Abstract
Background:
Randomized controlled clinical trials that have investigated minocycline as an adjunctive treatment for major depressive disorder have proved promising. Data from two studies were pooled to evaluate more definitively whether the addition of minocycline to standard treatment for major depressive disorder leads to an improvement of depressive symptoms when compared with placebo.
Methods:
Both studies were multi-site, double-blinded, placebo-controlled trials of minocycline 200 mg/day added to treatment as usual during a 12-week period. The primary outcome measure was change in depressive symptoms (Montgomery–Asberg Depression Rating Scale in Dean et al. and Hamilton Depression Rating Scale in Husain et al.). Secondary outcomes were change in depression severity (Montgomery–Asberg Depression Rating Scale for Dean et al. and 9-item Patient Health Questionnaire in Husain et al.), anxiety severity (Hamilton Anxiety Rating Scale in Dean et al. and Generalized Anxiety Disorder 7-item scale in Husain et al.) and functional status, which were also evaluated as potential mediators on the primary outcome.
Results:
A total of 112 participants were included in the pooled data (Dean et al., n = 71; Husain et al., n = 41). A significant change from baseline to week 12 was noted in depressive symptoms – differential change (Placebo vs Minocycline): 9.0, 95% confidence interval = [4.2, 13.9], Cohen’s D (95% confidence interval): 0.71 [0.29, 1.14], p < 0.001 – anxiety severity – differential change (Placebo vs Minocycline): 0.38, confidence interval = [0.00, 0.75], Cohen’s D (95% confidence interval): 0.41 [0.00, 0.82], p = 0.050) and functional status – differential change (Placebo vs Minocycline): 1.0, 95% confidence interval = [0.4, 1.5], Cohen’s D (95% confidence interval): 0.76 [0.34, 1.19], p = 0.001). Duration of illness, current use of benzodiazepine and pain medication were identified as moderators, whereas functional status as a mediator/predictor.
Conclusion:
The improvement of depressive symptoms, anxiety severity and functional status is promising and suggests that minocycline has potential as an adjunctive treatment for major depressive disorder. However, further studies are warranted to confirm therapeutic effects of minocycline in major depressive disorder.
Trial registrations:
NCT02263872, registered October 2014, and ACTRN12612000283875, registered March 2012.
Keywords
Major depressive disorder (MDD) is the leading cause of disability worldwide, with significant morbidity, mortality and social cost, with a 12-month and lifetime prevalence ranging, respectively, from 5.5% to 5.9% and 11.1% to 14.6% (Bromet et al., 2011). Despite marked prevalence and disease burden, a lack of knowledge about the aetiology and pathophysiology of MDD remains (Malhi and Mann, 2018), contributing to poorer treatment outcomes (Otte et al., 2016). According to the STAR*D study, remission rates of patients with MDD are low, reaching 50% after two treatment steps, while the likelihood of relapse is high, ranging from 40% to 70% (Warden et al., 2007). Likewise, a previous network meta-analysis evaluating pharmacological treatments for MDD patients in a primary health care setting reported a small effect size (ES) for antidepressants relative to placebo (Linde et al., 2015). Therefore, there is a great need for identification of new pharmacological treatment approaches.
Promising pre-clinical and clinical investigations have implicated inflammatory processes in mood disorders, in particular MDD (Leonard and Maes, 2012; Maes, 1995; Rosenblat et al., 2014). A significant number of individuals with MDD subgroup present higher levels of pro-inflammatory and immune-regulatory cytokines coupled with changes in diverse acute-phase proteins, which play a role in the onset and recurrence of depressive episodes and staging as well (Köhler et al., 2017; Maes and Carvalho, 2018). The current hypothesis suggests that depression arises from activated immune-inflammatory and oxidative pathways, which are insufficient by the compensatory immune-regulatory system (CIRS), leading to neuroprogression. This suggests that new treatments of depression should attenuate the activated immune, oxidative and neuroprogressive pathways and/or reinforce the CIRS including stimulating neuroprotective processes (Leonard and Maes, 2012; Maes and Carvalho, 2018). Due to this association, there is mounting evidence from recent clinical trials and meta-analysis suggesting a role for anti-inflammatory medications in treating depressive symptoms (Fond et al., 2014; Husain et al., 2017b; Köhler et al., 2017; Rosenblat and McIntyre, 2018).
Minocycline is a broad-spectrum tetracycline antibiotic with putative neuroprotective properties through anti-inflammatory and antioxidant effects as well as neuroprotective mechanisms including enhanced neurogenesis (Miao et al., 2018; Soczynska et al., 2012; Yong et al., 2004). This medication exerts effects in multiple interacting systems involved in the pathophysiology of mood disorders and may, therefore, reverse depressive states associated with activated immune-inflammatory, oxidative and neuroprogressive pathways (Dean et al., 2012). Minocycline is anti-inflammatory, by suppressing microglial activation, and inhibition of subsequent inflammatory mediator synthesis (Kitayama et al., 2011). It is also an antioxidant, by suppressing free radical generation and scavenging free radicals (Kraus et al., 2005). Minocycline promotes neurogenesis and is anti-apoptotic, through caspase-dependent and independent pathways, decreasing the release of apoptotic agents from mitochondria and raising the release of anti-apoptotic factors (Plane et al., 2010). Finally, minocycline modulates glutamate transmission and prevents cell death following NMDA treatment (Soczynska et al., 2012).
Despite these promising therapeutic properties, to date, few trials evaluating the potential antidepressant effects of minocycline have been conducted. Three randomized control trials (RCTs), two open-label studies and a case report were published, and 11 studies were ongoing, from which the three RCTs were included in their meta-analysis (Rosenblat and McIntyre, 2018). A large ES (SMD = −0.78) with moderate heterogeneity of the pooled sample (I2 = 62%) was found in the meta-analysis. In general, the medication was well tolerated by participants treated with minocycline in comparison with placebo for MDD participants. Recently, from those 11 ongoing studies, an RCT and an open-label study were published later with similar outcomes (Murrough et al., 2018; Savitz et al., 2018). Both studies concluded that minocycline might be an efficacious adjunctive treatment for bipolar depression, reducing depressive symptoms. Notably, there were sample size limitations in studies either included in the meta-analysis or published later.
In lieu of these sample size limitations, as well as the small number of RCTs evaluating the antidepressant effects of minocycline in MDD, the present pooled analysis was proposed with two studies with similar responses included in the meta-analysis (Dean et al., 2017; Husain et al., 2017a). The choice for a pooled analysis was selected over traditional meta-analysis to increase statistical power for both subgroup analysis and detection of treatment effects through large study samples (van der Steen et al., 2008). Similar trial designs allow direct pooling of data allowing for detailed analysis of subgroups. In summary, minocycline led to improvements in depressive symptoms in both studies, but significant outcomes were found only in Husain et al. (2017a). The primary hypothesis of both original studies was that adjunctive treatment with minocycline for MDD would lead to an improvement of depressive symptoms at the end of a 12-week treatment period. Secondary hypotheses were that 12 weeks of treatment would reduce the severity of both depression and anxiety symptoms, clinician-rated global impression, and functioning in the group treated with minocycline compared with a placebo.
Given the indicative findings from these studies and the clinical need to have robust findings supporting effective antidepressant treatment to govern therapeutic choice, pooling data from these both RCTs would be useful. Thus, this study aimed to evaluate whether the addition of minocycline to standard treatment, as conducted in both studies, would lead to an improvement of depressive symptoms when compared with placebo.
Overview
The two studies were multi-site, double-blinded, placebo-controlled trials evaluating 200 mg/day of minocycline added to treatment as usual (TAU) for MDD. The study led by Dean et al. (2017) was conducted at (1) Barwon Health and The Geelong Clinic and (2) The Melbourne Clinic in Victoria, Australia, in collaboration with Deakin University and The University of Melbourne. A third trial site was located in Bangkok, Thailand, in collaboration with Chulalongkorn University. The trial period was from August 2013 to August 2015. The study was approved by the Human Research Ethics Committee (HREC; #11/146) and registered on the Australian and New Zealand Clinical Trials Registry (ACTRN12612000283875). The complete study protocol (Dean et al., 2014) and the original results have been published (Dean et al., 2017).
The study led by Husain et al. (2017a) was conducted at Abbasi Shaheed Hospital, Karwan-e-Hayat Hospital, Civil Hospital, and the Institute of Behavioural Sciences in Karachi, Pakistan. The trial period was from October 2014 to March 2016. The study was approved by the Institutional Review Board (IRB) of the Karachi Medical and Dental College and Dow University of Health Sciences, Pakistan, and registered on Clinicaltrials.gov (NCT02263872). The complete study protocol (Husain et al., 2015), as well as the original results, has been published (Husain et al., 2017a). Participants from both studies provided written informed consent prior to commencing the study.
Participants
The current pooled data included a total of 112 participants; 71 recruited by Dean et al. (2017), and 41 recruited by Husain et al. (2017a). Both RCTs had similar inclusion and exclusion criteria. Participants were eligible to be included if they were (1) aged 18 years or over; (2) had the capacity and willing to consent to the study; (3) had a current diagnosis and treatment for MDD according to DSM-IV-TR and screened using the Mini International Neuropsychiatric Interview (MINI) (and at least a score of 25 in Montgomery–Asberg Depression Rating Scale (MADRS) in Dean et al. (2017) study); (4) if female, of childbearing potential and sexually active, had been using effective contraceptive methods other than contraceptive pills (contraindicated due to drug interaction); and (5) they had a stable treatment TAU period previous to baseline (at least 2 weeks in Dean et al. (2014) and 4 weeks, or 6 weeks for fluoxetine in Husain et al. (2015)).
Both RCTs also had similar exclusion criteria, such as (1) presence of relevant or suspected medical illness or systemic disorder, (2) prior history of intolerance or allergy to any tetracycline, (3) concomitant penicillin therapy, (4) concomitant anticoagulant therapy and (5) pregnancy or breastfeeding. The study led by Dean et al. (2017) had the additional following exclusion criteria: (1) current diagnosis for type I or II bipolar disorder according to DSM-IV-TR, (2) three or more failed adequate trials of antidepressant therapy or electroconvulsive therapy for the current MDD, (3) prior tetracycline use within 2 months of the treatment, (4) treatment with more than 5 mg beta-carotene or more than 300 μg retinol equivalent, (5) treatment with isotretinoin or etretinate and (6) current enrolment in another clinical study. Husain et al. (2017a) had the additional exclusion criteria: (1) presence of seizure disorder, (2) current use of valproic acid at baseline, (3) diagnosis of substance abuse disorder (except caffeine and nicotine) or dependence within last 3 months according to DSM-IV-TR and (4) the presence of primary psychotic disorder.
Participants were withdrawn from both studies if they (1) had poor compliance (defined by Dean et al. (2014) as seven consecutive days of medication discontinuation), (2) ceased effective contraceptive methods, (3) became pregnant or (4) had adverse effects during the trial period that required withdrawal from the trial.
Randomization, concealment and investigational product
Participants from both trials were assigned randomly and consecutively to treatment with minocycline or placebo. Randomization was conducted according to the Consolidated Standards of Reporting Trials (CONSORT) and outlined in the original publications (Dean et al., 2017; Husain et al., 2017a). There was no further stratification in both studies.
Participants from Dean et al. (2017) were instructed to take one capsule (100 mg), twice a day, with food during all 12 weeks trial, while participants from Husain et al. (2017a) had started at a dose of 100 mg within the first 2 weeks and then, until week 12, the dose was increased to 200 mg once a day.
Outcome measures
Demographic data included in this study were age, gender, height, weight and body mass index (BMI), duration of illness since diagnosis and current medication at baseline. Adverse effects from both studies were also recorded at each visit using specific scales designed on both studies.
The primary outcome for both studies was the change from baseline to week 12 on depression, with ratings made at baseline, weeks 2, 4, 8 and 12. However, the primary measures used differed between studies. Dean et al. (2017) used the MADRS, while Husain et al. (2017a) used the 17-item Hamilton Depression Rating Scale (HAM-D). Notably, the MADRS and HAM-D are highly correlated, and as such, MADRS scores were transformed to align with HAM-D scores in accordance with Leucht and colleagues’ equipercentile linking analysis (Leucht et al., 2018).
Secondary outcomes for anxiety severity also differed between studies. For anxiety symptoms, Dean et al. (2017) used the Hamilton Anxiety Rating Scale (HAM-A), a questionnaire developed to measure the severity of anxiety symptoms, and Husain et al. (2017a) used the Generalized Anxiety Disorder 7-item scale (GAD-7). There is a high correlation between both measurements (r = 0.852) (Garcia-Campayo et al., 2010). Husain et al. (2017a) also used an additional depression tool to measure severity other than the primary outcome (9-item Patient Health Questionnaire, PHQ-9). MADRS is a standard tool for assessment of depression severity, also highly correlated with the PHQ-9 (r = 0.794; Hawley et al., 2013) and was used to provide that variable in Dean et al. (2017). Composite scores were calculated for the severity of both depression and anxiety to harmonize scores from both studies by averaging the Z scores for each instrument in each study. Global clinical and functional status were also collected with Clinical Global Impression–Severity (CGI-S) and Clinical Global Impression–Improvement (CGI-I) in both studies at baseline (CGI-S only) and weeks 2, 4, 8 and 12 (CGI-I).
Remission and response rates in primary outcomes were also assessed at the end of the study. Remission was defined as HAM-D score equal or lower than 7 (or its equivalent) at week 12, while of treatment response was defined as a change in HAM-D score of 50% or more at 12 weeks in comparison with baseline.
Potential modifiers were also assessed as moderators and mediators in the treatment. The choice of potential modifiers was based on evidence in the literature about their role of each of them in depression and its treatment. Thus, clinical and demographical characteristics were included as possible moderators, such as age, duration of illness (i.e. chronicity), clinical and functional status at baseline (CGI-S), gender and current medication, whereas the severity of anxiety and depression from baseline to week 12, as well as improvement in clinical and functional status (CGI-I) from week 2 to week 12, was included as mediators/predictors.
Statistical analysis
All outcomes from minocycline and placebo groups from both studies were pooled into one data set. Main primary and secondary outcomes analyses were based on all randomized participants with at least one follow-up measurement (modified intention to treat, ITT).
All continuous variables were explored prior to analysis. Between-group comparisons were performed by independent t test for normally distributed and by Mann–Whitney test for non-normally distributed variables. Dichotomous variables, such as gender, current medication, and side effects, were assessed by chi-square test or Fisher’s exact test.
The intervention effects were estimated using generalized estimating equations (GEEs). To assess the impact of the intervention on primary or secondary outcomes, visit by treatment two-way interactions was examined in a model that included the fixed categorical effects of treatment, visit and treatment-by-visit interaction. An unstructured covariance structure was used for modelling within-participant autocorrelation and a robust sandwich approach was used for model standard error estimation. Differential changes from baseline to week 12, estimated from the two-way interaction of visit by treatment in the GEEs, were used to test between-group differences. ESs and their 95% confidence intervals (CIs) were calculated using Cohen’s D, and its effects being interpreted as small (0.20–0.49), medium (0.50–0.79) or large (⩾0.80) (Cohen, 2013). It is important to highlight that ES was the primary focus for the main outcome comparison in the present pooled analysis as the main outcome scales, described above, were different in the two RCTs.
To evaluate heterogeneity between studies, the study site by treatment two-way interactions were also examined in a GEE model including study site, visit and treatment and their interaction, as well as the main effect of the study site.
Logistic regressions were carried out to compare remission and response rates between the intervention and control groups from the pooled data. Odds ratios (ORs) with 95% CIs were reported.
Possible moderators and mediators were also evaluated. To model, continuous ordinal covariates were centred around their respective medians, and binary factors were coded as −0.5 and 0.5. Finally, GEEs similar to a model implemented for the main analyses were performed to evaluate moderator/mediator of treatment effect. Potential moderator/mediator was evaluated one at the time through separate GEE models. The GEE models included the fixed categorical effects of treatment, the logarithm of visit time point as a covariate (i.e. log(time), the potential moderator/mediator as a covariate, all possible two-way interactions and the three-way interaction of treatment, log(time) and the candidate moderator or mediator). The three-way interaction examines the effect of each causal predictor of treatment on the outcome measure, across time in the study, for each treatment time (Kraemer et al., 2001). As such the three-way interaction was primarily examined to investigate the role of potential moderator/mediators. The p values for all overall three-way interactions were reported using a model-based nested χ2 test, alongside corresponding beta coefficients that measured the causal impact of the potential moderator/mediator through intervention on outcome and 95% CIs were reported. For descriptive purpose, data were split based on the median of outcome percent change (absolute change from baseline divided by baseline value) and summary descriptive for both mediator and moderator was reported. The analysis was conducted using SPSS Statistics v23 (IBM Corp, 2015). The statistical significance level used was ⩽0.05, two-tailed.
Results
Demographics and baseline comparison of groups
A total of 231 potential participants were screened in both studies, of which 112 were randomized to receive either minocycline or placebo. All participants included completed at least one post-baseline visit. From those participants randomized, 91 participants completed the 12-week treatment. The CONSORT flow chart illustrates the participant flow from both trials and how they were pooled in this study (Figure 1).
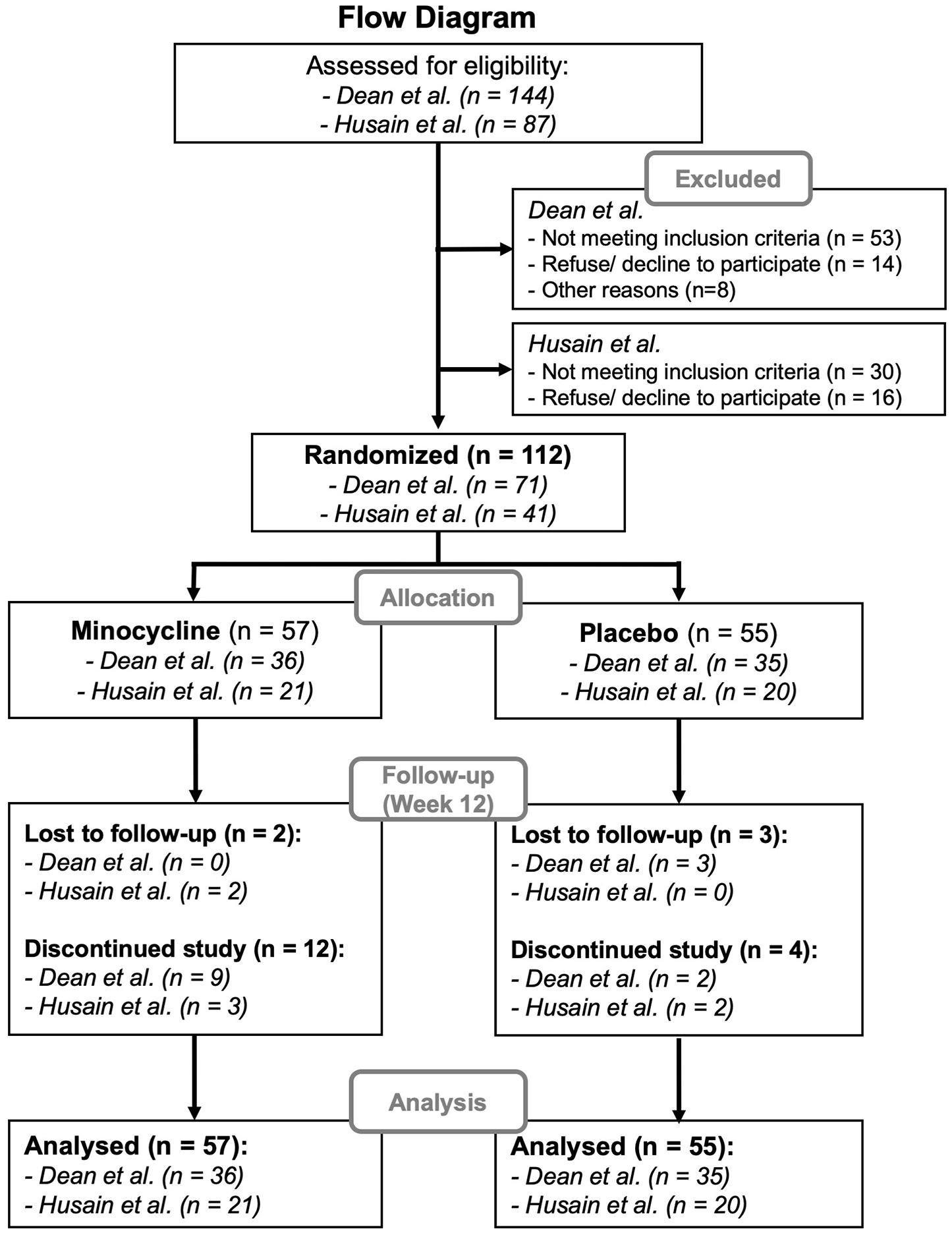
CONSORT diagram.
Participants did not differ significantly on any baseline demographics, clinical characteristics or baseline medication. The mean duration of illness was 9.0 (±10.1) years and 11.2 (±12.4) years in placebo and minocycline arms from the pooled data, respectively, suggesting chronic MDD. No significant difference was found between both arms for BMI, weight and current psychiatric medication (Table 1).
Baseline characteristics of participants.
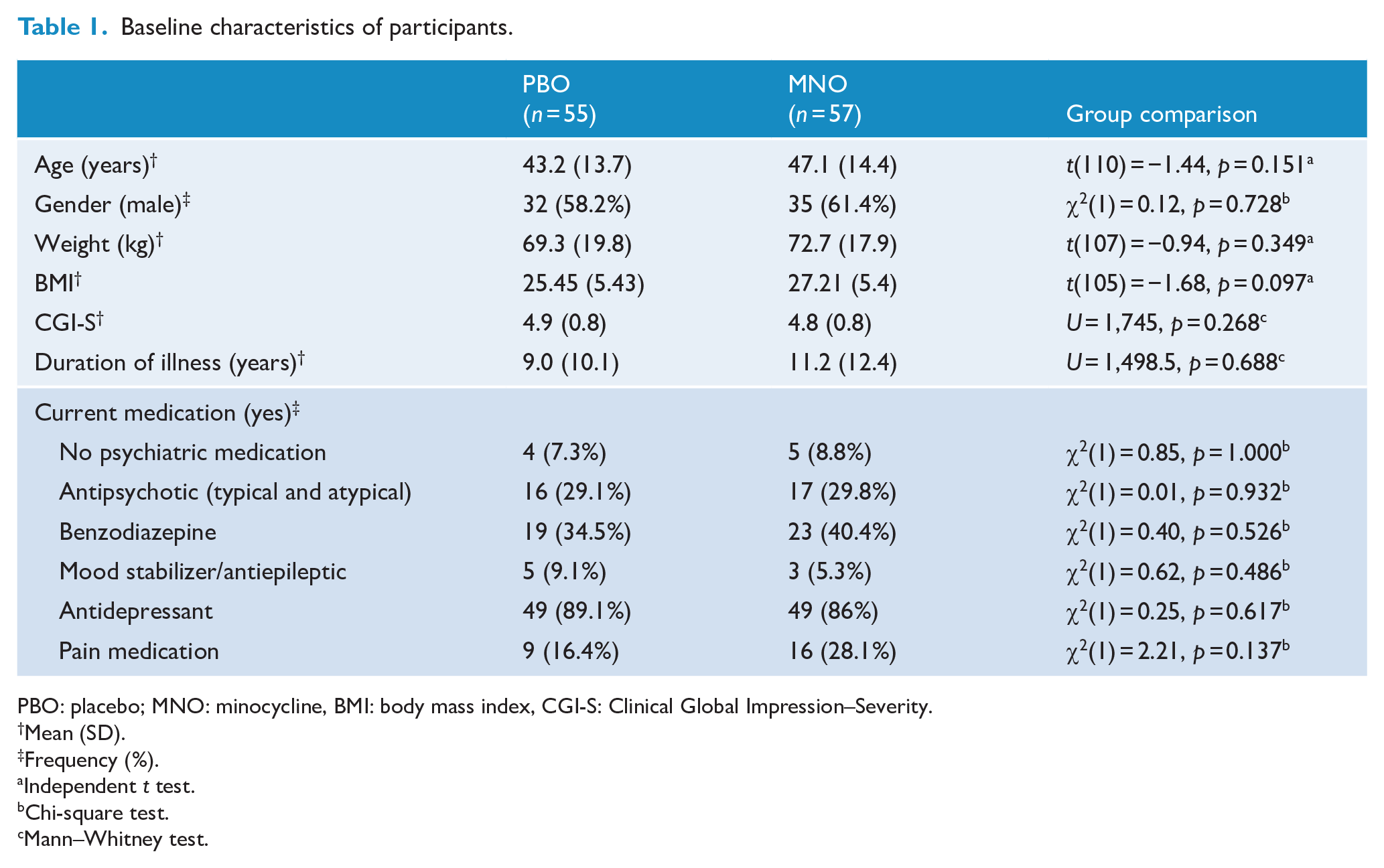
PBO: placebo; MNO: minocycline, BMI: body mass index, CGI-S: Clinical Global Impression–Severity.
Mean (SD).
Frequency (%).
Independent t test.
Chi-square test.
Mann–Whitney test.
Participants had moderate to severe depression with baseline primary outcome of 27.4 (±7.6) in placebo and 27.9 (±8.8) in the minocycline group. No difference was found between both arms, t(110) = 0.285, p = 0.776. In regard to secondary outcomes, mean score at baseline for depression severity was 0.12 (±0.93) in the placebo and −0.10 (± 1.05) in minocycline group, and for anxiety severity was 0.01 (±1.1) in placebo and 0.01 (±0.9) in minocycline group. Both depression and anxiety severity scores were similar at baseline with no difference between in both arms of the pooled data, respectively, t(110) = −1.14, p = 0.256 and t(110) = −0.08, p = 0.934.
Outcome measures
Based on GEE models (Table 2), there was a significant change in the primary outcome, depression, at week 12, between the placebo and minocycline groups – differential change (PBO vs MNO) from baseline: 9.0, confidence interval [CI] = [4.2, 13.9]; ES (95% CI) = 0.71 [0.29, 1.14], p < 0.001. Differences between groups were also identified in the middle point visits, 4-week visit – differential change (PBO vs MNO) from baseline: 4.6, CI = [1.2, 8.1], ES (CI) = 0.46 [0.06, 0.88], p = 0.009 – and 8-week visit – differential change (PBO vs MNO) from baseline: 6.0; CI = [1.9, 10.1]; ES (CI) = 0.62 [1.9, 10.1], p = 0.004 (Figure 2(a)).
Primary and secondary outcomes based in the Generalized Estimated Model (GEE) from baseline to week 12.
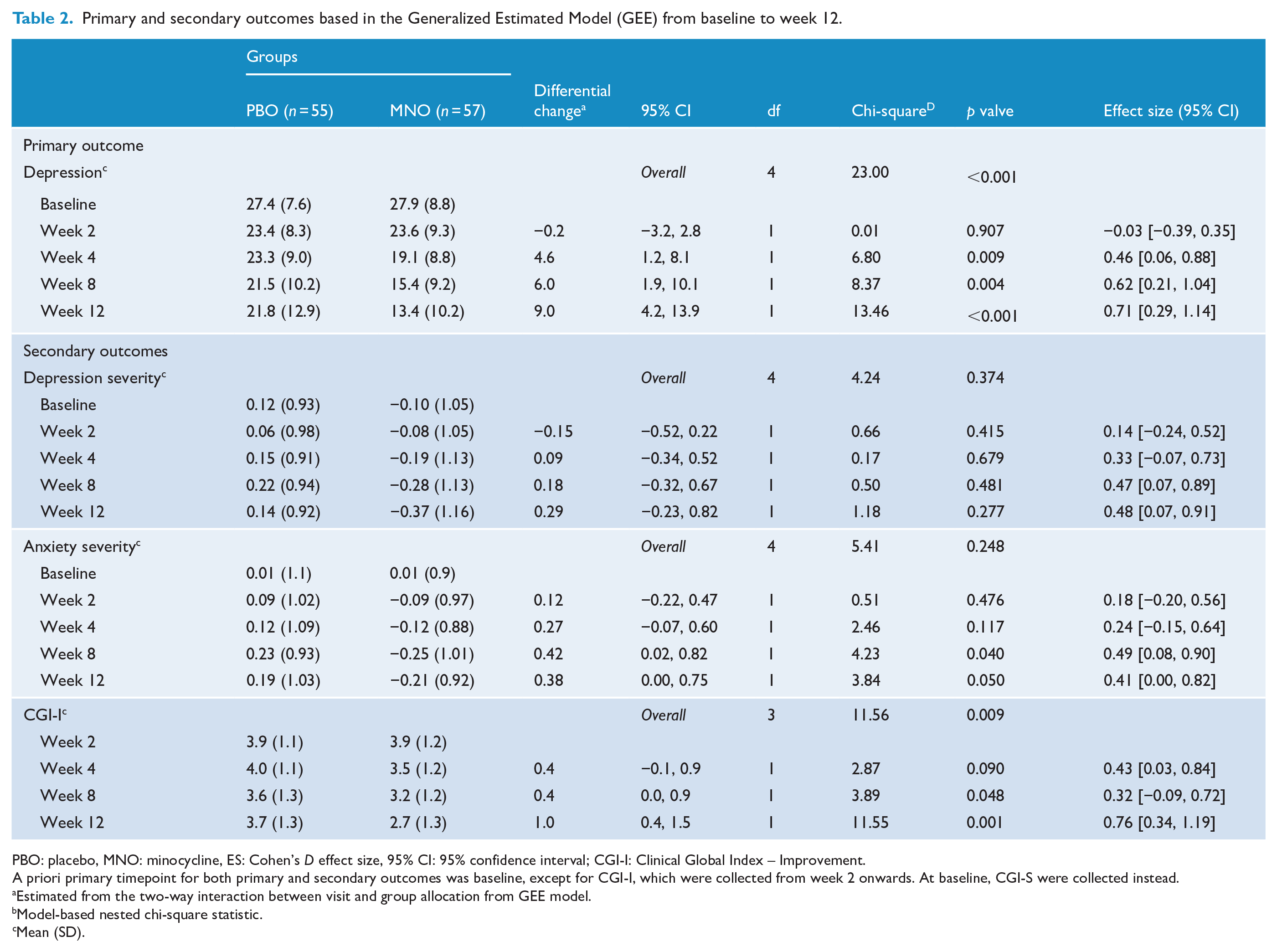
PBO: placebo, MNO: minocycline, ES: Cohen’s D effect size, 95% CI: 95% confidence interval; CGI-I: Clinical Global Index – Improvement.
A priori primary timepoint for both primary and secondary outcomes was baseline, except for CGI-I, which were collected from week 2 onwards. At baseline, CGI-S were collected instead.
Estimated from the two-way interaction between visit and group allocation from GEE model.
Model-based nested chi-square statistic.
Mean (SD).
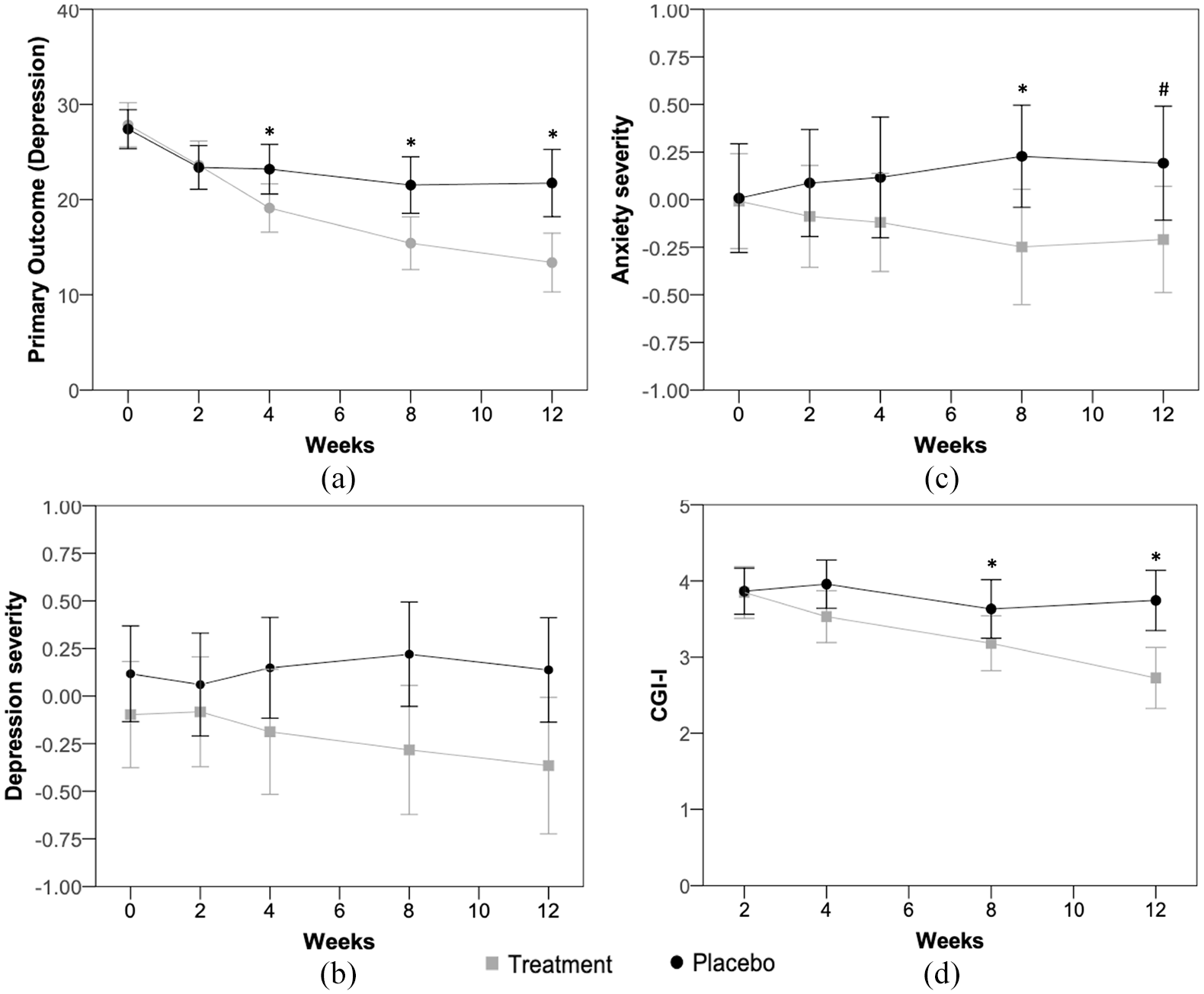
Estimated mean trajectories and their confidence intervals (95% CI) in primary and secondary outcomes across 12-week treatment. (a) Mean trajectory and its confidence interval (95% CI) in primary outcome (depression) from week 0 to week 12. (b) Mean trajectory and its confidence interval (95% CI) in secondary outcome (depression severity) from week 0 to week 12. (c) Mean trajectory and its confidence interval (95% CI) in secondary outcome (anxiety severity) from week 0 to week 12. (d) Mean trajectory and its confidence interval (95% CI) in secondary outcome (Clinical Global Index – Improvement) from week 2 to week 12.
Regarding secondary outcomes, no significant differences were found across follow-up visits or at the end of treatment for depression severity (Figure 2(b)). Anxiety severity outcomes showed significant between-group differential change (PBO vs MNO) from baseline of 0.38, CI = [0.00, 0.75]; ES (CI) = 0.41 [0.00, 0.82], p = 0.050. It was also significant at 8 weeks; differential change (PBO vs MNO) from baseline of 0.42, CI = [0.02, 0.82]; ES (CI) = 0.49 [0.08, 0.90], p = 0.040. No between-group significant differences were noted either at 2 or 4 weeks visits (Figure 2(c)).
No differences were noted at baseline for CGI-S between groups, t(110) = −0.93, p = 0.754 (Table 1). There was a significant improvement in the CGI-I from week 8 onwards, maintained until week 12 – between-group differential change (PBO vs MNO) from baseline: 1.0, CI = [0.4, 1.5]; ES (CI) = 0.76 [0.34, 1.19], p = 0.001 (Table 2 and Figure 2(d)).
Remission and response
According to definitions of treatment response (HAM-D score reduction of >50%), 56.8% of the minocycline group responded to treatment in comparison with 23.4% in the placebo group. Minocycline-treated participants were 2.14 times more likely to respond to minocycline than placebo-treated – χ2(1) = 10.6; OR (CI) = 2.14 [1.26, 3.63], p = 0.001. There was also a greater proportion of participants that showed remission (HAM-D scores equal to or lower than 7) in the minocycline group (31.8%), compared with the placebo arm (10.6%). Participants were approximately 2.2 times more likely to reach remission of symptoms if they received minocycline in comparison with those who received placebo – χ2(1) = 6.17, OR (CI) = 2.22 [1.02, 4.82], p = 0.013. There was no significant difference in the total number of adverse events reported by participants between groups (Table 3).
Frequency of side effects.
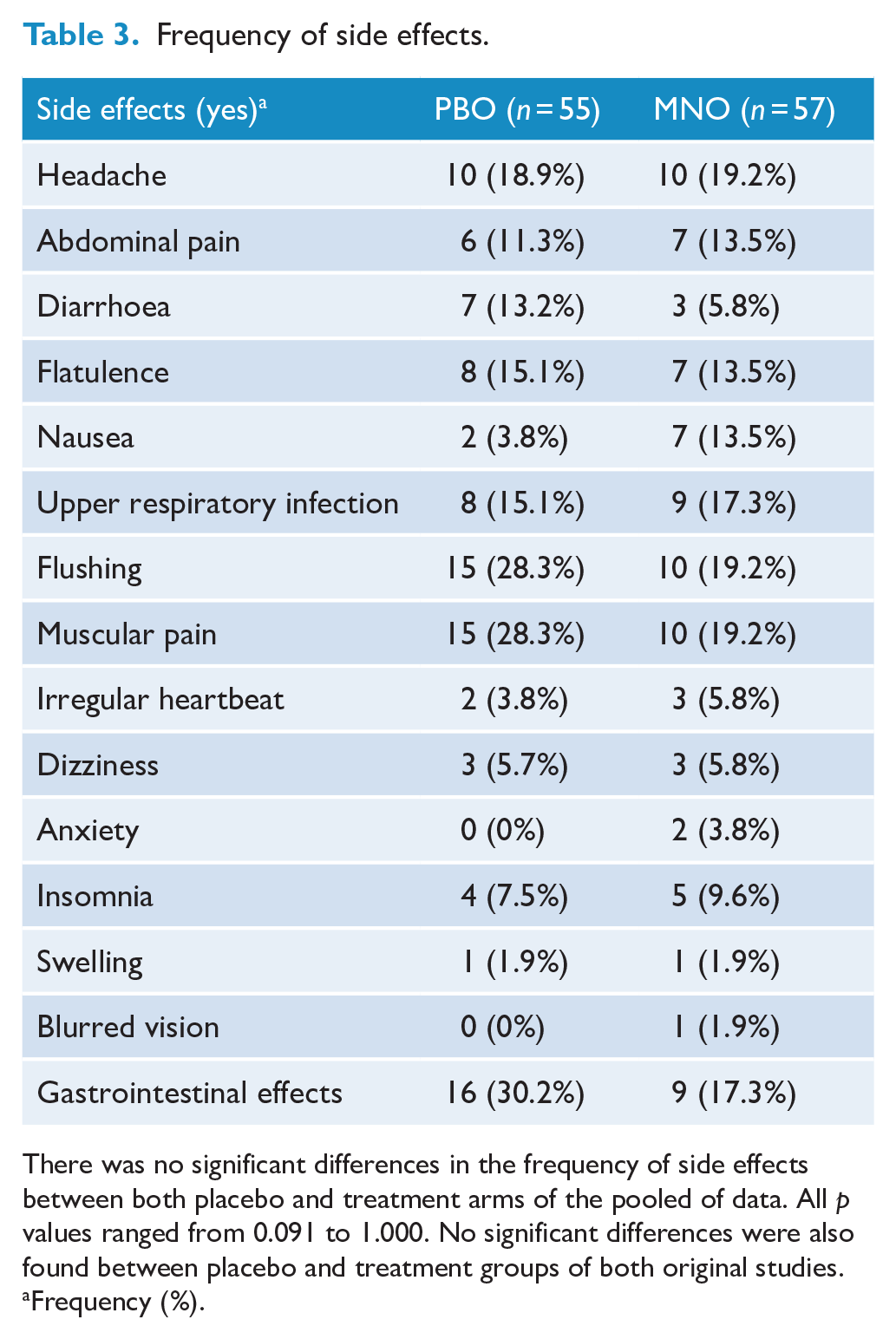
There was no significant differences in the frequency of side effects between both placebo and treatment arms of the pooled of data. All p values ranged from 0.091 to 1.000. No significant differences were also found between placebo and treatment groups of both original studies.
Frequency (%).
Moderators and mediators analyses of primary outcome from baseline
As illustrated in Table 4, a longer duration of illness – χ2 = 10.14, ß (CI) = −0.3 [−0.49, −0.12], p = 0.001 – and current use of pain medication – χ2 = 6.62, ß (CI) = −7.57 [−13.34, −1.81], p = 0.010 – were treatment modifiers, indicating that participants with longer duration of illness and current use of pain medication presented larger reductions in depression symptoms throughout the treatment. In addition to the above GEE, which detected a larger reduction in the minocycline arm in comparison with the placebo arm, a larger reduction in depression was observed in minocycline compared with placebo in the subgroup with longer duration of illness. Conversely, current non-use of benzodiazepines was correlated with larger reductions of depression differential change in minocycline group compared with placebo – χ2 = 5.47, ß (CI) = 6.56 [1.06, 12.06], p = 0.019. There was a trend towards a moderating effect of age – χ2 = 3.57, ß (CI) = −0.15 [−0.31, 0.001], p = 0.059 – indicating that older participants in the minocycline group presented a larger reduction in depression (i.e. from baseline to week 12) when compared with placebo. No other significant modifier effects of clinical and demographical data were found (Table 4).
Examining effect modifiers of treatment effect on study primary outcome.
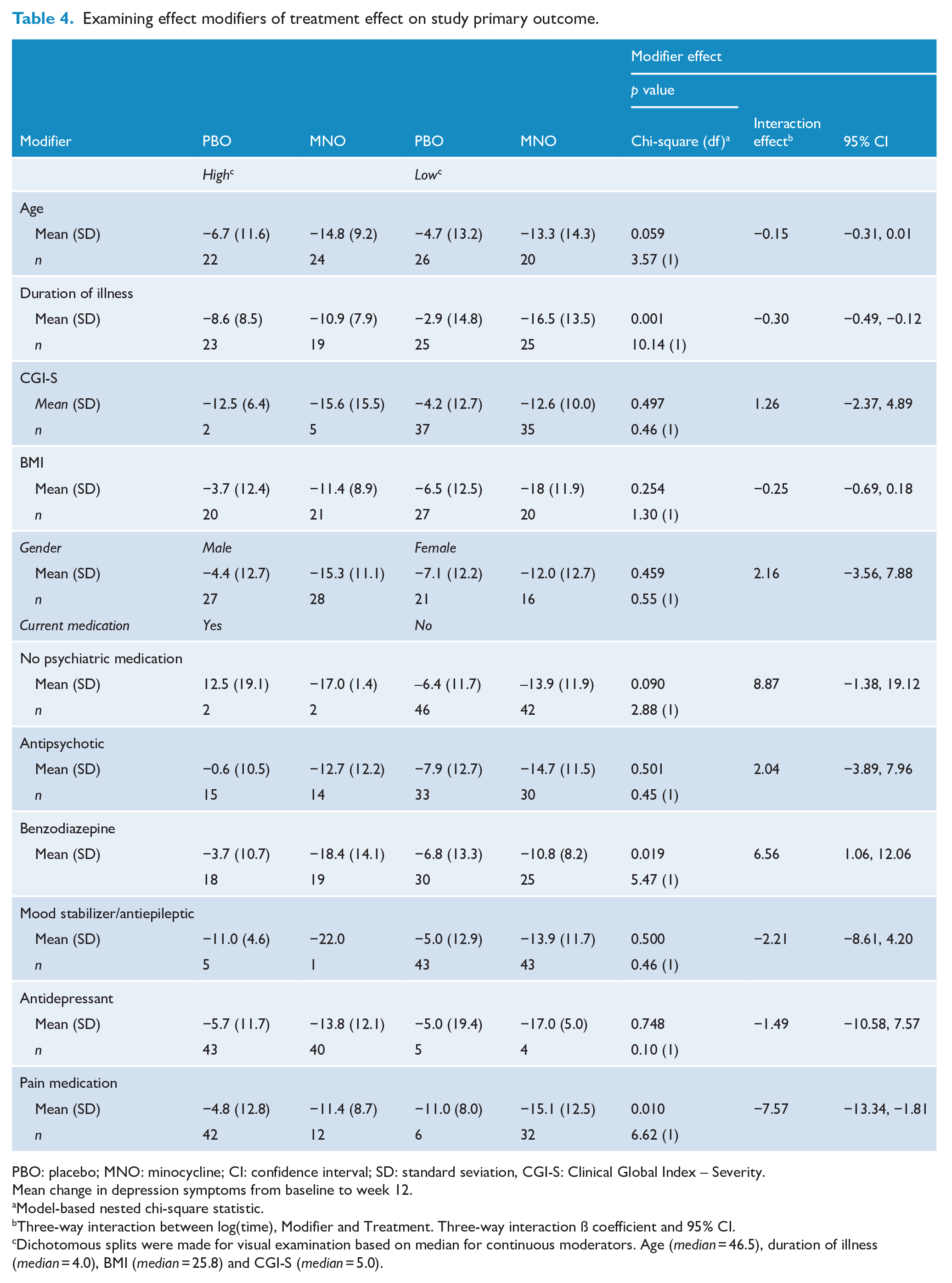
PBO: placebo; MNO: minocycline; CI: confidence interval; SD: standard seviation, CGI-S: Clinical Global Index – Severity.
Mean change in depression symptoms from baseline to week 12.
Model-based nested chi-square statistic.
Three-way interaction between log(time), Modifier and Treatment. Three-way interaction ß coefficient and 95% CI.
Dichotomous splits were made for visual examination based on median for continuous moderators. Age (median = 46.5), duration of illness (median = 4.0), BMI (median = 25.8) and CGI-S (median = 5.0).
Secondary outcomes, such as depression and anxiety severity, were not significant mediators/predictors of treatment effect in primary outcome, respectively, χ2 = 0.01, ß (CI) = 0.10 [−2.40, 2.60], p = 0.939, and χ2 = 0.80, ß (CI) = 1.11 [−1.32, 3.53], p = 0.370. Conversely, change in global clinical and functional status from weeks 2 to 12, measured by CGI-I, was a significant predictor of treatment effect during the trial – χ2 = 9.14, ß (CI) = 4.71 [1.65, 7.76], p = 0.003 (Table 5), which means that a larger reduction in depression was directly associated with reductions in functional and clinical status.
Examining mediators/predictors of treatment effect on study primary outcome.
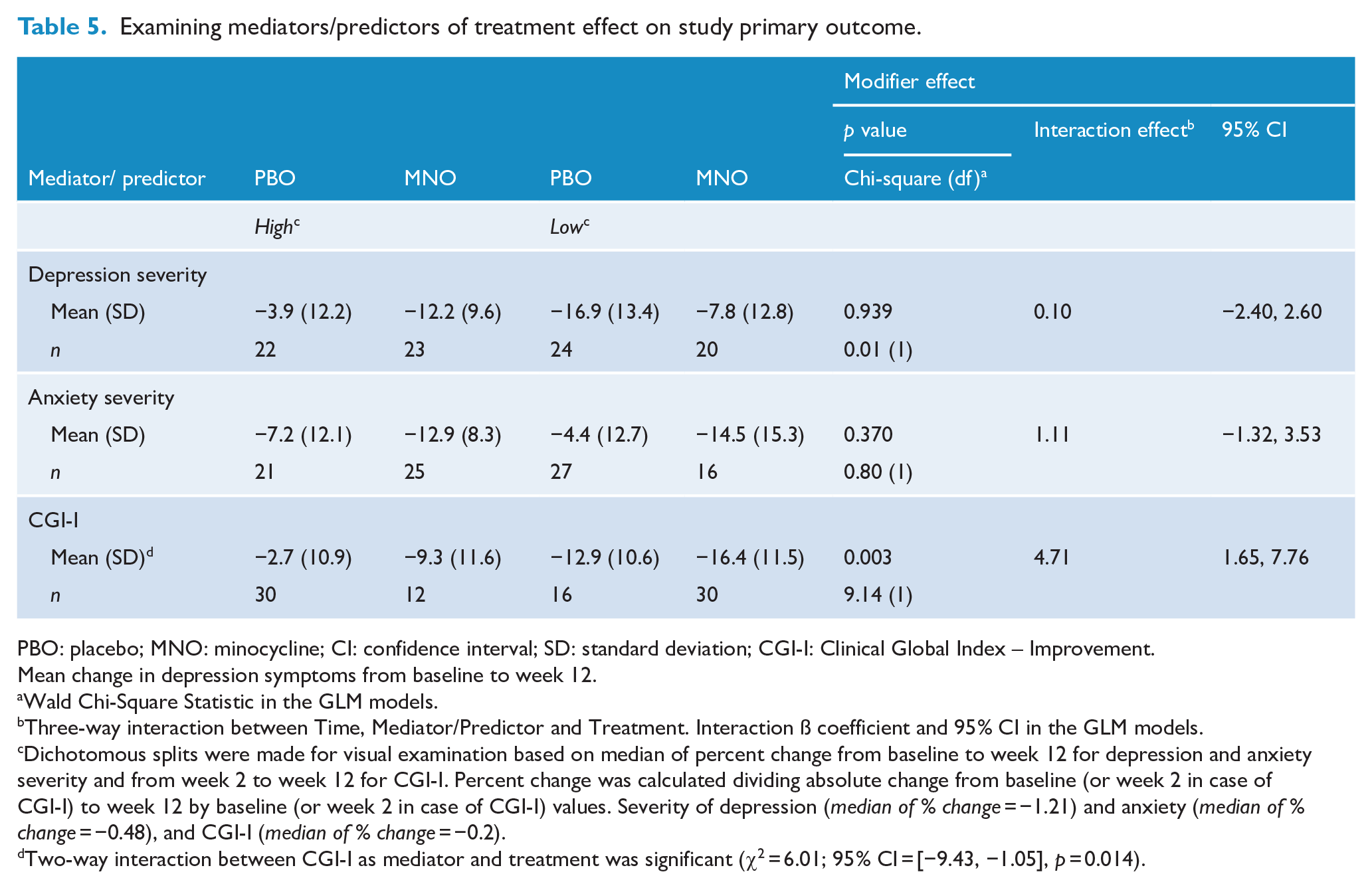
PBO: placebo; MNO: minocycline; CI: confidence interval; SD: standard deviation; CGI-I: Clinical Global Index – Improvement.
Mean change in depression symptoms from baseline to week 12.
Wald Chi-Square Statistic in the GLM models.
Three-way interaction between Time, Mediator/Predictor and Treatment. Interaction ß coefficient and 95% CI in the GLM models.
Dichotomous splits were made for visual examination based on median of percent change from baseline to week 12 for depression and anxiety severity and from week 2 to week 12 for CGI-I. Percent change was calculated dividing absolute change from baseline (or week 2 in case of CGI-I) to week 12 by baseline (or week 2 in case of CGI-I) values. Severity of depression (median of % change = −1.21) and anxiety (median of % change = −0.48), and CGI-I (median of % change = −0.2).
Two-way interaction between CGI-I as mediator and treatment was significant (χ2 = 6.01; 95% CI = [−9.43, −1.05], p = 0.014).
Investigating between-study heterogeneity
In order to evaluate heterogeneity between studies, we examined two-way interactions treatment × site in GEE models including study site, visit and treatment as factors (see Supplement Table S1). There was no statistically significant treatment × site interaction for primary (p = 0.068) and secondary outcomes, such as depressive and anxiety severity (respectively, p = 0.820 and p = 0.814), and clinical and functional outcomes, measured by CGI-I (p = 0.448). For the primary outcome, intervention effects at 2-, 4-, 8- and 12-week visits were not significantly different between both studies, showed in between-group differential change from baseline, estimated from time × treatment interaction. The effects were larger for Husain et al. (2017a) in comparison with Dean et al. (2017) (see Supplement Table S2). These findings indicated that the study site had no obvious between-study heterogeneity effect and as such pooling data from both studies in order to estimate the overall treatment effect were methodologically valid.
Discussion
The principal findings of this pooled data study from two RCTs of minocycline for MDD indicate that 12 weeks treatment with adjunctive minocycline resulted in reduction in depression (primary outcome), anxiety severity and improved clinician-rated global impression. With the exception of depression severity (secondary outcome), significant changes were found for all secondary outcomes at the end of the trial. A large proportion of participants treated with minocycline in both studies either responded to minocycline (56.8%) or achieved remission (31.8%) at the end of the trial in comparison with the placebo group (respectively, 25.5% and 10.6%). Finally, depression chronicity, as well as current use of benzodiazepines and pain medications, was identified as potential moderators, whereas clinical and functional status was identified as potential mediators/predictors of treatment effects. Given that concomitant pain and chronic depression are predictors of poorer outcomes, that these groups did better on minocycline is clinically meaningful, caveats regarding secondary outcomes notwithstanding.
The present results add to growing evidence that minocycline confers antidepressant effects. Emadi-Kouchak et al. (2016) reported significant improvement in depressive symptoms in HIV patients treated with minocycline in comparison with placebo controls following a 6-week trial. Similar results were found for depression in bipolar disorder patients (measured by both HAM-D and MADRS) and CGI-S in patients treated with minocycline after an 8-week period (Soczynska et al., 2017). A completed study yet to be published (NCT01659320) also found a significant outcome for reduction in depressive symptoms in the minocycline-treated group (Rosenblat and McIntyre, 2018). In addition, minocycline has shown to be beneficial for treating depressive-like behaviours and symptoms in early schizophrenia and bipolar depression (Chaudhry et al., 2012; Murrough et al., 2018; Savitz et al., 2018).
With regard to the primary outcomes, a significant change was found at the end of the treatment phase, and at weeks 4 and 8. Medium ESs were found at 4- and 8-week visits, with an increasing trend until the end of treatment. Originally, both studies were powered to achieve a medium ES and these findings could be considered clinically significant. A response criterion of 0.40 or higher may be clinically significant in clinical trials for antidepressant medication (Faries et al., 2000). Of the two original studies, only Husain et al. (2017b) found significant differences at the end of 12-week period treatment. One possible explanation for this is the variation in depression severity across the two cohorts. During baseline evaluation, in the trial conducted by Dean et al. (2017), 31 participants (43.7%) presented with an equivalent HAM-D score equal to or higher than 25, while in the trial conducted by Husain et al. (2017a), 32 participants (78%) presented with that score. According to Cleare et al. (2015), the severity of depressive symptoms is a potential variable when determining whether antidepressants are beneficial to a patient. Generally speaking, individuals with severe depressive symptoms are likely to respond better to medication, whereas individuals with only mild or moderate symptoms usually respond less well (Fournier et al., 2010). In line with this notion, effective antidepressant treatment may exert superior efficacy in a patient cohort with total HAM-D scores ⩾25. Notably, in the present pool of data, 56.3% of all participants presented with a score equal to or larger than 25 in the HAM-D scale in baseline.
Significant outcomes were also found for anxiety severity and clinician-rated global impression. There were differences between groups for anxiety severity at follow-up visits at 8 weeks. The changes observed in clinical, functional status and anxiety severity are noteworthy in the present pooled analysis. Initially, the focus of both studies was an improvement in depression symptoms; however, minocycline might be affecting other biological mechanisms linked with improvements in global impression, anxiety, functionality and quality of life (Dean et al., 2017). A recent RCT investigated the clinical effects of sertraline in primary care system found similar outcomes: greater improvements in anxiety, quality of life and clinical and functional outcomes in comparison with depressive symptoms (Lewis et al., 2019). Anxiety and depression may co-occur and the former could be primarily responsible for poor outcomes related to quality of life and both clinical and functional outcomes. Possibly, improvements in depressive outcomes could occur through improvements in quality of life and anxiety symptoms rather than reduced severity of depression (Lewis et al., 2019; Zhou et al., 2017).
In animal models, minocycline has been shown to reduce anxiety-like behaviours in three different behavioural assessments (open field tests, light-dark box and elevated maze zero; Majidi et al., 2016). Similarly, Liu et al. (2018) identified that minocycline might exert anxiolytic effects modulating neuroinflammation through peroxisome proliferator-activated receptor gamma/nuclear factor kappa B (PPAR-γ/NF-κB) pathway and, by extension, be used as an adjunctive treatment in stress-induced anxiety disorders. However, to date, no studies have been published investigating the use of minocycline as treatment for anxiety symptoms among humans. Thus, further studies might be necessary to investigate the effects of minocycline in general well-being, functionality and improvement of anxiety symptoms.
While there were no statistically significant differences between groups regarding the severity of depression, a trend towards decrease in severity could be observed in the treatment arm of the pooled data, in special among more severe participants, whereas in the placebo arm, symptoms tended to be stable independently of the severity of depression. The severity of symptoms could be related to several factors, including chronicity of illness, the severity of symptoms at baseline and previous treatment attempts. Due to the chronicity of the illness, change in the severity of symptoms of depression may take longer than the investigated 12-week period, as highlighted above, and is strongly associated with poor response to placebo (Fournier et al., 2010). In the same direction, it is important to highlight that the duration of the illness might have a moderator effect in response to treatment. As described above, older patients and those with longer illness duration from first onset were less likely to respond to treatment. Either early or late onset of MDD increases treatment resistance as well as longer duration of the illness, constituting possible predictors of response to antidepressant treatments (De Carlo et al., 2016; Nelson et al., 2013; Perlman et al., 2019).
Several mechanisms may explain the association between inflammation and depression and, by extension, the effects of minocycline as a treatment to improve depressive symptoms. As discussed by Husain et al. (2017a), minocycline may produce antidepressant effects inhibiting microglia and immune cell activation via multiple mechanisms, which exert a central role in the neuroinflammatory process. This process reduces the expression of several pro-inflammatory cytokines and regulates anti-inflammatory cytokines, as well as chemokines, lipid mediators of inflammation. Higher levels of pro-inflammatory cytokines are directly related with mood change and depression severity, and minocycline, alone or in combination with conventional antidepressants, may exert antidepressant properties. Another hypothesis suggests that the kynurenine pathway, which has been shown involved in the pathophysiology of mood disorders, may be modulated by minocycline (Soczynska et al., 2012). As observed in animal models, this treatment may block the upregulation of pro-inflammatory cytokines, which is accompanied by decreased depressive-like behaviours (Liu et al., 2015). It is also possible that minocycline normalizes gut dysbiosis which plays a role in depression by affecting gut permeability and the Toll-like receptor complex leading to increased immune and oxidative processes and neuroinflammation (Maes et al., 2008; Slyepchenko et al., 2016).
In addition, it is important to highlight the role of pain medication as a treatment modifier in this study. As identified in the moderator analysis, participants who were using pain medication presented larger reductions in depressive symptoms than those who were not. This may indicate an interaction in inflammatory pathways that may be targeted by minocycline. Walker et al. (2014) noted that increased levels of pro-inflammatory cytokines are associated with MDD and the sensitization of nociceptors, elevating pain sensitivity. In this way, participants who reported the use of pain medication might present higher levels of inflammatory levels prior to the use of the medication and such use could be directly or indirectly related to a decrease in inflammatory biomarkers. Thus, participants who were taking both pain medication and minocycline throughout the treatment might have experienced larger reduction in depressive symptoms than those who were taking minocycline only.
The results of this study indicate that minocycline could be a potential adjunctive treatment for individuals with MDD. The medication presents several advantages, which could be translated into clinical practice, including a relatively good safety profile with low rates of severe adverse effects, as shown in Table 4. There were no significant differences in the side effects reported compared to the placebo group. Moreover, minocycline is also readily available and is an affordable off-patent medication with a low propensity to produce antibiotic resistance (Husain et al. (2017a)). Finally, this study demonstrates not only a significant improvement in depression symptoms but also a significant improvement in anxiety severity and clinician-rated global impression.
To date there are only two published studies of minocycline for MDD which is insufficient to derive meaningful conclusions via meta-analysis. Both RCTs had relatively small sample sizes and combining their data increased the sample size, enabling subgroup analysis. Given the promise of minocycline as an adjunctive antidepressant, there is benefit in exploring these two studies sooner than is permissible by a traditional meta-analysis. Some extra strengths include the ability to analyse data in detail, the use of up-to-date data, the possibility to avoid biases associated with use of aggregate data and to investigate new hypotheses with the raw data, which would not be possible with a traditional meta-analysis (Simmonds et al., 2005).
However, several limitations need to be taken into consideration such as the small sample size in both trials, and a limited number of published and ongoing studies investigating the antidepressant effects of minocycline. Taking this into consideration, the present pooling of data had the following strengths: (1) a larger sample size, including data from two different RCTs; (2) homogeneity of study designs, including daily medication intake, both inclusion and exclusion criteria and highly correlated primary and secondary outcomes; (3) homogeneity in clinical and sociodemographic characteristics in both groups; and (4) identification of both primary and secondary significant outcomes as well as possible moderators and mediators of treatment response in comparison to placebo. Although this was a relatively large study, sample sizes for biological analyses were smaller in both studies and biomarkers measured were not comparable between studies. Moreover, the role of other psychiatric comorbidities was not taken into consideration in the present pooling of data. In future, a larger number of RCTs and larger samples are necessary to confirm our findings. Moreover, the inclusion of a larger number of biomarkers in larger samples would be useful.
Supplemental Material
Table_S1 – Supplemental material for Minocycline as adjunctive treatment for major depressive disorder: Pooled data from two randomized controlled trials
Supplemental material, Table_S1 for Minocycline as adjunctive treatment for major depressive disorder: Pooled data from two randomized controlled trials by Robson Zazula, Muhammad Ishrat Husain, Mohammadreza Mohebbi, Adam J Walker, Imran B Chaudhry, Ameer B Khoso, Melanie M Ashton, Bruno Agustini, Nusrat Husain, JFW Deakin, Allan H Young, Michael Berk, Buranee Kanchanatawan, Chee H Ng, Michael Maes, Lesley Berk, Ajeet B Singh, Gin S Malhi and Olivia M Dean in Australian & New Zealand Journal of Psychiatry
Supplemental Material
Table_S2 – Supplemental material for Minocycline as adjunctive treatment for major depressive disorder: Pooled data from two randomized controlled trials
Supplemental material, Table_S2 for Minocycline as adjunctive treatment for major depressive disorder: Pooled data from two randomized controlled trials by Robson Zazula, Muhammad Ishrat Husain, Mohammadreza Mohebbi, Adam J Walker, Imran B Chaudhry, Ameer B Khoso, Melanie M Ashton, Bruno Agustini, Nusrat Husain, JFW Deakin, Allan H Young, Michael Berk, Buranee Kanchanatawan, Chee H Ng, Michael Maes, Lesley Berk, Ajeet B Singh, Gin S Malhi and Olivia M Dean in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
This work was supported in part by an Academic Scholars Award to MIH from the Department of Psychiatry, University of Toronto. The authors would like to thank the Cooperative Research Centre (CRC) for Mental Health. This report represents independent research funded by the National Institute for Health Research (NIHR) Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. Olivia M Dean is a R.D. Wright NHMRC Biomedical Career Development Fellow [APP1145634]. The original study by Dean et al. was supported by the Brain and Behavior Foundation and an unrestricted joint award from the Australasian Society for Bipolar and Depressive Disorders and Servier. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: R.Z. reports received a scholarship from CAPES [PDSE - 1.187966/2018-01]. M. I. H. reports have received grant support from the Brain and Behavior Research Foundation, Stanley Medical Research Institute and the Physician’s Services Incorporated Foundation and he is also PI on a clinical trial sponsored by COMPASS Pathways Limited. M.M. reported no biomedical financial interests or potential conflicts of interest. A.J.W. reports have been supported by a Trisno Family Fellowship and received grant support from Deakin University. I.B.C. reported no biomedical financial interests or potential conflicts of interest. A.B.K. reported no biomedical financial interests or potential conflicts of interest. M.M.A. reports receiving grant/research support from Deakin University, Australasian Society for Bipolar Depressive Disorders, Lundbeck, Australian Rotary Health, Ian Parker Bipolar Research Fund and Cooperative Research Centre for Mental Health and PDG Geoff and Betty Betts Award from Rotary Club of Geelong. B.A. reported no biomedical financial interests or potential conflicts of interest. N.H. reported no biomedical financial interests or potential conflicts of interest. J.F.W.D. reported no biomedical financial interests or potential conflicts of interest. A.H.Y. reports have been employed by King’s College London; Honorary Consultant SLaM (NHS UK). Paid lectures and advisory boards for the following companies with drugs used in affective and related disorders: Astrazenaca, Eli Lilly, Lundbeck, Sunovion, Servier, Livanova, Janssen, Allegan, Bionomics, Sumitomo Dainippon Pharma. Consultant to Johnson & Johnson. Consultant to Livanova. Received honoraria for attending advisory boards and presenting talks at meetings organized by LivaNova. Principal Investigator in the Restore-Life VNS registry study funded by LivaNova. Principal Investigator on ESKETINTRD3004: ‘An Open-label, Long-term, Safety and Efficacy Study of Intranasal Esketamine in Treatment-resistant Depression’. Principal Investigator on ‘The Effects of Psilocybin on Cognitive Function in Healthy Participants’. Principal Investigator on ‘The Safety and Efficacy of Psilocybin in Participants with Treatment-Resistant Depression (P-TRD)’. No shareholdings in pharmaceutical companies. Grant funding (past and present): NIMH (USA), CIHR (Canada), NARSAD (USA), Stanley Medical Research Institute (USA), MRC (UK), Wellcome Trust (UK), Royal College of Physicians (Edin), BMA (UK), UBC-VGH Foundation (Canada), WEDC (Canada), CCS Depression Research Fund (Canada), MSFHR (Canada), NIHR (UK) and Janssen (UK). M.B. is supported by a National Health and Medical Research Council (NHMRC) Senior Principal Research Fellowship (1059660 and 1156072). B.K. reported no biomedical financial interests or potential conflicts of interest. C.H.N. reported no biomedical financial interests or potential conflicts of interest. M.M. reported no biomedical financial interests or potential conflicts of interest. L.B. reported no biomedical financial interests or potential conflicts of interest. A.B.S. reported no biomedical financial interests or potential conflicts of interest. G.S.M. reported no biomedical financial interests or potential conflicts of interest. O.M.D. is a R.D. Wright NHMRC Biomedical Career Development Fellow [APP1145634] and has received grant support from the Brain and Behavior Foundation, Simons Autism Foundation, Stanley Medical Research Institute, Deakin University, Lilly, NHMRC and ASBDD/Servier and she has also received in kind support from BioMedica Nutraceuticals, NutritionCare and BioCeuticals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The Dean et al. (2017) clinical trial was funded by the Brain and Behavior Foundation and Australasian Society for Bipolar and Depressive Disorders/Servier grant and the authors gratefully acknowledge their support. The ![]() trial was part funded by the Pakistan Institute of Living and Learning and the National Institute of Health Research Biomedical Research Centre at the South London and Maudsley NHS Foundation Trust and King’s College London. The authors would like to acknowledge the service support of Barwon Health, Deakin University, The Melbourne Clinic, The University of Melbourne and Chulalongkorn University.
trial was part funded by the Pakistan Institute of Living and Learning and the National Institute of Health Research Biomedical Research Centre at the South London and Maudsley NHS Foundation Trust and King’s College London. The authors would like to acknowledge the service support of Barwon Health, Deakin University, The Melbourne Clinic, The University of Melbourne and Chulalongkorn University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
