Abstract
Objectives:
Garcinia mangostana Linn. (“mangosteen”) pericarp contains bioactive compounds that may target biological pathways implicated in schizophrenia. We conducted a double-blind randomized placebo-controlled trial evaluating the efficacy of adjunctive mangosteen pericarp, compared to placebo, in the treatment of schizophrenia.
Methods:
People diagnosed with schizophrenia or schizoaffective disorder (Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition), recruited across 2 sites (Brisbane and Victoria, Australia), were randomized to receive 24 weeks of adjunctive mangosteen pericarp (1,000 mg/day) or matched placebo. The primary outcome measure was the Positive and Negative Symptom Scale total score. Secondary outcomes included positive and negative symptoms, general psychopathology, clinical global severity and improvement, participant reported overall improvement, depressive symptoms, functioning, quality of life, and safety data at 24 and 28 weeks (4 weeks postdiscontinuation). Data were collected from July 2016 to February 2019.
Results:
Baseline assessments were conducted on 148 people (mangosteen = 74, placebo = 74); data analyses were conducted on 136 (92%) participants with postbaseline data. The treatment group had significantly higher symptom severity compared to placebo, and both groups significantly improved on all symptom, functioning, and quality of life measures over time. No between-group differences were found for the rate of change between baseline and 24 or 28 weeks.
Conclusion:
Despite promising preclinical and clinical work, our results do not support mangosteen pericarp extract as an adjunctive treatment for schizophrenia or schizoaffective disorder.
Introduction
Schizophrenia is a complex and heterogeneous neuropsychiatric disorder. Lifetime prevalence is estimated to be 1%, with several factors conferring additional risk. Diagnostic features include “positive” symptoms (delusions and hallucinations), “negative” symptoms (affective flattening and social withdrawal), and “cognitive” symptoms such as memory and attention deficits. Consistent with this broad constellation of symptoms, the clinical manifestations of schizophrenia can be diverse. Moreover, schizophrenia is associated with substantial illness burden. 1 Even with adequate treatment, up to one-third of people with the condition still experience ongoing psychotic symptoms, 2 with many more experiencing persistent problems with functioning and quality of life. 3
While monoaminergic (particularly dopaminergic) dysfunction is associated with psychotic disorders, 4 the underlying pathophysiology in schizophrenia is still incompletely understood. Accumulating evidence indicates that nitro-oxidative stress, 5 immune-inflammatory activation, 6 mitochondrial and bioenergetic dysfunction, 7 neurogenesis, 8 and apoptosis 9 may also play a role. Although currently available antipsychotic treatments are effective, there are well-known adverse effects 2 and therapeutic shortfalls. In particular, negative and cognitive symptom domains are largely unresponsive to antipsychotic medications. 10 It has been previously proposed that targeting immune-inflammatory, nitro-oxidative stress, and neurotrophic pathways with adjunctive pharmacotherapies may improve therapeutic outcomes. 11 –13
The thick pericarp or rind of the tropical fruit Garcinia mangostana Linn., commonly known as mangosteen, contains an array of bioactive compounds such as xanthones and flavonoids. Mangosteen has long been used in traditional medicine and more recently has been the subject of both preclinical and clinical investigations. Preclinical investigations of the neurobiological activities of mangosteen have revealed a range of actions that potentially target the theorized pathophysiological pathways of schizophrenia. Numerous studies have demonstrated antioxidant, anti-inflammatory, and neuroprotective effects of both the pericarp and selected compounds (most commonly α- and γ-mangostin; see the studies of Ashton et al 14 and Do and Cho 15 ), aligning with oxidative stress and impaired inflammatory responses in both the acute and chronic phases of schizophrenia, 16,17 as well as other serious mental illnesses such as bipolar disorder (e.g., see the studies of Do and Cho 15 and Goldsmith et al 17 ). In vitro studies have demonstrated that α- and γ-mangostin in particular attenuate lipopolysaccharide-induced promotion of the proinflammatory signaling cascade, reducing interleukin 1β, interferon γ, tumor necrosis factor α, mitogen-activated protein kinase, nuclear factor κB, and nitric oxide synthase. 18,19 Notably, α-mangostin was also found to attenuate interleukin 6 and cyclooxygenase 2 in vitro, 19 findings which are supported by in vivo murine data. 20 Mangosteen pericarp extracts have been found to inhibit cell death, improve free radical scavenging, 21 and increase glutathione and serotonin in vitro. 22 Studies have also demonstrated free radical scavenging by increased acetylcholinesterase and reduction in malondialdehyde production 23 and reduced cognitive impairment in mice in vivo. 22
With regard to behavioral investigations, evidence of antidepressant and procognitive effects have been seen in animal models of schizophrenia and depression, 24,25 suggesting investigation of efficacy in both the depressive symptoms of schizophrenia and in mood disorders may be warranted. Small clinical trials have investigated effects of mangosteen on biological markers as well as behavior in humans, demonstrating positive changes to inflammatory and antioxidant markers, 26 –28 insulin levels, 29 and mood in healthy overweight adults. 30 No apparent adverse effects have been reported, suggesting a positive safety and tolerability profile. 14,15 Current theories around the pathophysiology of schizophrenia, preclinical data demonstrating the bioactivity of mangosteen, and available clinical and safety data point to the potential of mangosteen as an adjunctive treatment of schizophrenia.
Objectives and Hypotheses
The aim of this study was to determine, via a randomized, double-blind, placebo-controlled trial, whether 24 weeks of adjunctive treatment with 1,000 mg/day of mangosteen pericarp would improve clinical outcomes in people with schizophrenia or schizoaffective disorder compared to placebo.
Primary hypothesis
It was hypothesized that adjunctive mangosteen pericarp treatment would be superior to placebo in the treatment of overall psychopathology in schizophrenia, as assessed by mean change over time in the total score of the Positive and Negative Syndrome Scale (PANSS 31 ) from baseline to week 24 (completion of the treatment phase and primary end point).
Secondary hypotheses
It was hypothesized that 24 weeks of adjunctive mangosteen pericarp treatment would be superior to placebo in reducing severity of general, positive, and negative symptoms of schizophrenia (PANSS general [PANSSG], positive [PANSSP], and negative [PANSSN] subscale scores) and in reducing depressive symptoms, functioning, and quality of life. It was further hypothesized that there would be no difference in adverse events between the 2 groups over the 24 weeks of the trial.
An exploratory aim was to delineate whether any improvements in response to mangosteen pericarp that are observed at 24 weeks are sustained to 28 weeks (4 weeks postdiscontinuation).
Methods
Trial Design
The study design was a placebo-controlled, double-blind, randomized controlled trial (RCT). The study protocol has been published. 32 Participants provided written informed consent. Human ethics approval was received from Barwon Health Human Research Ethics Committee (HREC), Geelong, Victoria, (reference number 15/26); and Metro South Health Service District HREC, Queensland (reference number HREC/16/QPAH/15), Australia. The trial was conducted in accordance with Good Clinical Practice guidelines, Australian Clinical Trial guidelines, 33 and the National Statement on Ethical Conduct in Human Research. 34 Data collection commenced July 2016 and was completed February 2019. The study was prospectively registered with the Australian and New Zealand Clinical Trials Registry (ACTRN12616000859482).
Participants
Participants were recruited from sites in Geelong, Victoria, and Brisbane, Queensland. Both sites recruited from mental health services in the surrounding districts. The Geelong site also recruited from the community, using advertising, flyers, and contact with potential referral sources. Participants were reimbursed for travel costs to 20 AUD/visit.
Eligibility criteria
Study inclusion criteria were that participants were aged 18 years or older and met Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition 35 ) criteria for schizophrenia or schizoaffective disorder and scored ≥54 on the PANSS and/or ≥3 on the Clinical Global Impressions severity of illness scale (CGI-S 36 ). If there was a delay of >7 days between screening and baseline assessments or baseline assessment and medication commencement, the inclusion scales (PANSS and CGI-S) were readministered to ensure the participants still met eligibility criteria. If on psychotropic therapy, the regime needed to have been stable for 4 weeks prior to study entry. They also needed to be using effective contraception if female, sexually active, and of childbearing age; be able to speak, read, write, and understand the English language; have a current treating physician; and have capacity to consent to the study and comply with study procedures.
Participants were excluded if they had a known or suspected clinically unstable systemic medical disorder; were pregnant or breastfeeding; had any contraindications or intolerance to mangosteen pericarp or any of the trial preparations; or were currently enrolled in another clinical trial.
Withdrawal criteria
Participants were withdrawn if they ceased taking their trial medication for ≥7 consecutive days; if they ceased effective contraception or became pregnant; if they withdrew consent; or at the discretion of the researcher given adverse events or loss to follow-up. Dose changes to existing medications or addition or removal of an agent were allowed. Serious adverse events (SAEs) did not require automatic withdrawal from the study.
Interventions
The interventional product was 1,000 mg/day of mangosteen pericarp (two 500 mg capsules, once a day). The content of investigational product was provided by VitalXan (Adelaide, Australia) and produced from the inner rind of the mangosteen fruit as described in the protocol paper. Placebo tablets were produced with identical capsules and were identical in appearance, color, and taste to the active capsules. The mangosteen pericarp and matching placebo capsules (Pharmaceutical Packaging Professionals, Australia) were packed in identical bottles labelled with the anonymous participant ID (using the randomization schedule). The medication was dispensed at baseline and weeks 4, 8, 12, 16, and 20. Participants were instructed to take 2 capsules once a day (at any time, as long as this was consistent) with food due to evidence that dietary fat increases bioavailability of α-mangostin. 37 Unused medication was returned to the researchers.
Outcomes
Following referral, face-to-face consenting and eligibility screening, the baseline measures were collected and follow-up assessments conducted every 4 weeks until week 28. The primary end point was week 24 (end of treatment) with week 28 a postdiscontinuation visit.
Primary outcome
The primary outcome of the study was change in PANSS total score (PANSST), from baseline to the end of treatment (week 24). The PANSS is a 30-item scale evaluating positive symptoms, negative symptoms, and general psychopathology symptoms of schizophrenia. 31 All items are rated on a 7-point scale (1 = absent to 7 = extreme), with a total score range of 30 to 210.
Secondary outcomes
Secondary outcomes included PANSSP, PANSSN, and PANSSG subscales; clinician rated overall symptom state (CGI-S and CGI-Global Improvement); participant reported symptom state (Patient Global Improvement) 36 ; depressive symptoms (Montgomery Åsberg Depression Rating Scale [MADRS]) 38 ; functioning (Global Assessment of Functioning Scale [GAF] 39 and the Longitudinal Interval Follow-up Evaluation-Range of Impaired Functioning Tool [LIFE-RIFT] 40 ); and quality of life (Quality of Life Enjoyment and Satisfaction Questionnaire [Q-LES-Q]). 41
Adherence was estimated via capsule counts of medication returned by participants. Safety was assessed by monitoring and recording of adverse events (including SAEs), elicited by the open-ended question “Do you have any specific worries or complaints about your health in general?” The Abnormal Involuntary Movement Scale (AIMS), a standard rating scale for neuroleptic-based clinical trials, was administered at every interview. 36 We used a total score of the first 7 items to ascertain an index of observed movements.
Assignment of Interventions
Allocation to treatment arm was randomly assigned in a 1:1 ratio (active to placebo) using permutated block randomization, stratified by site (Brisbane and Geelong). An independent researcher developed a computer-generated randomization (randomization.com). Participants, trial clinicians, investigators, and statisticians were blinded to treatment allocation. The randomization plan was sent by the independent researcher to the investigational product manufacturer to package the active and placebo products according to participant ID and allocation, in identical packaging. The clinical trial pharmacy held a copy of the randomization list in case of unblinding requests, with another copy kept in a sealed envelope by the investigators in case of emergency. Participants were notified which arm of the study they took part in after the completion of all participants in the study. The investigators and statistician were unblinded after the statistical analysis was completed.
Statistical Methods
A sample size of 50 per group (100 total) was identified a priori as providing 90% power to detect a medium effect size with α at the 0.05 level. A medium effect size was chosen given this was the smallest effect size seen in the pilot RCT (for the GAF). 32 To allow for attrition, the final target sample size was increased to 75 per group (total 150).
The analyses were conducted in accord with the International Conference on Harmonization E9 statistical principles. 42 Results are presented based on the Consolidated Standards of Reporting Trials. 43 Baseline characteristics and participant flow for all those who are randomized are described using descriptive statistics. Analyses were based on all randomized participants with at least 1 postbaseline observation and are therefore based on a modified intention-to-treat (mITT).
The primary efficacy analysis involved the assessment of average treatment group differences for the PANSST over the entire treatment period using a linear mixed effects model (LMM) with a restricted maximum likelihood estimator as generated using the R (version 3.6.1) lme4 (version 1.1-21) package. The model comprised random effects for the intercepts and slopes and included the variables group, visit, site, and group by visit. Unstructured covariance matrix was used for the random slopes. From these models, we also estimated average group differences (±95% confidence intervals [CIs]) in change of PANSST scores from baseline to week 24 (end of treatment), baseline to week 28 (postdiscontinuation), and week 24 (end of treatment) to week 28 (postdiscontinuation). This approach to statistical analysis was also adopted for the secondary outcome measures including symptoms, functioning, and quality of life. A range of adaptations to the LMMs were adopted for sensitivity analyses.
Results
Cohort Characteristics
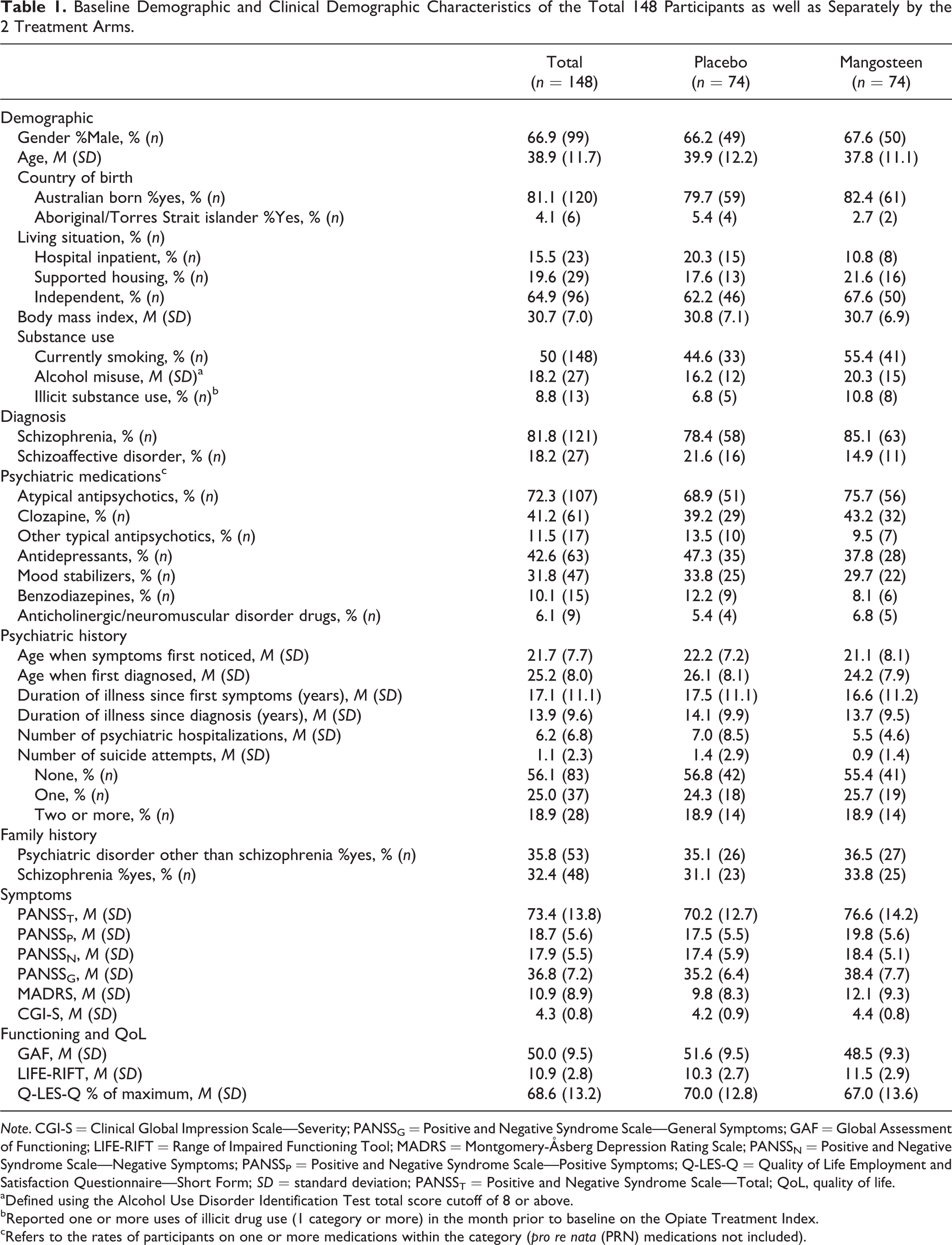
Of 150 people recruited, 148 participants were randomized and had baseline data (see Table 1 and Supplementary Table 1). Approximately, two-thirds of participants (65.5%, n = 97) were from the Brisbane site. The majority of participants were male (66.9%, n = 99), with ages ranging from 18 to 69 years (M = 38.9, SD = 11.7). Most participants were Australian born (81.1%, n = 120) and living independently in the community (64.9%, n = 96).
Baseline Demographic and Clinical Demographic Characteristics of the Total 148 Participants as well as Separately by the 2 Treatment Arms.
Note. CGI-S = Clinical Global Impression Scale—Severity; PANSSG = Positive and Negative Syndrome Scale—General Symptoms; GAF = Global Assessment of Functioning; LIFE-RIFT = Range of Impaired Functioning Tool; MADRS = Montgomery-Åsberg Depression Rating Scale; PANSSN = Positive and Negative Syndrome Scale—Negative Symptoms; PANSSP = Positive and Negative Syndrome Scale—Positive Symptoms; Q-LES-Q = Quality of Life Employment and Satisfaction Questionnaire—Short Form; SD = standard deviation; PANSST = Positive and Negative Syndrome Scale—Total; QoL, quality of life.
aDefined using the Alcohol Use Disorder Identification Test total score cutoff of 8 or above.
bReported one or more uses of illicit drug use (1 category or more) in the month prior to baseline on the Opiate Treatment Index.
cRefers to the rates of participants on one or more medications within the category (pro re nata (PRN) medications not included).
With regard to baseline clinical characteristics of the cohort (see Table 1), just over 80% had a diagnosis of schizophrenia with the remainder diagnosed with schizoaffective disorder. The average age of onset of symptoms was 21.7 (SD = 7.7) years, whereas the average age of formal diagnosis was 25.2 (SD = 8.0) years. The average length of time since diagnosis was 13.9 (SD = 9.6) years. The average number of hospitalizations prior to baseline was 6.2 (SD = 6.8), with the maximum being 50 hospitalizations. Just under 44% of participants (n = 65) reported having one or more suicide attempts during their lifetime.
Table 1 also summarizes symptom levels at baseline. Mean ratings on the PANSS subscales were in line with what would be expected of a cohort with schizophrenia spectrum disorder. 31 The mean MADRS rating indicated the presence of mild depression. The rating on the CGI-S indicated that patients were moderately unwell. The average rating on the GAF indicated moderate to severe symptoms and impairments in functioning.
Participant Flow
Of the 148 who were randomized and had baseline assessment, 12 cases (8.1%) did not have any postbaseline data; 9 of these cases were in placebo (12.2%) and 3 received mangosteen pericarp (4.1%), χ2(1) = 3.27, P = 0.07 (see Figure 1). In accordance with the mITT, these cases were excluded from the proceeding analyses.
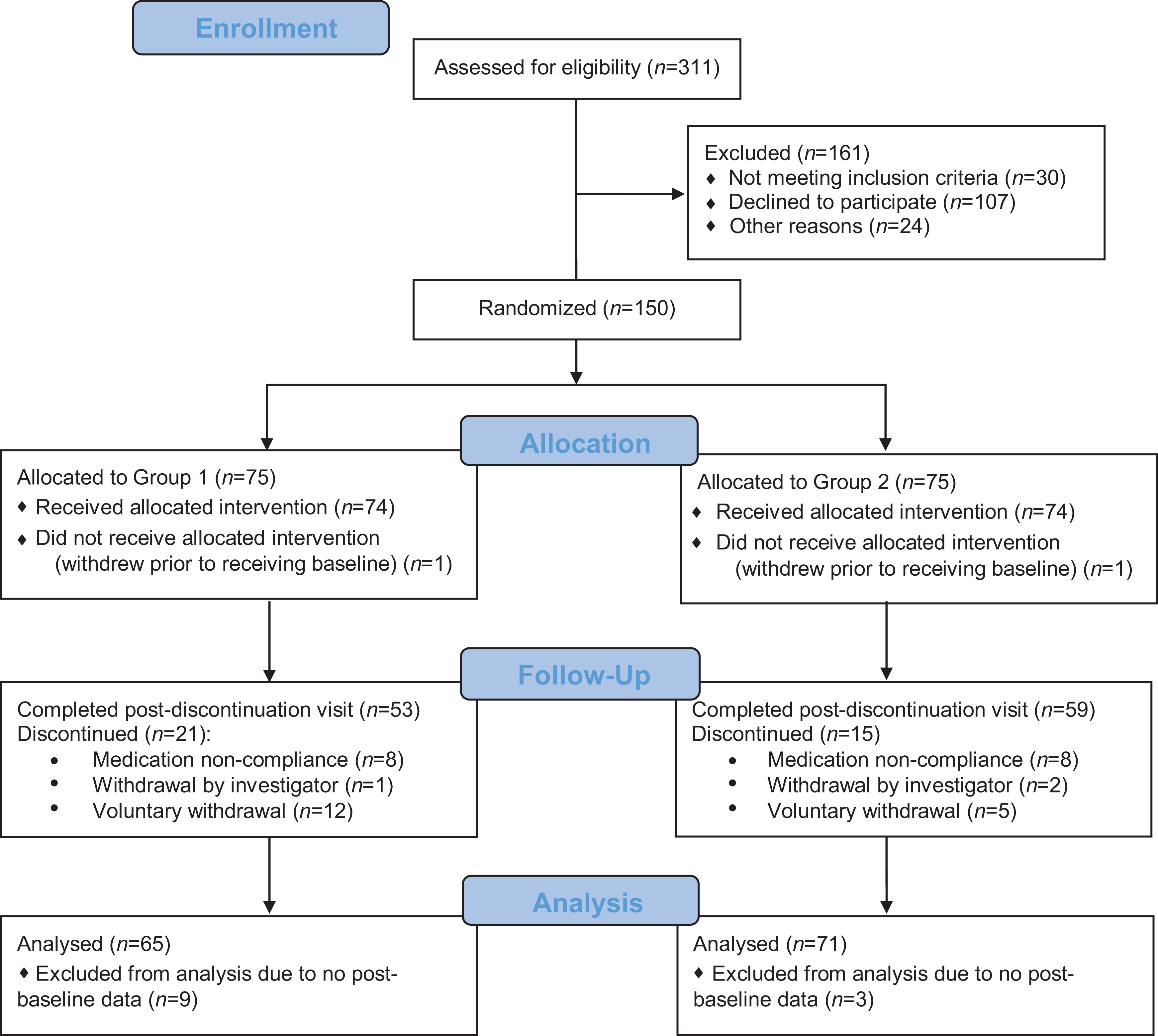
CONSORT diagram. CONSORT = Consolidated Standards Of Reporting Trials.
Of the 136 included in the analysis, 84.6% (n = 115) had data at the primary end point and 82.4% (n=112) had data at the postdiscontinuation visit. There were no significant differences between the groups with respect to rates of lost to follow-up at the primary end point, χ 2(1) = .21, P = 0.65, or at postdiscontinuation, χ2(1) = 0.06, P = 0.81. Those who did or did not have data at either primary end point or postdiscontinuation data did not differ significantly on any demographic, clinical, functioning, or quality of life variable with the exception of indigenous status.
Primary Outcome—PANSST
Figure 2 displays the mean (±95% CI) change from baseline at each of the 7 follow-up time points for placebo and treatment with respect to PANSST. Parameter estimates from the LMM shown in Tables 2 and 3 comprise the estimates pertaining to group differences in change from baseline to week 24, baseline to week 28, and between week 24 and week 28. With respect to PANSST, treatment had significantly higher symptom severity compared to placebo, P = 0.01, and both groups demonstrated decline in symptom severity over the 28 weeks, P < 0.001. The group by visit interaction was not significant, P = 0.42.
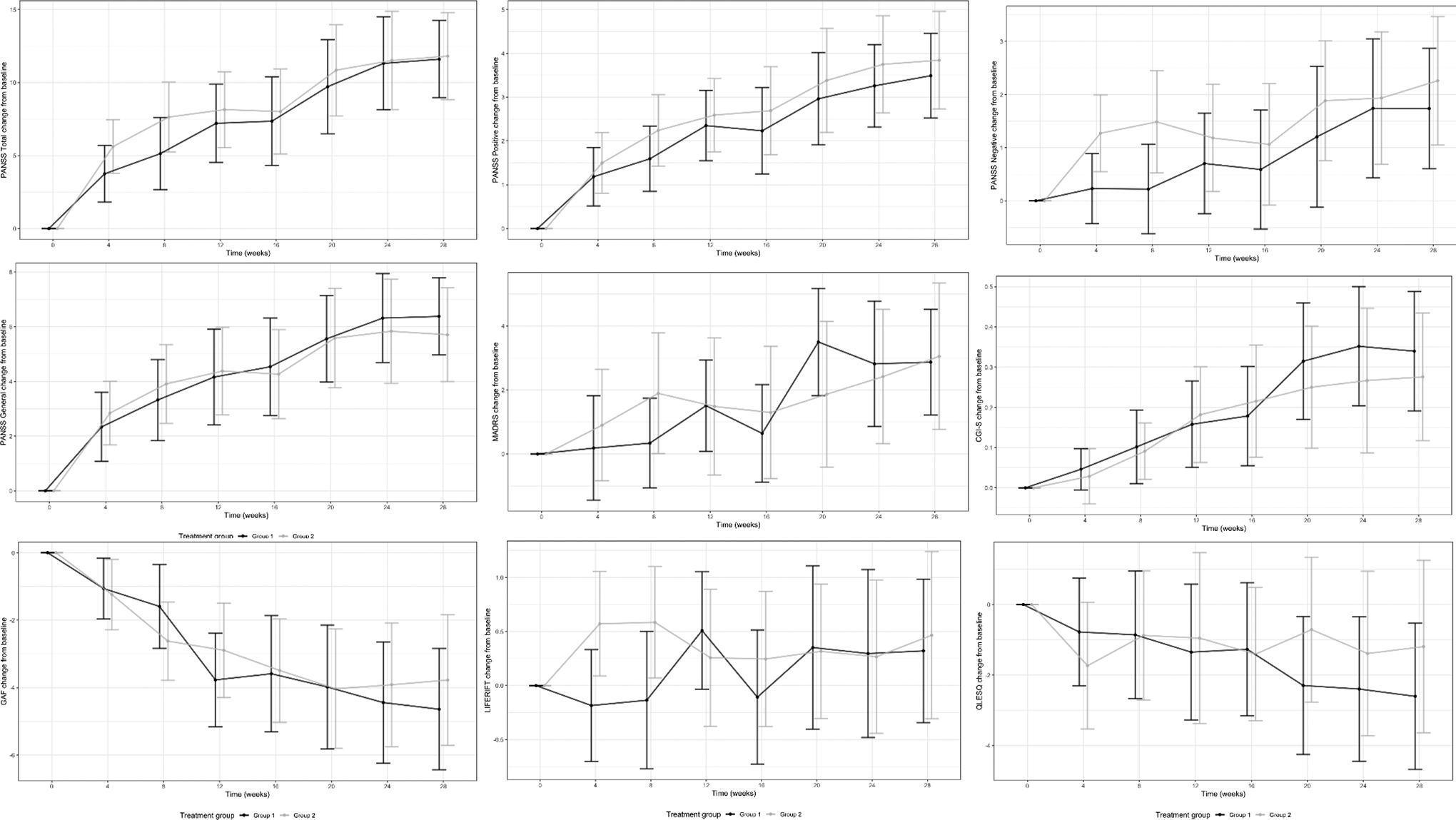
Mean change from baseline (±95% confidence intervals) for the 2 groups over 28 weeks on clinical, functioning, and quality of life measures. Group 1: placebo; Group 2: mangosteen. PANSS = Positive and Negative Syndrome; MADRS = Montgomery-Åsberg Depression Rating Scale; CGI-S = Clinical Global Impression Scale—Severity; GAF = Global Assessment of Functioning; LIFE-RIFT = Range of Impaired Functioning Tool; Q-LES-Q = Quality of Life Employment and Satisfaction Questionnaire—Short Form.
Parameter Estimates from Linear Mixed Models with Restricted Maximum Likelihood Estimator and Random Slopes and Random Intercepts for Clinical, Functioning, and Quality of Life Measures.
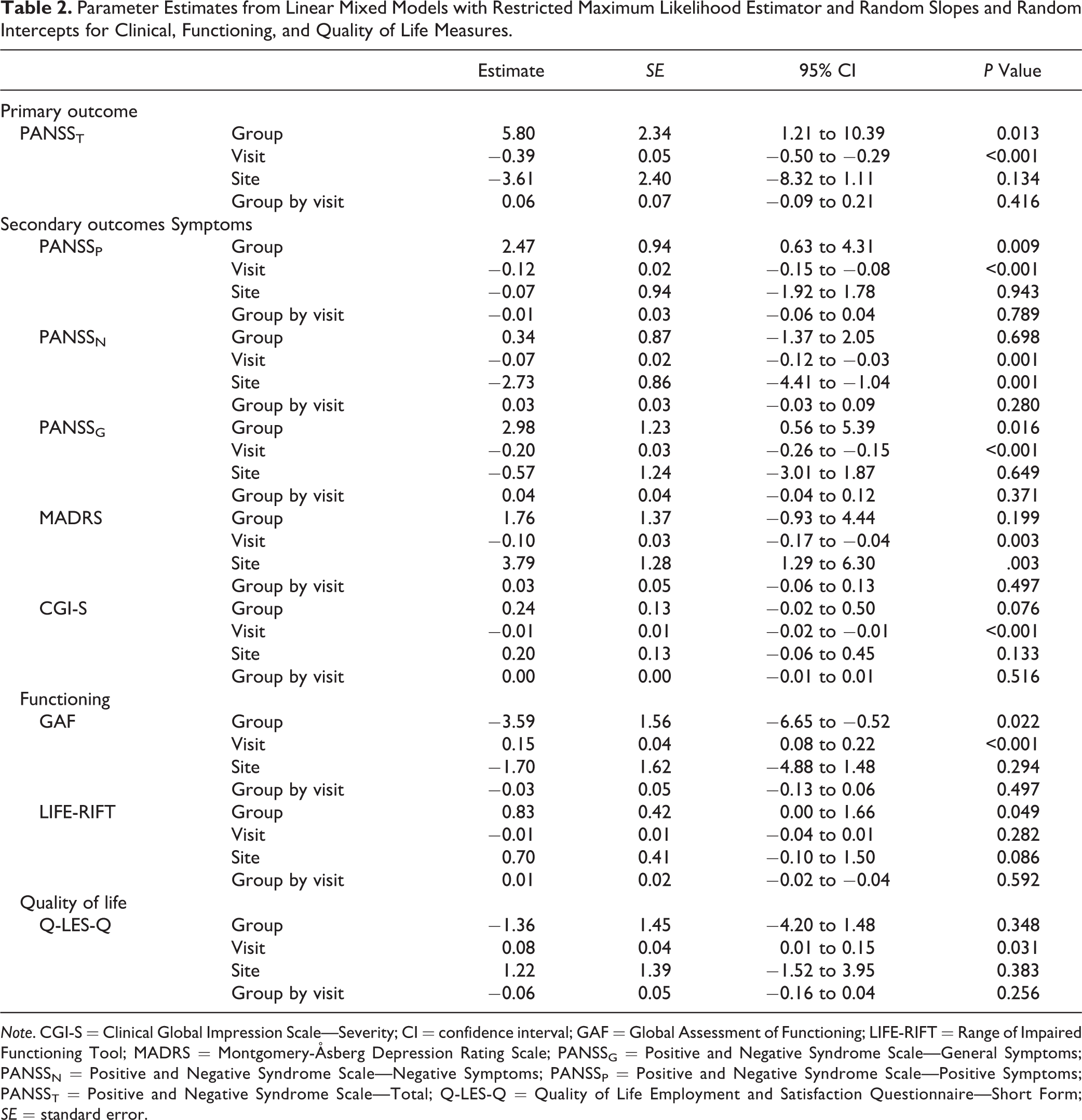
Note. CGI-S = Clinical Global Impression Scale—Severity; CI = confidence interval; GAF = Global Assessment of Functioning; LIFE-RIFT = Range of Impaired Functioning Tool; MADRS = Montgomery-Åsberg Depression Rating Scale; PANSSG = Positive and Negative Syndrome Scale—General Symptoms; PANSSN = Positive and Negative Syndrome Scale—Negative Symptoms; PANSSP = Positive and Negative Syndrome Scale—Positive Symptoms; PANSST = Positive and Negative Syndrome Scale—Total; Q-LES-Q = Quality of Life Employment and Satisfaction Questionnaire—Short Form; SE = standard error.
Mean Change (95% Confidence Intervals) from Baseline to Week 24 (End of Treatment), Baseline to Week 28, and Week 24 to Week 28.
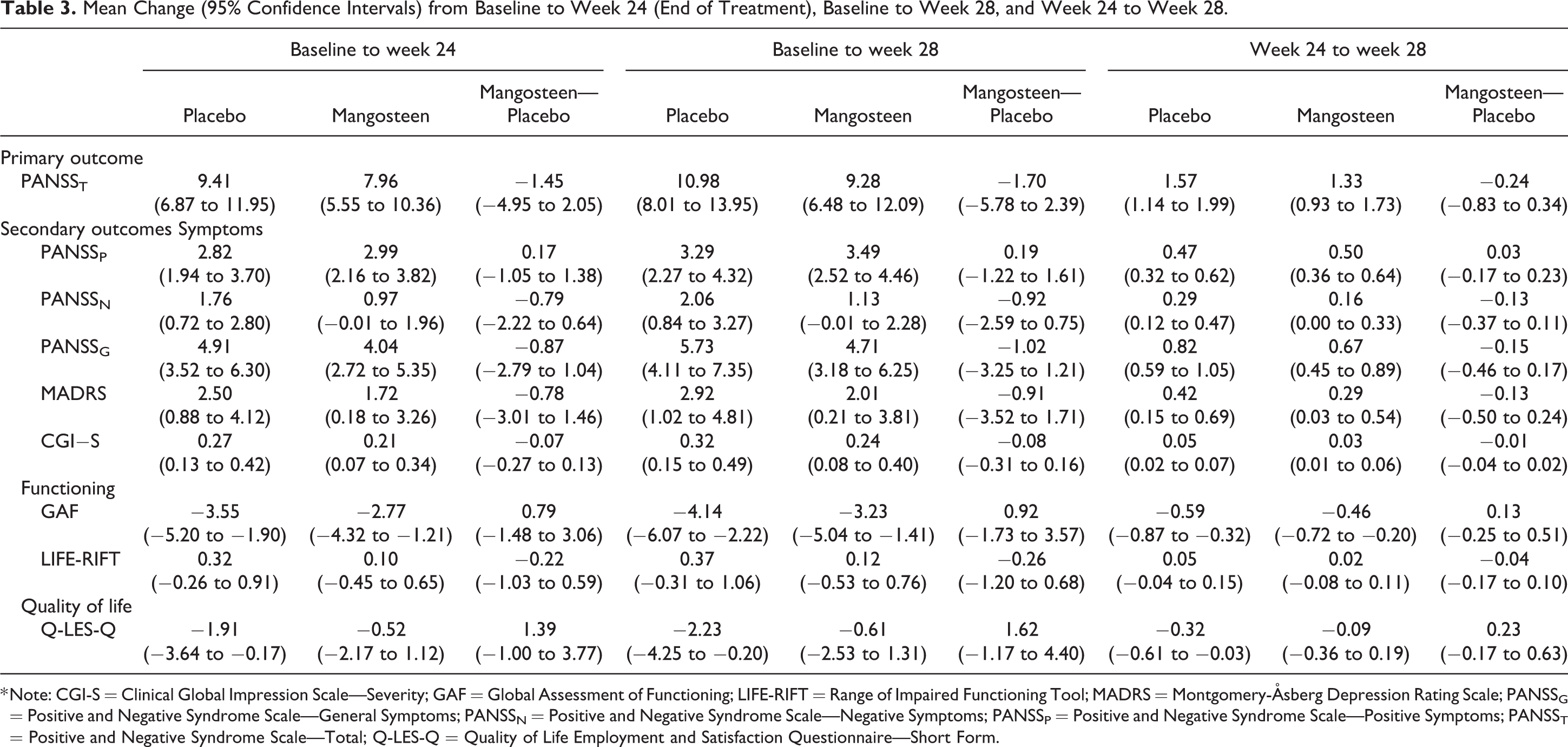
* Note: CGI-S = Clinical Global Impression Scale—Severity; GAF = Global Assessment of Functioning; LIFE-RIFT = Range of Impaired Functioning Tool; MADRS = Montgomery-Åsberg Depression Rating Scale; PANSSG = Positive and Negative Syndrome Scale—General Symptoms; PANSSN = Positive and Negative Syndrome Scale—Negative Symptoms; PANSSP = Positive and Negative Syndrome Scale—Positive Symptoms; PANSST = Positive and Negative Syndrome Scale—Total; Q-LES-Q = Quality of Life Employment and Satisfaction Questionnaire—Short Form.
Secondary Outcomes
For the secondary outcomes of PANSSP, PANSSG, and GAF, both group and visit were significantly associated with the outcomes; however, the interaction group by visit showed no significant different between the 2 groups (see Table 2). While the treatment group had significantly more severe symptoms and poorer functioning than placebo, both groups demonstrated comparable significant improvement over time. The groups did not significantly differ in terms of rates of change from baseline to week 24, baseline to week 28, and between weeks 24 and 28 (see Table 3).
For the PANSSN,, MADRS, CGI-S, and the Q-LES-Q, there were no significant group differences and all participants improved significantly over time. The interaction of group by visit was not significant for these variables. For the LIFE-RIFT, all participants showed significant improvement over time.
Sensitivity and Supplementary Analyses
Given the observed differences between the groups (i.e., in symptom severity and functioning), a range of adaptations to the LMM were adopted for sensitivity analyses. These included modeling change of scores from baseline and including or excluding baseline as a covariate and/or a baseline-by-time interaction. We also ran the basic LMM model separately for both those with a CGI-S baseline score in the 3 to 4 (mild to moderately ill) range and 5 to 6 (markedly to severely ill) range to determine whether there was an integration between group over time. The results of these models were very similar to the primary analyses, therefore the simplest model was presented.
Adherence and Safety Outcomes
Adherence and adverse events data were analyzed for all participants (including from participants who did not complete a postbaseline visit). Returned bottles of capsules (93% were returned) were counted and indicated that adherence was 94%. Regarding adverse events, there was no difference between groups on the rates of reported psychiatric, gastrointestinal, nervous system, or other adverse events, nor on SAEs or those adverse events deemed possibly related to the investigational product (see Supplementary Table 2). The treatment group reported more respiratory adverse events treatment n=24 (32.4%) versus placebo (n=13 (17.6%), χ2(1) = 4.36, P = 0.04); however none of these were deemed as potentially related to the investigational product by study investigators, with the majority of the reports being cold and flu (n = 32 of 37). Abnormal movements (AIMS) improved in both groups significantly over time, P < 0.01, and there was no significant interaction, P = 0.73. Average AIMS scores were <1 indicating a lack of observed tardive dyskinesia in the groups.
Discussion
Overall, we found that 24 weeks of adjunctive mangosteen pericarp (1,000 mg/day) was not superior to placebo for the treatment of schizophrenia and schizoaffective disorder. There were no significant group by time differences in the primary and secondary outcomes at the primary end point nor at the postdiscontinuation time point. The findings of the present study do not support the use of mangosteen pericarp for the adjunctive treatment of schizophrenia and schizoaffective disorder, despite promising preclinical and clinical findings. 14 Safety information indicated the only difference between groups was a higher rate of respiratory symptoms in the treatment group. These had not been rated as potentially treatment related and may be a type 1 error or associated with pre-existing between group differences (as seen in severity of illness measures).
Current treatment options for schizophrenia are of limited efficacy; they also have well-known, and significant, adverse effects impacting the cardiac, endocrine, and central nervous systems. 2 As such, balancing tolerability and efficacy can be challenging, with medication nonadherence one of the key contributors to symptom relapse. 2 Finding new effective primary or adjunctive pharmacotherapies that are also safe and tolerable remains an urgent research priority. Although mangosteen has been found to have a good safety profile, 14 this study did not find evidence of clinical efficacy in the recruited participant group. Despite the careful trial design, baseline differences were found between the groups, with the intervention group having greater severity of symptoms at baseline than placebo. Both groups significantly improved during the course of the trial and demonstrating additional benefits with mangosteen pericarp were difficult when the adjunctive placebo group had such large reductions in symptoms.
This study has a number of strengths, lending weight to our findings. It was a rigorously designed efficacy trial. The targeted sample size was achieved, ensuring power to detect differences between groups, particularly given the sample size allowed for greater attrition than seen in this study. Staff were well trained, while site was stratified to account for any potential differences in participant characteristics across the sites.
There are also a number of limitations. The study was powered to detect a medium, but not small, effect size. The heterogenous nature of schizophrenia makes treatment selection and subsequent episode recovery difficult. The inclusive trial recruitment criteria reflects real-world clinical practice where clinicians are biologically blind to the potential response of any given agent. This highlights the need for progress in biological psychiatry to target subgroups (i.e., a priori cohort enrichment) of people likely to respond based on their pathophysiological profile (e.g., inflammatory status, age, or body mass index) in combination with specific clinical factors (e.g., length of illness and severity of presentation; history of hospitalizations and suicide attempts). Future assessment of blood-based biological markers of oxidative stress and antioxidant defense as well as inflammation may be fruitful.
The dose or duration of the intervention may not have been sufficient to demonstrate an effect. Apart from the pilot work showing a large effect size at the dose and duration used in this study, clinical data to guide dosing of the product are lacking, and this could be explored in future research. Similarly, the method of preparation, or variation in the product given its fruit origin, may have impacted on the investigational product. In general, nonstandardization of the extraction process across studies to date is a methodological limitation. 14
Conclusions
These overall negative results do not support the use of Garcinia mangostana Linn. pericarp extract as an adjunctive treatment for schizophrenia or schizoaffective disorder, however future research could explore efficacy in targeted subgroups. Given the preclinical evidence of antidepressant and procognitive effects, 24,25 further investigations may also be warranted to determine whether there may be a role for mangosteen pericarp in the treatment of other psychiatric conditions such as bipolar disorder. As such, we currently have a clinical trial running investigating mangosteen pericarp for bipolar depression. 44 In the interests of improving the clinical outcomes of people living with schizophrenia, it is essential that high quality clinical trials of safe therapeutic agents continue to improve the health and psychosocial outcomes for people with psychotic disorders.
Supplemental Material
Supplemental Material, sj-doc-1-cpa-10.1177_0706743720982437 - Adjunctive Garcinia mangostana Linn. (Mangosteen) Pericarp for Schizophrenia: A 24-Week Double-blind, Randomized, Placebo Controlled Efficacy Trial: Péricarpe d’appoint Garcinia mangostana Linn (mangoustan) pour la schizophrénie : un essai d’efficacité de 24 semaines, à double insu, randomisé et contrôlé par placebo
Supplemental Material, sj-doc-1-cpa-10.1177_0706743720982437 for Adjunctive Garcinia mangostana Linn. (Mangosteen) Pericarp for Schizophrenia: A 24-Week Double-blind, Randomized, Placebo Controlled Efficacy Trial: Péricarpe d’appoint Garcinia mangostana Linn (mangoustan) pour la schizophrénie : un essai d’efficacité de 24 semaines, à double insu, randomisé et contrôlé par placebo by Alyna Turner, Andrea Baker, Olivia M. Dean, Adam J. Walker, Seetal Dodd, Susan M. Cotton, James G. Scott, Bianca E. Kavanagh, Melanie M. Ashton, Ellie Brown, John J. McGrath and Michael Berk in The Canadian Journal of Psychiatry
Supplemental Material
Supplemental Material, sj-docx-1-cpa-10.1177_0706743720982437 - Adjunctive Garcinia mangostana Linn. (Mangosteen) Pericarp for Schizophrenia: A 24-Week Double-blind, Randomized, Placebo Controlled Efficacy Trial: Péricarpe d’appoint Garcinia mangostana Linn (mangoustan) pour la schizophrénie : un essai d’efficacité de 24 semaines, à double insu, randomisé et contrôlé par placebo
Supplemental Material, sj-docx-1-cpa-10.1177_0706743720982437 for Adjunctive Garcinia mangostana Linn. (Mangosteen) Pericarp for Schizophrenia: A 24-Week Double-blind, Randomized, Placebo Controlled Efficacy Trial: Péricarpe d’appoint Garcinia mangostana Linn (mangoustan) pour la schizophrénie : un essai d’efficacité de 24 semaines, à double insu, randomisé et contrôlé par placebo by Alyna Turner, Andrea Baker, Olivia M. Dean, Adam J. Walker, Seetal Dodd, Susan M. Cotton, James G. Scott, Bianca E. Kavanagh, Melanie M. Ashton, Ellie Brown, John J. McGrath and Michael Berk in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Alyna Turner and Andrea Baker are joint first authors. John J. McGrath and Michael Berk are the joint senior authors.
Acknowledgments
All authors gratefully acknowledge their support. We wish to thank the participants who took part in the study, the health services including Barwon Health, the West Moreton Hospital and Health Service, the Metro North Mental Health Service, Metro South Mental Health Service, Gold Coast Mental Health Service, the Melbourne Clinic, and the Community and Research Network.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A.T. AT reports no disclosures. A.B. reports no disclosures. O.M.D. is a R.D. Wright NHMRC Biomedical Career Development Fellow (APP1145634) and has received grant support from the Brain and Behavior Foundation, Simons Autism Foundation, Stanley Medical Research Institute, Deakin University, Lilly, NHMRC, and ASBDD/Servier. She has also received in kind support from BioMedica Nutraceuticals, NutritionCare, and Bioceuticals. A.J.W. has previously received grant/research support from a Trisno Family Fellowship and Deakin University. S.D. has received grant support from Stanley Medical Research Institute. S.M.C. has received grant support from the NHMRC, the Stanley Medical Research Institute, BeyondBlue, Movember, The University of Melbourne, Australian Catholic University, ARHRF, and Mental Illness Research Fund (Victoria Department of Human Services). She is supported by a NHMRC Senior Research Fellowship (APP1136344). J.G.S. is supported by a National Health and Medical Research Council Practitioner Fellowship Grant (APP1105807), has been a speaker for Janssen Cilag, Lundbeck, Servier, and Shire Pharmaceuticals, and served as a consultant to Janssen Cilag, Lundbeck and Roche. B.E.K. has received grant/research support from the Australian Government Research Training Program, Deakin University, Ian Scott Mental Health PhD Scholarship, Australian Rotary Health, and the International Society for the Study of Personality Disorders. M.M.A. has received grant/research support from Deakin University, Australasian Society for Bipolar Depressive Disorders, Lundbeck, Australian Rotary Health, Ian Parker Bipolar Research Fund, Cooperative Research Centre for Mental Health and PDG Geoff, and Betty Betts Award from Rotary Club of Geelong. E.B. has received grant/research support from the University of Melbourne, the Australian Department of Health, Western Victoria PHN, and the University of the West of England, United Kingdom. M.B. has received grant/research support from the NIH, Cooperative Research Centre, Simons Autism Foundation, Cancer Council of Victoria, Stanley Medical Research Foundation, Medical Benefits Fund, National Health and Medical Research Council Senior Principal Research Fellowship (1059660 and 1156072), Medical Research Futures Fund, Beyond Blue, Rotary Health, A2 milk company, Meat and Livestock Board, Woolworths, Avant and the Harry Windsor Foundation; has been a speaker for Astra Zeneca, Lundbeck, Merck, Pfizer; and served as a consultant to Allergan, Astra Zeneca, Bioadvantex, Bionomics, Collaborative Medicinal Development, Lundbeck Merck, Pfizer, and Servier—all unrelated to this work. This study was funded by the Stanley Medical Research Institute, MD, USA. J.J.M. is supported by grant APP1056929 from the John Cade Fellowship from the National Health and Medical Research Council.
ORCID iD
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
