Abstract

Keywords
Introduction
Based on global cancer statistics in 2020, liver cancer is one of the most common cancers, reported as the sixth most common of all cancer incidents and the fourth leading cause of cancer-related deaths in the world. 1 Risk factors that can cause or increase the probability of liver cancer incidence include hepatitis B or C virus (HBV or HCV) infections, aflatoxin exposures, obesity, type 2 diabetes mellitus, heavy alcohol drinking, and tobacco smoking. 2 There are an increasing number of experimental mouse models used in liver cancer research to investigate its pathogenesis and assess novel therapeutics. For several decades, the most widely used chemical to induce liver cancer in mice is diethylnitrosamine (DEN), which targets the liver where it is metabolically activated by centrilobular hepatocytes into alkylating agents that can form mutagenic DNA adducts and finally can result in the development of cancer. 3
Due to the limitations and problems of conventional pharmaceutical agents, the nanotechnology has a significant role in the field of medicine and drug delivery, has come to be called “nanomedicine”. 4 Gallium is a metal of atomic number 31 in group IIIA in the periodic table of elements, presents at a concentration of 5–15 mg/kg in the earth’s crust, and obtains during the extraction of aluminum and zinc ores as a byproduct. 5 The chemical features of the Ga3+ ion and its ability to interfere with numerous cellular functions, such as apoptosis, DNA replication, and repair, made this metal very attractive for biomedical scientists, paving the way to several therapeutic applications. 6 The therapeutic properties of gallium led to the development of novel formulations for the treatment of various disorders in humans, including cancer, autoimmune diseases, accelerated bone resorption, and infectious diseases. Numerous studies reported that gallium has several diagnostic and therapeutic applications as it has antineoplastic,7–9 antibacterial,10–12 antifungal, 13 immunosuppressive, and anti-inflammatory activities.14,15 Moreover, radioactive gallium (67Ga citrate) was found to be localized in malignant cells and therefore 67Ga scan used for the detection of malignant tumors in patients. 16
Cetrimonium bromide ([(C16H33)N(CH3)3]Br; cetyl tri-methyl ammonium bromide; hexadecyltrimethylammonium bromide; CTAB) is a quaternary ammonium surfactant used as a topical antiseptic and has a tremendous inhibitory effect on osteosarcoma and therefore may play a variety of roles in cancer treatment. 17
Therefore, the present study was conducted to evaluate the efficiency of the novel compound copper-cetyl tri-methyl ammonium bromide combined with gallium oxide nanoparticle complex (Cu-CTAB + GaO-NPs) to reduce the incidence of hepatocellular carcinoma. The work comprises both in vivo and in vitro studies. The in vitro cytotoxic effect of this compound has been investigated using human hepatocellular carcinoma (HepG-2) cell line. The compounds were further promoted into in vivo studies, using DEN-induced liver tumors in the male rat model, to validate whether the novel complex (Cu-CTAB + GaO-NPs) could be used as a promising strategy for liver cancer therapy.
Methods
As mentioned above, this study was aimed to evaluate the ameliorative effect of Cu-CTAB + GaO-NPs against hepatocellular carcinoma induced by DEN in rats. The next paragraphs will describe chemicals and in vivo and in vitro studies used.
Chemicals
Diethyl nitrosamine, cetyl tri-methyl ammonium bromide, copper dichloride, and gallium oxide were obtained from Sigma Aldrich Chemical Co., St. Louis, Mo., USA. All these chemicals and reagents used were of analytical grade. Assay kits for testing alanine aminotransferase (ALT), aspartate transaminase (AST), total protein, iron concentration, and α-l–fucosidase were supplied by Biodiagnostics (Egypt). ELISA Kits for alpha-fetoprotein (AFP), transforming growth factor-β1 (TGF-β1), and caspase 3 (casp-3) were obtained from Cusabio Biotech, China; Elabscience, China; and MyBioSource, USA; respectively.
Experimental animals
Adult (20 weeks old) male Wister rats weighing about 140 ± 20 gm were obtained from the Nile Pharmaceutical Co., Cairo, Egypt. The animals were kept under standard laboratory conditions of light/dark cycle (12/12 h), a temperature of 25 ± 2°C and humidity of 60 ± 5%, and were housed in cages with free access to food and drinking water. Animals were fed on standard pellet diet, and sterilized water was provided ad libitum. All animal procedures were carried out in accordance with the Ethics Committee of The National Center for Radiation Research and Technology in accordance with the “Guide for the Care and Use of Laboratory Animals” published by the US National Institute of Health (NIH publication, No. 85-23, 1996).
Methods
Chemical studies
Synthesis of cetyl tri-methyl ammonium bromide copper complex
Cetyl tri-methyl ammonium bromide (CTAB) was synthesized according to Badawi et al. 18 Firstly, CTAB was prepared by refluxing 2 M amounts of the cationic surfactant (CTAB) and 1 mole of copper dichloride (CuCl2) in ethanol for 6 h. Then, the resultant crystals were washed twice with petroleum ether and recrystallized from methanol, and finally dried under vacuum at 50°C for 4 h.
Synthesis of gallium oxide nanoparticles
Nanoparticles of gallium oxide (GaO-NPs) were prepared by grinding gallium oxide powder to the nano-sized particles using ball mill at 200 r/min for 8 h and then suspended by mixing 0.25 gm of gallium oxide nanoparticles with 100 mL of distilled water using ultra-sonication for 30 min.
Synthesis of Cu-CTAB + GaO-NP complex
The newly synthesized Cu-CTAB + GaO-NP complex was prepared by suspending 0.25 gm of gallium oxide nanoparticles in 3 gm of Cu-CTAB complex dissolved in 100 mL distilled water using ultra-sonication for 30 min and then the characterization of complex was detected by using transmission electron microscopy (TEM) to determine size and morphology.
Characterization of Cu-CTAB + GaO-NP complex
To ensure that the new synthesized Cu-CTAB + GaO-NP complex was in nanoscale and to study its physicochemical properties, a panel of analyses was conducted at the Nanotechnology Unit, Egyptian Petroleum Institute (Egypt), for nano-verification of the novel Cu-CTAB + GaO-NP complex, which are as follows:
Transmission electron microscopy (TEM)
A sample for transmission electron microscopy (TEM) of the synthesized Cu-CTAB + GaO-NP was prepared by placing a drop of it on carbon-coated copper grids and allowing water to evaporate. The shape and size of complex were determined from TEM micrographs. The software (Advanced Microscopy Techniques, Danvers, MA) for the digital TEM camera was calibrated for size measurements of the nanoparticles according to Gurunathan et al., 19 while the spectrum absorbance of Cu-CTAB + GaO-NPs was analyzed by using ultraviolet-visible (UV-Vis) spectroscopy (JASCO UV-Vis spectrophotometer model V-750) in a measurement range of 200–600 nm. Then, a sample of Cu-CTAB + GaO-NPs was analyzed for size distribution throughout dynamic light scattering (DLS) by photon correlation spectroscopy using a Zetasizer ZS instrument (Malvern Instruments Ltd., Malvern, UK).
Evaluation of therapeutic antitumor efficacy of biosynthesized Cu-CTAB + GaO-NP complex
The present study was designed to comprise a series of in vitro and in vivo investigations as following:
In vitro study
The antitumor effects of Cu-CTAB, GaO-NPs, and combination of them were investigated on the viability of the human liver cancer cell line (HepG-2) using cell viability assay by crystal violet staining method.
In vivo study
Determination of the Median Lethal Dose (LD50)
To evaluate the LD50 of the synthesized compounds (Cu-CTAB combined with GaO-NPs and GaO-NPs alone), ascending oral doses of the novel tested complex (Cu-CTAB + GaO-NPs) and gallium oxide nanoparticles (GaO-NPs) were administered into male Wister rats (6 rats/group) weighing about 120–150 gm. Mortality incidence was recorded 24 h after administration. The optimum selected dose for evaluating the in vivo antitumor activity of the novel synthesized composite and gallium oxide nanoparticles alone was calculated approximately as (1/10) of their LD50 values, whereas LD50 of Cu-CTAB was reviewed in previous literature. 18
Biochemical studies
Experimental Design:
Eighty Wister rats weighing 120–150 gm were equally divided randomly into the following eight groups: • Group 1 (control): Untreated control group of healthy rats. • Group 2 (Cu-CTAB): Rats received 6.5 mg/kg body weight of Cu-CTAB orally five times per week for 4 weeks, according to Badawi et al.
18
• Group 3 (GaO-NPs): Rats were treated orally with GaO-NPs (3.3 mg/kg b. w.) five times per week for 4 weeks by intragastric gavage, according to Abdalla et al.
20
• Group 4 (Cu-CTAB + GaO-NPs): Rats received 16.3 mg/kg b. w. of complex orally (five times per week) for 4 weeks. • Group 5 (DEN): Rats received 20 mg/kg b. w. of DEN orally (five times per week for 6 weeks) and left alive for 4 weeks, according to Darwish and EL-Boghdady.
21
• Group 6 (DEN + Cu-CTAB): Rats received DEN as in group 5 and then were administrated Cu-CTAB orally five times per week for 4 weeks as in group 2. • Group 7 (DEN + GaO-NPs): Rats received DEN as in group 5 and then were treated with GaO-NPs orally five times per week for 4 weeks as in group 3. • Group 8 (DEN + complex): After rats received DEN as in group 5, they were treated with tested complex orally five times per week for 4 weeks.
Twenty-four hours after the last treatment, all rats were anesthetized with the recommended anesthetic dose of urethane (1.15 g/kg) 22 through intraperitoneal injection and then scarified and blood samples were obtained via heart puncture and collected in plain vacutainer tubes. Then samples were left to coagulate and serum was obtained after centrifuged at 4,000 rpm for 10 min for biochemical analysis. Liver was excised from animals in all groups. Part of the collected livers was homogenized (10% w/v) in phosphate-buffered saline (0.02 M sodium phosphate buffer with 0.15 M sodium chloride, pH 7.4) using glass homogenizer producing homogenates which were used for biochemical and molecular analyses, and the other part was quickly rinsed in saline solution and then kept in 10% formalin for histopathological examination.
Biochemical investigations
The activities of serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) and the concentrations of iron and total protein in the serum were measured calorimetrically using commercial assay kits (Biodiagnostic, Egypt) according to the manufacturer’s instructions.
The concentrations of alpha-fetoprotein (AFP) and transforming growth factor-β1 (TGF-β1) in serum were measured by using commercial sandwich ELISA Kit (Cusabio Biotech, China and Elabscience, China, respectively) according to the manufacturer’s instructions. While serum α-L–fucosidase activity was measured calorimetrically using commercial assay kits (Biodiagnostic, Egypt) according to the manufacturer’s instructions.
Lipid peroxidation was measured in the plasma and liver tissue homogenate according to the method of Yoshioka et al., 23 and total thiobarbituric acid reactive substances (TBARS) were expressed as malondialdehyde (MDA). In brief, liver homogenates (10% w/v) were mixed with 10% TCA (ice cold), centrifuged at 800 g for 10 min, then the supernatant was mixed with thiobarbituric acid (TBA) to develop the color, boiled for 10 min at 100°C, cooled at room temperature, and finally optical density was read at 535 nm. Also, the activity of superoxide dismutase (SOD) in the red blood corpuscles and liver tissue and the activity of arginase in the liver tissue were determined calorimetrically using commercial assay kits (BioVision, USA) according to the manufacturer’s instructions.
Detection of caspase-3 by quantitative real time PCR (qRT-PCR)
Primer sequences for quantitative RT-PCR analysis.

Histopathological examination
Following rat sacrificing, the liver was rapidly dissected, excised, and rinsed in saline solution and then fixed in 10% formalin solution for at least 3 days at 4°C. Following fixation, the specimens were dehydrated in ascending series of alcohol (70% methyl, 70% ethyl, and absolute (95–100%) ethyl), cleared in xylene and then embedded in paraffin at 60°C. Sections of 5 microns thickness were cut and stained with hematoxylin and eosin and examined by using a light microscope.
Statistical analysis
Statistical analysis was performed using IBM SPSS software (version 23.0; IBM Corp., Armonk, NY, USA), and data were presented as means ± SE. Data were analyzed with one-way analysis of variance (ANOVA) followed by post hoc test (LSD) for multiple comparisons. The criterion for significance was p < 0.05.
Results
Characterization of gallium oxide nanoparticles combined with tetrahalocuprate complex
The prepared gallium oxide nanoparticles combined with tetrahalocuprate complex were characterized by using transmission electron microscopy (TEM) to determine their size and morphology. The images obtained from TEM showed that the sample of complex appeared as regular spheres and distributed homogeneously (Figure 1(a)). The formation of Cu-CTAB + GaO-NPs was confirmed by UV–visible spectroscopy with a sharp peak of maximum absorbance at 265 nm, indicating the synthesis of Cu-CTAB + GaO-NPs (Figure 1(b)). Finally, DLS measurement of the hydrodynamic effective diameter of the synthesized Cu-CTAB + GaO-NPs showed that the zeta average diameter was measured to be 127.2 nm with 0.957 poly dispersity index (PDI) indicating the homogeneity and uniform dispersion of the synthesized BAs-ZnNPs (Figure 1(c)). Characterization of Cu-CTAB + GaO-NP complex. (a): Transmission electron microscopic image of Cu-CTAB + GaO-NPs; (b): UV absorbance of Cu-CTAB + GaO-NPs; (c): dynamic light scattering of Cu-CTAB + GaO-NP complex.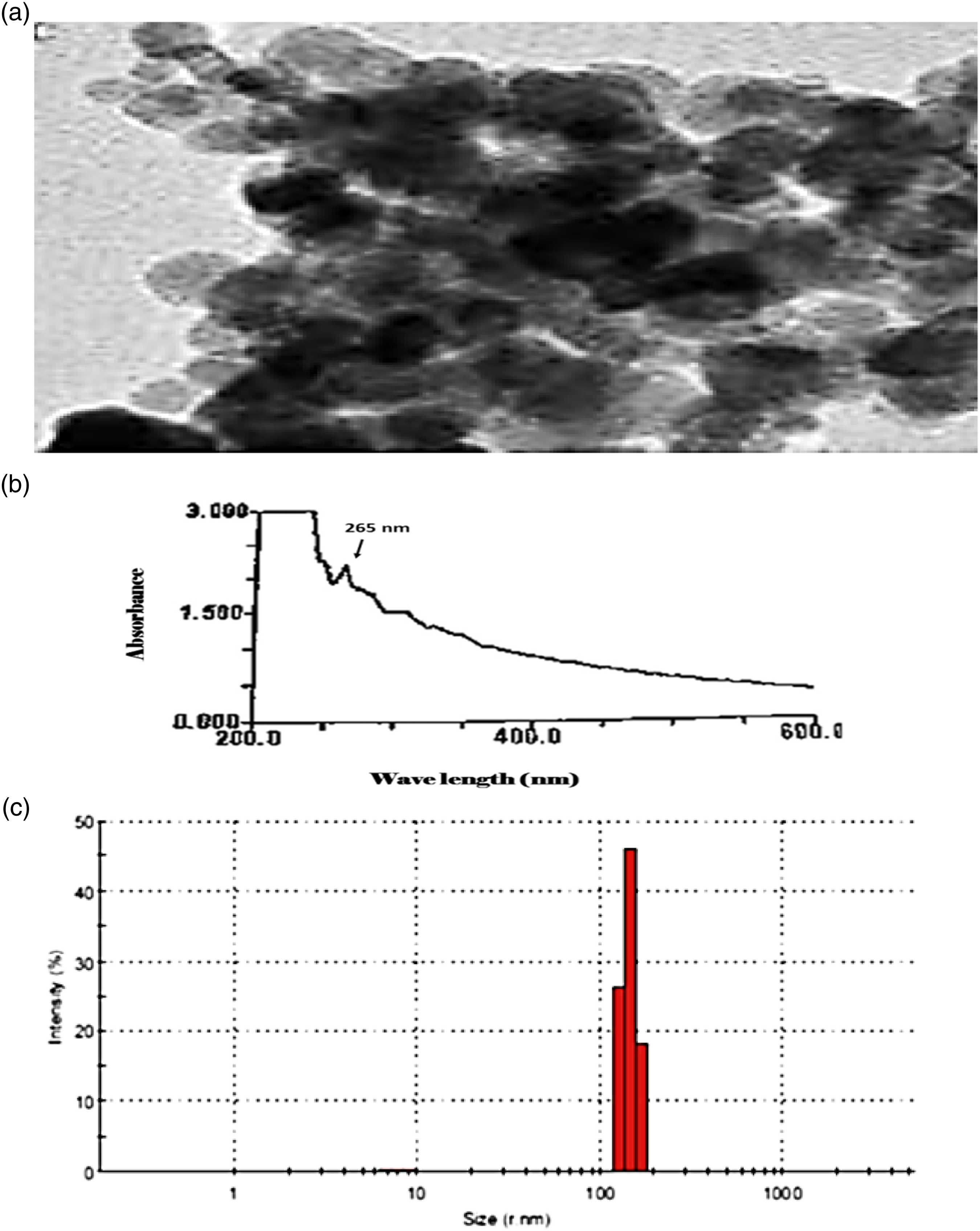
In vitro study (cell viability tests)
The in vitro anticancer cytotoxic activities of complex (Cu-CTAB-GaO-NPs) and gallium oxide nanoparticles (GaO-NPs) alone were investigated on hepatocellular carcinoma human cell line (HepG-2) using crystal violet dye. The cytotoxic activity was expressed as reduction of cell viability relative to control. The 50% growth inhibitory concentration (IC50) values were indicated in Figure 2 and Supplementary Tables S1, S3. The novel complex exhibited the strongest in vitro anticancer activity against HepG-2, as it recorded the lowest value (0.2 μg/ml) comparable to that of Cu-CTAB (11.07 μg/ml)
18
and gallium oxide nanoparticles alone (360 μg/ml) against this hepatocellular carcinoma human cell line (HepG-2). Cell viability test. (a): Antitumor activity of Cu-CTAB combined with GaO-NPs against HepG-2 cell line; (b): antitumor activity of gallium oxide nanoparticles against HepG-2 cell line.
In vivo studies
Determination of median lethal dose (LD50) value
The LD50 of Cu-CTAB + GaO-NPs complex, Cu-CTAB, and GaO-NPs alone were 163 mg/kg b. w., 65 mg/kg b. w., and 33 mg/kg b. w., respectively.
Biochemical markers
Results of all estimated biochemical parameters are reported in Supplementary Table S3. Our results revealed that the activities of liver enzymes (ALT and AST) were significantly increased, but serum total protein concentration was significantly decreased in DEN-intoxicated rats when compared to control ones, while after administration of Cu-CTAB, GaO-NPs, or Cu-CTAB + GaO-NPs complex, the activities of liver enzymes (ALT and AST) were significantly decreased while serum total protein concentration was significantly increased in DEN + Cu-CTAB, DEN + GaO-NP, or DEN + Cu-CTAB + GaO-NP groups when compared to the DEN group, as shown in Figure 3. Liver function tests. (a): Serum ALT activity (U/L) in different groups; (b): serum AST activity (U/L) in different groups; (c): serum total protein concentration (g/dL) in different groups. Data are expressed as mean ± SE (n = 10). a: Significant compared to the control, b: significant compared to the Cu-CTAB group, c: significant compared to the GaO-NP group, d: significant compared to the Cu-CTAB + GaO-NP group, e: significant compared to the DEN group, (p < 0.05).
Also, results of the current study showed that the tumor markers such as serum AFP concentration and α-L–fucosidase activity were significantly decreased after the treatment with Cu-CTAB, GaO-NPs, or Cu-CTAB + GaO-NP complex in DEN + Cu-CTAB, DEN + GaO-NP, and DEN + Cu-CTAB + GaO-NP groups when compared to the DEN group, as shown in Figures 4(a),(c). Moreover, the concentration of TGF-β1 in serum was significantly decreased while serum iron concentration was significantly increased in DEN + Cu-CTAB, DEN + GaO-NP, or DEN + Cu-CTAB + GaO-NP groups when compared to the DEN group, as shown in Figures 4(b),(d). Serum tumor and inflammatory markers. (a): Serum AFP concentration (ng/mL) in different groups; (b): serum TGF-β1 concentration (pg/mL) in different groups; (c): serum α-L–fucosidase activity (U/L) in different groups; (d): serum iron concentration (μmol/L) in different groups. Data are expressed as mean ± SE (n = 10). a: Significant compared to the control, b: significant compared to the Cu-CTAB group, c: significant compared to the GaO-NP group, d: significant compared to the Cu-CTAB + GaO-NP group, e: significant compared to the DEN group, (p < 0.05).
Level of lipid peroxidation was evaluated by measuring the level of MDA either in liver tissues or plasma, while the antioxidant status was evaluated by measuring SOD activity either in liver tissues or RBCs. Our results showed that the level of MDA was significantly increased, and SOD activity was significantly decreased in the DEN group when compared to the control group. While after the treatment with Cu-CTAB, GaO-NPs, or Cu-CTAB + GaO-NPs complex in DEN + Cu-CTAB, DEN + GaO-NP, and DEN + Cu-CTAB + GaO-NP groups, the level of MDA was significantly decreased, while SOD activity was significantly increased when compared to the DEN group, as shown in Figure 5. Oxidative stress and antioxidant markers. (a): Level of MDA in liver tissues (blue color) (nmol/gm tissue) and plasma (red color) (nmol/L) in different groups; (b): activity of SOD in liver tissues (blue color) (U/gm tissue) and RBCs (red color) (U/mL) in different groups. Data are expressed as mean ± SE (n = 10). a: Significant compared to the control, b: significant compared to the Cu-CTAB group, c: significant compared to the GaO-NP group, d: significant compared to the Cu-CTAB + GaO-NP group, e: significant compared to the DEN group, (p < 0.05).
Results of the present study also showed that the activity of arginase and the level of caspase-3 mRNA in liver tissues were significantly increased in DEN + Cu-CTAB, DEN + GaO-NP, and DEN + Cu-CTAB + GaO-NP groups when compared to the DEN group, as shown in Figure 6. Liver mitotic and apoptotic markers. (a): Activity of arginase in liver tissues (U/gm tissue) in different groups; (b): activity of caspase-3 in liver tissues (U/gm tissue). Data are expressed as mean ± SE (n = 10). a: Significant compared to the control, b: significant compared to the Cu-CTAB group, c: significant compared to the GaO-NP group, d: significant compared to the Cu-CTAB + GaO-NP group, e: significant compared to the DEN group, (p < 0.05).
Histopathological findings
Histopathological examination was performed in the liver of all groups of animals that were investigated in the present study. Tissue specimens of the liver from control rats (Figure 7(a)) showed normal histological structure of the central vein and surrounding hepatocytes. Also, Cu-CTAB (Figure 7(b)), GaO-NPs (Figure 7(c)), and Cu-CTAB + GaO-NPs (Figure 7(d)) treated rats showed no histopathological alteration. By contrast, liver specimens from DEN-treated rats revealed severe damage of hepatocytes which was characterized by focal area of anaplastic hepatocytes with other cells forming acini associated with fibroblastic cell proliferation dividing the degenerated and necrosed hepatic parenchyma into nodules (Figure 7(e)). On the other hand, liver specimens from DEN + Cu-CTAB-treated rats showed degenerative change which was noticed in the hepatocytes adjacent and surrounding the dilated central vein (Figure 7(f)); while liver specimens from DEN + GaO-NP-treated rats showed inflammatory cell infiltration with fibroblastic cell proliferation dividing the degenerated hepatocytes into nodules in association with dilatation in the portal vein (Figure 7(g)). In DEN + Cu-CTAB + GaO-NP-treated rats, the degenerated hepatocytes in the parenchyma were divided into nodules by proliferated fibroblasts and inflammatory cell infiltration with dilated portal vein (Figure 7(h)). Photomicrograph of liver tissues of male rats of (a): the control group, showing normal histological structure of central vein (CV) and surrounding hepatocytes (h) (H&E, X40).; (b): the Cu-CTAB group, showing normal histological structure (H&E, X40); (c): the GaO-NP group, showing normal histological structure (H&E, X40); (d): the Cu-CTAB + GaO-NP group, showing normal histological structure (H&E, X40); (e): the DEN group, showing focal area of anaplastic hepatocytes with other cells forming acini (1) arrow (H&E, X40); (f): the DEN + Cu-CTAB group, showing dilatation in central vein (CV) with degeneration in surrounding adjacent hepatocytes (arrow) (H&E, X40); (g): the DEN + GaO-NPs group, showing fibrosis (f) with inflammatory cell infiltration (m) dividing the degenerated hepatocytes cells into nodules with dilatation in portal vein (PV) (H&E, X40); (H): the DEN + Cu-CTAB + GaO-NPs group, showing fibrosis (f) with inflammatory cell infiltration (m) dividing the degenerated hepatocytes (d) into nodules with dilatation in portal vein (PV) (H&E, X40).
Discussion
Hepatocellular carcinoma (HCC) is one of the most frequent malignancies and a leading cancer-related cause of death worldwide; therefore, there is an emergency need for improvements in the therapeutic approach during the last decades. 25 The present study was designed to synthesize and evaluate the activity of the novel complex Cu-CTAB + GaO-NPs against HCC. Accordingly, in the current study, the cytotoxic effects and the biological activities of complex (Cu-CTAB combined with GaO-NPs), Cu-CTAB, and GaO-NPs alone were evaluated both in vitro and in vivo.
Results obtained from in vitro study revealed that both tested complex and gallium oxide nanoparticles alone affect cell viability of HepG-2 cancer cell line with IC50 0.2 μg/mL and 360 μg/mL, respectively. These results are in harmony with early studies which observed the in vitro anticancer activity of gallium compounds against many cancer cell lines, including those for melanoma, 26 lymphoma, 27 colorectal adenocarcinoma, 28 breast carcinoma, 29 and hepatocellular carcinoma. 30 Also, the cytotoxicity effects of Cu-CTAB were determined on many cancer cell lines such as prostate cancer 31 and osteosarcoma. 17
In vivo experiments were estimated on male Wister rats, which were administrated diethyl nitrosamine (DEN) as a potent hepatocellular carcinogen and then treated with Cu-CTAB or/and GaO-NPs to evaluate the chemotherapeutic efficacy of the tested compounds against hepatocellular carcinoma. Several studies used DEN to induce hepatocellular carcinoma in an animal model, as it was found that DEN causes hepatic cellular injury through the generation of reactive oxygen species (ROS) and DNA damage. 32 In this study, rats intoxicated with DEN showed a significant elevation in the activities of liver enzymes, ALT and AST, along with a significant decrease in the concentration of total proteins and severe histopathological liver deterioration, indicating the liver damage. These observations are consistent with previous studies, in which rats treated with DEN showed pronounced biochemical and histopathological hepatic fibrotic alterations.33,34 Also, the results obtained from this study showed that rats intoxicated with DEN had a significant increase in the levels of MDA and a significant decrease in SOD activity as compared with the control group. Our results are in accordance with previous studies which reported that rat treatment with DEN caused oxidative stress as assigned by the increase in MDA and fall in SOD content.32,35–38
In the present study, however, rats suffering from hepatocellular carcinoma treated with Cu-CTAB, GaO-NPs, and Cu-CTAB + GaO-NPs complex showed a significant decrease in serum activities of AST and ALT and the levels of MDA in liver and plasma while the concentration of serum total protein and SOD activity in the liver and RBCs were increased compared to the DEN-injected group. The obtained results may be attributed to the cytotoxicity of estimated compounds to tumor cells which resulted in diminished cellular damage and permeability of cell membrane, as consistent with previous studies.6,39 Also, results of the current study revealed that the concentrations of serum AFP and TGF-β1 along with serum α–L–fucosidase activity were significantly decreased in rats treated with Cu-CTAB, GaO-NPs, and Cu-CTAB + GaO-NP complex when compared to the DEN-injected group, indicating the antitumor activity of Cu-CTAB, GaO-NPs, and Cu-CTAB + GaO-NP complex. Several studies reported the antitumor efficacy of Cu-CTAB17,31 or GaO-NPs,6,40 but this is the first study that showed the antitumor efficacy of Cu-CTAB + GaO-NP complex. The antitumor activity of gallium depends mainly on the mimic action of Ga3+ with Fe3+, and therefore gallium acts on cellular iron-dependent processes at various points to disrupt tumor growth. 9 This explains the increased levels of serum iron in GaO-NP and Cu-CTAB + GaO-NP groups when compared to control or DEN + GaO-NP and DEN + Cu-CTAB + GaO-NP groups when compared to the DEN group. It was reported that gallium modifies DNA three-dimensional structure and modulates protein synthesis. Also, it inhibits the activity of several enzymes, such as DNA polymerases, ribonucleotide reductase, ATPases, and tyrosine-specific protein phosphatase. 41 Schachter et al. 42 documented that the high concentrations of CTAB displace phospholipids along the cell membrane, and formed holes cause the cell to be too permeable and to die, which may attribute to its antitumor activity. Another study reported that CTAB inhibited tumor formation and delayed tumor growth by inducing caspase-activated apoptosis by depolarization of the mitochondrial membrane potential and inhibition of H+-ATP synthase activity, therefore decreasing ATP levels in the cell. 43 In an agreement with our results, the levels of caspase-3 mRNA in liver tissues were significantly increased in rats treated with Cu-CTAB, GaO-NPs, and Cu-CTAB + GaO-NP complex when compared to the DEN-injected group, which emphasize their antitumor activities.
Arginase is a manganese-containing enzyme present mainly in the liver, and it catalyzes the final step in the urea cycle to get rid of toxic ammonia by converting L-arginine to L-ornithine and urea. 44 In the current study, the activity of arginase in all groups intoxicated with DEN had a significant decrease in respect to normal rats. Disorder of cellular metabolism is one of the critical marks for cancer production and expansion. Thus, the carcinogenic DEN decreases the arginase activity to prevent arginine deprivation because cancer cells need abnormal quantities of varied amino acids for their distinct metabolism to maintain high proliferative rates and resist some cell death signals. 45 The current work also agreed with Chrzanowska et al. 46 who elucidated that arginase activity in HCC was lower than in the cirrhotic liver, and the decrease in arginase activity was associated with impaired cirrhotic liver function estimated. On the other hand, Cu-CTAB, GaO-NPs, and Cu-CTAB + GaO-NP complex in the current work showed an increase in arginase activity after administration for DEN groups. This may indicate the potential of Cu-CTAB, GaO-NPs, and Cu-CTAB + GaO-NP complex as anti-proliferative treatments by decreasing the metabolism of tumor cells and consumption of arginine.
Histopathological sections of the liver tissues of the DEN-intoxicated group showed histopathological alterations in the architecture with respect to the control group as focal area of anaplastic hepatocytes with other cells forming acini associated with fibroblastic cell proliferation dividing the degenerated and necrosed hepatic parenchyma into nodules, which is consistent with previous studies.36,47 While treating DEN-induced HCC rats with Cu-CTAB, GaO-NPs, or Cu-CTAB + GaO-NP complex revealed a degenerative change in the hepatocytes.
There are some limitations that need to be addressed regarding the present study. First of all, the size of the study is limited, and additional large-scale studies are needed to confirm this finding, also power analysis for sample size calculation was not carried out. Secondly, other indicators such as animal weight variations after and before treatment, disease index score, and beside to test the blood index, it is better to provide imaging approaches, such as MRI, US, or others, to monitor the HCC in live rats need to be examined in the further studies to prove the ameliorative effect of the Cu-CTAB + GaO-NP complex. Third, safety study of the Cu-CTAB + GaO-NPs complex should be carried out, and histological analysis must be performed for all the vital organs and compared with control healthy mice to validate our findings. Also, caspase-3 is an apoptotic marker; however, for mitochondrial dysfunction caspase-9 is equally important, thus there is a need to be evaluated together. Finally, toxic effects of Cu-CTAB, GaO-NPs, or Cu-CTAB + GaO-NP complex should be tested in normal cell lines.
Conclusions
To the best of our knowledge, this is the first report in the literature that addresses the ameliorative effect of the novel Cu-CTAB + GaO-NPs complex against DEN-induced HCC in rats. The present study demonstrated that both GaO-NPs and Cu-CTAB + GaO-NP complex exhibit cytotoxic effects against DEN-induced HCC which proved biochemically and histopathologically in in vivo study, while Cu-CTAB + GaO-NP complex exhibited the strongest in vitro anticancer activity against HepG-2, comparable to that of Cu-CTAB and GaO-NPs alone, as proved by in vitro study, which would in turn speculates a possible therapeutic role of the novel Cu-CTAB + GaO-NP complex.
Supplemental Material
Supplemental material - In vitro and in vivo studies of a newly synthesized copper-cetyl tri-methyl ammonium bromide combined with gallium oxide nanoparticles complex as an antitumor agent against hepatocellular carcinoma
Supplemental material for In vitro and in vivo studies of a newly synthesized copper-cetyl tri-methyl ammonium bromide combined with gallium oxide nanoparticles complex as an antitumor agent against hepatocellular carcinoma by Fatma SM Moawed, Riham Abdel-Hamid Haroun, Eman S Abou Zaid, Somya Z Mansour, Abdel-Fattah M Badawi, and Eman I Kandil in International Journal of Immunopathology and Pharmacology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Availability of data and material
All data generated or analyzed during this study are included in this published article.
Ethics approval
The present study was approved by the Animal Ethics Care Committee of the National Centre for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Authority, Cairo, Egypt (approval no. 19A/20). All animal procedures were carried out in accordance with the Ethics Committee of The National Center for Radiation Research and Technology in accordance with the “Guide for the Care and Use of Laboratory Animals” published by the US National Institute of Health (NIH publication, No. 85-23, 1996).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
