Abstract
Objective:
The aim of this study was to investigate the effects of halofuginone (HF) on gastric cancer cells and whether the combination of HF and trametinib has synergistic effects.
Introduction:
Halofuginone, a natural small molecule isolated from the plant Dichroa febrifuga, has been found to have anticancer activity in a variety of cancers, but few studies on HF in gastric cancer.
Methods:
cell viability was performed using the CellTiterGlo assay and apoptosis and cell cycle analysis was performed by Annexin V-FITC staining and PI staining. We also analyzed RNA sequencing and differentially expressed genes was shown using the heatmap. Western blot and qPCR were carried out to determine the expression of pro-apoptotic and anti-apoptotic proteins.
Results:
HF inhibited proliferation and induced apoptosis in gastric cancer cells in a dose-dependent manner. HF induced the expression of p-ERK in gastric cancer cells. HF and trametinib synergistically inhibited the gastric cancer cell proliferation. Trametinib inhibited HF-induced p-ERK expression. HF reduced anti-apoptotic protein Mcl-1 expression and reduced trametinib-induced upregulation of Mcl-1 expression.
Conclusion:
HF exerts its anti-cancer effects in gastric cancer and has a synergistic inhibition with trametinib, which may provide a novel therapeutic strategy for gastric cancer.
Introduction
Gastric cancer is the fifth most common cancer and the third most common cause of cancer death worldwide. 1 Traditionally, surgery has been the cornerstone of curative-intent treatment for gastric cancer. However, the realization that gastric cancer is often a systemic disease, even at an early-stage presentation, has led to the development and integration of various systemic treatment modalities, including chemotherapy, targeted therapy, and immunotherapy. The advent of targeted therapies has revolutionized the treatment paradigm for gastric cancer. Trastuzumab, a monoclonal antibody against HER2, as a standard treatment option for HER2-positive gastric cancer, showing a significant survival benefit when combined with chemotherapy. 2 Immunotherapy has also emerged as a game-changer in the treatment of gastric cancer. The KEYNOTE-062 trial explored the role of pembrolizumab in the first-line treatment of advanced gastric cancer, and subsequent studies have further refined the use of immunotherapy, combining it with chemotherapy or other targeted agents to enhance efficacy.3,4
Against this backdrop of continuous evolution in systemic treatment for gastric cancer, chemotherapy has long been used in the neoadjuvant, adjuvant, and metastatic settings and remains the backbone of treatment in advanced settings, but chemotherapy may cause side effects and drug resistance. Recent research has also focused on novel combinations and strategies to overcome resistance and improve treatment outcomes. Additionally, efforts are being made to identify predictive biomarkers more accurately to guide personalized treatment decisions. 5 Plant-derived natural products with fewer side effects and high efficacy have played an increasing role in cancer treatment. 6 Moreover, many clinically proven anticancer drugs are plant-derived natural products. 7
Halofuginone (HF), a derivative of febrifugine, was originally isolated from the plant Dichroa febrifuga. 6 It has been shown that HF exhibits significant anticaner activity in a variety of cancers, such as colon cancer, 8 breast cancer, 6 liver cancer, 9 lung cancer, 10 prostate cancer, 11 pancreatic cancer, 12 melanoma, 13 and ovarian cancer, 14 but also showed synergistic effect with other cancer treatments, chemotherapy, 15 radiotherapy, 16 and reverse effects on drug-resistance in cancers, 17 suggesting that HF may be a potentially effective natural product for cancer treatment. HF shows its anticancer effect by inhibiting tumor cell proliferation and metastasis, inducing apoptosis, autophagy, and cell cycle arrest, effects on synergistic sensitization, reversing drug resistance, and so on.
Mutations of KRAS have also been identified in many human cancers and result in constitutive activation of downstream signaling pathways mediated by phosphatidylinositol 3-kinase and mitogen-activated protein kinases (MAPK). 18 The median incidence of a KRAS mutation was 6.5% ranging from 0% to 29% in gastric cancer. The median incidence of KRAS mutations was similar in studies from the East and the West (East: 6%, ranging from 0% to 20%; West: 7.5%, ranging from 0% to 29%) in gastric cancer. Trametinib, a selective allosteric inhibitor of MEK1/MEK2, demonstrated preclinical and clinical activity in KRAS-mutant non-small cell lung cancer. However, patients on trametinib had more clinically significant adverse events. 19
Although studies have reported that HF shows anticancer activity in a variety of cancers, there are few studies on HF in gastric cancer. In addition, there are few studies about the efficacy of trametinib in KRAS-mutated gastric cancer. In this study, the effects of HF on the proliferation and signaling pathway in gastric cancer cells, the effects of trametinib on the gastric cancer cell proliferation with KRAS mutation, and whether the combination of HF and trametinib has synergistic effect was investigated. The results showed that HF inhibited the gastric cancer cell proliferation, induced cell apoptosis and the expression of p-ERK. The combination of HF and trametinib had a synergistic effect on inhibiting the gastric cancer cell proliferation. This study may provide a novel therapeutic strategy for gastric cancer.
Materials and methods
Reagents and antibodies
Human gastric cancer cell lines AGS and NCI-N87 were purchased from the American Type Culture Collection (ATCC, Manassas, VA, United States). RPMI-1640 medium, fetal bovine serum, and penicillin-streptavidin were purchased from Gibco, Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Antibodies against Mcl-1, Bax, Bcl-2, phospho-p44/42 MAPK (p-ERK1/2; Thr202/Tyr204), and p-Akt were obtained from Cell Signaling Technology (Boston, MA, USA). ERK1/2, Akt, and β-actin were obtained from Proteintech Group, Inc. (Wuhan, Hubei, China).
Cell lines and cell culture
Human gastric cancer cell lines AGS (KRAS (c.35G>A), p.G12D) and NCI-N87 were cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum and 1% penicillin-streptavidin at 37°C in 5% CO2.
Cell viability assay and colony formation assay
Cell viability was performed using the CellTiterGlo (CTG, Promega, Madison, WI) assay. Cells were plated in 96-well plates at a density of 8 × 103 cells per well, and treated with different concentrations of HF for 1, 2, 3, 4, and 5 days at 37°C. CTG was added for 30 min after HF treatment, and luminescence intensity was measured by a microplate reader. As the colony formation assay, cells were seeded into six-well plates at a density of 2 × 103 cells per well, treated with HF, fixed and stained with crystal violet solution, and took photos with a camera.
Cell apoptosis analysis
Cells were cultured and treated with HF (0, 0.05, and 0.2 μM) for 24 h followed by staining with Annexin V-FITC and PI (Beijing 4A Biotech Co., Ltd, Beijing, China) for apoptosis analysis and staining with PI (Beijing 4A Biotech Co., Ltd, Beijing, China) for cell cycle analysis. Data analysis was performed using NovoExpress v1.3.4.
RNA-Seq analysis
AGS cells were incubated with DMSO or HF (0, 0.05, and 0.2 μM) for 24 h, and total RNA was extracted and submitted to Novogene (Beijing, China) for Illumina NovaSeq 6000 sequencing. Differentially expressed genes (DEGs; log2 FC ⩾ 1, p < 0.05) was shown using the heatmap. ClusterProfiler R package (3.8.1) was used to test the statistical enrichment of DEGs in KEGG pathways.
Quantitative real-time polymerase chain reaction (qPCR) analysis
Cells were cultured in six-well plates, and the total RNA of cells was isolated using Trizol (Beyotime Biotechnology, Shanghai, China). cDNA was obtained using TransScript® II All-in-One First-Strand cDNA Synthesis SuperMix for qPCR (One-Step gDNA Removal; TransGen Biotech Co., Ltd., Beijing, China) according to the manufacturer’s instructions. The GAPDH was used as the reference gene. The primer sequences are protein tyrosine phosphatase, receptor type, R (PTPRR)-sense (5′-CAGCCAGCCGAATTCTCACA-3′), PTPRR-antisense (5′-AGTTCCATGACGCGGAATATCAA-3′), GAPDH-sense (5′-GCACCGTCAAGGCTGAGAAC-3′), and GAPDH-antisense (5′-TGGTGAAGACGCCAGTGGA-3′). The qPCR was carried out using AceQ®qPCR SYBR Green Master Mix (Vazyme, Nanjing, China) according to the manufacturer’s instructions. The 2−ΔΔCT method was used to calculate relative gene expression. The control was defined as gene expression = 1. Relative expression of the gene was expressed as fold-change compared with the control.
Western blot analysis
Cells were washed twice with cold PBS, and cell lysates were obtained using RIPA lysis buffer (50 mM Tris (pH 7.4), 150 mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, sodium orthovanadate, sodium fluoride, EDTA, and leupeptin; Beyotime Biotechnology, Shanghai, China) containing protease inhibitor PMSF (1 mM). The protein concentration was determined using the BCA protein assay (Beyotime Biotechnology, Shanghai, China). Equal amounts of proteins (20–30 µg/well) were separated on SDS-PAGE and transferred to polyvinylidene fluoride membranes (Immobilon®-P Transfer Membrane, Millipore) using wet transfer. After blocking in 5% skim milk in TBS-Tween-20 (TBST) for 1 h at room temperature, the membrane was then incubated with primary antibodies overnight at 4°C. Then the membrane was washed with TBST, and incubated with horseradish peroxidase-conjugated anti-rabbit or anti-mouse secondary antibodies at room temperature for 1 h, followed by washing with TBST. Finally, immunoreactive bands were detected by enhanced chemiluminescence reagent.
Statistical analysis
Data statistical analysis was performed using GraphPad Prism 8.0. The apoptosis data were analyzed using two-tailed Student’s t test. A p value < 0.05 was considered as statistically significant. All data subjected to statistical evaluation were gathered from at least three independent repeats of experiments.
Results
HF inhibited cell viability in gastric cancer cells
To investigate whether HF could show an antiproliferative effect on gastric cancer, two gastric cancer cell lines, AGS and NCI-N87 cells, were treated with different concentrations of HF for 5 days. Results indicated that HF significantly inhibited the viability of two gastric cancer cell lines, with IC50 of 0.07 μM in AGS cell and 0.06 μM in NCI-N87 cell (Figure 1(a) and (b)). Two gastric cancer cells were treated with different concentrations of HF (0, 0.05, and 0.20 μM) for 5 days. As shown in Figure 1(c) and (d), HF inhibited AGS and NCI-N87 cell viability in a dose- and time-dependent manner. In addition, HF markedly inhibited colony formation of the two cell lines in a dose-dependent manner (Figure 1(e) and (f)). These results suggested that HF could remarkably inhibit AGS and NCI-N87 proliferation.
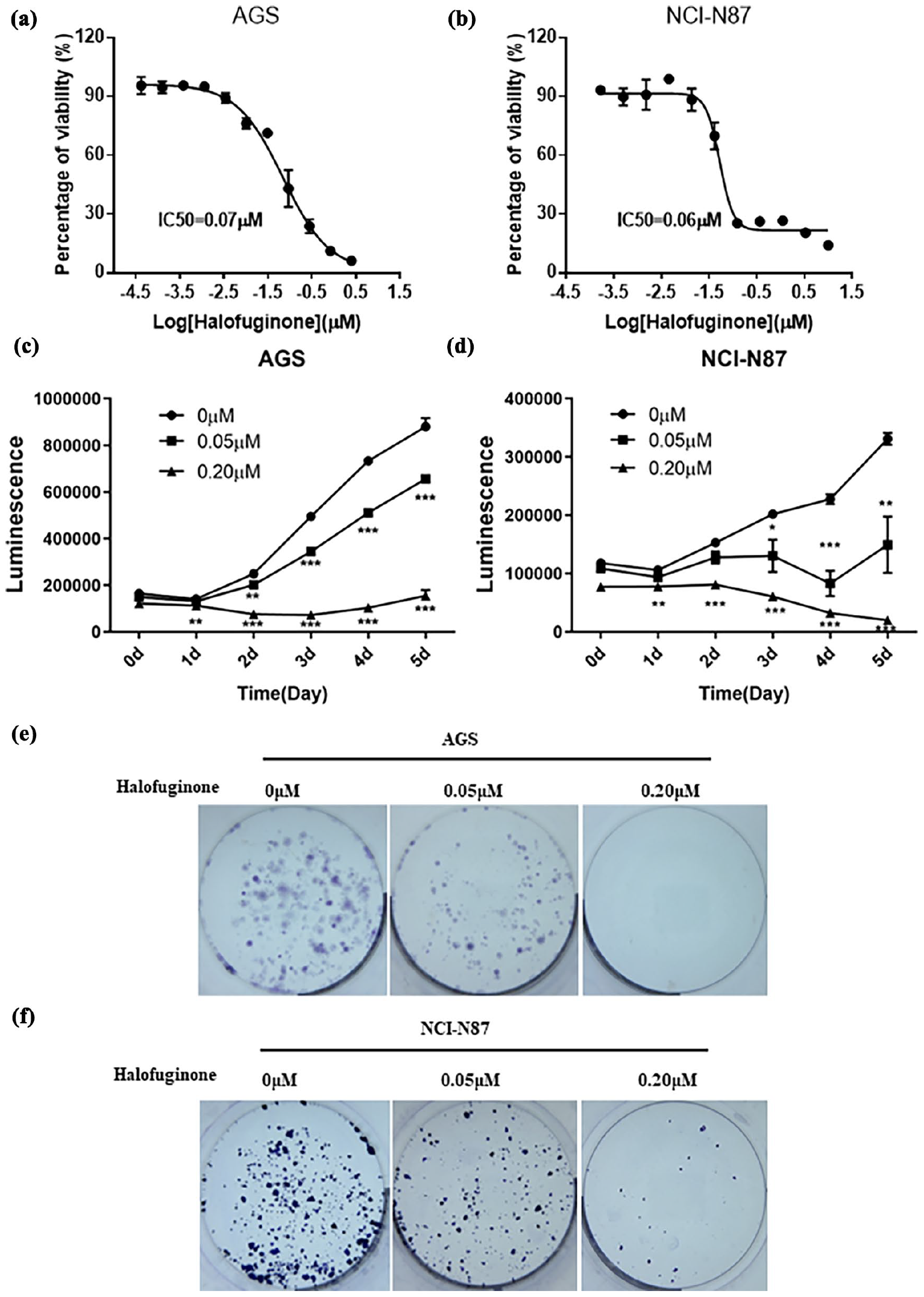
Halofuginone significantly inhibited cell growth in AGS and NCI-N87 cells. (a) and (b) The dose-response curves and IC50 values of halofuginone in AGS and NCI-N87 cells, respectively. (c) and (d) The effects of halofuginone on cell viability in AGS and NCI-N87 cells, respectively. (e) and (f) The effects of halofuginone on colony formation in AGS and NCI-N87 cells, respectively. Cells were stained with crystal violet solution. Compared with control, *p < 0.05, **p < 0.01, ***p < 0.001.
HF induced apoptosis in gastric cancer cells
To explore the possible ways that HF inhibited gastric cancer cell proliferation, the effects of HF on cell apoptosis were determined. The results showed that HF induced apoptosis in two gastric cancer cell lines, AGS cell and NCI-N87 cell, in a dose-dependent manner (Figure 2(a)–(d)). Next, the expression of apoptosis-related proteins, pro-apoptotic protein Bax, and anti-apoptotic proteins Bcl-2 and Mcl-1 was further determined using western blot. HF could significantly down-regulate the expression of the anti-apoptotic protein Mcl-1 in AGS cells (Figure 2(e) and (f)), and anti-apoptotic proteins Bcl-2 and Mcl-1 in NCI-N87 cells (Figure 2(g) and (h)).

Halofuginone induced apoptosis in AGS and NCI-N87 cells. (a–d) Flow cytometry analysis of apoptosis by Annexin V-FITC/ PI staining in AGS and NCI-N87 cells with halofuginone for 48 h. (e–h) Western blotting analysis of the expression of apoptosis-related proteins in AGS and NCI-N87 cells with halofuginone for 24 h or 48 h. β-actin was used as a loading control. Compared with control, *p < 0.05, **p < 0.01, ***p < 0.001.
HF induced the expression of p-ERK and p-Akt in gastric cancer cells
We explored how HF regulates cancer-related key signaling pathways in gastric cancer cells. MAPK/extracellular signal-regulated kinase (ERK) pathway is reported to be associated with cell proliferation, differentiation, migration, senescence, and apoptosis. 20 The MAPK/ERK pathway is an attractive therapeutic target for gastric cancer. 21 Phosphorylated AKT has been implicated in the deregulation of apoptosis and proliferation. Overexpression of p-AKT is considered to be a therapeutic target for treating malignant tumors. 22 To assess the effects of HF on the MAPK/ERK pathway and p-Akt in gastric cancer cells, western-blot analysis was performed on two gastric cancer cells. As shown in Figure 3, the expression of p-ERK and p-Akt in two gastric cancer cell lines was significantly up-regulated after the cells were treated with HF for 24 h or 48 h, indicating that HF induced the expression of p-ERK and p-Akt in gastric cancer cells.

Halofuginone induced the expression of p-ERK and p-Akt in AGS and NCI-N87 cells. (a–d) Western blotting analysis of the protein expression of p-ERK and ERK in AGS and NCI-N87 cells with halofuginone for 24 h or 48 h. (e–h) Western blotting analysis of the protein expression of p-Akt and Akt in AGS and NCI-N87 cells with halofuginone for 24 h or 48 h. β-actin was used as a loading control. Compared with control, *p < 0.05, **p < 0.01, ***p < 0.001.
HF altered genome-wide gene expression in gastric cancer cells
To better explore the effects of HF on gene expression in gastric cancer cells, RNA sequencing was performed. DEGs were shown by a volcano map (Figure 4(a) and (b)). Compared with the control, there were 596 up-regulated genes and 354 down-regulated genes when AGS cells were treated with 0.05 μM HF, and there were 2880 up-regulated genes and 2081 down-regulated genes when AGS cells were treated with 0.20 μM HF. Expression changes of apoptosis-related genes were shown on a heatmap (Figure 4(c)). In addition, KEGG enrichment analysis was performed. As shown in Figure 4(e), DNA replication was the most abundant gene enrichment pathway when AGS cells were treated with 0.20 μM HF.

Halofuginone induced genome-wide gene alteration in AGS cells. (a) and (b) Volcano maps show DEGs in 0.05 μM halofuginone versus DMSO, and 0.20 μM halofuginone versus DMSO, respectively. (c) Heatmap shows expression changes of apoptosis-related DEGs. From left to right, DMSO, 0.05 μM halofuginone, and 0.20 μM halofuginone, respectively. (d) and (e) KEGG enrichment analysis in 0.05 μM halofuginone versus DMSO, and 0.20 μM halofuginone versus DMSO, respectively.
HF and trametinib synergistically inhibited gastric cancer cell proliferation
We found the synergistic effect of HF and trametinib (Figure 5(a) and (b)), but no synergistic effect of HF and Akt inhibitor Ipatasertib on gastric cancer cell proliferation (Figure 5(c) and (d)). A drug combination is usually classified as synergistic, antagonistic, or non-interactive. To quantify the degree of drug synergy, several models have been proposed, such as those based on the Highest single agent model (HSA), the Loewe additivity model (Loewe), the Bliss independence model (Bliss), and the Zero Interaction Potency (ZIP). 23 To evaluate the synergistic effect of HF and trametinib, and HF and Akt inhibitor Ipatasertib on gastric cancer cell proliferation, four models, HSA, Loewe, Bliss, and ZIP were used. The four models all showed that HF and trametinib had a synergistic effect on inhibiting gastric cancer cell proliferation (Figure 5(e) and (f)), but HF and Akt inhibitor Ipatasertib had no synergistic effect (Figure 5(g) and (h)).

Synergy effects of halofuginone and trametinib on cell viability in AGS cells. (a) and (b) The dose-response curves of AGS cells treated with the combination of halofuginone and trametinib. (c) and (d) The dose-response curves of AGS cells treated with the combination of halofuginone and Akt inhibitor Ipatasertib. (e) and (f) The heatmap and surface plot show the degree of halofuginone and trametinib synergy using four models, the Highest single agent model (HSA), the Loewe additivity model (Loewe), the Bliss independence model (Bliss), and the Zero Interaction Potency (ZIP). (g) and (h) The heatmap and surface plot show the degree of halofuginone and Akt inhibitor Ipatasertib synergy using four models, the HSA, the Loewe, the Bliss, and the ZIP. (i) The effects of halofuginone and trametinib on the protein expression of Mcl-1, ERK, and p-ERK. (j) DEGs associated with MAPK/ERK phosphorylation processes were identified by RNA-seq. (k) The effects of HF on PTPRR gene expression were verified by qPCR. **p < 0.01 and ***p < 0.001.
To further explore the molecular mechanism of the synergistic inhibitory effect of HF and trametinib on gastric cancer cell proliferation, the expression of p-ERK, ERK, and anti-apoptotic protein Mcl-1 was further detected by western-blot. As shown in Figure 5(i), HF induced the upregulation of p-ERK expression in AGS cells. Compared with HF, trametinib inhibited HF-induced p-ERK expression. Compared with trametinib, HF did not significantly change the expression of p-ERK. The combination of HF and 10 nM trametinib reduced p-ERK expression compared with 10 nM trametinib. HF, trametinib, or the combination of HF and trametinib had no significant effect on the expression of ERK protein. For the anti-apoptotic protein Mcl-1, HF reduced Mcl-1 expression, trametinib upregulated Mcl-1 expression, and HF reduced trametinib-induced upregulation of Mcl-1 expression, indicating that the combination of HF and trametinib may induce apoptosis, consistent with the synergistic effect of the combination described above. Collectively, it demonstrated that the combination of HF and trametinib exhibited synergistic inhibitory effects in gastric cancer cells and this combination might provide a novel therapeutic strategy for gastric cancer.
To better understand the possible regulatory pathways of HF-induced p-ERK expression in gastric cancer cells, RNA-Seq was performed. DEGs associated with MAPK/ERK phosphorylation processes were identified by RNA sequencing. The results showed that HF induced the down-regulation of PTPRR, a negative regulatory key gene upstream of the ERK signaling pathway in AGS cells (Figure 5(j)). The effect of HF on PTPRR gene expression was further verified by qPCR. The results showed that HF downregulated PTPRR expression, consistent with the RNA sequencing results (Figure 5(k)). These results suggest that HF may induce p-ERK expression in AGS cells by down-regulating PTPRR.
Discussion
Studies have shown that HF inhibits the growth of cancer cells. HF inhibited hepatocellular carcinoma HepG2 cell proliferation, with an IC50 of 72.7 nM for 72 h, 9 colon cancer DLD-1 cells, with an IC50 of 60.89 nM for 48 h, 24 and lung cancer NCI-H460 and NCI-H1299 cells, with an IC50 of 70 nM and 60nM, respectively. 17 In this study, our results showed that HF inhibited gastric cancer cell AGS and NCI-N87 proliferation, with an IC50 of 70 nM and 60 nM, respectively, similar to the IC50 reported in the above studies.
Studies have indicated that HF induces apoptosis in a variety of cancer cells, such as colon cancer, 25 lung cancer, 17 breast cancer, 6 and liver cancer 9 etc. Similar to these studies, our results showed that HF induced apoptosis in gastric cancer cells. We further determined the effects of HF on the expression of apoptosis-related proteins, pro-apoptotic protein Bax, and anti-apoptotic proteins Bcl-2 and Mcl-1. It indicated that HF could significantly reduce the expression of anti-apoptotic protein Mcl-1 in gastric cancer cells, which is consistent with reports that HF down-regulated Mcl-1 expression in hepatocellular carcinoma HepG2 cell line 9 and multiple myeloma cell lines. 15 This may indicate that Mcl-1 plays an important role in HF-induced cancer cell apoptosis. In addition, HF reduced trametinib-induced upregulation of Mcl-1 expression in combination with HF and trametinib, indicating that Mcl-1 may play a crucial role in synergistic growth inhibition.
The PTPRR gene with the structure of the extracellular domain, single transmembrane domain, and intracellular catalytic domain can regulate the dephosphorylation of downstream MAPK steering cell proliferation, differentiation, and other functions.26,27 PTPRR can dephosphorylate p-ERK, thus inhibiting the MAPK pathway. 28 In this study, the results showed that HF reduced PTPRR mRNA expression and induced phosphorylation of ERK. We hypothesized that HF may induce phosphorylation of ERK by down-regulating PTPRR. The MAPK/ERK pathway is subject to a large number of negative feedback loops, including direct phosphorylation by ERK1/2 (and RSK2) as well as transcriptionally induced feedback regulators, such as DUSPs and Sprouty proteins. 29 More evidence is needed to confirm these complex processes.
The MAPK/ERK pathway is an attractive therapeutic target for gastric cancer. 21 Trametinib was the first mitogen-activated protein/extracellular signal-regulated kinase (MEK) inhibitor approved for use in the treatment of advanced BRAFV600 mutant melanoma as a single agent and in combination with BRAF inhibitor, dabrafenib. 30 Inhibition of phosphorylated ERK, Ki-67, and an increase in p27 were used as biomarkers of MEK inhibitor activity. 30 Trametinib reduced the p-ERK level in gastric cancer cells. 21 Cutaneous toxicities were the most common side effects of using trametinib monotherapy. 30 In this study, the results showed that HF and trametinib synergistically inhibited gastric cancer cell proliferation. Therefore, the combination of HF and trametinib may reduce the dosage of trametinib, reducing the side effects of trametinib. This combination may provide a novel therapeutic strategy for gastric cancer. Given that trametinib monotherapy in KRAS-mutant cancers is limited by dose-dependent toxicities, the combination with HF may enable reduced trametinib dosage while maintaining efficacy. This is particularly significant for gastric cancer patients with compromised tolerance to chemotherapy, as demonstrated by our in vitro data showing that HF reduces trametinib-induced upregulation of the anti-apoptotic protein Mcl-1, potentially overcoming drug resistance mechanisms. Clinically, this strategy could be prioritized for KRAS-mutated gastric cancer subtypes, which currently lack targeted therapies beyond chemotherapy.
This study also has several limitations. This study exclusively relies on human gastric cancer cell lines (AGS and NCI-N87) in in vitro experiments, lacking validation in animal models. Cell line results may not fully reflect the complexity of tumor microenvironments and drug responses in vivo. The KRAS mutation status of the cell lines (AGS: KRAS G12D mutation; NCI-N87: wild-type KRAS) limits the generalizability of findings to other genetic subtypes of gastric cancer (e.g. HER2-positive, Epstein-Barr virus-associated types). Although this study suggests that HF induces ERK phosphorylation by downregulating PTPRR, the specific regulatory pathway remains incompletely clarified. The synergistic mechanism between HF and trametinib primarily focuses on Mcl-1 and ERK pathways, but potential interactions with other signaling networks (e.g. PI3K/Akt, JAK/STAT) remain uninvestigated, limiting a comprehensive understanding of their combined effects. Trametinib’s clinical application is limited by adverse effects (e.g. skin toxicity, gastrointestinal reactions). The study does not evaluate whether combination therapy can reduce trametinib dosage to mitigate side effects, requiring further preclinical safety assessments. RNA sequencing was performed only in AGS cells, lacking validation in other gastric cancer lines, which may affect the universality of differentially expressed gene (DEG) analysis. The study does not correlate molecular findings (e.g. p-ERK, Mcl-1 expression) with clinical patient data (e.g. tumor tissue samples, survival outcomes), weakening the direct relevance of basic research to clinical practice.
Conclusions
In summary, this study shows that HF inhibited the proliferation, induced the apoptosis of gastric cancer cells, and showed a synergistic effect with trametinib on inhibiting gastric cancer cell proliferation. Trametinib inhibited HF-induced p-ERK expression. HF reduced anti-apoptotic protein Mcl-1 expression and reduced trametinib-induced upregulation of Mcl-1 expression. Therefore, the combination of HF and trametinib may provide a novel therapeutic strategy for gastric cancer. From a translational standpoint, this study underscores the value of repurposing natural products in oncology. We propose that future investigations should prioritize clinical trials evaluating HF-trametinib combinations in KRAS-mutated gastric cancer, alongside correlative studies to monitor PTPRR and Mcl-1 as response biomarkers. Addressing these gaps will not only advance our understanding of ERK signaling dynamics but also pave the way for more tolerable, mechanism-based therapies in gastric cancer.
Footnotes
Author contributions
Sun Y.M. and Wang P: data curation; methodology; writing-original draft. Bai X.F. and Tian L: data curation. Zhong Y.X.: investigation; writing; formal analysis; conceptualization; methodology; project administration; writing-review and editing.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Beijing Hope Run Special Fund of Cancer Foundation of China [LC2020A25]; CAMS Innovation Fund for Medical Sciences [2021-12M-1-022]; and Wu Jieping Medical Foundation [2022-081-ZZ].
Ethics approval
Ethical approval for this study was obtained from Ethics Committee of National Cancer Cencer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College (AA/274-3476).
Informed consent
Written informed consent was obtained from legally authorized representatives before the study.
