023). Because of the strong correlation between cognitive and physical impairments and depression (Di Nicola et al., 2023), elderly people are at greater risk of developing depression (Cai et al., 2023). Furthermore, depression, other mental health conditions (e.g., schizophrenia and anxiety disorders), and perceived stress are all predictors of suicide in elderly populations (Obuobi-Donkor et al., 2021). Among the elderly, women are more likely to experience depression than men (Abdoli et al., 2022), but suicide is more common in elderly men than in elderly women, with older age, male sex, and White race representing the most common demographic characteristics for suicide risk (Conwell et al., 2002).
Cognitive dysfunction, along with physical impairments, is likely to represent a crucial factor underlying impaired psychosocial functioning in older populations (Polyakova et al., 2014). Mounting evidence has shown significant clinical differences between depressive disorders that first manifest in adolescence or early adult life (i.e., early-onset depression) compared with an initial presentation in middle age or late life (i.e., late-onset depression) (Schweitzer et al., 2002). Within elderly populations, patients with late-onset depression experience more extensive cognitive impairment than patients with early-onset or a history of depression (Hashem et al., 2017; Schweitzer et al., 2002; Zenebe et al., 2021). Cognitive issues, such as memory and cognitive processing alterations, can cause anxiety in elderly patients and confound depressive symptoms (Zenebe et al., 2021). Additionally, depression can also lead to cognitive deficits and precede neurodegenerative disorders such as all-cause dementia, vascular dementia, and Alzheimer’s disease (Diniz et al., 2013; Schweitzer et al., 2002; Tsuno and Homma, 2009). The prevalence of neurological or psychiatric comorbidities can result in undertreatment of depression in elderly populations due to underdiagnosis and misrecognition of depressive symptoms (Mitchell et al., 2010; Park and Unutzer, 2011).
MDD may be comorbid with various psychiatric, neurological, and somatic conditions, including anxiety (Fava et al., 2000), Alzheimer’s disease and other types of dementia (Asmer et al., 2018), Parkinson’s disease (Marsh, 2013), cardiovascular disease (Van der Kooy et al., 2007), diabetes (Kreider, 2017), and obesity (Blasco et al., 2020; Deischinger et al., 2020), and the frequency of comorbidity between major depressive episodes and other psychiatric and somatic disorders significantly increases with age (Arve et al., 1999). Thus, treatment of patients with MDD and comorbidities may require multiple medications (Kennedy et al., 2009). Drug–drug interactions (DDIs) can reduce the efficacy of an antidepressant or other medications and increase the risk of adverse events (AEs) (Chen et al., 2018; Kennedy et al., 2009; Lackamp et al., 2016). Polypharmacy is common among older adults, and it is important to be mindful of relevant DDIs when selecting an antidepressant treatment (Charlesworth et al., 2015). Antidepressants such as selective serotonin reuptake inhibitors (SSRIs) can interact (typically via cytochrome P450 [CYP] activity modulation) with a number of frequently prescribed medications for common medical conditions (e.g., clopidogrel, antidiabetic agents, and opioid analgesics) (Manolopoulos et al., 2012; Wiese, 2011). Furthermore, SSRIs and serotonin and norepinephrine reuptake inhibitors have not demonstrated substantial efficacy in improving cognitive symptoms or altered psychosocial functioning associated with MDD (Conradi et al., 2011; Hammer-Helmich et al., 2018; Perini et al., 2019), further demonstrating the need for an effective and well-tolerated antidepressant for elderly patients with MDD (Lackamp et al., 2016).
Vortioxetine is an efficacious and well-tolerated multimodal antidepressant that has been authorized for use in the treatment of adults with MDD since 2013 in the United States and European Union (Sanchez et al., 2015). It is marketed in more than 80 countries globally (Zhang et al., 2022). Vortioxetine directly and indirectly influences multiple neurotransmitter systems relevant to the neurobiology of MDD and does not alter CYP enzyme activity, reducing the risk of clinically significant DDIs (Gonda et al., 2019). Vortioxetine has demonstrated efficacy across the spectrum of symptoms experienced by patients with MDD, including depressive, cognitive, and physical symptoms, as well as anxiety and functional impairment (Baldwin et al., 2016; Christensen et al., 2018; Christensen et al., 2023a; Iovieno et al., 2021; McIntyre et al., 2016; McIntyre et al., 2021; Thase et al., 2016). Vortioxetine is one of the few antidepressants with demonstrated dose-response efficacy, with the highest therapeutic benefit being achieved at 20 mg. In a dedicated randomized placebo-controlled study of adults with MDD aged ⩾65 years, vortioxetine demonstrated significant improvements in cognitive function and depressive symptoms over an 8-week period (Katona et al., 2012).
Vortioxetine has also shown effectiveness in older patients with MDD and comorbid early-stage dementia (Christensen et al., 2023b; Cumbo et al., 2022). Improvements in depressive symptoms, cognitive function, daily and global functioning, and health-related quality of life (HRQoL) have been demonstrated in this population. These studies have demonstrated the efficacy and safety of vortioxetine in a variety of patients with MDD, yet there remains a need to determine its sustained effectiveness in the treatment of elderly patients with MDD in routine clinical practice over a longer treatment period.
The RELIEVE study was initiated to complement and extend existing clinical evidence for the use of vortioxetine in the treatment of MDD (Mattingly et al., 2022b). The study assessed the real-world effectiveness of vortioxetine in a heterogeneous patient population and revealed significant and sustained improvements in patient functioning, depressive and cognitive symptoms, and HRQoL across a period of 6 months (Mattingly et al., 2022b). The aim of this subgroup analysis of data from the RELIEVE study was to examine the effectiveness, safety, and tolerability of vortioxetine in a real-life clinical setting in the subgroup of patients aged ⩾ 65 years.
Methods
Study design
RELIEVE was a 24-week, noninterventional, multinational (US, Canada, Italy, and France), prospective study in patients with MDD initiating treatment with vortioxetine (5–20 mg) in routine clinical practice (ClinicalTrials.gov identifier: NCT03555136). Patients were prescribed treatment with vortioxetine at their physician’s discretion according to the locally approved label. The study design and eligibility criteria have been previously described in detail in the RELIEVE primary publication (Mattingly et al., 2022b). This post hoc analysis reports data from the subgroup of elderly participants (defined as those aged ⩾ 65 years). The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines (Mattingly et al., 2022b). Local ethics committee approval for the study was obtained at all participating sites, and all patients provided written informed consent for participation (Mattingly et al., 2022b).
Study assessments and analyses
Elderly participants were assessed during visits at baseline, week 12 (±4 weeks), and week 24 (±4 weeks). Baseline demographics and history of MDD were collected at baseline, while outcomes assessing the effectiveness of vortioxetine were performed at each subsequent study visit. Analyses of outcome measures are limited to those included in the RELIEVE study. Primary effectiveness analysis (i.e., functioning) was assessed using the Sheehan Disability Scale (SDS), a validated tool for assessing functional impairment in patients with MDD (Mattingly et al., 2022b). SDS total scoring ranges from 0 to 30, with higher scores indicating greater impairment in functioning (Sheehan and Sheehan, 2008). An improvement of ⩾4 points in SDS total score is considered meaningful for patients (Sheehan and Sheehan, 2008).
Secondary effectiveness analyses included changes from baseline to weeks 12 and 24 in SDS functioning domain subscores (social life, family life/home responsibilities, and work/school), depression severity, cognitive symptoms, and HRQoL. Depression severity was assessed by patients using the Patient Health Questionnaire-9 (PHQ-9), scored from 0 to 27, with higher scores indicating more severe symptoms (Kroenke et al., 2001), and the Clinical Global Impressions – Severity (CGI-S) scale, scored from 1 to 7, with higher scores indicating greater disease severity (Busner and Targum, 2007). Cognitive performance was assessed using the Digit Symbol Substitution Test (DSST), scored from 0 to 133, with higher scores indicating better performance (Anand et al., 2022). DSST explores a variety of cognitive skills, including attention, processing speed, spatial perception, visual scanning, and executive function, and has been used to evaluate cognitive changes in patients with MDD (Di Nicola et al., 2022; Jaeger, 2018). The severity of self-reported subjective cognitive symptoms over the previous 7 days was evaluated using the Perceived Deficits Questionnaire–Depression-5 item (PDQ-D5), scored from 0 to 20, with higher scores indicating more severe symptoms (Lam et al., 2018). Finally, EuroQoL 5 Dimensions 5 Levels (EQ-5D-5L) was used for evaluating patient HRQoL, scored from 0 to 1, with a higher score indicating better health (Mattingly et al., 2022b). AEs were recorded using the Medical Dictionary for Regulatory Activities version 4.2.1.
Statistics
The primary and secondary analysis population included all enrolled participants who initiated vortioxetine treatment and completed the baseline visit and ⩾1 follow-up assessment visit (full analysis set). Least squares (LS) mean changes in assessment scores from baseline at weeks 12 and 24 were estimated using linear mixed models for repeated measures. LS mean changes were adjusted for clinically relevant baseline variables including age, sex, educational level, duration of current depressive episode, and depression severity (PHQ-9). Except for SDS total score (where an SDS work/school domain score was imputed based on an average of the other two domains for those not at work/school), missing data were not imputed. Safety variables were analyzed over the entire study period for patients who received at least one dose of vortioxetine (safety population). Analyses were performed with the statistical software R®, version 3.6.1.
Results
Patient disposition and baseline characteristics
Of the 737 patients included in the RELIEVE study, 130 were aged ⩾ 65 years, with a mean (SD) participant age of 71.4 (5.3) years in this subgroup (Table 1). The majority of elderly patients were female (68.5%) and White (85.4%). Among the four countries included in the RELIEVE study, Italy had the highest proportion of elderly patients (30.3%, 70/231), comprising 53.8% (70/130) of the total elderly subgroup population. Compared with the overall study population, a higher proportion of the elderly subpopulation had at least one comorbidity, and, separately, a higher proportion had cardiovascular disease. Elderly participants reported an average of 16.7 years since initial MDD diagnosis, with a mean duration of their current major depressive episode at baseline of 62.2 weeks, which were both higher than the overall RELIEVE population (Table 2). Baseline assessment scores were similar in the elderly subgroup compared with the overall RELIEVE population except for the DSST score (cognitive performance). Mean SDS total score at baseline was 18.3, indicating moderate functional deficits. Mean PHQ-9 score at baseline was 15.4, indicating moderately severe depression. The elderly subgroup had a lower mean DSST score than the overall RELIEVE population (30.3 vs. 42.6), which can indicate cognitive decline associated with aging. Vortioxetine was used as first-, second-, or later-line treatment in 41.5%, 40.8%, and 17.7% of elderly patients, respectively. Higher proportions of elderly patients were prescribed anxiolytics, hypnotics, antipsychotics, and anticonvulsants than patients in the overall RELIEVE population. The mean (SD) starting dose for elderly patients was 9.1 mg (9.1), with 43.1% starting on 5 mg and 40.8% starting on 10 mg.
Patient demographic and general clinical characteristics at baseline in the RELIEVE study overall population and subpopulation aged ⩾ 65 years (full analysis set).
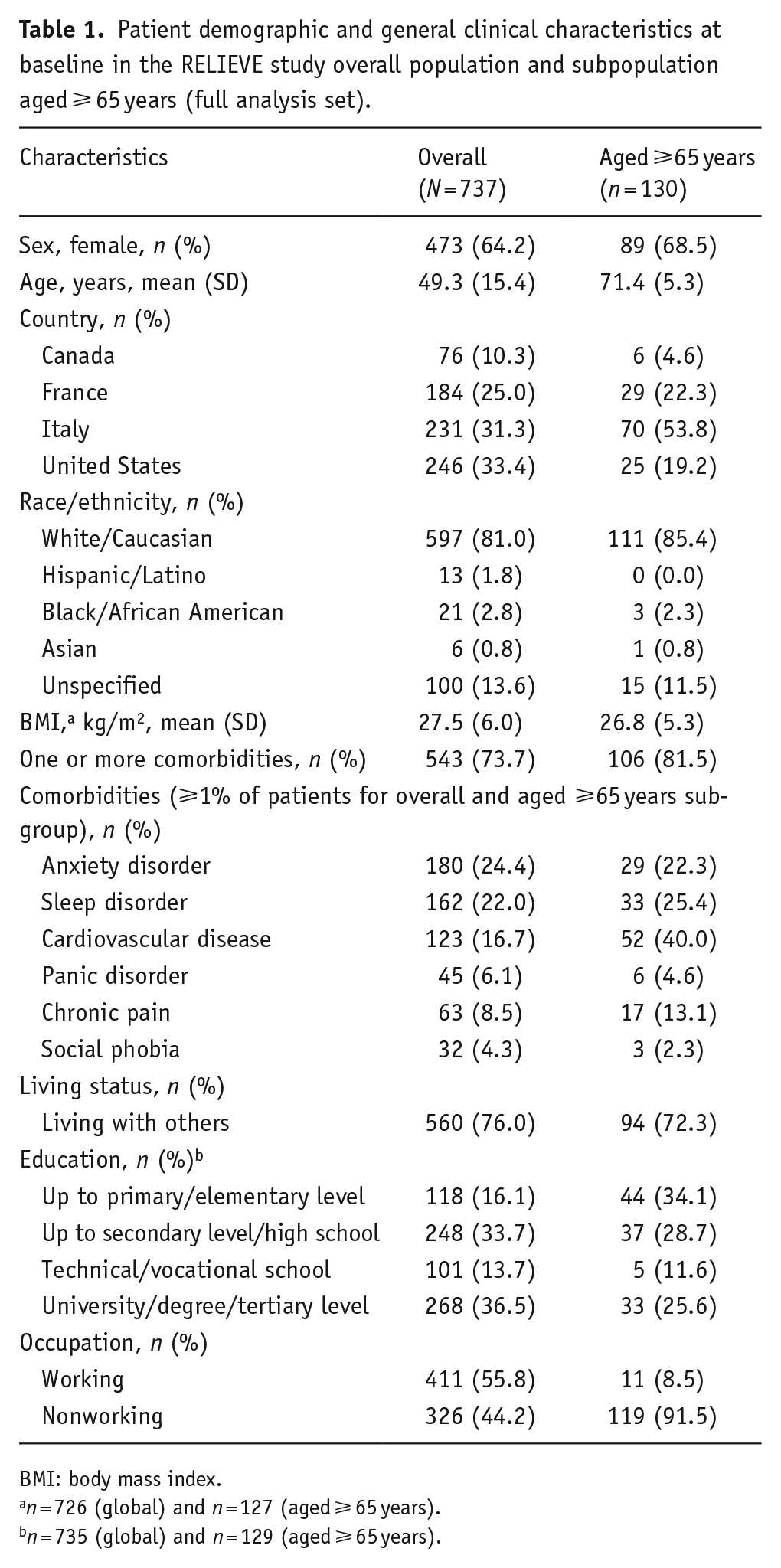
BMI: body mass index.
n = 726 (global) and n = 127 (aged ⩾ 65 years).
n = 735 (global) and n = 129 (aged ⩾ 65 years).
Patient MDD-related clinical characteristics at baseline in the RELIEVE study overall population and subpopulation aged ⩾ 65 years (full analysis set).
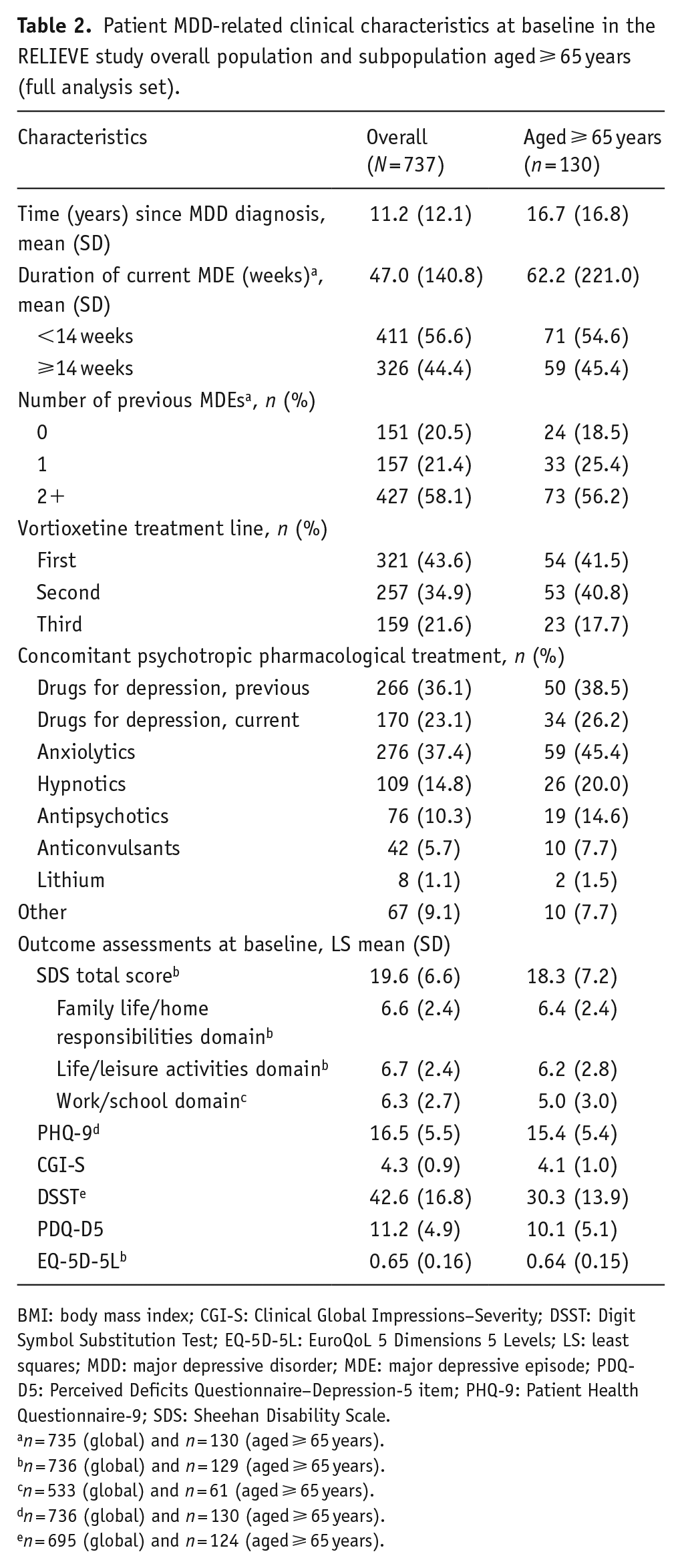
BMI: body mass index; CGI-S: Clinical Global Impressions–Severity; DSST: Digit Symbol Substitution Test; EQ-5D-5L: EuroQoL 5 Dimensions 5 Levels; LS: least squares; MDD: major depressive disorder; MDE: major depressive episode; PDQ-D5: Perceived Deficits Questionnaire–Depression-5 item; PHQ-9: Patient Health Questionnaire-9; SDS: Sheehan Disability Scale.
n = 735 (global) and n = 130 (aged ⩾ 65 years).
n = 736 (global) and n = 129 (aged ⩾ 65 years).
n = 533 (global) and n = 61 (aged ⩾ 65 years).
n = 736 (global) and n = 130 (aged ⩾ 65 years).
n = 695 (global) and n = 124 (aged ⩾ 65 years).
Effectiveness
After initiating vortioxetine treatment, patients showed clinically meaningful functional improvement (decrease of ⩾4 points in SDS total score), with LS mean SDS total score significantly decreased by 4.7 points from baseline at week 12 and 6.5 points at week 24 (p < 0.01 for both time points) (Figure 1). Statistically significant reductions from baseline were also observed for all three SDS subdomain scores (family life/home responsibilities, social life, and work/school) for both time points (p < 0.01 for all changes at weeks 12 and 24). Moreover, the proportion of patients with severe functional impairment (SDS total score > 20) decreased from 45.7% at baseline to 21.4% and 13.5% at weeks 12 and 24, respectively (Figure 2). This reduction occurred concomitantly with an increase of patients with mild or minimal functional impairment; after 24 weeks, more than 50% of patients reported mild or minimal functional impairment compared with only 19.4% at baseline.
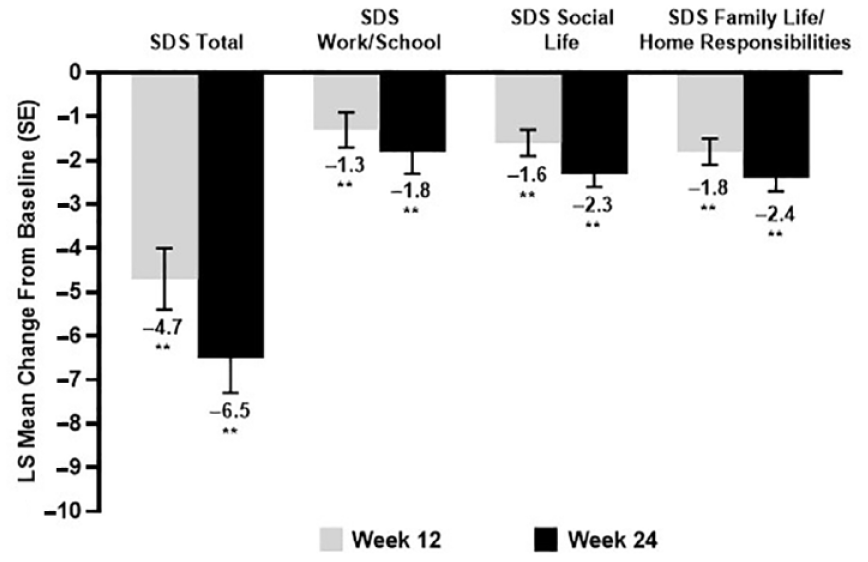
Change in adjusted LS mean scores from baseline to weeks 12 and 24 for SDS total score and SDS subscores.
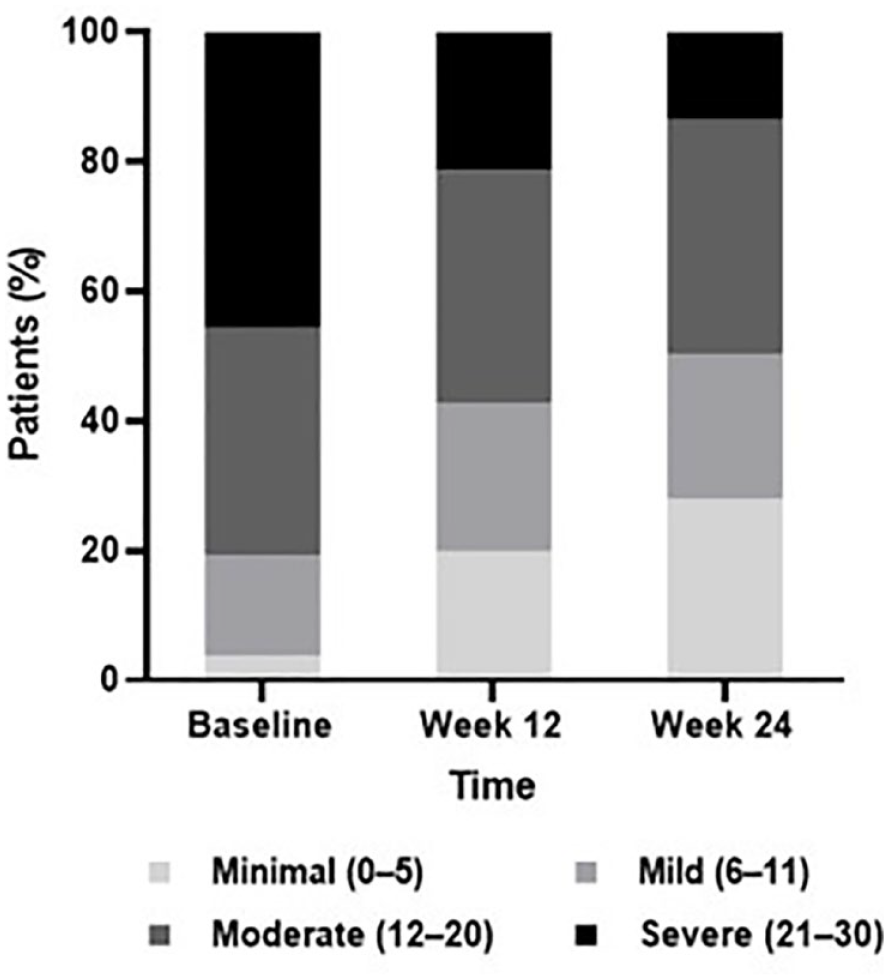
Proportion of patients according to SDS total score functional impairment category at baseline and weeks 12 and 24. Data represented as proportion of patients in each SDS total score category (Minimal, Mild, Moderate, and Severe) at baseline and after 12 and 24 weeks of vortioxetine treatment (full analysis set). N = 129 at baseline, N = 126 at week 12, and N =111 at week 24. Note not all patients were followed up at both time points; a subset of patients did not report SDS scores at both time points.
Improvements were seen across cognitive function parameters. LS mean PDQ-5 scores were reduced over the treatment period (Figure 3(a); p < 0.05 for both time points), demonstrating improvement in cognitive function (Lam et al., 2018), and LS mean DSST score increased by >4 points over the 24-week treatment period (Figure 3(b); p = 0.02 and p < 0.01 at weeks 12 and 24, respectively), indicating significant improvement. Significant improvements in depression severity, HRQoL, and cognitive function were also seen at weeks 12 and 24. PHQ-9 scores were significantly reduced, indicating, on average, a transition from moderately severe to mild depression severity (Figure 4(a); p < 0.01 for both time points) (Yoon et al., 2014). Furthermore, patients had reduced LS mean CGI-S scores at weeks 12 and 24 (Figure 4(b); p < 0.01 for both time points), indicating, on average, an overall improvement from moderately ill to mildly ill (Busner and Targum, 2007). Improvements in HRQoL were observed over the treatment period, as EQ-5D-5L utility index scores increased over 24 weeks of vortioxetine treatment (Figure 4(c); p < 0.01 for both time points).
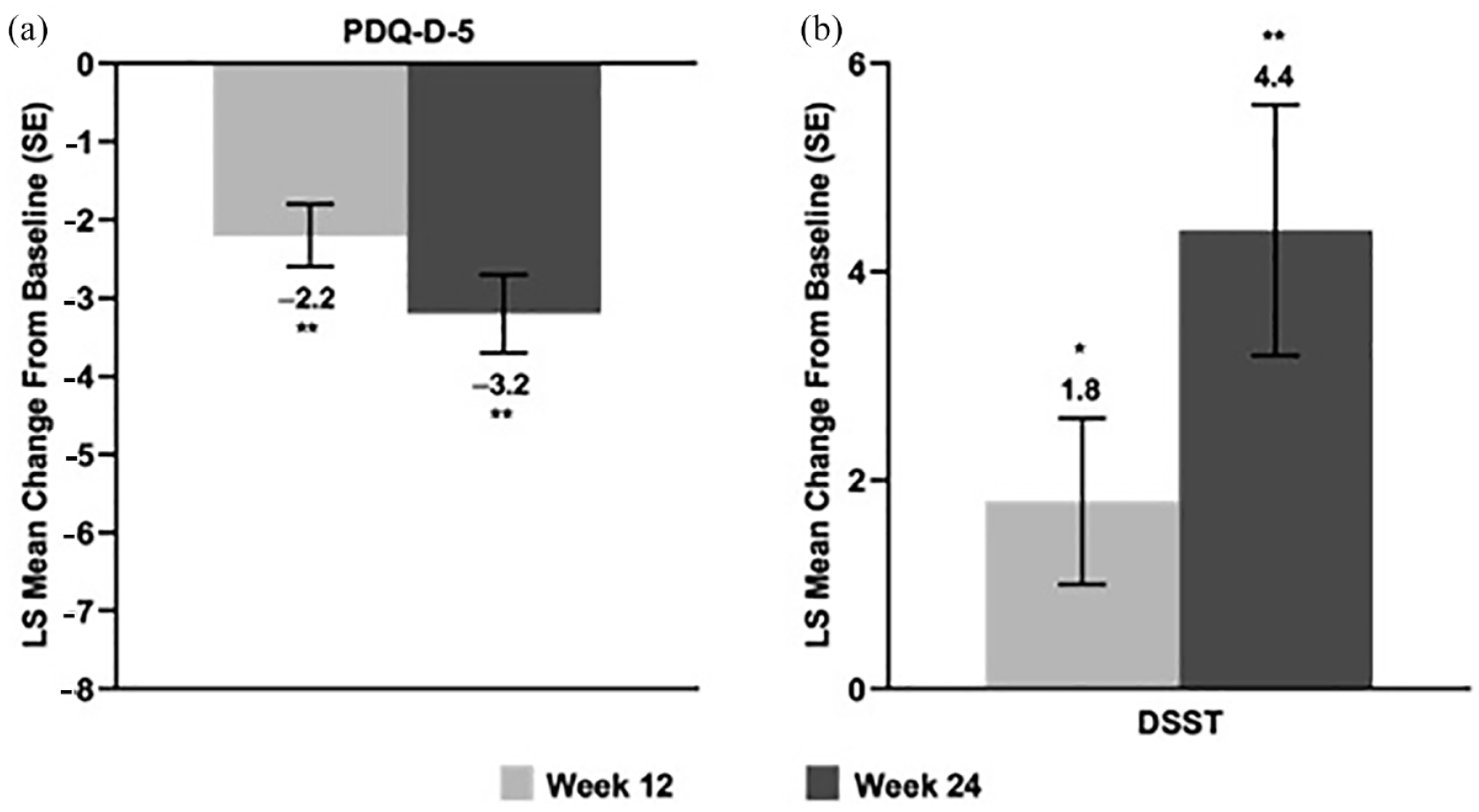
Cognition: Adjusted LS mean change (SE) from baseline in (a) cognitive symptoms (PDQ-D-5) and (b) cognitive performance (DSST) at weeks 12 and 24. PDQ-D-5 and DSST LS mean baseline scores (SD) were: PDQ-D-5, 11.24 (4.9); DSST, 42.6 (16.8). PDQ-D-5 score range 0–20; DSST score range 0–133.
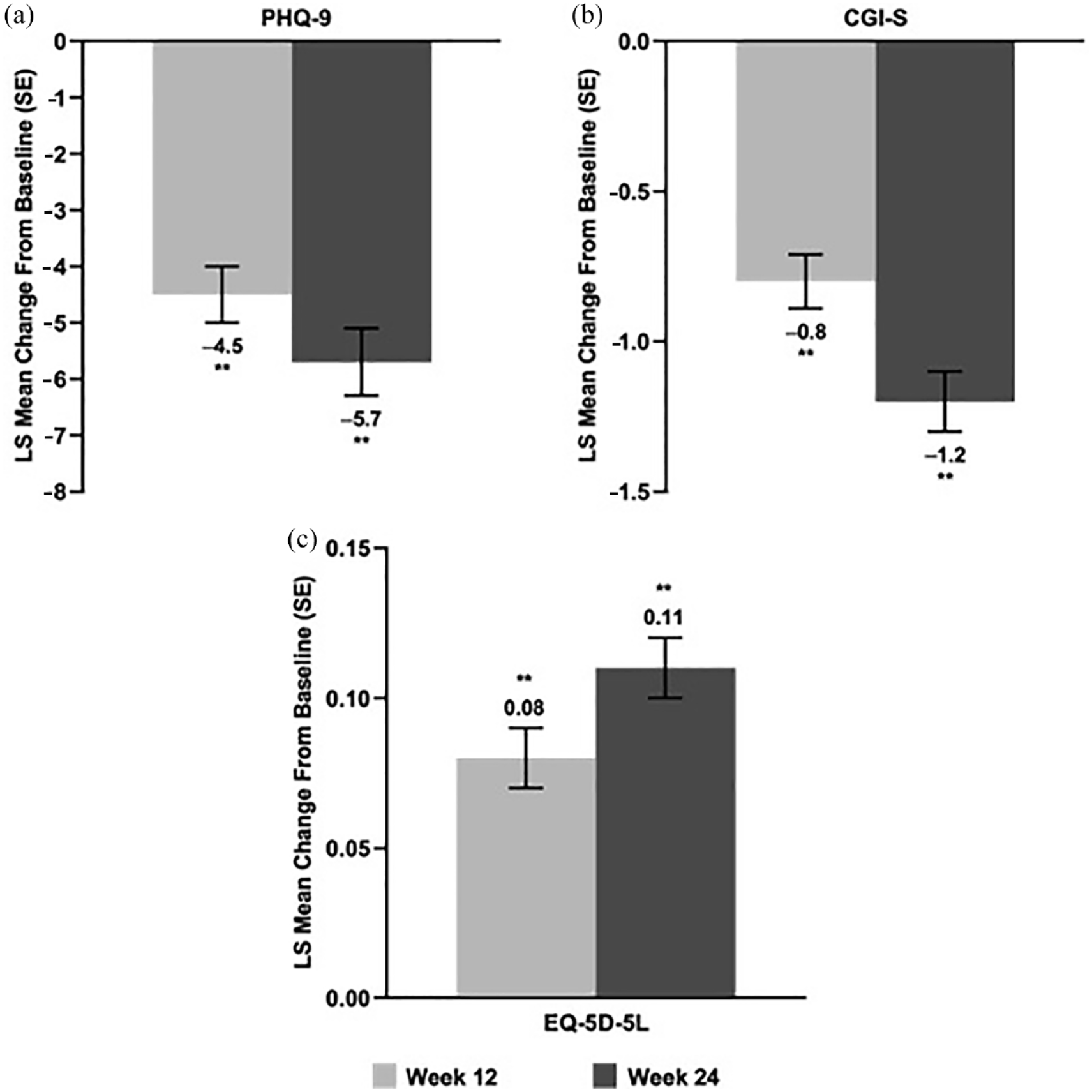
Disease severity and quality of life: Adjusted LS mean change (SE) from baseline to weeks 12 and 24 in (b) PHQ-9, (b) CGI-S, and (c) EQ-5D-EL. PHQ-9, CGI-S, and EQ-5D-5L LS mean baseline scores (SD) were: PHQ-9, 16.5 (5.5); CGI-S, 4.3 (0.9); EQ-5D-5L, 0.7 (0.2). PHQ-9 score range 0–27; CGI-S score range 1–7; EQ-5D-5L score range 0–1.
Safety and tolerability
At least, one AE was observed in 23.1% of elderly patients compared with 21.2% of all patients in the RELIEVE study. A total of 3.8% of patients reported a serious AE, similar to the incidence observed in the overall population (3.9%). Most AEs occurring in ⩾2 patients were nausea (n = 8, 6.2%), pruritus (n = 3, 2.3%), anxiety (n = 2, 1.5%), acute cholecystitis (n = 2, 1.5%), dizziness (n = 2, 1.5%), headache, (n = 2, 1.5%), and prescribed overdose (n = 2, 1.5%). Nine patients (6.9%) discontinued vortioxetine at week 12, and 6 more (4.6%) discontinued treatment at week 24. The most common reasons overall for discontinuation were lack of tolerability (n = 4) and lack of effectiveness (n = 6). No new safety signals specific to elderly patients were observed.
Discussion
Older adults are disproportionately affected by depression as well as other psychiatric and medical comorbidities, and they often experience more significant depression-related functional and cognitive impairments compared with younger patients (Cai et al., 2023; Schweitzer et al., 2002; World Health Organization, 2023; Zenebe et al., 2021). While previous studies have demonstrated the efficacy of vortioxetine in the elderly population with MDD in randomized controlled trials (RCTs) (Katona et al., 2012; Nomikos et al., 2017), it is clinically relevant to understand the real-world effectiveness of vortioxetine to help guide the long-term treatment of MDD in elderly patients. This study provides clinically meaningful, real-world evidence supporting improvements in patient functioning among elderly patients with MDD treated with vortioxetine, as well as improved depression severity, cognitive functioning (both cognitive performance and cognitive symptoms), and HRQoL sustained over a 6-month treatment period.
Depression has been shown to significantly affect psychosocial functioning in elderly populations (Cai et al., 2023). Additionally, depression-related deficits in functioning can persist despite amelioration of depressive symptoms with current treatments. We observed clinically relevant improvement from baseline in overall patient functioning in this group of elderly patients with MDD treated with vortioxetine, consistent with previous observations in the general MDD patient populations from RCTs and real-world studies (De Filippis et al., 2022; Di Nicola et al., 2022; Florea et al., 2017; Mattingly et al., 2022a; Mattingly et al., 2022b; Polosan et al., 2022; Wang et al., 2022). We also found clinically meaningful functional improvements from baseline in social situations, with family/home responsibilities and at work that were sustained at week 24, along with improvement in depression severity.
Importantly, cognitive dysfunction has been shown to partially underlie the impairments in psychosocial functioning observed in patients with MDD. Therefore, understanding and treating cognitive symptoms in patients with MDD are of particular importance, especially for elderly patients, as cognitive functional deficits are a common symptom of depression (Cai et al., 2023; Fiske et al., 2009), and depression can be a prodromal symptom or risk factor for dementia-related diseases such as Alzheimer’s disease (Diniz et al., 2013; Schweitzer et al., 2002; Tsuno and Homma, 2009). The findings of improved cognitive symptoms and function from this analysis are consistent with previous studies of vortioxetine in elderly patients. A previous RCT in elderly patients with MDD treated with vortioxetine (5 mg) found a significant improvement in cognitive performance (DSST) scores, which was not observed in the active comparator (duloxetine) group (Katona et al., 2012). In addition, a 12-week study evaluated the effectiveness of vortioxetine in patients with MDD and comorbid early-stage dementia (Christensen et al., 2023b). All patients initiated vortioxetine treatment at 5 mg, with forced up-titration to 10 mg at day 8 followed by flexible dosing within the approved range of 5–20 mg/day. Patients with MDD and early-onset dementia (Mini-Mental State Examination-2 total score of 20–24) reported a significant improvement in depressive symptom severity from week 1 onward. Cognitive performance as assessed by DSST and Rey Auditory Verbal Learning Test significantly improved, with significant improvements in functioning and HRQoL also observed (Christensen et al., 2023b). More than 50% of the patients were treated with vortioxetine 20 mg by the end of the study, providing supporting evidence that the highest dose yields the most pronounced therapeutic effects, if tolerated, in elderly patients with MDD. Finally, an analysis of real-world evidence in South Korea assessed the effectiveness and safety of vortioxetine in a cohort of patients with MDD and comorbid Alzheimer’s disease (Cumbo et al., 2022). Patients receiving vortioxetine showed an overall clinical improvement, including significantly improved depressive and cognitive symptoms (Cumbo et al., 2022).
The current study showed statistically significant improvement by week 12 compared to baseline in cognitive performance (DSST) and cognitive symptoms (PDQ-D-5) in a real-world, multinational elderly population. Further, these improvements in cognitive function were sustained over 6 months, as DSST and PDQ-D-5 scores continued to improve over the course of treatment. Taken together, these data provide supporting evidence for the effectiveness of vortioxetine in elderly patients with MDD, including those with dementia.
The improvements from baseline for depressive symptoms and HRQoL presented here are consistent with previously reported studies. In a dedicated RCT in elderly patients with MDD, patients received 5 mg/day of vortioxetine for 8 weeks and showed significantly improved 12-item Health Status Questionnaire scores for the domains of health perception, mental health, and energy relative to placebo (Florea et al., 2015; Katona et al., 2012). In addition, RCT data have effectively demonstrated clinical improvement in depression symptoms in a variety of patients with MDD who were treated with vortioxetine as assessed by the PHQ-9 and CGI-S scales (Almeida et al., 2023; Alvarez et al., 2012; De Filippis et al., 2022), which have been supported by results from the real-world RELIEVE study (Mattingly et al., 2022b). Consistent with these previous studies, significant improvements in depression severity were seen at weeks 12 and 24 in elderly patients. These results support the clinical effectiveness of vortioxetine in reducing symptoms of depression in potentially difficult-to-treat and complicated patient subgroups.
Additionally, vortioxetine was well tolerated in this cohort of elderly patients, and the safety results were consistent with the known tolerability profile of vortioxetine. Safety results reported here were similar to those reported in the recent MEMORY study (mean age of 70.3 years) in patients with comorbid and early-stage dementia, as well as those reported in a recent study of patients with MDD and comorbid Alzheimer’s disease receiving vortioxetine in routine care settings in South Korea (mean age of 77.7 years), with nausea and abdominal pain being the most commonly reported AEs (Christensen et al., 2023b; Cumbo et al., 2022). Moreover, the tolerability results presented here are particularly important for this patient subgroup because elderly patients tend to have more comorbidities and be at a greater risk for polypharmacy-related AEs.
Strengths of this study are its long-term follow-up of elderly patients and significant collection of important patient health data, such as detailed individual medical histories and both physician- and patient-reported information on the current depressive episode, including functioning and HRQoL data. The elderly patient subgroup did display some differences in the baseline characteristics compared with the overall RELIEVE population including increased clinical severity, comorbidities, and general treatment resistance. Compared with the overall RELIEVE population, the elderly subpopulation showed differences in clinical severity, with more years since initial MDD diagnosis, with generally longer duration of current major depressive episode at baseline. Notably, a longer duration of illness is associated with higher levels of treatment resistance in patients with MDD (Altamura et al., 2015). The elderly patient subgroup also presented with higher incidences of cardiovascular disease and chronic pain, and literature has also demonstrated that increasing age may confer a lesser and slower response to antidepressant treatment (Strawn et al., 2023). Improvements in assessments from baseline in the elderly cohort appeared reduced compared to those reported in the overall RELIEVE cohort, that is, all patients aged ⩾ 18 years (Mattingly et al., 2022b).
The primary limitations of the study were those that are inherent in all real-world studies, including its open-label study design, lack of a placebo or comparator group, and lack of intensive patient monitoring. Moreover, MDD was not diagnosed by standardized criteria, as patients were diagnosed based on local criteria and physician judgments. Additionally, while the current analysis focused on a specific subgroup of patients, it is part of the larger global RELIEVE study. The RELIEVE study recruited patients in four countries, which may limit generalizability to broader elderly populations.
Conclusions
This study provides supporting evidence for the effectiveness and safety of vortioxetine in treating elderly patients with MDD in a real-world setting over a 6-month period, which is consistent with the efficacy and safety profile observed in other clinical studies of vortioxetine. Elderly patients with MDD showed improvements from baseline in patient functioning, depressive symptoms, cognitive performance and cognitive symptoms, and HRQoL. Vortioxetine was generally well tolerated in elderly patients with MDD.
Footnotes
Acknowledgements
Under direction of the authors, medical writing assistance was provided by Robert Nichol, PhD, and Leandra Dang, PharmD, on behalf of Syneos Health Medical Communications, LLC. H. Lundbeck A/S provided funding to Syneos Health for support in writing this manuscript.
Data availability statement
The data supporting the findings of this study are included in the article; further inquiries can be directed to the corresponding author.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MDN is/has been a consultant and/or a speaker for and/or has received research grants from Angelini, Idorsia Pharmaceuticals, Janssen, Lundbeck, Neuraxpharm, and Otsuka. AR and MCC are employees of H. Lundbeck A/S, and MA was an employee at time of study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by H. Lundbeck A/S.
