Abstract
Objective:
To evaluate the safety and effectiveness of isoniazid preventive therapy (IPT) in pregnant women living with HIV (WLWH) through a systematic review and meta-analysis.
Introduction:
Isoniazid preventive therapy (IPT) is recommended for preventive treatment of tuberculosis in high risk groups. However, evidence on its role in pregnant WLWH remains scarce.
Methods:
We performed a systematic review and meta-analysis to pool randomized controlled trials (RCTs) as well as non-randomized studies (NRS) where IPT was administered to pregnant WLWH (PROSPERO ID: CRD42024618836). PubMed, Embase, and Cochrane Central databases were searched for relevant articles, until November 15, 2024. Statistical analysis was performed using R Software v4.4.1 and a random-effects model was applied to pool risk ratios (RRs) along with 95% confidence intervals.
Results:
Five studies with a total of 45,402 patients (mean age = 30 years) were included. The risk of maternal mortality was significantly decreased in pregnant WLWH exposed to IPT compared with the control group (RR 0.42; 95% CI 0.20–0.92; p = 0.03). However, the risks of other outcomes including composite adverse pregnancy outcome (RR 0.90; 95% CI 0.56–1.43; p = 0.66), prematurity (RR 0.86; 95% CI 0.46–1.60; p = 0.63), low birthweight (RR 0.99; 95% CI 0.69–1.42; p = 0.95), very low birthweight (RR 1.28; 95% CI 0.39–4.23; p = 0.55), congenital anomalies (RR 1.48; 95% CI 0.59–3.75; p = 0.41), and hepatotoxicity (RR 0.99; 95% CI 0.71–1.37; p = 0.93) were comparable between the two groups.
Conclusion:
IPT in pregnant WLWH significantly reduces maternal mortality without increasing adverse pregnancy outcomes. These findings support the continued use of IPT during pregnancy, with careful monitoring for hepatotoxicity, and highlight its potential role as an important maternal health intervention in high TB/HIV burden regions.
Keywords
Introduction
Tuberculosis (TB) prevention among WLWH is of high priority in the endemic world. 1 Pregnancy and HIV are considered independent risk factors for Tuberculosis (TB). It is a major non-obstetric cause of maternal mortality in pregnancy, especially in poor healthcare setups, and is associated with poor outcomes in newborns.2,3 Therefore, WHO recommends antiretroviral therapy (ART) in all persons with HIV, including in pregnancy. 4 Provision of Isoniazid treatment is considered to bring down the risk of both active incidence and latent episodes of TB infection, thus providing the greatest relief to HIV-positive patients and pregnant females being exposed to TB.5,6 Further, Isoniazid Preventive therapy (IPT) is also found to have a better safety profile, compared to other TB medicines, especially in immunocompromised patients, including pregnant women.3,7
Recently, a database cohort study has shown better pregnancy outcomes and reduction in TB risk among patients exposed to IPT along with ART compared to ART alone. 8 Previously cluster-randomized trials conducted in Brazil revealed the systematic implementation of IPT within HIV clinics offered with ART led to a notable 31% decline in the incidence of TB. 9 Current studies have also indicated better protection by IPT in HIV-positive patients against TB with or without using ART.10,11 For this reason, WHO recommends preventive therapy by Isoniazid for up to 6 months to people with either prior TB or living with HIV. 4 According to 2018 guidelines for Latent Tuberculosis infection (LTBI) isoniazid therapy is strongly recommended in pregnancy. However, clinical decision-making is critical to judge the suitable time for the commencement of preventive therapy as pregnancy has twice the higher risk of hepatotoxicity compared to non-pregnant females. Thus, constant monitoring is required alongside baseline assessment and liver function tests. 12
Earlier systematic reviews that evaluated the safety of IPT among pregnant women found inconsistent evidence of IPT and adverse outcomes of pregnancy. 13 However, no review has been done to identify the outcome of IPT among pregnant WLWH. A recent secondary analysis by Gupta et al. 14 found a higher rate of adverse events with the initiation of IPT in the first trimester of pregnancy alarming the crucial need for new knowledge to fill the gaps of information in previous recommendations and guidelines. Therefore, we performed a systematic review and meta-analysis focused on maternal and fetal outcomes following IPT exposure in pregnant WLWH compared to other recommended regimens or with no treatment.
Methods
The protocol of this review is registered with the International Prospective Register of Systematic Reviews (PROSPERO) under ID: CRD42024618836. The systematic review was conducted according to the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions and the PRISMA reporting guidelines for systematic reviews. 15
Data source and search strategy
A comprehensive literature search was performed to retrieve potentially relevant articles from three major databases, including PubMed, Embase, and the Cochrane Library from inception to November 15, 2024. Keywords and their synonyms were combined with the Boolean operators “AND” and “OR.” The literature search was done independently by two reviewers (S.S. and H.K.), followed by a manual review of the references in the included studies, previous related meta-analyses, and narrative reviews to identify all the possible articles. We did not apply any time or language restrictions in our search. A reference and suggestion list of relevant articles was thoroughly evaluated to look for any missed articles. The detailed search strategy is provided in Supplemental Table 1.
Study selection and data extraction
The studies identified through the literature search on the above databases were imported into EndNote software, and duplicates were removed. Two authors (S.S. and H.K.) independently carried out primary screening based on their titles and abstracts, followed by the review of the full-text articles of the included articles for secondary screening. In cases of disagreement, the third author (A.) was consulted. Data extraction of the baseline characteristics and individual outcome measures from the result section of each included study was extracted into a pretest Excel sheet, which included information such as the first author’s name, publication year, country of origin, study design, sample size (intervention or control groups), race, mean age, Female sex (%), and follow-ups for baseline characteristics.
Eligibility criteria
The aim of this review was structured using the PICO framework. Participants (P) were defined as pregnant WLWH, either already pregnant at enrollment or who became pregnant during the intervention period. The intervention arm (I) included women provided with isoniazid preventive therapy (IPT) initiated during pregnancy. The comparator arm (C) included pregnant WLWH who did not receive IPT, received placebo, or no preventive therapy. Outcomes (O) assessed included both pregnancy and maternal events. Pregnancy outcomes were: (1) composite adverse pregnancy outcome, defined as spontaneous abortion (<20 weeks gestation), ectopic pregnancy, stillbirth (⩾20 weeks gestation), or induced abortion; (2) prematurity (<37 weeks gestation); (3) low birth weight (<2500 g); (4) very low birth weight (<1500 g); and (5) congenital anomaly. Maternal outcomes included (1) maternal mortality and (2) hepatotoxicity events.
Exclusion criteria were as follows: (1) studies that included non-pregnant women, HIV-negative women, or women with major co-morbidities unrelated to HIV/TB (e.g. malignancy, renal failure); (2) studies in which TB preventive regimens were used without isoniazid; (3) studies lacking a comparator arm (placebo, no therapy, or non-IPT); (4) non-original research such as reviews, meta-analyses, case reports, editorials, or commentaries; (5) conference abstracts and non–peer-reviewed preprints, to ensure inclusion of complete and quality-assured data. No language restrictions were applied. Access restrictions (open access vs subscription-only) did not bias inclusion, as all full texts were obtained through institutional access, interlibrary loan, or direct author contact.
Quality assessment
The Cochrane Collaboration tool for assessing the risk of bias in randomized studies (RoB 2) was used to assess the risk of bias in included randomized controlled trials (RCTs). 16 Nonrandomized studies were scrutinized with the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool. 17 In the RoB 2 assessment, each trial is graded as high risk, low risk, or with some concerns across five different domains. Meanwhile, the ROBINS-I tool assesses the risk of bias in non-randomized studies across seven domains: confounding, participant selection, intervention classification, deviations from intended interventions, missing data, outcome measurement, and selection of reported results, grading bias as low, moderate, serious, critical, or with no information. Two authors independently performed the quality assessment (S.S, H.K.), with disagreements resolved by consensus with the third author (M.A). Publication bias was considered but not formally assessed with funnel plots or Egger’s regression test, as the small number of included studies (<10 per outcome) renders these methods unreliable. 18
Statistical analysis
This systematic review and meta-analysis were carried out employing a random effects model using a pooled risk ratio with a 95% Confidence Interval (CI) for binary outcomes. The statistical analyses were conducted using R Software v4.4.1 using the “meta” package and function “metabin” for the dichotomous outcomes. Sensitivity analyses were performed using a leave-one-out approach, sequentially excluding each study to assess its influence on pooled estimates and to determine whether findings were driven by any single large study. Forest plots were employed to visualize the effect sizes of individual trials and the aggregated overall effect measure. To assess heterogeneity, I 2 statistical calculations were analyzed. While, for the assessment of significant relations, criteria of p-value < 0.05 and I 2 > 25% were set.
Results
Study selection
A total of 445 articles were initially identified through the electronic database search. The initial screening performed based on titles and abstracts after removing duplicate articles resulted in 32 articles for full-text review. The full text review led to the exclusion of 27 articles. Finally, five studies met our inclusion criteria and were included in the meta-analysis.8,14,19–21 The details of our literature search and study selection process are provided in the PRISMA flow diagram (Figure 1).
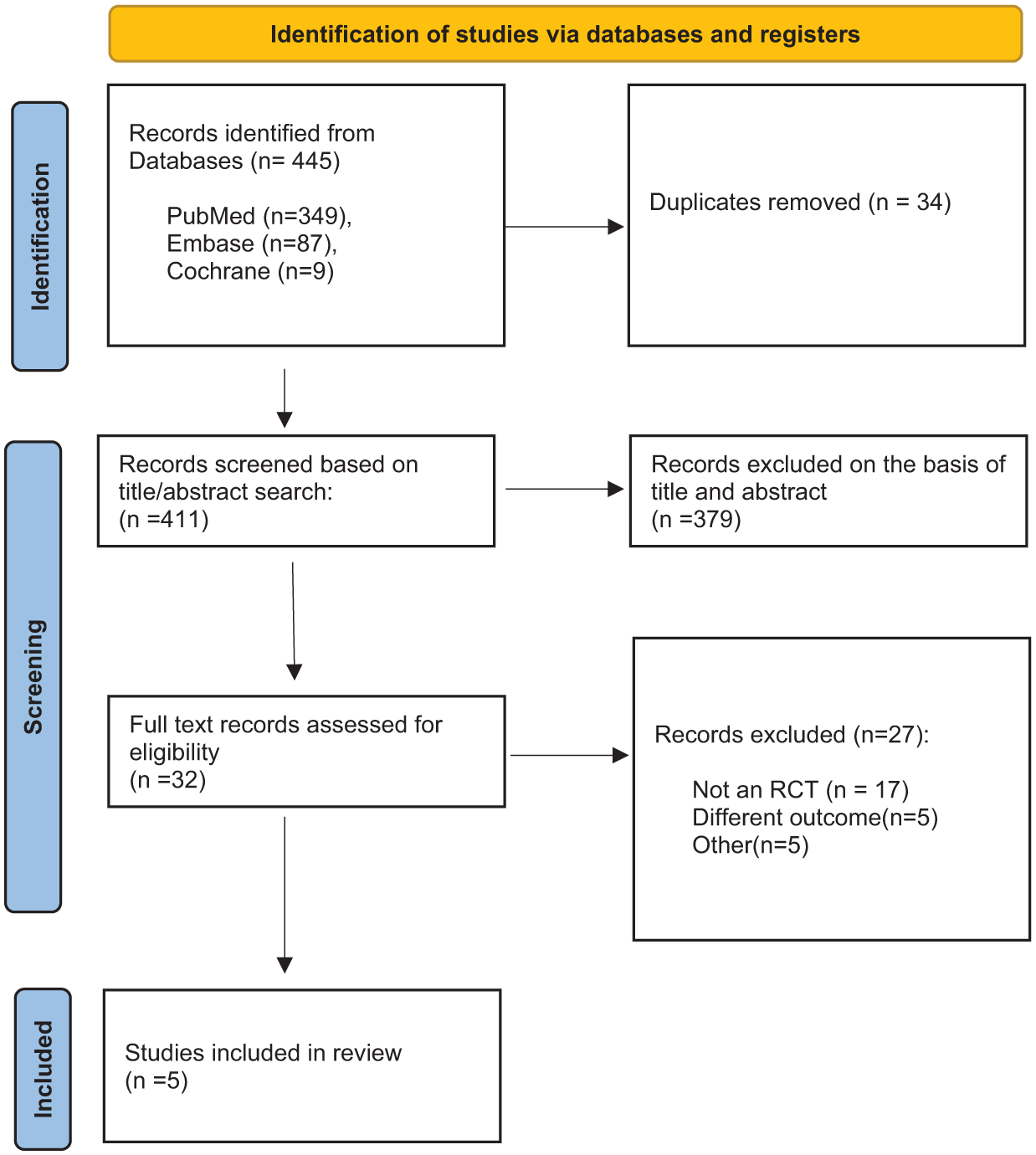
PRISMA flow chart of the screening process.
Baseline characteristics
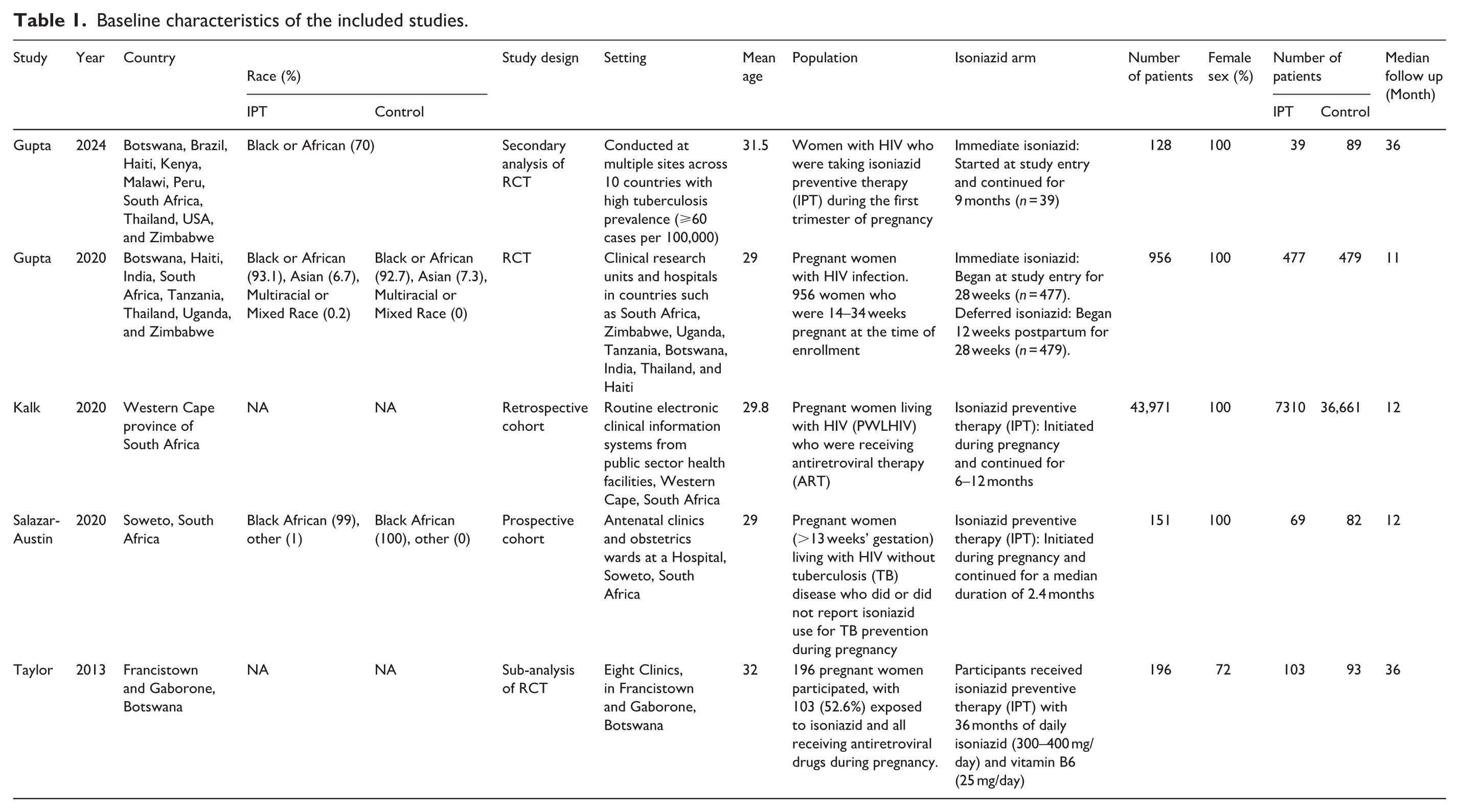
Our systematic review and meta-analysis included 5 studies with a total of 45,402 patients. The mean age of the patients was 30.2 years and the mean follow up period was 21 months. All the studies included pregnant WLWH; four specifically enrolled pregnant women,8,14,19,21 while one study included women who became pregnant during the trial. 20 Three out of five studies were conducted in Africa,8,19,20 while the remaining two had participants from multiple countries with a high prevalence of tuberculosis (⩾60 cases per 100,000 population).14,21 Among the included studies, Gupta et al. 21 represented the principal TB APPRISE (IMPAACT P1078) randomized controlled trial. Gupta et al. 14 reported a secondary analysis of outcomes in women from the deferred arm who conceived while receiving postpartum IPT, and was therefore included as a separate study. Theron et al. 22 provided a sub-analysis of the same TB APPRISE population and was classified accordingly. The detailed baseline characteristics of the included studies are given in Table 1.
Baseline characteristics of the included studies.
Pregnancy outcomes
Composite adverse pregnancy outcome
The risk of composite adverse pregnancy outcome was lesser in IPT exposed females as compared to the control group, but this difference did not achieve statistical significance (RR 0.90; 95% CI 0.56–1.43; p = 0.66; Figure 2). The high heterogeneity (I2 = 61%) observed was reduced to zero after excluding Gupta et al. 21 (Supplemental Figure 1).

Forest plot comparing the outcome of composite adverse pregnancy outcome for IPT exposed HIV-infected pregnant females to control group.
Prematurity
The risk of prematurity was found comparable between the two groups (RR 0.86; 95% CI 0.46–1.60; p = 0.63; Figure 3). Excluding either Gupta et al. 21 or Salazar et al. reduced the significant heterogeneity (I2 = 56%) to zero (Supplemental Figure 2).

Forest plot comparing the outcome of prematurity for IPT exposed HIV-infected pregnant females to control group.
Low birthweight
There was no significant difference in the outcome of low birthweight between the two groups (RR 0.99; 95% CI 0.69–1.42; p = 0.95; Figure 4). Leave one out analysis showed that excluding Kalk et al. or Gupta et al. 21 reduced the heterogeneity from 62% to zero (Supplemental Figure 3).

Forest plot comparing the outcome of low birthweight for IPT exposed HIV-infected pregnant females to control group.
Very low birthweight
The risk of very low birthweight was comparable between the two groups (RR 1.28; 95% CI 0.39–4.23; p = 0.55; Figure 5). Low heterogeneity (I2 = 0%) present confirmed the consistency of the results between different studies.
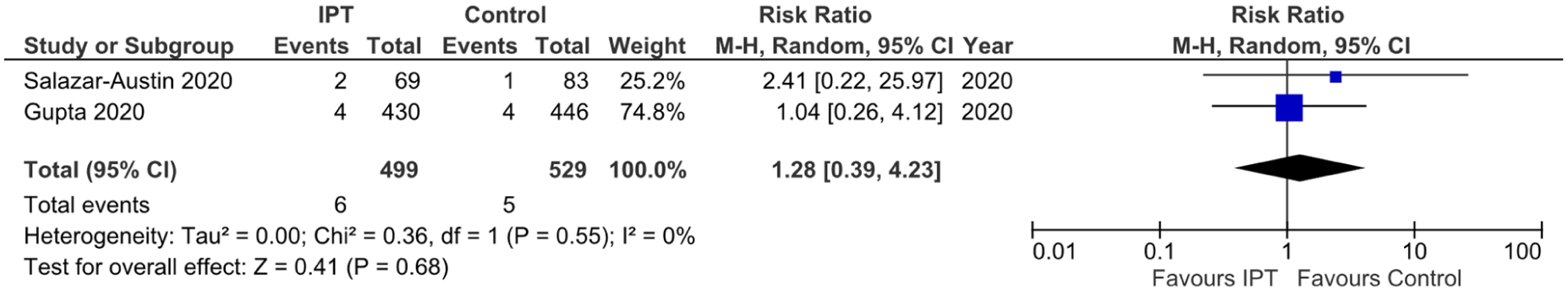
Forest plot comparing the outcome of very low birthweight for IPT exposed HIV-infected pregnant females to control group.
Congenital anomaly
There was no significant difference in the risk of congenital anomalies between the two groups (RR 1.48; 95% CI 0.59–3.75; p = 0.41; Figure 6) Insignificant heterogeneity (I2 = 0%) confirmed the consistency of results.
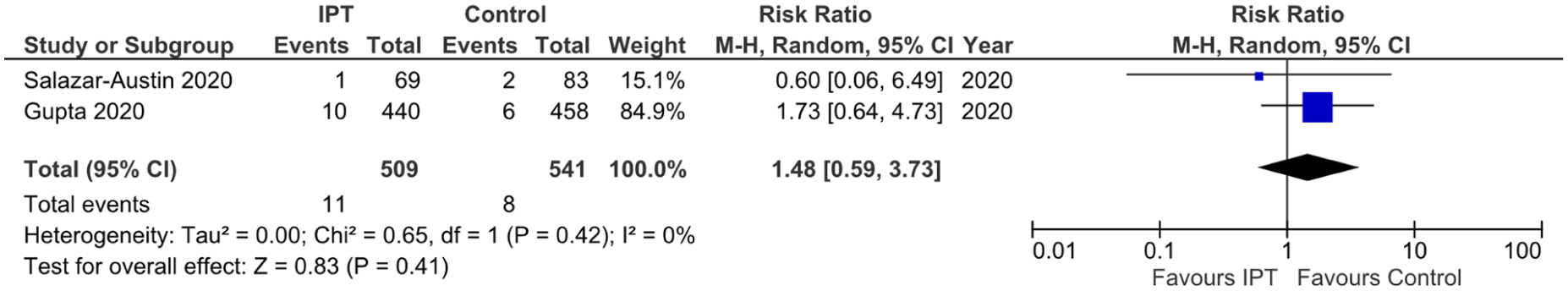
Forest plot comparing the outcome of congenital anomaly for IPT exposed HIV-infected pregnant females to control group.
Maternal outcomes
Maternal mortality
The risk of maternal mortality was significantly less in females exposed to IPT during the gestational period as compared to the control group (RR 0.42; 95% CI 0.20–0.92; p = 0.03; Figure 7). There was no heterogeneity across the studies for this outcome (I2 = 0%), indicating that the observed effect sizes were consistent and any variation was likely due to random chance rather than true differences between studies.

Forest plot comparing the outcome of maternal mortality for IPT exposed HIV-infected pregnant females to control group.
Hepatotoxicity
There was no significant difference in the risk of hepatotoxicity between the IPT-exposed and non-exposed females during their pregnancy (RR 0.99; 95% CI 0.71–1.37; p = 0.93; Figure 8). Low heterogeneity (I2 = 0%) further validated the consistency of findings between different studies.

Forest plot comparing the outcome of hepatotoxicity for IPT exposed HIV-infected pregnant females to control group.
Quality assessment
The trial by Gupta et al. 21 was considered to be of some concern due to missing outcome data. On the other hand, three out of four non-randomized studies were rated at moderate risk of bias8,14,19 and one was considered at serious risk of bias. 20 The detailed results of the risk of bias assessment are provided in the Supplemental Table 2.
Sensitivity analysis
Leave-one-out sensitivity analysis demonstrated that exclusion of either Gupta et al. 21 or Kalk et al., 8 which together contributed the largest sample sizes, caused minor shifts in effect estimates but did not alter the overall direction or statistical significance of the findings. This suggests that although larger studies carried more weight, the conclusions were not dependent on any single study. The detailed results of the sensitivity analysis are provided in the Supplemental Figures 1 to 3.
Discussion
This systematic review and meta-analysis is the first to evaluate the outcomes of IPT in pregnant WLWH. The results demonstrated that maternal mortality was significantly lower in females exposed to IPT during pregnancy compared to the control group, with no heterogeneity across studies. However, there were no significant differences in composite adverse pregnancy outcomes, prematurity, low birth weight, or congenital anomalies between the IPT-exposed and control groups.
In the study by Gupta et al., 21 six women died of which two were in the immediate group and four in the deferred group, with incidence rates of 0.40 and 0.78 per 100 person-years, respectively. The primary cause of death for four women (one in the immediate group and three in the deferred group) was liver failure, and all deaths occurred postpartum. Two of these women had received isoniazid (one from each group), while all were also on efavirenz–tenofovir–emtricitabine at the time of death. 23 However, IPT was not found to be associated with maternal mortality in adjusted survival analyses (aHR 0.75; 95% CI, 0.46–1.22), though there was a significant association with severe liver injury (aHR 1.51; 95% CI, 1.18–1.93), despite a low overall number of events (n = 127). 8 In contrast, our study found a significantly lower risk of maternal mortality in females exposed to IPT during pregnancy compared to the control group (RR 0.42; 95% CI 0.20–0.92; p = 0.03). The consistency of this outcome was confirmed by no heterogeneity across studies (I2 = 0%), suggesting the observed effect sizes were reliable and not due to random chance.
One randomized controlled trial (RCT) found more adverse pregnancy outcomes in the IPT group, possibly because nearly all participants in both the immediate and delayed IPT arms were on combination antiretroviral therapy (CART) at baseline. 23 Gupta et al. 21 observed higher adverse outcomes, particularly among women exposed to IPT during the first trimester. These included spontaneous abortion (<20 weeks gestation) and stillbirths, which varied depending on the trimester of exposure. 14 The composite adverse pregnancy outcome rate was higher in the immediate IPT group compared to the deferred group (23.6% vs 17.0%, difference: 6.7 percentage points; 95% CI: 0.8–11.9). 23 Our meta-analysis found that IPT-exposed females had a lower risk of composite adverse pregnancy outcomes compared to the control group, though the difference was not statistically significant (RR 0.90; 95% CI 0.56–1.43; p = 0.66). This contrasts with Gupta et al. (2024) who reported higher rates of adverse outcomes in the immediate IPT group compared to the deferred group. 23 The variability may be due to the timing of IPT initiation, with first-trimester exposure being a key factor. 14
Ngongondo et al. 24 noted that hepatotoxicity during IPT and ART was higher in women with pre-existing conditions such as elevated AST/ALT levels (23%), HIV/HBV co-infection (6%), or significant liver fibrosis or cirrhosis (3%) at baseline. Additionally, pregnancy itself was found to increase the risk of hepatotoxicity regardless of prior ART or Nevirapine exposure. 25 The study’s rigorous monthly liver enzyme monitoring may have led to an increased detection of hepatotoxicity, potentially including transient hepatic adaptations misclassified as injury. However, another study found no significant difference in hepatotoxicity between IPT and non-IPT groups. 8
We observed no significant difference in hepatotoxicity between IPT-exposed and control groups (RR 0.99; 95% CI 0.71–1.37; p = 0.93). This aligns with Kalk et al., who reported similar findings. However, Ngongondo et al. 24 noted increased hepatotoxicity in women with pre-existing liver conditions, suggesting that baseline health may influence outcomes. More detailed insights into hepatotoxicity from the TB APPRISE trial were recently provided by Montepiedra et al., 23 who found that the risk of hepatotoxicity was influenced not only by isoniazid exposure but also by the type of concomitant antiretroviral regimen and by pharmacogenetic factors. These findings suggest that individual variability, including genetic predisposition and ART selection, may partially explain the heterogeneity in hepatotoxicity outcomes observed across studies. 23
Gupta et al., 14 reported that while isoniazid exposure during conception and the first trimester was linked to spontaneous abortion, it was not associated with preterm birth or low birth weight. Other studies provided contrasting results. Salazar-Austin et al. 19 found no significant difference in low birth weight or size for gestational age between IPT-exposed and unexposed groups, noting their cohort primarily included women with less advanced HIV disease. Similarly, Taylor et al. reported no significant association between IPT or ART exposure and adverse outcomes like preterm delivery, low birth weight, or congenital abnormalities. Their study involved long-term IPT use combined with vitamin B6 supplementation. 20 In our study, the risk of prematurity was comparable between the IPT and control groups (RR 0.86; 95% CI 0.46–1.60; p = 0.63), consistent with findings by Salazar-Austin et al. 19 and Taylor et al., 20 who also found no significant association between IPT exposure and prematurity. Our analysis addressed high heterogeneity by excluding Gupta et al. 21 or Salazar-Austin et al., which reduced the variability to zero.
Theron et al. identified an increased risk of low birth weight and composite adverse pregnancy outcomes in women using IPT during pregnancy compared to those deferring IPT until postpartum, even after adjusting for maternal factors like nutritional status, HIV suppression and other clinical and demographic variables, highlighting a potentially independent effect of IPT on pregnancy outcomes. 22 Conversely, Salazar-Austin et al. 19 and Kalk et al. 8 reported lower risks of adverse pregnancy outcomes in the IPT-exposed group after controlling for confounding factors, suggesting a potential protective effect of IPT. Our study found no significant difference in low birth weight between the IPT and control groups (RR 0.99; 95% CI 0.69–1.42; p = 0.95). This result aligns with Salazar-Austin et al., 19 who found no association between IPT and low birth weight after controlling for confounders. These findings underscore the complexity of assessing IPT’s impact on pregnant WLWH and highlight the need for further studies to clarify these associations.
The studies we included in this review varied significantly, which influenced our results. Differences in maternal health conditions, the timing of IPT initiation during pregnancy, and overall HIV management practices contributed to these inconsistencies. For example, Gupta et al. 14 found more adverse outcomes in the IPT group, likely because most participants were on combination antiretroviral therapy (CART) at the start. 23 In contrast, Salazar-Austin et al. 19 observed fewer complications, possibly due to their cohort having less advanced HIV disease. To better understand these variations, we conducted a leave-one-out analysis. When we excluded Gupta et al., 21 the heterogeneity for composite adverse pregnancy outcomes dropped from 61% to zero. Similarly, for prematurity outcomes, removing either Gupta et al. 21 or Salazar-Austin et al. reduced heterogeneity from 56% to zero. This helped confirm that while individual studies showed differing results, our overall conclusions were more consistent once the impact of these outliers was addressed.
This review and meta-analysis study provides valuable insights into a critical public health concern by examining the impact of IPV on pregnant WLWH, a population often underserved in research. Its strengths lie in that this study is the first meta-analysis to examine the outcomes of IPT in this population. To address variability in the findings, we conducted leave-one-out analysis to identify which studies had the greatest influence on the results. This method clarified inconsistencies, enhancing the reliability of our conclusions with a solid foundation for future research and clinical guidelines.
This review has several limitations. First, the small number of included studies, with only 3–4 studies contributing to certain outcomes, reduces the statistical power and limits the conclusiveness of pooled estimates. Second, our analysis combined randomized controlled trials with non-randomized studies; while this was necessary to capture all available evidence, the differences in study design, data quality, and risk of bias reduce the certainty of pooled estimates. In particular, the observational study relied on routine electronic health records with >12% missing data, which may introduce bias. Third, the heterogeneity in study designs, populations, and treatment protocols presented challenges in synthesizing consistent conclusions, while key factors such as nutritional status and HIV suppression were not uniformly reported. Moreover, although we performed leave-one-out analyses to explore sources of heterogeneity, the small number of studies makes these results exploratory rather than definitive. Fourth, publication bias could not be formally assessed using funnel plots or Egger’s test, as fewer than 10 studies were available per outcome; this further reduces the certainty of the evidence. Finally, the focus on high-burden TB settings, particularly sub-Saharan Africa, may limit generalizability to other regions. Despite these limitations, the consistent finding of reduced maternal mortality across studies is noteworthy and highlights the need for larger, high-quality trials to confirm and expand upon these observations.
It is important to note that all participants in the TB APPRISE trial received pyridoxine (vitamin B6) and prenatal multivitamins until 40 weeks postpartum. This supplementation may have mitigated toxicity risks and could influence the generalizability of the findings to settings where such supplementation is not uniformly provided. Furthermore, additional insights have come from randomized TB prevention trials in which pregnancy was an exclusion criterion at enrollment. These include the BRIEF TB trial, TEMPRANO, and BOTUSA. While such trials were not included in our meta-analysis given our prespecified eligibility criteria, their secondary analyses provide complementary evidence.
Looking ahead, future research should focus on large-scale, randomized controlled trials that follow standardized protocols to clarify the mixed findings in this review. These studies must account for factors such as nutritional status, pre-existing liver conditions, and HIV viral suppression, as these may influence IPT outcomes.19,23 It’s crucial to identify the optimal timing for IPT initiation during pregnancy, as this may impact adverse pregnancy outcomes. 14 Moreover, investigating strategies to mitigate the risks of hepatotoxicity, such as enhanced monitoring or adjunctive treatments like vitamin B6, could improve safety.24,25 Research on patient adherence and socio-economic factors influencing treatment access would provide valuable insights into real-world effectiveness. Expanding studies to regions outside high-burden TB areas would also be beneficial for understanding the broader applicability of current guidelines.8,20 Long-term studies assessing both maternal and infant health outcomes will be essential for refining recommendations and improving care for pregnant WLWH globally.
Conclusion
In conclusion, this systematic review and meta-analysis show that IPT during pregnancy significantly reduces maternal mortality, with no clear differences in adverse pregnancy outcomes compared to controls. While findings were generally consistent, variability related to IPT timing, maternal health status, and study design warrants cautious interpretation. Careful monitoring for hepatotoxicity, particularly in women with underlying liver risk factors, remains essential. Importantly, a post-hoc analysis of the TB APPRISE trial has provided early evidence of infant growth effects, underscoring the need for further long-term studies across diverse settings to better define the maternal and infant safety of IPT and refine global recommendations. 26
Supplemental Material
sj-docx-1-iji-10.1177_03946320261427990 – Supplemental material for Outcomes of isoniazid preventive therapy in pregnant women living with HIV: A systematic review and meta-analysis
Supplemental material, sj-docx-1-iji-10.1177_03946320261427990 for Outcomes of isoniazid preventive therapy in pregnant women living with HIV: A systematic review and meta-analysis by Sufyan Shahid, Hasiba Karimi, Bilal Ahmad, Usama Hafeez, Biruk Demisse Ayalew and Abdullah in International Journal of Immunopathology and Pharmacology
Footnotes
Author contributions
S.S.: Conceptualization, Writing, Analysis.
H.K.: Writing and reviewing.
B.A.: Screening, Risk of Bias Assessment, Writing.
U.H.: Screening, Risk of Bias Assessment, Writing and reviewing.
B.D.A: Writing and Reviewing.
A.: Analysis, Supervision, Writing and Reviewing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
No ethical approval was required for this study design, as all data were obtained from publicly available sources.
Informed consent
“Consent to participate” does not apply to this research.
Data availability
All data were obtained from publicly available sources in this manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
