Abstract
Objective:
Bipolar disorder (BD) confers elevated suicide risk and associates with misaligned circadian rhythm. Real-time monitoring of objectively measured sleep is a novel approach to detect and prevent suicidal behavior. We aimed at understanding associations between subjective insomnia and actigraphy data with severity of suicidal ideation in BDs.
Methods:
This prospective cohort study comprised 76 outpatients with a BD aged 18 to 65 inclusively. Main measures included 10 consecutive days of wrist actigraphy; the Athens Insomnia Scale (AIS); the Montgomery–Åsberg Depression Rating Scale (MADRS); the Quick Inventory of Depressive Symptoms-16, self-rating (QIDS-SR-16); and the Columbia Suicide Severity Rating Scale. Diagnoses, medications, and suicide attempts were obtained from chart review.
Results:
Suicidal ideation correlated moderately with subjective insomnia (AIS with QIDS-SR-16 item 12 ρ =0.26, P = 0.03; MADRS item 10 ρ = 0.33, P = 0.003). Graphical sleep patterns showed that suicidal patients were enriched among the most fragmented sleep patterns, and this was confirmed by correlations of suicidal ideation with actigraphy data at 2 visits. Patients with lifetime suicide attempts (n = 8) had more varied objective sleep (a higher standard deviation of center of daily inactivity [0.64 vs. 0.26, P = 0.01], consolidation of daily inactivity [0.18 vs. 0.10, P = <0.001], sleep offset [3.02 hours vs. 1.90 hours, P = <0.001], and total sleep [105 vs. 69 minutes, P = 0.02], and a lower consolidation of daily inactivity [0.65 vs. 0.79, P = 0.03]).
Conclusions:
Subjective insomnia, a nonstigmatized symptom, can complement suicidality screens. Longer follow-ups and larger samples are warranted to understand whether real-time sleep monitoring predicts suicidal ideation in patient subgroups or individually.
Introduction
Bipolar disorder (BD) is associated with one of the highest suicide risks among psychiatric disorders. In long-term cohort studies and representative register–based studies, 4% to 20% of patients with BD have been reported to die by suicide. 1,2 One third to half of BD patients have a history of at least 1 suicide attempt (SA). 3 –5 Risk factors for SAs among BD patients include female gender, younger illness onset, depressive polarity of episodes, comorbid anxiety disorder, substance use disorder or cluster B personality disorder, and completed suicide in a first degree family member. 6 Unfortunately, risk factors that present a trait or are based on cross-sectional information have shown relatively low potency to predict suicidal behavior. 6 This is why predictors that fluctuate over time in intensity are of interest, including suicidal ideation (SI) and sleep. 7 Identification of increased SI or sleep problems in real-time could facilitate prevention of suicidal behavior among this high-risk population.
Several features of rest–activity rhythm have been proposed to associate with suicidality even after adjusting for depressive symptoms. 8,9 For preventive intervention, of recent interest is quality of sleep, especially insomnia and nightmares. 9 –13 In a veteran’s study, insomnia was reported in nearly half of the last doctor’s visits preceding suicide death, 14 suggesting it may be a less stigmatized indicator of suicide risk than psychiatric symptoms. The link between insomnia and/or nightmares 9 and suicide has been proposed to be in the dysfunction of the hypothalamic–pituitary–adrenal axis and hyperarousal. 10,15 The theoretical models for suicide research and prevention might profit from shifting focus from monitoring experiences of disturbed affect to consideration of fundamental aspects of vitality and psychomotor activity. 16 However, previous work has focused predominantly on associations between lifetime SAs and sleep parameters. While the predictors of ideation and attempt can be different, sleep parameters should be linked specifically with SI for prediction and prevention of SAs. Also, while insomnia and nightmares indicate problems with sleep, they are very unspecific in describing the actual characteristics of rest–activity rhythm to be targeted in treatment.
Recently, several authors have suggested that a misaligned/disrupted circadian rhythm is highly prevalent among the patients with a BD; 17,18 this is defined as an irregular, delayed/advanced, or non-24-hour sleep–wake rhythm with respect to the light–dark cycle. Given the high prevalence of both sleep problems and suicidality, a cohort of patients with BD is suitable to observe associations between sleep problems and SI, with an enrichment of both.
In this study, we aimed to further understand the association between sleep problems and the severity of SI in BD. We used both subjective insomnia and objective actigraphy data to obtain information about individual- and group-level characteristics of sleep that associate with SI. To our knowledge, this is the first study involving BD patients to examine the link between both subjective insomnia and objective sleep patterns with SI. Based on the literature supporting a relationship between subjective sleep problems and SAs trans-diagnostically, as well as a high prevalence of both circadian rhythm disturbance and suicidality in BD, we hypothesized that sleep problems would associate with severity of SI in BD.
Methods
Participants
Participants were recruited as consenting patients referred by clinicians from the Bipolar Disorders Clinic of the Douglas Mental Health University Institute (DMHUI), Montreal, Canada. Data were collected between April 2016 and June 2018. Eligibility for participation required a clinical diagnosis of BD according to DSM-5 criteria, 19 ongoing treatment at the DMHUI outpatient Bipolar Disorders Clinic, capacity to consent, and age between 18 and 65 years inclusively. Patients were excluded if they required hospitalization. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. All procedures involving human subjects/patients were approved by the Ethics Committee of the DMHUI. Written informed consent was obtained from all patients.
Procedure
Study data were collected and managed using REDCap electronic data capture tools hosted at the DMHUI. 20 Participating patients were already diagnosed with BD at the DMHUI, and medical files were reviewed for confirmation of diagnoses by a specialist in psychiatry. Information on current medication was obtained from medical records and confirmed by the patient.
Measures of Mood and Subjective Sleep
Evaluation of current mood included interviews, and surveys, completed over a course of 2 weeks. In an interview at baseline (V1) and after 2 weeks (V2), the researcher asked about mood during the past 7 days and rated mean severity of depressive symptoms using the Montgomery–Åsberg Depression Rating Scale (MADRS) 21 and the peak severity of manic symptoms using the Young Mania Rating Scale (YMRS). 22 The participants also provided subjective reports of depressive symptoms during the past 7 days by completing the Quick Inventory of Depressive Symptoms-16, self-rating (QIDS-SR-16) 23 at V1 and V2; the response to QIDS-SR-16 items 1 to 4 was summed to describe subjective insomnia during the past week. The participants responded to questions on sleep during the past month in the Athens Insomnia Scale (AIS) 24 at V1. AIS sleep score was calculated from the sum of items 1 to 5, AIS day score from the sum of items 6 to 8, and AIS total score from the sum of items 1 to 8. The current mood episode at both visits was investigator-determined (OL) using DSM-5 criteria, taking into account all information in the medical files, symptom scores, and the interview. A stable phase was defined as the absence of any DSM-5 episode and could thus include depressive, hypomanic, or mixed symptoms not fulfilling the severity or duration criteria of a DSM-5-defined episode. We used the Social and Occupational Functioning Scale (SOFAS) 19 to measure functionality.
Descriptors of Current Suicidality
Information on current suicidality was collected as part of the interviews and surveys completed at V1 and V2. Information on the Severity of SI during the past week was based on the response to the QIDS-SR-16 item 12, and MADRS item 10, which were available for all patients. We complemented the survey with the Columbia Suicide Severity Rating Scale (C-SSRS)—Adult/Adolescent (≥12 years) Quick Screen 25 after initial recognition of an association between rest–activity rhythms and suicidality; the scale was available for a subgroup of patients (n = 26).
Descriptors of Lifetime Suicidality
Information on lifetime suicidality was available from the medical files and as part of the survey. We defined the categorical descriptor Lifetime history of SA(s)’ as the presence of either patient-reported or medical-file documented lifetime SA(s).
Actigraphy Data/Descriptors of Objectively Measured Sleep
Upon entry into the study (V1), all participants were outfitted with GENEActiv actigraphy devices (ActivInsights) for measurement of (in)activity and were instructed to wear the devices continuously for 2 weeks. Sleep variables were produced from actigraphy data (for detailed methods and validation see references 26 –28 ). Briefly, Center of daily inactivity (CenDI) describes the mean direction of inactivity, measured as the daily distribution of sleep in radian angle of the sleep vector. CenDI informs about sleep phasing, which reflects chronotype. Consolidation of daily inactivity (ConDI) describes the mean length of the sum vector corrected for total duration of sleep across 24 hours, informing about the degree of daily sleep consolidation within a single night time sleep bout. Lower consolidation indicates more fragmentation of sleep. Both values were computed using the Circular Statistics Toolbox for MATLAB. We also calculated onset, offset, and total duration of sleep as described previously. 26,27 The standard deviations (SDs) of the aforementioned variables were also calculated to describe the degree of their day-to-day variation.
Categorization of Medication
We consulted the medical files for the current medication and confirmed with the participants at V2. For analysis, we categorized the medication (see Buyukkurt et al. 29 for a complete list of medications) and then used this classification to compare patients taking/not taking medication of a given compound class. This information is used for descriptive purposes. Given the effect of lithium in decreasing suicidality and regulating rest–activity rhythms, 30 we compared patients with and without lithium.
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics version 23.0 (SPSS Inc.). Normality of distribution for each dimensional variable was tested using Shapiro–Wilk test. Since most of the dimensional variables were not normally distributed, all correlation coefficients were computed using two-tailed Spearman’s rank order, and we used Mann–Whitney U-test to compare medians between categorical groups. Despite multiple testing, for descriptive purposes, significant relationships were defined by P values <0.05. A linear regression model was done with MADRS item 10 as a dependent variable and age, ConDI, and MADRS score without item 10 (SI) as independent variables.
Results
The Characteristics of the Sample
Of 130 patients offered participation, 81 consented to enroll and completed V1. A total of 76 patients were included in analyses after removing incomplete data due to dropout relating to hospitalization (n = 2) or withdrawal (n = 2), or insufficient actigraphy data (n = 1). The median age was 49 (interquartile range [IQR] = 19.75) years, and half (n = 37, 49%) were women. Clinical diagnoses were as follows: 40 patients (53%) had BD type I, 32 (42%) had BD type II, 3 (4%) had a schizoaffective disorder, bipolar type, and 1 (1%) had BD not otherwise specified. Current episode assessment at V1 showed that most patients were stable (n = 38, 50%) or had a major depressive episode (n = 29, 38%), while 3 had a hypomanic episode, 3 major depressive episode (MDE) and (hypo)manic symptoms, and 3 hypomania and depressive symptoms. Twenty (26%) were using lithium, 48 (63%) had anticonvulsants, 52 (68%) antipsychotics, and 37 (49%) had antidepressants. Polypharmacy was common; 56 (74%) patients were treated with medication from at least 2 of these classes.
The median severity of current depressive symptoms at V1 was in the category of mild depressive symptoms (median MADRS score 14, range 0 to 45, IQR = 20.75). The median severity of the manic symptoms was in the category of no manic symptoms (median YMRS score 5, range 0 to 30, IQR = 9.00). Median SOFAS was 55.0 (IQR = 27.0).
Prevalence of Current and Lifetime Suicidality
Lifetime SAs were reported for 8/76 (11%) patients. Using all sources of information (survey, interview, and mood chart during the study), any current SI (report of suicidal thoughts with or without intent to die, as opposed to having no wish to die or the feeling that life is not worth living) was reported in 33 patients (43%), and 5 of them had lifetime SAs.
Correlates of Current SI
AIS total and day scores during the past month and the QIDS-SR-16 sleep score for the past week correlated moderately with SI at V1 and V2 (Table 1).
Correlations of Subjective Sleep with Suicidal Ideation in a Sample of Patients with a Bipolar Disorder.
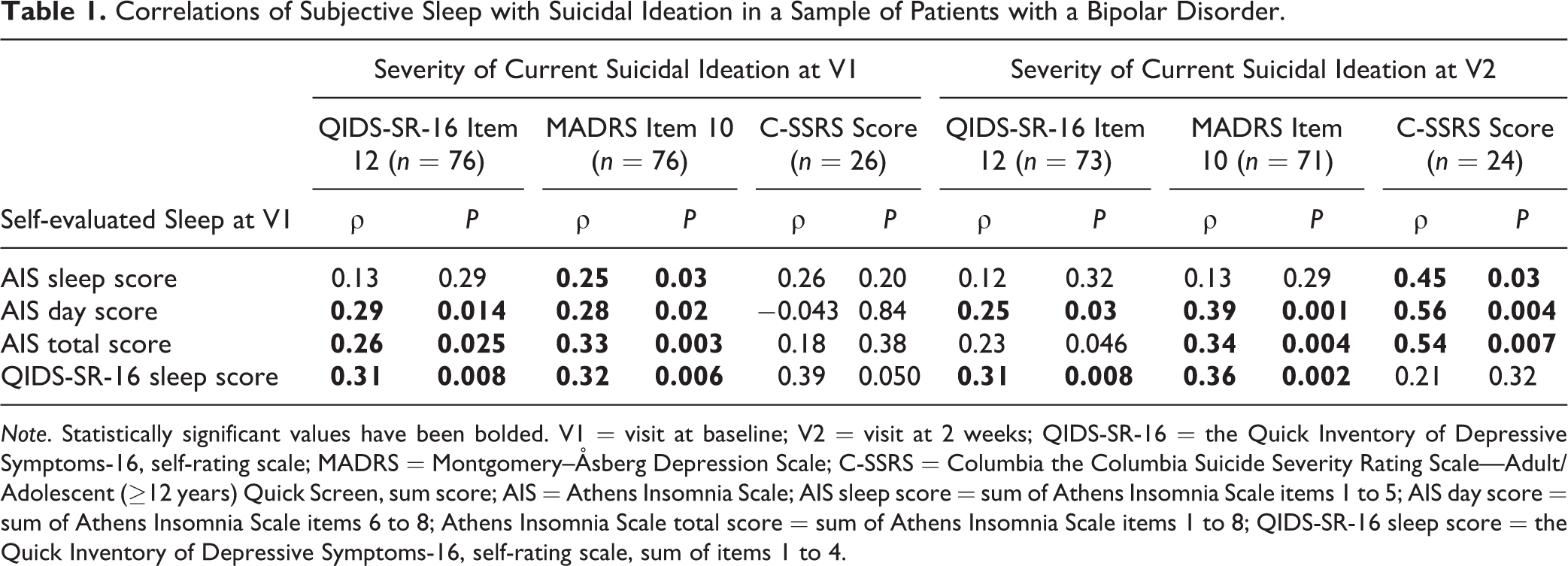
Note. Statistically significant values have been bolded. V1 = visit at baseline; V2 = visit at 2 weeks; QIDS-SR-16 = the Quick Inventory of Depressive Symptoms-16, self-rating scale; MADRS = Montgomery–Åsberg Depression Scale; C-SSRS = Columbia the Columbia Suicide Severity Rating Scale—Adult/Adolescent (≥12 years) Quick Screen, sum score; AIS = Athens Insomnia Scale; AIS sleep score = sum of Athens Insomnia Scale items 1 to 5; AIS day score = sum of Athens Insomnia Scale items 6 to 8; Athens Insomnia Scale total score = sum of Athens Insomnia Scale items 1 to 8; QIDS-SR-16 sleep score = the Quick Inventory of Depressive Symptoms-16, self-rating scale, sum of items 1 to 4.
Correlations between actigraphy measures and SI were weak to moderate (Table 2). The following sleep variables were associated with increased SI measured by at least 1 measure for SI: less consolidated sleep, more variation in consolidation, earlier onset, and later offset. However, comparing findings across different measures for SI as well as findings at 2 visits, the correlations were not consistent. Depressive symptoms, lower functionality, and anxiety correlated with SI (Table 3).
Correlations of Actigraphy Data with Suicidal Ideation at V1 and V2 in a Sample of Patients with a Bipolar Disorder.
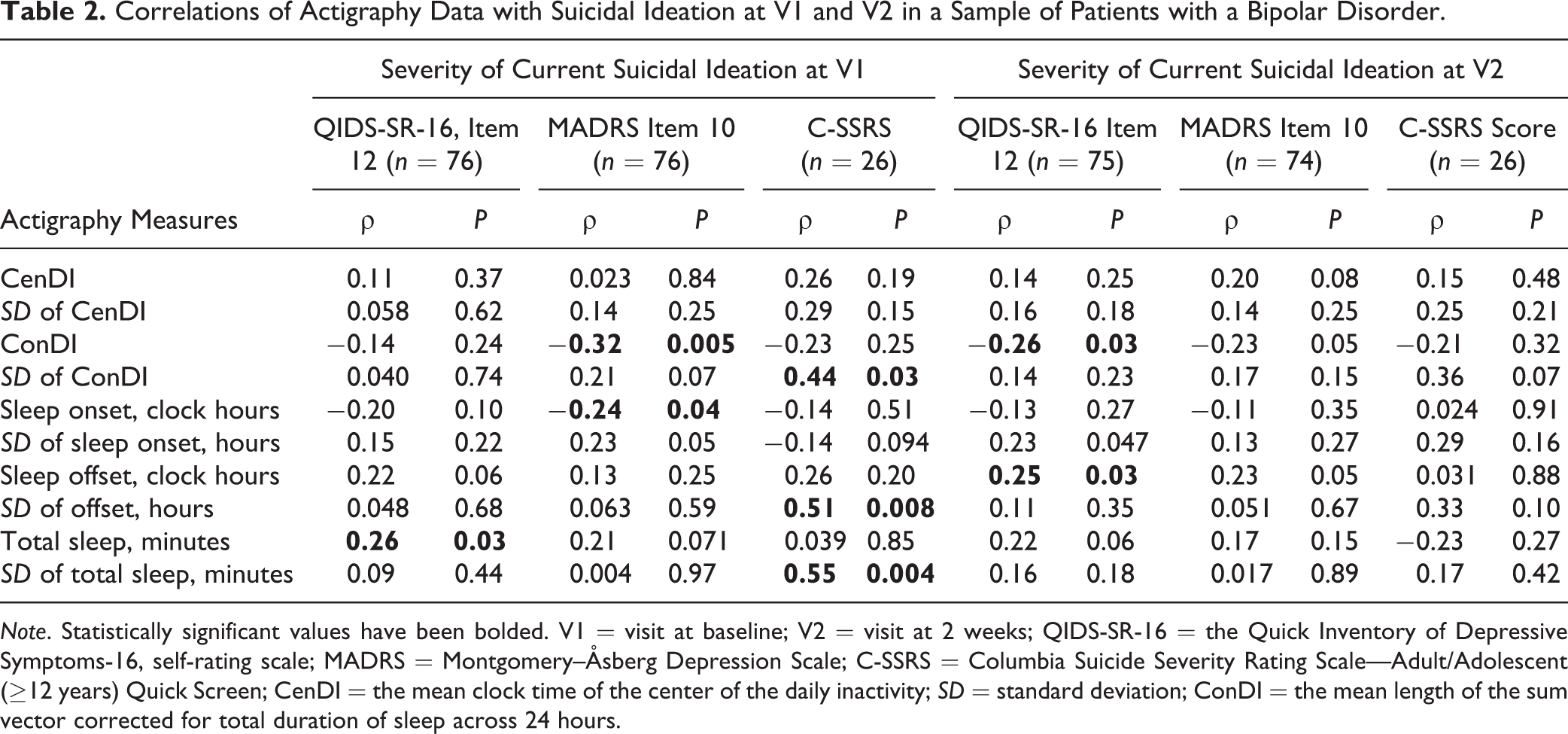
Note. Statistically significant values have been bolded. V1 = visit at baseline; V2 = visit at 2 weeks; QIDS-SR-16 = the Quick Inventory of Depressive Symptoms-16, self-rating scale; MADRS = Montgomery–Åsberg Depression Scale; C-SSRS = Columbia Suicide Severity Rating Scale—Adult/Adolescent (≥12 years) Quick Screen; CenDI = the mean clock time of the center of the daily inactivity; SD = standard deviation; ConDI = the mean length of the sum vector corrected for total duration of sleep across 24 hours.
Correlations of Other Clinical Features with Suicidal Ideation.
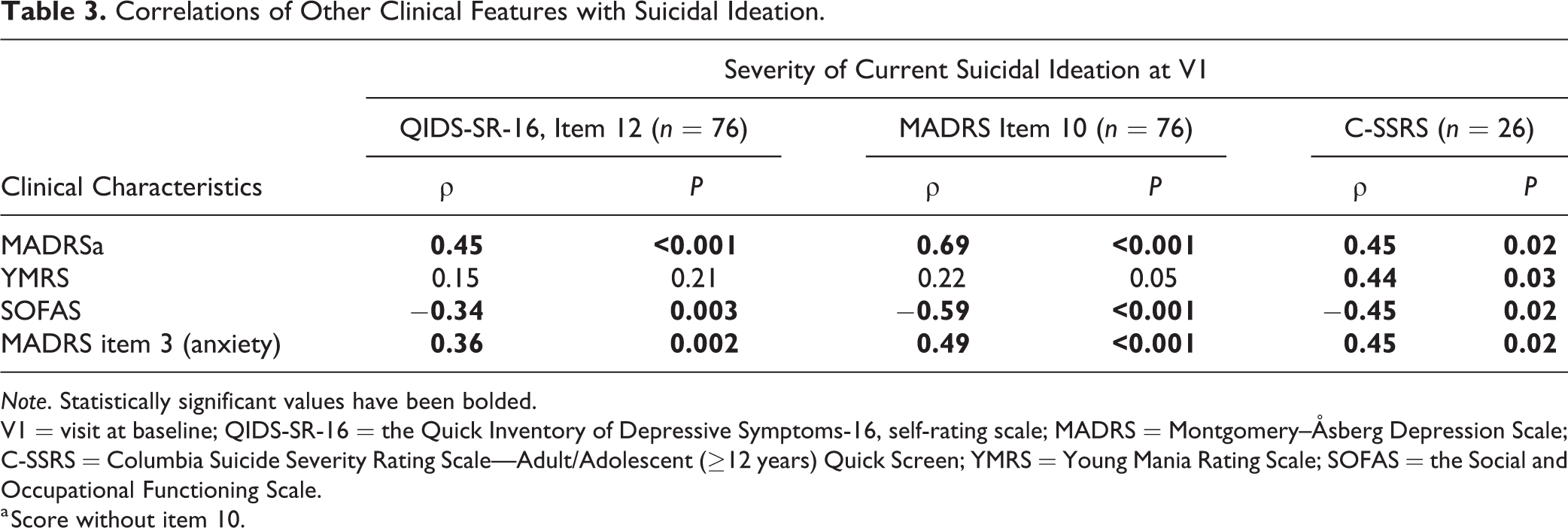
Note. Statistically significant values have been bolded.
V1 = visit at baseline; QIDS-SR-16 = the Quick Inventory of Depressive Symptoms-16, self-rating scale; MADRS = Montgomery–Åsberg Depression Scale; C-SSRS = Columbia Suicide Severity Rating Scale—Adult/Adolescent (≥12 years) Quick Screen; YMRS = Young Mania Rating Scale; SOFAS = the Social and Occupational Functioning Scale.
a Score without item 10.
To further characterize the association of severity of suicidality with actigraphy data, we divided patients into 4 groups based on multiple information sources (interview, survey, and mood chart). The groups were: no SI = 0 (n = 45), life is not worth living but no SI = 1 (n = 17), SI but no plan = 2 (n = 5), and SI and plan = 3 (n = 9). The boxplot of ConDI in these groups (Figure 1) as well as a display of the individual patients’ inactograms across a 10-day recording period ranked according to the degree of sleep fragmentation (= decreasing ConDI; Supplemental Material 1) shows how patients with no SI are highly variable in their ConDI and similar to those who report feeling that life is not worth living. Patients with SI belong to those with the most fragmented sleep.
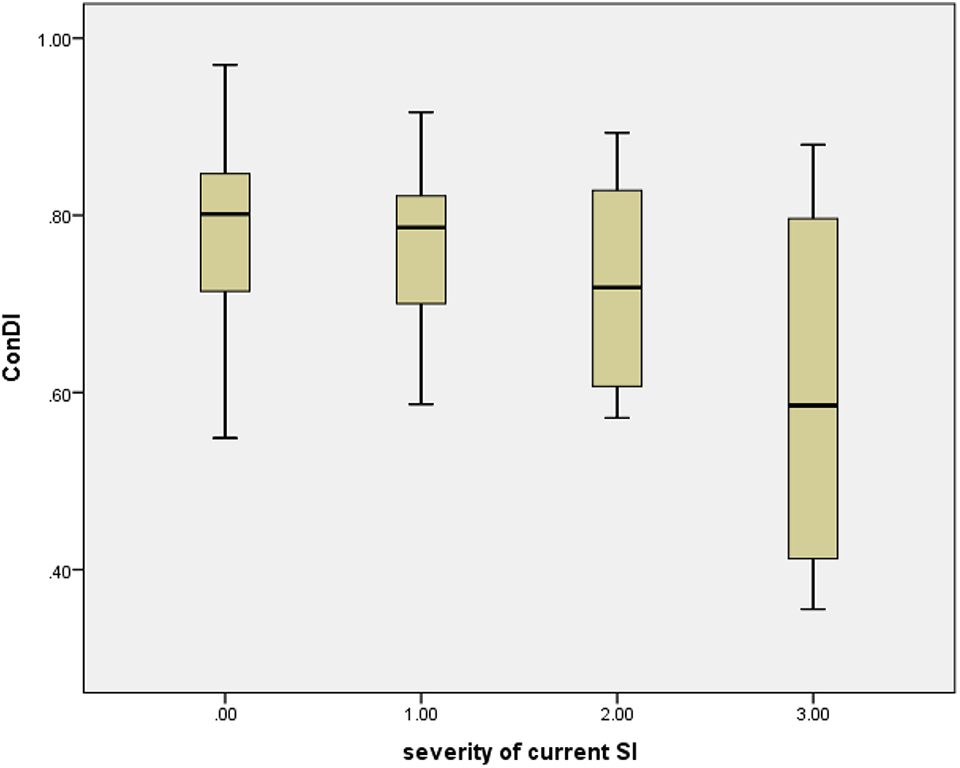
Consolidation of daily inactivity (ConDI) in 4 groups of patients with increasing level of suicidal ideation (SI). Legend: severity of current SI: 0 = no, 1 = feeling that life is not worth living; 2 = SI without intent to die; 3 = severe SI.
Correlations of Sleep Descriptors with Lifetime Suicidality
Patients with a lifetime history of SA had a higher SI score (MADRS item 10 1.5 vs. 0.0, P = 0.03; C-SSRS 2.0 vs. 0.0, P = 0.02). Subjective report of sleep did not differ between patients with and without lifetime SA. However, patients with lifetime SAs (n = 8) had more varied sleep (a higher SD of CenDI, ConDI, sleep offset, and total sleep), and a lower ConDI (Table 4). The display of inactograms ordered by increasing SD of sleep consolidation corroborates this finding as patients with a lifetime SAs were concentrated among the most fragmented sleep (Supplemental Material 2).
Comparison of Sleep between Patients with and without Lifetime Suicide Attempts.
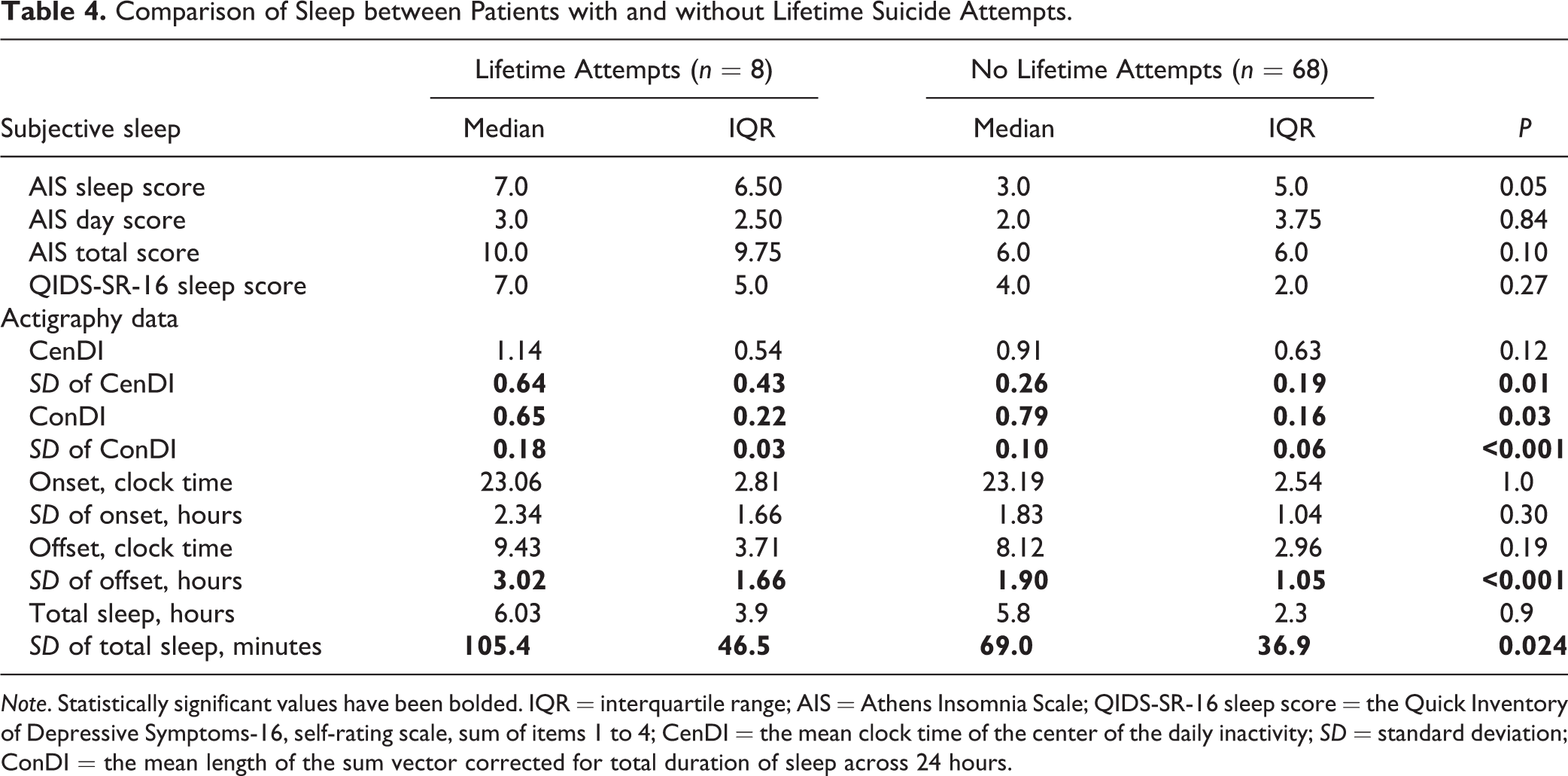
Note. Statistically significant values have been bolded. IQR = interquartile range; AIS = Athens Insomnia Scale; QIDS-SR-16 sleep score = the Quick Inventory of Depressive Symptoms-16, self-rating scale, sum of items 1 to 4; CenDI = the mean clock time of the center of the daily inactivity; SD = standard deviation; ConDI = the mean length of the sum vector corrected for total duration of sleep across 24 hours.
We compared severity of SI between other subgroups (gender; patients with and without lithium; type I and II BD) but found no differences in these comparisons (data not shown).
A Regression Model
In a linear regression model with MADRS item 10 as the dependent variable, ConDI (β = −0.34, P = 0.003) remained statistically significant after controlling for age (β = 0.10, P = 0.36) and presence of lifetime suicidality (standardized β = 0.21, P = 0.07). However, after controlling for depressive symptoms, depressive symptoms were the strongest correlate (β = 0.51, P = <0.001), followed by lifetime SAs (β = 0.18, P = 0.06), ConDI (β = −0.18, P = 0.08), and age (β = 0.027, P = 0.77). For the latter model, R = 0.65, P < 0.001.
Discussion
In this analysis including 76 patients with a BD, current SI associated moderately with subjective insomnia. Objectively, the patients with the most severe current SI showed disrupted sleep (low and variable ConDI). Moreover, patients with lifetime SAs were concentrated among the patients with the most fragmented sleep and late sleep offset. Thus, fragmented/nonconsolidated sleep characterized patients with risk of suicidality, in line with some previous work in other populations. 11 –13 Notably, the effect of sleep fragmentation on SI remained a trend even after controlling for the depressive symptoms.
A link between fragmented sleep and SI is supported by a previous study showing an association between higher nocturnal wakefulness and SI in a sample of depressed patients. 31 A recent study comparing patients with a BD and a lifetime history of SAs (n = 57) or not (n = 90) to healthy controls (n = 89) did not find statistically significant difference in fragmentation index between patients with and without lifetime SAs. 8 This might be due to methodological differences: They used commercial software to produce a fragmentation index, calculated as the amount of time associated with movement during the sleep period, expressed as percentage. Sleep period is calculated based on sleep onset and offset, which are less reliably estimated in fragmented sleep. We addressed this using recent open-source algorithms, which are device independent, have been validated against polysomnography, and we added use of a moving time window as described in detail previously. 26
Late sleep offset was found to associate with lifetime suicidality in our sample, consistent with results from a large Korean study. 32 This finding necessitates further work given the small number of patients with SAs in our data. Furthermore, the finding differs from the association between early wakening and previous SAs in a larger actigraphy study involving BD patients. 8
While depressive symptoms and anxiety were robust correlates of SI, sleep problems are supposedly less stigmatizing for screening purposes. Strikingly, it is estimated that 40% to 60% of suicide completers seek help for sleep problems during their final year, 33,34 while up to 78% of patients who died by suicide had denied SI in preceding clinical encounters. 35 Other studies have shown that nightmares, 9 subjective insomnia, early sleep offset, and vigorous circadian rhythm predict SI or SA independent of depressive symptoms. 8 Similarly, it has been shown that sleep complaints associate with SA independent of psychiatric diagnosis. 36
Our results underscore the clinical importance of recognizing and understanding the role of efficient individualized detection and treatment of sleep problems to prevent suicide. We expect either direct causality, sleep having an impact on SI, or a shared underlying cause for disturbed sleep and SI. In both cases, improvement of sleep can be expected to have a positive impact on SI. Based on our data, subjective insomnia may be an important state factor indicative of suicide risk. However, the extent to which it predicts immediacy of risk remains uncertain as self-evaluated sleep over both the past week and month correlated with current SI. Additionally, objectively measured fragmented sleep cannot be described as a specific marker of acute suicide risk given that both patients with current SI and those without current SI had fragmented sleep. As such, an alternative interpretation may be that features of rest–activity rhythm could indicate trait sensitivity to external factors and/or weakened internal rhythmic strength among those with risk for suicide.
A major strength of this study is the use of objective actigraphy measures derived from wrist-worn accelometric device data to describe rest–activity rhythms. 37 Actigraphy is very well validated to objectively track a patient’s rest–activity patterns in the home environment, is optimal for real-time monitoring, and permits evaluation of everyday stressors that could potentiate both insomnia and SI. This is the first study to examine the link between objective sleep and SI in BD with a registration of inactivity for a period that allows pattern analysis. 38 Although our findings do not support universal cutoffs for increased suicide risk based on objective sleep parameters in primary prediction and prevention of suicidality, we cannot exclude potential utility at an individual or patient subgroup level. Artificial intelligence designed to collect both symptom scores and actigraphy data for detection and notification in real time about an increase in individual risk factors for suicidality may prove clinically useful. Accordingly, larger data sets and longer follow-up of the temporal relation between sleep and SI as well as other predictors of SI are a requisite to understand whether real-time monitoring may be beneficial.
Our study was limited by an insufficiently long data collection period to capture suicidal behavior and the exclusion of several other well-established predictors of SI. It is known that illness phase predicts SA, the highest risk in BD being during mixed states and major depressive episodes. 39,40 A longer time spent in these high-risk states is likely to explain a higher cumulative incidence of SA. Comorbid personality disorders or other predisposing characteristics may increase the risk of SAs by adding to the time patients spend in depressive episodes or by moderating the risk directly. 41 Depressive symptoms, hopelessness, impulsiveness, and arousal/sleep patterns, as predictors of SI, are strongly correlated. All of these risk factors associate with acute insomnia, vulnerability to react to external stressors with insomnia, and chronic sleep problems. Thus, longer follow-ups are necessary to understand causality between sleep problems, rest–activity rhythms, and increased SI or suicidal behavior.
Further limitations in our methodology deserve consideration. While information on the presence and severity of SI was available from QIDS-SR-16 and MADRS for the total data set, a more accurate measure for SI, C-SSRS was only available for a subgroup. However, use of complementary methods to assess presence and severity of current SI, as well as lifetime suicidality, may be considered a strength. The 3 measures for the severity of current SI differ somewhat in their grading from the feeling that life is not worth living to having an active plan to commit suicide. This is likely to explain at least partly the differences in the statistical significance of the findings, while consistency is robust in the direction and approximate size of the correlations. In addition, we did not complete a diagnostic interview to confirm the diagnosis of BDs; however, since a transdiagnostic approach to suicidal behavior is increasingly used in research (for recent examples, see Britton et al. and Podlogar et al. 42,43 ), this is unlikely to bias our findings. Further, information pertaining to medications and compliance was not included in analysis which may be of particular interest given the supposed circadian rhythm stabilizing effect of lithium. 30 Given the lack of previous data with a focus on sleep patterns/ inactivity, this is an exploratory study. For descriptive purposes, we use P < 0.05; we consider as main findings those that show consistent significance in several descriptors of sleep and SI. We also present a regression model to evaluate the proportion of SI explained by rest–activity rhythm. The generalizability of our study is limited by the removal of suicidal patients requiring hospitalization from the study, the tertiary care setting, and the single site sampling. Last, comparability to other studies is limited by the use of nontraditional actigraphy markers. Our markers were selected based on the validation of actigraphy data with patient-reported sleep onset/offset and our preference to focus on inactivity rather than activity to mitigate confounders. However, to facilitate reproducibility, we have published algorithms for inactograms 26 and used open-source algorithms for the dimensional measures.
To conclude, insomnia as a nonstigmatized symptom can complement depressive symptoms and anxiety as a signal for potential SI. Unstable rest–activity rhythms correlated with lifetime suicidality. Clinical trials using psychotherapy, chronobiological treatments, and individualized psychoeducation specifically aiming at improving subjective insomnia, and sleep pattern aberrations are warranted to assess the potential of a sleep focus in prevention of suicide. Furthermore, analysis of long-term wrist actigraphy data that may involve approaches based on artificial intelligence could additionally provide objective real-time predictors for SI at the individual level.
Supplemental Material
Supplement_1_ConDI - Suicidal Ideation and Insomnia in Bipolar Disorders: Idéation suicidaire et insomnie dans les troubles bipolaires
Supplement_1_ConDI for Suicidal Ideation and Insomnia in Bipolar Disorders: Idéation suicidaire et insomnie dans les troubles bipolaires by Lia Bertrand, Clément Bourguignon, Serge Beaulieu, Kai-Florian Storch and Outi Linnaranta in The Canadian Journal of Psychiatry
Supplemental Material
Supplement_2_lt - Suicidal Ideation and Insomnia in Bipolar Disorders: Idéation suicidaire et insomnie dans les troubles bipolaires
Supplement_2_lt for Suicidal Ideation and Insomnia in Bipolar Disorders: Idéation suicidaire et insomnie dans les troubles bipolaires by Lia Bertrand, Clément Bourguignon, Serge Beaulieu, Kai-Florian Storch and Outi Linnaranta in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
We present individual sleep data in the panel. Other data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to being from a vulnerable population. None of funding sources participated in study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the article for publication.
Acknowledgments
We thank all study participants and the personnel of the Bipolar Disorders Clinic at the DMHUI Mental Health University Institute for help in data collection, and Sybille Saury, Olivia Crescenzi, Josée Martin, and Vasile Catana for help in data management.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Fonds de Recherche du Québec—Santé (FRQS) #8400863 and 252872 (OL), and CIHR #366858 and NSERC: RGPIN-2015-04034 (K-FS), and the Mach-Gaensslen Foundation Bursary/Edward Beatty Research Award/Dr. Clarke K. McLeod Memorial Scholarship (LB).
Supplemental Material
The supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
