Abstract
Objective
Airway inflammation is a prominent feature of asthma and may play an important role in disease pathophysiology. Despite the increasing incidence of asthma worldwide, reliable diagnostic biomarkers are lacking and widely lead to asthma misdiagnosis. Neutrophil–lymphocyte ratio (NLR) is a biomarker of systemic inflammation, in addition to NLR–alanine aminotransferase ratio (NAR) and NLR–albumin ratio (NBR). The aim of this study was to evaluate associations of NLR, NAR, and NBR with diagnosis of childhood asthma to determine if they can aid clinical childhood asthma diagnosis.
Methods
This retrospective case-control study included 89 children with asthma and 53 healthy children from the Wuxi Children’s Hospital affiliated with Nanjing Medical University. We applied various statistical tests to the dataset: Mann–Whitney U test to compare characteristics of the case and control groups; chi-squared test to compare categorical variables; Kruskal–Wallis test to compare statistical differences of asthma indicators among groups; receiver operating characteristic (ROC) curves to assess the diagnostic value of indices; and Spearman correlation analysis to evaluate relationships between NLR and lactate dehydrogenase, albumin, aspartate transaminase, and alanine transaminase levels.
Results
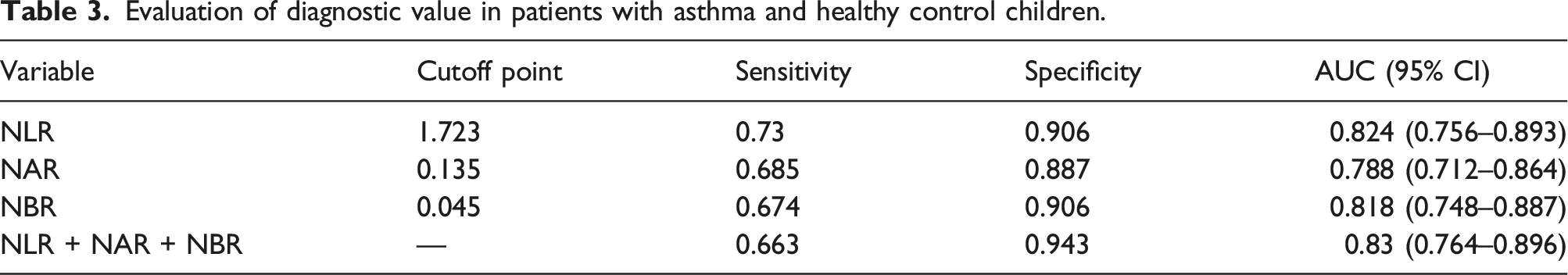
Compared with controls, the asthma case group had significantly higher white blood cell (p < 0.01), neutrophil, lactate dehydrogenase, C-reactive protein, and NLR levels (p < 0.01) and significantly lower lymphocyte (p = 0.001), platelet (p = 0.039), and albumin levels (p = 0.04). We determined optimal cutoff levels for several metrics: 1.723 for NLR, with sensitivity of 0.73 and specificity of 0.906; 0.135 for NAR, with sensitivity of 0.685 and specificity of 0.887; and 0.045 for NBR, with sensitivity of 0.674 and specificity of 0.906. The areas under the curve (AUCs) were 0.824 for NLR, 0.788 for NAR, 0.818 for NBR, and 0.83 for the combination of NLR + NAR + NBR.
Conclusion
The combination of NLR, NAR, and NBR biomarkers distinguished asthmatic ones suffering from exacerbation of the condition from healthy children. Thus, our results indicate NLR + NAR + NBR could be used as a clinical biomarker for asthma in children.
Keywords
Introduction
Asthma is a common and complex disorder characterized by inflammation and remodeling of the airway, leading to reversible airflow obstruction. 1 In most countries, the prevalence of asthma is increasing. The World Health Organization estimates that asthma affects more than 300 million people and accounts for a total of 250,000 deaths each year worldwide, including children. 2 Similar trends occur in China, with 2013 data indicating a prevalence of 3.0% in children aged 0–14 years. 3 Long-term uncontrolled asthma can lead to many complications, including chronic obstructive pulmonary disease and pulmonary heart disease, contributing to poor patient prognosis and generally harming human health. Thus, asthma is a serious global health problem that imposes heavy global economic and social burdens. 4
As childhood asthma progresses, untimely diagnosis and treatment can affect growth and development, limit physical activity, and lead to a poor prognosis. Asthma diagnosis mainly depends on clinical symptoms, objective signs, and lung function tests. However, this method has limitations, and it is difficult to complete lung function tests for patients <5 years old. Therefore, Fractional concentration of Nitric Oxide in the exhaled air (FeNO) is widely used to diagnose asthma, especially in children and particularly in pre-school–aged children. 5 The heterogeneity of asthma creates many diagnostic, prognostic, treatment, and management challenges, as disease pathogenesis is highly complex and the underlying cellular and molecular mechanisms remain poorly understood. Currently, this diagnostic test for asthma is still insufficient to clinically confirm or exclude disease. 6 Further, recent studies show that asthma may be overdiagnosed, with 30%–35% of asthma diagnoses representing misdiagnoses.7,8 Additional estimates suggest that 20%–70% of all asthma patients in the community may be misdiagnosed, partially due to unclear description of asthma symptoms in patients. 9 Therefore, more accurate diagnosis methods are needed for asthma. Reliable, easy to obtain, low-risk, clinically relevant, and ideally low-cost biomarkers, such as routine blood parameters, would be invaluable to improve the reliable detection rate of asthma and patient outcomes.10,11
Inflammation plays an important role in the mechanism of asthma pathophysiology. 12 Asthma involves various immune cells, including eosinophils, T lymphocytes, macrophages, and neutrophils. 13 Importantly, T lymphocytes involved in normal and asthmatic airway mucosa are activated and produce high levels of cytokines after antigenic stimulation or during acute asthma exacerbations, which control the inflammatory cell profile (e.g., neutrophils). 14 Further, neutrophil counts may be higher in sputum during acute asthma exacerbations in children. 15 Inflammatory infiltrates are mainly composed of eosinophils but also include mast cells, basophils, neutrophils, monocytes, and macrophages. T2-type inflammation occurs in >80% of children and in the majority of adults with asthma. 12
Routine blood index ratios can provide clinically useful diagnostic biomarkers. 16 For example, neutrophil–lymphocyte ratio (NLR) is a biomarker of generalized inflammation in pulmonology. Huang et al. 17 showed that the NLR values are a reasonable and easy-to-use marker for asthma. Here, we explored whether values of routine blood indexes are associated with exacerbations of asthma in children to determine if such indexes could be used as diagnostic biomarkers.
Materials and methods
Study population
This retrospective study included 89 children with bronchial asthma (aged <14 years) diagnosed in Wuxi Children’s Hospital affiliated with Nanjing Medical University from January 2019 to May 2020 as the case group. The control group included 53 healthy children who were seen for a routine health checkup or vaccination in Wuxi Children’s Hospital affiliated with Nanjing Medical University. There are pathophysiological differences between preschool children and school-aged children with asthma. In the former group, non-atopic episodes due to neutrophil infection can predominate, whereas in the latter group, airway eosinophilia, type-2 inflammation can occur prevalently, so age may be a factor affecting the disease. 18 Eighty-nine children with asthma were divided into mild, moderate, severe, and clinical remission groups based on clinical examination, pulmonary function testing, fractional concentration of Nitric Oxide in the exhaled air (FeNO), and methacholine bronchial provocation tests. Asthma was defined as the presence of symptoms with <16.0 mg/mL inhaled methacholine, which induced a 20% decrease in forced expiratory volume in one second (FEV1) (PC20). 19 Exclusion criteria included concomitant chronic respiratory disease, immunological disorders, cancer, current immunosuppression, or hepatic cirrhosis. Stable asthma was defined as the absence of symptoms of an acute exacerbation of asthma and lung function maintained for at least 4 weeks. This study was approved by the ethics committee of Wuxi Children’s Hospital Affiliated with Nanjing Medical University (WXCH2021-06–006).
Clinical and laboratory assays
Patient characteristics were collected from electronic medical records. After an 8-h overnight fast, venous blood samples (5 mL) were collected from each participant and placed in EDTA-K2 anticoagulant tubes and drying tubes. Whole blood routine parameters were analyzed in a Sysmex XE-5000 Automatic Hematology Analyzer (Sysmex Corp, Kobe, Japan). Analysis of liver function was carried out with a Beckman AU5800 Automatic Analyzer (Beckman Coulter Inc, Brea, CA, USA). Hematologic index neutrophil–lymphocyte ratio (NLR), NLR–alanine aminotransferase ratio (NAR), NLR–albumin ratio (NBR), and hemoglobin–platelet ratio (HPR) were acquired indirectly through calculation.
Statistical analysis
SPSS version 26.0 (SPSS Inc, Chicago, IL, USA) was used for statistical analysis. The Mann–Whitney U test was used to compare characteristics of the case and control groups. Medians (with interquartile ranges) are reported for continuous variables.
The Chi-squared test was used to compare categorical variables. The Kruskal–Wallis test was performed to compare statistical differences of asthma indicators among groups. Spearman’s correlation analysis was used to assess correlations between NLR and other laboratory parameters. Receiver operating characteristic (ROC) curves were generated to determine diagnostic accuracy. Values of p < 0.05 were considered statistically significant.
Results
Differences between patients with asthma and healthy children
Characteristics of study participants.

AbbreviationsMild, mild asthma exacerbation; Moderate, moderate asthma exacerbation; Severe, severe asthma exacerbation; Remission, clinical remission; WBC, white blood cell; HB, hemoglobin; PLT, platelet; HPR, hemoglobin–platelet ratio; NLR, neutrophil–lymphocyte ratio; N, neutrophil; LY, lymphocyte; CRP, C-reactive protein; AST, aspartate transaminase; ALT, alanine transaminase; LDH, lactate dehydrogenase; ALB, albumin; NAR, NLR–alanine aminotransferase ratio; NBR, NLR–albumin ratio
P*: Significance between healthy control group and patients with asthma.
p**: Significance between mild, moderate, and severe asthma versus remission groups.
The case group was further divided into an acute asthma exacerbation group (n = 81) and clinical remission group (n = 8). The acute asthma exacerbation group included 54 patients with mild asthma exacerbation, 17 patients with moderate asthma exacerbation, and 10 patients with severe asthma exacerbation. Among all asthma exacerbation subgroups, the severe asthma exacerbation group had highest levels of NLR [7.19 (3.02–11.68)], NAR [0.56 (0.23–0.92)], and NBR [0.16 (0.08–0.25)] (Figure 1). The clinical remission group had higher levels of NLR, NAR, and NBR than the control group but lower levels than all asthma exacerbation groups, including mild, moderate, and severe asthma groups (Figure 2). Comparison of NLR (a), NAR (b), and NBR (c) among asthma exacerbation groups (mild, moderate, severe). Comparison of NLR (a), NAR (b), and NBR (c) among asthma subgroups (control, exacerbation, remission).

Correlation analysis
Correlation analysis between NLR and other indicators in patients with asthma and healthy control children.

Diagnostic accuracy of NLR, NAR, and NBR
ROC curves distinguished patients with asthma from healthy subjects (Figure 3 and Table 3). The optimal NLR cutoff level was 1.723, with sensitivity of 0.73 and specificity of 0.906. The optimal NAR cutoff level was 0.135, with sensitivity of 0.685 and specificity of 0.887. The optimal NBR cutoff level was 0.045, with sensitivity of 0.674 and specificity of 0.906. AUC was 0.824 for NLR, 0.788 for NAR, 0.818 for NBR, and 0.83 for NLR + NAR + NBR. ROC curve analysis of NLR, NAR, and NBR alone or in combination in patients with asthma and healthy control children. Evaluation of diagnostic value in patients with asthma and healthy control children.
ROC curves for NLR, NAR, and NBR distinguished the severe asthma group from asthma subgroups (Figure 4 and Table 4). Among NLR, NAR, and NBR indexes, NLR with an optimal cutoff at 7.139 had the highest sensitivity of 0.6 and specificity of 0.924. NLR + NAR + NBR had a sensitivity of 0.6 and specificity of 0.924. AUC was 0.787 for NLR and 0.805 for NLR + NAR + NBR. ROC curve analysis of NLR, NAR, and NBR alone or in combination in severe asthma exacerbation and other asthma subgroups. Evaluation of diagnostic value in severe asthma exacerbation and other asthma subgroups.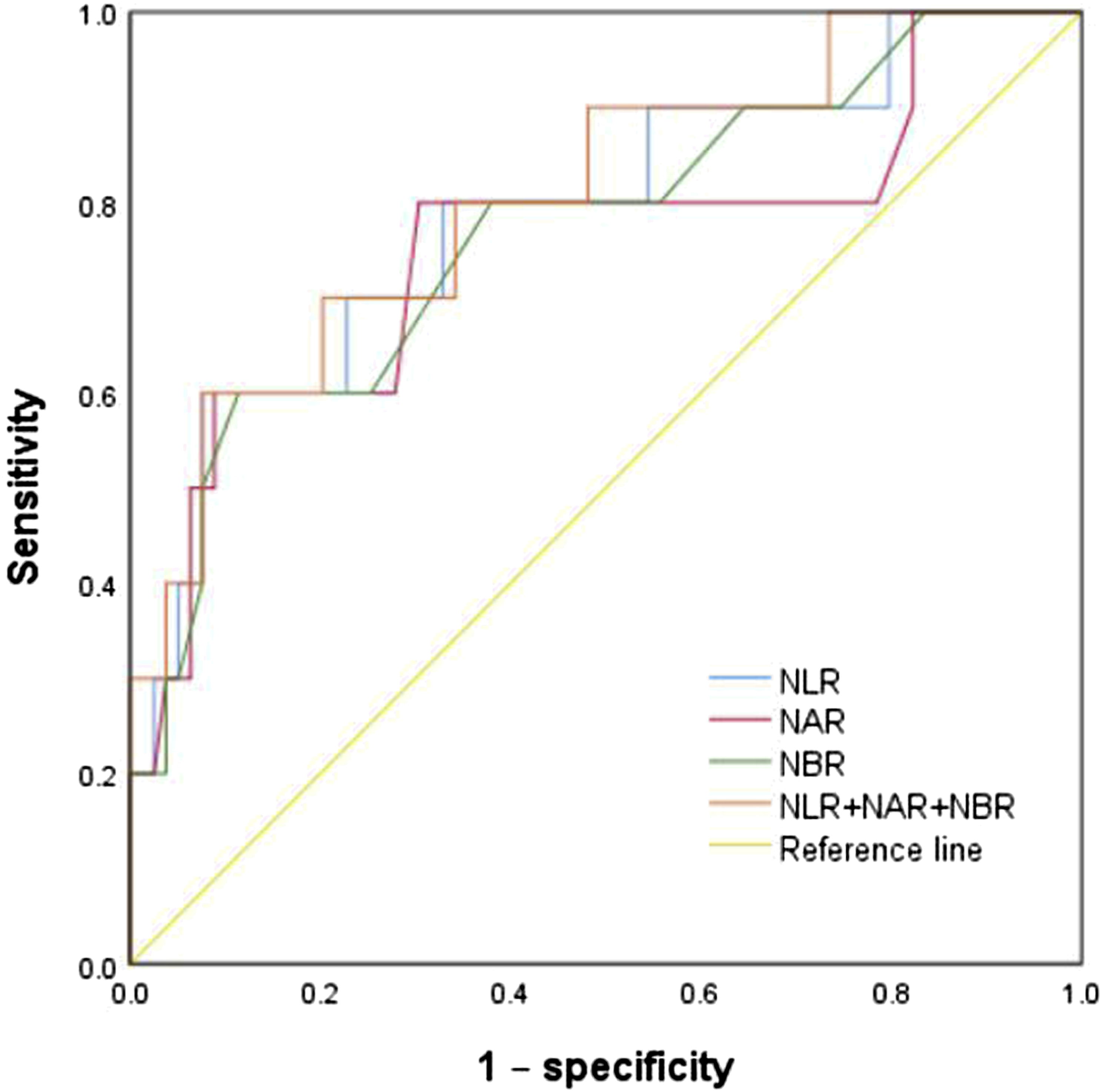

Discussion
In this study, we attempted to assess the levels of three significant indicators, NLR, NAR, and NBR, in childhood asthma to determine whether they could be useful biomarkers for early diagnosis. It is well known that asthma is a complex and heterogeneous disease with multiple phenotypes. 20 There is currently no gold standard for the diagnosis of asthma, and existing diagnostic modalities are probabilistic and take into account symptoms and variable expiratory airflow limitation. 21 In addition, fibreoptic bronchoscopy is risky and expensive for pediatric patients and may lead to tissue perforation and bleeding due to patient does not cooperate. Therefore, NLR, NAR, and NBR are non-invasive and secure method. For pediatric patients, this method requires very low blood volume and carries little risk, making measurement simple and allowing it to be used as a biomarker in asthma assessment and management alongside traditional clinical tools such as FeNO.
Zhu et al. 22 have found that the combined score of CRP level and NLR is a promising marker to distinguish children with exacerbated asthma from healthy children. Huang et al. 17 showed that children with asthma have a higher concentration of NLR. However, Bedolla-Barajas et al. have found that NLR did not differ between adult with asthma and control group. The level of NLR in children with asthma is increased, and age may be one of the influencing factors.
Neutrophils in sputum and bronchial lavage (BL) in asthmatic patients are associated with elevated levels of IL-5, IL-8, and proinflammatory mediators. Different triggers of severe acute asthma can recruit neutrophils through IL-8 production by activated macrophages or epithelial cells, leading to diffuse bronchial inflammation. 23 Th1/Th2 and Treg/Th17 imbalances have been found in patients with asthma, so asthma may be closely associated with activated T cells in the lungs. 24 Here we showed that patients with asthma have higher NLR, NAR, and NBR levels than healthy controls, suggesting that neutrophils and activated T cells may play an important role in the pathogenesis of asthma.
Total protein includes albumin and globulin. Synthesized by the liver, albumin is the main part of total protein and the most important protein in human plasma. Among its various physiological roles, serum albumin plays an important role in maintaining plasma colloid osmotic pressure, metabolism transport, and nutrition. 25 Decreased total protein and albumin levels are seen in some diseases with protein synthesis defects or excessive protein loss. ALB levels in asthmatic patients were significantly lower than those in the control group, which showed that the inflammatory of asthma might be related to nutritional insufficiency.
Platelets are involved in hemostasis and arterial thrombosis but also are involved in the physiological and pathophysiological processes of asthma.26,27 Platelets are involved in inflammatory responses and play an important role in platelet-dependent leukocyte recruitment. 27 LDH exists widely in human tissues, with the highest levels in kidney, followed by cardiac muscle and bone muscle. Serum LDH is generally considered an auxiliary diagnostic index of acute myocardial infarction. 28 In our study, compared with the control group, the case group had significantly higher LDH and significantly lower platelets. In addition, Spearman’s correlation analysis showed no correlation between NLR levels and AST, ALT, albumin, and LDH in asthma patients.
Our study had several limitations. First, it is a retrospective study and the study sample size was too small, second the lack of other inflammatory index such as eosinophils. Third, the age of the case group was statistically different from that of the control group. Finally, the group of asthmatic children was almost completely formed of children with asthma exacerbation.
Conclusions
The combination of NLR, NAR, and NBR improved our diagnostic ability to distinguish children with exacerbated asthma from healthy children. NLR, NAR, and NBR are a cheap and non-invasive test for patients that can be carried out in primary hospitals, offering potential as feasible and easily implemented asthma biomarkers. 29
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
