Abstract
Background
Gastric cancer with a high level of serum alpha fetoprotein (AFP) is uncommon and has unique clinicopathological features and a poorer prognosis. The aim of this research was to elucidate the clinicopathological and prognostic features of gastric cancer with a high level of AFP.
Methods
The sera from 1,286 patients with gastric cancer treated at Qilu Hospital of Shandong University from January 2004 to December 2008 were analyzed preoperatively for AFP, CEA and CA19-9 levels after excluding active or chronic hepatitis, liver cirrhosis and hepatocellular carcinoma as well as preoperative distant metastasis. Patients were divided into 2 groups: 86 serum AFP-positive patients and 1,200 serum AFP-negative patients according to a cutoff of 20 ng/mL. The clinicopathological features and prognostic factors were compared between the groups.
Results
A higher incidence of serosal invasion, lymph node metastasis and liver metastasis and poorer prognosis was observed in the AFP-positive group compared with the AFP-negative group (all p<0.05). Serum AFP showed the highest specificity (93.66%) and diagnostic accuracy (92.38%) for predicting liver metastasis among the 3 tumor markers examined. Multivariate survival analysis revealed that AFP positivity was an independent prognostic factor in all 1,286 gastric cancer patients. The prognosis of AFP-positive gastric cancer was poorer than that of AFP-negative gastric cancer (p<0.05).
Conclusions
A high level of serum AFP is an independent prognostic factor in gastric cancer and can be used for evaluating the prognosis of gastric cancers whether in the presence or absence of liver metastasis.
Introduction
Although alpha fetoprotein (AFP) is a well-known tumor marker of hepatocellular carcinoma and yolk sac tumor, its serum level can also be elevated in other cancers including primary gastric carcinoma (1). Since a case of AFP-producing gastric cancer with liver metastasis was first reported by Bourreille et al in 1970 (2), scattered reports have appeared on patients with AFP-producing gastric carcinoma. Some of them showed that AFP was associated with a poor prognosis and a high incidence of liver metastasis (3-5). However, most studies were case reports or had a small sample size, and controversy still exists about their conclusions. In this study, the complete data of 1,286 gastric cancer patients including 86 AFP-positive cases were analyzed retrospectively to clarify the comprehensive clinicopathological and prognostic features of gastric cancer with a high level of AFP, its correlation with liver metastasis, and the predictive value of a high serum AFP level in gastric cancer.
Materials and Methods
Materials
From January 2004 to December 2008, 1,286 patients with histologically confirmed primary gastric cancer who underwent gastrectomy at the Qilu Hospital of Shandong University were enrolled in the study. None of the patients received any adjuvant therapy before the operation or had any other AFP-producing conditions such as active or chronic hepatitis, liver cirrhosis and hepatocellular carcinoma, and none had preoperative distant metastases. Peripheral blood samples were obtained from each patient within 1 week before surgery. Serum AFP, CEA and CA19-9 levels were measured by a group of professional operators using a Roche electrochemiluminescence instrument Elecsys 2010.
Methods
The following parameters were examined: age, sex, type of operation, tumor location, tumor size, histological type, depth of gastric wall invasion, lymph node metastasis, vessel invasion, liver metastasis, postoperative chemotherapy and survival time. The diagnostic value of serum AFP, CEA and CA19-9 for liver metastasis was compared. The cutoff values for serum AFP, CEA and CA19-9 were 20 ng/mL, 5 ng/mL and 35 ng/mL, respectively, in accordance with the manufacturer's instructions. Tumor staging was done according to the TNM classification of the UICC (7th edition).
Follow-up
Postoperative follow-up included physical examination, chest radiography, abdominal ultrasonography, routine blood tests and tumor marker tests. Diagnostic imaging such as abdominal computed tomography (CT) and gastroscopy was performed if necessary. The patients were interviewed every 1 to 3 months in the first year, every 3 to 6 months for the next 2 years, and every 6 months thereafter until 5 years postoperatively. The diagnosis of postoperative recurrence was made based on patient history, physical examination, imaging results, pathological examination and findings at reoperation. This retrospective study was approved by the institutional review board of Qilu Hospital of Shandong University, with all participating patients signing an informed consent form.
Statistics
The association between serum AFP and clinicopathological factors was analyzed by means of the chi-square test. Cumulative survival rates were calculated according to the Kaplan-Meier method, and differences between survival curves were examined with the log-rank test. Significant variables in the univariate survival analysis were further assessed in multivariate survival analysis using the Cox proportional hazards model. The predictive value of each tumor marker was assessed using a multivariate logistic regression model. All significant variables observed in univariate logistic regression analysis were included in the multivariate logistic regression analysis. P values less than 0.05 were considered statistically significant. All statistical analyses were done with the SPSS 17.0 software.
Results
Preoperative serum AFP level and clinicopathological characteristics of AFP-positive gastric cancers
Preoperative serum AFP levels in the study patients ranged from 0.05 to 4,644.00 ng/mL, with a median value of 2.42 ng/mL. There were 86 patients in the AFP-positive (≥20 ng/mL) gastric cancer group (6.69%), the remaining 1,200 patients with normal serum AFP levels were assigned to the AFP-negative group. The mean ± SD values of serum AFP of patients with and without liver metastases were 49.96 ± 98.38 ng/mL and 18.08 ± 191.07 ng/mL, respectively. A higher mean AFP level was observed in patients with liver metastasis (Fig. 1) (p<0.05).
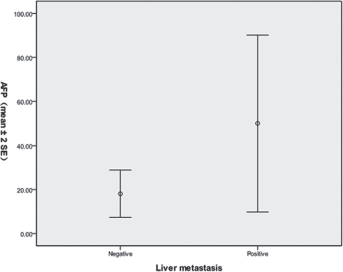
Comparison of AFP levels (mean ± 2SE) between patients with and without liver metastasis (p<0.05).
The comparison of clinicopathological characteristics between AFP-positive and AFP-negative gastric cancers is shown in Table I. Larger tumor size (>3 cm) and a higher incidence of serosal invasion (81.4% vs. 66.2%), lymph node metastasis (79.1% vs. 60.7%) and liver metastasis (6.98% vs. 1.5%) were observed in the AFP-positive group compared with the negative group (all p<0.05). Age, sex, type of operation, tumor location, histological type, vessel invasion and use of postoperative chemotherapy were similar in the AFP-positive and AFP-negative groups.
Comparison of clinicopathological characteristics between AFP-positive and AFP-negative gastric cancers
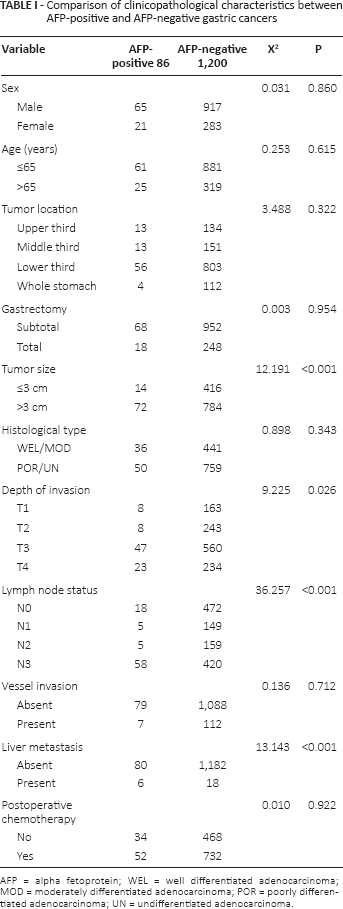
AFP = alpha fetoprotein; WEL = well differentiated adenocarcinoma; MOD = moderately differentiated adenocarcinoma; POR = poorly differentiated adenocarcinoma; UN = undifferentiated adenocarcinoma.
Diagnostic value of AFP for liver metastasis
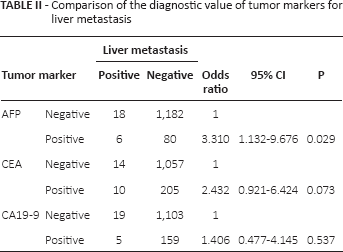
Figure 2 shows a comparison of the predictive value of 3 common tumor markers for liver metastasis. The sensitivity of AFP was 25% (6 of 24 patients) and the sera of 18 patients with liver metastasis were found to be false negative. The incidence of elevated AFP levels in patients without liver metastasis was 6.34% (80 of 1,262 patients). AFP had the highest specificity (93.66%) and accuracy (92.38%), and its sensitivity was slightly lower than that of CEA. Moreover, multivariate analysis by the logistic regression model indicated that patients with preoperative serum AFP above 20 ng/mL had the highest odds ratio (3.31; 95% CI 1.132-9.676) for predicting liver metastasis among the markers tested. CEA and CA19-9 did not add significant predictive information (p = 0.073 and p = 0.537, respectively) (Tab. II).
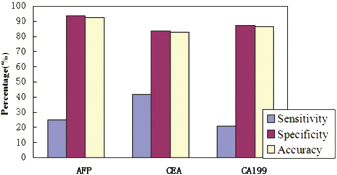
Comparison of diagnostic value for liver metastasis of each tumor marker. Sensitivity = (patients with liver metastasis with positive tests/all patients with liver metastasis tested) × 100. Specificity = (patients without liver metastasis with negative tests/all patients without liver metastasis tested) × 100. Diagnostic accuracy = (patients with liver metastasis with positive tests + patients without liver metastasis with negative tests)/(all patients tested) × 100.
Comparison of the diagnostic value of tumor markers for liver metastasis
Prognosis of serum AFP-positive gastric cancer
The overall 1-, 3- and 5-year survival rates of the AFP-positive group were 58.1%, 32.6% and 18.6%, while those of the AFP-negative group were 81.7%, 59% and 48.7%, respectively (p<0.05). The 1-, 3- and 5-year disease-free survival rates of the AFP-positive group were 51.2%, 23.3% and 16.3%, while those of the AFP-negative group were 76.3%, 54.5% and 44.2%, respectively (p<0.05). The AFP-positive group had a significantly lower overall survival rate (Fig. 3A) and disease-free survival rate (Fig. 3B) than the AFP-negative group. Multivariate survival analysis revealed that a high serum AFP level was an independent prognostic factor for both overall survival and disease-free survival (Tab. III).
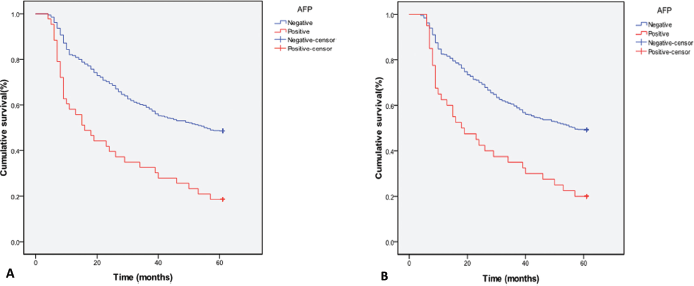
(
Univariate and multivariate survival analyses in 1,286 gastric cancer patients
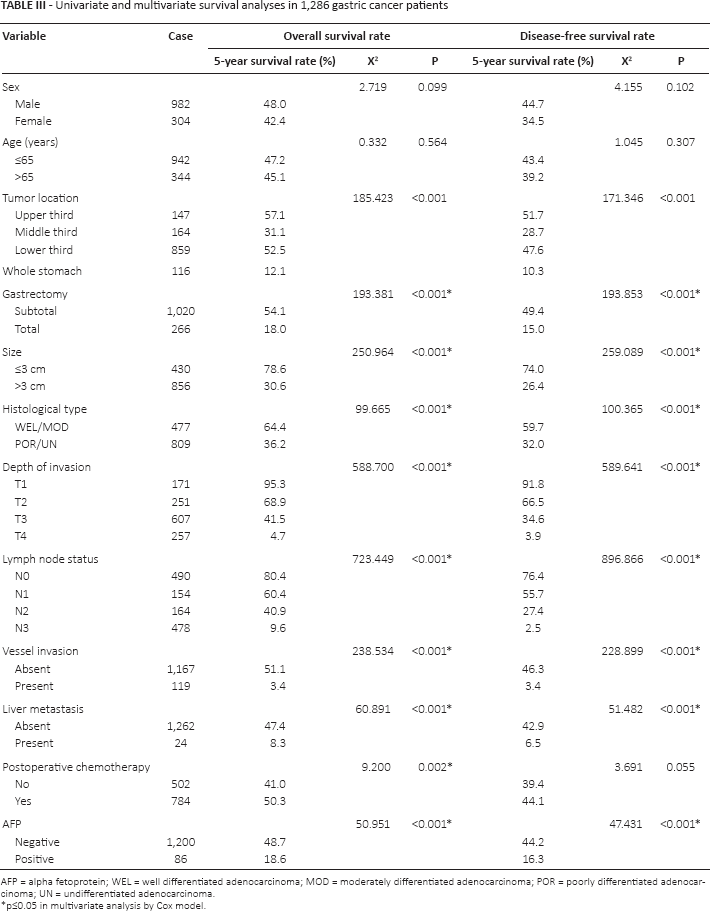
AFP = alpha fetoprotein; WEL = well differentiated adenocarcinoma; MOD = moderately differentiated adenocarcinoma; POR = poorly differentiated adenocarcinoma; UN = undifferentiated adenocarcinoma.
p≤0.05 in multivariate analysis by Cox model.
There was a significantly higher incidence of liver metastasis in the AFP-positive group than the AFP-negative group (6.98% vs. 1.5%). All 6 AFP-positive patients with liver metastasis died within 1 year. The survival rate of AFP-positive gastric cancer patients was lower than that of AFP-negative patients with liver metastasis (Fig. 4A). Also among patients without liver metastasis, the survival rate of AFP-positive patients was lower than that of AFP-negative patients (Fig. 4B).
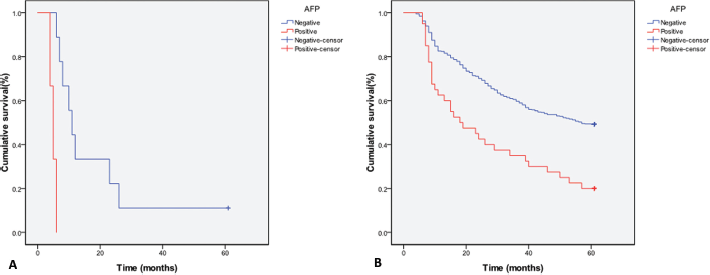
(
Univariate logistic regression analysis showed that prognosis was influenced by tumor location, type of operation, tumor size, histological type, depth of gastric wall invasion, lymph node metastasis, vessel invasion, postoperative chemotherapy and serum AFP level (Tab. III). Multivariate logistic regression analysis revealed that serum AFP was an independent prognostic factor in gastric cancer patients without liver metastasis (Tab. III).
Discussion
AFP was originally found in the human fetus and is normally produced in the fetal liver and yolk sac (6). It is considered to be a useful tumor marker in hepatocellular carcinoma and yolk sac tumors in clinical practice. Some studies have shown that other tumors can also produce AFP, and gastric cancer was one of the most common (7, 8). However, the exact mechanism remains controversial. Ishikura et al (9) proposed a new entity, hepatoid adenocarcinoma of the stomach (HAS), given that the stomach and liver were both embryologically derived from the primitive foregut of the embryo. The authors suggested that some gastric cancers may produce a large amount of AFP in the same way as hepatocellular carcinoma when there is an abnormality during differentiation (9). Ooi et al (10) on the other hand suggested that AFP-producing gastric cancers were not always derived from hepatoid differentiation, and that the AFP in primary gastric cancer might be a fetal protein specific for the gastrointestinal tract.
HAS or AFP-producing gastric cancer has been reported to be associated with more liver and lymph node metastasis (4, 5), and in this study we found a higher incidence of liver metastasis, lymph node metastasis and serosal invasion in the AFP-positive group of patients. No other clinicopathological features were correlated with serum AFP level except tumor size larger than 3 cm. Vascular invasion was also reported to be more common in AFP-producing gastric cancer (4, 11), but the difference of vessel invasion was not statistically significant in this study. The incidence of liver metastasis in the AFP-positive group was significantly higher than that in the AFP-negative group. Chang et al (12) reported that the incidence of synchronous liver metastasis in AFP-positive gastric cancer was 31.8%. Another group reported the incidence of synchronous liver metastasis in AFP-positive gastric cancer to be 6.7% (5). Our study found an incidence of 6.98%, which is obviously low with respect to the report of Chang et al. The discrepancies in incidence might be due to our excluding cases with synchronous liver metastasis, which cannot be performed with radical gastrectomy.
Yamashita et al (11) reported that AFPGC had a significantly poorer prognosis than common gastric cancers, and Chun and Kwon (3) came to the same conclusion. The Yamashita group proposed that AFP had an immunosuppressive function in the human body. In its presence, cancer cells grow faster and a cancer embolus can develop because of vessel invasion, resulting in distant metastasis (11). However, there is still controversy about the predictive value of serum AFP. Ucar et al (12) found that serum AFP was not related to the prognosis of gastric cancer, while Lin et al (4) reported that a high level of serum AFP was associated with short survival time. Hirajima et al (13) reported that AFP positivity was not an independent prognostic factor in gastric cancer patients. However, their analysis included a small number of patients with AFP-producing gastric cancers, and a larger sample size was needed. There were 86 patients with AFP-producing gastric cancer included in our study, which has been the largest sample as far as we know, and the results showed that serum AFP level independently influenced both the overall and disease-free survival rates.
Shimada et al (14) reported that AFP is useful for detecting and predicting liver metastases in gastric cancer, while Choi et al (15) reported that serum CEA rather than AFP was associated with liver recurrence. Our results showed that serum AFP had the best specificity and accuracy in predicting liver metastasis, and was more predictive than serum CEA and CA19-9. Furthermore, whether with or without liver metastasis, the prognosis of AFP-positive gastric cancer was always poorer than that of AFP-negative gastric cancer. Multivariate logistic analysis in patients without liver metastasis revealed that serum AFP was an independent prognostic factor, and patients with high levels of serum AFP had a poorer prognosis than those in whom AFP was negative. All these findings suggested that AFP positivity can be used to evaluate the prognosis of patients with gastric cancer. Special attention should therefore be paid to patients with high levels of serum AFP: we need to arrange regular visits with these patients more frequently and carry out examinations including CT, MRI, PET-CT and others whenever necessary. When their conditions are monitored in a timely and accurate manner, these patients can be appropriately treated with postoperative chemotherapy, radiotherapy, biotherapy and other treatments and gain benefit from them.
Several limitations of this study should be noted. First, it is a retrospective study in which patients underwent gastrectomies by several different surgeons, so it cannot be excluded that human confounding factors were present. Second, although a large number of gastric cancers were included, the number of patients with liver metastases was small. Finally, given that postoperative chemotherapy may vary in terms of opportunities and strategies, an obvious barrier to survival analysis cannot be eliminated in this study.
In conclusion, gastric cancer with a high level of serum AFP was associated with a high incidence of serosal invasion, lymph node metastasis and liver metastasis and a poor prognosis. Preoperative serum AFP levels could have predictive value for the development of liver metastasis in patients with gastric cancer. AFP positivity therefore is an independent prognostic factor in gastric cancer. It can be used to evaluate prognosis both in the presence and absence of liver metastasis.
Footnotes
Financial support: This study was supported by a research grant (No. Y2008C38) from the Natural Sciences Foundation of Shandong Province.
Conflict of interest: The authors have no conflict of interest related to this paper.
