Abstract
Objective:
Pregnant women are often concerned about the impact of medication use on their pregnancy, such as congenital abnormalities. This study examined the rate of adherence to and persistence with antidepressant medications during pregnancy based on the class of antidepressants prescribed.
Methods:
Women who gave birth between 2012 and 2015 in Alberta, Canada; had ≥1 diagnosis of depression within 1 year of preconception in outpatient physician claims, emergency department, or hospitalization administrative data; and were adherent (medication possession ratio ≥80%) to ≥2 consecutive antidepressant prescriptions during the preconception year (
Results:
During pregnancy, 834 (44.7%; 95% CI, 42.4% to 47.0%) women discontinued antidepressants. Among those continuing antidepressants, the overall rate of adherence was 62.6% (95% CI, 59.4% to 65.7%). The rate differed significantly by medication class (
Conclusions:
Adherence to and persistence with antidepressants is low during pregnancy and varies by medication class. Low adherence and persistence can interfere with a therapeutic effect of antidepressants, which may contribute to the worsening of depression symptoms.
Depression is one of the most common psychiatric disorders in childbearing women. 1 –3 Approximately, 1 in 5 women experience a depressive disorder during their lifetime, 2 –4 with 10% to 15% of women meeting the diagnostic criteria for a major depressive disorder at any point during pregnancy. 5 –7 Untreated depression during pregnancy is associated with the adverse health effects on mothers and infants, involving poor maternal self-care, fetal growth restriction, preterm birth, and postpartum depression. 6,8 –10 Given the high prevalence of depression amongst women of childbearing age and the high rate of unplanned pregnancies, 1,2,11 it is inevitable that a number of women will become pregnant while taking antidepressants or need to start taking antidepressants during pregnancy as nonpharmacological treatments (psychotherapy) are not always sufficiently effective. Adherence to prescribed antidepressants is important to achieve a therapeutic effect of the medications. However, there is evidence that pregnant women often do not adhere to their prescribed antidepressants, leading to a high rate of depression relapse (up to 68.0%) during pregnancy. 1,7,12,13
Failure to adhere to antidepressants may arise from a high level of anxiety amongst pregnant women about the safety of antidepressants for their baby. 14,15 More specifically, women are often concerned about the impact of medication use on their pregnancy, such as the perceived risk of congenital abnormalities 14,15 ; however, they do not necessarily consider the impact of untreated depression on their health or the health of their fetus. 10,14 Some women may tend to overestimate the teratogenic risks associated with prescribed medication use during pregnancy, reflecting a widespread belief that the harm of medication use during pregnancy always outweighs benefits. 13 Because of this misperception, many women face the difficult decision of whether they should continue taking their prescribed antidepressant throughout pregnancy, which may lead them to permanently or temporarily discontinue their prescribed medications during pregnancy. 15,16 Other factors that may affect medication intake practices include perceived or real side effects of medication, perceived severity of disease, perceived intensity of medication, and information/advice obtained from friends, relatives, and health care providers. 1,15
A recent study conducted in the general population shows that people with a depressive disorder have differential medication intake practices, including adherence (the extent to which a patient acts in accordance with the prescribed interval and the dose of a dosing regimen) and persistence (the duration of time from initiation to discontinuation of therapy) depending on the therapeutic class of antidepressant prescribed. 17,18 However, how adherence to and persistence with antidepressants are influenced by medication class have not been evaluated in the context of pregnancy. The assessment of adherence and persistence patterns across antidepressant classes during pregnancy may provide insight into the antidepressant intake behaviour in pregnant women. Thus, this study examined the rate of adherence to and persistence with antidepressant medications overall, stratified based on their pharmacological class, during pregnancy.
Methods
This retrospective cohort study used deidentified, population-based administrative data holdings maintained by Alberta Health and Alberta Health Services. These data holdings include the delivery record from the Alberta Perinatal Health Program (APHP), hospitalization data from the Discharge Abstract Database (DAD), outpatient physician claims data (PC), emergency department data from the National Ambulatory Care Reporting System (NACRS), and prescription medication data from the Pharmaceutical Information Network (PIN). These databases were originally created for health care management and monitoring functions (e.g., insurance claim and remunerating physicians) under the universal health care system in Canada, and they are suitable for research purposes. 19 Deterministic linkage using the personal health number was used to link records from these multiple data sets. Ethics approval was obtained from the Conjoint Health Research Ethics Board at the University of Calgary.
Figure 1 outlines how the cohort was identified. This study included data ranging from the 1 year prior to conception to 1 year postpartum and included the duration of pregnancy (for all deliveries that occurred between 2012 and 2015). Women who delivered a live or stillborn infant >20 weeks of gestation between 2012 and 2015 were identified in APHP data. Then, amongst this cohort, women with depression were identified from DAD, NACRS, and physician claims data using the validated case definitions proposed by Fiest et al.
20
This included ≥1 diagnosis of depression using the

Flowchart of cohort identification and selection.
Using the information on antidepressant prescriptions retrieved from PIN, antidepressants were classified into 4 classes using the Anatomical Therapeutic Chemical (ATC) Classification System. 21 The antidepressant classes examined included nonselective monoamine reuptake inhibitors (including tricyclic and tetracyclic compounds), selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine inhibitors (SNRIs), and other (atypical) antidepressants (see online supplemental material). No monoamine oxidase inhibitors were prescribed in this cohort.
Adherence to antidepressants was measured by the medication possession ratio (MPR), which assessed the proportion of a given time period where medication supply was available. 22 The MPR was calculated by summing the total supply of medication dispensed between the first refill date to the second last refill date divided by the difference between the last and first dispensed date. 22 The MPR method was chosen because of its stronger association with negative health outcomes than other alternative measures of adherence. 22 Women were classified as adherent if the MPR was ≥80%—a cutoff that has a good predictive ability for hospitalization in the general population. 23 For women on polytherapy, an average MPR estimate was calculated and assigned to each medication class. 22
Persistence with antidepressants, which corresponds to the continuity of prescribed treatment over several prescriptions, 18 was calculated at each month of pregnancy, starting from the second month to the ninth month. Persistence was calculated by subtracting the duration of pregnancy period from the sum of the total supply of medication dispensed between the first refill date to the second last refill date. Women were classified as persistent if the difference was ≤30 days (i.e., prescription refill gap). 24,25
Women who had at least 2 consecutive prescriptions for the same class of antidepressant and who were adherent (MPR ≥0.8) to antidepressants at any time during the 1 year preceding conception were eligible for this study (
Eligible women were categorized as being on monotherapy or polytherapy. Due to the small sample size of women receiving polytherapy in our data set, adherence and persistence across specific antidepressant classes were assessed only for those women who were on monotherapy. Adherence and persistence during pregnancy were calculated among those women who were adherent during 1 year preconception and had at least 1 prescription dispensed during pregnancy. The women who were adherent during preconception but did not fill a prescription from the date of conception (i.e., no single prescription dispensed during pregnancy) were classified as discontinuing medication from the beginning of pregnancy. This group of women was analysed separately to assess whether the discontinuation of medication differed by antidepressant class.
The adherence rate overall and for specific classes of antidepressants during pregnancy was calculated, and chi-square tests were used to assess whether the rates of adherence to antidepressant differed by the medication class. Adherence rates, overall and for specific antidepressant classes, were further calculated for each trimester (gestational age, <12 weeks, 13-24 weeks, ≥25 weeks) and were compared using chi-square tests for trend. Similarly, the rate of persistence overall and to specific antidepressant classes was calculated at each month of pregnancy, starting from the second month of pregnancy to the ninth month. Chi-square tests were used to assess whether the rates of persistence to antidepressants differed by the antidepressant class in each month of pregnancy. The trend of persistence rates between the second and ninth months of pregnancy was assessed using chi-square tests for trend. The proportion of women who resumed their medication during the first year postpartum was also calculated. The adherence rate, overall and stratified by antidepressant class, was also calculated for the first year postpartum and for different time frames within the first year postpartum (1-3, 3-6, 6-9, and 9-12 months). All analyses were performed using STATA/IC 14.1. An alpha level of 5% was used to assess statistical significance.
Results
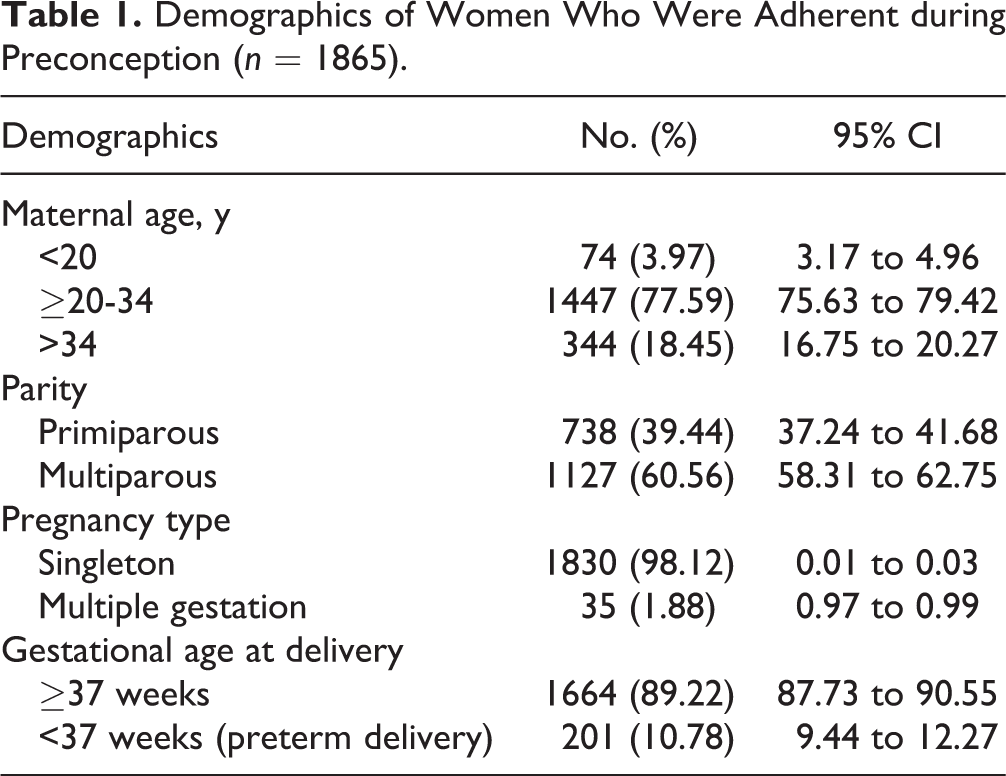
Of the 159,059 deliveries occurring in Alberta between 2012 and 2015, 11,220 women (7.0%) had at least 1 health care contact for depression in the year prior to conception. In total, 2703 women (24.1% of depressed women and 1.7% of all pregnant women) had at least 2 consecutive prescriptions for the treatment of depression in the year prior to pregnancy: SSRIs were the most prescribed (72.5%) and nonselective monoamine reuptake inhibitors were the least prescribed monotherapy antidepressants (3.3%). Among women with at least 2 consecutive prescriptions, 1865 (68.9%) women were adherent to their medication. Of these, 1600 were on monotherapy and 265 were on polytherapy (Figure 1). Among adherent women, over three-fourths of women were between the ages of 20 and 34 years and approximately two-fifths of women were primiparous (Table 1).
Demographics of Women Who Were Adherent during Preconception (
As shown in Table 2, amongst previously adherent women receiving monotherapy, 51.1% (95% CI, 48.6% to 53.6%) discontinued (i.e., no single prescription dispensed) their antidepressants during pregnancy. The rate of discontinuation differed by antidepressant class (
Adherence to Antidepressant Medication during Pregnancy and 1 Year Postpartum (
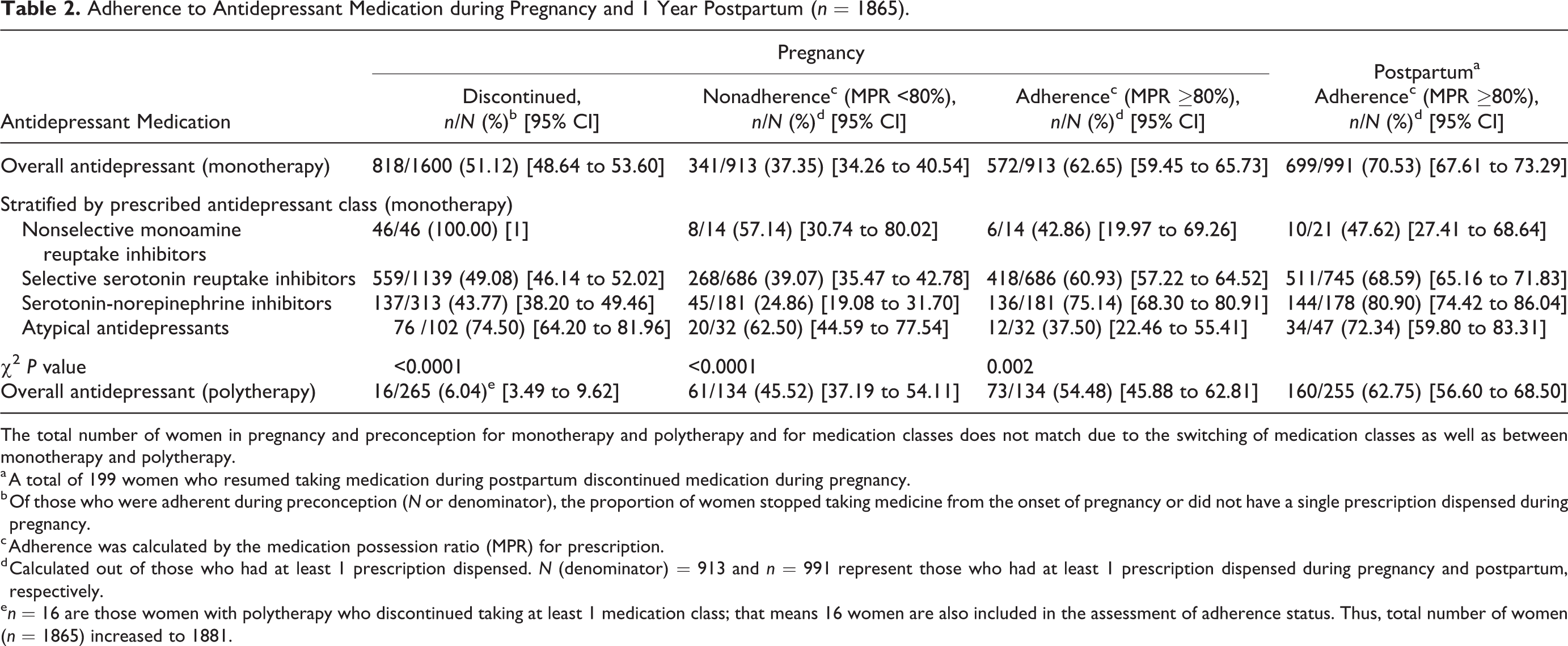
The total number of women in pregnancy and preconception for monotherapy and polytherapy and for medication classes does not match due to the switching of medication classes as well as between monotherapy and polytherapy.
a A total of 199 women who resumed taking medication during postpartum discontinued medication during pregnancy.
b Of those who were adherent during preconception (
c Adherence was calculated by the medication possession ratio (MPR) for prescription.
d Calculated out of those who had at least 1 prescription dispensed.
e
The overall rate of adherence during pregnancy among those who were adherent during preconception was 62.6% (95% CI, 59.4% to 65.7%) for women taking monotherapy antidepressants. The adherence rate was significantly different across antidepressant classes (
Adherence to Antidepressant Medication across First, Second, and Third Trimesters of Pregnancy.

a Adherence was calculated by the medication possession ratio (MPR) for prescription.
b At least 1 prescription dispensed for antidepressant medication.
As seen in Figure 2, persistence to antidepressants substantially decreased for each medication class as the duration of pregnancy progressed. The overall persistence rate was 84.3% (95% CI, 81.7% to 86.6%) at the third month of pregnancy, and it was less than 50.0% at the eighth or ninth month of pregnancy (48.5%; 95% CI, 45.2% to 51.8% or 40.8%; 95% CI, 37.5% to 44.1%, respectively). The trend of persistence rates between the second to ninth month of pregnancy was significantly different (
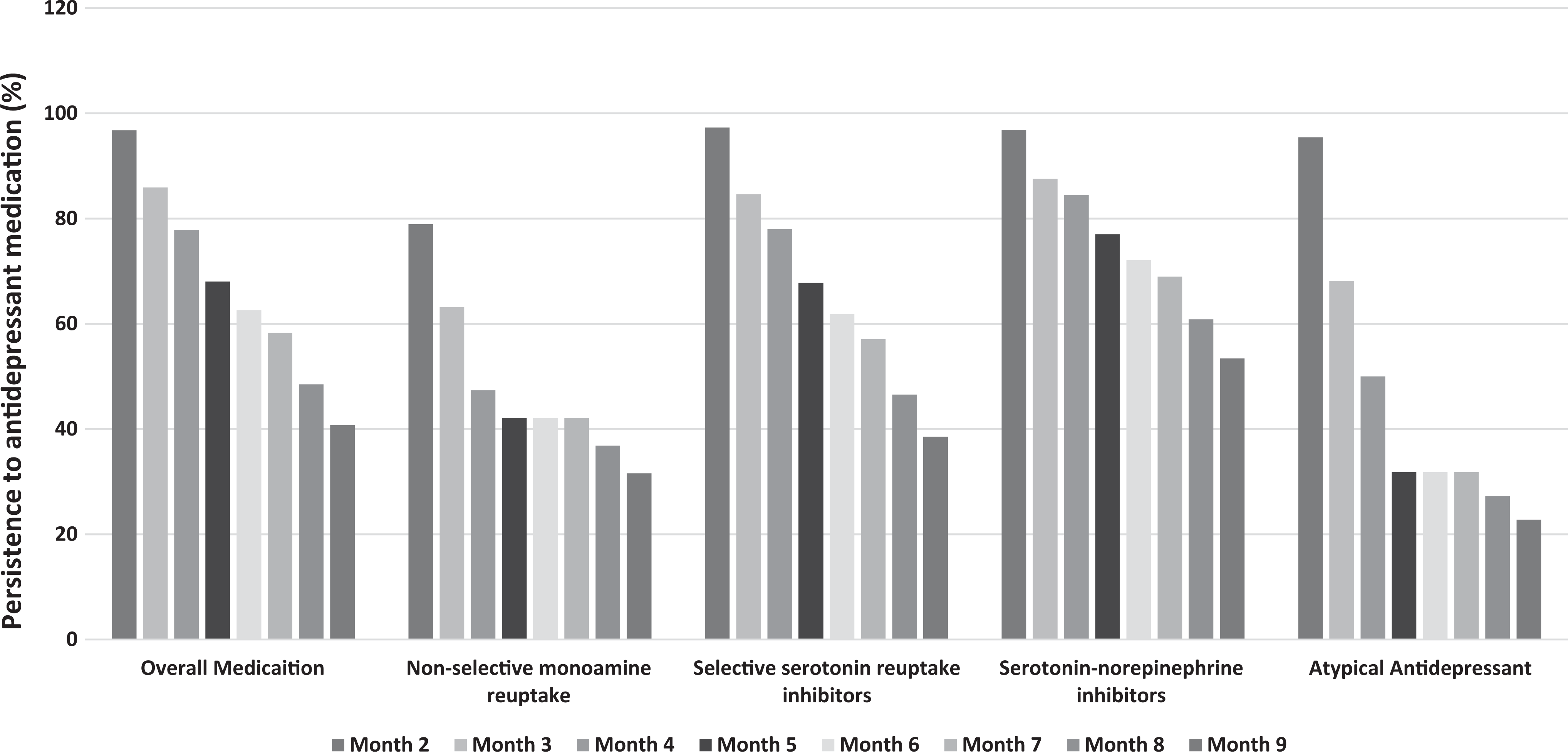
Persistence to antidepressant medication during pregnancy by antidepressant class (monotherapy,
Almost one-quarter of women who discontinued their antidepressants during pregnancy resumed taking their medications during the first year postpartum (24.33%; 95% CI, 21.42% to 27.41%). The adherence rate to monotherapy antidepressants was 70.5% (95% CI, 67.6% to 73.3%) during the first year postpartum. Similar to the adherence pattern observed during pregnancy, the rate was highest for SNRIs (80.9%; 95% CI, 74.4% to 86.0%) and lowest for nonselective monoamine reuptake inhibitors (47.6%; 95% CI, 27.4% to 68.6%) (Table 2). Adherence rates across different time frames during the first year postpartum (1-3, 3-6, 6-9, and 9-12 months) were similar.
Discussion
This study examined the rate of adherence to and persistence with using monotherapy antidepressants based on the class of antidepressants prescribed during pregnancy, using population-based administrative databases in Alberta, Canada. Nearly half of women who were previously adherent during the year prior to pregnancy discontinued using antidepressants during pregnancy (i.e., no single prescription was dispensed). Among those who continued using antidepressants during pregnancy, adherence to and persistence with using antidepressants was low and differed by the class of antidepressants prescribed. Overall, over one-third of women did not adhere to antidepressants, and persistence in using antidepressants during pregnancy remained below 50%—both adherence and persistence were highest for women taking SNRIs and lowest for women taking nonselective monoamine reuptake inhibitors or atypical antidepressants.
The observed low adherence and persistence to antidepressants is comparable to existing literature in perinatal populations. 1,12,26 The rates of nonadherence that range from 33% to 56% and the nonpersistence rate of 44% at 6 months in general populations, including nonpregnant women, are also comparable to our rates. 27 –29 However, unlike other studies, our adherence and persistence calculation was restricted to those women who were adherent in the year prior to pregnancy; thus, lower nonadherence and nonpersistence rates compared to those seen in previous studies were expected. The restriction of the analysis among those women who were adherent prior to pregnancy also supports the interpretation that nonadherence and nonpersistence can at least partially be attributed to the perceived risks of medications during pregnancy rather than other causes of nonadherence such as adverse effects, perceived lack of efficacy, stigmatization, and so on. While the reasons for medication nonadherence and nonpersistence cannot be assessed in studies that rely exclusively on the use of administrative data, our study adds to the literature that adherence and persistence vary by the medication class. However, SNRIs, the second line of treatment for severe major depression, 30 are often prescribed when the other classes of antidepressants including, SSRIs, are inadequately effective. Health care providers may have communicated this information to the women taking SNRIs; thus, they may anticipate greater disadvantages of discontinuing these medications, potentially explaining the relatively higher rate of adherence to and persistence with using SNRIs compared to other classes of antidepressants observed in this study. It is difficult to interpret our findings regarding women with complete discontinuation of antidepressants during pregnancy as this likely comprises a heterogeneous group of women—those who followed the advice of their physician to discontinue their antidepressants and maintain close follow-up or observation; those who made an informed decision to switch to psychotherapy, a first choice of treatment for mild to moderate major depression during pregnancy, as recommended by the Canadian Network for Mood and Anxiety Treatment (CANMAT) 30 ; and those who discontinued their medication upon pregnancy recognition with or without consultation with their care provider. As study databases only contain information on filled prescriptions and not all written but unfilled prescriptions, we have no mechanism to identify these cases.
The overall lower adherence to antidepressants during pregnancy compared to the preconception and postpartum periods may be at least partially due to the extra social support and urgency to offer psychotherapy that may accompany pregnancy and make discontinuation of antidepressants possible. Furthermore, the lower adherence to antidepressants in the first trimester is likely related to women’s overall perception that medication use during organogenesis negatively affects fetal development. However, as half of pregnancies are unplanned, it is also possible that some women who were adherent to medications in the first trimester were taking medication prior to pregnancy recognition.
CANMAT recommends that the prescribed antidepressants should continue at least 6 to 12 months after remission, including during the perinatal period. 30 However, continuing antidepressants during pregnancy remains a challenge as the risks associated with taking antidepressants during pregnancy are not completely understood. 1,30 –32 Nevertheless, if the continued use of antidepressants is recommended (based on a clinical judgement that possible risks associated with not taking the antidepressants outweigh the risks associated with taking them), adherence to and persistence with antidepressants are important to allow them to be effective. For this reason, strategies that potentially improve adherence to and persistence with prescribed antidepressants should be used from the time they are originally prescribed, taking into consideration our findings that adherence to and persistence with medication differ by the classes of antidepressant prescribed. Useful strategies may involve evidence-based teratogenic risk counselling of prescribed medications, detailed education focusing on the importance of good adherence for the health of both the mother and fetus, informed decision making on choosing the appropriate medication, and continuous monitoring of medication intake practices with extra support for women on certain antidepressants. 15,33 –35
This study used population-based data in the province of Alberta, Canada; thus, the observed findings are more likely to be generalizable to other provinces in Canada and other high-income countries with similar health care systems. This study provides more conservative estimates of adherence and persistence as estimates were calculated considering the second last date of prescription refilled as a last day of therapy. 22,36 However, assessing adherence and persistence through the analysis of prescription refill data may not reflect true adherence and persistence as our adherence and persistence calculations assumed patients take the prescriptions they refill. Furthermore, individual circumstances (e.g., severity of depression symptoms, the response of medication in helping manage depression, women’s participation in treatment decisions, and care providers’ confidence, encouragement, and information they provided to women) may partly play a role in adherence and persistence patterns observed across the classes of antidepressants. 1,13,15,34 However, due to the unavailability of data on these circumstances, our study was unable to explore this. Furthermore, assessment of hospitalization data, including pregnancy outcomes and health resource use, is needed to understand the risk associated with the discontinuation of or nonadherence to antidepressants. If women who switched medication classes or therapy types (monotherapy and polytherapy), when they became pregnant, had different adherence and persistent patterns, our finding may not be representative to them.
In conclusion, amongst previously adherent women, adherence to and persistence with using antidepressants during pregnancy are low, and both adherence and persistence vary by medication classes. Adherence to and persistence with prescribed antidepressants are important to achieve a therapeutic benefit of antidepressants. Low adherence and persistence to prescribed antidepressants during pregnancy (in the absence of other alternative management strategies such as psychotherapy and close follow-up) may put women at increased risk for developing the severe symptoms of depression, consequently exposing fetuses and newborns to the negative impacts of perinatal depression. It may also place some women at risk of adverse events related to antidepressant treatment while diminishing benefits they may receive from such treatment. The low adherence to and persistence with using antidepressants could be improved by conducting more research on drug safety during pregnancy and translating research evidence into treatment decisions and patient education. Differences in adherence and persistence by antidepressants may suggest the need of extra support for women on certain antidepressants.
Supplemental Material
Supplemental Material, 2018-04-30_-_Supplementry_material - Adherence to and Persistence with Antidepressant Medication during Pregnancy: Does It Differ by the Class of Antidepressant Medication Prescribed?
Supplemental Material, 2018-04-30_-_Supplementry_material for Adherence to and Persistence with Antidepressant Medication during Pregnancy: Does It Differ by the Class of Antidepressant Medication Prescribed? by Kamala Adhikari, Scott B. Patten, Sangmin Lee, and Amy Metcalfe in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
Kamala Adhikari is supported by the Vanier Canada Graduate Scholarship and the Alberta Innovates Studentship Award. Amy Metcalfe is supported by a Canadian Institutes of Health Research New Investigator Award. These awards/scholarships were not for this project; due to this, we report as no financial support for this research. We would, however, like to acknowledge these awards as it is a requirement of receiving them.
Data Access
Supplementary file has been submitted to provide additional information about this research. Data types such as statistical data file, statistical codes, text files, tables, additional charts, and graphs necessary to understand the research will be provided upon the request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
