Abstract
Objectives:
Plant-based medicines have had a long-standing history of use in psychiatric disorders. Highly quantified and standardized extracts or isolates may be termed “phytoceuticals,” in a similar way that medicinal nutrients are termed as “nutraceuticals.” Over the past 2 decades, several meta-analyses have examined the data for a range of plant-based medicines in the treatment of psychiatric disorders. The aim of this international project is to provide a “meta-review” of this top-tier evidence.
Methods:
We identified, synthesized, and appraised all available up to date meta-analyses... of randomized controlled trials (RCTs) reporting on the efficacy and effectiveness of individual phytoceuticals across all major psychiatric disorders.
Results:
Our systematic search identified 9 relevant meta-analyses of RCTs, with primary analyses including outcome data from 5,927 individuals. Supportive meta-analytic evidence was found for St John’s wort for major depressive disorder (MDD); curcumin and saffron for MDD or depression symptoms, and ginkgo for total and negative symptoms in schizophrenia. Kava was not effective in treating diagnosed anxiety disorders. We also provide details on 22 traditional Chinese herbal medicine formulas’ meta-analyses (primarily for depression studies), all of which revealed highly significant and large effect sizes. Their methodology, reporting, and potential publication bias were, however, of marked concern. The same caveat was noted for the curcumin, ginkgo, and saffron meta-analyses, which may also have significant publication bias.
Conclusions:
More rigorous international studies are required to validate the efficacy of these phytoceuticals before treatment recommendations can be made. In conclusion, the breadth of data tentatively supports several phytoceuticals which may be effective for mental disorders alongside pharmaceutical, psychological therapies, and standard lifestyle recommendations.
Introduction
Data from the World Health Organization (WHO) reveal a continuing increase in the use of herbal and plant-based medicines, with an estimated 80% of the world population utilizing them as part of primary health care treatment for a range of physical ailments. 1 In the United States, a national household survey has shown that herbal/natural medicines, along with dietary supplements, were the most commonly utilized complementary therapies for psychiatric disorders, with St John’s wort (Hypericum perforatum L.) and kava (Piper methysticum) as a treatment of mood disorders being the most used in this population. 2 This therapeutic approach is broadly under the umbrella of Traditional, Complementary, and Integrative Medicine as defined by the WHO. 3 The specific therapeutic application of natural products produced via pharmaceutical Good Manufacturing Practice, standardized and optimized, and in some cases purified (e.g., curcumin) or slightly modified (e.g., n-acetyl cysteine or 5-hydroxytryptophan) can be further subdivided into the terms “nutraceuticals” 4 and “phytoceuticals.”
Phytoceuticals have a range of psychoactive effects that include putative antidepressant, anxiolytic, nootropic (cognitive enhancing), sedative, hypnotic, and analgesic properties. 5 The diverse mechanisms of action for these plant-based medicines primarily involve alteration of neurotransmitter synthesis and degradation, agonism and antagonism of neuroreceptors or inhibition of neurotransmitters reuptake proteins, or supporting the homeostatic function of the hypothalamic pituitary adrenal (HPA) axis. 5 For example, kava, through its main active compounds, kavalactones, acts as a γ-aminobutyric acid pathway agonist, 6 whereas St John’s is a known monoamine reuptake inhibitor, although its modulation of HPA activity is also evident (note that preclinical data may not directly translate into pharmacodynamic effects at relevant human doses). 7 Further, phytoceuticals such as ginkgo (Ginkgo biloba) may exert antioxidant, anti-inflammatory, and antiplatelet activities, while increasing blood–brain barrier permeability, providing a potential adjunctive supportive treatment for brain disorders. 8 Traditional Chinese herbal medicine (TCHM) formulas are based on traditional medical knowledge, involving a range of different plant-based medicines often combined for both pharmacodynamic activity in addition to enhanced bioavailability (via the addition often of herbal “envoys” such as licorice root [Glychrrhiza glabra] and ginger rhizome [Zingiber officinale] to increase digestive absorption of the constituents). 9 Conventional mechanistic understandings of these agents in this context are, however, somewhat limited.
A recent review has examined the evidence of 23 plant-based medicines for 11 psychiatric disorders with human clinical trials; these include kava (Piper methysticum), passionflower (Passiflora spp) galphimia (Galphimia glauca), and chamomile (Matricaria recutita) for anxiety disorders; St John’s wort (Hypericum perforatum), curcumin from turmeric (Curcuma longa), and saffron (Crocus sativus) for major depressive disorder (MDD); while ginkgo (Ginkgo biloba) was studied as an adjunctive treatment in schizophrenia. 10 However, an assessment of top-tier meta-analytic evidence via a “meta-synthesis” has not been conducted, and would provide far more definitive data. An example can be found in our sister-review on nutraceuticals, 11 which included 33 meta-analyses of placebo-controlled RCTs involving 10,951 individuals, covering a range of nutrients including ω-3 fatty acids, methylfolate, and n-acetyl cysteine.
The aim of this meta-review was to aggregate and evaluate the top-tier evidence for the efficacy of plant-based medicines in the treatment of psychiatric disorders. To achieve this, we identified, synthesized, and appraised all available data from meta-analyses of randomized controlled trials (RCTs) examining psychiatric outcomes for all phytoceuticals across various psychiatric disorders. Along with providing an overview of the efficacy of these agents across different disorders, we also aimed to explore which dosages and symptomatic targets were most appropriate in order to provide clinical guidelines.
Methods
The search strategy and data synthesis were conducted in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement [6] and followed a preregistered protocol (PROSPERO: CRD42018105880).
Systematic Search
The title and key word search algorithm are presented in Table 1. The systematic search was conducted using Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews, Health Technology Assessment Database, Allied and Complementary Medicine, PsycINFO, and Ovid MEDLINE(R), from inception until January 2020.
PICO (Participants, Interventions, Comparisons, Outcomes) Systematic Search Strategy.

A search of Google Scholar was conducted using the same key words to identify any additional relevant articles. Reference lists of included articles were also searched.
Eligibility Criteria
Eligibility criteria were organized in accordance with the PICO (Participants, Interventions, Comparisons, Outcomes) reporting structure, as described above.
Participants
We included studies of individuals with common and severe mental disorders: These included depressive disorders (including any clinical depression, diagnosed MDD or bipolar disorder (or mania), depression in pregnancy, in old age, or as a comorbidity to chronic health conditions); all diagnosed anxiety disorders; schizophrenia (examining total symptoms along with positive, negative, general symptoms, and tardive dyskinesia); states at risk for psychosis (examining attenuated psychotic symptoms, negative symptoms, transition to psychosis, and functioning); and attention-deficit hyperactivity disorder.
All studies of the above conditions were eligible provided that at least 75% of the sample had a confirmed mental illness or at-risk state, ascertained by either clinical diagnostic history or reaching established thresholds on validated screening measures. Reports examining mental health outcomes in general population studies were only included if they met the above criteria. Studies examining neurodegenerative disorders (e.g., dementia) or neurodevelopmental disorders (e.g., autism, intellectual disability) were not included. It was decided via consensus that meta-analyses involving TCHMs would not be included in the main meta-synthesis analyses and forest plots due to concerns over continual highly significant findings (P < 0.01) or large effect sizes (d > 0.8). These studies were, however, included in tabular form to provide an overview of the results (and to illustrate the consistent positive findings and methodological concerns).
Interventions and comparisons
All plant-based interventions were considered for this meta-review, used either as adjunctive treatment or monotherapy. These could be either whole plant medicines or isolated constituents. Because this study aimed to provide a meta-review of top-tier evidence, only meta-analyses of RCTs were included.
Outcomes
All data on mental health outcomes (including changes in clinical measures, response rates) from meta-analyses of RCTs examining phytoceuticals for any eligible disorder were included in this meta-review. A meta-analysis was classified as eligible if: (a) it had clearly stated inclusion, intervention, and comparison criteria aligned with the participant, intervention, and comparison criteria listed above; (b) it reported a systematic search with a screening procedure; (c) it had used systematic data extraction and reported pooled continuous or categorical outcome data from more than one study.
Where overlapping meta-analyses of a given intervention for a specific outcome/disorder existed, the most recently updated meta-analysis was used, provided it captured more than 75% of the trials in the earlier version. Where older meta-analyses presented unique findings, through inclusion of a greater number of studies or use of particular subgroup analyses, these data were used as secondary analyses for our meta-review.
Quality Assessment of Included Meta-analyses
The quality of eligible meta-analyses was assessed using “A Measurement Tool to Assess Systematic Reviews” Version 2 (AMSTAR-2). 12 This is an updated version of the original AMSTAR designed to better capture review quality and confidence in findings. AMSTAR-2 assesses 16 constructs, which all indicate the quality of a systematic review/meta-analysis.
Data Extraction and Analysis
For each study, we manually extracted effect size data as standardized mean differences (SMDs) with 95% confidence intervals compared to placebo conditions, along with the P value. Data were initially extracted by 2 authors (J.S. and W.M.) and then cross-checked for quality with duplicate data extraction by an additional author (M.A.).
In line with conventional interpretations, SMDs were classified as negligible (<0.2), small (0.2 to 0.4), medium (0.4 to 0.8), or large (>0.8). In cases where continuous outcomes were reported as weighted mean differences or raw mean differences, these were recalculated into an SMD (Hedges’ g) using Comprehensive Meta-Analysis 3.0. The SMDs were also adjusted in a uniform direction to indicate whether the intervention was either a positive or negative (in relation to placebo). Where meta-analyses had applied fixed-effects models to calculate the effect size of the phytoceutical compared to placebo, these were also recalculated using a random-effects model, such that SMDs across supplements/disorders could be meaningfully compared.
For both primary and secondary analyses, we also extracted the number of participants (N), along with the number of trials/comparisons (n) from which the pooled effect size was derived. Additionally, heterogeneity was quantified using the I 2 statistic and categorized as low (I 2 < 25%), moderate (I 2 = 25% to 50%), or high (I 2 > 50%). Where reported, all relevant study characteristics were also extracted, specifically with regard to the phytoceutical used (including formulation and dose), the sample and the diagnostic details, and any relevant subgroup analyses. The potential impact of publication bias was assessed when sufficient data for appropriate analyses were available.
Results
Systematic Search Results
The search revealed 882 results. Title and abstract screening removed 527 articles, with 103 being articles reviewed in full by the 3 reviewing academics (Figure 1). Of these, 72 were ineligible. Thus, in total, eligible data from 9 independent main meta-analyses of RCTs of plant-based medicines, and 22 TCHM formulas for the treatment of psychiatric disorders were included for this meta-review (Table 2).
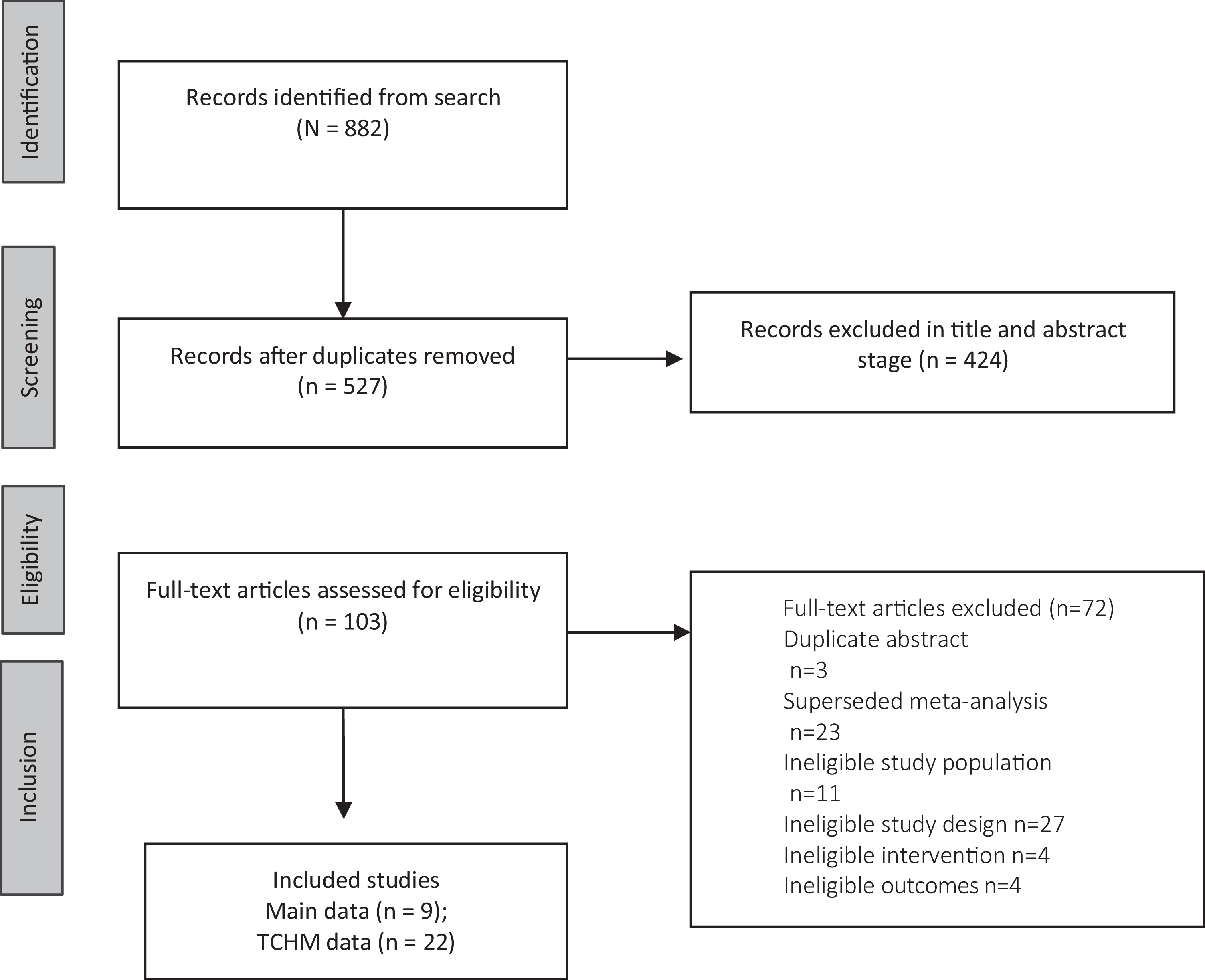
PRISMA flow diagram. PRISMA = Preferred Reporting Items for Systematic Reviews and Meta-Analyses; TCHM = traditional Chinese herbal medicine.
Key Evidence Summaries for Plant-based Medicines.
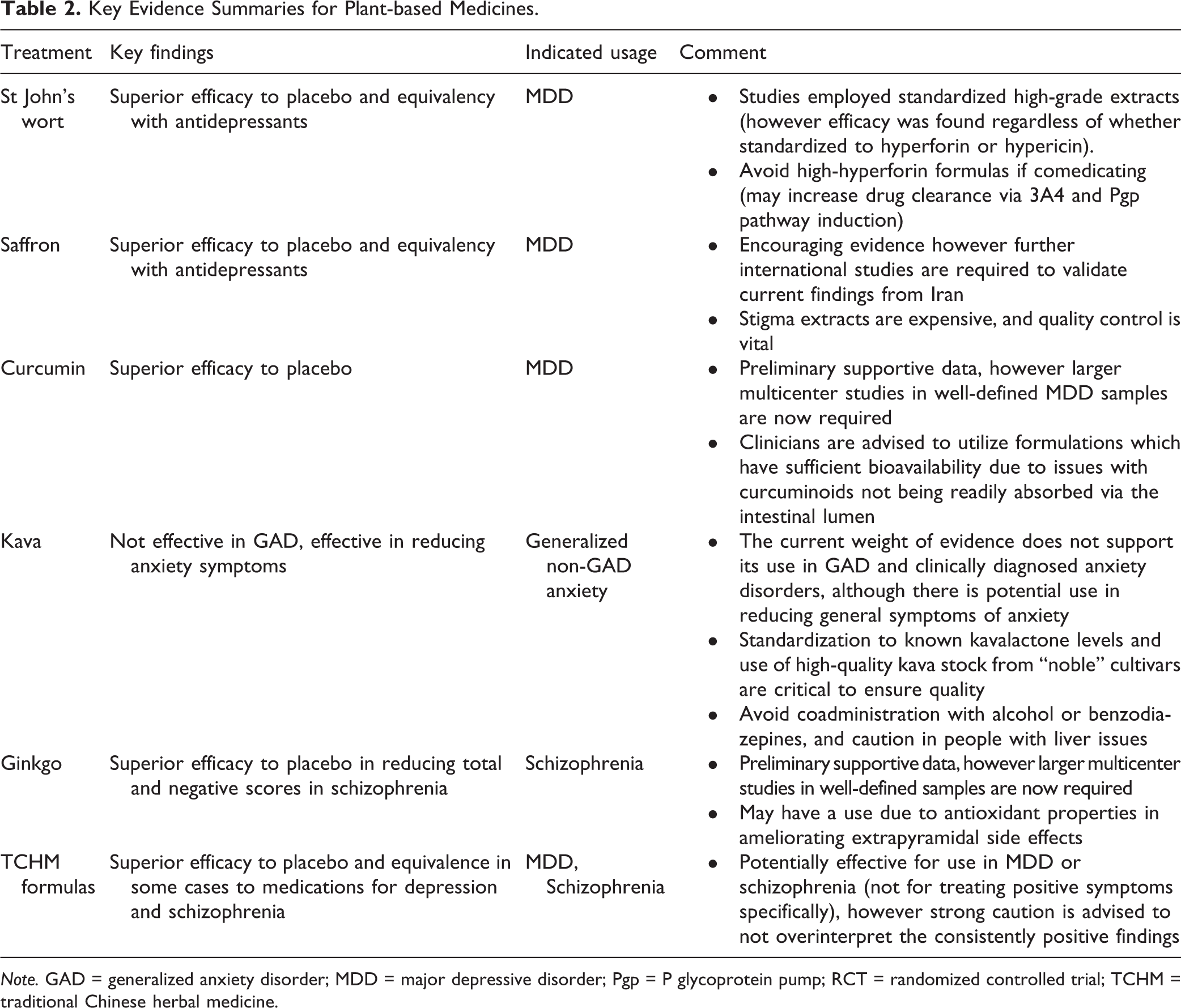
Note. GAD = generalized anxiety disorder; MDD = major depressive disorder; Pgp = P glycoprotein pump; RCT = randomized controlled trial; TCHM = traditional Chinese herbal medicine.
Meta-analyses of RCTs with primary analyses including outcome data revealed 5 plant-based medicines focusing on 4 disorders. These were St John’s wort for MDD 13 and curcumin for MDD or depression symptoms, 14,15 saffron for MDD or depression/anxiety symptoms, 16 kava for anxiety disorders, 17,18 and ginkgo for total and negative symptoms in schizophrenia (in addition to effects on tardive dyskinesia). 19 –21 The total sample consisted of 5,927 individuals. Meta-analyses were typically based on monotherapy interventions administered in conjunction with “usual care” (without specifying treatment regimens) or as an adjunctive treatment to a specific class of psychotropics (e.g., selective serotonin reuptake inhibitors in depression or antipsychotics in schizophrenia). See below for details on TCHM formula study characteristics and outcome data.
Quality Assessment of the Included Meta-analyses
The quality assessment of the data revealed that the individual meta-analyses fulfilled between 8 and 28 of the AMSTAR-2 criteria (median: 21, mean: 21.6). A qualitative assessment of the studies contained within the individual meta-analyses showed that all were RCTs (meeting our inclusion criteria); see Figure 2. A preponderance of the research was conducted in the United States and Europe for St John’s wort, Europe and Australia for kava, Iran for saffron, China for ginkgo, and a range of countries for curcumin.
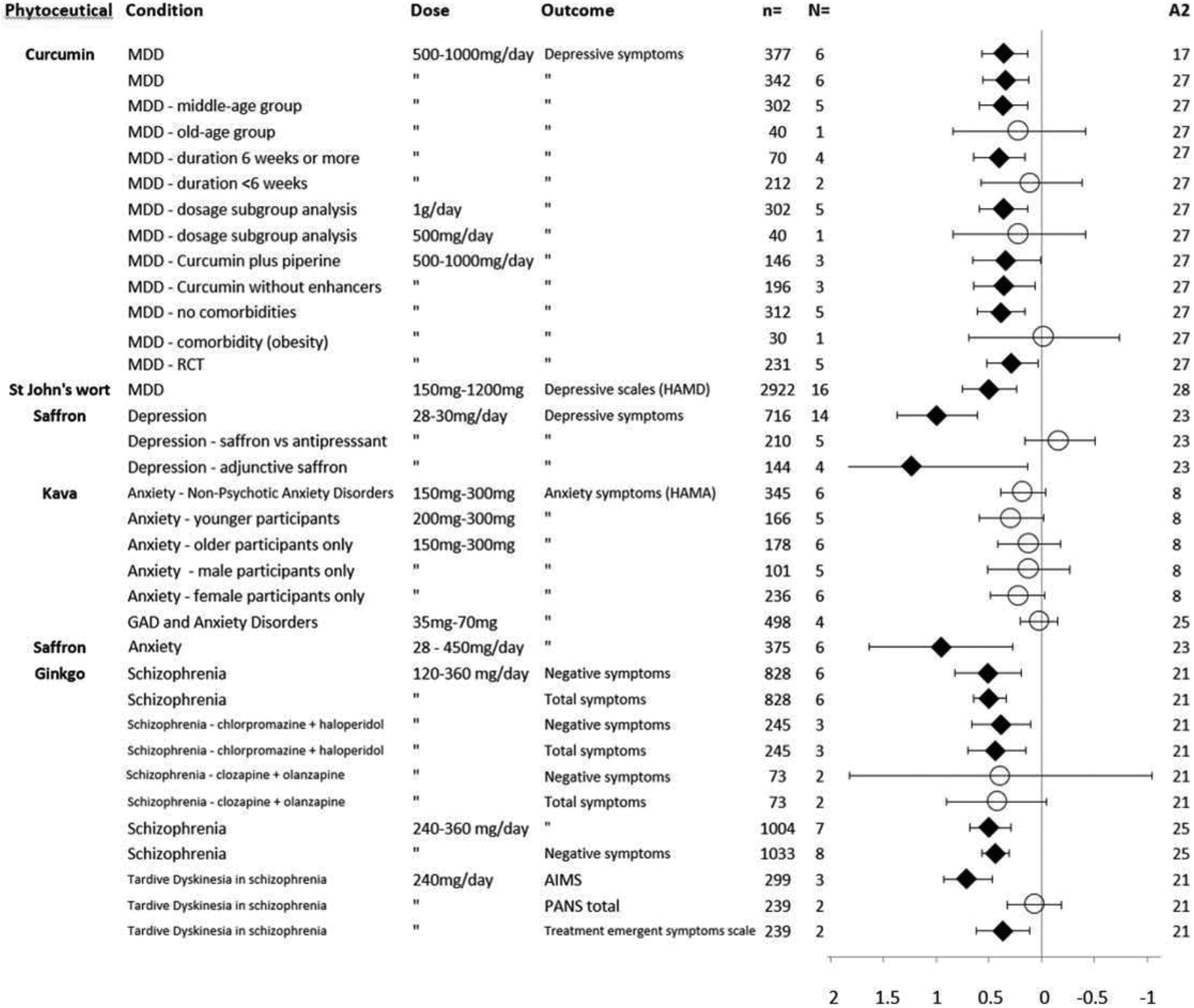
Effects of phytoceuticals in psychiatric disorders, shown as standardized mean difference with 95% CI. Circles represent no significant difference from placebo; diamonds represent P ≤ 0.05 compared to placebo; A2 = AMSTAR-2 total score; CI, confidence interval; GAD = generalized anxiety disorder; MDD = major depressive disorder.
Efficacy and Effectiveness Data
Depression
Figure 2 shows the meta-review forest plot. St John’s wort studies revealed a significant reduction of depressive symptoms in MDD compared to placebo (N = 16, SMD = 0.49, 0.23 to 0.74, P < 0.05; I 2 = 89%). 13 Preparations studied varied, having differing levels of hypericin and hyperforin (typically 900 mg per day divided into 2 or 3 doses). As noted by the I 2 value, there was obvious high heterogeneity in data. Trials were mainly conducted in Europe and the United States, with study lengths between 6 and 8 weeks, while sample sizes were between 30 and 570. In respect to saffron, a significant reduction in depressive symptoms was found (N = 14, SMD = 0.99, 0.61 to 1.37, P < 0.001; I 2 = 82%), 16 while once again, significant data heterogeneity was revealed. Saffron preparations studied were commonly standardized to crocin, or for one particular product to lepticrosalides® (a nonspecific measurement of bioactive compounds present in saffron, including safranal and crocin isomers). Trials were predominantly conducted in Iran, with study lengths between 4 and 8 weeks, while sample sizes ranged between 30 and 68. A positive finding was also revealed in support of curcumin for all studies included which assessed depressive symptoms (N = 6, SMD = 0.34, 0.13 to 0.56, P = 0.002; I 2 = 0%). 15 Two meta-analyses are included in the meta-review due to differing subsample data (n = 377 and n = 342); however, both had similar effect sizes, and heterogeneity was very low. Sample sizes were between 30 and 111, with trial lengths between 4 and 8 weeks using 500 mg to 1 g of curcumin (commonly 1 g of curcumin per day), with the addition of piperine or prepared via a formulation designed to enhance bioavailability. These phytoceuticals were considered to have a good safety profile, although St John’s wort has some clinical considerations regarding potential drug interactions, and certain potential adverse effects (see Table 2 for details).
Anxiety
The only plant-based medicines with meta-analytic evidence were kava and saffron. In respect to kava’s anxiolytic effect, the most recent data suggest an absence of statistical significance for the treatment of a mixture of anxiety disorders (and in particular for GAD; N = 4, SMD = −0.02, −0.19 to 0.16, P = 0.059; I 2 = 80%). 18 The data displayed marked heterogeneity. Trial lengths varied between 3 and 8 weeks with sample sizes between 35 and 100, and study extracts were standardized between 35 and 240 mg of kavalactones per day. An older meta-analysis of a specific kava extract (WS1490) also revealed a trend toward significance for treatment of a mixture of diagnosed anxiety disorders (N = 6, 0.18, −0.03 to 0.40, P = 0.07; I 2 = not available). 22 Study lengths were between 4 and 28 weeks, sample sizes ranged from 40 to 100 participants, and used 150 mg or 300 mg per day of the WS1490 kava extract. No significant finding was revealed when subgroup analyses were performed based on age (older/younger) or gender (male or female; P values all >0.05, see Figure 2). While studies revealed no major instance of adverse effects, some previous concerns have been noted over certain extracts having a negative effect on the liver (see Table 2 for details). A meta-analysis concerning saffron for anxiety outcomes revealed a significant effect in favor of the plant medicine over placebo (N = 6, SMD = 0.95, 0.27 to 1.63, P = 0.006; I 2 = 89%). 16
Schizophrenia
Three meta-analyses involving ginkgo providing data sufficiently unique to meet inclusion criteria. The most recent data showed that adjunctive use of ginkgo was superior to placebo in reducing total and negative symptoms of schizophrenia (N = 7, SMD = 0.49, 0.30 to 0.69, P < 0.001; I 2 = 42% and N = 8, SMD = 0.44, 0.32 to 0.57, P < 0.001; I 2 = 0%, respectively 20 ). It should be noted that meta-analytic data were not available assessing positive symptoms. As indicated via the I 2 scores, absent to moderate statistical heterogeneity was revealed. An older meta-analysis containing additional studies also supported ginkgo for both total and negative symptoms with an SMD in both cases of 0.5 in 6 studies (0.18 to 0.81, P < 0.05; I 2 = 67% and 0.36 to 0.64, P < 0.05; I 2 = 7%, respectively). 19 Further separate subanalyses of adjunctive treatment of ginkgo with chlorpromazine and haloperidol, and also clozapine and olanzapine, revealed a significant effect over placebo. Finally, a separate meta-analysis revealed mixed evidence for use of ginkgo in treating tardive dyskinesia, assessed via the Abnormal Involuntary Movements Scale (N = 3, SMD = 0.70, 0.47 to 0.93, P < 0.001; I 2 = 0%), Positive and Negative Syndrome Scale (N = 2, SMD = −0.06, 0.20 to −0.31, P = 0.67; I 2 = 62%), and the Treatment Emergent Symptoms Scale (N = 2, SMD = 0.37, 0.16 to 0.63, P = 0.004; I 2 = 0%). 21 The ginkgo studies in the meta-analyses used 120 to 360 mg of the standardized “EGb” extract. Study lengths were between 8 and 16 weeks, and sample sizes ranged from 29 to 568 participants. Ginkgo is generally regarded as a safe phytoceutical, although caution can be extended for couse with anticoagulants and prior to surgery due to mild anticoagulant effects.
Traditional Chinese herbal medicine formulas
A total of 22 meta-analyses were revealed via our search criteria concerning TCHMs for the treatment of depressive disorders (N = 20) and schizophrenia (N = 2; see Table 3). The number of studies included in the meta-analyses ranged from 7 to as many as 296 individual studies. The formulas studied included Chaihu-Shugan-San, Xiao Yao San, Ganmai Dazao, Wuling, Shuganjieyu, Wendan Decoction, Jiu Wei Zhen Xin (see supplementary Table 1). These were administered via decoction, granule, or capsule delivery. As mentioned in the Methods section, only the broad results were highlighted due to concerns over the reporting and methodological strengths of the studies included in the meta-analyses. All publications reported highly significant results in favor of the Chinese herbal medicine, and in some cases TCHMs were superior to standard antidepressants such as fluoxetine. Adverse events from these interventions were reported as very low, and in many adjunctive studies the Chinese herbal formulas were reported incidentally to ameliorate some of the medication side effects.
Traditional Chinese Herbal Medicines.

Discussion
This meta-review aggregated and evaluated all the recent top-tier evidence from meta-analyses of RCTs examining the efficacy of plant-based medicines for psychiatric disorder symptoms. We identified 9 eligible meta-analyses published from 2005 onward with primary analyses including 5,927 individuals with psychiatric disorders randomized to individual plant-based medicines or placebo control conditions. For a narrative summary of results and clinical considerations, see Table 3.
The interventions with the strongest evidentiary support were for St John’s wort for MDD (supported also by pooled analysis of key European study data 43 ), curcumin, and saffron for MDD and depression symptoms, and ginkgo for total and negative symptoms. Kava was not effective in treating diagnosed anxiety disorders, with an absence of statistical significance in GAD, while some supportive data for saffron for this application were evident. These results in the main reflects the position held in the most recent Canadian Network for Mood and Anxiety Treatment guidelines. 44 The data concerning kava should also be further taken into context due to a recently published large RCT involving the phytoceutical in the treatment of GAD, which revealed a nonsignificant effect compared to placebo 45 on the Hamilton Anxiety Rating Scale. However, it can be noted that research has generally supported kava in nonclinical populations (potentially for more “situational” anxiety as a short-term anxiolytic). All 22 meta-analyses of Chinese herbal medicine formulas (primarily for depression studies) revealed highly significant and large effect sizes, but their methodologies, reporting, and potential publication bias were of marked concern. Additionally, an important caveat is that many studies included in the curcumin, ginkgo, and saffron meta-analyses may have had publication bias (in respect to negative studies potentially not being published). Additional more rigorous international studies are therefore required to validate these results.
While many strengths of this meta-review are evident, including the collation of tier-1 meta-analytic RCT data via a rigorous systematic search process, some limitations are noted. First, studies were confined to the English language, and some research may have been excluded. Further, some of the data from studies contained within the individual meta-analyses had poor reporting and weak methodological standards, small samples, or absence of constituent standardization, thus some degree of caution needs to be applied, most notably in respect to TCHM formulas 46 and saffron. A final consideration is that regarding the data including within the meta-analyses, these commonly derived data from individual RCTs using “completer” analyses, and not more statistically acceptable intention-to-treat data. Due to this, there is presently limited clinical utility of the results in the meta-review in making treatment determinations. More research is required to firmly validate TCHM formulas and saffron across more diverse jurisdictions, in addition to more research on more newly studied phytoceuticals such as curcumin.
We also note that other plant medicines for certain psychiatric disorders with supportive or mixed RCT evidence exist that have not been subjected to meta-analysis, in particular for other disorders such as bipolar depression. These include for affective disorders: lavender (Lavandula spp.), roseroot (Rhodiola rosea), catmint (Nepeta menthoides), dodder (Cuscuta spp.), Galphimia (Galphimia glauca), chamomile (Matricaria recutita), passionflower (Passiflora incarnata), ginkgo (Ginkgo biloba), ashwagandha (Withania somnifera; also revealing procognitive effects in bipolar disorder), and mangosteen (Garcinia mangostana) for schizophrenia and mood disorders. 10 Isolated RCTs have also been conducted for St John’s wort in the treatment of social phobia and obsessive compulsive disorder (OCD) and somatoform (all NS), with other positive yet less convincing data for valerian (Valeriana officinalis) or milk thistle (Silybum marianum) in OCD 10 ; while there are promising mood-modulating candidates such as kanna (Sceletium tortuosum) 47 –49 and a standardized lavender oil, Silexan. 50 Although these data are encouraging, further replicated research of these are needed in order to subject them to meta-analysis.
In conclusion, this “meta-synthesis” of the data from 9 meta-analyses showed positive findings for a variety of plant-based medicines in a range of psychiatric disorders, albeit limited by the quality of source data. The breadth of data supports that several phytoceuticals may be an effective option for mental disorders, alongside pharmaceutical, psychology therapies, and standard lifestyle recommendations. Further robust RCTs across various jurisdictions are required to firmly validate these findings.
Supplemental Material
Supplemental Material, sj-docx-1-cpa-10.1177_0706743720979917 - Plant-based Medicines (Phytoceuticals) in the Treatment of Psychiatric Disorders: A Meta-review of Meta-analyses of Randomized Controlled Trials: Les médicaments à base de plantes (phytoceutiques) dans le traitement des troubles psychiatriques: une méta-revue des méta-analyses d’essais randomisés contrôlés
Supplemental Material, sj-docx-1-cpa-10.1177_0706743720979917 for Plant-based Medicines (Phytoceuticals) in the Treatment of Psychiatric Disorders: A Meta-review of Meta-analyses of Randomized Controlled Trials: Les médicaments à base de plantes (phytoceutiques) dans le traitement des troubles psychiatriques: une méta-revue des méta-analyses d’essais randomisés contrôlés by Jerome Sarris, Wolfgang Marx, Melanie M. Ashton, Chee H. Ng, Nicole Galvao-Coelho, Zahra Ayati, Zhang-Jin Zhang, Siegfried Kasper, Arun Ravindran, Brian H. Harvey, Adrian Lopresti, David Mischoulon, Jay Amsterdam, Lakshmi N. Yatham and Michael Berk in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Data access is not applicable to this meta-review.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.Sarris has received either presentation honoraria, travel support, clinical trial grants, book royalties, or independent consultancy payments from: Integria Healthcare & MediHerb, Pfizer, Scius Health, Key Pharmaceuticals, Taki Mai, Fiji Kava, FIT-BioCeuticals, Blackmores, Soho-Flordis, Healthworld, HealthEd, HealthMasters, Kantar Consulting, Grunbiotics, Australian Natural Therapeutics Group, Research Reviews, Elsevier, Chaminade University, International Society for Affective Disorders, Complementary Medicines Australia, SPRIM, Terry White Chemists, ANS, Society for Medicinal Plant and Natural Product Research, Sanofi-Aventis, Omega-3 Centre, the National Health and Medical Research Council, CR Roper Fellowship. C.N. had served as a consultant for Lundbeck, Grunbiotics, Servier, Janssen-Cilag, Wyeth and Eli Lilly; received research grant support from Wyeth and Lundbeck; and speaker honoraria from Servier, Lundbeck, Bristol-Myers Squibb, Organon, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Astra-Zenaca, Wyeth, and Pfizer. M.B. has received Grant/Research Support from the NIH, Cooperative Research Centre, Simons Autism Foundation, Cancer Council of Victoria, Stanley Medical Research Foundation, Medical Benefits Fund, National Health and Medical Research Council, Medical Research Futures Fund, Beyond Blue, Rotary Health, A2 milk company, Meat and Livestock Board, Woolworths, Avant, and the Harry Windsor Foundation; has been a speaker for Astra Zeneca, Lundbeck, Merck, Pfizer; and served as a consultant to Allergan, Astra Zeneca, Bioadvantex, Bionomics, Collaborative Medicinal Development, Lundbeck Merck, Pfizer, and Servier. DM has received research support from Nordic Naturals. He has provided unpaid consulting for Pharmavite LLC and Gnosis USA, Inc. He has received honoraria for speaking from the Massachusetts General Hospital Psychiatry Academy, Blackmores, Harvard Blog, and PeerPoint Medical Education Institute, LLC. He has received royalties from Lippincott Williams & Wilkins for published book “Natural Medications for Psychiatric Disorders: Considering the Alternatives.” B.H. over the past 3 years has participated in advisory boards, received honoraria from Servier, and received research funding from Servier, Lundbeck, Deakin University, Cannabis Science Inc, and HG&H Pharmaceuticals. AL has received either presentation honoraria or clinical trial grants from Arjuna Natural Ltd, Dolcas-Biotech LLC, Pharmactive Biotech Products SL, Ixoreal Biomed, Metagenics Australia, and EuroPharma Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: J. Sarris is supported by a National Health and Medical Research Council (NHMRC) Clinical Research Fellowship (APP1125000); M. Berk is supported by a NHMRC Senior Principal Research Fellowship (APP1059660 and APP1156072). W. Marx is supported by a Deakin University postdoctoral fellowship. Z.-J. Zhang is supported by the National Key R&D Program of China (2018YFC1705801). N. Galvao-Coelho is supported by the CAPES Foundation of the Ministry of Education of Brazil (Research Grant 88887.466701 / 2019-00).
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
