Abstract
In total, 20 fungal spore types were identified which are commonly encountered in the studied surface soil samples. The presence and abundance of the recovered fungal spore were observed in relation to the different vegetation types namely Forestland, Grassland and Swamp in the Pobitora Wildlife Sanctuary (PWS). The studies reveal that both coprophilous and non-coprophilous fungal spores, such as Sporormiella, Ascodesmis, Saccobolus, Glomus, Nigrospora and Tetraploa, are regularly encountered in the studied samples and significant which could be a strong proxy and complementary to pollen for the palaeoherbivory and palaeoecology analysis in the PWS and to correlate the other parts of the region.
Introduction
The Quaternary period witnessed many global climatic changes that occurred in the Holocene epoch, such as the Holocene Climate Optimum (HCO), which occurred at about 9000 to 5000 years BP, the Medieval Warm Period (MWP), which occurred around 950 to 1250 CE, and Little Ice Age (Alley et al., 1997; Mayewski et al., 2004; Tripathi et al., 2021). It is well established that pollen is a reliable proxy to reconstruct the past vegetation and climate at the regional and global levels. However, among non-pollen palynomorphs, especially the fungal spore is also identified and characterised as a strong proxy for the palaeoenvironmental in relation to the palaeoherbivory analysis. In the last few years, the fungal spore encountered in the same palynological slide as identified and utilised to interpret the palaeoecology and palaeoherbivory analysis palynological slide is identified and characterised as a powerful and complimentary proxy of pollen. For the palaeomycological analysis, the study on the fungal spore preservation of modern peat, soil and lake deposits is very essential and useful (Gill et al., 2009; Kramer et al., 2010; Montoya et al., 2010; Mudie et al., 2010; van Geel, 2001; van Geel et al., 1989). In the last three decades, work was done on Quaternary sediment on the fossil fungal remains from different parts of Europe and Siberia (Aptroot & van Geel, 2006; Buurman et al., 1995; Kuhry, 1997; Pals et al., 1980; van Geel, 2001; van Geel & Aptroot, 2006; van Geel et al., 1989, 2003), Kenya (van Geel et al., 2011), Zambia (Jarzen & Elsik, 1986; Wolf, 1966, 1967), Uganda (Lejju et al., 2006) and Netherlands (van Geel et al., 2003). In India, fungal spores recovered in Madhya Pradesh (Quamar, 2015), Gujarat (Sharma, 1974), Mizoram (Mandaokar et al., 2008), Tripura (Prasad & Ramesh, 1983) and Assam (Basumatary et al., 2020, 2024; Basumatary & McDonald, 2017; Pandey et al., 2023). This study will be helpful for the accurate identification of the fungal spore recovered from the sediment, which could be precisely utilised for the interpretation of the palaeoherbivory and palaeoecology analysis in PWS and other regions of the continents.
Study Area, Vegetation and Wildlife
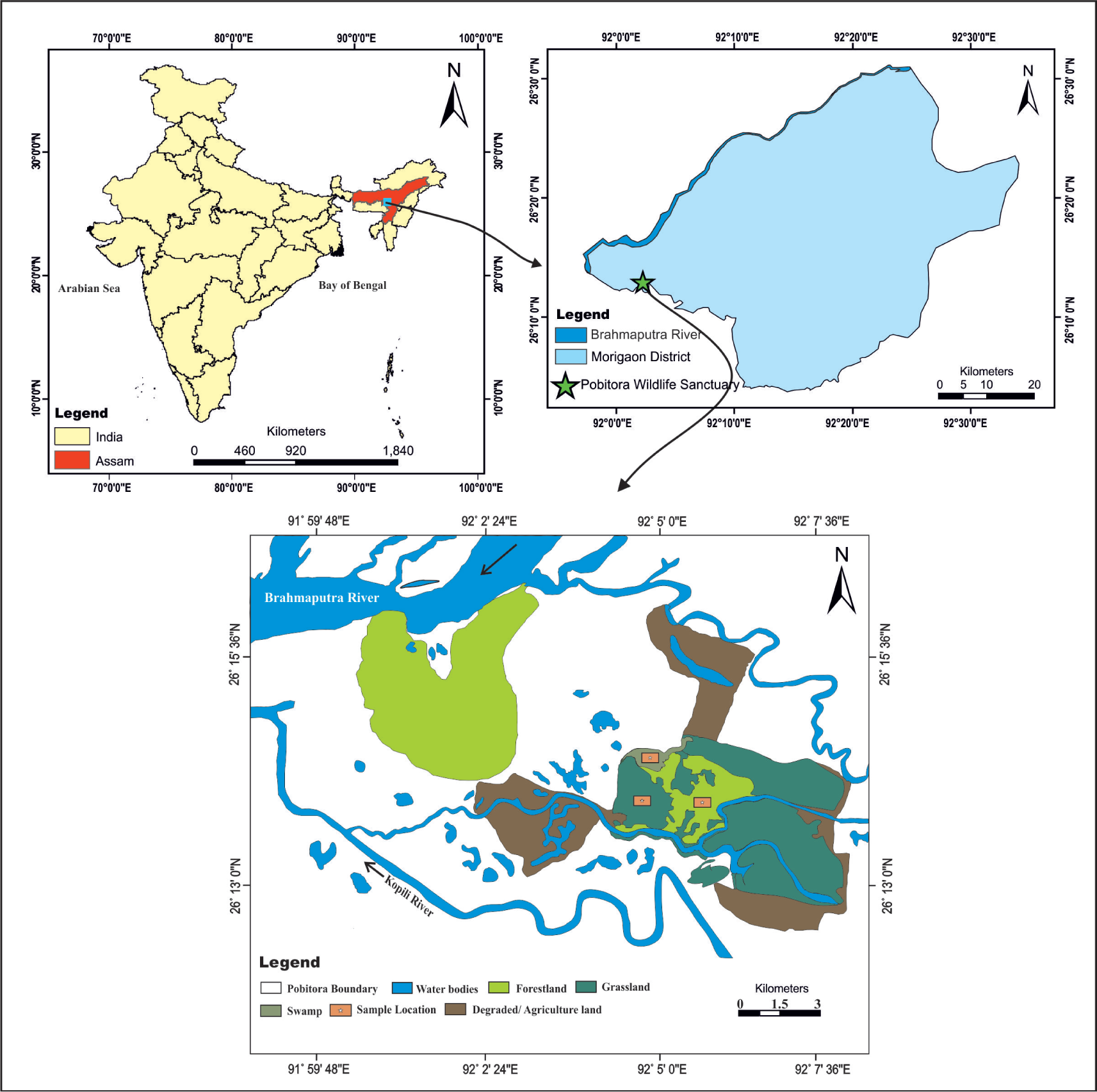
The Pobitora Wildlife Sanctuary (PWS; 26˚ 12’N to 26˚ 15’ N and 92˚ 2’ to 92˚ 5’E) is located on the flood plains of Mighty Brahmaputra River in the Morigaon district of Assam and the river Kopili is also flowing close to the wildlife sanctuary (Figure 1). It covers about 38.85 km2 and is situated 40 km from Guwahati city. It was declared as a reserve forest in 1971 and a wildlife sanctuary on 16 July 1987 (Areendran et al., 2021). The sanctuary is rich in biodiversity and comes under one of the biodiversity hotspot regions of the world known as Indo–Burma hotspot region (Basumatary et al., 2014). The vegetation of the area is tropical mixed deciduous forest, swamp and grassland. Major area of this sanctuary is covered with forestland, grassland followed by swamps. The major tree elements in this sanctuary are Lagerstroemia parviflora, Syzygium cumini, Dillenia indica, Bombax ceiba, Terminalia belliricum and Semecarpus anacardium (Figure 2a). The floor of the forest is covered by different species of Poaceae, Asteraceae, Convolvulaceae and Euphorbiaceae. The ferns Gleichenia dichotoma, Adiantum caudatum and Polypodium vulgare are the most common members of the forest assemblage (Champion & Seth, 1968). The grassland areas within the wildlife sanctuary are scattered and are composed of both tall and short grasses, chiefly Phragmites karka, Arundo donax, Cenchrus ciliaris and Microstegium ciliatum (Figure 2b). The vegetation of the swamp is dominated by marshy and aquatic taxa such as Cyperus rotundus, Potamogeton pectinatus, Nymphaea alba, Ludwigia sedioides and Nymphoides indica (Figure 2c). Some trees and shrubs are present, such as B. ceiba, Careya arborea, D. indica and Albizia lebbeck, which are scattered within the grassland. The sanctuary is a major attraction and habitat of Indian one-horned rhinoceros (Rhinoceros unicornis) (Figure 2d) besides, rhino other animals, like Asiatic buffalo (Bubalus arnee) and wild boar (Sus scrofa), are also living in this sanctuary. It is also a home for more than 2000 migratory birds, and various reptiles and some big lizards are also living in this grassland (Figure 2e), other large and medium-sized mammalian herbivores include monkey (Rhesus macaques).
Location Map showing the study site (Modified after Bhatta & Saikia, 2011).
Field photographs from Pobitora Wildlife Sanctuary; a. Tropical Forest inside Pobitora Wildlife Sanctuary; b. A view of Grassland in Pobitora Wildlife Sanctuary; c. View of Swamp with Asiatic Buffalo (Bubalus arnee) within Pobitora Wildlife Sanctuary; d. A view of Rhino (Rhinoceros unicornis) during feeding; e. A view of lizard within the grassland; f. Collection of soil samples inside Pobitora Wildlife Sanctuary.

Climate and Soil
The climate of this region is governed by the Indian monsoon (Northeast and Southwest Monsoon). The maximum temperature ranges between 33–28˚C in summer and the minimum temperature is 3–4˚C. This region is sensitive to the Indian monsoon due to which rainfall occurs throughout the year, within the range of 2000–3000 mm. The relative humidity is very high and ranges between 76% and 95% (Konwar et al., 2009). The soil varies from site to site. In forest areas, the soil is mainly sandy loam and rich in humus, sandy soil is present in the grassland habitat but is low in organic matter and clayey soil predominates in the swamp area.
Material and Method
Fieldwork
During the month of December 2022, the first (SV) and second (SKB) authors visited the site and a total of 20 surface soil samples 6 from forestland, 6 from grassland and 8 from swamp areas were collected randomly from the PWS for palynological analysis (Figure 2f).
Laboratory Work
For the fungal spore analysis from the surface soil samples, we use the standard acetolysis method (Erdtman, 1953). To create a composite pie diagram of fungal spores, a total of 170–220 fungal spores were counted per slide from palynoassemblage and divided into coprophilous and non-coprophilous fungal spores. Identification and characterisation of fungal spores were performed from published literature (Basumatary et al., 2017, 2024; Basumatary & McDonald, 2017; van Geel et al., 2003, 2011). Microscopic observation and photo-documentation of fungal spores were made using an Olympus BX-61 microscope with a DP-25 digital camera under 40X magnification (Plate I).
Result
In total, 20 fungal spore types were identified and characterised in the surface soil samples of PWS. The high diversity and different characteristics of fungal spores have been observed in the palynological slide, which could be a strong proxy and complimentary to the pollen proxy for the interpretation of palaeoecology and palaeoherbivory. The description of fungal spores is based on morphological features, such as shape and size (Gelorini et al., 2011; Kuhry, 1985; van Geel, 2001; van Geel et al., 2011).
Forestland
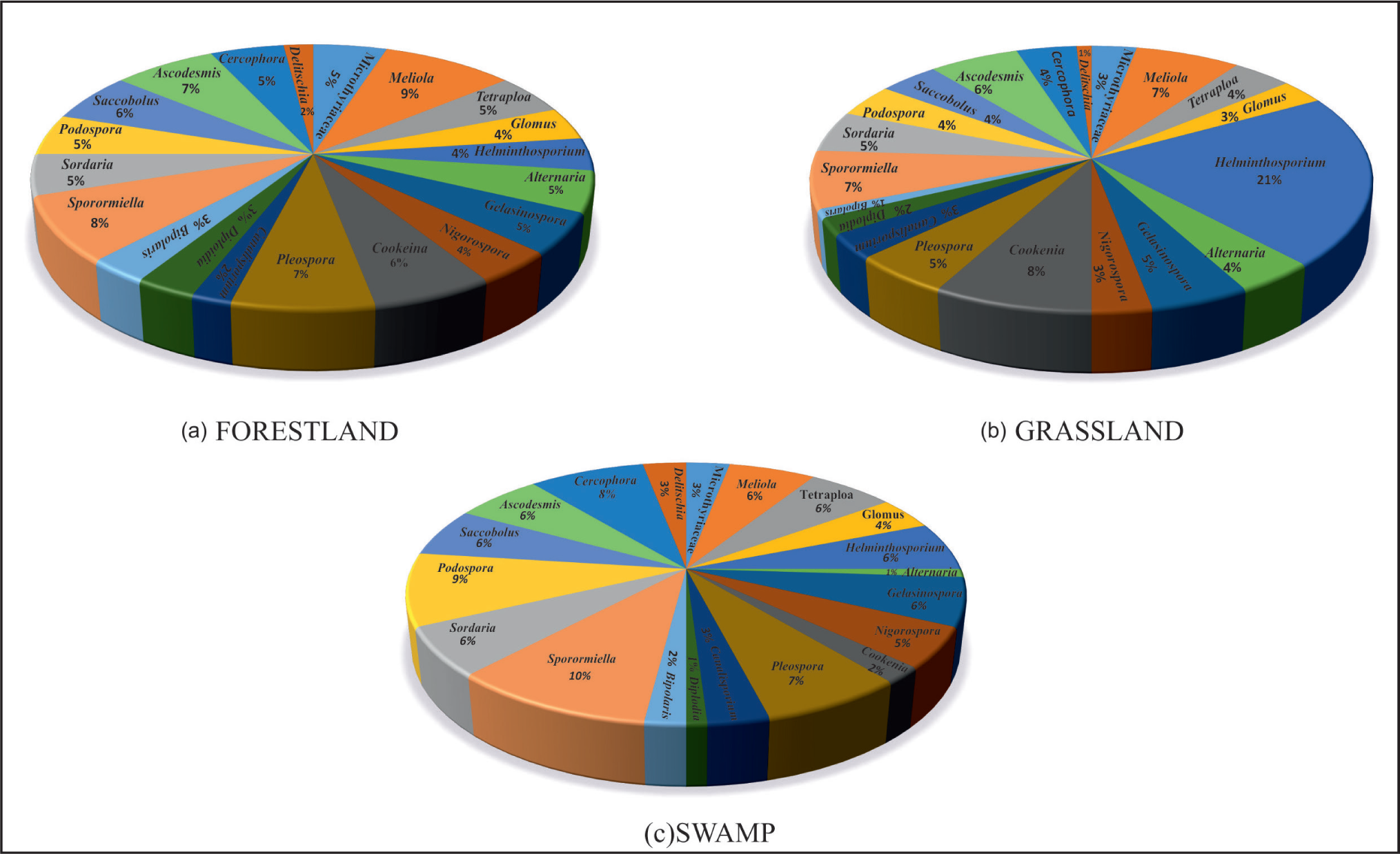
Non-coprophilous fungal spores are dominant at an average value of 62% over coprophilous fungal spores, which are at an average value of 38%. Among non-coprophilous fungal spores, Meliola is recorded at an average value of 9% followed by Pleospora (7%) and Cookeina (6%). Similarly, among coprophilous fungal spores, Sporormiella is recorded at an average value of 8% followed by Ascodesmis (7%) and Saccobolus (6%) (Figure 3a).
Composite Pie diagram of fungal spore in relation to different vegetation types of Pobitora Wildlife Sanctuary; a. Distribution of fungal spore in Forestland; b. Distribution of fungal spore in Grassland; c. Distribution of fungal spore in Swamp.
Grassland
Non-coprophilous fungal spores are dominant at a value of 69% over coprophilous fungal spores at 31%. Among non-coprophilous fungal spores, Helminthosporium recorded a value of 21% followed by Cookeina (8%) and Meliola (7%). Among coprophilous fungal spores, the Sporormiella exhibited a value of 7% followed by Ascodesmis (6%) and Delitschia (1%) (Figure 3b).
Swamp
Non-coprophilous fungal spores (52%) showed predominance over coprophilous fungal spores (48%). Pleospora is the non-coprophilous fungal spore with the highest recorded average value of 7% followed by Tetraploa (6%) and Glomus (4%). Among coprophilous fungal spores, Sporormiella is recorded at an average value of 10% followed by Podospora (9%) and Cercophora (8%) (Figure 3c).
Description and Illustration of Fungal Spore
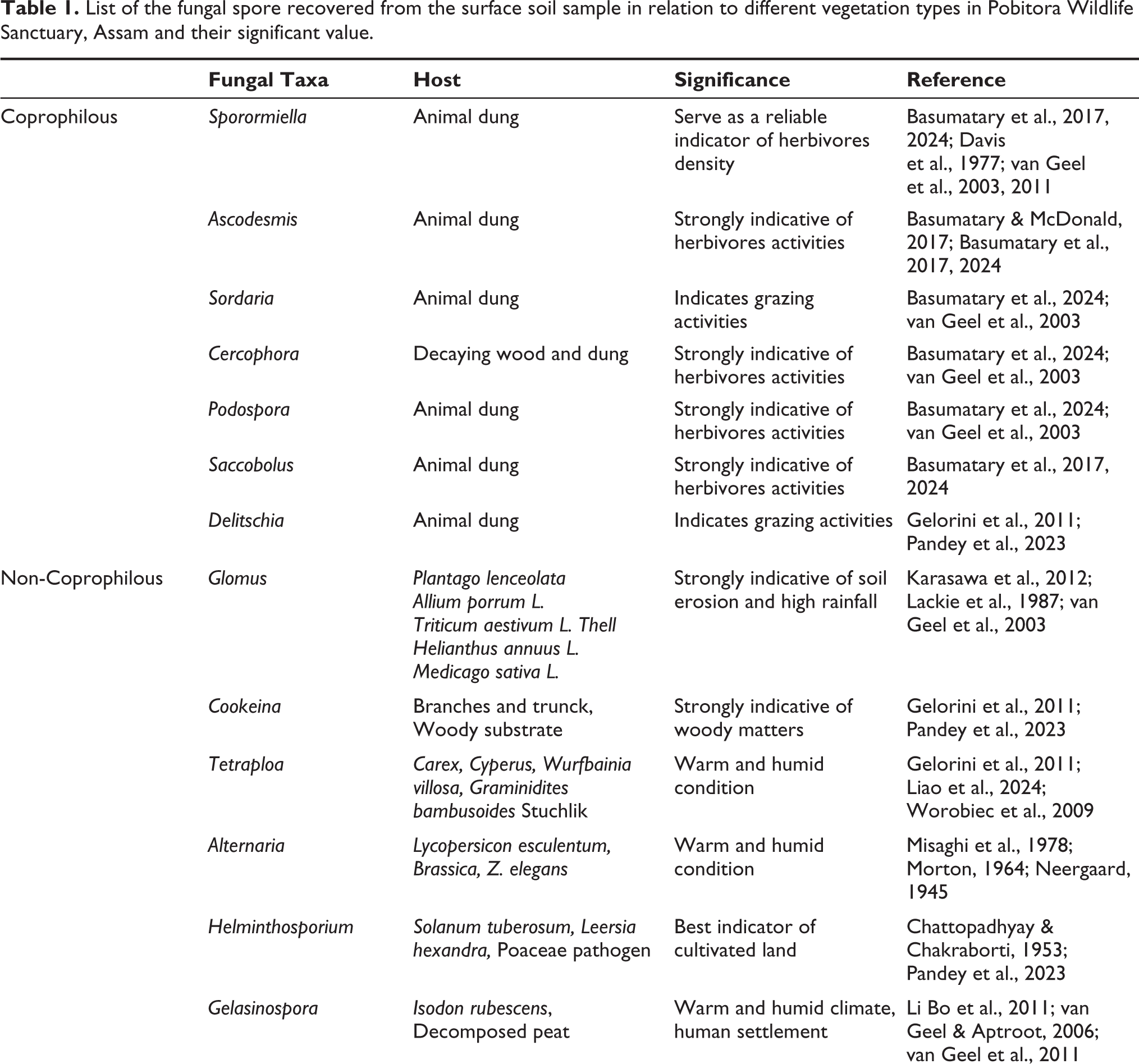
Ascospores globose to subglobose, walled thick, brown to dark brown, 35–38 µm diam. The presence of Ascodesmis on dung strongly indicates herbivores activity (Gelorini et al., 2011; van Geel et al., 2003).
1. Ascodesmis; 2. Sordaria; 3. Sporormiella; 4. Podospora; 5. Cercophora; 6. Saccobolus; 7. Cookeina; 8. Tetraploa; 9. Glomus; 10. Gelasinospora; 11. Meliola; 12. Delitschia; 13. Alternaria; 14. Helminthosporium; 15. Pleospora; 16. Bipolaris; 17. Canalisporium; 18. Microthyriaceae; 19. Nigrospora; 20. Diploidia.

Ascospores ellipsoidal, light to dark brown, 32–36 × 16–20 µm2 and smooth thick-walled, one pore at the apical end and other smaller pores on the basal end. Sordaria is a type of coprophilous fungal spore, which occurs on dung substrate and in soil (Bell, 1983; Hanlin, 1990).
Ascospores subglobose, cylindrical, dark brown, multiseptate, 17–19 µm diam. Sporormiella is a strong indicator of herbivores activity (Perrotti & van Asperen, 2019; van Geel et al., 2011).
Ascospores ellipsoidal with blunt conical shape, light to dark brown, single-celled, 36–40 × 14–18 µm2 in size with one pore at the apical end and the basal end. Podospora typically grows on domestic and wild animals’ dung and is a promising indicator of herbivores (van Geel et al., 2003).
Ascospores tapered at the apical end and terminated at the basal side, 31–40 × 12–18 µm2 in size and having one pore at the apical end of 0.8 µm diam. Cercophora fungal spore grows on herbaceous stems, leaves and decomposing wood. Cercophora are good indicator of the presence of herbivores (Basumatary & McDonald, 2017; van Geel et al., 2003).
Ascospores ellipsoidal or ovoidal broadly fusiform, hyaline thick-walled, smooth, brown, 20–25 × 11–15 µm2 diam. Saccobolus occurs on both vegetable material and herbivore animal’s dung (Basumatary et al., 2024; Doveri, 2014).
Ascospores dark brown, ellipsoid to fusiform, with two equal and symmetrical cells 25–30 × 5–15 μm2 and slightly thick-walled. Cookeina is mostly found in tropical and subtropical regions. Cookeina is frequently seen on fallen branches, trunks (Bera et al., 2008; Gelorini et al., 2011).
Conidia ellipsoid to rectangular, light to dark brown, 4–5 columns with 5 cells to each column, 40–45 × 25–30 µm2 diam, highly thick-walled. Tetraploa is distributed in tropics and subtropics. It is mostly found on leaf and stems of host plants (Ellis, 1971; Gelorini et al., 2011).
Chlamydospores globose to subglobose, smooth thick-walled, in most cases, hyphae are attached but they are also observed in the clusters. The spores of Glomus have a highly variable size of 12–60 µm. The Glomus is very common in soil because it is an endomycorrhizal fungus occurring on the roots of a variety of host plants and is strongly indicative of soil erosion and high rainfall activity (Basumatary & McDonald, 2017; Basumatary et al., 2024; van Geel et al., 2003).
Ascospores ellipsoidal to subglobose, 30 × 21 µm2 and brown to dark brown, the surface is covered with hyaline pits having 1 µm diam, thick-walled. Gelasinospora generally grows on excreta and decaying wood under relatively dry conditions (Lundqvist, 1972; van Geel & Aptroot, 2006).
Ascospores smooth, thick-walled, brown, ellipsoid to oblong, unevenly and asymmetrically four-celled, slightly curved, septa 25–30 × 12–16 μm2. Meliola is mostly found as parasites on the leaves and stems of a variety of hosts plant Acacia (Kokwaro, 1999).
Ascospores smooth, thick-walled, ellipsoidal to widely fusiform, asymmetrically two-celled, brown to dark brown, 22–27 × 8–11 μm2. Delitschia mostly grows on dung of herbivores (Bell, 1983) and is extremely indicative of grazing activity.
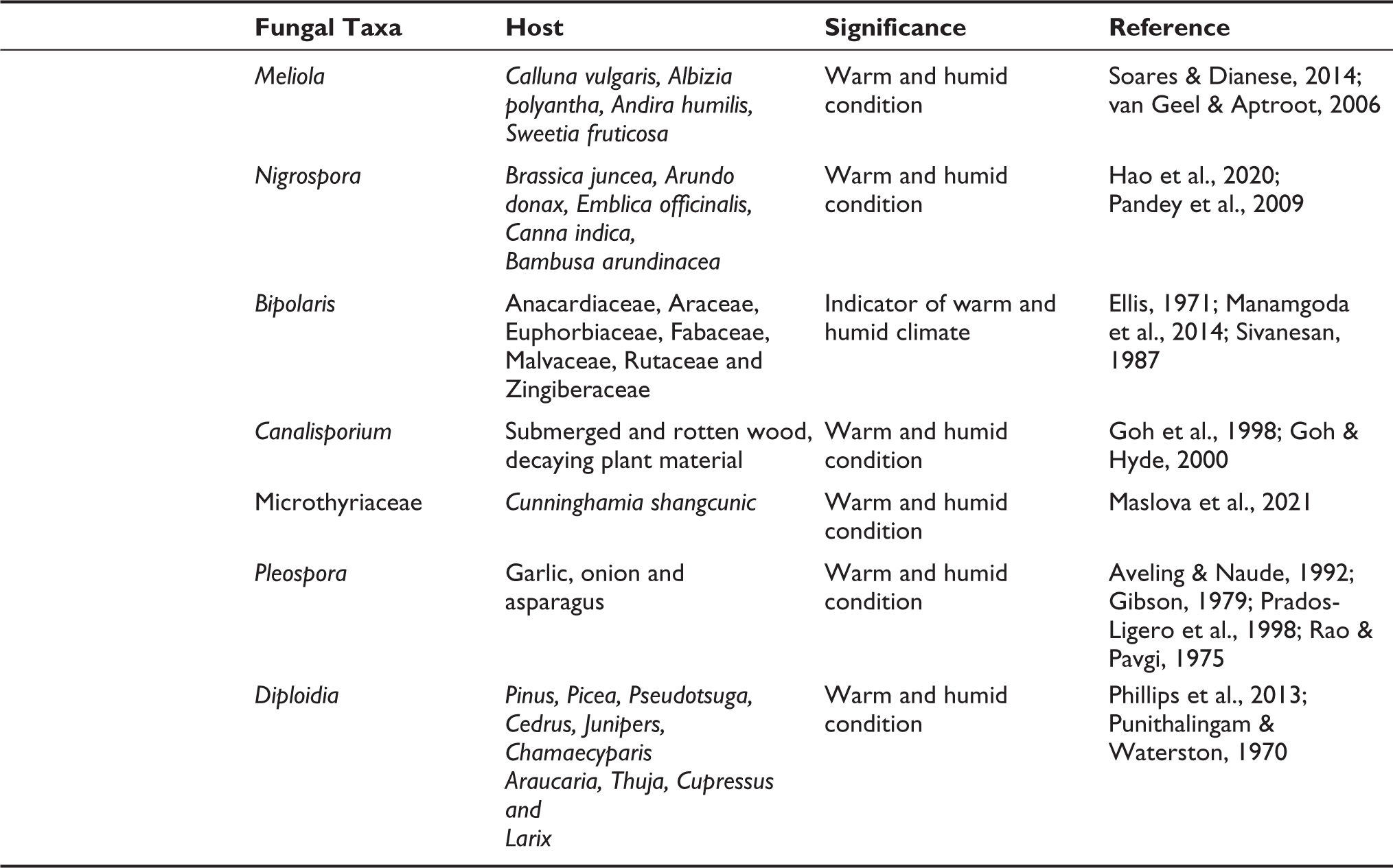
Conidia broader at apex and tapers down towards the base, 35–40 × 10–16 µm2 diam. The tri-septate spore has four to five longitudinal septa split into chambers. It is generally pathogenic in a wide variety of plants under warm and humid conditions.
Conidia septate multicellular (five- or more-celled), 60–120 µm long, brown to dark brown, parallel and thick-walled, club shaped. It is strongly indicative of humid and warm conditions and cultivated land (van Geel et al., 2011).
Shape muriform, 25–30 × 10–15 µm2 and brown, Pleospora-type consists of two forms: (i) with 3 transverse and 4–5 longitudinal septa and (ii) with 6 transverse and 4–6 longitudinal septa (Wei et al., 2021).
Conidia large, euseptate 70–75 × 18–23 µm2, and is found on dead plants and the colour ranges from light brown to dark brown (Duveiller et al., 2002).
Conidia ellipsoid to oval-shaped, pale to dark brown, 33–40 × 25–32 μm2 and smooth and thick-walled, restricted at the septa. Species of Canalisporium are common saprophytes on rotten and submerged wood and have a global tropical distribution (Goh et al., 1998; Goh & Hyde, 2000).
Globose or disc-shaped bodies, ascomata 45–50 × 65–76 µm2 and about 105–120 µm high, oblong or elongated, with a longitudinal cleft-like opening, black to dark brown. The presence of Microthyriaceae is strongly suggestive of high rainfall, dense forest vegetation under warm and humid conditions (Basumatary et al., 2024).
Spherical, black, hyaline spore 25–30 μm2 diam. Nigrospora is a cosmopolitan fungus that is more prolific in warm climates. It is typically found in soil and decomposing plant matter. Single conidium (14–20 µm in diameter) at their apex. Conidia are black, solitary and unicellular.
Ascospores ellipsoidal, light brown to dark brown. It clearly indicates a warm, muggy climate.
Discussion
In total, 20 fungal spore types were identified from the surface soil samples in relation to different vegetation types in PWS. The detailed morphology of the fungal spore is observed and characterised along with their significant value (Table 1). It is a preliminary study of fungal spores in surface soil samples in relation to the different vegetation types in PWS. This is partially due to the fact that it is typically challenging to identify a microfossil at the species or genus level. The majority of biological researchers specialise in just one species of organisms, and identifying (micro-) fragments of those organisms preserved in sediment samples already demands a great deal of specialised knowledge and experience (Prager et al., 2006). The implication of fungal spores to reconstruct the past climate and vegetation has continuously increased in recent years. The growth of fungal spores directly depends on climatic factors, such as temperature, precipitation and favourable places like sediment and dung. They show a strong indication of past activities of herbivorous animals and climatical and vegetational oscillations that took place in an area. Several previous studies evident that fungal spores are local in origin (van Asperen et al., 2021; van Geel, 2001; van Geel & Aptroot, 2006). It is well established that the presence or absence of herbivorous animals can be identified by the presence of coprophilous fungal spores, especially Sporormiella in the sediments (Gelorini et al., 2011; Loughlin et al., 2021; van Geel & Aptroot, 2006).
List of the fungal spore recovered from the surface soil sample in relation to different vegetation types in Pobitora Wildlife Sanctuary, Assam and their significant value.
Forestland
The six-surface soil sample from the Forestland area reveals that the fungal spore in the palynoassemblage both coprophilous and non-coprophilous fungal spores consistently encountered. The coprophilous fungal spore, such as Sporormiella, Ascodesmis and Saccobolus in palynoassemblage, is regularly observed, which is indicative of the herbivore’s activity in the wildlife sanctuary (Basumatary et al., 2024; van Geel et al., 2003). The coprophilous fungal spores are local in origin and travel short distances where sporulation takes place (Basumatary et al., 2024; Davis et al., 1977; van Geel et al., 2003). The study shows that Ascodesmis, Saccobolus and Sporormiella are more frequent in the studied sample, and they are therefore the most robust indicator of grazing activities (Basumatary & McDonald, 2017; Cugny et al., 2010). In particular, Sporormiella (including also possibly Sporormia and Preussia part-spores, van Geel & Aptroot, 2006) has been shown to be a valuable indicator of past grazers and density (Burney et al., 2003; Davis, 1987; Davis & Shafer, 2006; van Geel et al., 2003). Other coprophilous fungal spores, such as Cercophora, Podospora, Sordaria and Delitschia, were also consistently present in the studied sample and suggestive of the herbivore’s activity. It must be noted that Cercophora are indicative of woodland and grassland environments (Basumatary & McDonald, 2017). Non-coprophilous fungal spores, such as Alternaria, are good pathogens associated with herbaceous plants (Misaghi et al., 1978). The presence of Tetraploa in the palynoassemblage is observed which generally grows on the leaf base and stem (Ellis, 1971). Canalisporium in the assemblage is an indication of a warm and humid environment in the region. Other fungal spores, like Nigrospora, Gelasinospora, in the assemblage are strongly indicative of open land vegetation which is consistently present in the studied samples (Gelorini et al., 2011). The frequency of coprophilous fungal spores, namely Sporormiella, Ascodesmis and Saccobolus, was relatively higher in swamp areas as compared to the dense forest and grassland.
Grassland
In the grassland, the fungal spores both coprophilous and non-coprophilous fungal spores are regularly exhibited in the palynoassemblage. However, coprophilous fungal spores, especially Sporormiella, Ascodesmis and Saccobolus, are also consistently represented in the palynoassemblage as the forestland and swamp areas samples in the wildlife sanctuary. The regular presence of these fungal spores is strongly indicative of the herbivore’s activities in the grassland area. Non-coprophilous fungal spores, such as Helminthosporium and Alternaria, are commonly observed in the palynoassemblage and are significantly pathogenic and associated with Poaceae (Chattopadhyay & Chakraborti, 1953). The presence of Tetraploa in the assemblage is marked which generally grow on the leaves of grasses and associated herbs (Worobiec et al., 2009) which commonly encountered in the studied palynoassemblage.
Swamp
It is observed that the non-coprophilous fungal spore are dominate over the coprophilous fungal spore in the palynoassemblage. Non-coprophilous fungal spores, such as Tetraploa, Glomus, Alternaria, Diploidia and Bipolaris, are regularly exhibited in the assemblage and suggestive of warm and humid climatic conditions in the region. The coprophilous fungal spores, such as Sporormiella, Ascodesmis, Sordaria, Podospora and Saccobolus, are also consistently encountered and indicative of herbivore’s activity in the wildlife sanctuary. The preserved fungal spores originated from the surrounding areas and transported through the rainwater. The swamp area is serving as a centre place for wildlife and birds which come for drinking water. Among non-coprophilous fungal spores, Tetraploa is a type of saprophytic fungi and associated with the host plant Cyperaceae (a marshy taxa), which is indicative of the perennial waterlogged condition in the PWS. The presence of Glomus along with hyphae is frequent in the studied assemblages and the type of endomycorrhizal fungi that grow on the plant roots in the variety of host plants and their presence in the studied sample might be transported from the surrounding areas which is suggestive of the soil erosion in response to the high rainfall activity. The regular presence of Microthyriaceae in the palynoassemblage is marked and significant where the presence of Microthyriaceae is strongly indicative of the warm and humid climatic condition in the region (Basumatary et al., 2017, 2024; Basumatary & McDonald, 2017; van Geel et al., 1989, 2003).
Conclusion
A total of 20 fungal spore types both coprophilous and non-coprophilous were recovered from surface soil samples which is a prerequisite for accurate identification and for the interpretation of palaeoecology and palaeoherbivory analysis. It is observed that the depositional pattern of fungal spores varies in relation to different vegetation types and activities of herbivores in the PWS. Based on this study, it is characterised that the fungal spores both coprophilous and non-coprophilous from the swamp samples could be a reliable baseline for the reconstruction of palaeoecology in relation to palaeoherbivory analysis in PWS and its neighbouring areas.
Footnotes
Acknowledgements
We are grateful to Prof. Mahesh G. Thakkar, Director of the Birbal Sahni Institute of Palaeosciences, India, for providing the laboratory facilities needed for this study. We are grateful to Prof. Dhruv Sen Singh, Head, Department of Geology, University of Lucknow for library facilities. We also thankful to Department of Science and Technology, New Delhi for their financial support (DST No - EEQ/2021/000846). We also grateful to forest department, Govt. of Assam for their permission and guidance during our field visit in PWS. We thank the handling editor and two anonymous reviewers for their insightful suggestions to improve our manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received financial support for the research from department of science and technology, New Delhi (DST no - EEQ/2021/000846) Govt. of India.
