Abstract
Background and objectives
Metformin, an oral hypoglycemic drug, has been suggested to possess antitumour activity in several types of cancers. Additionally, interleukin-8 (IL-8) has been reported to be involved in the development and metastasis of many cancers. However, the effect of metformin on IL-8 expression in hepatocellular carcinoma (HCC) remains unclear. Therefore, this study aimed to investigate whether metformin could inhibit IL-8 expression to exert an inhibitory effect on HCC progression.
Materials and methods
The IL-8 levels were measured in the plasma of 159 HCC patients (86 men, 73 women; average age 56 years) and in the culture supernatant of HCC cells (Hep3B and HuH7) using flow cytometry. In addition, the protein expression levels of IL-8 were also validated by the Human Protein Atlas (HPA) database. The prognostic value of IL-8 was evaluated using the Kaplan–Meier Plotter database. The association between IL-8 expression and immune checkpoints was estimated using the TIMER and The Cancer Genome Atlas (TCGA) databases. What’s more, bioinformatics analysis, western blotting, and transwell assays were conducted to illustrate the molecular mechanism of metformin (≤1 mM) on IL-8 in HCC.
Results
IL-8 expression was found to be increased in the plasma of HCC patients, which is consistent with the expression of IL-8 in HCC cells and tissues. High expression of IL-8 was significantly related to poor prognosis. In addition, IL-8 was positively correlated with immune checkpoints in HCC. Notably, we found that low-dose metformin could inhibit the secretion of IL-8 by HCC cells and the migration of HCC cells. Mechanistically, low-dose metformin significantly suppresses HCC metastasis mainly through the AMPK/JNK/IL-8/MMP9 pathway.
Conclusion
The results indicate that low-dose metformin can inhibit HCC metastasis by suppressing IL-8 expression. Targeting the AMPK/JNK/IL-8 axis may be a promising treatment strategy for patients with HCC metastasis.
Introduction
Hepatocellular carcinoma (HCC) is the main type of primary liver cancer and is the third leading cause of cancer-associated deaths worldwide. 1 Although remarkable advances have been achieved in the treatment of HCC, the trends for 5-year survival of HCC patients remain poor. 2 High metastasis and recurrence rates are the dominant reasons for the poor outcomes of HCC patients. Hence, it is urgent to investigate the potential metastasis-regulating mechanisms and explore novel HCC treatment approaches to improve patient survival.
Metformin, the prescribed medication for treating obesity-associated type 2 diabetes (T2D), has been widely used in the clinic for more than 60 years 3 and has been studied in various cancer clinical trials, with doses ranging from 250 mg/day to 2000 mg/day. 4 Mounting evidence suggests that metformin may exert important anticancer effects in multiple cancers, including colorectal cancer, cervical cancer, and bladder cancer.5–7 Specifically, metformin can inhibit colorectal cancer progression by regulating the levels of proinflammatory cytokines. 8 Zhao et al. 9 noted that metformin could inhibit the IL-6-induced epithelial-mesenchymal transition (EMT) by blocking STAT3 phosphorylation in lung adenocarcinoma. Although previous studies have noted that metformin can inhibit the progression of HCC, 10 the underlying mechanisms by which metformin inhibits HCC remain to be further clarified.
Interleukin-8 (IL-8, also known as CXCL8), which contains the ELR (Glu-Leu-Arg) motif, is a crucial proinflammatory CXC chemokine, and is secreted by inflammatory and tumour cells. 11 Elevated IL-8 expression was related to advanced tumour stage, vascular invasion, and could indicate the clinical outcomes of multiple tumours.12,13 Fan et al. 14 found that IL-8 was a putative diagnostic and prognostic biomarker in prostate cancer. Meanwhile, studies have pointed out that the overexpression of IL-8 has been related to poor outcome with regard to immune checkpoint blockade. 15 However, the relationship between metformin and IL-8 expression remains elusive.
Thus, the aim of this study was to investigate the impact of metformin on IL-8 expression and its influence on the metastasis of HCC cells. Here, we demonstrate that low-dose metformin (≤1 mM) can inhibit the secretion of IL-8 by HCC cells through the AMPK/JNK pathway. Additionally, it significantly suppresses HCC metastasis via the IL-8-MMP9 pathway. Our data reveal that low-dose metformin may be a promising treatment strategy for patients with HCC metastasis.
Materials and methods
Cell culture
Human HCC cell lines (Hep3B, HepG2, and HuH7) and the normal liver cell line LO2 were acquired from the Chinese Academy of Sciences Cell Bank (Shanghai, China). All cells were grown in a complete DMEM medium (VivaCell, Shanghai, China) with 10% fetal bovine serum (FBS; CLARK Bioscience) at 37°C and 5% CO2.
Collection of patient samples
HCC patients (
Detection of cytokines in the peripheral blood of HCC patients
The levels of 12 cytokines (IFN-α, IFN-γ, TNF-α, IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p70, and IL-17) in the plasma of patients were measured using cytokine detection kits (Qingdao Raisecare Biological Technology, Qingdao, China). First, 2 mL peripheral blood collected by EDTA-K2 anticoagulant tube was centrifuged at 4°C for 10 min at 1000×g, and plasma was absorbed for later use. Then, 25 μL of experimental buffer, 25 μL of cytokine capture antibody-coated fluorescent microspheres, 25 μL of plasma specimens, and 25 μL of cytokine-detecting antibodies were added to the flow tubes and incubated at 37°C for 2 h. Besides, 25 μL of SA-PE was added and incubated for 0.5 h with shaking at room temperature. After washing with 500 μL of washing buffer, 200 μL of washing buffer was added to the reaction tubes. The expression levels of 12 cytokines were identified by DxFLEX flow cytometry (Beckman Coulter).
Cell counting Kit-8 (CCK-8)
HCC cells were seeded in 96-well plates (5000 cells/well) and cultured for 24 h. Then, different concentrations of metformin (MedChemExpress, NJ, USA) or pharmacological inhibitors (Supplemental Table 1) were added to each well and cultured for 72 h. 10 μL of CCK-8 reagent (Meilunbio, Dalian, China) was added to each well. After 2 h of incubation at 37°C, OD values were measured at a wavelength of 450 nm.
Apoptosis analysis
The apoptotic effect of metformin on human HCC cell lines (Hep3B and HuH7) was evaluated by flow cytometry using the FITC-Annexin V/PI apoptosis detection kit (Meilunbio, Dalian, China). Briefly, after treatment with different doses of metformin (0.1, 0.3, 1.0, and 3.0 mM) for 48 h, HCC cells were harvested and dispersed into cell suspensions with EDTA-free trypsin (Corning, NY, USA). The 1 × 106 cells were stained with 5 µL of annexin V-fluorescein isothiocyanate (FITC) and 5 µL of propidium iodide (PI) in the dark for 15 min. Apoptotic cells were detected by DxFLEX flow cytometry. The flow cytometric data were analyzed using FlowJo V10 software (FlowJo, LLC).
Migration assays
Cell migration assays were examined using 24-well plates (8.0-μm pore size, Corning, NY, USA). Hep3B and HuH7 cells were harvested and suspended in 200 μL of serum-free medium (1 × 105) and transferred to the upper chamber, while 600 μL of medium containing 20% FBS was added to the lower chamber. Following incubation for 48 h, the invading cells were fixed with 4% polyformaldehyde (Servicebio, Wuhan, China), stained with crystal violet (Beyotime Biotechnology, Shanghai, China), and counted in 5 random view fields under a light microscope.
Western blotting analysis
Total protein was extracted from HCC cells with RIPA lysis buffer containing protease inhibitors (SolarBio Life Science, Beijing, China). Quantification of protein was conducted with BCA Protein Assay Kit (Beyotime Biotechnology, Shanghai, China). Equal amounts of protein samples (40 μg) were separated by SDS-PAGE (NCM Biotech, Suzhou, China) and wet-transferred onto PVDF membranes at a constant 250 mA for 120 min at 4°C. 5% skim milk was utilized to block the membranes. The membranes were incubated with the corresponding primary antibodies at 4°C overnight. Then, the membranes were incubated with secondary antibodies. Membranes were detected using the BCIP/NBT Alkaline Phosphatase Color Development Kit (Beyotime Biotechnology, Shanghai, China) or NcmECL Ultra Kit (NCM Biotech, Suzhou, China). A digital camera (ZHIHENG Intelligent Technology, Nanjing, China) was utilized to visualize the protein bands. GAPDH levels were used for data normalization. All the antibodies utilized in this study are listed in Supplemental Table 2.
Bioinformatic analysis
The associations between IL-8 mRNA expression and the clinicopathological features of HCC patients were evaluated using The Cancer Genome Atlas (TCGA) database. The Human Protein Atlas (HPA) database 16 was queried for immunohistochemical data regarding IL-8 expression in HCC. Additionally, the prognostic value of IL-8 expression was assessed using the Kaplan‒Meier Plotter database. The TIMER database 17 was utilized to investigate the associations between IL-8 and 6 inhibitory checkpoints (PDCD1, CTLA4, BTLA, HAVCR2, TIGIT, and LAG3) in HCC. Subsequently, gene set enrichment analysis (GSEA) 18 was applied to elucidate the potential molecular mechanisms of IL-8 in HCC. The correlations of IL-8 expression with the expression levels of EMT-related molecules (MMP9 and VIM) and immune checkpoint ligands (PDL1 and PDL2) were estimated by the TCGA database.
Statistical analysis
All quantitative measurements were repeated three times and are presented as the means ± SD in this study. All data were statistically analyzed using GraphPad Prism (version 8.0.1 Windows version, GraphPad Software, San Diego). The Student’s t-test was used for comparison between two groups. One-way analysis of variance (ANOVA) was applied to compare differences in multiple groups. Pearson’s correlation test was applied to evaluate the correlation of the data with R version 4.2.1. The log-rank test was utilized to plot survival curves.
Results
Plasma IL-6 and IL-8 were significantly elevated in HCC patients
The expression levels of 12 cytokines in the peripheral blood of HCC patients.

IL-8 was highly expressed in HCC tissues and cells and related to poor prognosis
Next, the IL-6 and IL-8 secretion levels were examined in HCC cell lines using flow cytometry. The results showed that IL-8 secretion levels were significantly elevated in human HCC cells compared with those in normal hepatocyte cells (Figure 1(a)). In contrast, significantly decreased secreted protein levels of IL-6 were observed in human HCC cells (Supplemental Figure 1), which was not consistent with previous studies. Thus, IL-8 was selected for further study. Then, immunohistochemistry (IHC) analysis revealed that IL-8 was overexpressed in HCC tissues compared with that in normal hepatic tissues in the HPA database (Figure 1(b)). Additionally, analysis of clinicopathological parameters from the TCGA database showed that high IL-8 expression was related to the patient’s Child–Pugh grade ( The expression levels and prognostic value of IL-8 in HCC. (a) IL-8 secretion levels were upregulated in HCC cell lines compared with those in normal hepatocyte cells. (b) IHC staining images from HPA showed higher protein expression levels of IL-8 in HCC tissues than in normal hepatic tissues. (c) HCC patients with high IL-8 expression suffered worse overall survival, progression-free survival, and disease-specific survival according to Kaplan‒Meier plotter databases. (d) High IL-8 expression was correlated with shorter overall survival in different subgroups, including male, T1, hepatitis virus, stage Ⅰ, stage Ⅱ, and stage Ⅲ + Ⅳ. HR, hazard ratio; * Associations of IL-8 expression with clinicopathological characteristics in HCC patients. aDisease-free intervals. *

Furthermore, Kaplan‒Meier curves were utilized to evaluate the correlation between IL-8 expression and the clinical prognosis of HCC patients. As shown in Figure 1(c), high IL-8 expression was significantly related to worse overall survival, progression-free survival, and disease-specific survival in HCC patients. In addition, the relationship between IL-8 expression and overall survival in different subgroups was analyzed. The results showed that high IL-8 expression was related to poor prognosis in multiple subgroups, including male, T1, hepatitis virus, stage I, stage II, and stage III + IV (Figure 1(d)). Collectively, these results indicate that IL-8 may serve as a biomarker for prognostic prediction in HCC patients.
IL-8 was positively correlated with immune checkpoints
The tumour microenvironment (TME) of HCC is highly heterogeneous and complex with the expression levels of immune inhibitory checkpoints have crucial effects on the prognosis of cancer patients.
19
To explore the role of the IL-8-influenced inhibitory checkpoints, the TIMER database was utilized to estimate the underlying relationships between IL-8 expression and inhibitory checkpoints in HCC. The analysis showed that IL-8 expression was positively correlated with several inhibitory checkpoints, including PDCD1 (r = 0.263, Correlations of IL-8 expression with checkpoint molecule levels and the TME in HCC. (a) IL-8 was positively associated with inhibitory checkpoints in HCC by TIMER databases. (b) IL-8 was positively associated with tumour-infiltrating immune cells in HCC by ssGSEA. (c) The IL-8 expression was positively associated with PDL1 and PDL2 expression in the TCGA database.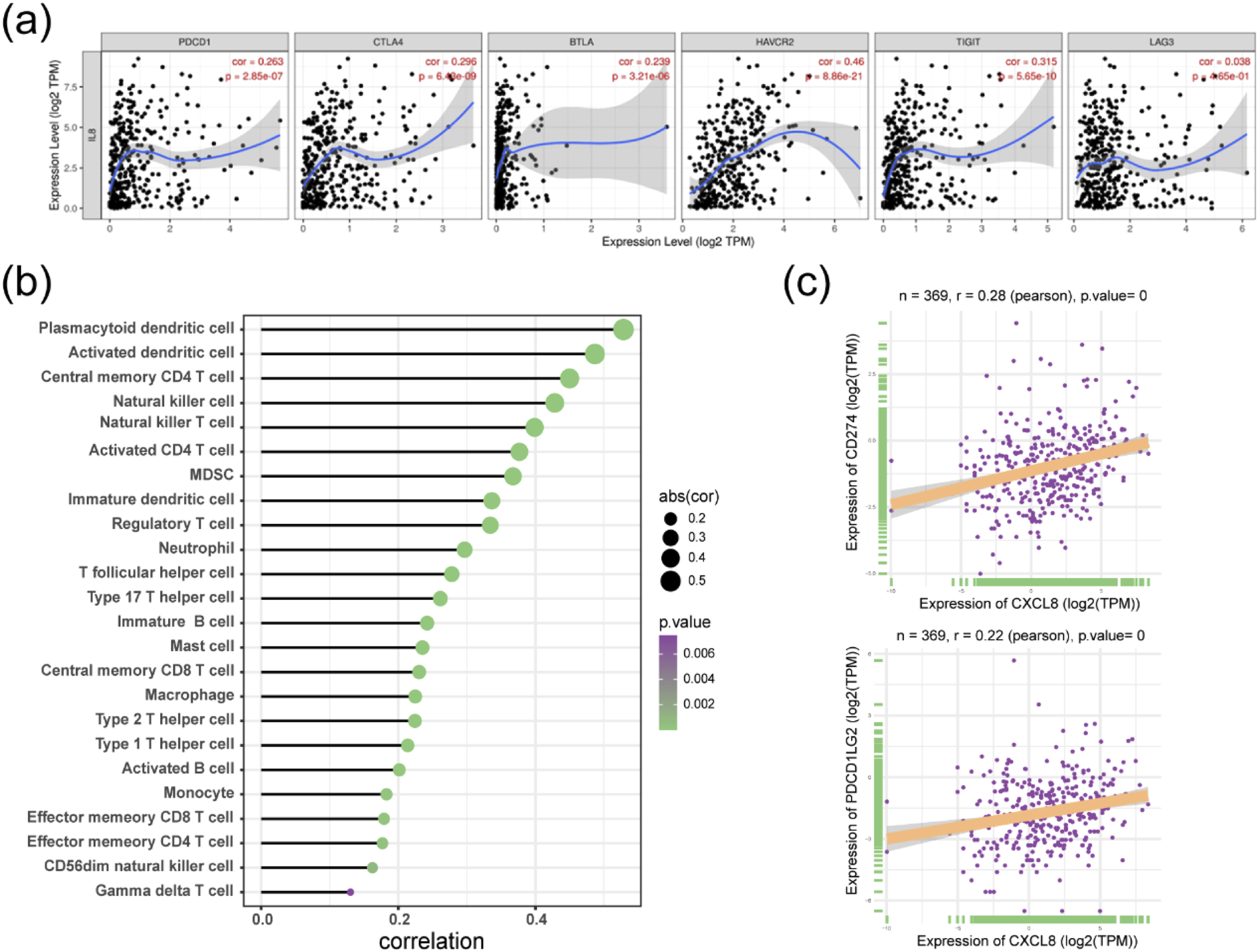
Low-dose metformin inhibited the expression of IL-8 in HCC cells
Accumulating studies have implicated that metformin plays a crucial anticancer role in many cancers.
20
To explore the effect of different concentrations of metformin on apoptosis and proliferation of HCC cells, apoptosis and CCK-8 assays were performed. The results unveiled that when the concentration of metformin was 3 mM, the total apoptosis rate of HCC cells was considerably increased and the proliferation ability of cells was decreased compared with other concentrations (Figure 3(a) and (b)). Unexpectedly, low concentration of metformin (≤1 mM) had no effect on cell apoptosis and proliferation, and the secreted protein levels of IL-8 were significantly decreased in a time- and concentration-dependent manner (Figure 3(c)). Thus, low-dose metformin was selected for further investigation. Low-dose metformin suppressed the expression of IL-8 in HCC cells. (a) Flow cytometric analysis was performed to evaluate the effect of metformin on apoptosis in HCC cells. (b) The viability of HCC cells was assessed using the CCK-8 assay. (c) The level of IL-8 secretion by HCC cells was analyzed using flow cytometry. *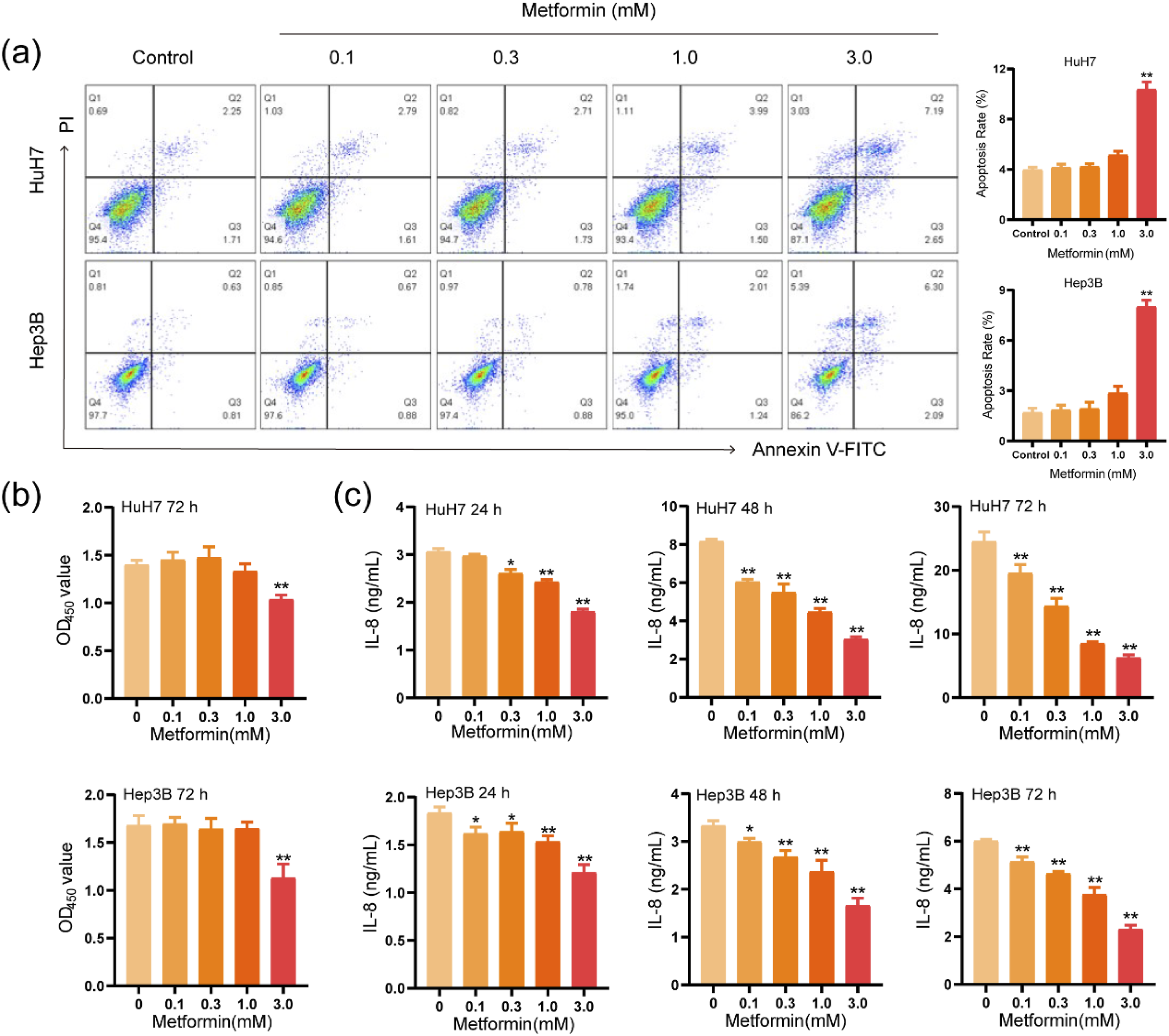
Low-dose metformin inhibited IL-8 expression through the AMPK and JNK pathways
Moreover, specific pharmacological inhibitors were used to determine the underlying mechanisms by which low-dose metformin inhibits IL-8. Surprisingly, the results showed that the screened concentrations of inhibitors didn’t affect cell growth (Figure 4(a)). However, both the JNK inhibitor (SP600125) and metformin significantly reduced the level of IL-8 secretion compared to the control and other inhibitors (Figure 4(b)). Some studies have unveiled that metformin can affect cancer progression through the AMPK and JNK signalling pathways.21,22 Thus, western blots were utilized to evaluate the effects of low-dose metformin on the expression levels of AMPK and JNK pathways in HCC cells. The results uncovered that low-dose metformin dose-dependently promoted phosphorylated AMPK (p-AMPK) expression and inhibited phosphorylated JNK (p-JNK) expression, but the levels of total AMPK and JNK weren’t changed (Figure 4(c)). Besides, the addition of compound C (the AMPK inhibitor, TargetMol, USA) effectively reversed the downregulation of p-JNK induced by low-dose metformin (Figure 4(d)). These results imply that low-dose metformin inhibits IL-8 expression via the AMPK/JNK signalling pathways. Low-dose metformin inhibited the expression of IL-8 through the AMPK/JNK pathways. (a) The viability of HCC cells after treatment with various inhibitors was determined using the CCK-8 assay. (b) Flow cytometry was utilized to measure IL-8 secretion levels in HCC cells after treatment with different inhibitors. (c) Effects of low-dose metformin on the AMPK and JNK pathways in HCC cells. (d) After treatment of HCC cells with metformin (1 mM) and metformin (1 mM) + compound C (1 μM) for 24 h, the expression levels of p-AMPK and p-JNK were measured by western blot. *
Low-dose metformin inhibited HCC cell migration through IL-8
Furthermore, the migration abilities of HCC cells were evaluated with low-dose metformin, anti-IL-8 monoclonal antibody (αIL-8, BioLegend), or IL-8 cytokine (TargetMol, USA) combination. When cells were treated with low-dose metformin, cell migration was reduced, and the effects were dose dependent. Similar results were gained when HCC cells were treated with αIL-8 (Figure 5(a)). In addition, when HCC cells were treated with low-dose metformin and IL-8, IL-8 reversed the inhibitory effect of low-dose metformin on cell migration (Figure 5(b)). These results showed that low-dose metformin inhibited IL-8-mediated HCC cell migration. Effects of low-dose metformin and IL-8 on the migration of human HCC cells. (a) Transwell assays were utilized to detect the migration ability of cells treated with metformin and αIL-8 (5 µg/mL). (b) The effects of low-dose metformin (1 mM) and metformin (1 mM) + IL-8 (20 ng/mL) on the migration of HCC cells were investigated by transwell assays. **
Low-dose metformin inhibited HCC cell migration by inhibiting MMP9
Next, we investigated the key molecules involved in the regulation of migration by metformin and IL-8. Bioinformatics analysis revealed that IL-8 significantly affected EMT (Figure 6(a)) and its expression was positively related to MMP9 and VIM expression (Figure 6(b)). Subsequently, the effect of low-dose metformin on EMT was explored. After treatment with low-dose metformin, HCC cells showed a significant reduction in MMP9 protein expression. However, there was no significant difference in the protein levels of E-cadherin and VIM (Figure 6(c)). Moreover, IL-8 increased MMP9 protein expression and counteracted the inhibitory effect of metformin on MMP9 protein expression (Figure 6(d) and (e)). As shown in Figure 6(f), IL-8 promotes the migration of HCC cells, and this effect can be reversed by adding the MMP9 inhibitor (JNJ0966, TargetMol, USA). Taken together, low-dose metformin could reduce HCC cell migration by inhibiting IL-8-MMP9 signalling axis. Low-dose metformin inhibited HCC cell migration by downregulating MMP9. (a) GSEA analysis of IL-8 and EMT. (b) Correlation analysis between IL-8 expression and MMP9 and VIM expression in TCGA databases. (c) Detection of EMT markers in HCC cells treated with low-dose metformin by western blotting. (d)–(e) Western blot assays were utilized to assess the protein expression levels of MMP9 in HCC cells under different conditions. (f) The effects of IL-8 and IL-8 + JNJ0966 on the migration of HCC cells were investigated by transwell assays. (metformin 1 mM; IL-8 20 ng/mL; JNJ0966 10 μM) *
Discussion
HCC is one of the most common malignant tumours, with a low survival rate globally. The specific mechanism of HCC occurrence has not been fully elucidated, due to its complexity and diversity of the pathogenesis,23–26 and clinical treatments still lack effective therapeutic strategies. Metformin is currently used as a first-line drug treatment for T2D. Specifically, accumulative studies have implicated that high-dose metformin may exert anticancer activity in multiple cancers, especially in HCC. 27 Nevertheless, in a recent randomized trial, 1 year of low-dose metformin significantly decreased the polyp and adenoma risk in patients undergoing polypectomy for single or multiple colorectal adenomas, 28 suggesting that low-dose metformin was more effective and appropriate than high-dose metformin. The main mechanism for the antitumour activity of metformin may be the inhibition of mitochondrial respiration, leading to intracellular energy consumption. 29 Recently, Yuan et al. 30 found that metformin therapy can significantly prolong the survival of HCC patients with T2D and reduce the recurrence rate, indicating the pivotal role of metformin in the treatment of HCC. In parallel, IL-8, a chemotactic cytokine, has been demonstrated to assist HCC progression by regulating self-renewal of tumour-initiating cells and angiogenesis.31,32 IL-8 is also related to HCC cell EMT and HCC cell invasion of blood vessels, promoting HCC metastasis. 33 Studies have reported that metformin can modulate IL-8 in human endometrial stromal cells. 34 Nevertheless, the relationship between metformin and IL-8 in HCC remains elusive. Herein, our study uncovered that IL-8 was upregulated and related to poor prognosis in HCC. Treatment with low-dose metformin reduced IL-8 expression in HCC cells.
EMT has a pivotal effect on the occurrence and metastasis of HCC. When EMT occurs, epithelial tumour cells undergo significant morphological and phenotypic changes, including cytoskeletal recombination and altered cell polarity, making tumour cells more invasive. 35 In addition, MMP9, as a member of the endopeptidase family of proteins, has been found to promote EMT, leading to increased invasiveness and metastasis of HCC cells. 36 Hence, it is crucial to investigate the related genes and pathways affecting EMT for the treatment of HCC. Specifically, recent researches have found that the AMPK and JNK signalling pathways are involved in EMT in many cancers. Chang et al. 37 found that elevated CLDN1 expression could promote the cancer progression by regulating EMT via AMPK/TGF-β signalling in head and neck squamous cell carcinoma. Besides, APOC1 participates in the progression of breast cancer through EMT and the MAPK/JNK pathway. 38 What’s more, the research showed that activation of AMPK by metformin alleviated the suppressive H3K9me2-mediated epithelial genes silencing during EMT and inhibited the metastasis of lung cancer, indicating that metformin showed promising prospects in inhibiting tumour metastasis. 39 In this study, the results showed that low-dose metformin significantly suppressed HCC metastasis by inhibiting MMP9 through the AMPK/JNK/IL-8 pathway. These data lead to a better understanding of the underlying anticancer effect of low-dose metformin.
PD-L1 is a main immune checkpoint molecule and has a negative regulatory effect on T-cell activity. High PD-L1 expression is related to poor outcomes in many cancers.40,41 Furthermore, the binding of PD-L1 to the PD-1 receptor on activated CTLs inhibited T-cell receptor signalling cascades and CTL activity. Therefore, attempts have been made to restore CTL activity by blocking PD-L1/PD-1 signalling. Recently, some studies uncovered that metformin can promote the immunotherapy of HCC by enhancing the efficacy of anti-PD-1. 42 The combination of anti-PD-1 antibody and metformin showed superior antitumour efficacy than monotherapy. 43 These results imply that metformin can enhance the antitumour effect of immune checkpoint inhibitors and may be a potential treatment option for HCC. In addition, studies have uncovered that IL-8 may be related to poor outcome with regard to immune checkpoint blockade. 15 Yuen et al. 44 pointed out that IL-8 was related to reduced clinical benefit of PD-L1 blockade. In addition, high levels of IL-8 were related to high neutrophil infiltration, poor T-cell function, and weak antigen presentation in a large-scale retrospective analysis. 45 In line with the prior studies of others, we observed that IL-8 was positively associated with inhibitory checkpoints and was positively related to PDL1 and PDL2 expression in HCC using databases. Hence, IL-8 may be used as a promising prognostic biomarker for HCC patients treated with immune checkpoint inhibitors.
However, there are some limitations to our study. In the future, we will collect plasma samples from two groups of HCC patients: those without T2D and those with T2D who are taking metformin. We will detect IL-8 levels in both groups and analyze the prognosis of patients, including metastasis and survival. Additionally, the effect and mechanism of metformin and IL-8 on immune checkpoint blockade in HCC need to be further evaluated.
Conclusion
In summary, IL-8 was overexpressed and associated with poor prognosis in HCC. Additionally, IL-8 expression was found to be positively associated with immune checkpoints in HCC. Notably, our findings demonstrate that low-dose metformin (≤1 mM) can inhibit IL-8 secretion and migration of HCC cells. Mechanistically, low-dose metformin significantly suppressed HCC migration via the AMPK/JNK/IL-8/MMP9 pathway. The results indicated that low-dose metformin is a possible therapeutic agent for patients with HCC metastasis. What’s more, the study provides new insights into understanding the potential mechanisms of low-dose metformin in alleviating HCC development and metastasis.
Supplemental Material
Supplemental Material - Low-dose metformin suppresses hepatocellular carcinoma metastasis via the AMPK/JNK/IL-8 pathway
Supplemental Material for Low-dose metformin suppresses hepatocellular carcinoma metastasis via the AMPK/JNK/IL-8 pathway by Chengwen Zhao, Lu Zheng, Yuting Ma, Yue Zhang, Chanjuan Yue, Feng Gu, Guoping Niu, and Yongqiang Chen in International Journal of Immunopathology and Pharmacology
Footnotes
Author contributions
CWZ, LZ, YTM, FG, GPN, and YQC designed and discussed this research. CWZ, LZ, YTM, YZ, and CJY carried out the experiments. FG and GPN analyzed the data. CWZ, LZ, YTM, and YQC prepared the figures and wrote the manuscript. All authors contributed to the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Natural Science Foundation of Jiangsu Province, China (grant number BK20210081); the Medical Scientific Research Program of Jiangsu Provincial Health Commission, China (grant number M2021025); the Key Research and Development Program of Xuzhou City, China (grant numbers KC23179); the Medical Youth Reserve Talents Training Program of Xuzhou Health Commission, China (grant number XWRCHT20220006); and the Key Medical Disciplines of Jiangsu Province’s 14th Five-Year Plan, China (grant number ZDXK202237).
Ethical statement
Data availability statement
All data generated or analyzed during the study are included in the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
