Abstract
Background:
The optimal lumateperone dose for bipolar depression remains uncertain.
Aims:
To examine its dose–response relationship for efficacy and safety.
Methods:
We systematically searched major databases to 1 July 2025. Efficacy outcomes included change in depression severity, global illness severity, quality of life, responder, and remitter rates. Safety outcomes included all-cause dropout, discontinuations due to adverse event (AE), treatment-emergent AE, mania, suicidality, extrapyramidal symptoms, body weight, lipid profile, and fasting glucose. A one-step dose–response meta-analysis generated effect sizes, reported as standardized mean differences (SMDs) and risk ratios (RRs) with 95% confidence intervals (CIs).
Results:
Three randomized controlled trials involving 1454 patients showed that a 42-mg daily dose of lumateperone significantly improved depressive symptoms (SMD = −0.26; 95% CI: −0.51, −0.02), Clinical Global Impression-Bipolar-Severity (CGI-BP-S) overall bipolar illness (SMD = −0.31; 95% CI: −0.45, −0.16), CGI-BP-S bipolar depression (SMD = −0.33; 95% CI: −0.48, −0.17), quality of life (SMD = 0.22; 95% CI: 0.07, 0.36), and responder rate (RR = 1.27; 95% CI: 1.05, 1.53), but not remitter rate (1.06; 95% CI: 0.81, 1.38). Compared with placebo, discontinuation due to AE significantly increased at the 42 mg dose (RR = 3.12; 95% CI: 1.68, 5.80), but not at 28 mg (1.58; 95% CI: 0.25, 9.89). Moreover, dropout rates (42 mg RR = 1.15; 95% CI: 0.76, 1.73) and other safety outcomes did not exhibit a dose–response trend.
Conclusions:
Preliminary evidence suggests that 42 mg daily of lumateperone may provide clinical benefit in bipolar depression, yet the higher rate of AE-related discontinuation warrants caution in practice. However, current data remain limited, requiring further studies to establish the optimal dosing range balancing efficacy and safety.
Introduction
Bipolar disorder is defined by the presence of a manic episode (bipolar I disorder) or a hypomanic episode (bipolar II disorder), with or without a depressive episode (Voelker, 2024). The age of onset is usually between 15 and 25 years with depression being the most common first symptom, and approximately 75% of overall symptom time is comprised depressive episodes or symptoms (Nierenberg et al., 2023). During the depressive phase, patients often experience diminished interest in activities, persistent fatigue, and occasional suicidal ideation, which can substantially erode daily functioning and social engagement (McIntyre et al., 2020). A global record indicates that bipolar disorder has a lifetime prevalence of nearly 2.4% (Carvalho et al., 2020) and ranks as the 28th leading cause of years lived with disability worldwide (Ferrari et al., 2022). These underscore the importance to effectively treat the depressive symptoms of bipolar disorder.
Despite this need, pharmacotherapeutic options for bipolar depression remain relatively limited (Keramatian et al., 2023). For example, a guideline by the Canadian Network for Mood and Anxiety Treatments and the International Society for Bipolar Disorders lists only quetiapine and lurasidone as first-line antipsychotics for bipolar I depression, whereas quetiapine alone is recommended first-line for bipolar II depression (Yatham et al., 2018). Before 2021, there were only four pharmacological treatments (cariprazine, lurasidone, olanzapine–fluoxetine combination, and quetiapine) approved by the US Food and Drug Administration (US FDA) to treat acute bipolar depression (Husain and Yatham, 2023). This situation highlights the urgent need for developing expanded options to treat bipolar depression. Lumateperone is a newer antipsychotic that has attracted attention in recent years due to the Calabrese et al. (2021) study, which reported its effectiveness in reducing depressive symptoms compared with placebo. In 2021, lumateperone became the fifth drug approved by the US FDA to treat bipolar I depression (Husain and Yatham, 2023). A recent meta-analysis found that lumateperone significantly improved depressive symptoms in bipolar disorder, but it only examined the 42 mg dose, while omitting possible benefits from lower doses such as 28 mg (Peng et al., 2024). Therefore, it is unclear whether lower doses may provide clinically meaningful effects. Additionally, the meta-analysis did not assess for treatment efficacy at different time points during the 6-week treatment course. Lastly, the US FDA has also approved lumateperone for the treatment of bipolar II depression (Husain and Yatham, 2023); however, some data (e.g., the study by Suppes et al. (2023)) suggested limited efficacy in the bipolar II population. Hence, understanding the effects of lumateperone in treating bipolar I and II disorders remain a critical issue.
One effective way to clarify these uncertainties is a dose–response meta-analysis, which would examine how different doses affect clinical outcomes to guide decisions about the most appropriate course of treatment (Lin et al., 2023; Yu et al., 2024). For example, a dose–response meta-analysis conducted on lurasidone in bipolar depression concluded that doses between 40 and 60 mg yielded better results than higher doses, striking a balance between efficacy and safety (Lin et al., 2024). Based on these considerations, this study aimed to systematically review randomized controlled trials (RCTs) of lumateperone and utilized a dose–response meta-analysis approach. Specifically, we concentrated on the therapeutic effectiveness of lumateperone for alleviating depressive symptoms and related outcomes. In addition, we evaluated the medication’s safety profile by examining dropout rates, adverse events (AEs), and metabolic effects, thereby providing a clearer perspective on lumateperone’s overall risk-benefit balance for clinical practice.
Method
Search strategy and study selection
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (Appendix) for this study (Page et al., 2021). The study protocol was registered with the International Platform of Registered Systematic Review and Meta-Analysis Protocols (INPLASY202510048).
We performed a thorough search of PubMed, Embase, Cochrane CENTRAL, and ClinicalTrials.gov from each database’s inception through 1 July 2025. We applied the search terms (lumateperone OR ITI-007 OR ITI-722) AND (depress* OR bipolar OR affective OR mood) without language restriction. We also manually searched the references in the literature. The search method was in line with the Population, Intervention, Comparator, Outcomes, Study Design (PICOS) framework: (1) Population: individuals with bipolar disorder currently experiencing a major depressive episode; (2) Intervention: lumateperone; (3) Comparator: placebo; (4) Outcomes: changes in depression severity and dropout rates as primary interests; and (5) Study Design: RCT.
Our goal was to locate RCTs evaluating the efficacy and safety of lumateperone for bipolar depression. The inclusion criteria were (1) RCTs that compared placebo with lumateperone (administered alone or in combination). We assumed placebo to be a zero dose of lumateperone; therefore, studies comparing lumateperone with other active agents would not yield dose–response data. (2) Participants had a bipolar depression diagnosis established by standard criteria (e.g., the Diagnostic and Statistical Manual of Mental Disorders). (3) RCTs reporting pre- and posttreatment depression severity using a validated scale (e.g., the Montgomery–Asperger Depression Rating Scale (MADRS)) to evaluate lumateperone. We excluded studies if (1) they compared lumateperone only with other active treatments, omitting a placebo arm; (2) they enrolled patients with diagnoses other than bipolar disorder (e.g., schizophrenia); (3) they did not measure depressive symptoms as an outcome; or (4) they duplicated data from the same research protocol. In cases of multiple articles originating from the same dataset, only the one with the largest sample size and most comprehensive information was included.
Two reviewers (CWH and YCC) independently screened the titles and abstracts to identify studies that merited further review. Each then independently examined the full-text articles. Any disagreement at this stage was resolved by a third reviewer (PTT).
Data extraction and quality assessment
Two reviewers (CWH and YCC) separately extracted data from each eligible article. Extracted data included publication details, study design, participant characteristics (diagnostic criteria, age, sex, sample size), treatment approach (dosing schedule and duration), symptom scales, and geographic location. We focused on efficacy and safety outcomes. For efficacy, the primary variable of interest was the change in depression severity, assessed using the MADRS. Secondary efficacy outcomes included (1) global illness severity, such as the change in Clinical Global Impression-Bipolar-Severity (CGI-BP-S) overall bipolar illness and depression scores; (2) quality of life, such as scores on the Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form (Q-LES-Q-SF); (3) responder rate (MADRS total score ⩾ 50% decrease from baseline); and (4) remitter rate (MADRS total score ⩽ 12). The primary safety metric was the all-cause dropout rate, calculated by dividing the number of participants who discontinued the trial (for any reason) by the total randomized sample. Secondary safety outcomes included (1) discontinuations due to AEs, (2) any treatment-emergent adverse events (TEAEs), (3) mania or hypomania AEs, (4) suicidal ideation or behavior AEs, and (5) extrapyramidal symptom (EPS). These events were defined as any AEs reported over the course of the study. In addition, we were interested in metabolic effects—including body weight and laboratory values for total cholesterol, high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, triglycerides, and fasting glucose—which were treated as supplementary safety outcomes. If the information was not found in the published article, we contacted the corresponding author for clarification.
Risk of bias in each included trial was assessed by two reviewers (YCC and CWH) using the Cochrane Handbook methodology (Risk of Bias 2; Higgins et al., 2024). Any disagreements were addressed through discussion with a third reviewer (PTT).
Data synthesis and statistical analysis
All statistical analyses were performed using the dosresmeta package (version 2.0.1; Crippa, Discacciati and Bottai, 2019) in R software (version 4.3.2; (R Core Team, R Foundation for Statistical Computing, Vienna, Austria). For continuous variables, we computed pre- to posttreatment changes and expressed them as standardized mean differences (SMDs) with 95% confidence intervals (CIs). For categorical variables, we calculated event counts and converted these to risk ratios (RRs) with 95% CIs. We explored whether lumateperone, compared with placebo, exhibited a dose–dependent relationship for both primary and secondary outcomes by using a one-stage random-effects dose–response meta-analysis (Orsini et al., 2006). Dose–response curves were modeled with restricted cubic splines (three knots) at fixed percentiles (10%, 50%, and 90%; Desquilbet and Mariotti, 2010); a simple linear model was also fitted for comparison. Model comparisons were evaluated using the Akaike information criterion and the Bayesian information criterion. Goodness-of-fit statistics were calculated, and the obtained coefficient of determination (R-squared) indicated the proportion of effect-size variability that could be explained by dose.
We conducted four additional assessments with focus on the primary efficacy outcome. First, we performed a leave-one-out analysis to evaluate the impact each individual study had toward the overall findings. Second, following a post-hoc systematic review, we identified a RCT that included both bipolar and unipolar depression (ClinicalTrials.gov, 2020). We incorporated its data into our dose–response meta-analysis and recalculated the dose–response relationship. Third, we investigated temporal trends of lumateperone versus placebo across varying doses using methods analogous to the basic analysis described above. Fourth, we were interested in whether dose and efficacy patterns were similar for bipolar I and II disorders, so we conducted a subgroup analysis stratified by bipolar subtype. To gauge heterogeneity in the one-stage dose–response meta-analysis, we employed a variance partition coefficient (an extension of the I-squared statistic) (Crippa et al., 2019). We assessed the certainty of the evidence for each primary outcome using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework. GRADE categorizes the overall certainty of a body of evidence as high, moderate, low, or very low, allowing ratings to be downgraded or upgraded according to prespecified criteria (Shao et al., 2023). All tests were two-sided, and p-values < 0.05 were considered statistically significant.
Results
The literature search process is shown in the flowchart (eFigure 1), and the complete set of search strategies is shown in eTable 1. Three hundred and six articles underwent initial screening, which involved removing duplicates and excluding studies based on title and abstract review, and 20 articles remained. Further full-text examination (see eTable 2 for exclusion details) yielded three eligible articles (Calabrese et al., 2021; ClinicalTrials.gov, 2015; Suppes et al., 2023). These three RCTs encompassed 1454 total participants, of whom 905 received lumateperone and 549 received placebo (Table 1). Across all studies, 804 (55%) participants were male, and mean ages ranged from 41 to 46 years. All included studies administered a 6-week treatment course with daily doses ranging from 28 to 42 mg. For the primary outcomes, each trial utilized the MADRS to measure depression severity and recorded the number of dropouts. In addition, we identified a RCT involving participants with both bipolar and unipolar depression (ClinicalTrials.gov, 2020), the details of which are also shown in Table 1.
Characteristics of included studies.
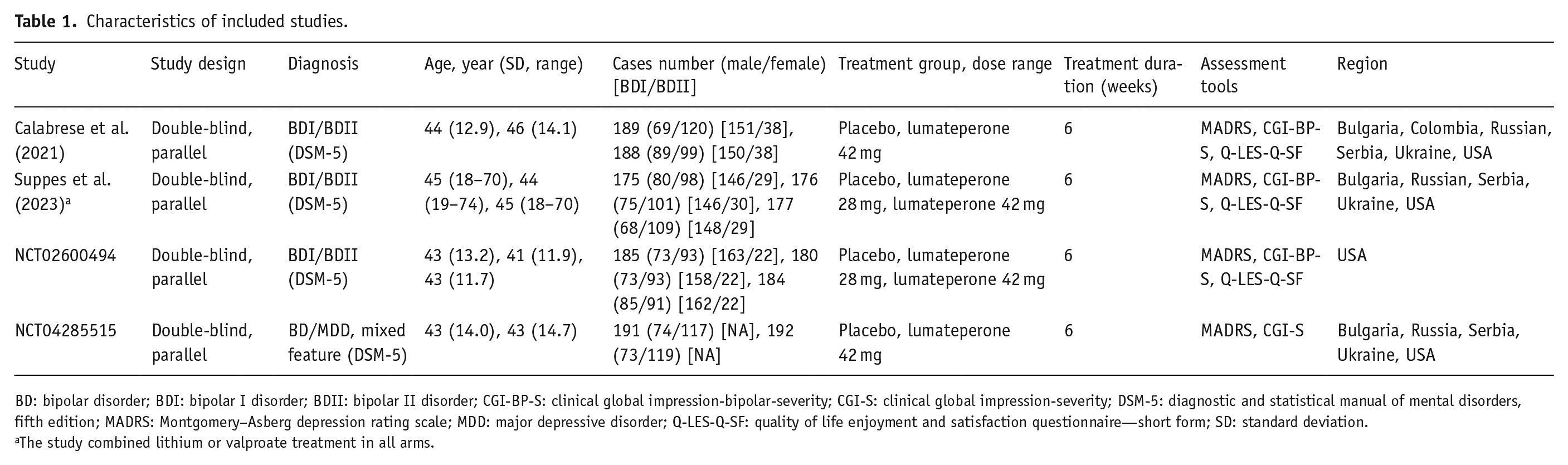
BD: bipolar disorder; BDI: bipolar I disorder; BDII: bipolar II disorder; CGI-BP-S: clinical global impression-bipolar-severity; CGI-S: clinical global impression-severity; DSM-5: diagnostic and statistical manual of mental disorders, fifth edition; MADRS: Montgomery–Asberg depression rating scale; MDD: major depressive disorder; Q-LES-Q-SF: quality of life enjoyment and satisfaction questionnaire—short form; SD: standard deviation.
The study combined lithium or valproate treatment in all arms.
Regarding the four continuous measures of efficacy, the therapeutic effect of lumateperone over placebo increased with higher doses (Table 2). Specifically, the 42 mg dose of lumateperone significantly improved depression severity (MADRS: SMD = −0.26; 95% CI: −0.51 to −0.02; Figure 1), global illness severity (CGI-BP-S overall bipolar illness: SMD = −0.31; 95% CI: −0.45 to −0.16; CGI-BP-S depression: SMD = −0.33; 95% CI: −0.48 to −0.17; eFigure 2(a) and (b)), and quality of life (Q-LES-Q-SF: SMD = 0.22; 95% CI: 0.07–0.36; eFigure 2(c)). In contrast, maximum therapeutic effects for responder and remitter rates peaked at 28 mg, followed by a plateau. Compared to placebo, the responder rate was RR: 1.34 (95% CI: 1.07–1.68) at 28 mg and RR: 1.27 (95% CI: 1.05 to 1.53) at 42 mg (eFigure 2(d)), and the remitter rate was RR: 1.08 (95% CI: 0.79–1.46) at 28 mg and RR: 1.06 (95% CI: 0.81–1.38) at 42 mg (eFigure 2(e)).
Estimated effect sizes from dose–response meta-analysis.
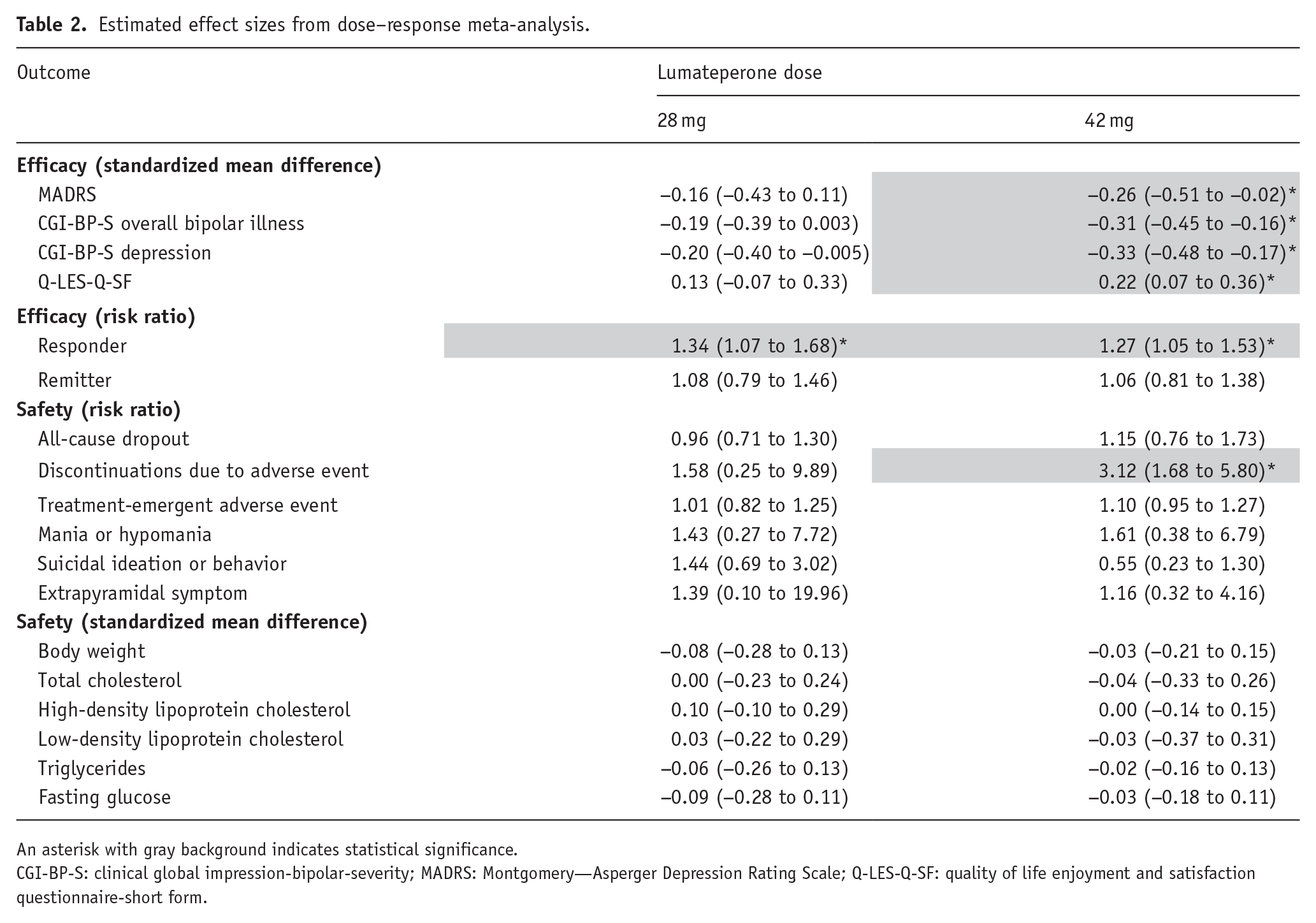
An asterisk with gray background indicates statistical significance.
CGI-BP-S: clinical global impression-bipolar-severity; MADRS: Montgomery—Asperger Depression Rating Scale; Q-LES-Q-SF: quality of life enjoyment and satisfaction questionnaire-short form.
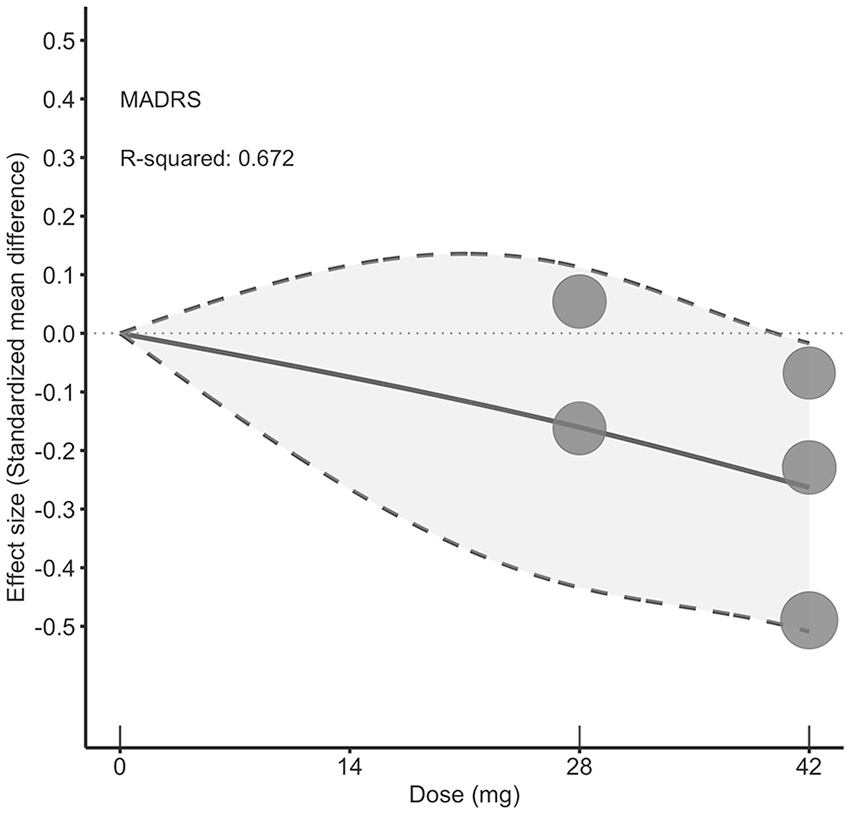
Dose–response relationship between daily lumateperone doses and depression symptoms. Solid line: pooled point estimates; dotted line: 95% confidence band around the pooled curve, illustrating the statistical uncertainty (narrower where data are abundant, wider where sparse); open circles: outcome markers for all included studies, with size representing the reciprocal of the standard error of the effect size; x-axis: short vertical lines indicate the lumateperone dose of the included studies, with 0 mg defined as placebo.
We conducted four additional analyses focusing on primary efficacy outcomes. First, leave-one-out sensitivity testing consistently identified the 42 mg dose of lumateperone as yielding the greatest therapeutic benefit, even though the magnitude of the pooled effect size varied modestly depending on which trial was excluded (eFigure 3). Second, when we incorporated data from the RCT that included both bipolar and unipolar depression, 42 mg remained the lumateperone dose with greatest therapeutic effect compared with placebo (eFigure 4). Third, evaluating changes over time revealed that only the 42 mg dose showed a significant increase in efficacy over the study period (3 weeks SMD: −0.10; 95% CI: −0.20 to −0.001; 6 weeks SMD: −0.18; 95% CI: −0.30 to −0.07; eFigure 5). Fourth, comparing dose–response trends for different bipolar subtypes showed a generally positive relationship between dose and effect in both bipolar I and II disorders (Figure 2). However, in bipolar I disorder, significant therapeutic efficacy was found at an even lower dose (28 mg) compared to the primary outcome (28 mg: SMD: −0.36; 95% CI: −0.60 to −0.13; 42 mg: SMD: −0.53; 95% CI: −0.77 to −0.29), whereas in bipolar II disorder, statistical significance was not reached at either the 28 or 42 mg doses (28 mg: SMD: −0.09; 95% CI: −0.88 to 0.70; 42 mg: SMD: −0.50; 95% CI: −1.78 to 0.78).
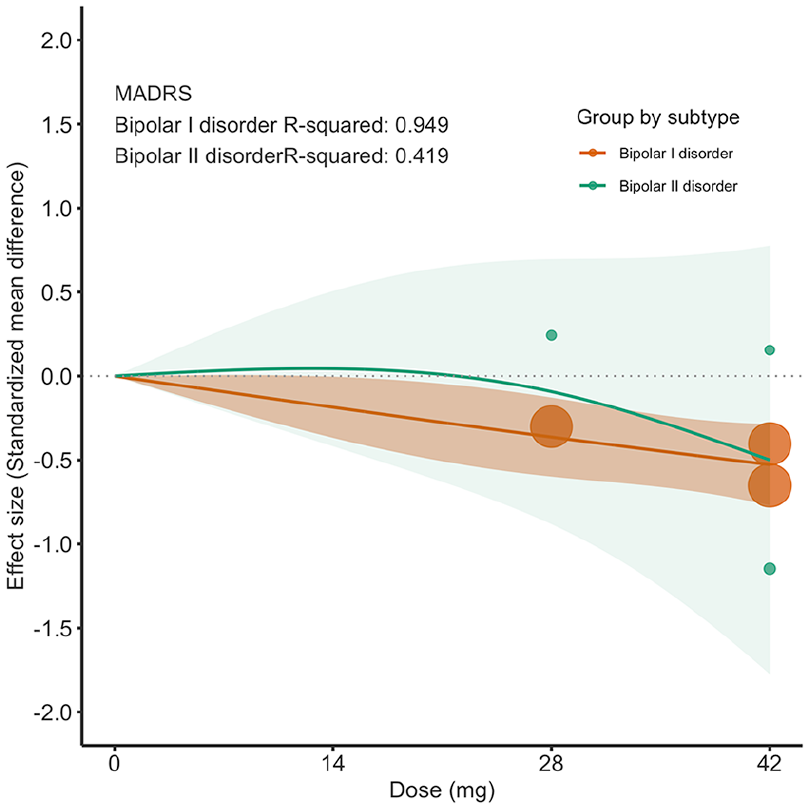
Subgroup analysis by bipolar depression subtypes for dose–response relationship between daily lumateperone doses and depression symptoms. Solid line: pooled point estimates; color-shaded areas: 95% confidence band around the pooled curve, illustrating the statistical uncertainty (narrower where data are abundant, wider where sparse); open circles: outcome markers for all included studies, with size representing the reciprocal of the standard error of the effect size; x-axis: 0 mg defined as placebo.
With respect to the all-cause dropout rates, no clear dose–response relationship emerged when comparing lumateperone to placebo (Figure 3 and Table 2). Among all AE outcomes, only discontinuations attributed to AEs reached statistical significance at the 42 mg dose (RR: 3.12; 95% CI: 1.68–5.80; eFigure 6(a) and Table 2). According to US FDA reports (U.S. Food and Drug AdministrationAdministration USFaD 2021), the most common AE-related reasons for discontinuation of lumateperone were headache, migraine, dizziness, and nausea/vomiting, each reported in approximately 0.5%–0.7% of patients. Other AE outcomes, such as TEAEs, mania or hypomania AEs, suicidal ideation or behavior AEs, and EPS did not show any specific dose-related increases (eFigure 6(b)–(e) and Table 2). Supplementary safety outcomes—body weight, total cholesterol, HDL-cholesterol, LDL-cholesterol, triglycerides, and fasting glucose—likewise exhibited no dose-dependent changes (eFigure 7(a)–(f) and Table 2). A detailed quality appraisal using the Cochrane Risk of Bias 2 tool is provided in eTable 3 and eFigure 8. Two trials were rated as having a low risk of bias (Calabrese et al., 2021; Suppes et al., 2023), while one study (ClinicalTrials.gov, 2015) raised concerns due to its unpublished status and incomplete outcome reporting compared with its study protocol, such as the absence of data on bipolar I and bipolar II disorders. Variance partition coefficients for the primary outcomes are available in eFigure 9. Model comparison fit statistics for the current restricted cubic spline and linear models are summarized in eTable 4. According to the GRADE assessment, the certainty of evidence for the primary outcomes was low (eTable 5).
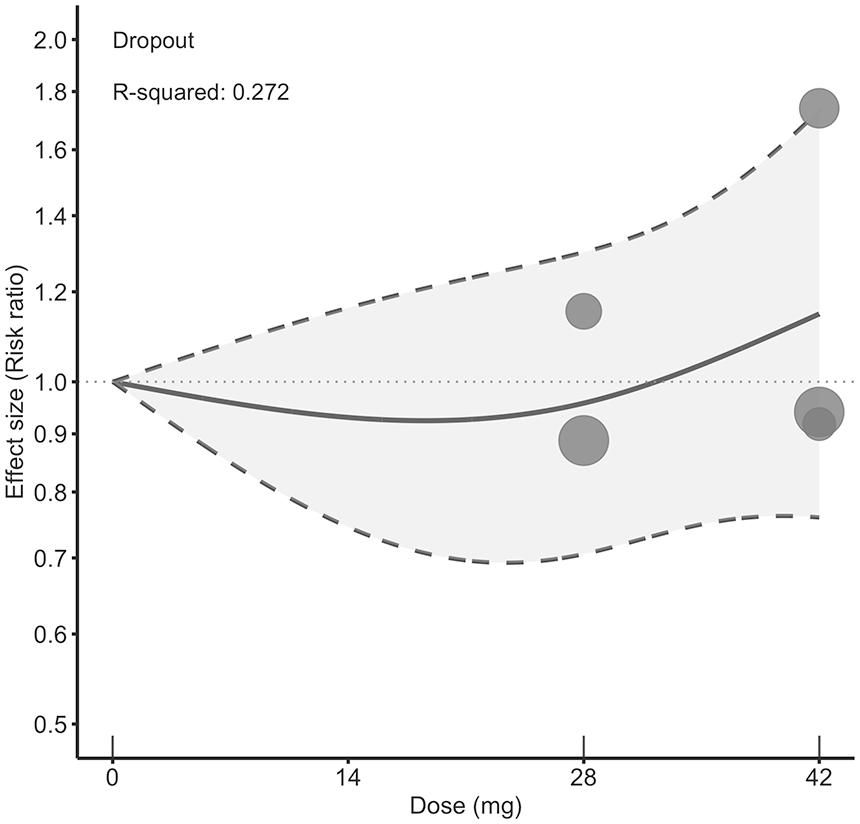
Dose–response relationship between daily lumateperone doses and dropout rate. Solid line: pooled point estimates; dotted line: 95% confidence band around the pooled curve, illustrating the statistical uncertainty (narrower where data are abundant, wider where sparse); open circles: outcome markers for all included studies, with size representing the reciprocal of the standard error of the effect size; x-axis: short vertical lines indicate the lumateperone dose of the included studies, with 0 mg defined as placebo.
Discussion
This meta-analysis evaluated the efficacy and safety of different doses of lumateperone. In terms of efficacy, our results showed that lumateperone at a dose of 42 mg significantly improved depressive symptoms, global illness severity, quality of life, and responder rate relative to placebo. In comparison, the 28 mg dose did not show a statistically significant improvement under our analytical model except for responder rate. Time-course analysis further supported these conclusions: the 42-mg dose produced progressive reductions in depressive symptoms over 6 weeks of treatment, whereas the 28-mg dose did not follow the same trajectory. Subgroup analysis by bipolar disorder subtype showed that 42 mg was the most effective dose in treating bipolar I disorder and bipolar II disorder. In terms of safety, the only significant risk with the 42 mg dose was discontinuation due to AEs. No clear dose–response association emerged for other safety outcomes including all-cause dropout rates, TEAEs, mania or hypomania, suicidal ideation or behavior, EPS, body weight, total cholesterol, HDL-cholesterol, LDL-cholesterol, triglycerides, and fasting glucose. Overall, lumateperone demonstrated good tolerability.
Our findings suggest that for the treatment of bipolar depression, 42 mg (rather than 28 mg) of lumateperone may be required to achieve a clinically meaningful therapeutic benefit, which aligns with the US FDA-approved dose. A reasonable pharmacological explanation is that lumateperone exerts its antidepressant effects through two main mechanisms: antagonism of 5-HT2A receptors and antagonism of postsynaptic D2 receptors (Davis and Correll, 2016; Kantrowitz, 2020; Titulaer et al., 2022). We hypothesize that fully activating both pathways is crucial for optimal antidepressant efficacy. However, lumateperone’s affinity for the 5-HT2A receptor is significantly greater than its affinity for the D2 receptor (approximately 60 to; Kantrowitz, 2020; Snyder et al., 2015). Therefore, at lower doses, lumateperone appears to act primarily through its 5-HT2A antagonism, a mechanism that can produce a first-line antidepressant effect (Davis and Correll, 2016; Sowa-Kućma et al., 2024). At higher doses, the drug likely leverages dopaminergic modulation by antagonizing postsynaptic D2 receptors, thereby offering a second and more pronounced antidepressant mechanism (Davis and Correll, 2016; Sowa-Kućma et al., 2024). This hypothesis is indirectly supported by responder rate data, which suggest that for many patients, 28 mg is sufficient to reduce depressive symptoms by approximately 50% (responders; Suppes et al., 2023), presumably through 5-HT2A blockade. However, the 42 mg dose regimen appears necessary to fully engage the drug’s effect on the postsynaptic D2 pathway, thereby achieving more prominent clinical symptom improvement, as assessed by the MADRS or CGI-BP-S. Nonetheless, neither dose achieved statistical superiority over placebo on the stringent remission endpoint of MADRS ⩽ 12 (Table 2). This underscores the therapeutic challenge of bipolar depression: achieving full symptomatic remission with a single agent is difficult (Husain and Yatham, 2023), and simple dose escalation is often insufficient. eTable 6 summarizes the efficacy and safety of the five US FDA-approved treatments for bipolar depression, and illustrates that combination regimens, such as olanzapine (antipsychotic) plus fluoxetine (antidepressant), may be needed to attain higher remission rates than lumateperone (antipsychotic) monotherapy (Yildiz et al., 2023), a finding that merits careful clinical consideration.
Regarding safety, all-cause dropout and TEAEs rates did not appear to be clearly dose related. Critically, mania or hypomania and suicidal ideation or behavior—both critical concerns when treating bipolar depression—did not display significant dose-related trends. It is also noteworthy that lumateperone is relatively well tolerated for EPS, possibly due to its unique pharmacological profile as a postsynaptic D2 receptor antagonist and presynaptic D2 partial agonist (Greenwood et al., 2021; Kantrowitz, 2020). This dual mechanism may enable a more nuanced modulation of dopaminergic signaling, thereby reducing the EPS risk commonly associated with traditional antagonists (Blair, 2020; Dedic et al., 2019; Snyder et al., 2015). Nonetheless, discontinuations due to AEs were significantly higher with the 42-mg dose than with placebo, a finding that should alert clinicians to monitor tolerability closely. Furthermore, compared with placebo, lumateperone at 42 mg as well as at lower doses, did not materially affect metabolic parameters such as body weight, lipid profile, or fasting glucose. Taken together with evidence that antidepressant efficacy tends to improve as dose increases, these data suggest the possibility of an optimal therapeutic window above the currently studied ceiling of 42 mg. A useful analogy can be drawn from a recent dose–response meta-analysis of lurasidone, another antipsychotic for bipolar depression, which identified an efficacy “sweet spot” between 40 and 60 mg despite some trials testing doses up to 120 mg (Lin et al., 2024). Current studies have not found an upper therapeutic dose limit for lumateperone, and it seems reasonable that doses above 42 mg of lumateperone may result in greater therapeutic effects without substantially compromising safety. However, this assumption remains speculative due to the lack of sufficient data. Future RCTs specifically designed to evaluate higher doses of lumateperone are needed to validate this hypothesis.
One notable aspect of this study concerns the dose–response relationship of lumateperone in distinct bipolar subtypes, especially since lumateperone has received FDA approval for treating depressive episodes associated with bipolar I or bipolar II disorder (Abuelazm et al., 2023). In bipolar I disorder, higher doses consistently translated into steady therapeutic gains. By contrast, in bipolar II disorder, the effect was negligible below 28 mg and appeared to improve only near 42 mg, yet no dose achieved statistical significance. Although the FDA label includes bipolar II disorder (Abuelazm et al., 2023), our findings do not substantiate this indication. One plausible explanation is insufficient statistical power stemming from the small number of bipolar II participants: only two of the three eligible RCTs reported subtype-specific outcomes (Calabrese et al., 2021; Suppes et al., 2023), and one of these showed suboptimal efficacy in bipolar II depression (Suppes et al., 2023). Until larger, adequately powered RCTs are completed, current evidence cannot reliably endorse 42 mg lumateperone—or any dose—as evidence-based therapy for bipolar II disorder. Moreover, our dose–response curve hints that doses above 42 mg might confer additional benefit. Future research should therefore explore higher dosing regimens and delineate the patient subgroups most likely to respond.
A key strength of our analysis is its focus on lumateperone’s efficacy within a rigorous dose–response framework. The largest SMD observed—approximately 0.30 at the 42 mg dose—corresponds to a small effect size, especially when contrasted with lurasidone, which achieves an SMD of roughly 0.60 at 40–60 mg (Lin et al., 2024). Nevertheless, compared with other agents commonly used for bipolar depression, such as quetiapine, olanzapine, and cariprazine, lumateperone provides a comparatively favorable safety profile, particularly with respect to weight gain and EPS (Yildiz et al., 2023). Clinicians might therefore prioritize lumateperone when metabolic or motor-tolerability is a primary concern. However, several limitations warrant attention. First, only three RCTs of lumateperone in bipolar depression met our inclusion criteria, and despite an exhaustive search that included FDA sources, conclusions regarding certain secondary outcomes (e.g., discontinuation due to AEs) were based on evidence from only two studies. Accordingly, we will maintain this work as a living systematic review (INPLASY202510048), and the present findings should be regarded as exploratory. Second, owing to the small number of included studies, our ability to perform subgroup analyses for specific demographic factors (e.g., pediatric patients (ClinicalTrials.gov, 2024) was constrained. Third, despite the lack of direct pharmacodynamic data, our model suggests the optimal dose may exceed 42 mg, yet no RCT has tested higher doses. Future trials should examine escalated regimens (e.g., 56–64 mg) to determine whether they provide additional benefit. Fourth, lumateperone has not been directly compared with other active treatments (e.g., lurasidone or quetiapine) in head-to-head RCTs, leaving its relative efficacy uncertain. Such trials would also create a foundation for network meta-analysis. Fifth, the lack of individual participant data (IPD) prevented us from performing IPD meta-analyses, which are essential for stratifying results by factors such as age or baseline body mass index given the substantial variability in AE tolerability across these subgroups. Finally, data on sedation were unavailable in the included trials. Future RCTs should prospectively designate sedation and EPS, both major clinician concerns, as core safety endpoints to ensure robust and clinician-relevant evidence synthesis.
Conclusions
This preliminary dose–response meta-analysis suggests that a dose of 42 mg of lumateperone may be required to effectively treat bipolar depression. At this dose, patients experienced significant improvements in depressive symptoms, global illness severity, and quality of life, while rates of all-cause dropout, TEAEs, mania, suicidality, body weight gain, lipid abnormalities, and fasting glucose elevations remained comparable with placebo. However, discontinuations due to AEs were more frequent, underscoring the need for careful monitoring and caution in practice. Furthermore, based on our dose–response model, doses above 42 mg may potentially produce greater efficacy, although such treatment regimens have not yet been examined. Well-powered, dose-finding trials are therefore needed to evaluate higher dosing strategies and to delineate the optimal therapeutic window that best balances efficacy and safety for lumateperone.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251364389 – Supplemental material for Dose-response efficacy and safety of lumateperone in bipolar depression: A preliminary meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-jop-10.1177_02698811251364389 for Dose-response efficacy and safety of lumateperone in bipolar depression: A preliminary meta-analysis of randomized controlled trials by Chih-Wei Hsu, Yu-Kang Tu, Kuo-Chuan Hung, Chih-Sung Liang, Ping-Tao Tseng and Yang-Chieh Brian Chen in Journal of Psychopharmacology
Footnotes
Acknowledgements
The authors thank Ms. Pei-Ying Yang for technical support.
Author’s contributions
C-WH conceived the idea for this study. C-WH led the study design with Y-CBC. C-WH and Y-CBC contributed to the data acquisition and extraction. C-WH performed statistical analyses. C-WH drafted the manuscript first; Y-CBC, Y-KT, K-CH, C-SL, and P-TT revised the manuscript. All authors contributed important intellectual content during manuscript revision, had full access to all study data, and accepted the responsibility to submit the manuscript for publication.
Data sharing statement
The data supporting the findings of this study are available from the corresponding author, CWH, upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Taiwan National Science and Technology Council (112-2314-B-182-070-MY3) and the funding sources had no role in the design of the study. C-WH is supported by Taiwan National Science and Technology Council (109-2314-B-182A-009-MY2, 111-2314-B-182A-027-, and 112-2314-B-182-070-MY3) and the Chang Gung Medical Foundation (CMRPG8N0881, CMRPG8P0631, CORPG8P0561, and BMRPJ30).
Ethics approval
Institutional review board approval was not required because this article uses published data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

