Abstract
Suicidal ideation and behavior are among the most severe psychiatric presentations, warranting emergency room visits and psychiatric admission for higher levels of care. In the United States, suicide rates continue to climb, especially in younger patients, and the continued psychosocial stressors of the COVID-19 pandemic may further exacerbate this crisis. Suicidal ideation and behavior are core features of a major depressive episode, but there are limited treatment options to rapidly redress these life-threatening symptoms. Racemic ketamine and its S-enantiomer, esketamine, are N-methyl-D-aspartate receptor antagonists and glutamate modulators that have robust antidepressant efficacy in treatment-resistant major depressive disorder and bipolar depression. Additionally, both ketamine and esketamine have demonstrated rapid-acting antisuicidal efficacy in major mood disorders. In August 2020, this culminated in a first-in-class approval of Spravato® (intranasal esketamine) for the treatment of major depressive disorder with acute suicidal ideation and behavior. In this article, we review the literature in support of the antisuicidal efficacy of ketamine and esketamine.
Keywords
Introduction
Recurrent thoughts of death, or suicidal ideation (SI), is one of the core symptoms of major depressive disorder (MDD). In 2010, 73% of suicide attempts in people with MDD occurred during a major depressive episode (MDE).
1
SI and behavior are also often reported in schizophrenia, substance use disorders, and other neuropsychiatric illnesses. Strikingly, the incidence of suicide has steadily increased over the last 20 years,
2
and in 2019, 4.8% of U.S. adults reported serious thoughts of suicide.
3
Individuals that later die by suicide show a greater amount of healthcare visits, specifically in emergency and inpatient settings.
4
Current treatments to reduce the occurrence of SI and behavior, such as antidepressants, lithium, clozapine, psychotherapy, electroconvulsive therapy, and transcranial magnetic stimulation, are not immediate nor effective for all patients. Moreover, selective serotonin reuptake inhibitors come with a warning for children and adolescents for potential
Racemic ketamine, and its S-enantiomer esketamine, are N-methyl-D-aspartate (NMDA) receptor antagonists and are hypothesized to exert their rapid antidepressant effects via enhanced glutamate signaling. 7 Where typical antidepressants take weeks-to-months for maximal efficacy, (es)ketamine exerts antidepressant effects within hours of administration, making it distinctly suited to treat acute SI. 8 (Es)ketamine may also treat trans-diagnostic aspects of SI, such as hopelessness and anhedonia. 9 This opportunity to use (es)ketamine to rapidly treat SI at common sites of patient contact may reduce the national rate of suicidal behaviors.
The purpose of the present manuscript is to review the literature of ketamine and esketamine’s efficacy on suicidality. This selection of studies spans from inception until June 2022 and focuses on studies where suicidality was the main outcome and/or studies that specifically recruited participants with baseline suicidality.
Ketamine
As illustrated in Tables 1–6 and outlined below, subanesthetic dose ketamine treatments have been studied in multiple treatment settings for depressed and suicidal patients.
Ketamine's effects on suicidal ideation in the emergency department.

BHS, Beck Hopelessness Scale; BSS, Beck Scale for Suicidal Ideation; IV, Intravenous; MADRS-SI, Suicide item of Montgomery-Åsberg Depression Rating Scale; SSI, Scale for Suicidal Ideation.
Ketamine's effect on suicidal ideation in inpatient units.
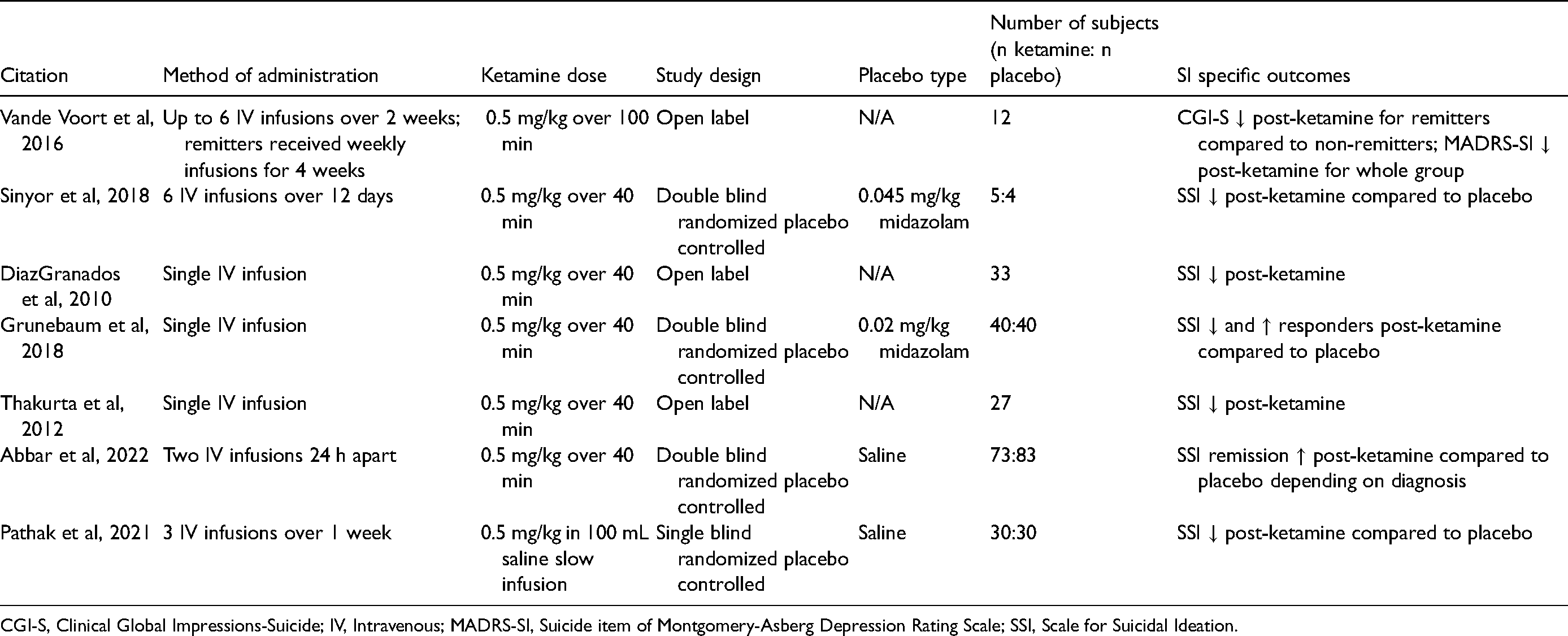
CGI-S, Clinical Global Impressions-Suicide; IV, Intravenous; MADRS-SI, Suicide item of Montgomery-Asberg Depression Rating Scale; SSI, Scale for Suicidal Ideation.
Ketamine's effects on suicidal ideation in outpatient clinics.

BSS, Beck Scale for Suicidal Ideation; ECT, Electroconvulsive Therapy; IM, Intramuscular; IV, intravenous; MADRS-SI, Suicide item of the Montgomery-Asberg Depression Rating Scale; QIDS-SR, Quick Inventory of Depression Symptomatology-Self Report; SI, Suicidal Ideation.
Ketamine's independent effects on depression and suicidal ideation.

BSS: Beck Scale for Suicidal Ideation; IV: Intravenous; MADRS–SI: Suicide item of the Montgomery–Asberg Depression Rating Scale; QIDS–SI: Suicide item of the Quick Inventory of Depression Symptomatology; SSI: Scale for Suicidal Ideation.
Ketamine's effects on suicidal ideation mediated by depression improvement.
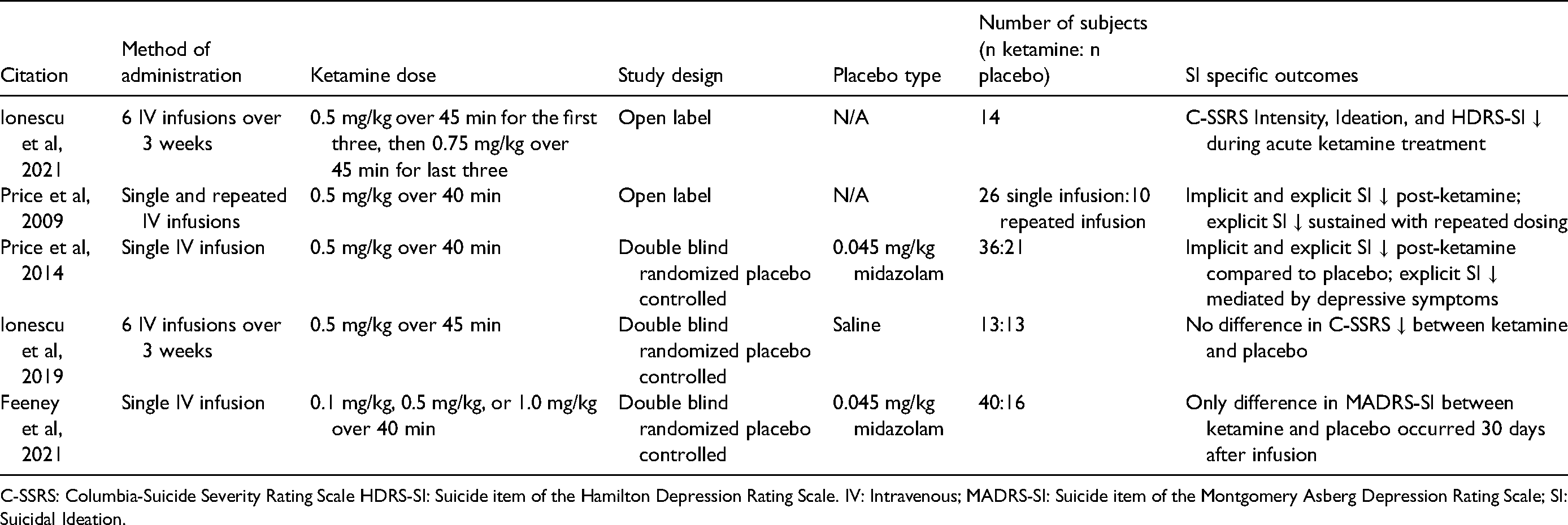
C-SSRS: Columbia-Suicide Severity Rating Scale HDRS-SI: Suicide item of the Hamilton Depression Rating Scale. IV: Intravenous; MADRS-SI: Suicide item of the Montgomery Asberg Depression Rating Scale; SI: Suicidal Ideation.
Meta-analyses of ketamine's antisuicidal effects.
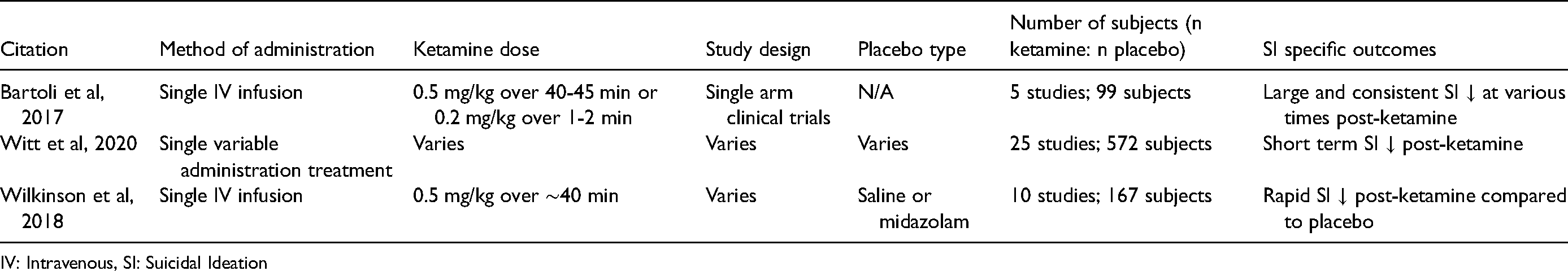
IV: Intravenous, SI: Suicidal Ideation
Ketamine’s Antisuicidal Efficacy (Subdivided by Location of Administration)
Emergency Settings
Due to its rapid-acting effects, ketamine has shown promise with patients in crisis presenting to emergency departments (ED) (Table 1).
Kashani et al’s ED study of patients with SI revealed that a single bolus infusion of ketamine reduced the percentage of participants hospitalized from 25.5% to 8.2% at the 10 day post-administration timepoint. 10 The greatest decreases in SI occurred at 40 minutes, as measured by the Scale for Suicidal Ideation (SSI), with a significant difference from baseline lasting throughout the 120-minute evaluation period. Yet, there was an incomplete reduction of SI, with 6.1% of participants having continued SI at the 10-day time point. Next, Burger and colleagues studied active-duty military patients with depression and acute suicidality awaiting inpatient psychiatric admission from the ED. 11 These participants were given a single infusion of either ketamine (0.2 mg/kg over 2 min) or saline placebo. 67% of the treatment group showed significant improvement in the Beck Scale for Suicidal Ideation (BSS) and the Beck Hopelessness Scale (BHS) compared to placebo at 4 hours post-infusion, and BHS remained significantly improved in the treatment group until discharge. Following discharge, both groups received treatment as usual and the differences between ketamine and placebo diminished, suggesting a transient antisuicidal response.
In another ED study of patients awaiting hospitalization, Domany and colleagues recruited participants with depression and SI. 12 These subjects were given a single ketamine (0.2 mg/kg over 5 minutes) or saline infusion; ketamine was associated with significant improvements over placebo in the BSS from 90 to 180 minutes after administration. The suicide item of the Montgomery-Åsberg Depression Rating Scale (MADRS-SI) also significantly improved in the ketamine group at 120 minutes, with 88% of the ketamine group scoring <2 at 90 minutes, compared to only 33% in the placebo group. Domany and colleagues also measured suicide-related Research Domain Criteria (RDoC) constructs: hopelessness, pessimistic thoughts, anxiety, and impulsivity. Hopelessness (BHS) decreased by 90.7% in the ketamine group, compared to 17.5% in the placebo group, at 3 days post-treatment. Pessimistic thoughts (MADRS-9) were significantly reduced at 15 minutes, 60 minutes, and 3 days post-infusion. Potential threat [Beck Anxiety Index (BAI)] improved by 82.4% with ketamine compared to 47.3% with placebo, and inner tension (MADRS-3) was significantly lower with ketamine at 30 minutes. In this small cohort, ketamine did not modify impulsivity [Barratt Impulsiveness Scale (BIS)]. The same group assessed the efficacy of intranasal ketamine in emergency room-presenting patients requiring psychiatric hospitalization for suicidal ideation independent of psychiatric diagnosis. 13 In this study, participants were given either a single fixed dose of 40 mg ketamine or saline intranasally. There was a significant group reduction in MADRS-SI scores 4 hours post-treatment, but no significant interactive (group-by-time) effects, as the ketamine group demonstrated decreased SI for up to 28 days after the treatment (of note, other treatment changes were permitted). Additionally, 80% of the ketamine group, compared to only 33% of the placebo group, had complete remission of SI (MADRS-SI = 0). This study replicates ketamine’s antisuicidal efficacy in Domany and colleagues' prior work, 12 and work from Burger and colleagues. 11
Taken together, these studies demonstrated the antisuicidal effects of low-dose ketamine administered acutely in the emergency department. The several-minute administration of ketamine, instead of the prototypical ≥40-minute slow infusion, appears to be rapidly effective in busy ED settings, where prolonged infusion monitoring might not feasible or practical due to staff burden, such as competing demands and/or shortages, but most importantly, facilitates immediate crisis stabilization, for those in critical need.
Psychiatric hospitals
Ketamine has also shown antisuicidal efficacy in the psychiatric inpatient milieu, where patients can be monitored for longer periods of time than in the ED (Table 2).
Vande Voort et al recruited patients with treatment-resistant major depressive disorder or bipolar disorder that were hospitalized for acute SI [based on the suicide item on the Patient Health Questionnaire-9 (PHQ-9)] and in a current MDE of 1–2 years. 14 Participants received 3 ketamine infusions (0.5 mg/kg over 100 minutes)/week for up to 2 weeks, and upon achieving remission (MADRS ≤9), were switched to once-weekly infusions for 4 weeks. They found that SI remitters had significant reductions from baseline to the end of the acute treatment phase. Next, in Sinyor et al, hospitalized suicidal patients in an active MDE were given 6 ketamine or midazolam infusions in addition to treatment as usual. 15 The ketamine group, who were more treatment resistant at baseline, had an 84% reduction in SSI and 68% reduction in MADRS-SI scores by the end of the study, with effects lasting for around a month. In comparison, the midazolam group’s SSI and MADRS-SI scores were reduced by 37% and 29%, respectively. In DiazGranados et al, MDD subjects admitted to an inpatient research unit were administered subanesthetic dose ketamine (0.5 mg/kg over 40 minutes) or saline placebo in a cross-over, double-blind design. 16 By the end of the infusion, SSI, MADRS-SI, and the suicide items of the Beck Depression Index (BDI-SI) and Hamilton Depression Rating Scale (HDRS-SI) significantly improved. The effect sizes were also larger for improvement of depression, anxiety, and hopelessness in patients with high baseline SI (SSI >3) compared to patients with lower baseline SI. In a similar inpatient setting, Grunebaum and colleagues randomized MDD subjects with clinically significant SI to either ketamine or midazolam. 17 The authors reported a 4.96-point greater improvement in SSI 24 hours after ketamine compared to midazolam. One day after the infusion, 55% of the ketamine group were SI responders compared to only 30% of the midazolam group. Midazolam non-responders then received open-label ketamine, resulting in significant improvements in SSI.
Thakurta et al administered a single subanesthetic dose ketamine infusion to treatment- resistant depression (TRD) inpatients. In their open-label design, they revealed a robust (80%) but transient (up to 4 hours) improvement in SSI. 18 Similarly, HDRS-SI was 70% reduced from baseline to 40 minutes, with significance persisting until 4 hours. Next, in a study from seven teaching hospitals, suicidal inpatients were randomized to receive two daily infusions of ketamine or saline. 19 Subjects were stratified into unipolar, bipolar, and other psychiatric disorders. Interestingly, at day 3, there was a significant effect of diagnosis, with bipolar patients driving the antisuicidal response. Psychological pain, as detected by the Physical and Psychological Pain Visual Analog Scale (PPP-VAS) mediated the antisuicidal response in this cohort. 19 Finally, Pathak and colleagues assessed participants in a MDE with current SI (Modified Scale for Suicidal Ideations >20) given 3 infusions of either ketamine or placebo. 20 They found significant reductions in SI from baseline to each day for the ketamine group, with the greatest reduction occurring 6 hours after the first infusion.
The studies outlined above illustrate ketamine’s efficacy for treating SI in an inpatient setting. Ketamine use in the inpatient milieu may allow for shorter admission periods and reduce the rate of suicidal behavior post-discharge.
Outpatient clinics
Suicidal thinking that does not necessitate emergency evaluation or inpatient hospitalization is a frequent grievance for many depressed patients in outpatient clinics (Table 3). Yet, it requires more rapid and robust response than what traditional antidepressants or psychotherapies provide, making ketamine an attractive candidate.
In Kheirabadi et al, depressed patients with either treatment resistance, SI, severe symptoms, or agitation received an acute series of 0.5 mg/kg intramuscular ketamine, 1 mg/kg oral ketamine, or ECT. 21 The authors found significant improvements in BSS after all treatments, with ketamine showing superiority at 24 hours after a single treatment and 2 weeks after a full treatment course. Overall, the antisuicidal effects of these treatments lasted for approximately one month post-treatment. Next, McIntyre et al conducted a naturalistic study of treatment-refractory unipolar and bipolar depressed subjects given 4 subanesthetic dose ketamine infusions. 22 The first two infusions were 0.5 mg/kg, and dosing could be maintained or increased depending on change in Quick Inventory for Depression Symptomatology (QIDS) score. In participants with baseline SI, QIDS-SI significantly improved from baseline after each infusion and lasted for approximately one week after the final infusion. Murrough et al conducted a randomized, midazolam-controlled trial in outpatients with clinically significant SI, excluding those with current intent. 23 In comparison to midazolam, ketamine significantly lowered MADRS-SI and BSS at 24 hours and 48 hours post-infusion, respectively. However, the rapid antisuicidal effect was not sustained, as there were no differences in SI between treatment groups one week post-treatment. Next, Fan and colleagues conducted a midazolam controlled randomized clinical trial (RCT) in newly diagnosed cancer patients. 24 In comparison to midazolam, BSS and MADRS-SI were significantly lower after a single ketamine infusion on days 1 and 3. This provides initial evidence that ketamine can rapidly improve SI in the presence of significant medical comorbidities. Hu et al conducted a RCT in patients with severe MDD (HAMD-17 total score ≥24) and SI (HAMD-SI ≥1), excluding those with a suicide attempt during the current depressive episode. 25 These subjects received escitalopram (10 mg/day) and either subanesthetic dose ketamine or saline placebo for 4 weeks. From 2 to 72 hours post-infusion, QIDS-SI was significantly lower in the ketamine group. Finally, ultra-treatment resistant depressed patients, as defined by meeting three of the following criteria: major depressive episode, failed ≥5 antidepressant medications, failed or restrictions for ECT, and/or active suicidality, were administered six subanesthetic dose ketamine infusions (0.5 mg/kg) over 2–3 weeks. 26 Even in this highly refractory suicidal sample, 69% of patients with mild baseline SI had a score of 0 on SI subscales throughout treatment.
The above studies illustrate that ketamine is an effective treatment for suicidal thinking that does not necessitate crisis stabilization or inpatient hospitalization. In such cases, ketamine may be an effective adjunctive treatment, allowing patients to continue outpatient care and daily activities.
Relationship Between Antidepressant and Antisuicidal Effects (Independence vs Mediation)
Independence
The following studies illustrate ketamine’s distinct antisuicidal effects, resulting in improvement in SI alone, without improvement in other depressive symptoms (Table 4).
Grunebaum et al conducted a midazolam-controlled trial in bipolar participants with clinically significant SI (SSI ≥4). 27 In the ketamine group, SSI significantly differed at 4 hours, 1 day, 1 week, and 5 weeks post-infusion. 57% of participants given ketamine were first day responders and 43% were first day remitters, compared to only 11% responders and 11% remitters to midazolam. Interestingly, change in HDRS only explained 51% of the variance in SSI improvement. Next, in a cross-over design, participants with TRD and suicidal thinking (MADRS-SI ≥2) were given a single ketamine or midazolam infusion. 28 In the ketamine group, there were greater QIDS-SI and MADRS-SI improvements at 2 hours and 7 days post-infusion. Although there appeared to be significant carry-over effects between ketamine and midazolam, the greatest effect was observed at 7 days. After relapsing, participants received up to 6 open-label ketamine infusions (3 infusions/week over 2 weeks). MADRS-SI scores decreased throughout the first 3 infusions, and QIDS-SI significantly differed from baseline after all 6 infusions. After the repeated infusion course, 92% of participants had reduced SI, with 69% achieving full remission. 60% met antidepressant response criteria and received an additional 4 weekly infusions, which maintained the anti-suicidal response. Total MADRS change accounted for 40% of the variance in MADRS-SI change, and the peak in antidepressant and antisuicidal effects occurred at different times during the study. Additionally, the incremental improvement in SI after repeated infusions was not driven by antidepressant efficacy, as only 36% of antidepressant responders no longer had SI. Similarly, in a previously mentioned study by Pathak and colleagues, the time courses of SI and depression improvements after ketamine differed. 20
Continuing this trend of diverging antisuicidal versus antidepressant effects of ketamine, Zheng et al reported on patients with treatment-resistant unipolar or bipolar depression who received a six-infusion ketamine course.
29
They found significant improvement in SSI from baseline as early as 4 hours after the first infusion, which persisted throughout the treatment course. There were significant differences in SSI between responders and non-responders at days 5, 9, 10, and 11. Upon an analysis of only the unipolar participants, there were significant decreases in SSI in both responder and non-responder groups, with greater reduction occurring in the responder group at later time points.
30
The same group then assessed unipolar or bipolar participants with clinically significant SI who received 6 ketamine infusions.
31
57% had an anti-suicidal response (SSI-5 <2) at 24 hours after a single infusion, 65% after the sixth infusion, and 70.6% at the two-week naturalistic follow-up period. Time to SI relief in low (SSI <4) and high (SSI ≥4) ideation groups were around 3 and 6 days, respectively. After 4 hours, the low and high SI group response rate was 81.8% and 41.3%, respectively, and 24 hours after the last infusion response rates were 81.8% and 59.4%. A regression analysis revealed that response at the end of treatment was correlated with response at 4 hours after the first infusion. After controlling for baseline SI, HAMD change only explained 7.4% of the variance in ketamine’s antisuicidal efficacy, consistent with aforementioned studies. In Kang et al, treatment-refractory unipolar or bipolar depressed subjects, having failed ≥4 prior treatments, received 10 open-label ketamine infusions.
32
There was a significant improvement in suicidality in those with moderate baseline SI (MADRS-SI ≥2) one hour post-infusion, with no interaction with antidepressant response. Participants in both antidepressant responder and non-responder subgroups had significantly improved SI. Next, Can and colleagues tested variable dose (0.5-3.0 mg/kg) oral ketamine over 6 weeks in participants with chronic SI.
33
They found significant decreases in BSS after the treatment course with very large effect sizes, including continued improvement
Taken together, these studies support divergence in antisuicidal and antidepressant response, and a potential for somewhat distinct symptom etiologies. This suggests that the antisuicidal effects of ketamine may have trans-diagnostic utility, that is, not solely limited to treatment-resistant major depressive disorder or bipolar disorder.
Mediation
In contrast to the above section, some studies have observed enmeshment of SI and depression improvement, where reduction in suicidal thinking is mediated by overall depression improvement.
Ionescu and colleagues administered six ketamine infusions to subjects with TRD and chronic SI; the first three infusion doses were 0.5 mg/kg, and the last three doses were 0.75 mg/kg. 34 During the treatment, half of the participants had remitted SI Intensity [Columbia-Suicide Severity Rating Scale Intensity (C-SSRS)] and HDRS-SI scores (interestingly, HDRS-SI was positively correlated with dissociation). The increased dosage did not augment antisuicidal efficacy. At 3-month follow-up, 29% of acute phase responders maintained remission. By assessing C-SSRS Ideation, Intensity, HDRS-SI, and implicit association testing (IAT), Ionescu and colleagues found that SI improved throughout the treatment course, but these effects became non-significant after controlling for total HDRS score. Only 29% of those with remitted SI also had remission of depression symptoms, further complicating the relationship between SI and depression improvement. Next, in Price and colleagues, TRD participants were administered a single, open-label, 0.5 mg/kg ketamine infusion to study its effects on explicit (MADRS-10, BSS) and implicit (IAT) suicidal thinking. 35 In the portion of participants with clinically significant SI, 62% had MADRS-SI scores of 0–1, 23% had scores of 2–3, and 15% had scores ≥4 24 hours after the infusion, with moderate-to-very large effect sizes. The authors concluded that SI improvement was fully mediated by antidepressant response. “Escape = Me” IAT scores significantly correlated with BSS and trended with MADRS-SI but not total MADRS. Additionally, “Death = Me” IAT scores only had a trending relationship with BSS. Further analyses with a subset of participants who received subsequent dosing show maintenance of the antisuicidal effects through the 12-day treatment course. This study was replicated in 2014 by assessing implicit (IAT) and explicit (BSS, MADRS-SI, QIDS-SI) change after a single subanesthetic dose ketamine or midazolam infusion in TRD outpatients not at imminent risk for suicide. 36 At 24 hours post-infusion, explicit measures of SI were significantly decreased after ketamine compared to midazolam. At baseline, only 7% of participants were without SI; yet, at 24 hours post-treatment, 53% of ketamine participants scored zero on all three explicit measures, compared to 24% in the midazolam group. Price and colleagues found no difference in implicit SI between midazolam and ketamine, but ketamine improved “Escape = Me” from baseline whereas midazolam had no change, replicating findings from their earlier study. 35 They also found the greatest difference between ketamine and midazolam in participants with the highest SI. Baseline total MADRS scores did not moderate or predict subsequent SI improvement but change in MADRS at 4 and 24 hours fully mediated change in SI. As a final example of potential overlap in antisuicidal and antidepressant efficacies, in Grunebaum et al’s study discussed earlier, 17 improvement in the Profile of Mood States (POMS) mediated 33.6% of the improvement in SSI.
These studies challenge the independence of ketamine’s antisuicidal and antidepressant efficacy. However, it may be more complicated to fully parse out this relationship due to the strong collinearity between SI and depression. This emphasizes not only the need to continue using SI-specific questionnaires in clinical and research settings, but also to explore trans-diagnostic features of suicidal behavior, such as impulsivity, substance misuse, and non-lethal self-injury.
Negative Studies
While the preponderance of studies support ketamine’s anti-SI effects, an antisuicidal response has not been
Ionescu et al conducted a RCT in TRD medicated outpatients with chronic SI not at imminent risk of suicide who received 6 infusions of either ketamine or placebo.
37
The authors did not find a significant difference between ketamine and placebo in antisuicidal efficacy at 4 hours after each infusion or during the follow-up phase. The percentage of responders between the two groups was also non-significant. Ionescu and colleagues suggest that this result may be due to the greater treatment resistance and chronicity of their patients in comparison to other studies, and that higher doses of ketamine may be necessary to achieve antidepressant and/or antisuicidal response in this refractory population. Yet, there was a decrease in SI
While fewer in number than studies in support of ketamine’s antisuicidal efficacy, the negative trials indicate treatment response heterogeneity. To further understand this phenomenon, the field requires adequately powered biomarker-based studies to compare differences between groups with and without antisuicidal response to ketamine.
Meta-Analyses
Combining many of the above studies, meta-analyses have demonstrated that ketamine is effective at reducing SI in inpatient, outpatient, and emergency settings, 39 and in subjects with psychiatric disorders, 40 with varying effect sizes and heterogeneity (Table 6). Wilkinson and colleagues found that clinician-reported SI effects lasted up to a week compared to placebo, with varying results from self-reported measures. 41 They also reported that the early improvement in SI correlated with, but was not fully driven by, depression scores, accounting for 10–46% of the variance in SI change. Witt et al has also commented on the dearth of studies specifically reporting ketamine’s effect on suicidal behavior(s), rather than suicidal ideation alone. 40 Thus, inclusion of participants with suicidal behaviors may allow for a more direct comparison between the treatment types. Meta-analyses may be a well-powered avenue to understand subpopulations with varying levels of antisuicidal response as well as depression and SI entwinement.
Esketamine
The United States Food and Drug Administration (FDA) approval of intranasal esketamine as an adjunctive therapy for TRD in 2019 was shortly followed by a second indication for MDD with acute SI and behavior in 2020, a landmark first-in-class approval of a rapid-acting antisuicidal treatment for MDD.
Esketamine’s Antisuicidal Effects
One of the seminal studies leading to esketamine’s approval as an antisuicidal agent was conducted by Canuso and colleagues (Table 7). 42 This RCT investigated the efficacy of 4 weeks of twice-weekly intranasal esketamine (84 mg) versus saline placebo in MDD patients at high risk of suicide requiring inpatient hospitalization. They discovered significant improvements in MADRS-SI only 4 hours after the first dose; yet, other SI-related measures, eg, BSS and clinician global judgement of suicide risk, were not significantly different between groups. The percentage of participants in the esketamine versus placebo groups that had resolution of suicide risk (as defined by a clinician global judgement of suicide risk score of 0 or 1) was 21.1% and 9.7% four hours after the first dose and 40% and 6.5% the following day, respectively. To extend these initial observations, two identical phase 3 trials, ASPIRE I 43 and ASPIRE II, 44 were conducted with MDD participants with SI and intent requiring hospitalization. Though there were significant differences in depression scores between ketamine and placebo, there were no differences between groups on the various patient and clinician-rated modules of the Suicide Ideation and Behavior Assessment Tool (SIBAT). ASPIRE II also did not find significant between-group differences in suicidal thinking [Clinical Global Impression – Severity of Suicide – revised (CGI-SS-R)], with 86.3% of ketamine participants and 76.7% of placebo participants with a CGI-SS score of 0 or 1. In post-hoc analyses of data from the two ASPIRE studies, Canuso found that there was a greater MADRS benefit with esketamine at 24 hours after the first treatment in those with a suicide attempt in the prior month, as well as in those with MADRS scores above the median. 45 Like reports from the individual ASPIRE RCTs, there was no significant improvement in CGI-SS-R between treatment groups, though there were greater odds of improving MADRS-SI with esketamine compared to placebo plus standard of care. Finally, a recent case series demonstrated open-label antidepressant and antisuicidal efficacy of intranasal esketamine in adolescent TRD. 46 In this study, adolescents with TRD received a single administration of either subcutaneous or IV esketamine. At 24 hours post-administration, based on total MADRS and MADRS-SI, esketamine had large-to-very large antidepressant and antisuicidal efficacies.
Esketamine's antisuicidal effects and (es)ketamine meta-analyses.

CGI-S: Clinical Global Impressions-Suicide; IN: Intranasal; IV: Intravenous; MADRS-SI: Suicide item of the Montgomery-Asberg Depression Rating Scale; SC: Subcutaneous; SI: Suicidal Ideation.
In sum, esketamine appears to have pronounced acute antisuicidal effects, with lack of separation from placebo at later timepoints. Unlike many of the ketamine studies, which occurred in unmedicated patients, the Janssen-sponsored esketamine trials required newly initiated or optimized antidepressant medication, which may explain the significant antisuicidal response in the placebo groups. To date, there have been no published attempts to parse out whether the antisuicidal response of esketamine is mediated by overall depression improvement.
Comparative Efficacy
Few studies have discussed the comparative efficacy of racemic ketamine and esketamine. Vieira et al, conducted a RCT of IV racemic ketamine (0.5 mg/kg) and esketamine (0.25 mg/kg) in TRD patients with baseline suicidal ideation (MADRS-SI ≥1). 47 They found a significant reduction in MADRS-SI at 24 hours, lasting up to 7 days, but no significant between-group differences. Next, two meta-analyses reported that intranasal esketamine and IV ketamine are both effective at treating SI. Bahji et al reported increased same-day antisuicidal efficacy of ketamine and esketamine. 48 In critique of this approach, Xiong et al suggested that there are too few esketamine studies to compare the modalities directly. 49
Conclusion
In this review, we have presented mostly positive data on the antisuicidal effects of racemic ketamine and its FDA approved enantiomer esketamine. Several limitations exist when assessing (es)ketamine’s effect on suicidal ideation. Importantly, many of the studies reported in the present manuscript limit inclusion criteria to patients not at immediate risk of suicidal behavior, confining the conclusions that we can draw for more severely suicidal patients. This sample restriction may obscure some of the relationships between depression and suicidal ideation, and how (es)ketamine exerts its effects. Moreover, the field may benefit from close monitoring with standardized rating scales or specific SI-sensitive items to the rapid changes seen with (es)ketamine, such as the 5 item SSI, MADRS-SI, and BDI-SI9, 50, but these single Likert scale items do not provide the granularity needed to effectively assess the complexities of suicidal ideation. There also is no “gold standard” measure for assessing SI. To this point, Grunebaum and colleagues have suggested the use of computerized adaptive ratings due to their greater sensitivity and reduced variability compared to typically used clinical scales.
51
Aside from choosing the appropriate rating scales, an additional hurdle remains rapid
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) acknowledge funding from the National Institutes of Alcohol Abuse and Alcoholism
