Abstract
Keywords
Introduction
The duodenal papilla part consists of a common duct that joins the bile duct and pancreatic duct, the surrounding sphincter muscle, and the papillary bulge of the duodenal mucosa. 1 Duodenal papilla is an important part of the opening of the pancreatic duct and common bile duct, which participates in regulating the excretion of bile and pancreatic juice. Therefore, it is regarded as an important anatomical structure. Duodenal papilla is covered with two types of mucosa, the intestinal mucosa of the duodenal ampulla and the mucosa of the pancreaticobiliary ampulla. 2 In terms of pathology, there are protuberant lesions of duodenal papilla mucosa, of which endoscopic manifestations are nodular, polypoid, or giant lesions. For protuberant lesions of duodenal papilla mucosa, chronic inflammation is more common, and inflammation is more than malignant tumor. Duodenal papillary carcinoma is a cancer that occurs in duodenal papilla as well as bile ducts, pancreatic ducts, and their confluent ducts surrounded by the sphincter of the biliary orifice in the duodenal wall. 3 Duodenal papillary carcinoma is the least prevalent form of gastrointestinal malignancy, accounting for only 0.2%. 4 Futakawa et al. held that lesions in 60 patients with duodenal papillary carcinoma were mostly located at the common opening. 5 Kimura et al. also found 576 autopsy reports of duodenal papilla finding the highest incidence at the common opening, which is considered to be an important site of duodenal papillary carcinoma. 2 Duodenal papillary carcinoma was predominantly adenocarcinoma. The etiology remains to be unclear, which may be associated with p53, p21, K-ras gene mutations, chronic inflammation, genetics, and other gastrointestinal malignancies.6,7 Early symptoms are not obvious, and when the tumor obstructs the biliary outlet, it may show tumor-related consumption symptoms such as such as progressively worsening jaundice, abdominal pain, and emaciation and anemia. Surgery is the mainstay of treatment with poor prognosis.
Materials and methods
According to pathological types, patients were divided into duodenal papilla malignancy group and chronic mucositis group.
Inclusion criteria
Duodenal papilla malignancies and chronic mucositis confirmed by pathology; complete clinical data.
Exclusion criteria
Patients with duodenal papilla lesions with incomplete medical records; a history of other malignant tumors and benign obstruction of ampulla
Chronic duodenal papilla mucositis group
37 patients with chronic duodenal papilla mucositis who were diagnosed by CT scan performed in our hospital (The Second Affiliated Hospital Zhejiang University School of Medicine) between January 2012 and December 2019 were collected. There were 13 males and 24 females. The youngest was 40 years old and the oldest was 87 years old, with an average age of 61.32 ± 9.58 years. Duodenal papilla CT scan and pathology are shown in Figure 1. Endoscopic manifestations of duodenal papilla: the descending papilla of duodenum was enlarged, the mucosa at the nipple opening was rough and granular, and the biopsy texture was soft. In some patients, the duodenal papilla is brittle and the surface was congested. (a) Abdominal computed tomography: enlarged duodenal papilla in the descending part of duodenum, which showed soft tissue nodular shadows with regular appearance and homogeneous enhancement. (b) Pathology: (staining, H&E; magnification, ×100) duodenal papilla chronic mucosal inflammation.
Duodenal papillary carcinoma group
46 patients with pathologically confirmed duodenal papillary carcinoma in our hospital from January 2012 and December 2019 were collected. There were 30 males and 16 females, the youngest was 43 years and the oldest was 84 years, with an average age of 66.67 ± 9.79 years. Dual-papilla CT scan and pathology are shown in Figure 2. Endoscopic manifestations of duodenal papilla: the descending part of duodenum was swollen and bulging, the surface mucosa was rough, the boundary was still clear, and the opening was granular. In some patients, the duodenal papilla was brittle and the surface was congested. There was no obvious difference between benign and malignant duodenal papilla lesions under duodenoscopy. (a) Abdominal CT: enlarged duodenal papilla in the intestinal lumen of the descending duodenum, which was irregular in shape, shallowly lobulated, heterogeneously enhanced, and slightly hyperdense with annular fine lines at the edges. (b) Abdominal CT: dilatation of the common bile duct and pancreatic duct. (c) Pathology: (staining, H&E; magnification, ×200) duodenal papilla moderate-to-poorly differentiated adenocarcinoma, infiltrating the full thickness of the duodenal muscular layer, involving the common bile duct. CT: computed tomography.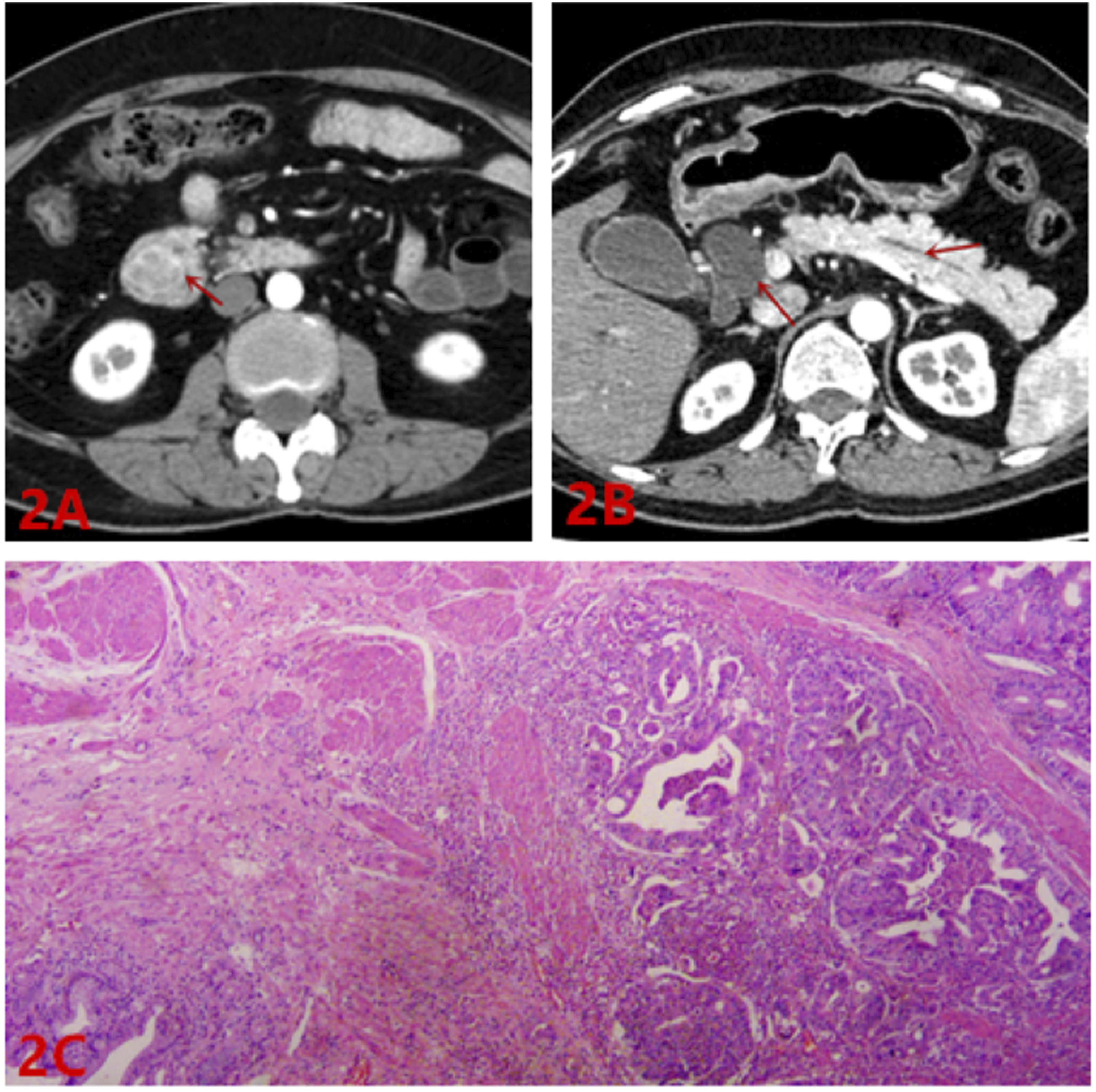
CT scan
Abdominal plain + triphasic enhanced scan was performed using Siemens 64-slice spiral CT scanner.
The scan range: up to the diaphragmatic dome and down to the upper pelvic margin. Scanning conditions: voltage 120 kV, current 200 mAs, scanning slice thickness 5 mm, slice spacing 5 mm, screw pitch 0.6. Puncture was performed via the elbow vein and 100 mL ioversol contrast agent (350 mg/mL) was injected at a rate of 3–4 mL/s. Arterial, venous, and delayed phase images were obtained 25 s, 65 s, and 110 s after the start of injection. Reconstructed images were transmitted from enhanced scans to Siemens background workstation.
Endoscopic and biopsies
All patients underwent duodenoscopic biopsy. Biopsy method: Olympus TJF-240 electronic duodenoscope was inserted from the mouth; through esophagus, stomach and duodenal bulb, it reaches the descending part of duodenum and finds duodenal papilla; by observing the condition of duodenal papilla, biopsy forceps were placed through duodenoscopy biopsy channel, and samples were taken from multiple points of biopsy forceps, and 2–3 tissues were clamped continuously at each clamp point. The biopsy forceps were pulled out and the tissues were placed in formalin solution.
The pathological manifestations of chronic inflammation of duodenal papilla mucosa were simple lymphocytes and plasma cells in lamina propria, intestinal glandular epithelium hyperplasia to intestinal villi atrophy and deformation to varying degrees, which accorded with chronic inflammation of mucosa. The pathological classification of duodenal papillary carcinoma group was adenocarcinoma.
Measurement index
The size and shape of duodenal papilla, CT value in each phase of plain scan and enhanced scan, enhancement uniformity, diameter of bile duct and pancreatic duct, presence of calculus at the lower end, bilirubin, white blood cell count, clinical jaundice, abdominal pain, fever, and other indicators were observed and compared between the two groups.
Statistical analysis
SPSS 22.0 software was used to process the data. Measurement data were expressed as mean ± SD and t test was adopted (There were few data in this study, but the variables were normally distributed, so t test was adopted). Enumeration data were expressed as n (%), χ2 test was used. p < .05 was considered as statistically significant.
Results
The majority of chronic duodenal papilla mucositis direct signs were mostly round with 75.68% (28/37), the maximum diameter of duodenal papilla was 11.41 ± 3.76 mm, enhanced uniformity was 72.97% (27/37), plain CT value was 37.43 ± 4.85, enhanced arterial phase CT value was 78.97 ± 18.86, and enhanced venous phase CT value was 88.32 ± 23.77. Indirect signs: the diameter of common bile duct was 11.67 ± 4.01 mm, and the diameter of pancreatic duct was 2.42 ± 1.10 mm. The lower end was accompanied by calculus 18.92% (7/37). Laboratory parameters CA19-9 (73.85 ± 197.43) ku/L, CEA (2.54 ± 0.97) μg/L, CA125 (2.54 ± 0.97) ku/L, direct bilirubin (10.90 ± 18.72) μmol/L, total bilirubin (27.19 ± 34.49) μmol/L, white blood cell count (7.82 ± 4.46) × 109/L, jaundice (18.92%, 7/37), abdominal pain (59.46%, 22/37), and fever (21.62%, 8/37).
Duodenal papillary carcinoma direct sign irregular shape was 45.65% (21/46), duodenal papilla maximum diameter was 18.52 ± 3.32 mm, enhanced uniformity was 56.52, 26/46. CT value of plain scan was 38.88 ± 4.30, arterial phase CT value of enhanced scan was 78.11 ± 14.41, venous phase CT value of enhanced scan was 80.98 ± 14.64. Indirect signs: the diameter of common bile duct was 19.06 ± 3.54 mm, and the diameter of pancreatic duct was 5.13 ± 2.71 mm. The lower end was accompanied by calculus 13.04% (6/46). Laboratory parameters CA19-9 (1247.11 ± 3365.41) ku/L, CEA (8.75 ± 22.69) μg/L, CA125 (21.4 2 ± 36.13) ku/L, direct bilirubin (68.82 ± 61.91) μmol/L, total bilirubin (118.44 ± 108.52) μmol/L, white blood cell count (9.13 ± 11.07) × 109/L, jaundice (89.13%, 41/46), abdominal pain (19.57%, 9/46), and fever (23.91%, 11/46).
Differences of Patients’ Information and CT.

CT: computed tomography.
Differences of Patients’ Clinical Features.

Discussion
Duodenal papilla is the common opening of the common bile duct and pancreatic duct, 7 which is the center connecting the liver, gallbladder, pancreas, and intestine. Therefore, it is also an important anatomical structure. Duodenal papilla lesions are common in inflammation, calculus and tumors, of which benign tumors are more common in adenomas, malignant tumors are more common in adenocarcinomas, and others such as lymphomas and stromal tumors are rare. 8 Due to the small size of duodenal papilla malignant tumor in the early stage, CT findings, clinical features and even endoscopic findings are often similar to chronic inflammation of duodenal papilla mucosa, which is easy to be confused. Therefore, it is difficult to diagnose accurately at an early stage. Secondly, chronic inflammation is more common for protuberant lesions of duodenal papilla mucosas, and inflammation is more than malignant tumor. In addition, no scholars have published the comparison of CT and clinical manifestations between chronic duodenal papilla mucositis and duodenal papillary carcinoma at present. In this study, MRCT, through thin-section scanning, enhanced scanning and laboratory examination, the imaging characteristics of duodenal papilla site and the comparison of benign and malignant lesions were displayed in more detail and statistically analyzed, including direct duodenal papilla signs (size, location, shape, density, etc.), indirect duodenal papilla signs (pancreatic duct bile duct dilatation, lower end with calculus) and clinical indicators (tumor markers CA19-9, CA125, CEA, blood routine (white blood cell count), bilirubin, with jaundice, with fever, with abdominal pain, age, gender), so as to detect duodenal papillary carcinoma earlier and provide more meaningful information for clinical diagnosis.
It has previously been reported in the literature that duodenal papillary carcinoma often occurs in middle-aged and elderly males, with a mean onset age of 50 years old and a peak age of 70 years old. 1 In the duodenal papillary carcinoma group, the mean age was 66.67 ± 9.79 years old, including 30 males and 16 females. The mean age of chronic duodenal papilla mucositis group was 61.32 ± 9.58 years old, including 13 males and 24 females. The mean age of duodenal papillary carcinoma group was higher than that of chronic duodenal papilla mucositis group, and there was a significant difference in age between the two groups, with the peak age of 60 years old or older, and the age significantly advanced, which was basically consistent with viewpoint of Chen Xiaoping et al. However, there was no statistical difference in gender between the two groups. Duodenal papilla malignancies are rare in clinical practice, and Gupta V et al. believed that jaundice may occur early in its clinical features, progressively worsen with disease progression, and present with symptoms such as abdominal pain. 8 Kumar M et al. held that duodenal papillary carcinoma is nonspecific in its clinical presentation, which may present as jaundice when the common bile duct is obstructed, but jaundice appears later. 9 In this study, duodenal papillary carcinoma group was accompanied by jaundice (89.13%, 41/46), abdominal pain (19.57%, 9/46), and fever (23.91%, 11/46). The chronic duodenal papilla mucositis group suffered from jaundice (18.92%, 7/37), abdominal pain (59.46%, 22/37), and fever (21.62%, 8/37). 89.13% cases in duodenal papillary carcinoma group presented with jaundice, while very few cases presented with jaundice in chronic duodenal papilla mucositis group. There was significant difference in jaundice between the two groups, but there was no significant difference in abdominal pain and fever, which was consistent with Gupta V et al.
Because duodenal papillary carcinoma lacks specific laboratory parameters, early diagnosis of duodenal papillary carcinoma is difficult. Beger HG et al. found that the positive rates of tumor markers CA19-9 and CEA in patients with duodenal papillary carcinoma were not high, but painless progressive jaundice could provide help for early diagnosis. 10 In this study, CA19-9 in duodenal papillary carcinoma group was 1247.11 ± 3365.41 ku/L, and 73.85 ± 197.43 ku/L in chronic duodenal papilla mucositis group, the difference between the two groups was statistically significant. There were no statistically significant differences in CEA, CA125 and white blood cell count between the two groups. Laboratory parameters are different from previous scholars' views. In duodenal papillary carcinoma group, the direct bilirubin was 6 8.82 ± 6 1.91 μmol/L and the total bilirubin was 118.44 ± 108.52 μmol/L. In chronic duodenal papilla mucositis group, the direct bilirubin was 10.90 ± 18.72 μmol/L and the total bilirubin was 27.19 ± 34.49 μmol/L. There were significant differences in direct bilirubin and total bilirubin between the two groups. It was considered that bilirubin increased might be associated with common bile duct obstruction caused by duodenal papillary carcinoma. 10
CT is beneficial to show the anatomy of the ampulla of Vater and the details of the lesion, and its localization diagnostic accuracy is high, which is an indispensable clinical diagnostic tool.11,12 Normal duodenal papilla CT showed semicircular or oval soft tissue density shadows protruding into the lumen from the medial wall of the descending duodenum. Horiguchi S et al. maintained that the maximum transverse diameter of duodenal papilla was generally about 5–10 mm.13,14 Chen WX et al. held that transverse papilla diameter of duodenal papillary carcinoma > duodenal papilla adenomatous lesions > chronic duodenal papilla mucositis lesions > maximum transverse diameter of normal duodenal papilla.15,16 In duodenal papillary carcinoma group, the maximum transverse diameter of the papilla was 18.52 mm ± 3.32 mm, the maximum transverse diameter of the papilla in chronic duodenal papilla mucositis group was 11.41 mm ± 3.76 mm, and there was a statistically significant difference in the maximum transverse diameter of the papilla between the two groups, which was consistent with the views of scholars Horiguchi S and Chen WX, namely, the maximum transverse diameter of the papilla of duodenal papillary carcinoma was larger than that of chronic duodenal papilla mucositis. Normal duodenal papilla showed conical or cylindrical nodular structures. 17 In this study, duodenal papillary carcinoma group had irregular papillae (45.65%, 2 1/46), while chronic duodenal papilla mucositis group had mostly round papillae, with irregular shape of 24.32% (9/37), there was statistical difference between the two groups. It can be seen that duodenal papilla type and size are particularly important in clinical diagnosis. Chang S et al. showed that duodenal papillary carcinoma showed homogeneous enhancement on CT in 75% of duodenal papilla cases, Diagnosis of duodenal papilla disease cannot be made by enhancing pattern. 18 In this study, 26/46 patients with duodenal papilla adenocarcinomas showed homogeneous enhancement; 27/37 patients with chronic duodenal papilla mucositis showed homogeneous enhancement, with no statistical difference between the two groups. There were no statistical differences in plain scan and arterial phase, portal venous phase CT value and duodenal papilla with calculus between the two groups, which were consistent with the views of Chang S et al. Therefore, in diagnosing duodenal papilla lesions, if duodenal papilla soft tissue density shadows are found to be homogeneously enhanced, the possibility of carcinogenesis cannot be ignored. Hawes RH et al. concluded that patients with duodenal papillary carcinoma often present with intrahepatic and extrahepatic bile duct dilatation and common bile duct and pancreatic duct dilatation on CT. 19 In this study, in the duodenal papillary carcinoma group, the diameter of common bile duct was 19.06 mm ± 3.54 mm and the maximum transverse diameter of pancreatic duct was 5.13 mm ± 2.71 mm; in the chronic duodenal papilla mucositis group, the diameter of common bile duct was 11.67 mm ± 4.01 mm and the maximum transverse diameter of pancreatic duct 2.42 mm ± 1.10 mm. There was significant difference in the diameter of common bile duct and pancreatic duct between the two groups, which was consistent with those in previous literature reports.
There are few data in this study, which still needs a lot of data to be further verified. The specific pathogenesis of duodenal papillary carcinoma and the specific indicators of early diagnosis are still the future research direction.
Conclusion
In summary, duodenal papillary carcinoma and chronic duodenal papilla mucositis have many similar manifestations in clinical manifestations, laboratory tests, and CT examinations. However, there are significant differences in duodenal papilla size, shape, diameter of common bile duct, pancreatic duct maximum long diameter, total bilirubin, direct bilirubin, CA19-9, jaundice, and age between the two groups. Therefore, comprehensive diagnosis should be combined with direct and indirect CT signs and laboratory and clinical manifestations of duodenal papilla, so as to improve the diagnosis of duodenal papillary carcinoma, and reduce missed diagnosis and misdiagnosis.
Footnotes
Author contributions
Ning Wang and Wenyan Wei: Conceptualization, Methodology, Formal analysis, Investigation, Writing - Original Draft; Weixing Mo: Validation, Investigation, Writing - Review & Editing; Qinghai Li: Statistical Analysis, Investigation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was waived by Hangzhou First People’s Hospital Medical Technology Clinical Application and Scientific Research Ethics Committee because our institution does not require ethical approval for analyzing cases or case series.
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
