Abstract
Previously, we identified that Celastrus orbiculatus, a traditional Chinese herbal medicine, exhibited prominent anti-inflammatory and anti-tumor activities. More recently, the formation of neutrophil extracellular traps (NETs) or NETosis has been recognized as a critical pathological event in the development of inflammatory and autoimmune diseases. The present study is aimed to explore the pharmacological effect of the total terpenoids from the stems of C. orbiculatus (TTC) on NETosis and underlying mechanisms, which may provide fundamental knowledge for future utilization of the Chinese medicine. Human neutrophils were isolated by density gradient centrifugation; lactase dehydrogenase (LDH) assay was used to detect cytotoxic effect of TTC on neutrophils. Moreover, we established phorbol-12-myristate-13-acetate (PMA)-induced NETosis. Quantitative and qualitative study of PMA-induced NET release was labeled by SYTOX™ Green. ROS production was determined by flow cytometry. The neutrophil NADPH oxidase (NOX) activity was assessed by lucigenin chemiluminescence assay, and the phosphorylation of NOX subunit was analyzed by immunoblot assay. TTC (5–80 μg.mL−1) had no predominant neutrophil cytotoxicity after 4 h exposure. PMA (200 ng.mL−1) significantly induced the formation of NETs after 4 h stimulus, whereas TTC dose-dependently (5–80 μg.mL−1) inhibited the process. TTC (40 μg.mL−1) blocked neutrophil elastase (NE) and myeloperoxidase (MPO) translocation from cytoplasm to nucleus and disrupted the formation of NET-associated deoxyribonucleic acid (DNA)–MPO and DNA–NE complexes. Moreover, TTC dose-dependently blocked PMA-mediated ROS production, and inhibited the NOX enzymatic activity of neutrophils upon PMA stimulus for 1 h. Finally, TTC suppressed PMA-induced phosphorylation of NOX subunit p40phox on Thr154 residue. TTC inhibited PMA-induced NOX phosphorylation, thereby suppressing NOX enzymatic activity and ROS generation in neutrophils undergoing NETosis. Consequently, TTC disrupted NETosis in the early stage of NOX-dependent NETs formation, which might serve as a promising anti-inflammatory agent by targeting suicidal NETosis.
Introduction
Traditional Chinese medicine (TCM) featured by plants of the genus Tripterygium and Celastrus both from Celastraceae family has shown great potential for treatment of inflammatory and autoimmune diseases. Today, TCM of Celastraceae family has garnered substantial interest as leadpoint for identification of novel phytochemicals.1,2 Triptolide, a well-established diterpenoid and prototypical nuclear factor-kappa (NF-κB) inhibitor, is originally isolated from Tripterygium wilfordii (Chinese: 雷公藤). 3 Although the efficacy of T. wilfordii for treating rheumatoid arthritis (RA) been recognized in controlled clinical trials,4,5 the herb has a very narrow therapeutic window and causes broad and severe toxicities.6,7 To this end, our lab has been focused on alternative herbal medicines from genus Celastrus that might possess better safety profiles than T. wilfordii.
Here, we introduce Celastrus orbiculatus (Chinese: 南蛇藤), or termed “Oriental bittersweet,” a high-climbing vine that is native to China and widely distributed in eastern Asia. The woody and twining stems of C. orbiculatus have been used as a folk medicine in treating a wide range of conditions.8–10 It is well documented by ancient Chinese physicians that the stems of C. orbiculatus can eliminate pathogenic factors mediated via wind and dampness, activate blood circulation and relieve pain, thereby improving the overall health of the tissues, organs, and joints. 11 The bulk of the bioactive constituents of C. orbiculatus are terpenoids and sesquiterpenes.2,12–16 Celastrol, a quinone methide triterpenoid isolated from the herb, has been extensively studied and assigned to treat autoimmune diseases, chronic inflammation, neurodegenerative diseases, and many types of cancers. 17 Previously, we have demonstrated that the ethyl acetate extract of the stems of C. orbiculatus exhibited significant anti-RA and anti-tumor activities in vivo and in vitro.18–21 Moreover, we have identified 11 kinds of primary chemical constituents from the stems of C. orbiculatus including six triterpenoids, two phenolics, two steroids, and a benzoquinone (Supplementary Table 1). 13
In comparison with conventional crude extract of Chinese herbal medicine, pharmaceutical agents made by enriched active components with controllable quality have become the mainstay of modernized Chinese medicine. Terpenoids constitute the largest class of natural products and are a rich reservoir of candidate compounds for drug discovery. Even though continued efforts are concentrated on characterization of purified terpenoid including triptolide and celastrol, we would like to attach more importance to terpenoid enriched fraction from the stems of C. orbiculatus. The total terpenoids of C. orbiculatus, which is referred to TTC, has been qualified by at least 60% content of terpenoid ingredients by our lab (China invention patent, license no. 200710025343.3).
Extracellular DNA traps (ETs) are part of the innate immune response and were first observed and described in activated neutrophils as neutrophil extracellular traps (NETs). In recent years, the phenomenon has been additionally identified in other immune cells such as eosinophils, mast cells, monocytes, or macrophages. 22 Much of the research on ETs has been conducted on neutrophils, most probably because neutrophils were the first to be associated with the production of such extracellular DNA structures. Moreover, neutrophils are the most abundant leukocytes in the blood and constitute the first line of host defense against invading pathogens. NETs were first discovered in 1996 as a pathway of cell death in neutrophils following chemical stimulation with phorbol-12-myristate-13-acetate (PMA) that was quite different from apoptosis and necrosis. 23 In 2004, Brinkmann et al. 24 further detailed the process of formation of NETs and termed it as NETosis, which is a neutrophil-specific suicidal biology through externalization of neutrophil nuclear DNA and granular contents for neutrophil sequestering pathogens and antimicrobial defense. Few years later, Fuchs et al. 25 examined that NETosis was completed 1–4 h after PMA stimulus and found that PMA-induced NETosis was dependent on reactive oxygen species (ROS) production, which was a beneficial suicide to the host defense. Apart from the best described suicidal NETosis, a groundbreaking study published in 2002 showed that neutrophils released NETs without exhibiting loss of nuclear or plasma membrane within 5–60 min, and it was independent of ROS. 26 Thus, vital NETosis has been described to be a rapid release of NETs. However, it still remains controversial whether and how suicidal and vital NETosis coexist. 27
The primary components of NETs are chromatin and histones conjugated with neutrophil proteases and peroxidases. At the initial stage of neutrophil activation, NADPH oxidase (NOX)-mediated oxidative burst has been implicated in degranulation of neutrophil proteases and peroxidases during NETosis. This is required for subsequent translocation of neutrophil elastase (NE) to the nucleus where NE cleaves histones and leads to chromatin decondensation, while myeloperoxidase (MPO) catalyzes the production of hypochlorous acid and regulates extracellular DNA release.28–30 Furthermore, citrullination of histone H3 (citH3) by peptidyl arginine deiminase 4 (PAD4) is central for NET formation and has been recognized as a biomarker of NETosis. 31 Currently, excessive formation of NETs has been considered as a dark side of neutrophils that plays pivotal role in tumor-associated thrombosis, hematogenous tumor metastasis, RA, autoimmune diseases, and so on.32,33 Thus, pharmacological approach to delay pathological NETosis has shed new light on the fight against inflammatory diseases.
In the present study, we aimed to ascertain whether TTC could serve as pharmacological antagonist of suicidal NETosis. PMA is a robust and well-defined inducer of NETosis by activating protein kinase C (PKC). In turn, PKC initiates membrane translocation and assembly of NOX subunits and further triggers NOX-dependent NET formation. 34 Therefore, NOX participates in the earlier stage of NETosis, and we further asked whether TTC could interfere with PMA-induced NOX activity. Our data suggest that TTC can effectively inhibit NOX-dependent NETosis, which may further advance the knowledge of C. orbiculatus in anti-inflammatory therapy highlighted by pathological NETosis.
Materials and methods
Preparation of TTC
Chinese materia medica C. orbiculatus farmed in the south-west areas of China was purchased from Zhixin Pharmaceutical Company (Batch number: 070510, Guangzhou, China) and further authenticated by Prof. Minjian Qin from China Pharmaceutical University (Nanjing, China). The method of TTC preparation has been authorized by our lab’s China invention patent (license no. 200710025343.3). In brief, 10 kg of the stem of C. orbiculatus was collected, chopped into small pieces, and soaked in six-fold volume of 90% ethanol for 1 h. The sample was subjected to refluxing extraction for a total of three times and ethanol was further evaporated in a rotary decompression tank. The concentrated extracts were suspended in water and sequentially refined by petroleum ether and ethyl acetate for three times, respectively. The ethyl acetate fraction was further purified by 20% ethanol and 220 g dried TTC was stored at 4°C. The content of total terpenoids was indicated by 3-acetyl-oleanolic acid and the content of TTC from three independent batches showed higher than 60% of the total extract. Dried TTC powder was completely dissolved in a small amount of dimethyl sulfoxide (DMSO) as a clear stock solution (100 mg.mL−1).Before use, it was diluted to the maximum concentration of working solution (800 μg.mL−1) using phenol-free RPMI-1640 medium after brief periods of sonication and filtered using a 0.22 μm syringe filter. Subsequently, 800 μg.mL−1 of working solution was further serially diluted to 400, 200, 100, and 50 μg.mL−1 in phenol-free RPMI-1640 medium. A 10-fold dilution of TTC in the culture plates was obtained as the final concentration less than 80 μg.mL−1. The same volume of diluted DMSO solution (final concentration: 0.08% v/v) served as vehicle control.
Isolation of neutrophils from peripheral human blood
Peripheral blood from healthy donors was collected in K2·ethylenediaminetetraacetic acid (EDTA) anticoagulant blood collection tubes and kept at room temperature before experiment. A signed informed consent was obtained from each donor, and the protocol was approved by Yangzhou Hospital of Chinese Medicine. The blood was used within 2 h of drawing from the donors. Neutrophils were isolated using PolymorphPrep™ density gradient media (Axis-Shield, Cat# 1114683) according to the manufacturer’s instructions with minor modifications. Red blood cells were then removed by ammonium–chloride–potassium (ACK) buffer (eBioscience, Cat# 00-4300-54). Isolated neutrophils washed by Hanks’ balanced salt solution (HBSS) and resuspended in RPMI 1640 cell culture media without phenol red. Purity of cells was >90% as determined by Wright–Giemsa staining.
Lactase dehydrogenase cytotoxicity assay
Freshly isolated neutrophils were resuspended at a density of 3 × 104 cells per well in a 96-well culture plates were treated with various concentrations of TTC (0, 5, 10, 20, 40, 80 μg.mL−1) for 4 and 10 h, and the potential cytotoxicity of TTC in neutrophils was determined by lactase dehydrogenase (LDH) release assay using LDH Cytotoxicity Assay Kit (Nanjing Jiancheng Bioengineering Institute, China, Cat# A020-2).
NETotic assay
Freshly isolated neutrophils were seeded at 3 × 104 cells per well in a 96-well black, transparent bottom microplates (Costar, Cat# 3603) in phenol red-free RPMI media. NETotic assay was initiated by 200 ng.mL−1 of PMA (Sigma-Aldrich, Cat# P8139) and monitored with fluorescence intensity of a membrane-impermeable DNA-binding dye SYTOX™ Green (Invitrogen, Cat# S7020). NETotic index was used to indicate the extent of DNA release. Briefly, cells were co-incubated with PMA and TTC (0, 5, 10, 20, 40, 80 μg.mL−1) for 4 h in the presence of SYTOX Green (1 μmol.L−1). As a negative control, DNase I (Thermo Scientific, Cat# 90083) was added to degrade the DNA releasing to make NETs ineffective, solidifying the evidence of NETosis induced by PMA. The readout from the cells incubated with nucleic acid stain and solvent DMSO served as control. The fluorescence intensity was measured by Enspire™ Multilabel Plate Reader (PerkinElmer, Ex/Em: 504/523 nm). To calculate NETotic index, fluorescence readout obtained from cells lysed with 0.5% (v/v) Triton X-100 was considered as 100% DNA release, and the index was calculated as the percentage of total values. NETotic index (%) = (RFU of vehicle control/experimental wells) × 100/RFU of Triton X-100 wells. 35
Fluorescence microscopic analysis of NETosis
The structure of NETosis was confirmed by imaging extracellular DNA stained by SYTOX Green. The histological colocalization of NE, MPO, and DNA was analyzed by immunofluorescence. Neutrophils were seeded at 1 × 106 cells per well on glass coverslips in 24-well plates and co-treated with or without 200 ng.mL−1 of PMA and 40 μg.mL−1 of TTC for 4 h. Subsequently, the cells were carefully washed with phosphate-buffered saline (PBS) and then fixed with pre-cooled acetone. For DNA staining, cells were incubated with 1 μmol.L−1 of SYTOX Green and coverslips were mounted onto glass slides with Prolong Gold mounting agent (Molecular Probes, Cat# P36930). Images were captured using the Olympus Fluorescence Microscope (Upright BX61) under 40× lens (400-fold magnification). For multiplex immunostaining assay, cells were permeabilized in 0.1% Triton X-100 and incubated with 1% bovine serum albumin (BSA)/PBS to block non-specific antibody binding, followed by incubation with the antibody against MPO (diluted 1:100, Abcam, Cat# ab45977) overnight at 4°C. After washing with PBS, cells were incubated with goat anti-rabbit IgG H&L Alexa Fluor-594 (diluted 1:200, Cat# ab150080) for 2 h at room temperature. The samples were further incubated with antibody against NE (diluted 1:100, Santa Cruz, Cat# sc-55549) overnight at 4°C. After washing with PBS, cells were incubated with goat anti-Mouse IgG H&L Alexa Fluor-488 (diluted 1:200, Cat# ab150113) for 2 h at room temperature. Nuclei were visualized with Hoechst 33342 (Invitrogen, Cat# H1399). After mounting, cells were visualized using Olympus Fluorescence Microscope (Upright BX61) under 100× oil immersion lens (1000-fold magnification).
Measurement of ROS
Measurement of intracellular ROS was performed using the peroxide-sensitive fluorescent probe 2′,7′-dichlorofluorescin diacetate (DCF-DA, Sigma-Aldrich, Cat# D6883). Neutrophils were plated at a density of 1 × 106 cells/well in six-well plates and incubated with 10–40 μg.mL−1 of TTC and PMA (200 ng.mL−1) for 1 h. Subsequently, the cells were collected and washed once with ice-cold PBS. Cells were collected into tubes and centrifuged at 1000 r/min for 5 min to obtain a cell pellet. After resuspension in a solution of phenol-free RPMI-1640 medium containing 20 μmol.L−1 of DCF-DA, neutrophils were incubated for 15 min at 37°C in the dark. DCF fluorescence was measured immediately with BD FACSCalibur flow cytometer using an FL1 detector having a bandpass filter with specifications of 530 ± 15 nm. In each analysis, 10,000 events were recorded. Data were analyzed using FACSDiva Version 6.1.3.
NOX activity assay
The NOX activity was performed with lucigenin (bis-N-methylacridinium nitrate) enhanced chemiluminescence method in the presence of its substrate NADPH. Lucigenin is a luminescence-generating reagent that interacts with NOX-derived superoxide anion and the emitted luminescence can be quantitatively analyzed by a luminometer. 36 Specificity for non-NOX-mediated superoxide production was confirmed by adding a pan-NOX inhibitor diphenylene iodonium (DPI). In short, aliquots of neutrophils at density of 3 × 104 cells/mL cultured in 24-well plates were incubated with 40 μg.mL−1 of TTC and PMA (200 ng.mL−1) in the presence or absence of DPI (Sigma-Aldrich, Cat# D2926) for 1 h. Subsequently, the cells were collected and washed once with ice-cold PBS. Cell pellets were then resuspended in 100 μL of ice-cold Krebs-HEPES buffer with 20 μmol·L−1 lucigenin (Sigma-Aldrich, Cat# M8010). 100 μL of cell suspension was transferred to 96-well white plates with solid bottom (Costar, Cat# 3912) and 20 μL of NADPH (Sigma-Aldrich, Cat# N7505) balanced with Krebs-HEPES buffer were sequentially added to the well at final concentrations of 500 μmol.L−1. Immediately, the luminescence was dynamically recorded at 1 min intervals for a total of 10 min of reaction time by Enspire Multilabel Plate Reader (PerkinElmer). The moment of adding NADPH into samples was defined as 0 min, and we accurately measured luminescence once the reaction time up to 1 min. After that, the plate reader automatically detected the luminescence at 1 min increments until 10 min. The slope with 10 min for the trend line was defined as the relative NOX activity, expressed as luminescence unit (RLU)/min.
Western blot analysis
Freshly isolated neutrophils were seeded at 1 × 107 cells per well in six-well plates and incubated with or without 10, 40 μg.mL−1 of TTC in the presence of PMA for 1 h. Subsequently, cells were centrifuged and washed once with ice-cold PBS and lysed with radioimmunoprecipitation assay (RIPA) buffer containing protease and phosphatase inhibitors. Equal amounts of cell lysates (25 μg) were resolved by 10% SDS-PAGE and transferred onto polyvinylidene difluoride (PVDF) membranes. Membranes were incubated with monoclonal antibodies against phospho-Thr154-p40phox (CST, Cat# 4311) at dilution of 1:1000, NCF-4 (Abcam, Cat# ab76158) at dilution of 1:1000, and GAPDH (Abcam, Cat# ab8245) at dilution of 1:10,000. Membranes were incubated with horseradish peroxidase (HRP)-conjugated anti-rabbit or mouse IgGs (Proteintech, Cat# SA00001-2 and SA00001-1) at dilution of 1:10,000 followed by enhanced chemiluminescence (Millipore, Cat# WBKLS0500) and visualized with a ChemiDoc XRS system (Bio-Rad).
Statistical analysis
Data are shown as x̅ ± SD and evaluated using either two-tailed Student’s t-test or one-way analysis of variance (ANOVA) followed by post hoc Dunnett’s test. Statistical analyses were performed using IBM SPSS 20.0. Scientific graphing was performed using GraphPad Prism 5.0. Differences where P < 0.05 were considered statistically significant.
Results
TTC has no predominant cytotoxic effect on neutrophils
Human neutrophil has a short life span and can only survive for 12–24 h in vitro, 37 even trivial chemical stimuli might be enough to induce significant biological effect on the cell function of neutrophil. Thus, it was critical to examine the potential neutrophil cytotoxicity of TTC. To this end, we treated freshly isolated neutrophils with various doses of TTC (0, 5, 10, 20, 40, 80 μg.mL−1) for 4 and 10 h, respectively, and cytotoxic effect of TTC on neutrophils was detected by LDH leakage assay. As a result, TTC (5–80 μg.mL−1) failed to show significant neutrophil cytotoxicity after 4 h treatment, whereas the maximum dose of TTC (80 μg.mL−1) displayed some cytotoxic effect upon 10 h exposure (P < 0.05, Figure 1). Taken together, TTC (5–40 μg.mL−1) shows a very low level of cytotoxic effect on human neutrophils in vitro.

The cytotoxicity of TTC on neutrophils. The fresh neutrophils were incubated with various concentrations of TTC (0, 5, 10, 20, 40, 80 μg.mL−1) for 4 and 10 h, and the cytotoxic effect of TTC on neutrophils was determined by LDH release assay. Data shown are expressed as mean values ± SD and represent the results of three independent experiments performed in triplicate wells. Multiple differences between control (0 μg.mL−1) and various doses of TTC (5, 10, 20, 40, 80 μg.mL−1) were analyzed by one-way ANOVA and post hoc Dunnett’s test. *, P < 0.05 between control and 80 μg.mL−1 of TTC treatment at 10 h.
TTC inhibits PMA-induced NETosis
To examine the potential effect of TTC on NETosis, we established PMA-induced NET formation in the presence or absence of TTC. To quantify the NET formation, we used SYTOX Green, a cell-impermeable fluorescent DNA dye, to measure the extent of extracellular DNA released from NETotic cells but not from viable, membrane-intact cells. Initially, TTC (5–80 μg.mL−1) treatment alone could not induce NETotic activity in resting neutrophils during 10 h of exposure time (Supplementary Figure 1). Next, we observed that 200 ng.mL−1 of PMA after 4 h stimulus substantially triggered up-regulation of NETotic index (P < 0.01), which could be successfully reversed by DNase I (P < 0.01). In addition, TTC (5, 10, 20, 40, 80 μg.mL−1) exerted dose-dependent inhibitory effect on PMA-induced NETosis (Figure 2(a)). To solidify this result, we further visualized the changes of neutrophil morphology after TTC treatment during PMA-induced NETosis. As a result, PMA caused typical delobulation and swelling of neutrophil nuclei with highly interlaced chromatin DNA. In consistent with our previous findings (Figure 2(b)), extracellular DNA scaffolds were suppressed by co-treatment of TTC (40 μg.mL−1). However, TTC failed to completely reverse the process of nuclear delobulation and minimize the swollen nucleus compared with normal neutrophils. Altogether, TTC exerts inhibitory potential on PMA-induced NETosis.

The effect of TTC on PMA-induced NET formation. (a) The neutrophils were activated by 200 ng.mL−1 of PMA and incubated with or without various concentrations of TTC (5, 10, 20, 40, 80 μg.mL−1) for 4 h. NETotic index was used to indicate the extent of DNA release during NETosis. Data shown are expressed as mean values ± SD and represent the results of three independent experiments performed in triplicate wells. Difference between control and PMA (200 ng.mL−1) was analyzed by Student’s t-test. ##, P < 0.01 between control and PMA. Multiple differences between PMA (200 ng.mL−1) and PMA + DNase I, PMA + TTC (5, 10, 20, 40, 80 μg.mL−1) were analyzed by one-way ANOVA and post hoc Dunnett’s test. *, P < 0.05 or **, P < 0.01 between PMA and PMA + DNase I, PMA + TTC. (b) The structures of NETs were observed by fluorescent microscopy using SYTOX™ Green staining in formalin-fixed neutrophils at 400× magnification, scale bar represents 100 μm.
TTC inhibits NET-associated DNA-NE and DNA-MPO complexes
PMA represents a potent stimulus of formation of NETs, which incorporates the translocation of granular proteins primarily composed of neutrophil proteases and peroxidases, including MPO and NE. To further verify whether TTC served as an antagonist of NETosis, we used multicolor immunofluorescence staining to locate MPO and NE in neutrophils. As we found in resting neutrophils, NE and MPO were distributed in the cytoplasm. However, once neutrophils were activated by PMA, NE and MPO began to translocate from the cytoplasm to the nucleus, release outside of the cell, and conjugate with chromatin DNA. Consistent with our expectations, TTC did not only attenuated NET-like texture, but also disrupted the formation of NET-associated DNA-MPO and DNA-NE complexes, which indicated a compromised degradation of chromatin DNA (Figure 3(a)). Neutrophils with released DNA backbone are morphologically recognized as NETs. However, a better way to detect true NETs is to look for DNA, histones, and active enzymes secreted by neutrophils all in close proximity. 38 Thus, we quantified the percentage of NET releasing cells accompanied with positive staining of DNA conjugated NE and MPO. As a result, PMA induced around 58.6% of NETosis rate while co-treatment of TTC could decrease the rate to 25.8% (Figure 3(b)). In sum, TTC can block the initiation of NETosis characterized with formation of DNA and granular proteins complexes.

The effect of TTC on PMA-induced formation of DNA-NE and DNA-MPO complexes. (a) NETs were further confirmed by a multicolor fluorescence immunostaining of DNA-NE and DNA-MPO complexes at 1000× magnification, scale bar represents 50 μm. (b) Counting of percentage of NETotic neutrophils with NET-like DNA structure accompanied with positive staining of NE and MPO. Data shown are expressed as mean values ± SD and represent the results of three independent slides with randomly selected three visual fields. Difference between control and PMA was analyzed by Student’s t-test. ##, P < 0.01 between control and PMA. Difference between PMA and PMA + TTC was analyzed by Student’s t-test. **, P < 0.01 between PMA and PMA + TTC.
TTC inhibits PMA-induced ROS generation
ROS production has been implicated PMA-induced NETosis; we therefore asked whether TTC could block PMA-induced oxidative damage. ROS levels were monitored by flow cytometry using DCF-DA, which could be oxidized to the fluorescent compound DCF and fluorescence intensity is proportional to the amount of ROS produced. As shown in Figure 4(a), it was found that PMA (200 ng.mL−1) significantly simulated an oxidative stress in neutrophils with 1 h (P < 0.05). Meanwhile, TTC (10, 20, and 40 μg.mL−1) exerted a dose-dependent inhibitory effect on PMA-induced ROS generation (Figure 4(b)), and higher concentration of TTC at 40 μg.mL−1 could achieve statistical significance (P < 0.05). Thus, TTC is able to downregulate the ROS levels in PMA-activated neutrophils.
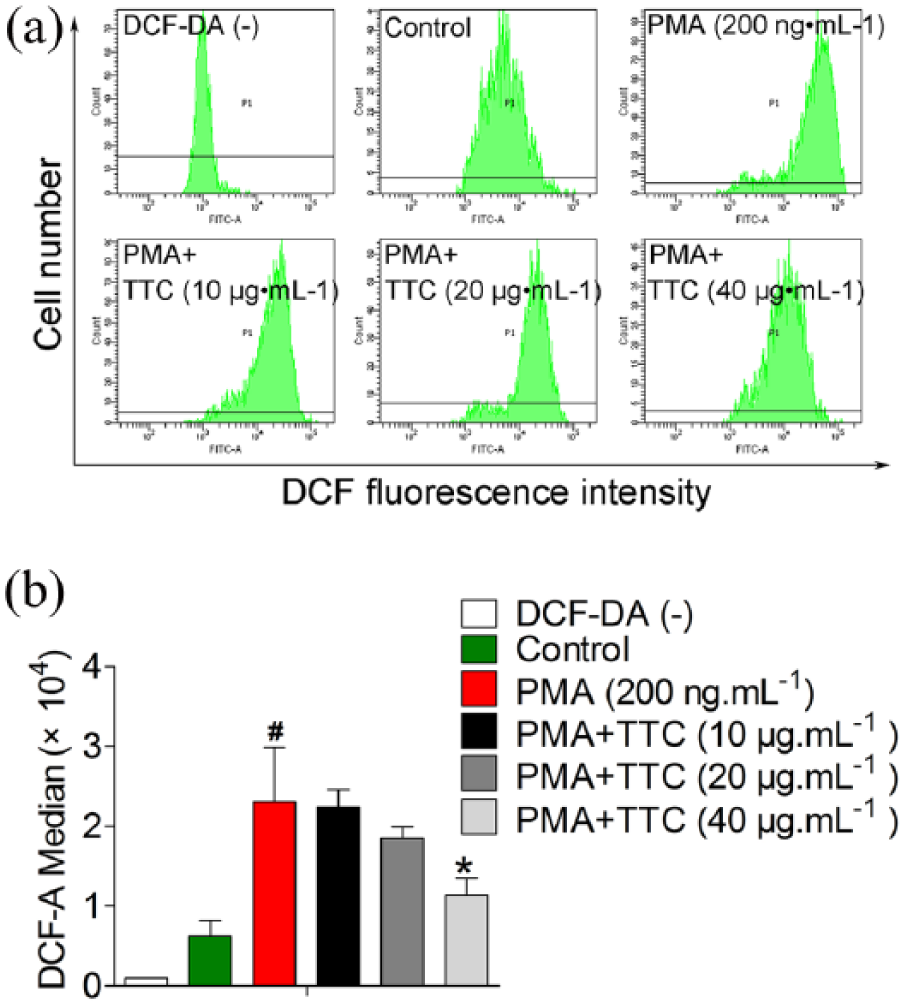
The effect of TTC on PMA-induced ROS generation. (a) Representative flow cytometry histograms of ROS levels in neutrophils, which were stained with or without DCF-DA (20 μmol.L−1, 15 min at 37°C) and treated with TTC (10, 20, 40 μg.mL−1) in the presence of PMA (200 ng.mL−1) for 1 h. (b) Quantified ROS levels indicated by fluorescence intensity of DCF in neutrophils. Data shown are expressed as mean values ± SD and represent the results of three independent experiments performed in triplicate wells. Difference between control and PMA was analyzed by Student’s t-test. #, P < 0.05 between control and PMA. Multiple differences between PMA and PMA + TTC were further analyzed by one-way ANOVA and post hoc Dunnett’s test. *, P < 0.05 between PMA and PMA + TTC (40 μg.mL−1).
TTC inhibits PMA-induced NOX activity
PMA has been extensively used as an agonist to activate NOX-dependent NETosis. To elucidate the underlying mechanisms of TTC-mediated inhibition of PMA-induced NETosis and ROS generation, we next aimed to examine the effect of TTC on PMA-induced NOX activity, which was detected by NOX-derived superoxide anions using lucigenin-amplified chemiluminescence assay. As shown in Figure 5(a) and (b), we found the activity of NOX was almost preserved in quiescent neutrophils but obviously excited upon 1 h of PMA stimulus (P < 0.01). Moreover, inhibition of NOX by DPI (10 μmol.L−1) completely abolished superoxide production. TTC (40 μg.mL−1) revealed a significant inhibitory effect on NOX activity and DPI could override the effect of TTC, suggesting that TTC specifically targeted for NOX activity. Therefore, our data demonstrate that inactivation of NOX is involved in TTC-mediated inhibitory effect on PMA-induced NETosis.

The effect of TTC on PMA-induced NOX activity. (a) NOX activity was detected using lucigenin chemiluminescence assay. The chemiluminescence readout was recorded every 10 min for a total of 10 min. (b) The NOX activity was expressed as luminescence unit (RLU)/min in 10 min. Data shown are expressed as mean values ± SD and represent the results of three independent experiments performed in triplicate wells. Multiple differences between control and PMA, DPI, TTC were analyzed by one-way ANOVA and post hoc Dunnett’s test. #, P < 0.05 between control and DPI. ##, P < 0.01 between control and PMA. Multiple differences between PMA and PMA + TTC, PMA + TTC + DPI were further analyzed by one-way ANOVA and post hoc Dunnett’s test. **, P < 0.01 between PMA and PMA + TTC, PMA + TTC + PDI.
TTC inhibits PMA-induced phosphorylation of p40phox subunit
To substantiate the role of reduced NOX activity in TTC-mediated inhibitory effect on PMA-induced NETosis, we checked whether phosphorylation status of NOX was required for TTC inhibiting NOX activity. The phosphorylation of cytosolic NOX subunit p40phox on Thr154 residue was thought to be a key event for NOX activation. 39 In agreement with our hypothesis, PMA strikingly increased the levels of phosphorylated p40phox, and treatment with TTC (10, 40 μg.mL−1) dose-dependently prevented this process (Figure 6). Collectively, TTC can interrupt NOX enzymatic activity through NOX signaling pathway.

The effect of TTC on PMA-induced phosphorylation of NOX subunit p40phox. The effect of TTC on PMA-induced phosphorylation of NOX subunit p40phox was analyzed by western blot. Protein levels were quantified with NIH ImageJ. Graphs show mean ± SD from at least three independent blots. Difference between control and PMA was analyzed by Student’s t-test. ##, P < 0.01 between control and PMA. Multiple differences between PMA and PMA + TTC were further analyzed by one-way ANOVA and post hoc Dunnett’s test. **, P < 0.01 between PMA and PMA + TTC (40 μg.mL−1).
Discussion
Given that neutrophils have the shortest life span among all leukocytes within few hours in vitro, 40 even a slight effect on cell viability upon chemical stimuli may have a profound impact on NET biology of neutrophils. Besides, neutrophils can die by various forms of cell death, and these forms of cell death appear to be transformable. 41 It is probably that there are specific subsets of neutrophils died by making NETs in culture microenvironments concurrent with other types of cell death. Therefore, we should carefully conclude that TTC exclusively targets NET biology of neutrophils. Extracellular DNA release during NET formation was tracked by impermeable DNA dye, an enhancement of fluorescence intensity might be overwhelmed by increased permeability of dye access to the interior DNA of dying neutrophils. To rule out the potential effect of TTC on the membrane integrity and viability of neutrophils, we therefore conducted LDH assay. LDH is a suggested biomarker that leaks from the cell upon cell plasma and has been widely used in in vitro toxicology studies. Our results showed that TTC had no detrimental effect on quiescent neutrophils and hardly induced NET formation under physiological conditions (Figure 1 and Supplementary Figure 1). This is prerequisite for TTC against NETosis in the setting of inflammatory and autoimmune disorders.
A variety of stimuli have been reported to induce NETosis in response to chemical and biological conditions. Moreover, NETosis occurs through several signaling mechanisms. 42 Certain immune factors and pro-inflammatory cytokines can drive NET formation, such as colony stimulating factor (G-CSF), interleukin-8 (IL-8), and bacterial lipopolysaccharide (LPS), but their ability to reliably reproduce NET formation as stable NET inducers is still controversial. 43 In comparison, the most commonly employed tool to model NETosis is chemical agent PMA. Moreover, the mechanism behind PMA-induced NETosis has been extensively studied. 44 Here, we used PMA to model NETosis in therapeutic scenarios targeting suicidal NETosis; however, it has several limits. First, it is not physiologically relevant. Second, for starters, many of the early experiments with NETs were done with high concentrations of PMA at levels that cells within the body are unlikely to ever encounter. 38 Finally, “vital NETosis” in early time points might be compromised by catastrophic explosion of PMA-induced suicidal NETosis. In our future directions, it is important to study the effect of TTC on physiological NETosis inducers. These findings will provide better understanding of C. orbiculatus as to how it works against NETosis upon different stimuli.
A complex degree of mechanisms is responsible for NET formation in different circumstances. Identification of NETs has proven challenging because there is no rigorous classification scheme that defines NETs biochemically and not morphologically. 38 So far, the molecular processes of PMA-induced NET formation have been fully characterized. It is well known that PMA activates PKC, which further phosphorylates several subunits of NOX, resulting in the assembly of NOX subunits into a functional complex to produce ROS. 29 The NOX family is a major source of cellular ROS production, and the isoform NOX2 is expressed principally in neutrophils. 45 NOX activity is controlled by a complex regulatory system and needs to be determined in entire complement of cellular machinery. Current methods used to measure NOX catalytic activity are based on indirectly detecting NOX-derived ROS that reacts with chemiluminescent probes such as lucigenin. In addition, lucigenin specifically reacts with superoxide, but not MPO-derived ROS. 46 Notably, a pan-NOX inhibitor DPI is used to rule out other sources of superoxide, thereby ensuring specificity for determining NOX activity. In the present study, we did not only measure the potential of TTC on PMA-induced ROS levels and NOX activity, but also tested the phosphorylation of NOX subunits. Thus, it helped to discriminate that TTC acted to be direct inhibitor of NOX enzymatic activity but not ROS scavenger that would reduce chemiluminescent signal. Therefore, we believe that TTC inhibits PMA-induced NETosis in NOX activity-dependent manner.
PMA and TTC are both natural occurred terpenoids, and PMA belongs to phorbol ester terpenoids while TTC consists of non-phorbol ester terpenoids. However, they might have entirely different mechanism of action on PKC biology. It is well known that PMA binds to PKC in a similar way to its natural ligand, diacylglycerol (DAG), and activate the kinase. In contrast, TTC might inhibit both the upstream and downstream mechanisms of PKC. As a representative component of TTC, celastrol has been reported to inhibit PKC maturation by disrupting the interaction between Hsp90 and Cdc37. 47 Moreover, celastrol has been found as a potent inhibitor of several members of NOX enzymes. 48 Therefore, TTC might inhibit the stabilization of PKC and directly impede the NOX activity independent of PKC activation. However, NOX activity is also involved in phagocytosis and other functions of neutrophils, and diverse stimuli engage different NET pathways. 42 NOX-independent mechanisms are also important for NET formation. For instance, calcium ionophores are commonly used to model NOX-independent NETosis. 35 Therefore, we have to confirm the stimuli-specificity shown by TTC in future.
In conclusion, our data indicate the efficacy and safety profile of TTC, a multi-terpenoid-based remedy against PMA-induced NETosis via NOX signaling pathway, which provides preliminary evidence for C. orbiculatus as anti-inflammatory medications.
Supplemental Material
Supplementary_Figure.1 – Supplemental material for The total terpenoids of Celastrus orbiculatus (TTC) inhibit NOX-dependent formation of PMA-induced neutrophil extracellular traps (NETs)
Supplemental material, Supplementary_Figure.1 for The total terpenoids of Celastrus orbiculatus (TTC) inhibit NOX-dependent formation of PMA-induced neutrophil extracellular traps (NETs) by Li Tao, Min Xu and Yanqing Liu in European Journal of Inflammation
Supplemental Material
Supplementary_Table.1 – Supplemental material for The total terpenoids of Celastrus orbiculatus (TTC) inhibit NOX-dependent formation of PMA-induced neutrophil extracellular traps (NETs)
Supplemental material, Supplementary_Table.1 for The total terpenoids of Celastrus orbiculatus (TTC) inhibit NOX-dependent formation of PMA-induced neutrophil extracellular traps (NETs) by Li Tao, Min Xu and Yanqing Liu in European Journal of Inflammation
Footnotes
Authors’ Note
L.T. and M.X. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Young Scientists Fund of the National Natural Science Foundation of China (no. 81803782), the Young Scientists Fund of Natural Science Foundation of Jiangsu Province (no. BK20170516), China Postdoctoral Science Foundation (no. 2017M611936), Jiangsu Postdoctoral Science Foundation (no. 1701185B), Yangzhou University for Science and Technology Innovation Project (no. 2017CXJ102), to L.T., and National Natural Science Foundation of China (no. 81773944), to Y.L..
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
