Abstract
The systemic inflammation plays a crucial role in carcinogenesis and cancer progression. Pretreatment lymphocyte-to-monocyte ratio (LMR) has been suggested to be associated with clinical outcomes in various malignancies. To evaluate the prognostic significance of pretreatment LMR on gastric cancer, we conducted a comprehensive literature search in PubMed, Embase, Web of Science, Cochrane Library, and ClinicalTrials.gov (Prospero Registration No. CRD42018087263). This meta-analysis included all studies evaluating the prognostic significance of pretreatment LMR on gastric cancer. The main outcome measures included overall survival (OS), progression-free survival (PFS), and the relationship between LMR and clinicopathological features. In total, 11 studies (12 cohorts) enrolling 14,262 patients with gastric cancer were included. The pooled estimates showed that elevated pretreatment LMR was significantly associated with better OS (hazard ratio (HR): 0.71, 95% confidence interval (CI): 0.58–0.83) and better PFS (HR: 0.71, 95% CI: 0.44–0.99). The elevated LMR was also significantly associated with young patients, female, low level of carcinoembryonic antigen (CEA), low level of carbohydrate antigen 19-9 (CA19-9), stage I–II, small tumor size, absence of lymph node metastasis, absence of vascular invasion, and absence of perineural invasion. In conclusion, the elevated pretreatment LMR predicted the better clinical outcomes in patients with gastric cancer.
Introduction
Gastric cancer remained the major cause of cancer-related mortality worldwide, with the annual incidence of nearly 1 million. 1 Currently, surgical resection was the main method for gastric cancer. Even though there is a great progress in surgical techniques and adjuvant therapy, the 5-year overall survival (OS) rate in patients with gastric cancer who underwent resection was only 50%–70%. 2 Prognostic indicators stratifying patients in the preoperative period were critical for optimizing gastric cancer management, decision-making, and improving clinical outcomes. Because patients with gastric cancer at the same TNM stage have different clinical outcomes, the prognostic indicators based on the TNM staging system still remain pessimistic. Therefore, additional prognostic indicators are needed to evaluate the risk of relapse better and to aid in the selection of individualized therapy.
There was growing consensus that systemic inflammation was involved in carcinogenesis and cancer progression. 3 The peripheral routine blood test has been proved to be indicators of systemic inflammation. This idea has led to a wide study of peripheral routine blood test in the hope of developing reliable, reproducible, and low-cost prognostic indicators. Several simple indictors based on routine blood tests, such as neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio (LMR), have been considered as potential prognostic indicators of gastrointestinal cancer. However, the prognostic significance of LMR on gastric cancer has not been investigated intensively. Several studies evaluating the relationship between preoperative LMR and clinical outcomes on gastric cancer have been published.4–6 On account of sample size and propose in the study design, the results remained inconclusive.
Given all this, we comprehensively searched the available studies and performed this systemic review and meta-analysis to evaluate the prognostic significance of LMR on gastric cancer. Moreover, we aimed to find out the relationship between LMR and clinicopathological features for optimizing the treatment of gastric cancer.
Methodology
Search strategy
Based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), we performed a comprehensive literature search in PubMed, Embase, Web of Science, Cochrane Systematic Reviews, and clinical trial database from inception to February 2018. Prior to the study, the protocol and search strategy were prespecified (Prospero Registration No. CRD42018087263). The following search terms were used: GC (gastric cancer, gastric neoplasms, stomach cancer, stomach neoplasms), LMR (lymphocyte monocyte ratio, lymphocyte to monocyte ratio), and MLR (monocyte lymphocyte ratio, monocyte to lymphocyte ratio). In addition, the references of related reviews and studies were also manually checked to recognize other potentially eligible studies. When necessary for further information, the corresponding authors were contacted.
Inclusion and exclusion criteria
All studies evaluating gastric cancer and LMR/MLR were considered eligible for meta-analysis. The inclusion criteria were as follows: (1) patients with gastric cancer were verified by pathological examination; (2) the prognostic significance of pretreatment LMR/MLR with OS or progression-free survival (PFS) was reported; (3) hazard ratio (HR) of LMR and 95% confidence interval (CI) were allowed or could be calculated by other data; (4) if MLR was reported, the LMR and other relevant data could be calculated.
The exclusion criteria applied were as follows: (1) case report, reviews, abstract, letters, expert opinions, experimental animal studies; (2) studies lacking basic information for calculating HR and 95% CI. When several papers repetitively reported the same cohort, or derived from the same database, we chose the most informative study for analysis.
Data extraction and quality assessment
We employed the Endnote bibliographic software (Endnote X7) to establish an electronic database and delete duplicate or uncorrelated records. Two trained investigators (Z.L. and Y.C.) reviewed the full texts of potential eligible studies independently. Information, including the first author’s name, year of publication, sample size, age, gender, country, treatment strategy, cut-off value, and HR with 95% CI for OS and PFS, was extracted and inserted into an Excel database.
Quality assessment of each study was independently performed in accordance with the Newcastle-Ottawa Quality Assessment Scale (NOS). The highest total scores were 9. Studies with NOS scores of ⩾6 were considered as high-quality studies. Any disagreements were resolved by consensus and discussion with other coauthors and the corresponding authors.
Statistical analysis
HR and 95% CI for LMR were directly extracted from the original study or from estimation in the light of the methods by Parmar et al.
7
If MLR was reported in the original article, HR or 95% CI for LMR was calculated as 1 divided by HR or 95% CI for MLR. If multivariate and univariate analyses were reported in the same study, we chose the multivariate analysis to generate the pooled estimates. I2 statistics was performed to assess the heterogeneity of the included studies. The pooled estimates were generated by the fixed-effects model analysis (inverse variance method) if a little heterogeneity (I2 < 50%) was found. If severe heterogeneity (I2 ⩾ 50%) was observed, the random-effects model (inverse variance method) was applied for analysis. We performed subgroup analysis and sensitivity analysis to explore the potential sources of heterogeneity when severe heterogeneity was observed. Assessment of publication bias was conducted by Egger’s test; trim and fill analysis was further performed to evaluate the prognostic significance of LMR. All statistical analyses were implemented using Stata 12.0 software (Stata Corporation, College Station, TX, USA), and a two-sided
Results
Description of the included studies
The titles and abstracts of 281 studies were retrieved by the initial search algorithm. After the initial review, 264 studies were excluded in accordance with our exclusion criteria, and only 17 studies were further evaluated. For the full-text information evaluation, we reviewed the remaining studies evaluating the prognostic significance of LMR on gastric cancer, which were published between 2014 and 2018. Of them, three did not provide the valuable results and three demonstrated the duplicate data. Thus, the rest of the 11 eligible studies (12 cohorts) involving 14,262 patients were included in this meta-analysis (PMID: 25139101; 28607596; 28741195; 28752404; 28706446; 25885254; 27229102; 29108302; 28860808; 28029656; 28915691), with sample size ranging from 91 to 3243 patients. One study included two cohorts and demonstrated the HR and 95% CI separately; 8 we marked them as Feng F. et al (A) and Feng F. et al (B).
Nine studies (10 cohorts) were from China, one from Japan, and one from Italy. LMR was based on the pretreatment routine blood test in all of the studies, which was calculated as absolute lymphocyte count divided by absolute monocyte count. HR in nine studies (nine cohorts) was produced by the multivariate analysis, and HR in two studies (three cohorts) by the univariate analysis. LMR was reported in seven studies (seven cohorts). MLR was reported in four studies (five cohorts). NOS score of each study was above 6.
LMR and OS on gastric cancer
In total, 11 studies (12 cohorts) involving 14,262 patients reported the data of LMR and OS on gastric cancer. The overall pooled estimates (HR: 0.71, 95% CI: 0.58–0.83) showed that elevated LMR was significantly associated with better OS on gastric cancer (Figure 1). Because severe heterogeneity was observed (I2 = 88.2%), we also performed subgroup analysis and sensitivity analysis to explore potential heterogeneity. Subgroup analysis was performed by sample size, main therapy, study location, clinical stage, and cutoff. The pooled estimates were not significantly changed (Table 1). When excluding any single trial, the pooled estimates were not significantly changed, indicating the credibility of our results (data not shown).

Forest plot showing meta-analysis results of the relationship between LMR and overall survival.
Pooled hazard ratio for overall survival and progression-free survival in the light of subgroup analyses.
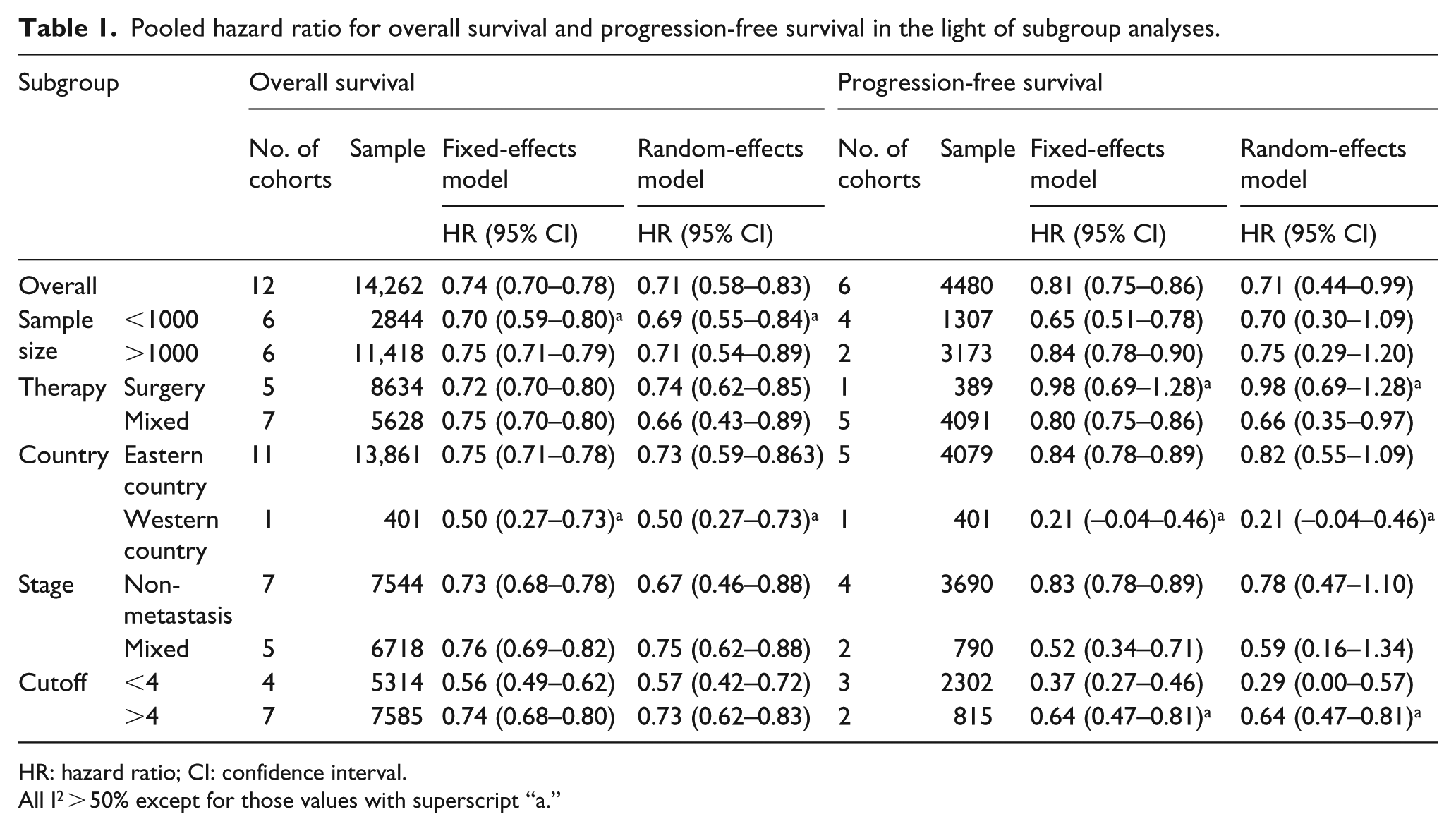
HR: hazard ratio; CI: confidence interval.
All I2 > 50% except for those values with superscript “a.”
LMR and PFS on gastric cancer
Six studies (six cohorts) involving 4376 patients reported the data of LMR and PFS on gastric cancer. The overall pooled estimates (HR: 0.71, 95% CI: 0.44–0.99) showed that elevated LMR was expected to have longer PFS (Figure 2). Because severe heterogeneity (I2 = 93.6%) was found, subgroup analysis and sensitivity analysis were also performed to explore potential heterogeneity. Subgroup analysis was performed as described above (Table 1). We also performed sensitivity analysis to explore the influence of a single cohort on the overall estimates. When excluding any single trial, the pooled estimates were not significantly changed (data not shown).
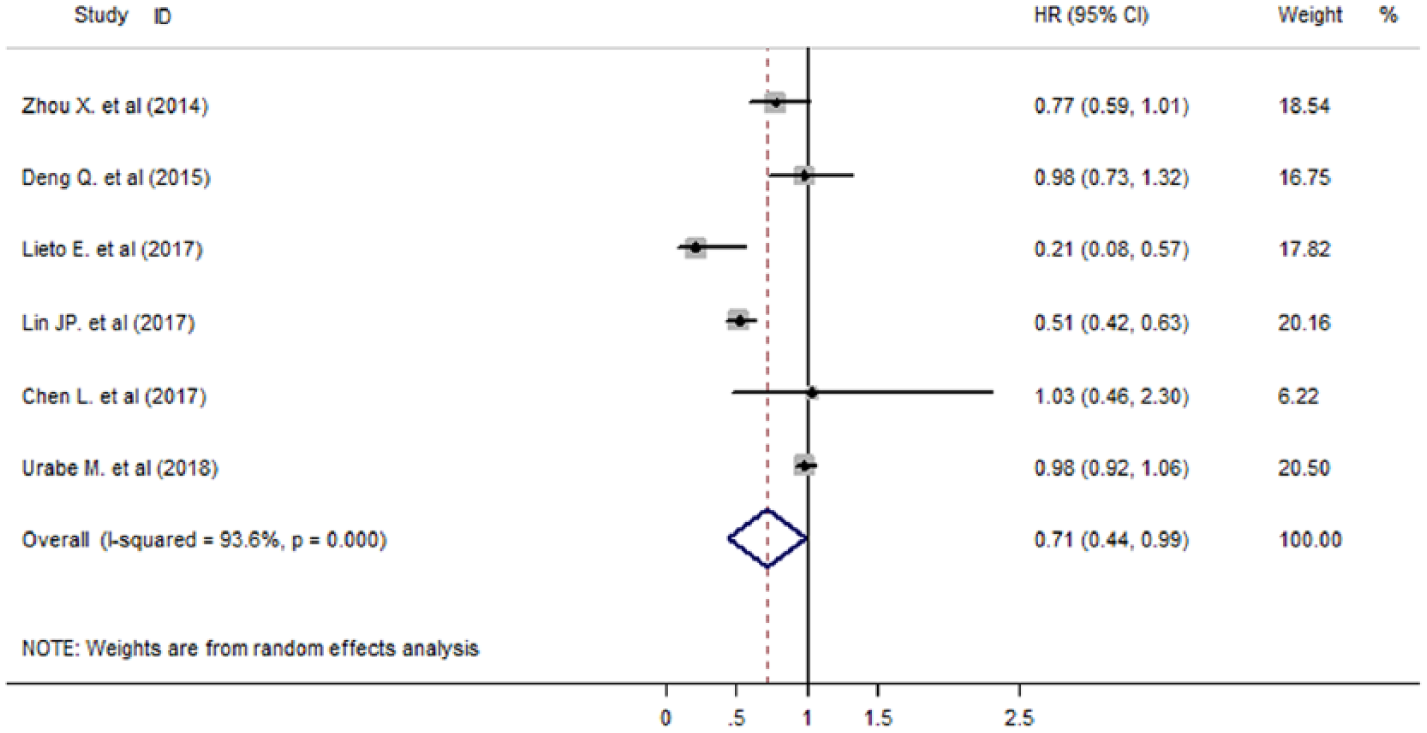
Forest plot showing meta-analysis results of the relationship between LMR and progression-free survival.
LMR and clinicopathological features
Aiming to explore the relationship between LMR and clinicopathological features, 10 clinicopathological features were identified (Table 2). The pooled estimates demonstrated that elevated LMR was significantly associated with young patients, female, carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA19-9), stage I–II, small tumor size, absence of lymph node metastasis, absence of vascular invasion, and absence of perineural invasion. There were no convincing evidences to prove the relationships between LMR and tumor differentiation.
Pooled hazard ratio for the relationship between LMR and clinicopathological features.
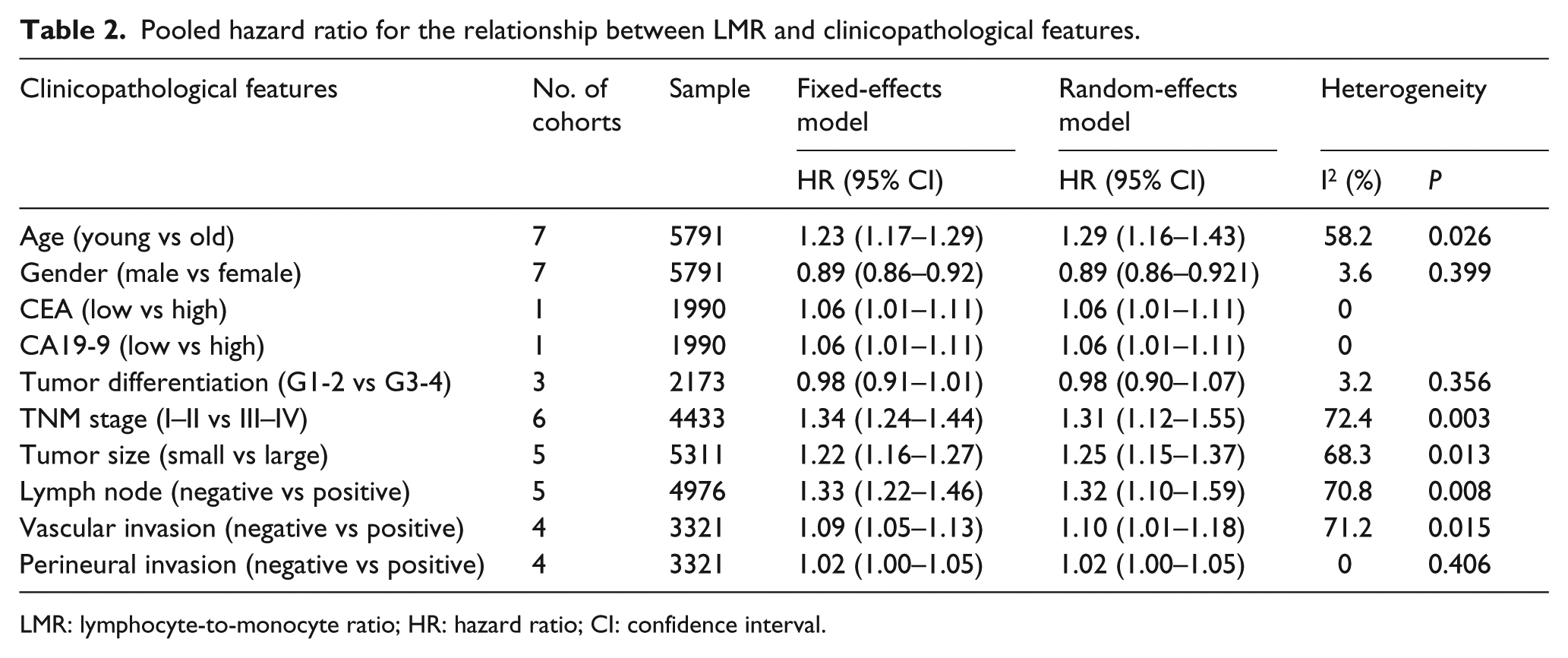
LMR: lymphocyte-to-monocyte ratio; HR: hazard ratio; CI: confidence interval.
Publication bias
Egger’s test was performed to evaluate overt publication bias in the meta-analysis. The
Discussion
Until now, the TNM staging system and tumor differentiation were the main approach for risk assessment of gastric cancer. However, they were imperfect methods for precisely predicting the prognosis. Additional prognostic indicators are necessary for improving the accuracy of the risk assessment system. Cumulative evidences demonstrated that systemic inflammation was significantly associated with carcinogenesis, cancer progression, and metastasis. Peripheral laboratory indicators including NLR, LMR, PLR, and systemic inflammation response index (SIRI) contributed to reflect systemic inflammation. 9 However, the prognostic significance of these biomarkers has been relatively ignored.
Several recent studies showed that pretreatment LMR was an independent prognostic indicator of various malignancies, such as colorectal cancer, breast cancer, and endometrial cancer. Even though severe heterogeneity was found in this study, our study demonstrated that elevated LMR was an independent prognostic indicator of better clinical outcomes in patients with gastric cancer. To the best of our knowledge, this meta-analysis is the most comprehensive assessment of the relationship between LMR and gastric cancer, which strongly supports the fact that the LMR was significantly associated with clinical outcome.
The exact mechanism of the relationship between the LMR and gastric cancer remained elusive. There were several interpretations for this relationship. The LMR reflected the balance of peripheral lymphocyte count and monocyte count. The elevated LMR had the relative lymphocytosis and monocytopenia. Lymphocytes, especially tumor-infiltrating lymphocytes (TILs), could reflect robust, sufficient immunity to cancer, thereby indicating better clinical outcome. On the contrary, there was increasing evidence that the monocytes suppress the host immune system and promote tumor burden. Taken together, the LMR was a useful indicator of the balance between the immune status of the host and the degree of tumor progression.
In conclusion, this systematic review and meta-analysis showed that elevated LMR was significantly associated with better clinical outcome, indicating that peripheral pretreatment LMR could be a useful and independent prognostic indicator of gastric cancer. Nevertheless, further large-scale and better-designed prospective studies are still needed to evaluate the relationship between LMR and gastric cancer.
Footnotes
Acknowledgements
Z.L. and Z.X. designed the study and wrote this manuscript. Z.L., Y.H., Y.W., H.R.K., Y.C., and R.Z. retrieved the database. Y.Z. and X.W. interpreted the data and proofread the final version. All of the authors have read and approved the final manuscript. Z.L. and Z.X. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
