Abstract
Background
Liver metastasis and chemotherapy resistance are two unfavorable factors for the outcomes of gastric cancer patients. ADHFE1 was identified as cancer-related molecule and candidate for early detection in gastric cancer. This study evaluated the significance of ADHFE1 in gastric cancer, aiming to explore a potential biomarker and therapeutic target for gastric cancer.
Methods
The expression of ADHFE1 was detected by polymerase chain reaction and compared between 143 gastric cancer patients and 54 individuals with benign lesions. The role of ADHFE1 in gastric cancer development and prognosis was assessed. The effect of ADHFE1 on gastric cancer cell growth and motility was evaluated by cell counting kit-8 and Transwell assay. The angiogenesis and cisplatin resistance were assessed by related markers and the half maximal inhibitory concentration values.
Results
Significant upregulation of ADHFE1 was observed in gastric cancer patients compared to individuals with benign lesions, which showed close association with the tumor node metastasis stage, lymph node metastasis, liver metastasis, and adverse prognosis of gastric cancer patients. Patients with liver metastasis showed higher serum ADHFE1 levels, which decreased after chemotherapy. Silencing ADHFE1 significantly suppressed gastric cancer cell proliferation, migration, invasion, and angiogenesis while its overexpression showed opposite effects. Moreover, the overexpression of ADHFE1 significantly improved the cisplatin resistance of gastric cancer cells.
Conclusion
Serum ADHFE1 served as an indicator for poor prognosis and liver metastasis of gastric cancer. ADHFE1 regulates gastric cancer cell progression, cisplatin resistance, and angiogenesis, suggesting its potential to serve as a therapeutic target.
Introduction
As one of the most common malignancies, gastric cancer possesses high incidence and mortality, and the age of onset tends to be younger. Gastric cancer is a multifactorial disease with high heterogeneous molecules and phenotypes, but the pathogenetic mechanism of gastric cancer remains unclear. In the early stage of gastric cancer, it is always asymptomatic, and most patients are diagnosed at advanced stages, which results in an unfavorable prognosis. 1 Gastrointestinal endoscopy combined with tissue biopsy is the golden standard for screening gastric cancer, but its application is limited to the disadvantages of invasiveness and high cost. 2 With the development of diagnostic technology, liquid biopsy has attracted special attention, which showed good compliance, and the samples for analyses are easily obtained.3,4 Recently, serum tumor markers, such as carcinoembryonic antigen (CEA) and carbohydrate antigen, have been employed in the clinic, but they have poor sensitivity and specificity.5,6 Additionally, distant metastasis is another adverse prognostic factor of gastric cancer. Liver metastasis accounts for the majority of distant metastasis in gastric cancer patients.7,8 According to the time point at which metastasis occurs, liver metastasis was divided into two types: simultaneous (occurred within 6 months after lesion resection) and heterochromia (occurred over 6 months after resection). 9 Chemotherapy is an effective treatment for patients with liver metastasis, but post-therapeutic recurrence and resistance still threaten patients’ outcomes. Predicting recurrence risk and therapy resistance could dramatically improve therapeutic efficiency; however, there is still a lack of sensitive biomarkers for this detection and prediction.
Alcohol dehydrogenase iron containing 1 (ADHFE1) has been identified as a cancer-related gene, which was demonstrated to promote tumor progression in breast cancer and colorectal cancer and was associated with the adverse prognosis of cancer patients.10–12 Several previous studies constructed various profiles of gastric cancer and screened several candidate biomarkers. For instance, a Weighted Gene Co-expression Network Analysis (WGCNA) study based on RNAseq datasets, GSE113255, GSE142000, GSE118897, and GSE130823 identified the increasing ADHFE1 level as a potential diagnostic marker for gastric cancer. 13 Another bioinformatic study revealed the prognostic significance of ADHFE1 in gastric cancer, where highly expressed ADHFE1 was found to show a close association with the poor survival of gastric cancer patients. 14 The specific function of ADHFE1 in other gastrointestinal tumors, such as colorectal cancer and esophagus cancer, was revealed.15–17 However, there has been a lack of direct evidence with clinical and experimental data confirming its significance in gastric cancer onset and development.
This study evaluated the expression and significance of ADHFE1 in gastric cancer, especially in patients with liver metastasis. Additionally, the response of ADHFE1 to chemotherapy and its regulatory effect of ADHFE1 on gastric cancer cells was also assessed to provide a potential therapeutic target.
Materials and methods
Study subjects
A total of 143 patients diagnosed with gastric cancer at The First Affiliated Hospital of Anhui Medical University North District were enrolled from January 2018 to December 2020. The inclusion criteria were: (a) patients who were primarily diagnosed with gastric cancer; (b) patients without history or complication of other malignancies; (c) patients who had not received radiotherapy, chemotherapy, or other anti-cancer therapy; (d) patients with completed clinical records; and (e) patients or their families had provided informed consent.
Another control group was composed of 54 patients receiving gastrointestinal endoscopy during the same period at The First Affiliated Hospital of Anhui Medical University North District and diagnosed with benign lesions by pathological examination. This group of patients had no history of other malignancies or precancerous lesions.
The study had been approved by the Ethics Committee of The First Affiliated Hospital of Anhui Medical University North District, and a follow-up survey was conducted by telephone or through outpatient review. If the patient died or the gastric cancer recurred, the time of death or recurrence was recorded. If patients showed no gastric cancer-related progression, the survival time was calculated according to the last follow-up time. The survival time was recorded in months, and the follow-up survey was up to December 2023.
Sample collection and preparation
Fasting peripheral venous blood was collected in the morning into a vacuum tube without anticoagulants. The serum was isolated by centrifugation at 3000 rpm for 10 min at room temperature within 4 h of blood collection. Isolated serum was stored at −80°C for the following analyses.
Serum tumor markers, including CEA, carbohydrate antigen 199 (CA199), and alpha fetoprotein (AFP), were analyzed by the DXI-800 autoanalyzer (Bechman Coulter, Brea, CA, USA).
Cell culture
The study employed a normal human gastric epithelial immortalized cell line, GSE-1 cell, and a human gastric adenocarcinoma (AGS) cell. Cells were obtained from the Beijing Institute for Cancer Research Collection and incubated with RPMI-1640 culture medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 g/mL streptomycin (Invitrogen). Cells were maintained at 37°C with 5% CO2 and were available reaching the logarithmic phase.
Cell transfection
Cells were transfected with the small interference RNA of ADHFE1 (si-ADHFE1) to knock down ADHFE1 or pcDNA 3.1-ADHFE1 for its overexpression (oe-ADHFE1) using Lipofectamine 2000 (Invitrogen). Cell transfection was performed at room temperature, and transfected cells were available for the following experiments after 48 h of cell transfection. The transfection efficiency was assessed by the expression of ADHFE1 using quantitative polymerase chain reaction (qPCR).
Real-time qPCR
Total RNA was extracted from serum and cells using Trizol reagent, and the purity and concentration were evaluated by the values of OD260/280 and OD260/230. cDNA was generated from isolated RNA using M-MuLV reverse transcriptase. PCR amplification was performed on the ABI PRISM 7900 system (Applied Biosystems, Foster City, CA, USA) with the assistance of a SYBR Green kit. GAPDH was used as the internal reference, and the 2−ΔΔCT method was used to calculate the relative expression of ADHFE1, CD31, CD34, and VEGF.
Cell proliferation assay
Cells were seeded into 96-well plates and supplied with a completed culture medium mainlining at 37°C with 5% CO2. The plates were incubated for 0, 24, 48, and 72 h, and then 10 μL CCK8 reagent was added to each well. Then, the absorbance at 450 nm (OD450) was measured with a microplate reader, and the time-OD450 curve was plotted representing the proliferation of cells.
Cell motility assay
Cells were seeded into the upper chambers of 24-well Transwell plates (Corning Inc., NY, USA), and the upper chamber was filled with FBS-free culture medium. The upper chamber was pre-coated with Matrigel before the invasion assay. The lower chamber was filled with a chemoattractant. Cell migration and invasion were evaluated by the number of cells on the subsurface of the upper chamber. Cells were fixed with glutaraldehyde and stained with violet crystal.
Evaluation of cell resistance to cisplatin
The viability of AGS cells was evaluated under 0, 2, 4, 6, 8, and 10 μg/mL cisplatin after 48 h cell transfection. The treatment of cisplatin was sustained for 24 h. The IC50 values were evaluated by non-linear regression analysis, and the higher IC50 values indicated the higher resistance of cells to cisplatin.
Statistical analyses
Difference comparison was conducted with the Student's t-test and one-way ANOVA (P < 0.05). The receiver operating characteristic (ROC) curve was performed to evaluate the discriminating value of ADHFE1 in liver metastasis of gastric cancer patients, and its correlation with patients’ clinicopathological features was evaluated by Chi-square test. The Kaplan–Meier curve and Cox regression analysis were carried out to assess the prognostic value of ADHFE1 in gastric cancer patients. The correlation of ADHFE1 with tumor markers, CEA, CA199, and AFP were evaluated by Pearson correlation analysis.
Results
Expression and prognostic value of ADHFE1 in gastric cancer
Compared with individuals with benign lesions, significant upregulation of ADHFE1 was observed in the serum of gastric cancer patients (Figure 1(a)). Based on the average serum ADHFE1 levels, gastric cancer patients were separated into a low-ADHFE1 group and a high-ADHFE1 group with 70 and 74 patients, respectively. The high-ADHFE1 group showed a poorer 5-year progression-free survival than that of the low-ADHFE1 group (log-rank P = 0.024, Figure 1(b)).
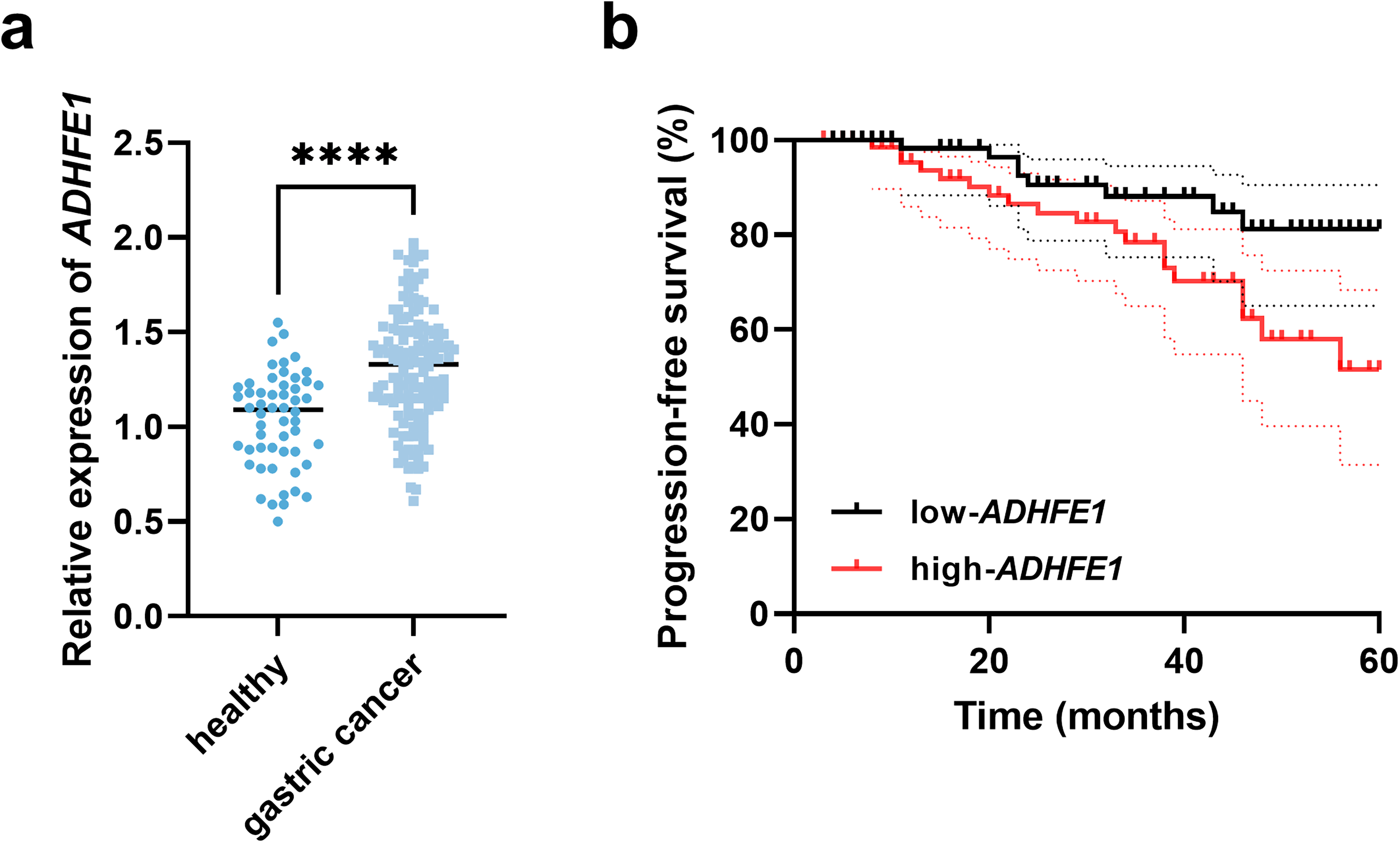
Expression of ADHFE1 and its association with 5-year progression-free survival of gastric cancer patients. (a) Serum expression of ADHFE1 in study subjects. (b) Kaplan–Meier curve based on serum ADHFE1 levels in gastric cancer patients. ****P < 0.0001.
Additionally, the high-ADHFE1 group showed a larger number of patients with advanced tumor node metastasis (TNM) stage (P = 0.014), liver metastasis (P < 0.001), and lymph node metastasis (LNM, P = 0.007), and these indexes were demonstrated to show significant correlations with ADHFE1 (Table S1). More importantly, ADHFE1 was identified to possess significant prognostic significance with the HR value of 4.363 (95% CI = 1.744–10.917), as well as TNM stage (HR = 3.706, 95% CI = 1.243–11.051), liver metastasis (HR = 4.048, 95% CI = 1.640–9.992), and LNM (HR = 10.231, 95% CI = 1.175–39.047, Table 1).
Multivariate Cox regression analysis evaluating adverse prognostic factors for gastric cancer.
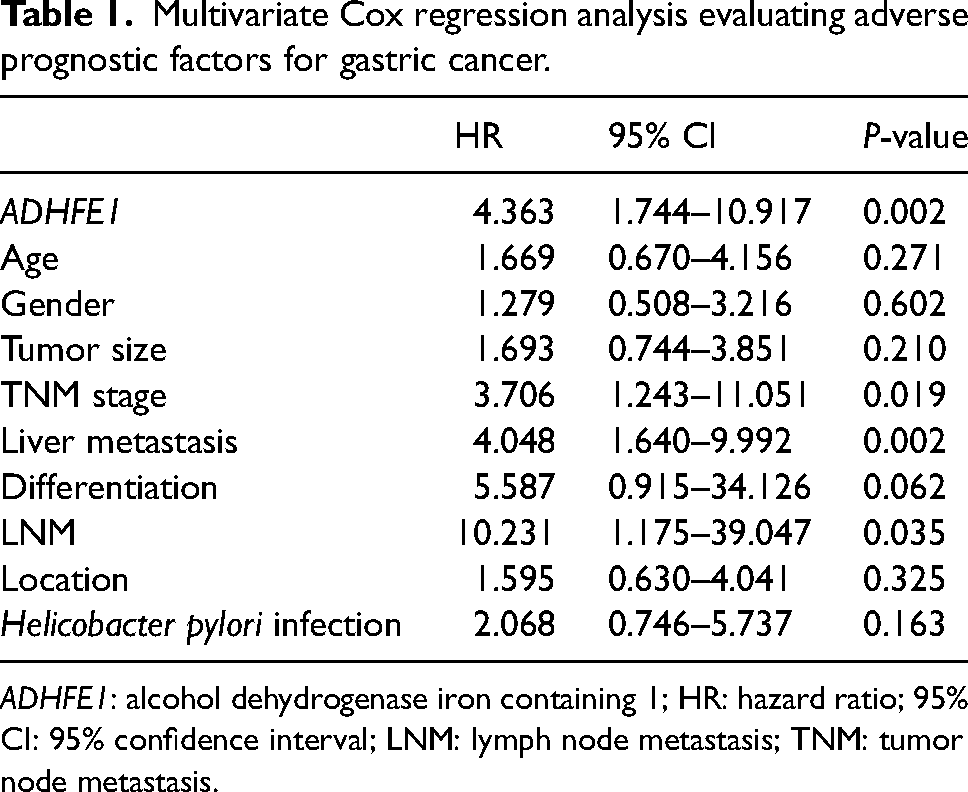
ADHFE1: alcohol dehydrogenase iron containing 1; HR: hazard ratio; 95% CI: 95% confidence interval; LNM: lymph node metastasis; TNM: tumor node metastasis.
Expression and significance of ADHFE1 in the liver metastasis of gastric cancer
There were 36 patients occurring liver metastasis among the enrolled gastric cancer patients, and the serum ADHFE1 level in patients with liver metastasis was significantly higher than that in patients without liver metastasis (Figure 2(a)). Serum ADHFE1 showed significant diagnostic value discriminated patients with liver metastasis with the sensitivity and specificity of 75.00% and 86.92%, respectively (area under the curve (AUC) = 0.863, Figure 2(b)). All these patients with liver metastasis received chemotherapy, after which the serum ADHFE1 decreased (Figure 2(c)).
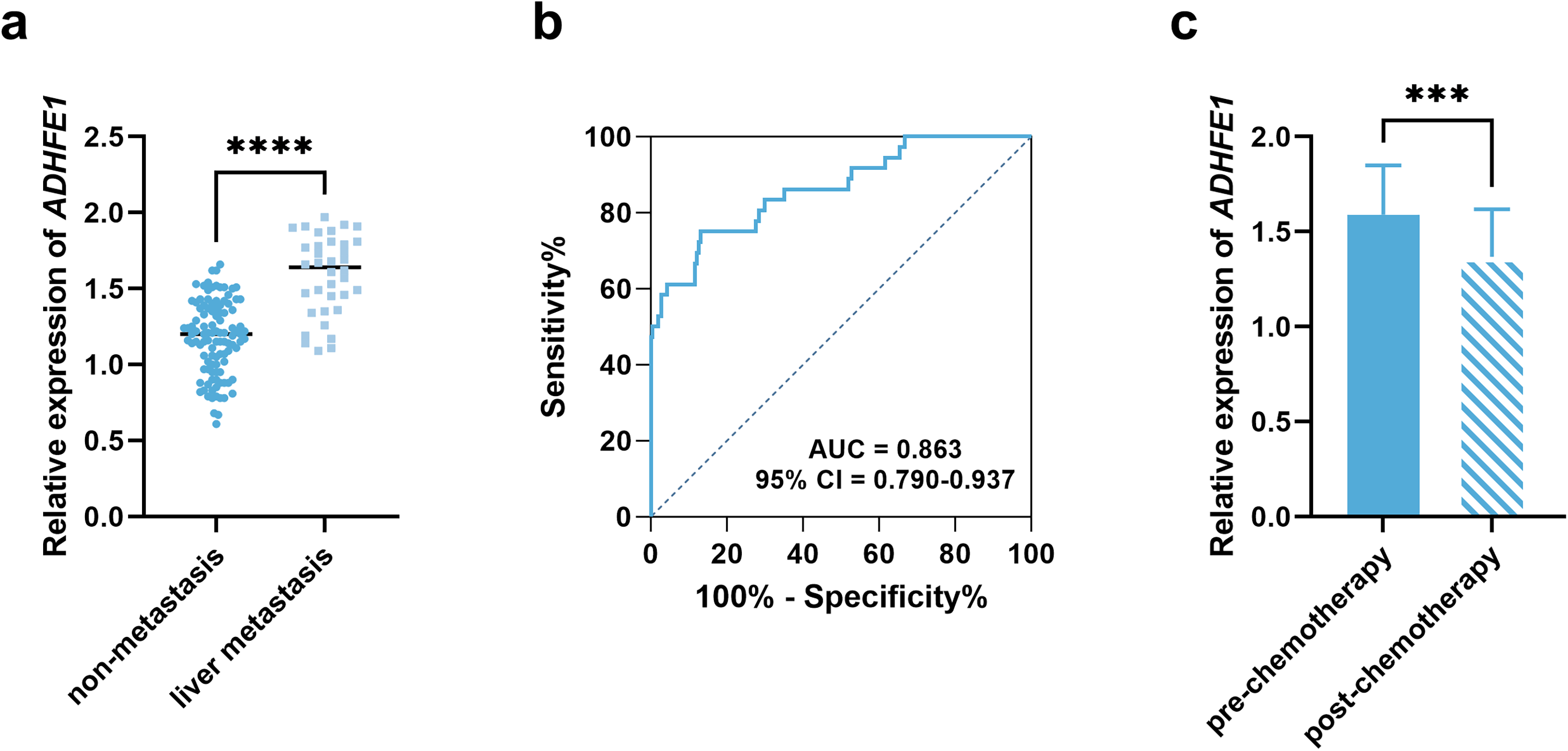
Expression of ADHFE1 and its association with liver metastasis and sensitivity to chemotherapy in gastric cancer patients. (a) Comparison of serum ADHFE1 level between gastric cancer patients with or without liver metastasis. (b) ROC curve discriminating gastric cancer patients with liver metastasis. (c) Comparison of serum ADHFE1 before and after chemotherapy in gastric cancer patients with liver metastasis. ***P < 0.001, ****P < 0.0001.
Patients with liver metastasis showed higher levels of serum tumor markers, including CEA (Figure 3(a)), CA199 (Figure 3(b)), and AFP (Figure 3(c)), and serum ADHFE1 was positively correlated with these markers (rCEA = 0.810, rCA199 = 0.788, rAFP = 0.765, Figure 3(d) to (f)).
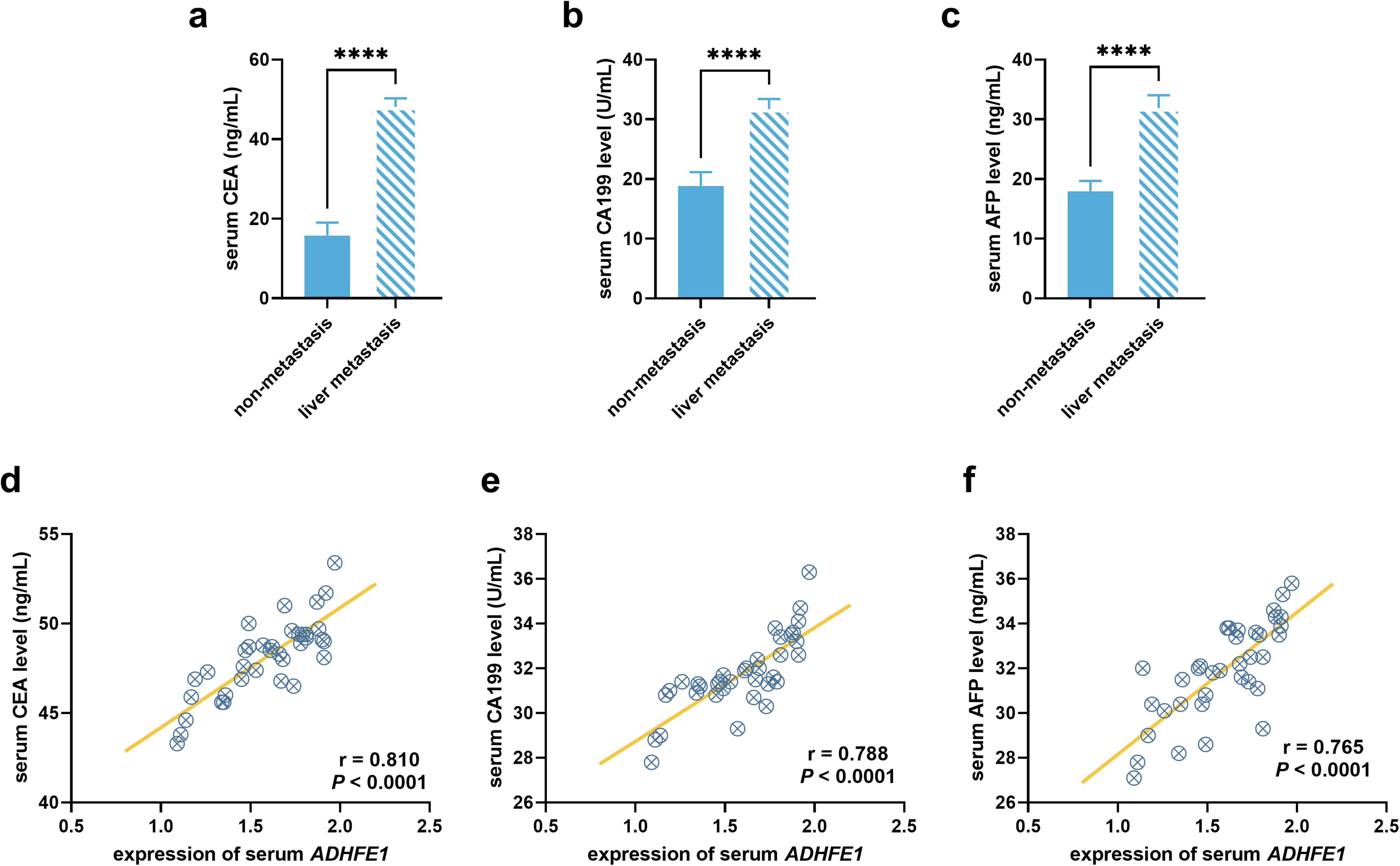
Comparison of tumor markers and their correlation with serum ADHFE1. Comparison of serum CEA (a), CA199 (b), and AFP (c) between gastric cancer patients with liver metastasis and without liver metastasis. Correlation of serum ADHFE1 levels with serum CEA (d), CA199 (e), and AFP (f) in gastric cancer patients with liver metastasis. ****P < 0.0001.
Regulatory effect of ADHFE1 on the biological behaviors of gastric cancer cells
Significant upregulation of ADHFE1 was also observed in AGS cells compared with GSE-1 cell (Figure S1). ADHFE1 was successfully knocked down and overexpressed in AGS cells (Figure 4(a)). Silencing ADHFE1 significantly suppressed the proliferation (Figure 4(b)), migration (Figure 4(c)), and invasion (Figure 4(d)) of AGS cells, while the overexpression of ADHFE1 exerted opposite effects. The knockdown of ADHFE1 reduced the resistance of AGS cells to cisplatin with the IC50 value decreasing from 7.62 to 5.05 μg/mL; the overexpression of ADHFE1 could improve the cisplatin resistance with the increasing IC50 value of 11. 58 μg/mL (Figure 4(e)). Moreover, silencing ADHFE1 significantly inhibited the angiogenesis of AGS cells, which was significantly enhanced by the overexpression of ADHFE1 (Figure 4(f)).
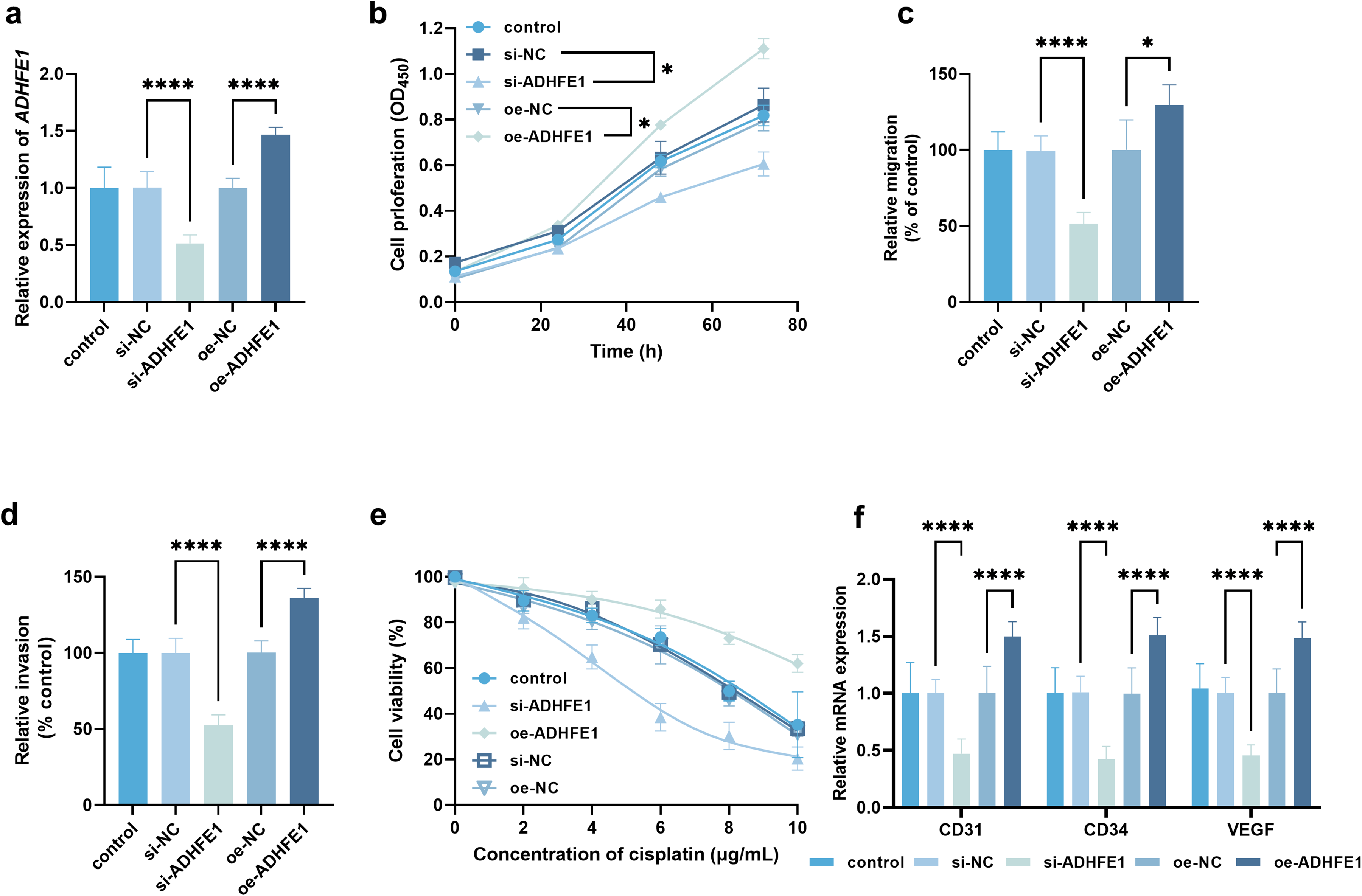
Regulatory effect of ADHFE1 on gastric cancer cellular processes and cisplatin resistance. (a) Regulation of ADHFE1 in AGS cells by cell transfection. Regulatory effect of ADHFE1 on cell proliferation (b), migration (c), and invasion (d). (e) Effect of ADHFE1 on cisplatin sensitivity of AGS cell. (f) Effect of ADHFE1 on angiogenesis-related markers. Cell motility, cisplatin sensitivity, and angiogenesis were evaluated after 24 h of cisplatin treatment. *P < 0.05, ****P < 0.0001.
Discussion
Significance of ADHFE1 in gastric cancer development
Molecular information provides a novel insight into precision medicine in oncology, which could help improve diagnosis accuracy and clinical prognosis. Increasing research has focused on biomarkers for human cancers, and several functional molecules have been identified, where ADHFE1 is one of the most prevalent candidates for various cancers. For example, the hypermethylation of ADHFE1 was demonstrated to promote cell growth and the cell cycle of colorectal cancer, which further modulate the tumor progression and influence patients’ prognosis.15,16 In breast cancer, ADHFE1 was revealed to drive disease progression via regulating metabolic programming.12,18 ADHFE1 was identified as a candidate for diagnostic biomarkers in gastric cancer as it was dysregulated. This study compared the serum ADHFE1 level between gastric cancer patients and individuals with benign lesions and observed its significant upregulation. Increasing serum ADHFE1 levels showed a close association with the advanced TNM stage and the occurrence of lymph node metastasis. An increasing serum ADHFE1 level was also associated with the adverse 5-year progression-free development. Additionally, in gastric cancer cells, silencing ADHFE1 showed a significant inhibitory effect on cell growth and metastasis. The proliferation, migration, and invasion of tumor cells are closely associated with tumor progression. All these findings indicated the potential involvement of ADHFE1 in the deterioration of gastric cancer. 19
Significance of ADHFE1 in liver metastasis of gastric cancer
As the enrolled gastric cancer patients included a part of patients at advanced stages, 36 patients with liver metastasis were included. More importantly, most patients with liver metastasis showed a relatively higher serum ADHFE1 level. Liver metastasis is one of the major reasons for the adverse outcomes of gastric cancer patients. Therefore, the prediction of liver metastasis is critical to improve the clinical prognosis. Previous studies demonstrated the predictive value of tumor markers for liver metastasis and other distant metastasis. CEA was reported to be associated with postoperative distant metastasis in colorectal cancer, but it showed a high rate of false-negative and false-positive results.20,21 CA199 is a specific marker for gastrointestinal tumors, which is upregulated in gastric cancer and colorectal cancer and could predict liver metastasis. However, the sensitivity of CA199 was relatively lower. 22 AFP is a diagnostic tumor marker for primary hepatic carcinoma and could indicate disease severity and therapeutic efficiency. 23 Recent studies have noticed the significance of AFP in predicting liver metastasis, but the specificity and sensitivity were unsatisfactory.24–26 Gastric cancer patients with liver metastasis in the present study showed higher levels of these tumor markers, and significant correlations of ADHFE1 with these tumor markers were revealed in patients with liver metastasis. Serum ADHFE1 could also discriminate patients with liver metastasis with a relatively high sensitivity and specificity. Furthermore, in vitro cell experiments revealed that ADHFE1 promoted angiogenesis. As the liver is supplied by a systemic dual blood system of arteries and portal veins, angiogenesis is critical for liver metastasis of human cancers.27,28 The regulatory effect of ADHFE1 on angiogenesis suggested that ADHFE1 might be a potential target for preventing liver cancer metastasis. Hence, ADHFE1 was identified as a biomarker for the development (especially of liver metastasis) and the prognosis of gastric cancer.
Significance of ADHFE1 in chemotherapy efficiency
Chemotherapy is commonly applied in patients with distant metastasis.29,30 All patients with liver metastasis in this study showed higher ADHFE1 levels, and the serum ADHFE1 level decreased after chemotherapy, indicating the sensitive response of ADHFE1 to chemotherapy. The increasing number of studies identified several molecules sensitive to chemotherapy, and these molecules also showed a significant regulatory effect on the chemotherapy resistance of tumor cells. For instance, circPVT1 showed significant upregulation in lung adenocarcinoma patients with chemotherapy resistance, and circPVT1 knockdown contributed to the resistance of lung adenocarcinoma cells to cisplatin. 31 Herein, silencing ADHFE1 increased the sensitivity of gastric cancer cells to cisplatin with decreased IC50 values, while the overexpression of ADHFE1 showed opposite effects. Therefore, ADHFE1 was identified as a regulator of chemotherapy for gastric cancer and can be considered as a potential therapeutic target for chemotherapy.
Limitation and outlook
As the study subjects included several patients at advanced stages, it is meaningless to evaluate the distinguishing value of ADHFE1 with the present groups. Hence, further investigations could focus on the significance of ADHFE1 in the detection of gastric cancer patients at early stages. Additionally, this study only focused on the function of ADHFE1 in gastric cancer cells, neglecting its potential role in normal gastric cells, which needs further attention.
Conclusion
Serum ADHFE1 served as a biomarker indicating severe development, liver metastasis, and poor prognosis in gastric cancer patients. ADHFE1 could regulate the growth, metastasis, angiogenesis, and cisplatin resistance of gastric cancer cells.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155251319784 - Supplemental material for Alcohol dehydrogenase iron containing 1 (ADHFE1) is associated with liver metastasis and regulates cellular function of gastric cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155251319784 for Alcohol dehydrogenase iron containing 1 (ADHFE1) is associated with liver metastasis and regulates cellular function of gastric cancer by Ling Qin, Fei Liu, Chen Sun, Zhang Zhang, Lingling Sun, Xingli Tang and Yunhong Xia in The International Journal of Biological Markers
Footnotes
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study protocol was approved by The Ethics Committee of The First Affiliated Hospital of Anhui Medical University North District and followed the principles outlined in the Declaration of Helsinki. In addition, informed consent was obtained from the participants involved.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
